CuII Complexes and Coordination Polymers with Pyridine or Pyrazine Amides and Amino Benzamides—Structures and EPR Patterns
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis of the Cu Complexes
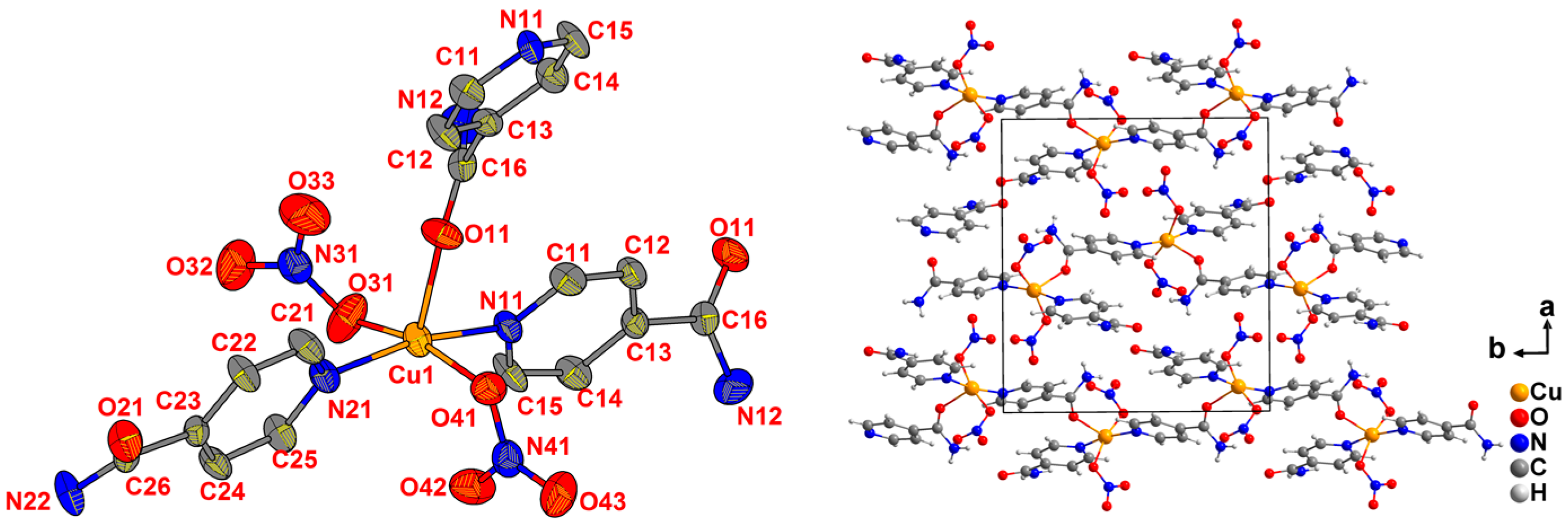
2.2. Crystal Structures from Single Crystal XRD
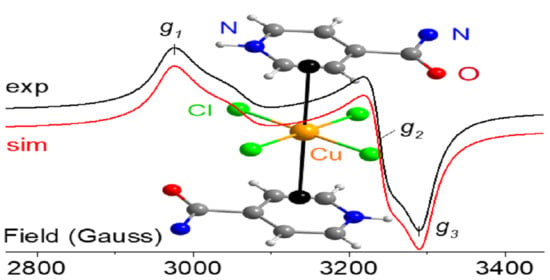
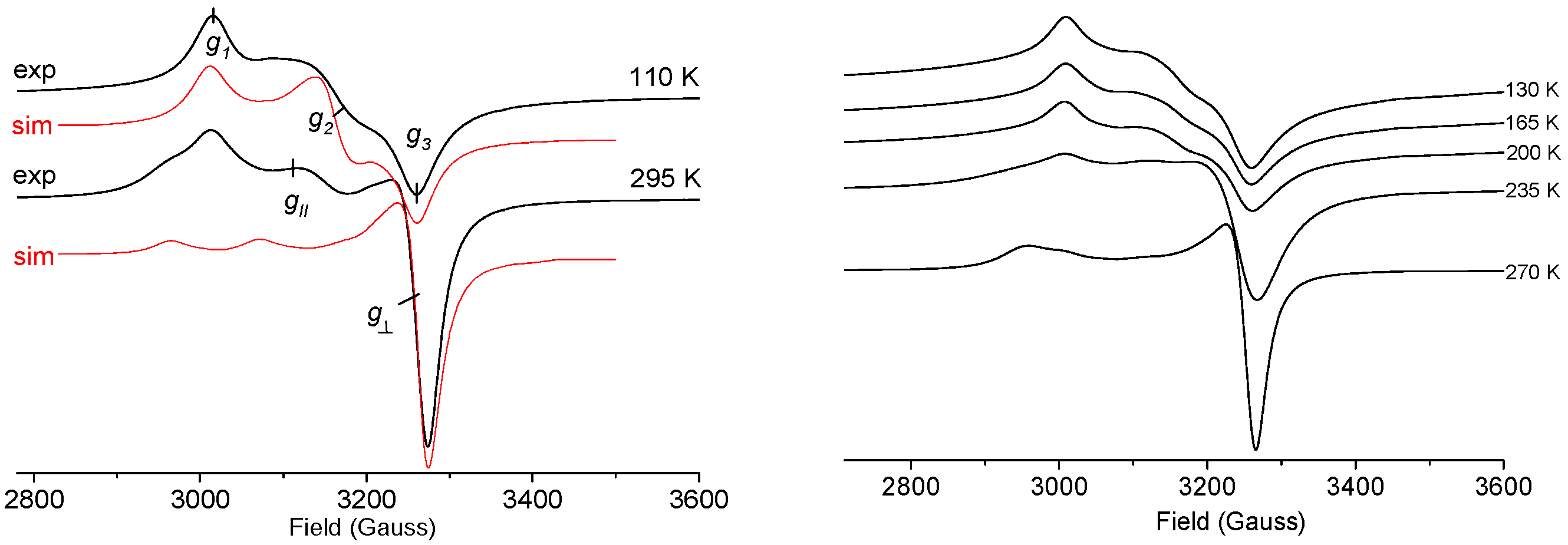
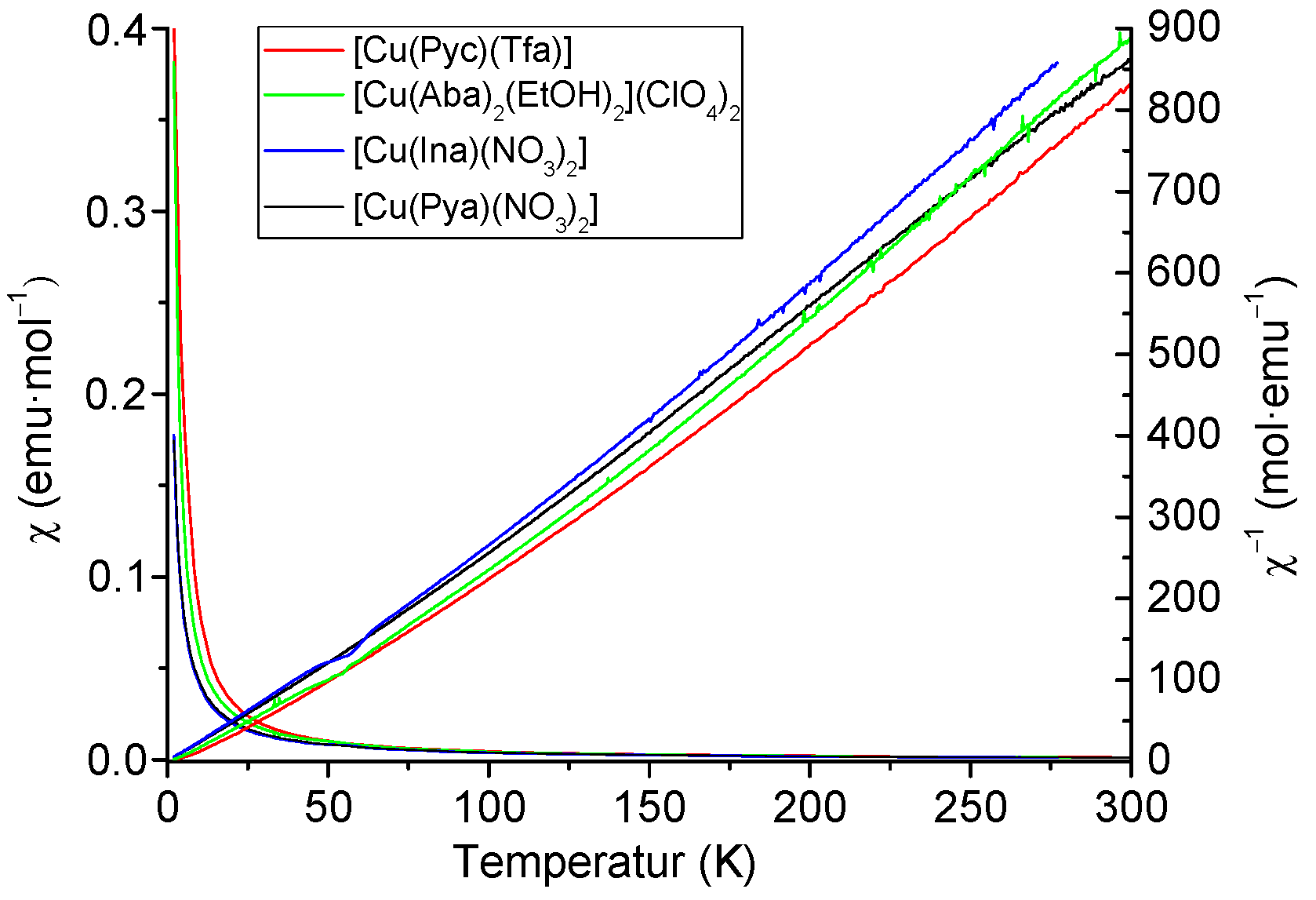
2.3. X-Band EPR Spectroscopy and Magnetic Measurements
2.4. Absorption Spectroscopy in the Solid and in Solution
3. Experimental Section
3.1. Methods and Instrumentation
3.2. Single Crystal Structure Determination
3.3. Powder X-ray Diffraction (PXRD)
3.4. Syntheses
3.4.1. General
3.4.2. Synthesis of the CuII Compounds—General Method
3.4.3. Further Syntheses
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Riascos-Rodríguez, K.; Marks, S.; Evans, P.G.; Hernández-Rivera, S.P.; Ruiz-Caballero, J.L.; Pinñero, D.; Hernaández-Maldonado, A.J. Lithium Functionalization Promoted by Amide-Containing Ligands of a Cu(pzdc)(pia) Porous Coordination Polymer for CO2 Adsorption Enhancement. Cryst. Growth Des. 2020, 20, 3898–3912. [Google Scholar] [CrossRef]
- Li, F.-C.; Li, X.-L.; Tan, L.-K.; Wang, J.-T.; Yao, W.-Z. Evans–Showell-type polyoxometalate-based metal–organic complexes with novel 3D structures constructed from flexible bis-pyrazine-bis-amide ligands and copper metals: Syntheses, structures, and fluorescence and catalytic properties. Dalton Trans. 2019, 48, 2160–2169. [Google Scholar] [CrossRef]
- Hearne, N.; Turnbull, M.M.; Landee, C.P.; van der Merwe, E.M.; Rademeyer, M. Halide-bi-bridged polymers of amide substituted pyridines and -pyrazines: Polymorphism, structures, thermal stability and magnetism. CrystEngComm 2019, 21, 1910–1927. [Google Scholar] [CrossRef]
- Kumar, S.; Sharma, R.P.; Venugopalan, P.; Ferretti, V.; Tarpin, M.; Sayen, S.; Guillon, E. New copper(II) niflumate complexes with N-donor ligands: Synthesis, characterization and evaluation of anticancer potential against human cell lines. Inorg. Chim. Acta 2019, 488, 260–268. [Google Scholar] [CrossRef]
- Kwiatek, D.; Kubicki, M.; Skokowski, P.; Gruszczyńska, J.; Lis, S.; Hnatejko, Z. Five subsequent new pyridine carboxamides and their complexes with d-electron ions. Synthesis, spectroscopic characterization and magnetic properties. J. Mol. Struct. 2019, 1178, 669–681. [Google Scholar] [CrossRef]
- Kwiatek, D.; Kubicki, M.; Belter, J.; Jastrząb, R.; Wiśniewska, H.; Lis, S.; Hnatejko, Z. Synthesis, spectroscopic characterization and antifungal activity studies of five novel complexes with pyridine carboxamides. Polyhedron 2017, 133, 187–194. [Google Scholar] [CrossRef]
- Nguyen, T.V.; Ong, T.D.; Lam, A.H.M.; Pham, V.T.; Phan, N.T.S.; Truong, T. Nucleophilic trifluormethylation of aryl boronic acid under heterogeneous Cu(INA)2 catalysis at room temperature: The catalytic copper-based protocol. Mol. Catal. 2017, 436, 60–66. [Google Scholar] [CrossRef]
- Chen, Y.; Li, L.; Li, J.; Ouyang, K.; Yang, J. Ammonia capture and flexible transformation of M-2(INA) (M = Cu, Co, Ni, Cd) series materials. J. Hazard. Mater. 2016, 306, 340–347. [Google Scholar] [CrossRef]
- Chen, Y.; Li, L.; Yang, J.; Wang, S.; Li, J. Reversible flexible structural changes in multidimensional MOFs by guest molecules (I2, NH3) and thermal stimulation. J. Solid Stat. Chem. 2015, 226, 114–119. [Google Scholar] [CrossRef]
- Zurowska, B. Structural and magnetic characterization of Cu–picolinate and Cu–quinaldinate and their mixed complexes with water or halides. Inorg. Chim. Acta 2014, 418, 136–152. [Google Scholar] [CrossRef]
- Wang, P.; Li, G.; Chen, Y.; Chen, S.; James, S.L.; Yuan, W. Mechanochemical interconversion between discrete complexes and coordination networks—Formal hydration/dehydration by LAG. CrystEngComm 2012, 14, 1994–1997. [Google Scholar] [CrossRef]
