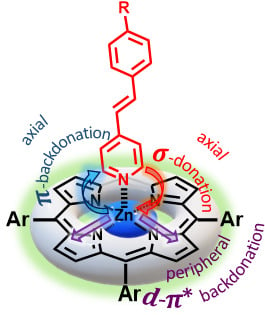
Second Order Nonlinear Optical Properties of 4-Styrylpyridines Axially Coordinated to A4 ZnII Porphyrins: A Comparative Experimental and Theoretical Investigation †
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis of L1 and L2 Axially Substituted A4 ZnII Porphyrins
2.2. 1H-NMR and UV–Vis Spectroscopy
2.3. Experimental and Theoretical Investigation of the Second Order NLO Properties
3. Materials and Methods
3.1. General
3.2. EFISH and THG Measurements
3.3. Computational Calculations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Di Bella, S. Second-order nonlinear optical properties of transition metal complexes. Chem. Soc. Rev. 2001, 30, 355–366. [Google Scholar] [CrossRef]
- Cariati, E.; Pizzotti, M.; Roberto, D.; Tessore, F.; Ugo, R. Coordination and organometallic compounds and inorganic-organic hybrid crystalline materials for second-order non-linear optics. Coord. Chem. Rev. 2006, 250. [Google Scholar] [CrossRef]
- Di Bella, S.; Dragonetti, C.; Pizzotti, M.; Roberto, D.; Tessore, F.; Ugo, R. Molecular Organometallic Materials for Optics; Springer: Berlin, Germany, 2010; Volume 28. [Google Scholar]
- Roberto, D.; Ugo, R.; Bruni, S.; Cariati, E.; Cariati, F.; Fantucci, P.; Invernizzi, I.; Quici, S.; Ledoux, I.; Zyss, J. Quadratic hyperpolarizability enhancement of para-substituted pyridines upon coordination to organometallic moieties: The ambivalent donor or acceptor role of the metal. Organometallics 2000, 19, 1775–1788. [Google Scholar] [CrossRef]
- Baccouche, A.; Peigné, B.; Ibersiene, F.; Hammoutène, D.; Boutarfäa, A.; Boucekkine, A.; Feuvrie, C.; Maury, O.; Ledoux, I.; Le Bozec, H. Effects of the metal center and substituting groups on the linear and nonlinear optical properties of substituted styryl-bipyridine metal(II) dichloride complexes: DFT and TDDFT computational investigations and harmonic light scattering measurements. J. Phys. Chem. A 2010, 114, 5429–5438. [Google Scholar] [CrossRef] [PubMed]
- Roberto, D.; Ugo, R.; Tessore, F.; Lucenti, E.; Quici, S.; Vezza, S.; Fantucci, P.; Invernizzi, I.; Bruni, S.; Ledoux-Rak, I.; et al. Effect of the Coordination to M(II) Metal Centers (M = Zn, Cd, Pt) on the Quadratic Hyperpolarizability of Various Substituted 5-X-1,10-phenanthrolines (X) Donor Group) and of trans-4-(Dimethylamino)-4′-stilbazole. Organometallics 2002, 21. [Google Scholar] [CrossRef]
- Roberto, D.; Tessore, F.; Ugo, R.; Bruni, S.; Manfredi, A.; Quici, S. Terpyridine Zn(II), Ru(III) and Ir(III) complexes as new asymmetric chromophores for nonlinear optics: First evidence for a shift from positive to negative value of the quadratic hyperpolarizability of a ligand carrying an electron donor substituent upon. Chem. Commun. 2002, 2, 846–847. [Google Scholar] [CrossRef]
- Tessore, F.; Roberto, D.; Ugo, R.; Pizzotti, M.; Quici, S.; Cavazzini, M.; Bruni, S.; De Angelis, F. Terpyridine Zn(II), Ru(III), and Ir(III) complexes: The relevant role of the nature of the metal ion and of the ancillary ligands on the second-order nonlinear response of terpyridines carrying electron donor or electron acceptor groups. Inorg. Chem. 2005, 44. [Google Scholar] [CrossRef]
- Kanis, D.R.; Lacroix, P.G.; Ratner, M.A.; Marks, T.J. Electronic Structure and Quadratic Hyperpolarizabilities in Organotransition-Metal Chromophores Having Weakly Coupled π-Networks. Unusual Mechanisms for Second-Order Response. J. Am. Chem. Soc. 1994, 116, 10089–10102. [Google Scholar] [CrossRef]
