Concise Review of Nickel Human Health Toxicology and Ecotoxicology
Abstract
:1. Nickel Occurrence and Uses
2. Nickel Exposure to Humans and Toxicokinetics
2.1. Nickel Exposures
2.1.1. Occupational
2.1.2. General Public
2.2. Toxicokinetics and Bioavailability of Nickel
2.2.1. Gastrointestinal
2.2.2. Respiratory
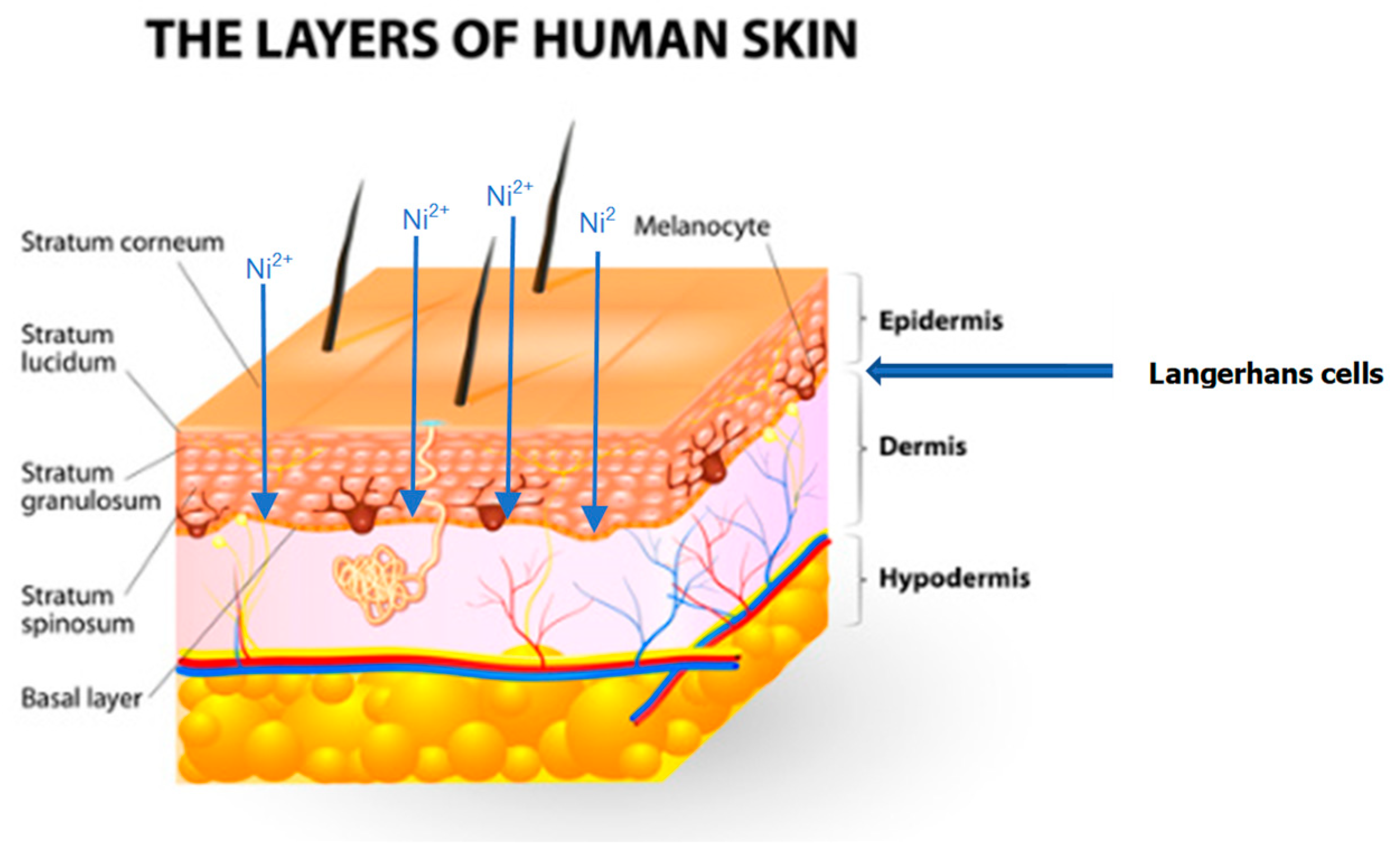
2.2.3. Dermal
2.3. Distribution, Metabolism and Excretion of Nickel
3. Toxicity of Nickel
3.1. Toxicity and Nickel Ion
3.2. Nickel Allergic Contact Dermatitis (NACD)
3.2.1. Prevalence in General and Clinical Populations
3.2.2. Induction vs. Elicitation
3.2.3. Mechanisms of Nickel ACD
3.2.4. Sources of Exposure: Nickel Release versus Content
3.2.5. Susceptible Populations
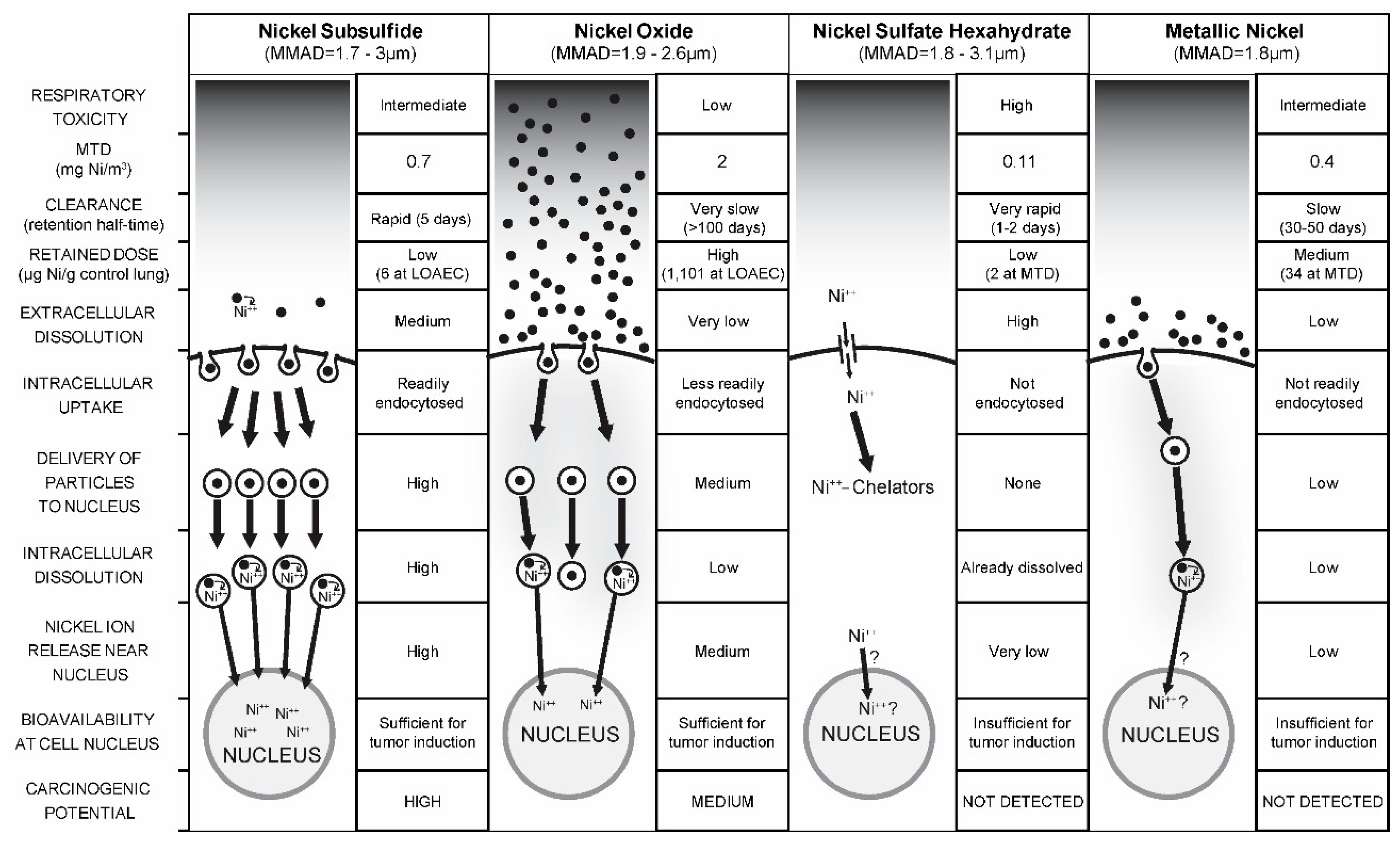
3.3. Nickel Carcinogenicity
3.3.1. Human and Animal Evidence for Nickel Carcinogenicity
3.3.2. Inhalation Exposure Route
3.3.3. Oral Exposure Route
3.3.4. Dermal Exposure Route
3.3.5. Other Exposure Routes
3.3.6. Nickel Compounds’ Genotoxic and Carcinogenic Mode of Action
3.4. Reproductive and Developmental Toxicity
3.4.1. Human Epidemiological Studies
3.4.2. Studies in Animals
3.4.3. Conclusions on Reproductive Toxicity
3.5. Non-Cancer Lung Effects
4. Nickel Exposure in the Environment
4.1. Exposure Sources in the Environment
4.2. Interactions between Nickel and Natural Chemical Parameters and the Concept of Bioavailability
4.2.1. Water
4.2.2. Sediment
4.2.3. Soil
4.3. Ecosystem-Specific Nickel Ecotoxicity
4.3.1. Identification, Screening and Aggregation of Nickel Ecotoxicity Data
4.3.2. Freshwater
4.3.3. Marine
4.3.4. Sediment
4.3.5. Soil
4.4. Mechanisms of Toxicity
4.5. Bioaccumulation and Trophic Transfer Potential
5. Emerging Issues
5.1. Mixtures
5.2. Nanoparticles
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Environmental Health Criteria 108. Nickel; Prepared as a Part of WHO’s International Programme on Chemical Safety; World Health Organization: Geneva, Switzerland, 1991; p. 383. [Google Scholar]
- European Chemicals Agency (ECHA). Annex 1–Background Document in Support of the Committee for Risk Assessment (RAC) for Evaluation of Limit Values for Nickel and Its Compounds in the Workplace; ECHA/RAC/A77-0-0000001412-86-189/F; European Chemicals Agency: Helsinki, Finland, 2018; pp. 1–211. [Google Scholar]
- Nickel Institute. Safe Use of Nickel in the Workplace: A Guide for Health Maintenance of Workers Exposed to Nickel, Its Compounds and Alloys, 3rd ed.; Nickel Institute: Brussels, Belgium, 2008. [Google Scholar]
- International Agency for Research on Cancer (IARC). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Nickel and Nickel Compounds Monograph; WHO Press: Geneva, Switzerland, 2017; Volume 100C, pp. 169–218. [Google Scholar]
- Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological Profile for Nickel; Department of Health and Human Services, Public Health Service: Atlanta, GA, USA, 2005; pp. 1–397.