- He, Y.-C.; Yang, J.; Yang, G.-C.; Kan, W.-Q.; Ma, J.-F. Solid-state single-crystal-to-single-crystal transformation from a 2D layer to a 3D framework mediated by lattice iodine release. Chem. Commun. 2012, 48, 7859–7861. [Google Scholar] [CrossRef] [PubMed]
- Lian, T.T.; Chen, S.-M. A new microporous Cu(II)-isonicotinate framework with 8-connected bcu topology. Inorg. Chem. Commun. 2012, 18, 8–10. [Google Scholar] [CrossRef]
- Monfared, H.H.; Vahedpour, M.; Yeganeh, M.M.; Ghorbanloo, M.; Mayer, P.; Janiak, C. Concentration dependent tautomerism in green [Cu(HL1)(L2)] and brown [Cu(L1)(HL2)] with H2L1 = (E)-N′-(2-hydroxy-3-methoxybenzylidene)-benzoylhydrazone and HL2 = pyridine-4-carboxylic (isonicotinic) acid. Dalton Trans. 2011, 40, 1286–1294. [Google Scholar] [CrossRef]
- Dakovic, M.; Jaglicic, Z.; Kozlevcar, B.; Popovic, Z. Association of copper(II) isonicotinamide moieties via different anionic bridging ligands: Two paths of ferromagnetic interaction in the azide coordination compound. Polyhedron 2010, 29, 1910–1917. [Google Scholar] [CrossRef]
- Dakovic, M.; Popovic, Z. Uncommon isonicotinamide supramolecular synthons in copper(II) complexes directed by nitrate and perchlorate anions. Acta Cryst. 2009, C65, m361–m366. [Google Scholar]
- Svorec, J.; Lörinc, S.; Moncol, J.; Melnik, M.; Koman, M. Structural and spectroscopic characterization of copper(II) tolfenamate complexes. Trans. Met. Chem. 2009, 34, 703–710. [Google Scholar] [CrossRef]
- Lörinc, S.; Švorec, J.; Melnik, M.; Koman, M. Structure and spectral characterisation of copper(II) meclofenamate complexes. Polyhedron 2008, 27, 3545–3548. [Google Scholar] [CrossRef]
- Kavalírova, J.; Korabik, M.; Stachová, P.; Moncol, J.; Sillanpää, R.; Lis, T.; Miklos, D.; Melník, M.; Mrozinski, J.; Valigura, D. Synthesis, spectral and magnetic properties of two different 2-nitrobenzoatocopper(II) complexes containing N,N-diethylnicotinamide. Polyhedron 2008, 27, 1333–1342. [Google Scholar] [CrossRef]
- Moncol, J.; Mudra, M.; Lönnecke, P.; Hewitt, M.; Valko, M.; Morris, H.; Svorec, J.; Melnik, M.; Mazur, M.; Koman, M. Crystal structures and spectroscopic behavior of monomeric, dimeric and polymeric copper(II) chloroacetate adducts with isonicotinamide, N-methylnicotinamide and N,N-diethylnicotinamide. Inorg. Chim. Acta 2007, 360, 3213–3225. [Google Scholar] [CrossRef]
- Pichon, A.; Lazuen-Garay, A.; James, S.L. Solvent-free synthesis of a microporous metal–organic framework. CrystEngComm 2006, 8, 211–214. [Google Scholar] [CrossRef]
- Atac, A.; Yurdakul, S.; Ide, S. Synthesis and vibrational spectroscopic studies of isonicotinamide metal(II) halide complexes. J. Mol. Struct. 2006, 783, 79–87. [Google Scholar] [CrossRef]
- Li, C.-B.; Liu, B.; Gao, G.-G.; Che, G.-B. Hydrogen bonding and π–π stacking in di-µ-isophthalato-bis[bis(isonicotinamide)copper(II)] trihydrate. Acta Cryst. 2005, E61, m1705–m1707. [Google Scholar]
- Moncol, J.; Kalinakova, B.; Svorec, J.; Kleinova, M.; Koman, M.; Hudecova, D.; Melnik, M.; Mazur, M.; Valko, M. Spectral properties and bio-activity of copper(II) clofibriates, part III: Crystal structure of Cu(clofibriate)2(2-pyridylmethanol)2, Cu(clofibriate)2(4-pyridylmethanol)2(H2O) dihydrate, and Cu2(clofibriate)4(N,N-diethylnicotinamide)2. Inorg. Chim. Acta 2004, 357, 3211–3222. [Google Scholar] [CrossRef]
- Goher, M.A.S.; Mautner, F.A. Spectral and structural characterization of copper(I) halide, nitrate and perchlorate complexes of pyrazine carboxamide (pyza) and X-ray crystal structure of polymeric [Cu(pyza)2I]n complex. Polyhedron 2000, 19, 601–606. [Google Scholar] [CrossRef]
- Aakeroy, C.B.; Beatty, A.M.; Leinen, D.S.; Lorimer, K.R. Deliberate combination of coordination polymers and hydrogen bonds in a supramolecular design strategy for inorganic/organic hybrid networks. Chem. Commun. 2000, 935–936. [Google Scholar] [CrossRef]