- Lucenti, E.; Cariati, E.; Dragonetti, C.; Manassero, L.; Tessore, F. Effect of the coordination to the “Os3(CO)11” cluster core on the quadratic hyperpolarizability of trans-4-(4′-X-styryl)pyridines (X = NMe2, t-Bu, CF3) and trans,trans-4-(4′-NMe2-phenyl-1,3-butadienyl. Organometallics 2004, 23. [Google Scholar] [CrossRef]
- Oudar, J.L. Optical nonlinearities of conjugated molecules. Stilbene derivatives and highly polar aromatic compounds. J. Chem. Phys. 1977, 67, 446–457. [Google Scholar] [CrossRef]
- Oudar, J.L.; Chemla, D.S. Hyperpolarizabilities of the nitroanilines and their relations to the excited state dipole moment. J. Chem. Phys. 1977, 66, 2664–2668. [Google Scholar] [CrossRef]
- Bruni, S.; Cariati, E.; Cariati, F.; Porta, F.A.; Quici, S.; Roberto, D. Determination of the quadratic hyperpolarizability of trans-4-[4-(dimethylamino)styryl]pyridine and 5-dimethylamino-1,10-phenanthroline from solvatochromism of absorption and fluorescence spectra: A comparison with the electric-field-induced second-harmon. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2001, 57, 1417–1426. [Google Scholar] [CrossRef]
- Pizzotti, M.; Annoni, E.; Ugo, R.; Bruni, S.; Quici, S.; Fantucci, P.; Bruschi, M.; Zerbi, G.; Zoppo, M. Del A multitechnique investigation of the second order NLO response of a 10,20-diphenylporphyrinato nickel(II) complex carrying a phenylethynyl based push-pull system in the 5- and 15-positions. J. Porphyr. Phthalocyanines 2004, 8, 1311–1324. [Google Scholar] [CrossRef]
- Morotti, T.; Pizzotti, M.; Ugo, R.; Quici, S.; Bruschi, M.; Mussini, P.; Righetto, S. Electronic characterisation and significant second-order NLO response of 10,20-diphenylporphyrins and their ZnII complexes substituted in the meso position with π-delocalised linkers carrying push or pull groups. Eur. J. Inorg. Chem. 2006, 1743–1757. [Google Scholar] [CrossRef]
- De Angelis, F.; Fantacci, S.; Sgamellotti, A.; Pizzotti, M.; Tessore, F.; Orbelli Biroli, A. Time-dependent and coupled-perturbed DFT and HF investigations on the absorption spectrum and non-linear optical properties of push-pull M(II)-porphyrin complexes (M = Zn, Cu, Ni). Chem. Phys. Lett. 2007, 447, 10–15. [Google Scholar] [CrossRef]
- Pizzotti, M.; Tessore, F.; Orbelli Biroli, A.; Ugo, R.; De Angelis, F.; Fantacci, S.; Sgamellotti, A.; Zuccaccia, D.; Macchioni, A. An EFISH, theoretical, and PGSE NMR investigation on the relevant role of aggregation on the second order response in CHCl3 of the push-pull chromophores [5-[[4′-(dimethylamino)phenyl]ethynyl]-15-[(4′′-nitrophenyl)ethynyl]-10,20diphenylporphyri. J. Phys. Chem. C 2009, 113. [Google Scholar] [CrossRef]
- Orbelli Biroli, A.; Tessore, F.; Righetto, S.; Forni, A.; Macchioni, A.; Rocchigiani, L.; Pizzotti, M.; Di Carlo, G. Intriguing Influence of −COOH-Driven Intermolecular Aggregation and Acid-Base Interactions with N,N-Dimethylformamide on the Second-Order Nonlinear-Optical Response of 5,15 Push-Pull Diarylzinc(II) Porphyrinates. Inorg. Chem. 2017, 56. [Google Scholar] [CrossRef] [Green Version]
- Annoni, E.; Pizzotti, M.; Ugo, R.; Quici, S.; Morotti, T.; Bruschi, M.; Mussini, P. Synthesis, electronic characterisation and significant second-order non-linear optical responses of meso-tetraphenylporphyrins and their ZnII complexes carrying a push or pull group in the β pyrrolic position. Eur. J. Inorg. Chem. 2005, 3857–3874. [Google Scholar] [CrossRef]