- National Toxicology Program (NTP). Final Report on Carcinogens Background Document for Metallic Nickel and Certain Nickel Alloys, Proceedings of theMeeting of the NTP Board of Scientific Counselors Report on Carcinogens Subcommittee, Durham, NC, USA, 13–14 December 2000; Technology Planning and Management Corporation: Durham, NC, USA, 2000; pp. 1–102. [Google Scholar]
- De Brouwere, K.; Buekers, J.; Cornelis, C.; Schlekat, C.E.; Oller, A.R. Assessment of indirect human exposure to environmental sources of nickel: Oral exposure and risk characterization for systemic effects. Sci. Total Environ. 2012, 419, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Sunderman, F.W., Jr.; Hopfer, S.M.; Sweeney, K.R.; Marcus, A.H.; Most, B.M.; Creason, J. Nickel absorption and kinetics in human volunteers. Proc. Soc. Exp. Biol. Med. 1989, 191, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, G.D.; Søderberg, U.; Jørgensen, P.J.; Templeton, D.M.; Rasmussen, S.N.; Andersen, K.E.; Grandjean, P. Absorption and retention of nickel from drinking water in relation to food intake and nickel sensitivity. Toxicol. Appl. Pharmacol. 1999, 154, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Goodman, J.E.; Prueitt, R.L.; Thakali, S.; Oller, A.R. The nickel ion bioavailability model of the carcinogenic potential of nickel-containing substances in the lung. Crit. Rev. Toxicol. 2011, 41, 142–174. [Google Scholar] [CrossRef] [PubMed]
- Oller, A.R.; Oberdörster, G. Incorporation of particle size differences between animal studies and human workplace aerosols for deriving exposure limit values. Regul. Toxicol. Pharmacol. 2010, 57, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Oller, A.R.; Oberdörster, G. Incorporation of dosimetry in the derivation of reference concentrations for ambient or workplace air: A conceptual approach. J. Aerosol. Sci. 2016, 99, 40–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oller, A.R.; Oberdörster, G.; Seilkop, S.K. Derivation of PM10 size-selected human equivalent concentrations of inhaled nickel based on cancer and non-cancer effects on the respiratory tract. Inhal. Toxicol. 2014, 26, 559–578. [Google Scholar] [CrossRef]
- Fullerton, A.; Menné, T. In vitro and in vivo evaluation of the effect of barrier gels in nickel contact allergy. Contact Dermat. 1995, 32, 100–106. [Google Scholar] [CrossRef]
- Fullerton, A.; Andersen, J.R.; Hoelgaard, A. Permeation of nickel through human skin in vitro—Effect of vehicles. Br. J. Dermatol. 1988, 118, 509–516. [Google Scholar] [CrossRef]
- Fischer, T. Occupational nickel dermatitis. In Nickel and the Skin: Immunology and Toxicology, 1st ed.; Maibach, H.I., Menné, T., Eds.; CRC Press: Baton Rouge, FL, USA, 1989; pp. 117–132. [Google Scholar]
- Wilkinson, D.S.; Wilkinson, J.D. Nickel allergy and hand eczema. In Nickel and the Skin: Immunology and Toxicology, 1st ed.; Maibach, H.I., Menné, T., Eds.; CRC Press: Baton Rouge, FL, USA, 1989; pp. 133–163. [Google Scholar]
- Fullerton, A.; Andersen, J.R.; Hoelgaard, A.; Menné, T. Permeation of nickel salts through human skin in vitro. Contact Dermat. 1986, 15, 173–177. [Google Scholar] [CrossRef]
- Tanojo, H.; Hostýnek, J.J.; Mountford, H.S.; Maibach, H.I. In vitro permeation of nickel salts through human stratum corneum. Acta Derm. Venereol. 2001, 212, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Hostynek, J.J.; Dreher, F.; Pelosi, A.; Anigbogu, A.; Maibach, H.I. Human Stratum corneum penetration by nickel: In vivo study of depth distribution after occlusive application of the metal as powder. Acta Derm. Venereol. 2001, 212, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Rezuke, W.N.; Knight, J.A.; Sunderman, F.W., Jr. Reference values for nickel concentrations in human tissues and bile. Am. J. Ind. Med. 1987, 11, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Dunnick, J.K.; Elwell, M.R.; Benson, J.M.; Hobbs, C.H.; Hahn, F.F.; Haly, P.J.; Cheng, Y.S.; Eidson, A.F. Lung toxicity after 13-week inhalation exposure to nickel oxide, nickel subsulfide, or nickel sulfate hexahydrate in F344/N rats and B6C3F1 mice. Fundam. Appl. Toxicol. 1989, 12, 584–594. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Agency). Scientific opinion on the risks to public health related to the presence of nickel in food and drinking water, EFSA Panel on Contaminants in the Food Chain (CONTAM). EFSA J. 2015, 13, 4002. [Google Scholar]
- Christensen, O.B.; Möller, H.; Andrasko, L.; Lagesson, V. Nickel concentration of blood, urine and sweat after oral administration. Contact Dermat. 1979, 5, 312–316. [Google Scholar] [CrossRef]
- Sunderman, F.W., Jr.; Aitio, A.; Morgan, L.G.; Norseth, T. Biological monitoring of nickel. Toxicol. Ind. Health 1986, 2, 17–78. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P. Human exposure to nickel. IARC Sci. Publ. 1984, 53, 469–485. [Google Scholar]
- Alinaghi, F.; Bennike, N.H.; Egeberg, A.; Thyssen, J.P.; Johansen, J.D. Prevalence of contact allergy in the general population: A systematic review and meta-analysis. Contact Dermat. 2019, 80, 77–85. [Google Scholar] [CrossRef]
- Warshaw, E.M.; Aschenbeck, K.A.; DeKoven, J.G.; Maibach, H.I.; Taylor, J.S.; Sasseville, D.; Belsito, D.V.; Fowler, J.F., Jr.; Zug, K.A.; Zirwas, M.J.; et al. Epidemiology of pediatric nickel sensitivity: Retrospective review of North American Contact Dermatitis Group (NACDG) data 1994–2014. J. Am. Acad. Dermatol. 2018, 79, 664–671. [Google Scholar] [CrossRef]
- Zenz, C.; Dickerson, O.B.; Horvath, E.P. Occupational Medicine, 3rd ed.; Mosby: St. Louis, MO, USA, 1994. [Google Scholar]
- Menné, T.; Veien, N.; Sjolin, K.-E.; Maibach, H.I. Systemic contact dermatitis. Dermatitis 1994, 5, 1–12. [Google Scholar] [CrossRef]
- Curtis, A.; Morton, J.; Balafa, C.; MacNeil, S.; Gawkrodger, D.J.; Warren, N.D.; Evans, G.S. The effects of nickel and chromium on human keratinocytes: Differences in viability, cell associated metal and IL-1α release. Toxicol. Vitr. 2007, 21, 809–819. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.M.; Bonefeld, C.M.; Poulsen, S.S.; Geisler, C.; Skov, L. IL-23 and T(H)17-mediated inflammation in human allergic contact dermatitis. J. Allergy Clin. Immunol. 2009, 123, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Sebastiani, S.; Albanesi, C.; Nasorri, F.; Girolomoni, G.; Cavani, A. Nickel-specific CD4(+) and CD8(+) T cells display distinct migratory responses to chemokines produced during allergic contact dermatitis. J. Investig. Dermatol. 2002, 118, 1052–1058. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Arakaki, R.; Yamada, A.; Tsunematsu, T.; Kudo, Y.; Ishimaru, N. Molecular Mechanisms of Nickel Allergy. Int. J. Mol. Sci. 2016, 17, 202. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Raghavan, B.; Müller, V.; Vogl, T.; Fejer, G.; Tchaptchet, S.; Keck, S.; Kalis, C.; Nielsen, P.J.; Galanos, C.; et al. Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel. Nat. Immunol. 2010, 11, 814–819. [Google Scholar] [CrossRef]
- Steinman, R.M.; Pack, M.; Inaba, K. Dendritic cells in the T-cell areas of lymphoid organs. Immunol. Rev. 1997, 156, 25–37. [Google Scholar] [CrossRef]
- Thyssen, J.P.; Johansen, J.D.; Menné, T. Contact allergy epidemics and their controls. Contact Dermat. 2007, 56, 185–195. [Google Scholar] [CrossRef]
- Thyssen, J.P.; Menné, T. Metal allergy—A review on exposures, penetration, genetics, prevalence, and clinical implications. Chem. Res. Toxicol. 2010, 23, 309–318. [Google Scholar] [CrossRef]