- Goher, M.A.S.; Abu-Youssef, M.A.M.; Mautner, F.A. 1D polymeric copper(II) complexes containing bridging tridentate pyrazinato and terminal chloro or azido anions. Synthesis, spectral, structural and thermal study of [CuCl(pyrazinato)(H2O)]n and [Cu(N3)(pyrazinato)(H2O)]n complexes. Polyhedron 1998, 17, 3305–3314. [Google Scholar] [CrossRef]
- Goher, M.A.S.; Al-Salem, N.A.; Mautner, F.A.; Klepp, K.O. A copper(II) azide compound of pyrazinic acid containing a new dinuclear complex anion [Cu2(N3)6]2–. Synthesis, spectral and structural study of KCu2(pyrazinato)(N3)4. Polyhedron 1997, 16, 825–831. [Google Scholar] [CrossRef]
- Goher, M.A.S.; Mautner, F.A. New Unexpected Coordination Modes of Azide and Picolinato Anions Acting as Bridging Ligands Between Copper(II) and Sodium or Potassium Ions. Synthesis, Crystal Structures and Spectral Characterizations of [MCu(picolinato)(N3)2]n (M = Na or K) Complexes. Polyhedron 1995, 14, 1439–1446. [Google Scholar] [CrossRef]
- Goher, M.A.S.; Mautner, F.A. Spectroscopic and Crystal Structure Study of NaCu(Picolinato)2(N3)(H2O)2. A Polymeric Structure Containing Simultaneous Bridging Pentadentate Picolinato Anion and µ(1,3) Azido Ligands Between Copper and Sodium Centred Polyhedra. Polyhedron 1994, 13, 2149–2155. [Google Scholar] [CrossRef]
- Sileo, E.E.; Morando, P.J.; Della Vedova, C.; Blesa, M.A. The Thermal Decomposition of Copper(II) Nicotinate and Isonicotinate. Thermochim. Acta 1989, 138, 233–239. [Google Scholar] [CrossRef]
- Sekizaki, M. The Crystal Structure of Bis(pyrazine-2-carboxamide)copper(II) Perchlorate. Acta Cryst. B 1973, B29, 327–331. [Google Scholar] [CrossRef]
- Mukherjee, R. Comprehensive Coordination Chemistry II; Chap. 6.6; Jon, A., McCleverty, J.A., Meyer, T.J., Eds.; Elsevier: Oxford, UK, 2004; Volume 6, p. 747910. [Google Scholar]
- Kaim, W.; Schwederski, B.; Klein, A. Bioinorganic Chemistry: Inorganic Elements in the Chemistry of Life, 2nd ed.; Wiley: Chichester, UK, 2013. [Google Scholar]
- Kaim, W.; Rall, J. Copper—A “Modern” Bioelement. Angew. Chem. Int. Ed. 1996, 35, 43–60. [Google Scholar] [CrossRef]
- Erxleben, A. Interactions of copper complexes with nucleic acids. Coord. Chem. Rev. 2018, 360, 92–121. [Google Scholar] [CrossRef]
- Girma, K.B.; Lorenz, V.; Blaurock, S.; Edelmann, F.T. Coordination chemistry of acrylamide. Coord. Chem. Rev. 2005, 249, 1283–1293. [Google Scholar] [CrossRef]
- Allan, J.R.; McCloy, B.; Paton, A.D. Preparation, thermal, structural and electrical studies of dichlorohexa(anthranilamide) cobalt(II) and dichloro(anthranilamide) copper(II). Thermochim. Acta 1994, 231, 121–128. [Google Scholar] [CrossRef]
- Ahuja, I.S.; Singh, R.; Rai, C.P. Complexes of Copper(II) with Nicotinic Acid and some Related Ligands. Trans. Met. Chem. 1977, 2, 257–260. [Google Scholar] [CrossRef]
- Damous, M.; Denes, G.; Bouacida, S.; Hamlaoui, M.; Merazig, H.; Darand, J.-C. Di-µ-chlorido-bis[(2-aminobenzamide-κ2N2,O)chloridocopper(II)]. Acta Cryst. E 2013, E69, m488. [Google Scholar] [CrossRef]
- Pérez, A.L.; Neuman, N.I.; Baggio, R.; Ramos, C.A.; Dalosto, S.D.; Rizzi, A.C.; Brondino, C.D. Exchange interaction between S = 1/2 centers bridged by multiple noncovalent interactions: Contribution of the individual chemical pathways to the magnetic coupling. Polyhedron 2017, 123, 404–410. [Google Scholar] [CrossRef]
- Halcrow, M.A. Jahn-Teller Distortions in Transition Metal Compounds, and Their Importance in Functional Molecular and Inorganic Materials. Chem. Soc. Rev. 2013, 42, 1784–1795. [Google Scholar] [CrossRef]
- Klein, A.; Butsch, K.; Elmas, S.; Biewer, C.; Heift, D.; Nitsche, S.; Schlipf, I.; Bertagnolli, H. Oxido-pincer complexes of copper(II)—An EXAFS and EPR study of mono- and binuclear [(pydotH2)CuCl2]n (n = 1 or 2). Polyhedron 2012, 31, 649–656. [Google Scholar] [CrossRef]
- Thakurta, S.; Roy, P.; Rosair, G.; Gómez-García, C.J.; Garribba, E.; Mitra, S. Ferromagnetic exchange coupling in a new bis(µ-chloro)-bridged copper(II) Schiff base complex: Synthesis, structure, magnetic properties and catalytic oxidation of cycloalkanes. Polyhedron 2009, 28, 695–702. [Google Scholar] [CrossRef]