- Tessore, F.; Orbelli Biroli, A.; Di Carlo, G.; Pizzotti, M. Porphyrins for Second Order Nonlinear Optics (NLO): An Intriguing History. Inorganics 2018, 6, 81. [Google Scholar] [CrossRef] [Green Version]
- Di Carlo, G.; Pizzotti, M.; Righetto, S.; Forni, A.; Tessore, F. Electric-Field-Induced Second Harmonic Generation Nonlinear Optic Response of A4 β-Pyrrolic-Substituted ZnII Porphyrins: When Cubic Contributions Cannot Be Neglected. Inorg. Chem. 2020, 59, 7561–7570. [Google Scholar] [CrossRef]
- Bajju, G.D.; Kundan, S.; Kapahi, A.; Gupta, D. Synthesis and Spectroscopic Studies of Axially Ligated Zn(II)5,10,15,20-meso-tetra(p-chlorophenyl)porphyrin with Oxygen and Nitrogen Donors. J. Chem. 2013, 2013, 135815. [Google Scholar] [CrossRef]
- Charisiadis, A.; Stangel, C.; Nikolaou, V.; Roy, M.S.; Sharma, G.D.; Coutsolelos, A.G. A supramolecular assembling of zinc porphyrin with a π-conjugated oligo(phenylenevinylene) (oPPV) molecular wire for dye sensitized solar cell. Rsc Adv. 2015, 5, 88508–88519. [Google Scholar] [CrossRef]
- Xie, M.; Bai, F.-Q.; Zhang, H.-X.; Zheng, Y.-Q. The influence of an inner electric field on the performance of three types of Zn-porphyrin sensitizers in dye sensitized solar cells: A theoretical study. J. Mater. Chem. C 2016, 4, 10130–10145. [Google Scholar] [CrossRef]
- Annoni, E.; Pizzotti, M.; Ugo, R.; Quici, S.; Morotti, T.; Casati, N.; Macchi, P. The effect on E-stilbazoles second order NLO response by axial interaction with M(II) 5,10,15,20-tetraphenyl porphyrinates (M = Zn, Ru, Os); a new crystalline packing with very large holes. Inorg. Chim. Acta 2006, 359, 3029–3041. [Google Scholar] [CrossRef]
- Cole, S.J.; Curthoys, G.C.; Magnusson, E.A. Ligand Binding by Metalloporphyrins. I. Thermodynamic Functions of Porphyriniron(II)-Pyridine Complexes. J. Am. Chem. Soc. 1970, 92, 2991–2996. [Google Scholar] [CrossRef]
- Levine, B.F.; Bethea, C.G. Molecular hyperpolarizabilities determined from conjugated and nonconjugated organic liquids. Appl. Phys. Lett. 1974, 24, 445–447. [Google Scholar] [CrossRef]
- Singer, K.D.; Garito, A.F. Measurements of molecular second order optical susceptibilities using dc induced second harmonic generation. J. Chem. Phys. 1981, 75, 3572–3580. [Google Scholar] [CrossRef]
- Kurtz, H.A.; Dudis, D.S. Quantum mechanical methods for predicting nonlinear optical properties. In Reviews in Computational Chemistry; 2007; Volume 12, pp. 241–279. [Google Scholar] [CrossRef]
- Di Carlo, G.; Orbelli Biroli, A.; Pizzotti, M.; Tessore, F.; Trifiletti, V.; Ruffo, R.; Abbotto, A.; Amat, A.; De Angelis, F.; Mussini, P.R. Tetraaryl ZnII porphyrinates substituted at β-pyrrolic positions as sensitizers in dye-sensitized solar cells: A comparison with meso-disubstituted push-pull ZnII porphyrinates. Chem. A Eur. J. 2013, 19, 10723–10740. [Google Scholar] [CrossRef]
- Di Carlo, G.; Orbelli Biroli, A.; Tessore, F.; Caramori, S.; Pizzotti, M. β-Substituted ZnII porphyrins as dyes for DSSC: A possible approach to photovoltaic windows. Coord. Chem. Rev. 2018, 358, 153–177. [Google Scholar] [CrossRef]
- Di Carlo, G.; Orbelli Biroli, A.; Pizzotti, M.; Tessore, F. Efficient sunlight harvesting by A4 β-Pyrrolic Substituted ZnII Porphyrins: A Mini-Review. Front. Chem. 2019, 7. [Google Scholar] [CrossRef]
- Di Carlo, G.; Caramori, S.; Casarin, L.; Orbelli Biroli, A.; Tessore, F.; Argazzi, R.; Oriana, A.; Cerullo, G.; Bignozzi, C.A.; Pizzotti, M. Charge Transfer Dynamics in β and Meso Substituted Dithienylethylene Porphyrins. J. Phys. Chem. C 2017, 121, 18385–18400. [Google Scholar] [CrossRef]