- Johansen, J.D.; Menné, T.; Christophersen, J.; Kaaber, K.; Veien, N. Changes in the pattern of sensitization to common contact allergens in Denmark between 1985–1986 and 1997–1998, with a special view to the effect of preventive strategies. Br. J. Dermatol. 2000, 142, 490–495. [Google Scholar] [CrossRef]
- European Commission (EC). EC Regulation No 552/2009 of 22 June 2009, Entry 27 in Annex XVII; European Union: Brussels, Belgium, 2009. [Google Scholar]
- European Committee for Standardisation (CEN). Reference Test Method for Release of Nickel from All Post Assemblies Which Are Inserted into Pierced Parts of the Human Body and Articles Intended to Come into Direct and Prolonged Contact with the Skin; EN 1811:2011+A1; European Union: Brussels, Belgium, 2015. [Google Scholar]
- Menné, T.; Rasmussen, K. Regulation of nickel exposure in Denmark. Contact Dermat. 1990, 23, 57–58. [Google Scholar] [CrossRef]
- Antico, A.; Soana, R. Chronic allergic-like dermatopathies in nickel sensitive patients. Results of dietary restrictions and challenge with nickel salts. Allergy Asthma Proc. 1999, 20, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Di Gioacchino, M.; Ricciardi, L.; De Pità, O.; Minelli, M.; Patella, V.; Voltolini, S.; Di Rienzo, V.; Braga, M.; Ballone, E.; Mangifesta, R.; et al. Nickel oral hyposensitization in patients with systemic nickel allergy syndrome. Ann. Med. 2014, 46, 31. [Google Scholar] [CrossRef] [PubMed]
- European Chemicals Agency (ECHA). Classification, Labelling and Packaging. Table of Harmonized Entries in Annex VI to CLP; European Chemicals Agency: Helsinki, Finland, 2018. [Google Scholar]
- International Agency for Research on Cancer (IARC). Nickel and nickel compounds. IARC Monogr. Eval. Carcinog. Risks Hum. 2012, 100C, 169–218. [Google Scholar]
- International Committee on Nickel Carcinogenesis in Man (ICNCM). Report of the International Committee on Nickel Carcinogenesis in Man. Scand. J. Work Environ. Health 1990, 16, 1–82. [Google Scholar] [CrossRef]
- National Toxicology Program (NTP). NTP toxicological and carcinogenesis studies of nickel oxide (CAS No. 1313-99-1) in F344/N rats and B6C3F1 mice (inhalation studies). Natl. Toxicol. Progr. Tech. Rep. Ser. 1996, 451, 1–381. [Google Scholar]
- National Toxicology Program (NTP). NTP toxicological and carcinogenesis studies of nickel subsulfide (CAS No. 12035-72-2) in F344/N rats and B6C3F1 mice (inhalation studies). Natl. Toxicol. Progr. Tech. Rep. Ser. 1996, 453, 1–365. [Google Scholar]
- NTP (National Toxicology Program). NTP toxicological and carcinogenesis studies of nickel sulphate hexahydrate (CAS No. 10101-97-0) in F344/N rats and B6C3F1 mice (inhalation studies). Natl. Toxicol. Progr. Tech. Rep. Ser. 1996, 454, 1–380. [Google Scholar]
- Heim, K.E.; Bates, H.K.; Rush, R.E.; Oller, A.R. Oral carcinogenicity study with nickel sulfate hexahydrate in Fischer 344 rats. Toxicol. Appl. Pharmacol. 2007, 224, 126–137. [Google Scholar] [CrossRef]
- Oller, A.R.; Kirkpatrick, D.T.; Radovsky, A.; Bates, H.K. Inhalation carcinogenicity study with nickel metal powder in Wistar rats. Toxicol. Appl. Pharmacol. 2008, 233, 262–275. [Google Scholar] [CrossRef]
- Oller, A.R. Respiratory carcinogenicity assessment of soluble nickel compounds. Environ. Health Perspect. 2002, 110, 841–844. [Google Scholar] [CrossRef]
- Sivulka, D.J. Assessment of respiratory carcinogenicity associated with exposure to metallic nickel: A review. Regul. Toxicol. Pharmacol. 2005, 43, 117–133. [Google Scholar] [CrossRef] [PubMed]
- Easton, D.F.; Peto, J.; Morgan, L.G.; Metcalfe, L.P.; Usher, V.; Doll, R. Respiratory cancer mortality in Welsh nickel refiners: Which nickel compounds are responsible. In Nickel and Human Health: Current Perspectives; Nieboer, E., Nriagu, J.O., Eds.; John Wiley & Sons, Inc.: New York, NY, USA, 1992; pp. 603–619. [Google Scholar]
- Grimsrud, T.K.; Berge, S.R.; Haldorsen, T.; Andersen, A. Exposure to different forms of nickel and risk of lung cancer. Am. J. Epidemiol. 2002, 156, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Arena, V.C.; Sussman, N.B.; Redmond, C.K.; Costantino, J.P.; Trauth, J.M. Using alternative comparison populations to assess occupation-related mortality risk. Results for the high nickel alloys workers cohort. J. Occup. Environ. Med. 1998, 40, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Arena, V.C.; Costantino, J.P.; Sussman, N.B.; Redmond, C.K. Issues and findings in the evaluation of occupational risk among women high nickel alloys workers. Am. J. Ind. Med. 1999, 36, 114–121. [Google Scholar] [CrossRef]
- Morfeld, P.; Groϐ, J.V.; Erren, T.C.; Noll, B.; Yong, M.; Kennedy, K.J.; Esmen, N.A.; Zimmerman, S.D.; Buchanich, J.M.; Marsh, G.M. Mortality among hardmetal production workers: German historical cohort study. J. Occup. Environ. Med. 2017, 59, e288–e296. [Google Scholar] [CrossRef] [PubMed]
- Marsh, G.M.; Buchanich, J.M.; Zimmerman, S.; Liu, Y.; Balmert, L.C.; Graves, J.; Kennedy, K.J.; Esmen, N.A.; Moshammer, H.; Morfeld, P.; et al. Mortality among hardmetal production workers: Pooled analysis of cohort data from an international investigation. J. Occup. Environ. Med. 2017, 59, e342–e364. [Google Scholar] [CrossRef] [PubMed]
- Marsh, G.M.; Buchanich, J.M.; Zimmerman, S.; Liu, Y.; Balmert, L.C.; Esmen, N.A.; Kennedy, K.J. Mortality among hardmetal production workers: US cohort and nested case-control studies. J. Occup. Environ. Med. 2017, 59, e306–e326. [Google Scholar] [CrossRef]
- Westberg, H.; Bryngelsson, I.-L.; Marsh, G.; Buchanich, J.; Zimmerman, S.; Kennedy, K.; Esmen, N.; Svartengren, M. Mortality among hardmetal production workers: The Swedish cohort. J. Occup. Environ. Med. 2017, 59, e263–e274. [Google Scholar] [CrossRef]
- Westberg, H.; Bryngelsson, I.-L.; Marsh, G.; Kennedy, K.; Buchanich, J.; Zimmerman, S.; Esmen, N.; Svartengren, M. Mortality among hardmetal production workers: Swedish measurement data and exposure assessment. J. Occup. Environ. Med. 2017, 59, e327–e341. [Google Scholar] [CrossRef]
- Dunnick, J.K.; Elwell, M.R.; Radovsky, A.E.; Benson, J.M.; Hahn, F.F.; Nikula, K.J.; Barr, E.B.; Hobbs, C.H. Comparative carcinogenic effects of nickel subsulfide, nickel oxide, or nickel sulfate hexahydrate chronic exposures in the lung. Cancer Res. 1995, 55, 5251–5256. [Google Scholar]
- Hueper, W.C. Experimental studies in metal cancerigenesis. IX. Pulmonary lesions in guinea pigs and rats exposed to prolonged inhalation of powdered metallic nickel. Arch. Pathol. 1958, 65, 600–607. [Google Scholar]
- Pott, F.; Ziem, U.; Reiffer, F.J.; Huth, F.; Ernst, H.; Mohr, U. Carcinogenicity studies of fibres, metal compounds, and some other dusts in rats. Exp. Pathol. 1987, 32, 129–152. [Google Scholar] [CrossRef]
- Wehner, A.P.; Dagle, G.E.; Milliman, E.M. Chronic inhalation exposure of hamsters to nickel-enriched fly ash. Environ. Res. 1981, 26, 195–216. [Google Scholar] [CrossRef]
- National Toxicology Program (NTP). NTP toxicology studies of cobalt metal (CAS No. 7440-48-4) in F344/N rats and B6C3F1/N mice and toxicology and carcinogenesis studies of cobalt metal in F344/NTac rats and B6C3F1/N mice (inhalation studies). Natl. Toxicol. Progr. Am. Tech. Rep. Ser. 2014, 581, 1–308. [Google Scholar]
- National Toxicology Program (NTP). NTP toxicology and carcinogenesis studies of cobalt sulfate heptahydrate (CAS No. 10026-24-1) in F344/N rats and B6C3F1/N mice (inhalation studies). Natl. Toxicol. Progr. Am. Tech. Rep. Ser. 1998, 471, 1–268. [Google Scholar]