- Klein, A.; Elmas, S.; Butsch, K. Oxido Pincer Ligands—Exploring the Coordination Chemistry of Bis(hydroxymethyl)pyridine Ligands for the Late Transition Metals. Eur. J. Inorg. Chem. 2009, 2009, 2271–2281. [Google Scholar] [CrossRef]
- Yraola, F.; Albericio, F.; Corbella, M.; Royo, M. [{Cu(pzPh)(Opo)}2(μ-Cl)2]: A new dinuclear copper(II) complex with a chloride bridge and mixed blocking ligands. Inorg. Chim. Acta 2008, 361, 2455–2461. [Google Scholar] [CrossRef]
- Koval, I.A.; Sgobba, M.; Huisman, M.; Lüken, M.; Saint-Aman, E.; Gamez, P.; Krebs, B.; Reedijk, J. A remarkable anion effect on the crystal packing of two analogous copper complexes from a thiophene-containing phenol-based ligand. Inorg. Chim. Acta 2006, 359, 4071–4078. [Google Scholar] [CrossRef]
- Kilner, C.A.; McInnes, E.J.L.; Leech, M.A.; Beddard, G.S.; Howard, J.A.K.; Mabbs, F.E.; Collison, D.; Bridgeman, A.J.; Halcrow, M.A. A crystallographic, EPR and theoretical study of the Jahn–Teller distortion in [CuTp2] (Tp– = tris{pyrazol-1-yl}hydridoborate). Dalton Trans. 2004, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.L.; Stevens, E.D.; O’Connor, C.I.; Majeste, R.J.; Trefonas, L.M. Magnetic Properties and Molecular Structure of Copper(II) Complexes of 2,3-Pyrazinedicarboxamide. Inorg. Chim. Acta 1983, 70, 151–158. [Google Scholar]
- Laussmann, T.; Grzesiak, I.; Krest, A.; Stirnat, K.; Meier-Giebing, S.; Ruschewitz, U.; Klein, A. Copper Thiocyanate Complexes and Cocaine—A Case of Black Cocaine. Drug Test. Anal. 2015, 7, 56–64. [Google Scholar] [CrossRef]
- Glowiak, T.; Podgórska, I. X-ray, Spectroscopic and Magnetic Studies of Hexaaquacopper(II) Di(diphenylphosphate) Diglycine. Inorg. Chim. Acta 1986, 125, 83–88. [Google Scholar] [CrossRef]
- Hathaway, B.; Billing, D. The electronic properties and stereochemistry of mono-nuclear complexes of the copper(II) ion. Coord. Chem. Rev. 1970, 5, 143–207. [Google Scholar] [CrossRef]
- Rieger, P.H. Electron Spin Resonance—Analysis and Interpretation; RCS Publishing: Cambridge, UK, 2007. [Google Scholar]
- Halcrow, M.A.; Kilner, C.A.; Wolowska, J.; McInnes, E.J.L.; Bridgeman, A.J. Temperature dependence of the electronic ground states of two mononuclear, six-coordinate copper(II) centres. New J. Chem. 2004, 28, 228–233. [Google Scholar] [CrossRef]
- Solanki, N.K.; Leech, M.A.; McInnes, E.J.L.; Mabbs, F.E.; Howard, J.A.K.; Kilner, C.A.; Rawson, J.M.; Halcrow, M.A. A crystallographic and EPR study of the fluxional Cu(II) ion in [CuL2][BF4]2 (L = 2,6-dipyrazol-1-ylpyridine). J. Chem. Soc. Dalton Trans. 2002, 1295–1301. [Google Scholar] [CrossRef]
- Mirzaei, M.; Lippolis, V.; Aragoni, M.C.; Ghanbari, M.; Shamsipur, M.; Meyer, F.; Demeshko, S.; Pourmortazavi, S.M. Extended structures in copper(II) complexes with 4-hydroxypyridine-2,6-dicarboxylate and pyrimidine derivative ligands: X-ray crystal structure, solution and magnetic studies. Inorg. Chim. Acta 2014, 418, 126–135. [Google Scholar] [CrossRef]
- Sheldrick, G.M. ShelXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A Found. Crystallogr. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. SHELXL-2017/1, Program for the Solution of Crystal Structures; University of Göttingen: Göttingen, Germany, 2017. [Google Scholar]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- STOE X-RED. Data Reduction Program; Version 1.31/Windows; STOE & Cie: Darmstadt, Germany, 2005. [Google Scholar]
- STOE X-SHAPE. Crystal Optimisation for Numerical Absorption Correction; Version 1.06/Windows; STOE & Cie: Darmstadt, Germany, 1999. [Google Scholar]
- STOE WinXPOW 1.07; STOE & Cie GmbH: Darmstadt, Germany, 2000.
- Williams, T.; Kelley, C.; Lang, R. Gnuplot 4.6—An Interactive Plotting Program. 2012. Available online: http://gnuplot.info/ (accessed on 23 November 2020).