- Di Carlo, G.; Orbelli Biroli, A.; Tessore, F.; Rizzato, S.; Forni, A.; Magnano, G.; Pizzotti, M. Light-Induced Regiospecific Bromination of meso-Tetra(3,5-di-tert-butylphenyl)Porphyrin on 2,12 β-Pyrrolic Positions. J. Org. Chem. 2015, 80, 4973–4980. [Google Scholar] [CrossRef] [PubMed]
- Orbelli Biroli, A.; Tessore, F.; Di Carlo, G.; Pizzotti, M.; Benazzi, E.; Gentile, F.; Berardi, S.; Bignozzi, C.A.; Argazzi, R.; Natali, M.; et al. Fluorinated ZnII Porphyrins for Dye-Sensitized Aqueous Photoelectrosynthetic Cells. Acs Appl. Mater. Interfaces 2019, 11, 32895–32908. [Google Scholar] [CrossRef] [PubMed]
- Lindsey, J.S.; Schreiman, I.C.; Hsu, H.C.; Kearney, P.C.; Marguerettaz, A.M. Rothemund and Adler-Longo reactions revisited: Synthesis of tetraphenylporphyrins under equilibrium conditions. J. Org. Chem. 1987, 52, 827–836. [Google Scholar] [CrossRef]
- Berardi, S.; Caramori, S.; Benazzi, E.; Zabini, N.; Niorettini, A.; Orbelli Biroli, A.; Pizzotti, M.; Tessore, F.; Di Carlo, G. Electronic Properties of Electron-Deficient Zn(II) Porphyrins for HBr Splitting. Appl. Sci. 2019, 9, 2739. [Google Scholar] [CrossRef] [Green Version]
- Covezzi, A.; Orbelli Biroli, A.; Tessore, F.; Forni, A.; Marinotto, D.; Biagini, P.; Di Carlo, G.; Pizzotti, M. 4D-π-1A type β-substituted ZnII-porphyrins: Ideal green sensitizers for building-integrated photovoltaics. Chem. Commun. 2016, 52. [Google Scholar] [CrossRef] [Green Version]
- Shirazi, A.; Goff, H.M. Carbon-13 and Proton NMR Spectroscopy of Four- and Five-Coordinate Cobalt(II) Porphyrins: Analysis of NMR Isotropic Shifts. Inorg. Chem. 1982, 3420–3425. [Google Scholar] [CrossRef]
- Tessore, F.; Roberto, D.; Ugo, R.; Mussini, P.; Quici, S.; Ledoux-Rak, I.; Zyss, J. Large, concentration-dependent enhancement of the quadratic hyperpolarizability of [Zn(CH3CO2)2(L)2] in CHCl3 on substitution of acetate by triflate. Angew. Chem. Int. Ed. 2003, 42. [Google Scholar] [CrossRef]
- Tessore, F.; Locatelli, D.; Righetto, S.; Roberto, D.; Ugo, R.; Mussini, P. An Investigation on the Role of the Nature of Sulfonate Ancillary Ligands on the Strength and Concentration Dependence of the Second-Order NLO Responses in CHCl3 of Zn(II) Complexes with 4,4′-trans-NC5H4CH=CHC6H4NMe2 and 4,4′-trans,trans-NC5H4(CH=CH)2C6H4NMe2. Inorg. Chem. 2005, 44, 2437–2442. [Google Scholar]
- Gouterman, M. Spectra of porphyrins. J. Mol. Spectrosc. 1961, 6, 138–163. [Google Scholar] [CrossRef]
- Nappa, M.; Valentine, J.S. The Influence of Axial Ligands on Metalloporphyrin Visible Absorption Spectra. Complexes of Tetraphenylporphinatozinc. J. Am. Chem. Soc. 1978, 100, 5075–5080. [Google Scholar] [CrossRef]
- Szintay, G.; Horváth, A. Temperature dependence study of five-coordinate complex formation of zinc(II) octaethyl and tetraphenylporphyrin. Inorg. Chim. Acta 2000, 310, 175–182. [Google Scholar] [CrossRef]
- Hansch, C.; Leo, A.; Taft, R.W. A Survey of Hammett Substituent Constants and Resonance and Field Parameters. Chem. Rev. 1991, 91, 165–195. [Google Scholar] [CrossRef]
- Zyss, J.; Chemla, D.S.; Nicoud, J.F. Demonstration of efficient nonlinear optical crystals with vanishing molecular dipole moment: Second-harmonic generation in 3-methyl-4-nitropyridine-1-oxide. J. Chem. Phys. 1981, 74, 4800–4811. [Google Scholar] [CrossRef]