- International Agency for Research on Cancer (IARC). Chromium, nickel and welding. IARC Monogr. Eval. Carcinog. Risks Hum. 1990, 49, 1–648. [Google Scholar]
- Uddin, A.N.; Burns, F.J.; Rossman, T.G.; Chen, H.; Kluz, T.; Costa, M. Dietary chromium and nickel enhance UV-carcinogenesis in skin of hairless mice. Toxicol. Appl. Pharmacol. 2007, 221, 329–338. [Google Scholar] [CrossRef]
- Lee, J.E.; Ciccarelli, R.B.; Jennette, K.W. Solubilization of the carcinogen nickel subsulfide and its interaction with deoxyribonucleic acid and protein. Biochemistry 1982, 21, 771–778. [Google Scholar] [CrossRef]
- Biggart, N.W.; Costa, M. Assessment of the uptake and mutagenicity of nickel chloride in Salmonella tester strains. Mutat. Res. 1986, 175, 209–215. [Google Scholar] [CrossRef]
- Chen, H.; Giri, N.C.; Zhang, R.; Yamane, K.; Zhang, Y.; Maroney, M.; Costa, M. Nickel ions inhibit histone demethylase JMJD1A and DNA repair enzyme ABH2 by replacing the ferrous iron in the catalytic centers. J. Biol. Chem. 2010, 285, 7374–7383. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, Y.; Ma, L.; Huang, S.; Wang, R.; Gao, R.; Wu, Y.; Shi, H.; Zhang, J. The alteration of miR-222 and its target genes in nickel-induced tumor. Biol. Trace Elem. Res. 2013, 152, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhou, Y.; Wu, Y.J.; Li, M.J.; Wang, R.J.; Huang, S.Q.; Gao, R.R.; Ma, L.; Shi, H.J.; Zhang, J. Hyper-methylated miR-203 dysregulates ABL1 and contributes to the nickel-induced tumorigenesis. Toxicol. Lett. 2013, 223, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Yang, L.; Yuan, J.; Yang, L.; Zhang, M.; Qi, D.; Duan, X.; Xuan, A.; Zhang, W.; Lu, J.; et al. MicroRNA-152 targets DNA methyltransferase 1 in NiS-transformed cells via a feedback mechanism. Carcinogenesis 2013, 34, 446–453. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Wang, Y.-F.; Huang, W.-R.; Huang, Y.-T. Nickel induces oxidative stress and genotoxicity in human lymphocytes. Toxicol. Appl. Pharmacol. 2003, 189, 153–159. [Google Scholar] [CrossRef]
- Åkerlund, E.; Cappellini, F.; Di Bucchianico, S.; Islam, S.; Skoglund, S.; Derr, R.; Wallinder, I.O.; Hendriks, G.; Karlsson, H.L. Genotoxic and mutagenic properties of Ni and NiO nanoparticles investigated by comet assay, γ-H2AX staining, Hprt mutation assay and ToxTracker reporter cell lines. Environ. Mol. Mutagen. 2018, 59, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Chen, J. Chronic nickel-induced DNA damage and cell death: The protection role of ascorbic acid. Environ. Toxicol. 2008, 23, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Benson, J.M.; March, T.H.; Hahn, F.F.; Seagrave, J.C.; Divine, K.K.; Belinsky, S.A. Short-Term Inhalation Study with Nickel Compounds; Final Report to NiPERA, Inc.; Lovelace Respiratory Research Institute: Albuquerque, NM, USA, 2002. [Google Scholar]
- Kawanishi, S.; Oikawa, S.; Inoue, S.; Nishino, K. Distinct mechanisms of oxidative DNA damage induced by carcinogenic nickel subsulfide and nickel oxides. Environ. Health Perspect. 2002, 110, 789–791. [Google Scholar] [CrossRef]
- Saplakoglu, U.; Iscan, M.; Iscan, M. DNA single-strand breakage in rat lung, liver and kidney after single and combined treatments of nickel and cadmium. Mutagen. Res. 1997, 394, 133–140. [Google Scholar]
- Werfel, U.; Langen, V.; Eickhoff, I.; Schoonbrood, J.; Vahrenholz, C.; Brauksiepe, A.; Popp, W.; Norpoth, K. Elevated DNA single-strand breakage frequencies in lymphocytes of welders exposed to chromium and nickel. Carcinogenesis 1998, 19, 413–418. [Google Scholar] [CrossRef] [Green Version]
- Danadevi, K.; Rozati, R.; Banu, B.S.; Grover, P. Genotoxic evaluation of welders occupationally exposed to chromium and nickel using the comet and micronucleus assays. Mutagenesis 2004, 19, 35–41. [Google Scholar] [CrossRef]
- Faccioni, F.; Franceschetti, P.; Cerpelloni, M.; Fracasso, M.E. In vivo study on metal release from fixed orthodontic appliances and DNA damage in oral mucosa cells. Am. J. Orthod. Dentofac. Orthop. 2003, 124, 687–694. [Google Scholar] [CrossRef]
- Hafez, H.S.; Selim, E.M.N.; Kamel Eid, F.H.; Tawfik, W.A.; Al-Ashkar, E.A.; Mostafa, Y.A. Cytotoxity, genotoxicity, and metal release in patients with fixed orthodontic appliances: A longitudinal in vivo study. Am. J. Orthod. Dentofac. Orthop. 2011, 140, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Miñano, E.; Ortiz, C.; Vicente, A.; Calvo, J.L.; Ortiz, A.J. Metallic ion content and damage to the DNA in oral mucosa cells of children with fixed orthodontic appliances. Biometals 2011, 24, 935–941. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, A.; Mullenders, L.H.F.; Schlepegrell, R.; Kasten, U.; Beyersmann, D. Nickel(II) interferes with the incision step in nucleotide excision repair in mammalian cells. Cancer Res. 1994, 54, 4045–4051. [Google Scholar] [PubMed]
- Hartwig, A.; Schlepegrell, R.; Dally, H.; Hartmann, M. Interaction of carcinogenic metal compounds with deoxyribonucleic acid repair processes. Ann. Clin. Lab. Sci. 1996, 26, 31–38. [Google Scholar] [PubMed]
- Hartwig, A.; Beyersmann, D. Enhancement of UV-induced mutagenesis and sister-chromatid exchanges by nickel ions in V79 cells: Evidence for inhibition of DNA repair. Mutat. Res. 1989, 217, 65–73. [Google Scholar] [CrossRef]
- Mayer, C.; Klein, R.G.; Wesch, H.; Schmezer, P. Nickel subsulfide is genotoxic in vitro but shows no mutagenic potential in respiratory tract tissues of BigBlueTM rats and MutaTM Mouse mice in vivo after inhalation. Mutat. Res. 1998, 420, 85–98. [Google Scholar] [CrossRef]
- Arrouijal, F.Z.; Hildebrand, H.F.; Vophi, H.; Marzin, D. Genotoxic activity of nickel subsulphide α-Ni3S2. Mutagenesis 1990, 5, 583–589. [Google Scholar] [CrossRef]
- Fletcher, G.G.; Rossetto, F.E.; Turnbull, J.D.; Nieboer, E. Toxicity, uptake, and mutagenicity of particulate and soluble nickel compounds. Environ. Health Perspect. 1994, 102 (Suppl. 3), 69–79. [Google Scholar]
- Kargacin, B.; Klein, C.B.; Costa, M. Mutagenic responses of nickel oxides and nickel sulfides in Chinese Hamster V79 cell lines at the xanthine-guanine phosphoribosyl transferase locus. Mutat. Res. 1993, 300, 63–72. [Google Scholar] [CrossRef]
- Danish Environmental Protection Agency (Danish EPA). Nickel and Nickel Compounds: Background Document in Support of Individual Risk Assessment Reports of Nickel Compounds Prepared in Relation to Council Regulation (EEC) 793/93; Danish Environmental Protection Agency: Copenhagen, Denmark, 2008. [Google Scholar]
- Nishimura, M.; Umeda, M. Induction of chromosomal aberrations in cultured mammalian cells by nickel compounds. Mutat. Res. 1979, 68, 337–349. [Google Scholar] [CrossRef]
- Sen, P.; Costa, M. Incidence and localization of sister chromatid exchanges induced by nickel and chromium compounds. Carcinogenesis 1986, 7, 1527–1533. [Google Scholar] [CrossRef] [PubMed]
- Oller, A.R.; Erexson, G. Lack of micronuclei formation in bone marrow of rats after repeated oral exposure to nickel sulfate hexahydrate. Mutat. Res. 2007, 626, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Dhir, H.; Agarwal, K.; Sharma, A.; Talukder, G. Modifying role of Phyllanthus emblica and ascorbic acid against nickel clastogenicity in mice. Cancer Lett. 1991, 59, 9–18. [Google Scholar] [CrossRef]