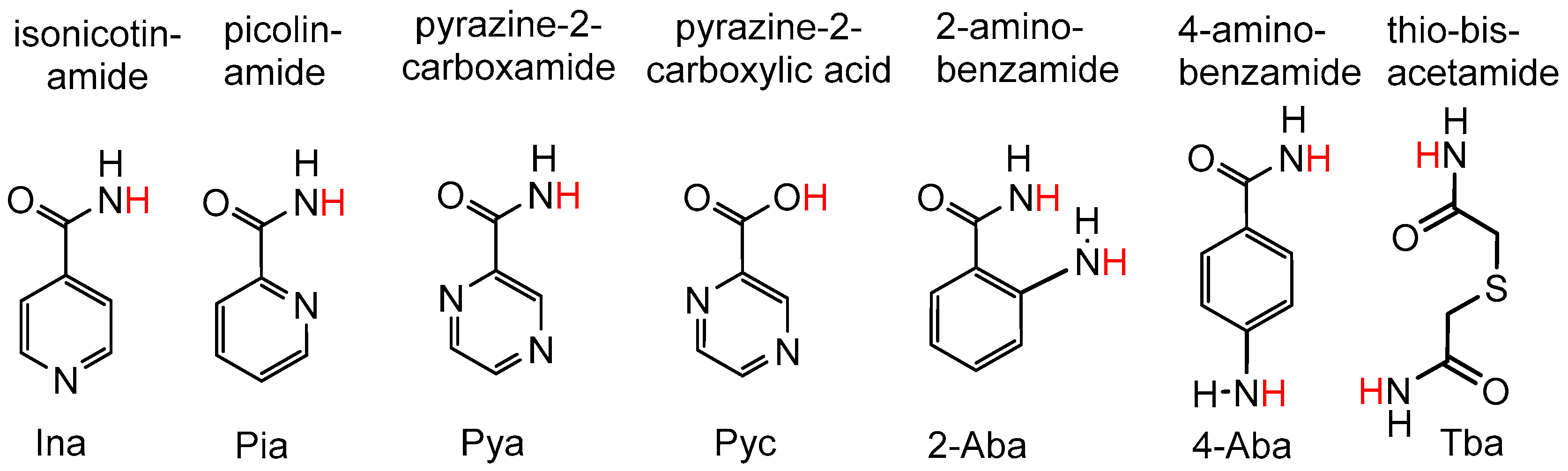



 . Yields were defined relative to the ligands (for details, see Experimental Section).
. Yields were defined relative to the ligands (for details, see Experimental Section).



 . Yields were defined relative to the ligands (for details, see Experimental Section).
. Yields were defined relative to the ligands (for details, see Experimental Section).
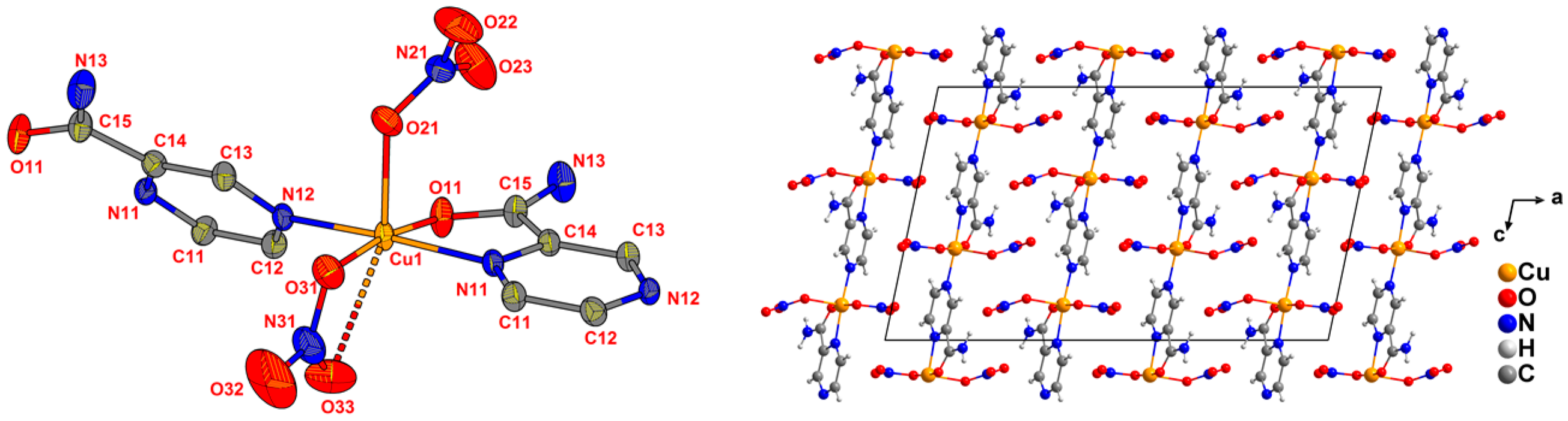
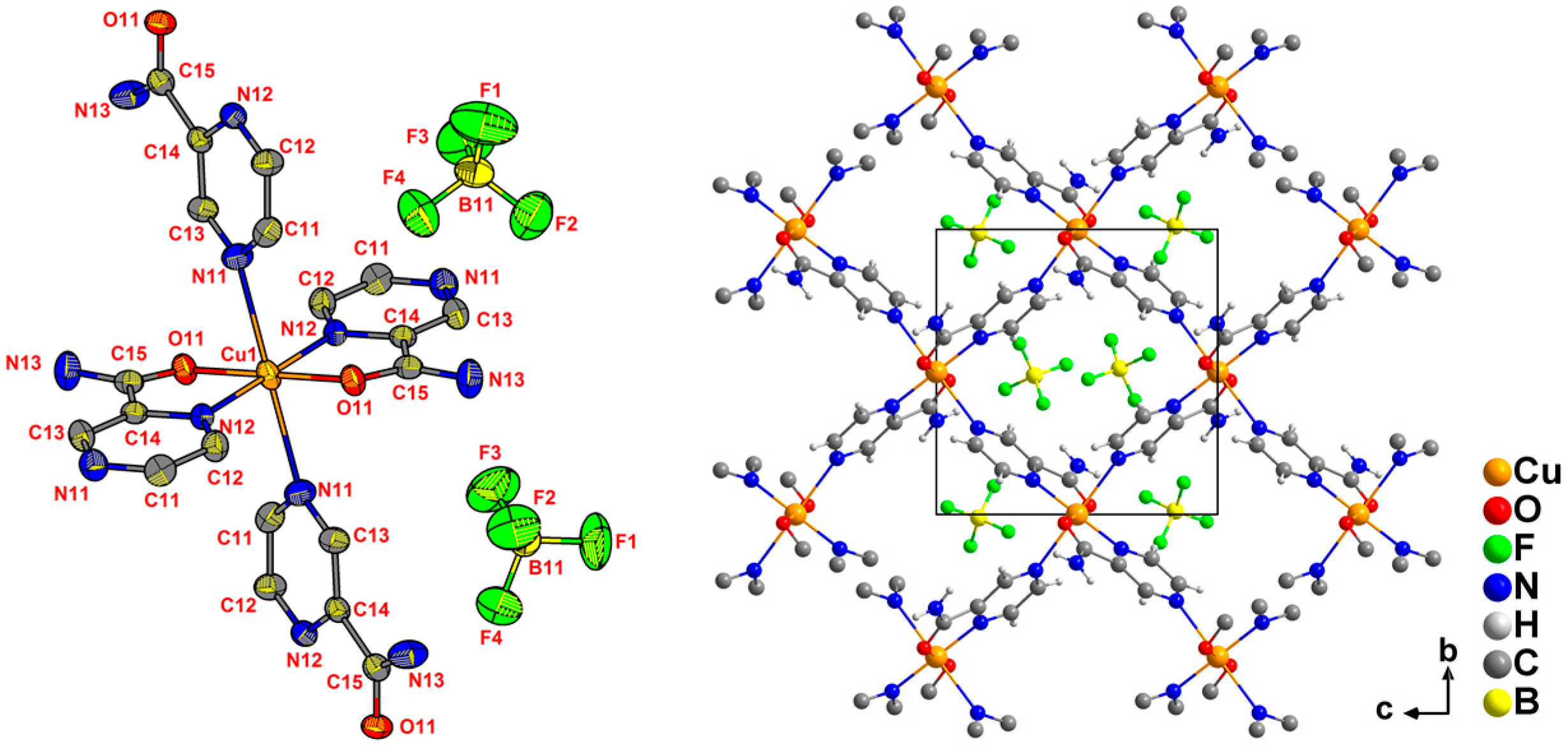
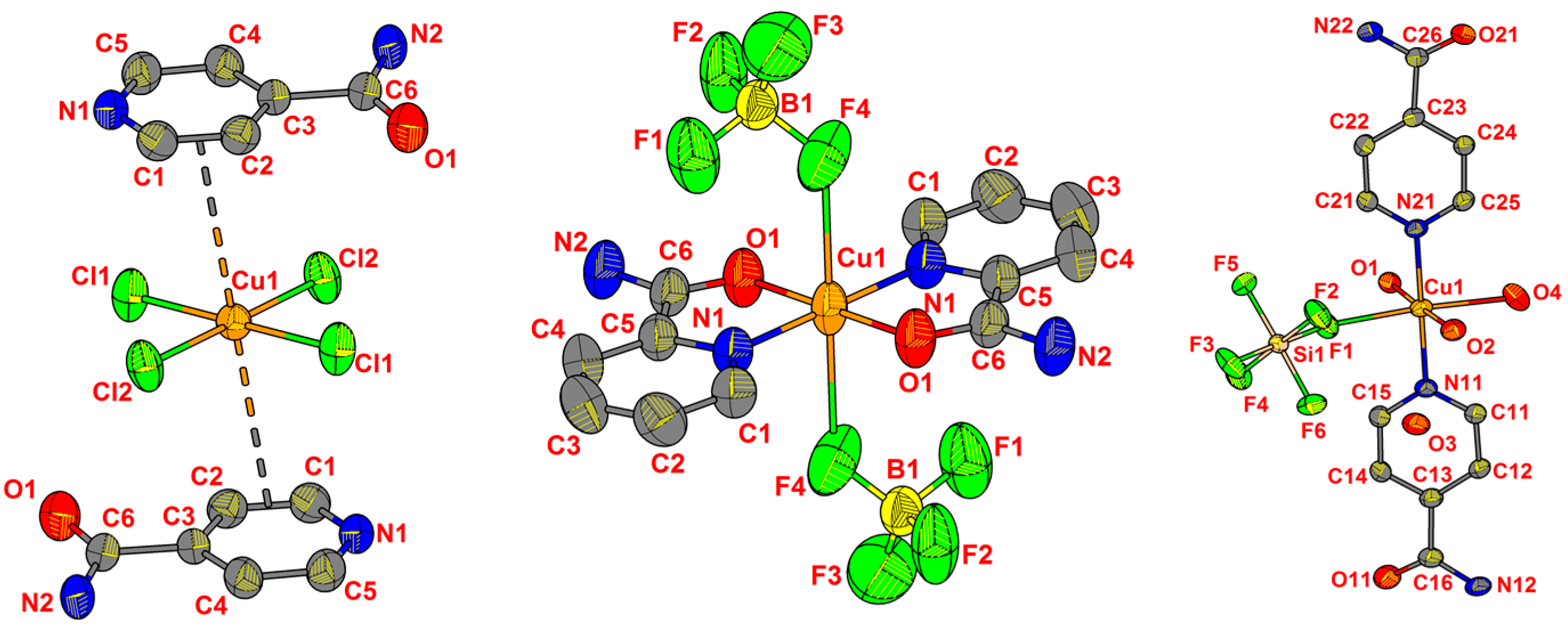

| Ligand | CuII Precursor | Compound | Formula (Weight) | Space Group |
|---|---|---|---|---|
| Ina | Cu(BF4)2∙6H2O | [Cu(Ina)4(H2O)2](BF4)2 (1) | C24H28B2Cu1F8N8O6 (761.69) | C2/c (No. 15) d |
| Ina | Cu(NO3)2∙3H2O | [Cu(Ina)2(NO3)2] (2) | C12H12Cu1N6O8 (431.81) | P212121 (No. 19) |
| Ina | Cu(NO3)2∙3H2O | [Cu(Ina)2(H2O)2(NO3)2] (3) | C12H16Cu1N6O10 (467.84) | P21/c (No. 14) e |
| Ina | Cu(BF4)2∙6H2O | [Cu(Ina)2(H2O)3(SiF6)]·H2O (4) | C12H20Cu1F6N4O6Si1 (521.94) | P-1 (No. 2) |
| Ina | [Cu(Ina)2(NO3)2] | [Cu(Ina)2Br2] (5) | C12H12Br2Cu1N4O2 (467.61) | P21 (No. 4) f |
| HIna+ a | CuCl2∙2H2O | (HIna)2[CuCl4]∙2H2O (6) | C12H18Cl4CuN4O4 (487.64) | P-1 (No. 2) |
| HIna+ a | CuCl2∙2H2O | (HIna)[Cu(H2O)Cl3] (7) | C6H9Cl3Cu1N2O2 (311.05) | P-1 (No. 2) |
| Pia | Cu(BF4)2∙6H2O | [Cu(Pia)2(BF4)2] (8) | C12H12B2Cu1F8N4O2 (481.41) | P-1 (No. 2) |
| Pya | Cu(BF4)2∙6H2O | [Cu(Pya)2](BF4)2 (9) | C10H10B2Cu1F8N6O2 (483.38) | P21/c (No. 14) |
| Pya | Cu(NO3)2∙3H2O | [Cu(Pya)2(H2O)(NO3)](NO3) (10) | C10H12Cu1N8O9 (451.80) | P-1 (No. 2) |
| Pya | Cu(NO3)2∙3H2O | [Cu(Pya)(NO3)2] (11) | C5H5Cu1N5O7 (310.67) | C2/c (No. 15) |
| Pyc− b | Cu(Tfa)2∙H2O | [Cu(Pyc)(Tfa)] (12) | C7H3Cu1F3N2O4 (299.65) | P212121 (No. 19) |
| 2-Aba | Cu(NO3)2∙3H2O | [Cu(2-Aba)2(NO3)2] (13) | C14H16Cu1N6O8 (459.86) | P21/n (No. 14) g |
| 4-Aba | Cu(NO3)2∙3H2O | [Cu2(4-Aba)2(H2O)3(NO3)3](NO3) (14) | C14H22Cu2N8O17 (701.46) | C2/c (No. 15) h |
| 4-Aba | Cu(ClO4)2∙6H2O | [Cu(4-Aba)2(EtOH)2](ClO4)2 (15) | C18H28Cl2Cu1N4O12 (626.88) | P21/c (No. 14) |
| 4-Aba | Cu(BF4)2∙6H2O | [Cu(4-Aba)2(EtOH)2](BF4)2 (16) | C18H28B2Cu1F8N4O4 (601.60) | P21/c (No. 14) |
| 4-HAba+ c | Cu(ClO4)2∙6H2O + HCl | [Cu(4-HAba)2Cl4] (17) | C14H18Cl4Cu1N4O2 (479.67) | P21/c (No. 14) |
| Tba | Cu(ClO4)2∙6H2O | [Cu(Tba)2](ClO4)2 (18) | C8H16Cl2Cu1N4O12S2 (558.80) | C2/m (No. 12) |
| Complex | Geometry a | T (K) | Cu–L1 b | Cu–L2 | Cu–L3 | Cu–L4 | Cu–L5 | Cu–L6 |
|---|---|---|---|---|---|---|---|---|
| L1–Cu–L2 | L2–Cu–L3 | L3–Cu–L4 | L1–Cu–L5 | L1–Cu–L6 | L1–Cu–L3 | L5–Cu–L6 | ||
| [Cu(Ina)4(H2O)2](BF4)2 (1) | OE | 298(2) | 2.024(3) N21 | 2.045(4) N11 | =N11 | =N21 | 2.392(5) O1 | 2.645(5) O2 |