- Willetts, A.; Rice, J.E.; Burland, D.M.; Shelton, D.P. Problems in the comparison of theoretical and experimental hyperpolarizabilities. J. Chem. Phys. 1992, 97, 7590–7599. [Google Scholar] [CrossRef]
- Zhao, Y.; Truhlar, D.G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other function. Theor. Chem. Acc. 2008, 120, 215–241. [Google Scholar] [CrossRef] [Green Version]
- Johnson, L.E.; Dalton, L.R.; Robinson, B.H. Optimizing calculations of electronic excitations and relative hyperpolarizabilities of electrooptic chromophores. Acc. Chem. Res. 2014, 47, 3258–3265. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16; revision, A.03; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]



| Compound | Ligand Band λmax (nm) | Soret or B Band λmax (nm) | QIV Band λmax (nm) | QIII Band λmax (nm) | QII Band λmax (nm) | QI Band λmax (nm) |
|---|---|---|---|---|---|---|
| L1 | 332 | |||||
| L2 | 376 | |||||
| TFP | 412 | 506 | 583 | 637 | 657 | |
| ZnTFP | 419 | 551 | 584 | |||
| ZnTFP-L1 | 321 | 419 | 551 | 585 | 627 | |
| ZnTFP-L2 | 419 | 480 | 550 | 585 | 617 | |
| TPP | 418 | 515 | 550 | 589 | 646 | |
| ZnTPP | 422 | 553 | 594 | |||
| ZnTPP-L1 | 319 | 421 | 553 | 594 | ||
| ZnTPP-L2 | 422 | 553 | 594 | |||
| TBP | 421 | 518 | 554 | 592 | 648 | |
| ZnTBP | 424 | 552 | 595 | |||
| ZnTBP-L1 | 329 | 424 | 552 | 595 | ||
| ZnTBP-L2 | 372 | 424 | 552 | 595 | ||
| ZnTNP | 306 | 438 | 559 | 605 | ||
| ZnTNP-L1 | 310 | 433 | 559 | 603 | ||
| ZnTNP-L2 | 305 | 434 | 559 | 603 |
| Compound | μ0β1907 (×10−48 esu) | μ0 calc (μ0 EF a) (D) | β1907 (β1907 EF b) (×10−30 esu) | β|| (β|| EF d) (×10−48 esu) |
|---|---|---|---|---|
| L1 | +310 | 2.67 | +116 | 11 |
| ZnTFP-L1 | −1680 | 0.29 | nd c | −3 |
| ZnTPP-L1 | −540 | 0.36 | nd | 7 |
| ZnTBP-L1 | −880 | 0.68 | nd | 7 |
| ZnTNP-L1 | −733 | 0.70 | nd | 12 |
| L2 | +250 | 6.59 | +38 d | 37 |
| ZnTFP-L2 | −980 | 10.63 (1.61) | −92 (2.42) | 75 (2.0) |
| ZnTPP-L2 | −280 | 9.84 (1.49) | −28 (0.74) | 65 (1.75) |
| ZnTBP-L2 | −860 | 9.64 (1.46) | −89 (2.34) | 64 (1.73) |
| ZnTNP-L2 | −606 | 9.31 (1.41) | −65 (1.71) | 60 (1.62) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tessore, F.; Di Carlo, G.; Forni, A.; Righetto, S.; Limosani, F.; Orbelli Biroli, A. Second Order Nonlinear Optical Properties of 4-Styrylpyridines Axially Coordinated to A4 ZnII Porphyrins: A Comparative Experimental and Theoretical Investigation. Inorganics 2020, 8, 45. https://doi.org/10.3390/inorganics8080045
Tessore F, Di Carlo G, Forni A, Righetto S, Limosani F, Orbelli Biroli A. Second Order Nonlinear Optical Properties of 4-Styrylpyridines Axially Coordinated to A4 ZnII Porphyrins: A Comparative Experimental and Theoretical Investigation. Inorganics. 2020; 8(8):45. https://doi.org/10.3390/inorganics8080045
Chicago/Turabian StyleTessore, Francesca, Gabriele Di Carlo, Alessandra Forni, Stefania Righetto, Francesca Limosani, and Alessio Orbelli Biroli. 2020. "Second Order Nonlinear Optical Properties of 4-Styrylpyridines Axially Coordinated to A4 ZnII Porphyrins: A Comparative Experimental and Theoretical Investigation" Inorganics 8, no. 8: 45. https://doi.org/10.3390/inorganics8080045
APA StyleTessore, F., Di Carlo, G., Forni, A., Righetto, S., Limosani, F., & Orbelli Biroli, A. (2020). Second Order Nonlinear Optical Properties of 4-Styrylpyridines Axially Coordinated to A4 ZnII Porphyrins: A Comparative Experimental and Theoretical Investigation. Inorganics, 8(8), 45. https://doi.org/10.3390/inorganics8080045