- Morita, T.; Asano, N.; Awogi, T.; Sasaki, Y.F.; Sato, S.; Shimada, H.; Sutou, S.; Suzuki, T.; Wakata, A.; Sofuni, T.; et al. Evaluation of the rodent micronucleus assay in the screening of IARC carcinogens (Groups 1, 2A and 2B), The summary report of the 6th collaborative study by CSGMT/JEMS.MMS. Mutat. Res. 1997, 389, 3–122. [Google Scholar] [CrossRef]
- Waksvik, H.; Boysen, M.; Brogger, A.; Klepp, O. Chromosome aberrations and sister chromatid exchanges in persons occupationally exposed to mutagens/carcinogens. In Chromosome Damage Repair; NATO Advanced Study Institutes Series (Series A: Life Sciences); Seeberg, E., Kleppe, K., Eds.; Springer: New York, NY, USA, 1981; Volume 40, pp. 563–566. [Google Scholar]
- Waksvik, H.; Boysen, M.; Hogetveit, C. Increased incidence of chromosomal aberrations in peripheral lymphocytes of retired nickel workers. Carcinogenesis 1984, 5, 1525–1527. [Google Scholar] [CrossRef]
- Morán-Martínez, J.; Monreal-de Luna, K.D.; Betancourt-Martínez, N.D.; Carranza-Rosales, P.; Contreras-Martínez, J.G.; López-Meza, M.C.; Rodríguez-Villareal, O. Genotoxicity in oral epithelial cells in children caused by nickel in metal crowns. Genet. Mol. Res. 2013, 12, 3178–3185. [Google Scholar] [CrossRef]
- Natarajan, M.; Padmanabhan, S.; Chiharajan, A.; Narasimhan, M. Evaluation of the genotoxic effects of fixed appliances on oral mucosal cells and the relationship to nickel and chromium concentrations: An in vivo study. Am. J. Orthod. Dentofac. Orthop. 2011, 140, 383–388. [Google Scholar] [CrossRef]
- Heravi, F.; Abbaszadegan, M.R.; Merati, M.; Hasanzadeh, N.; Dadkhah, E.; Ahrari, F. DNA damage in oral mucosa cells of patients with fixed orthodontic appliances. J. Dent. 2013, 10, 494–500. [Google Scholar]
- Westphalen, G.H.; Menezes, L.M.; Prá, D.; Garcia, G.G.; Schmitt, V.M.; Henriques, J.A.; Medina-Silva, R. In vivo determination of genotoxicity induced by metals from orthodontic appliances using micronucleus and comet assays. Genet. Mol. Res. 2008, 7, 1259–1266. [Google Scholar] [CrossRef]
- Zhou, D.; Salnikow, K.; Costa, M. Cap43, a novel gene specifically induced by Ni2+ compounds. Cancer Res. 1998, 58, 2182–2189. [Google Scholar] [PubMed]
- Salnikow, K.; An, W.G.; Melillo, G.; Blagosklonny, M.V.; Costa, M. Nickel-induced transformation shifts the balance between HIF-1 and p53 transcription factors. Carcinogenesis 1999, 20, 1819–1823. [Google Scholar] [CrossRef] [PubMed]
- Salnikow, K.; Blagosklonny, M.V.; Ryan, H.; Johnson, R.; Costa, M. Carcinogenic nickel induces genes involved with hypoxic stress. Cancer Res. 2000, 60, 38–41. [Google Scholar] [PubMed]
- Salnikow, K.; Davidson, T.; Costa, M. The role of hypoxia-inducible signaling pathway in nickel carcinogenesis. Health Perspect. 2002, 110 (Suppl. 5), 831–834. [Google Scholar] [CrossRef]
- Kang, Y.-T.; Hsu, W.-C.; Wu, C.-H.; Hsin, I.-L.; Wu, P.-R.; Yeh, K.-T.; Ko, J.-L. Metformin alleviates nickel-induced autophagy and apoptosis via inhibition of hexokinase-2, activating lipocalin-2, in human bronchial epithelial cells. Oncotarget 2017, 8, 105536–105552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, V.C.; Morse, J.L.; Zhitkovich, A. p53 activation by Ni(II) is a HIF-1α independent response causing caspases 9/3-mediated apoptosis in human lung cells. Toxicol. Appl. Pharmacol. 2013, 269, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Green, S.E.; Luczak, W.W.; Morse, J.L.; DeLoughery, Z.; Zhitkovich, A. Uptake, p53 pathway activation, and cytotoxic responses for Co(II) and Ni(II) in human lung cells: Implications for carcinogenicity. Toxicol. Sci. 2013, 136, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Di Bucchianico, S.; Gliga, A.R.; Åkerlund, E.; Skoglund, S.; Wallinder, I.O.; Fadeel, B.; Karlsson, H.L. Calcium-dependent cyto-and genotoxicity of nickel metal and nickel oxide nanoparticles in human lung cells. Part. Fibre Toxicol. 2018, 15, 32. [Google Scholar] [CrossRef] [PubMed]
- Bonin, S.; Larese, F.F.; Trevisan, G.; Avian, A.; Rui, F.; Stanta, G.; Bovenzi, M. Gene expression changes in peripheral blood mononuclear cells in occupational exposure to nickel. Exp. Dermatol. 2011, 20, 147–148. [Google Scholar] [CrossRef]
- Chen, H.; Ke, Q.; Kluz, T.; Yan, Y.; Costa, M. Nickel ions increase histone H3 lysine 9 dimethylation and induce transgene silencing. Mol. Cell. Biol. 2006, 26, 3728–3737. [Google Scholar] [CrossRef]
- Chen, H.; Kluz, T.; Zhang, R.; Costa, M. Hypoxia and nickel inhibit histone demethylase JMJD1A and repress Spry2 expression in human bronchial epithelial BEAS-2B cells. Carcinogenesis 2010, 31, 2136–2144. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Bai, Y.; Pu, H.; Gou, F.; Dai, M.; Wang, H.; He, J.; Zheng, T.; Cheng, N. Histone methylation in nickel-smelting industrial workers. PLoS ONE 2015, 10, e0140339. [Google Scholar] [CrossRef] [PubMed]
- Arita, A.; Niu, J.; Qu, Q.; Zhao, N.; Ruan, Y.; Nadas, A.; Chervona, Y.; Wu, F.; Sun, H.; Hayes, R.B.; et al. Global levels of histone modifications in peripheral blood mononuclear cells of subjects with exposure to nickel. Environ. Health Perspect. 2012, 120, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Cantone, L.; Nordio, F.; Hou, L.; Apostoli, P.; Bonzini, M.; Tarantini, L.; Angelici, L.; Bollati, V.; Zanobetti, A.; Schwartz, J.; et al. Inhalable metal-rich air particles and histone H3K4 dimethylation and H3K9 acetylation in a cross-sectional study of steel workers. Environ. Health Perspect. 2011, 119, 964–969. [Google Scholar] [CrossRef] [PubMed]
- Ke, Q.; Li, Q.; Ellen, T.P.; Sun, H.; Costa, M. Nickel compounds induce phosphorylation of histone H3 at serine 10 by activating JNK-MAPK pathway. Carcinogenesis 2008, 29, 1276–1281. [Google Scholar] [CrossRef] [Green Version]
- Ke, Q.; Ellen, T.P.; Costa, M. Nickel compounds induce histone ubiquitination by inhibiting histone deubiquitinating enzyme activity. Toxicol. Appl. Pharmacol. 2008, 228, 190–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, J.; Zhang, Y.; Chen, J.; Chen, H.; Lin, C.; Wang, Q.; Ou, Y. Nickel-induced histone hypoacetylation: The role of reactive oxygen species. Toxicol. Sci. 2003, 74, 279–286. [Google Scholar] [CrossRef]
- Kang, J.; Zhang, D.; Chen, J.; Lin, C.; Liu, Q. Involvement of histone hypoacetylation in Ni2+-induced bcl-2 down-regulation and human hepatoma cell apoptosis. J. Biol. Inorg. Chem. 2004, 9, 713–723. [Google Scholar] [CrossRef]
- Huang, X.; Frenkel, K.; Klein, C.B.; Costa, M. Nickel induces increased oxidants in intact cultured mammalian cells as detected by dichlorofluorescein fluorescence. Toxicol. Appl. Pharmacol. 1993, 120, 29–36. [Google Scholar]
- Huang, X.; Klein, C.B.; Costa, M. Crystalline Ni3S2 specifically enhances the formation of oxidants in the nuclei of CHO cells as detected by dichlorofluorescein. Carcinogenesis 1994, 15, 545–548. [Google Scholar] [CrossRef]
- Jadhav, S.H.; Sarkar, S.N.; Aggarwal, M.; Tripathi, H.C. Induction of oxidative stress in erythrocytes of male rats subchronically exposed to a mixture of eight metals found as groundwater contaminants in different parts of India. Arch. Environ. Contam. Am. Toxicol. 2007, 52, 145–151. [Google Scholar] [CrossRef] [PubMed]
- El-Habit, O.H.; Moneim, A.E. Testing the genotoxicity, cytotoxicity, and oxidative stress of cadmium and nickel and their additive effect in male mice. Biol. Trace Elem. Res. 2014, 159, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Merzenich, H.; Hartwig, A.; Ahrens, W.; Beyersmann, D.; Schlepegrell, R.; Scholze, M.; Timm, J.; Jöckel, K.-H. Biomonitoring on carcinogenic metals and oxidative DNA damage in a cross-sectional study. Cancer Epidemiol. Prev. Biomark. 2001, 10, 515–522. [Google Scholar]