| 91.7(1)° | 88.9(2)° | 91.7(1)° | 86.5(1)° | 93.4(1)° | 173.0(1)° | 180° | ||
| [Cu(Ina)2(NO3)2] (2) | SPy+1 | 298(2) | 1.987(5) O31 | 1.994(5) N21 | 2.002(5) N11 | 2.004(4) O41 | 2.314(4) O11 | - |
| 90.1(2)° | 87.7(2)° | 89.1(2)° | 78.9(2)° | - | 165.5(2)° | - | ||
| [Cu(Ina)2(H2O)2(NO3)2] (3) c | OE | 296(2) | 1.984(1) O2 | 1.992(1) N1 | 2.507(1) O3 | |||
| 90.0(1)° | 90.0(1)° | 95.6(1)° | 84.4(1)° | 180° | 180° | |||
| [Cu(Ina)2(H2O)3(SiF6)]·H2O (4) | OE | 298(2) | 1.969(2) O1 | 1.986(2) O2 | 1.999(2) N21 | 2.026(2) N11 | 2.409(2) F1 | 2.513(2) O4 |
| 90.1(1)° | 89.6(1)° | 88.2(1)° | 88.2(1)° | 84.9(1)° | 180° | 170.9(1)° | ||
| (HIna)2[CuCl4]∙2H2O (6) | SP+2 | 298(2) | 2.263(1) Cl1 | 2.270(1) Cl2 | =Cl1 | =Cl2 | 3.360(1) = (py) | =L5 |
| (HIna)[Cu(H2O)Cl3] (7) c | SPy | 298(2) | 1.980(4) O1 | 2.251(1) Cl2 | 2.268(1) Cl1 | 2.279(1) Cl3 | 2.923(1) Cl2′ | - |
| 87.4(1)° | 93.2(1)° | 92.6(1)° | - | - | 172.5(1)° | - | ||
| [Cu(Pia)2(BF4)2] (8) | OE | 150(2) | 1.935(3) O1 | 1.958(4) N1 | =O1 | =N1 | 2.589(3) F4 | =F4 |
| 83.3(2)° | 96.6(1)° | 83 (2)° | 89.7(1)° | 90.3(1)° | 180° | 180° | ||
| [Cu(Pya)2](BF4)2 (9) | OE | 298(2) | 1.957(2) O11 | 1.984(3) N12 | =O11 | =N12 | 2.447(3) N11 | =N11 |
| 82.2(1)° | 97.7(1)° | 82.2(1)° | 85.5(1)° | 94.4(1)° | 180° | 180° | ||
| [Cu(Pya)2(H2O)(NO3)](NO3) (10) | Spy+1 | 298(2) | 1.935(4) O11 | 1.938(4) O21 | 1.984(4) N21 | 1.989(4) N11 | 2.234(4) O5 | 2.818(4) O43 |
| 82.7(1)° | 97.6(1)° | 82.7(1)° | 94.3(2)° | 89.7(1)° | 172.1(1)° | 169.8(1)° | ||
| [Cu(Pya)(NO3)2] (11) | SPy | 298(2) | 1.953(2) O11 | 1.979(2) O31 | 1.998(2) N12 | 2.004(2) N11 | 2.238(2) O21 | 2.572(3) O33 |
| 81.6(1)° | 89.3(1)° | 92.3(1)° | 91.0(1)° | 93.6(1)° | 169.2(1)° | 136.9(1)° | ||
| [Cu(Pyc)(Tfa)] (12) d | SPy | 298(2) | 1.942(5) O1 | 1.944(7) O11 | 1.997(6) O12 | 2.065(8) N11 | 2.272(8) N12 | - |
| 88.8(2)° | 97.6(2)° | 92.7(3)° | 96.5(3)° | - | 171.3(2)° | - | ||
| [Cu(2-Aba)2(NO3)2] (13) | OE | 298(2) | 1.976(3) O1 | 2.010(4) N1 | =O1 | =N1 | 2.506(5) O3 | =O3 |
| 87.3(2)° | 92.7(2)° | 87.3(2)° | 92.8(1)° | 87.1(1)° | 180° | 180° | ||
| [Cu2(4-Aba)2(H2O)3(NO3)3](NO3) (14) e | OD | 298(2) | 1.942(4) O11 | 1.969(4) O21 | 2.016(4) N11 | 2.034(5) O21 | 2.262(4) O32 | 2.35(1) O23 |
| 89.1(2)° | 88.2(2)° | 93.3(2)° | 82.9(2)° | 95.2(3)° | 176.0(2)° | 143.1(3)° | ||
| [Cu(4-Aba)2(EtOH)2](ClO4)2 (15) | OE | 150(2) | 1.951(2) O11 | 2.077(4) N11 | =N11 | =O11 | 2.529(2) O21 | =O21 |
| 87.8(1)° | 92.1(1)° | 87.8(1)° | 94.4(1)° | 85.5(1)° | 180° | 180° | ||
| [Cu(4-Aba)2(EtOH)2](BF4)2 (16) | OE | 298(2) | 1.951(2) O11 | 2.062(3) N11 | =O11 | =N11 | 2.545(2) O1 | =O1 |
| 87.9(1)° | 92.1(1)° | 87.9(1)° | 87.3(1)° | 92.6(1)° | 180° | 180° | ||