- Chaschschin, V.P.; Artunina, G.P.; Norseth, T. Congenital defects, abortion and other health effects in nickel refinery workers. Sci. Total Environ. 1994, 148, 287–291. [Google Scholar] [CrossRef]
- Vaktskjold, A.; Talykova, L.; Chashchin, V.; Odland, J.; Nieboer, E. Small-for-gestational age newborns of female refinery workers exposed to nickel. Int. J. Occup. Med. Environ. Health 2007, 20, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Vaktskjold, A.; Talykova, L.; Chashchin, V.; Odland, J.; Nieboer, E. Spontaneous abortions among nickel-exposed female refinery workers. Int. J. Environ. Health Res. 2008, 18, 99–115. [Google Scholar] [CrossRef] [PubMed]
- Vaktskjold, A.; Talykova, L.; Chashchin, V.; Nieboer, E.; Thomassen, Y.; Odland, J. Genital malformations in newborns of female nickel-refinery workers. Scand. J. Work Environ. Health 2006, 32, 41–50. [Google Scholar] [CrossRef] [Green Version]
- Vaktskjold, A.; Talykova, L.; Chashchin, V.; Odland, J.; Nieboer, E. Maternal nickel exposure and congenital musculoskeletal defects. Am. J. Ind. Med. 2008, 51, 825–833. [Google Scholar] [CrossRef]
- Ebisu, K.; Bell, M.L. Airborne PM2.5 chemical components and low birth weight in the Northeastern and Mid-Atlantic regions of the United States. Environ. Health Perspect. 2012, 120, 1746–1752. [Google Scholar] [CrossRef]
- Ambrose, A.M.; Larson, P.S.; Borzelleca, J.F.; Hennigar, G.R., Jr. Long-term toxicologic assessment of nickel in rats and dogs. J. Food Sci. Technol. 1976, 13, 181–187. [Google Scholar]
- Smith, M.K.; George, E.L.; Stober, J.A.; Feng, H.A.; Kimmel, G.A. Perinatal toxicity associated with nickel chloride. Environ. Res. 1993, 61, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Research Triangle Institute (RTI). Fertility and Reproductive Performance of the P(0) Generation: Two-Generation Reproduction and Fertility Study of Nickel Chloride Administered to CD Rats in the Drinking Water; Final Report, RTI Master Protocol No. 182, Study Code No. RT85-NICL.REPRO, Project No. 472U-3228-07, EPA No. 68-01-7075; Research Triangle Institute: Research Triangle Park, NC, USA, 1988. [Google Scholar]
- Research Triangle Institute (RTI). Fertility and Reproductive Performance of the F(1) Generation: Two-Generation Reproduction and Fertility Study of Nickel Chloride Administered to CD Rats in the Drinking Water; Final Report; Research Triangle Institute: Research Triangle Park, NC, USA, 1988. [Google Scholar]
- Springborn Laboratories, Inc. (SLI). An Oral (Gavage) 1-Generation Reproduction Study of Nickel Sulfate Hexahydrate in Rats; Study No. 3472.3; Springborn Laboratories, Inc.: Spencerville, OH, USA, 2000. [Google Scholar]
- Springborn Laboratories, Inc. An Oral (Gavage) Two-Generation Reproduction Toxicity Study in Sprague-Dawley Rats with Nickel Sulfate Hexahydrate; Study No. 3472.4; Springborn Laboratories, Inc.: Spencerville, OH, USA, 2000. [Google Scholar]
- Ishimatsu, S.; Kawamoto, T.; Matsuno, K.; Kodama, Y. Distribution of various nickel compounds in rat organs after oral administration. Biol. Trace Elem. Res. 1995, 49, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Classification, Labelling and Packaging (CLP). Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on Classification, Labelling and Packaging of Substances and Mixtures; Amending and Repealing Directives 67/548/EEC and 1999/45/EC, and Amending Regulation (EC) No 1907/2006; European Union: Brussels, Belgium, 2008. [Google Scholar]
- Haber, L.T.; Bates, H.K.; Allen, B.C.; Vincent, M.J.; Oller, A.R. Derivation of an oral toxicity reference value for nickel. Regul. Toxicol. Pharmacol. 2017, 87 (Suppl. 1), S1–S18. [Google Scholar] [CrossRef]
- Saini, S.; Nair, N.; Saini, M.R. Embryotoxic and teratogenic effects of nickel in Swiss albino mice during organogenetic period. BioMed. Res. Int. 2013, 2013, 701439. [Google Scholar] [CrossRef] [PubMed]
- Akesson, B.; Skerfving, S. Exposure in welding of high nickel alloy. Int. Arch. Occup. Environ. Health 1985, 56, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Phillips, J.I.; Green, F.Y.; Davies, J.C.; Murray, J. Pulmonary and systemic toxicity following exposure to nickel nanoparticles. Am. J. Ind. Med. 2010, 53, 763–767. [Google Scholar] [CrossRef]
- California Environmental Protection Agency (Cal EPA). Nickel Reference Exposure Levels; Office of Environmental Health Hazard Assessment: Sacramento, CA, USA, 2012.
- Muir, D.C.F.; Julian, J.; Roberts, R.; Roos, J.; Chan, J.; Machle, W.; Morgan, W.K.C. Prevalence of small opacities in chest radiographs of nickel sinter plant workers. Br. J. Ind. Med. 1993, 50, 428–431. [Google Scholar] [CrossRef]
- Berge, S.R.; Skyberg, K. Radiographic evidence of pulmonary fibrosis and possible etiologic factors at a nickel refinery in Norway. J. Environ. Monit. 2003, 5, 681–688. [Google Scholar] [CrossRef]
- Miller, A.; Warshaw, R.; Nezamis, J. Diffusing capacity and forced vital capacity in 5003 asbestos-exposed workers: relationships to interstitial fibrosis (ILO profusion score) and pleural thickening. Am. J. Ind. Med. 2013, 56, 1383–1393. [Google Scholar] [CrossRef]
- Sorahan, T. Mortality of workers at a plant manufacturing nickel alloys, 1958–2000. Occup. Med. 2004, 54, 28–34. [Google Scholar] [CrossRef]
- Sorahan, T.; Williams, S.P. Mortality of workers at a nickel carbonyl refinery, 1958–2000. Occup. Environ. Med. 2005, 62, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Moulin, J.J.; Clavel, T.; Roy, D.; Danaché, B.; Marquis, N.; Févotte, J.; Fontana, J.M. Risk of lung cancer in workers producing stainless steel and metallic alloys. Int. Arch. Occup. Environ. Health 2000, 73, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Cragle, D.L.; Hollis, D.R.; Newport, T.H.; Shy, C.M. A Retrospective Cohort Mortality StudyAamon Workers Occupationally Exposed to Metallic Nickel Powder at the Oak Ridge Gaseous Diffusion Plant. In Nickel in the Human Environment, Proceedings of a JoInt. Symposium, IARC Scientific Publications No. 53, Lyon, France, 6–8 October March 1983; Sunderman, F.W., Jr., Ed.; International Agency for Research on Cancer: Lyon, France, 1984; pp. 57–63. [Google Scholar]
- Block, G.T.; Yeung, M. Asthma induced by nickel. JAMA 1982, 247, 1600–1602. [Google Scholar] [CrossRef] [PubMed]
- Estlander, T.; Kanerva, L.; Tupasela, O.; Keskinen, H.; Jolanki, R. Immediate and delayed allergy to nickel with contact urticaria, rhinitis, asthma and contact dermatitis. Clin. Exp. Allergy 1993, 23, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Bright, P.; Burge, P.S.; O’Hickey, S.P.; Gannon, P.F.G.; Robertson, A.S.; Boran, A. Occupational asthma due to chrome and nickel electroplating. Thorax 1997, 52, 28–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandez-Nieto, M.; Quirce, S.; Carnes, J.; Sastre, J. Occupational asthma due to chromium and nickel salts. Int. Arch. Occup. Environ. Health 2006, 79, 483–486. [Google Scholar] [CrossRef] [PubMed]
- United States Environmental Protection Agency (US EPA). Guidelines for Deriving Numerical National Water Quality Criteria for the Protection of Aquatic Organisms and Their Uses; Office of Research and Development, Environmental Research Laboratories: Duluth, MN, USA; Narragansett, RI, USA; Corvallis, OR, USA, 1985; pp. 1–59.
- Chau, Y.K.; Kulikovsky-Cordeiro, O.T.R. Occurrence of nickel in the Canadian environment. Environ. Rev. 1995, 3, 95–120. [Google Scholar] [CrossRef]
- Ankley, G.T.; Di Toro, D.M.; Hansen, D.J.; Berry, W.J. Technical basis and proposal for deriving sediment quality criteria for metals. Environ. Toxicol. Chem. 1996, 15, 2056–2066. [Google Scholar] [CrossRef]
- Merrington, G.; Peters, A.; Schlekat, C. Accounting for metal bioavailability in assessing water quality: A step change? Environ. Toxicol. Chem. 2016, 35, 257–265. [Google Scholar] [CrossRef]
- Peters, A.; Merrington, G.; Schlekat, C.; de Schamphelaere, K.; Stauber, J.; Batley, G.; Harford, A.; van Dam, R.; Pease, C.; Mooney, T.; et al. Validation of the nickel biotic ligand model for locally relevant species in Australian freshwaters. Environ. Toxicol. Chem. 2018, 37, 2566–2574. [Google Scholar] [CrossRef]
- Chapman, P.M. Environmental risks of inorganic metals and metalloids: A continuing, evolving scientific odyssey. Hum. Ecol. Risk Assess. 2008, 14, 5–40. [Google Scholar] [CrossRef]
- Deleebeeck, N.; De Schamphelaere, K.; Janssen, C. A bioavailability model predicting the toxicity of nickel to rainbow trout (Oncorhynchus mykiss) and fathead minnow (Pimephales promelas) in synthetic and natural waters. Ecotoxicol. Toxicol. Environ. Saf. 2007, 67, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Deleebeeck, N.; De Schamphelaere, K.; Janssen, C. A novel method for predicting chronic nickel bioavailability and toxicity to Daphnia magna in artificial and natural waters. Environ. Toxicol. Chem. 2008, 27, 2097–2107. [Google Scholar] [CrossRef]
- Deleebeeck, N.; De Schamphelaere, K.; Janssen, C. Effects of Mg2+ and H+ on the toxicity of Ni2+ to the uni-cellular green alga Pseudokirchneriella subcapitata: Model development and validation with surface waters. Sci. Total Environ. 2009, 407, 1901–1914. [Google Scholar] [CrossRef] [PubMed]
- Lock, K.; Van Eeckhout, H.; De Schamphelaere, K.; Criel, P.; Janssen, C. Development of a Biotic Ligand Model (BLM) predicting nickel toxicity to barley (Hordeum vulgare). Chemosphere 2007, 66, 1346–1352. [Google Scholar] [CrossRef] [PubMed]
- Schlekat, C.; DeSchamphelaere, K.; Van Genderen, E.; Antunes, P.; Stubblefield, W.; Rogevich, E. Cross-species extrapolation of chronic nickel Biotic Ligand Models. Sci. Total Environ. 2010, 408, 6148–6157. [Google Scholar] [CrossRef] [PubMed]
- Nys, C.; Janssen, C.; van Sprang, P.; de Schamphelaere, K. The effect of pH on chronic aquatic Ni toxicity is dependent on the pH itself: Extending the chronic Ni bioavailability models. Environ. Toxicol. Chem. 2016, 35, 1097–1106. [Google Scholar] [CrossRef] [PubMed]
- Deleebeeck, N.M.E.; Muyssen, B.; De Lander, F.; De Schamphelaere, K.A.C.; Janssen, C.R. Comparison of nickel toxicity to cladocerans in soft versus hard surface waters. Aquat. Toxicol. 2007, 84, 223–235. [Google Scholar] [CrossRef]
- Santore, R.C.; Di Toro, D.M.; Paquin, P.R.; Allen, H.E.; Meyer, J.S. Biotic ligand model of the acute toxicity of metals. 2. Application to acute copper toxicity in freshwater fish and Daphnia. Environ. Toxicol. Chem. 2001, 20, 2397–2402. [Google Scholar] [CrossRef]
- Paquin, P.R.; Gorsuch, J.W.; Apte, S.; Batley, G.E.; Bowles, K.C.; Campbell, P.G.C.; Delos, C.G.; Di Toro, D.M.; Dwyer, R.L.; Galvez, F.; et al. The biotic ligand model: A historical overview. Comp. BioChem. Physiol. C Toxicol. Pharmacol. 2002, 133, 3–35. [Google Scholar] [CrossRef]
- Nys, C.; Janssen, C.; De Schamphelaere, K. Evaluation of Acute Ni Bioavailability Models for Model and Non-Model Species; Faculty of Bioscience Engineering, Laboratory of Environmental Toxicology and Aquatic Ecology, Ghent University: Ghent, Belgium, 2015. [Google Scholar]
- Blewett, T.A.; Dow, E.; Wood, C.M.; McGeer, J.C.; Smith, D.S. The role of dissolved organic carbon concentration and composition in ameliorating nickel toxicity to early life-stages of the blue mussel Mytilus edulis. Ecotoxicol. Toxicol. Environ. Saf. 2018, 160, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Sherman, S. Influence of Ligand Complexation on Nickel Toxicity, Speciation and Bioavailability in Marine Waters. Master’s Thesis, Wilfrid Laurier University, Waterloo, ON, Canada, 2019. [Google Scholar]
- Blewett, T.A.; Smith, D.S.; Wood, C.M.; Glover, C.N. Mechanisms of nickel toxicity in the highly sensitive embryos of the sea urchin Evechinus chloroticus, and the modifying effects of dissolved organic carbon. Environ. Sci. Technol. 2016, 2, 1595–1603. [Google Scholar] [CrossRef] [PubMed]
- US EPA (United States Environmentmental Protection Agency). The Aquatic Life Ambient Freshwater Criteria–Copper; 2007 Revision (CAS Registry Number 7440-50-8); US Environmental Protection Agency, Office of Water, Office of Science and Technology: Washington, DC, USA, 2007.
- Warne, M.; Batley, G.E.; van Dam, R.A.; Chapman, J.C.; Fox, D.R.; Hickey, C.W.; Stauber, J.L. Revised Method for Deriving Australian and New Zealand Water Quality Guideline Values for Toxicants–Update of 2015 Version, Prepared for the Revision of the Australian and New Zealand Guidelines for Fresh and Marine Water Quality; Australian and New Zealand Governments and Australian State and Territory Governments: Canberra, Australia, 2018.
- Canadian Council of Ministers of the Environment (CCME). A Protocol for the Derivation of Water Quality Guidelines for the Protection of Aquatic Life 2007. In Canadian Environmental Quality Guidelines, 1999; Canadian Council of Ministers of the Environment: Winnipeg, MB, Canada, 2007. [Google Scholar]
- European Commission (EC). Nickel and Its Compounds (Final Revision Oct 12 2010) EQS Sheet; Prepared by Denmark, Danish Environmental Protection Agency on behalf of the European Union; European Union: Brussels, Belgium, 2010. [Google Scholar]
- European Commission (EC). Common Implementation Strategy for the Water Framework Directive (2000/60/EC) Guidance Document No. 27 Technical Guidance for Deriving Environmental Quality Standards; Technical Report for Approval by Water Directors-WD 2018-1-1; Office for Official Publications in the European Communities: Luxembourg, 2018. [Google Scholar]
- Paller, M.H.; Knox, A.S. Bioavailability of metals in contaminated sediments. EDP Sci. 2013, 1, 2001. [Google Scholar] [CrossRef]
- Besser, J.M.; Brumbaugh, W.G.; Ingersoll, C.G.; Ivey, C.D.; Kunz, J.L.; Kemble, N.E.; Schlekat, C.E.; Garman, E.R. Chronic Toxicity of Nickel-Spiked Freshwater Sediment: Variation in Toxicity Among Eight Invertebrate Taxa and Eight Sediments. Environ. Toxicol. Chem. 2013, 32, 2495–2506. [Google Scholar] [CrossRef] [PubMed]
- Vangheluwe, M.; Verdonck, F.; Besser, J.M.; Brumbaugh, W.G.; Ingersoll, C.G.; Schlekat, C.E.; Garman, E.R. Improving sediment quality guidelines for nickel: Development and application of predictive bioavailability models to assess chronic toxicity of nickel in freshwater sediments. Environ. Toxicol. Chem. 2013, 32, 2507–2519. [Google Scholar] [CrossRef] [PubMed]
- Di Toro, D.M.; Mahony, J.D.; Hansen, D.J.; Scott, K.J.; Carlson, A.R.; Ankley, G.T. Acid volatile sulfide predicts the acute toxicity of cadmium and nickel in sediments. Environ. Sci. Technol. 1992, 26, 96–101. [Google Scholar] [CrossRef]
- Hansen, D.J.; Berry, W.J.; Mahony, J.D.; Boothman, W.S.; Di Toro, D.M.; Robson, D.L.; Ankley, G.T.; Ma, D.; Yan, Q.; Pesch, C.E. Predicting the toxicity of metal-contaminated field sediments using interstitial concentrations of metals and acid-volatile sulfide normalizations. Environ. Toxicol. Chem. 1996, 15, 2080–2094. [Google Scholar] [CrossRef]
- Di Toro, D.M.; McGrath, J.A.; Hansen, D.J.; Berry, W.J.; Paquin, P.R.; Mathew, R.; Wu, K.B.; Santore, R.C. Predicting sediment metal toxicity using a sediment biotic ligand model: Methodology and initial application. Environ. Toxicol. Chem. 2005, 24, 2410–2427. [Google Scholar] [CrossRef]
- Vangheluwe, M.; Nguyen, L. Advanced Research on Nickel Toxicity in Sediments: Results Additional Species and Modelling; Final Report to NiPERA; Nickel Producers Environmental Research Association, Inc.: Durham, NC, USA, 2015. [Google Scholar]
- Costello, D.M.; Burton, G.A.; Hammerschmidt, C.R.; Rogevich, E.C.; Schlekat, C.E. Nickel phase partitioning and toxicity in field-deployed sediments. Environ. Sci. Technol. 2011, 45, 5798–5805. [Google Scholar] [CrossRef]
- Lock, K.; Janssen, C. Ecotoxicity of nickel to Eisenia fetida, Enchytraeus albidus, and Folsomia candida. Chemosphere 2002, 46, 197–200. [Google Scholar] [CrossRef]
- Smolders, E.; Oorts, K.; Van Sprang, P.; Schoeters, I.; Janssen, C.; McGrath, S.; McLaughlin, M. Toxicity of trace metals in soil as affected by soil type and aging after contamination: Using calibrated bioavailability models to set ecological soil standards. Environ. Toxicol. Chem. 2009, 28, 1633–1642. [Google Scholar] [CrossRef] [PubMed]
- Rooney, C.; Zhao, F.J.; McGrath, S. Phytotoxicity of nickel in a range of European soils: Influence of soil properties, nickel solubility, and speciation. Environ. Pollut. 2007, 145, 596–605. [Google Scholar] [CrossRef] [PubMed]
- European Commission (EC). European Union Risk Assessment Report (EU RAR) of Nickel and Nickel Compounds; Prepared by the Danish Environmental Protection Agency (DEPA); European Union: Brussels, Belgium, 2008. [Google Scholar]
- Binet, M.T.; Adams, M.S.; Gissi, F.; Golding, L.A.; Schlekat, C.E.; Garman, E.R.; Merrington, G.; Stauber, J.L. Toxicity of nickel to tropical freshwater and sediment biota: A critical literature review and gap analysis. Environ. Toxicol. Chem. 2017, 37, 293–317. [Google Scholar] [CrossRef] [PubMed]
- Gissi, F.; Stauber, J.L.; Binet, M.T.; Golding, L.A.; Adams, M.S.; Schlekat, C.E.; Garman, E.R.; Jolley, D.F. A review of nickel toxicity to marine and estuarine tropical biota with particular reference to the South East Asian and Melanesian region. Environ. Pollut. 2016, 218, 1308–1323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klimisch, H.-J.; Andreae, M.; Tillmann, U. A systematic approach for evaluating the quality of experimental toxicological and ecotoxicological data. Regul. Toxicol. Pharmacol. 1997, 25, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Moermond, C.; Kase, R.; Korkaric, M.; Ågerstrand, M. CRED-Criteria for reporting and evaluating ecotoxicity data. Environ. Toxicol. Chem. 2015, 35, 1297–1309. [Google Scholar] [CrossRef] [PubMed]
- Batley, G.E.; van Dam, R.; Warne, M.S.J.; Chapman, J.C.; Fox, D.R.; Hickey, C.W.; Stauber, J.L. Technical Rationale for Changes to the Method for Deriving Australian and New Zealand Water Quality Guideline Values for Toxicants–Update of 2014 Version; Prepared for the revision of the Australian and New Zealand Guidelines for Fresh and Marine Water Quality; Australian and New Zealand Governments and Australian State and Territory Governments: Canberra, Australia, 2018.