| [Cu(4-HAba)2Cl4] (17) f | OE | 298(2) | 1.957(6) O1 | 2.313(3) Cl1 | =O1 | =Cl | 2.867(2) Cl2 | =Cl2 |
| 89.6(2)° | 90.4(2)° | 89.6(2)° | 98.2(2)° | 81.8(2)° | 180° | 180° | ||
| [Cu(Tba)2](ClO4)2 (18) | OE | 298(2) | 1.976(2) O11 | =O11 | =O11 | =O11 | 2.625(1) S11 | =S11 |
| 90.9(1)° | 89.1(1)° | 90.9(1)° | 81.5(1)° | 98.4(1)° | 180° | 180° |
| Compound | T (K) | gav | g‖/g1 | g2 | g⊥/g3 | Δg | Spectral Geom. | CuII Geom. |
|---|---|---|---|---|---|---|---|---|
| Group I | ||||||||
| [Cu(Ina)2(NO3)2] (2) | 298 | 2.143 | 2.280 | 2.074 | 0.206 | axial | SPy+1 | |
| [Cu(Ina)4(H2O)2](BF4)2 (1) b | 298 | 2.118 | 2.260 | 2.048 | 0.212 | axial | OE | |
| (HIna)2[CuCl4].2H2O (6) | 298 | 2.132 | 2.266 | 2.082 | 2.047 | 0.219 | rhombic | SP+2 |
| [Cu(Ina)2(H2O)3(SiF6)]·H2O (4) | 298 | 2.154 | 2.326 | 2.068 | 0.258 | axial | OE | |
| [Cu(Ina)2(H2O)2(NO3)2] (3) | 298 | 2.156 | 2.297 | 2.085 | 0.212 | axial | OE | |
| [Cu(4-HAba)2Cl4] (17) | 298 | 2.175 | 2.286 | 2.169 | 2.066 | 0.219 | rhombic | OE |
| [Cu(Pya)(NO3)2] (11) | 298 | 2.179 | 2.372 | 2.083 | 0.289 | axial | SPy | |
| [Cu(Pya)2(H2O)(NO3)](NO3) (10) | 298 | 2.183 | 2.400 | 2.075 | 0.325 | axial | SPy+1 | |
| similar compounds | ||||||||
| [Cu(CCl3COO)2(MNA)2]∙2H2O c | 298 | 2.143 | 2.280 | 2.075 | 0.205 | axial | OE | |
| [Cu(Ina)2(µ-N,S-SCN)2] d | 298 | 2.20 | 2.27 | 2.07 | 0.200 | axial | OE | |
| [Cu(meclof)2(2-pyca)2] e | 298 | 2.142 | 2.290 | 2.068 | 0.222 | axial | OE | |
| [Cu(clof)2(4-pymeth)2(H2O)] 2H2O f | 298 | 2.128 | 2.271 | 2.054 | 0.217 | axial | SPy | |
| [Cu(clof)2(Et2nia)2] f | 298 | 2.132 | 2.289 | 2.053 | 0.236 | axial | g | |
| [Cu(clof)2(Ina)2] f | 298 | 2.153 | 2.285 | 2.087 | 0.198 | axial | g | |
| [Cu(tolf)2(Et2nia)2)(H2O)2] h | 298 | 2.130 | 2.294 | 2.048 | 0.246 | axial | OE | |
| [Cu(tolf)2(Nia)2] h | 298 | 2.157 | 2.319 | 2.076 | 0.243 | axial | g | |
| Group II | ||||||||
| [Cu(Pya)2](BF4)2 (9) | 298 | 2.140 | 2.204 | 2.139 | 2.078 | 0.126 | rhombic | OE |
| [Cu(4-Aba)2(EtOH)2](ClO4)2 (15) | 110 | 2.137 | 2.231 | 2.123 | 2.059 | 0.172 | rhombic | OE |
| [Cu(4-Aba)2(EtOH)2](ClO4)2 (15) | 298 | 2.111 | 2.200 | 2.066 | 0.133 | axial | OE | |
| [Cu(4-Aba)2(EtOH)2](BF4)2 (16) | 110 | 2.139 | 2.220 | 2.140 | 2.058 | 0.162 | rhombic | OE |
| [Cu(4-Aba)2(EtOH)2](BF4)2 (16) | 298 | 2.110 | 2.196 | 2.068 | 0.131 | axial | OE | |
| [Cu(TBA)2](ClO4)2 (18) | 298 | 2.130 | 2.228 | 2.080 | 0.147 | axial | OE | |
| similar compounds | ||||||||
| [Cu2(Ina)2(µ-1,1-N3)2(µ-1,3-N3)2] d | 298 | 2.17 | 2.22 | 2.07 | 0.150 | axial | SPy+1 | |
| [Cu(Ina)2(µ-1,1-N3)2(µ-O,O-SO4)]∙2H2O d | 298 | 2.17 | 2.24 | 2.18 | 2.09 | 0.150 | rhombic | SPy |
| Group III | ||||||||
| [Cu(Pyc)(Tfa)] (12) | 298 | 2.138 | 2.138 | 0 | isotropic | SPy | ||
| [Cu(Ina)2Br2] (5) | 298 | 2.111 | 2.111 | 0 | isotropic | SPy i | ||
| Group IV | ||||||||
| [Cu2(Aba)2(H2O)3(NO3)3](NO3) (14) | 298 | 2.141 | 2.190 | 2.050 | 0.112 | inv. axial | OD |
| Compound | μB | θCW (K) | EPR Symmetry | Symmetry Around CuII |
|---|---|---|---|---|
| [Cu(Ina)2(NO3)2] (2) | 1.50 | 36.9 | axial | SPy + 1 |
| [Cu(Pya)(NO3)2] (11) | 1.65 | 6.55 | isotropic | SPy |
| [Cu(Pyc)(Tfa)] (12) | 1.56 | 48.3 | isotropic | SPy |
| [Cu(Aba)2(EtOH)2](ClO4)2 (15) | 1.52 | 42.8 | axial | OE |
| Compound | λ (nm/cm−1) of d-d Bands | Colour of the Crystals | Symmetry Around CuII b |
|---|---|---|---|
| [Cu(Pya)2](BF4)2 (9) | 613/16,310 | blue | OE |
| [Cu(Pya)2(H2O)(NO3)](NO3) (10) | 640/15,630 | blue | SPy + 1 |
| [Cu(Pya)(NO3)2] (11) | 780/12,820 | blue | SPy |
| [Cu(Pyc)(Tfa)] (12) | 664/15,060 | turquoise | SPy |
| [Cu(Ina)2Br2] (5) | 691/14,470 | dark green | - b |
| similar compounds | |||
| [Cu(tolf)2(Et2nia)2)(H2O)2] c | 614/16,290 | blue | OE |
| [Cu(meclof)2(2-pyca)2] d | 615/16,260 | blue | OE |
| [Cu(meclof)2(Et2nia)2] d | 605/16,530 | blue | - e |
| [Cu(clof)2(4-pymeth)2(H2O)] 2H2O f | 620/16,130 | blue | SPy |
| [Cu(clof)2(Ina)2] f | 660/15,150 | blue | - e |
| [Cu(clof)2(Et2nia)2] f | 603/16,580 | violet | - e |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wackerbarth, I.; Widhyadnyani, N.N.A.T.; Schmitz, S.; Stirnat, K.; Butsch, K.; Pantenburg, I.; Meyer, G.; Klein, A. CuII Complexes and Coordination Polymers with Pyridine or Pyrazine Amides and Amino Benzamides—Structures and EPR Patterns. Inorganics 2020, 8, 65. https://doi.org/10.3390/inorganics8120065
Wackerbarth I, Widhyadnyani NNAT, Schmitz S, Stirnat K, Butsch K, Pantenburg I, Meyer G, Klein A. CuII Complexes and Coordination Polymers with Pyridine or Pyrazine Amides and Amino Benzamides—Structures and EPR Patterns. Inorganics. 2020; 8(12):65. https://doi.org/10.3390/inorganics8120065
Chicago/Turabian StyleWackerbarth, Ines, Ni Nyoman Agnes Tri Widhyadnyani, Simon Schmitz, Kathrin Stirnat, Katharina Butsch, Ingo Pantenburg, Gerd Meyer, and Axel Klein. 2020. "CuII Complexes and Coordination Polymers with Pyridine or Pyrazine Amides and Amino Benzamides—Structures and EPR Patterns" Inorganics 8, no. 12: 65. https://doi.org/10.3390/inorganics8120065
APA StyleWackerbarth, I., Widhyadnyani, N. N. A. T., Schmitz, S., Stirnat, K., Butsch, K., Pantenburg, I., Meyer, G., & Klein, A. (2020). CuII Complexes and Coordination Polymers with Pyridine or Pyrazine Amides and Amino Benzamides—Structures and EPR Patterns. Inorganics, 8(12), 65. https://doi.org/10.3390/inorganics8120065