- Schlekat, C.E.; Garman, E.R.; Vangheluwe, M.L.U.; Burton, G.A., Jr. Development of a bioavailability-based risk assessment approach for Ni in sediments. Integr. Environ. Assess. Manag. 2016, 12, 735–746. [Google Scholar] [CrossRef]
- Aldenberg, T.; Jaworska, J.S. Uncertainty of the hazardous concentration and fraction affected for normal species sensitivity distributions. Ecotoxicol. Toxicol. Environ. Saf. 2000, 46, 1–18. [Google Scholar] [CrossRef]
- Niyogi, S.; Brix, K.V.; Grosell, M. Effects of chronic waterborne nickel exposure on growth, ion homeostasis, acid-base balance, and nickel uptake in the freshwater pulmonate snail, Lymnaea stagnalis. Aquat. Toxicol. 2014, 150, 36–44. [Google Scholar] [CrossRef]
- Peters, A.; Merrington, G.; Leverett, D.; Wilson, I.; Schlekat, C.; Garman, E. Comparison of the chronic toxicity of nickel to temperate and tropical freshwater species. Environ. Toxicol. Chem. 2019. [Google Scholar] [CrossRef]
- Hommen, U.; Knopf, B.; Rüdel, H.; Schäfers, C.; De Schamphelaere, K.; Schlekat, C.; Garman, E.R. A microcosm study to support aquatic risk assessment of nickel: Community-level effects and comparison with bioavailability-normalized species sensitivity distributions. Environ. Toxicol. Chem. 2016, 35, 1172–1182. [Google Scholar] [CrossRef] [PubMed]
- Peters, A.; Simpson, P.; Merrington, G.; Schlekat, C.; Rogevich-Garman, E. Assessment of the effects of nickel on benthic macroinvertebrates in the field. Environ. Sci. Pollut. Res. Int. 2014, 21, 193–204. [Google Scholar] [CrossRef] [PubMed]
- European Union (EU). Directive 2013/39/EU of the European Parliament and of the Council. Off. J. Eur. Union 2013, 226, 338–436. [Google Scholar]
- Deforest, D.K.; Schlekat, C.E. Species sensitivity distribution evaluation for chronic nickel toxicity to marine organisms. Integr. Environ. Assess. Manag. 2013, 9, 580–589. [Google Scholar] [CrossRef] [PubMed]
- Gissi, F.; Stauber, J.L.; Binet, M.T.; Trenfield, M.A.; Van Dam, J.W.; Jolley, D.F. Assessing the chronic toxicity of nickel to a tropical marine gastropod and two crustaceans. Ecotoxicol. Toxicol. Environ. Saf. 2018, 159, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Gissi, F.; Stauber, J.; Reichelt-Brushett, A.; Harrison, P.L.; Jolley, D.F. Inhibition in fertilisation of coral gametes following exposure to nickel and copper. Ecotoxicol. Toxicol. Environ. Saf. 2017, 145, 32–41. [Google Scholar] [CrossRef] [Green Version]
- Gissi, F.; Reichelt-Brushett, A.J.; Chariton, A.A.; Stauber, J.L.; Greenfield, P.; Humphrey, C.; Salmon, M.; Stephenson, S.A.; Cresswell, T.; Jolley, D.F. The effect of dissolved nickel and copper on the adult coral Acropora muricate and its microbiome. Environ. Pollut. 2019, 250, 792–806. [Google Scholar] [CrossRef]
- European Chemicals Agency (ECHA). Guidance on Information Requirements and Chemical Safety Assessment, Chapter R.10: Characterisation of Dose [Concentration]-Response for Environment; European Chemicals Agency: Helsinki, Finland, 2008. [Google Scholar]
- Vandegehuchte, M.B.; Roman, Y.E.; Nguyen, L.T.; Janssen, C.R.; De Schamphelaere, K.A. Toxicological availability of nickel to the benthic oligochaete Lumbriculus variegatus. Environ. Int. 2007, 33, 736–742. [Google Scholar] [CrossRef] [Green Version]
- Brumbaugh, W.G.; Besser, J.M.; Ingersoll, C.G.; May, T.W.; Ivey, C.D.; Schlekat, C.E.; Garman, E.R. Preparation and characterization of nickel-spiked freshwater sediments for toxicity tests: Toward more environmentally realistic testing conditions. Environ. Toxicol. Chem. 2013, 32, 2482–2494. [Google Scholar]
- Chandler, G.T.; Schlekat, C.E.; Garman, E.R.; He, L.; Washburn, K.M.; Stewart, E.R.; Ferry, J.L. Sediment nickel bioavailability and toxicity to estuarine crustaceans of contrasting bioturbative behaviors—An evaluation of the SEM-AVS paradigm. Environ. Sci. Technol. 2014, 48, 12893–12901. [Google Scholar] [CrossRef]
- Costello, D.M.; Burton, G.A.; Hammerschmidt, C.R.; Taulbee, W.K. Evaluating the performance of diffusive gradients in thin films (DGTs) for predicting Ni sediment toxicity. Environ. Sci. Technol. 2012, 46, 10239–10246. [Google Scholar] [PubMed]
- Nguyen, L.T.; Burton, G.A., Jr.; Schlekat, C.E.; Janssen, C.R. Field measurement of nickel sediment toxicity: Role of acid volatile sulfide. Environ. Toxicol. Chem. 2011, 30, 162–172. [Google Scholar] [CrossRef] [PubMed]
- Costello, D.M.; Burton, G.A. Response of stream ecosystem function and structure to sediment metal: Context dependency and variation among endpoints. Elem. Sci. Anth. 2014, 2, 30. [Google Scholar] [CrossRef]
- Costello, D.M.; Hammerschmidt, C.R.; Burton, G.A. Nickel partitioning and toxicity in sediment during aging: Variation in toxicity related to stability of metal partitioning. Environ. Sci. Technol. 2016, 50, 11337–11345. [Google Scholar] [CrossRef] [PubMed]
- Mendonca, R.M.; Daley, J.M.; Hudson, M.L.; Schlekat, C.E.; Burton, G.A.; Costello, D.M. Metal oxides in surface sediment control nickel bioavailability to benthic macroinvertebrates. Environ. Sci. Technol. 2017, 51, 13407–13416. [Google Scholar] [CrossRef] [PubMed]
- Hale, B.; Gopalapillai, Y.; Pellegrino, A.; Jennett, T.; Kikkert, J.; Lau, W.; Schlekat, C.; McLaughlin, M.J. Validation of site-specific soil Ni toxicity thresholds with independent ecotoxicity and biogeochemistry data for elevated soil ni. Environ. Pollut. 2017, 231 Pt 1, 165–172. [Google Scholar] [CrossRef]
- Brix, K.; Schlekat, C.; Garman, E. The mechanisms of nickel toxicity in aquatic environments: An adverse outcome pathway (AOP) analysis. Environ. Toxicol. Chem. 2017, 36, 1128–1137. [Google Scholar] [CrossRef]
- He, E.; Qiu, H.; Dimitrova, K.; Van Gestel, C.A. A generic biotic ligand model quantifying the development in time of Ni toxicity to Enchytraeus crypticus. Chemosphere 2015, 124, 170–176. [Google Scholar] [CrossRef]
- Luo, S.Q.; Plowman, M.C.; Hopfer, S.M.; Sunderman, F.W. Mg(2+)-deprivation enhances and Mg(2+)-supplementation diminishes the embryotoxic and teratogenic effects of Ni2+, Co2+, Zn2+ and Cd2+ for frog embryos in the FETAX assay. Ann. Clin. Lab. Sci. 1993, 23, 121–129. [Google Scholar]
- Gopalapillai, Y.; Hale, B.; Vigneault, B. Effect of major cations (Ca2+, Mg2+, Na+, K+) and anions (SO42−, Cl−, NO3−) on Ni accumulation and toxicity in aquatic plant (Lemna minor L.): Implications for Ni risk assessment. Environ. Toxicol. Chem. 2013, 32, 810–821. [Google Scholar] [CrossRef]
- Shahzad, B.; Tanveer, M.; Rehman, A.; Cheema, S.A.; Fahad, S.; Rehman, S.; Sharma, A. Nickel; whether toxic or essential for plants and environment—A review. Plant Physiol. Biochem. 2018, 132, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Schlekat, C.E.; McGeer, J.C.; Blust, R.; Borgmann, U.; Brix, K.; Bury, N.; Couillard, Y.; Dwyer, R.L.; Luoma, S.N.; Robertson, S.; et al. Bioaccumulation: Hazard identification of metals and inorganic metal substances. In Assessing the Hazard of Metals and Inorganic Metal Substances in Aquatic and Terrestrial Systems; Adams, W.J., Chapman, P.M., Eds.; SETAC Press: Pensacola, FL, USA, 2006; pp. 55–89. [Google Scholar]
- DeForest, D.; Schlekat, C.; Brix, K.; Fairbother, A. Secondary poisoning of nickel. Integr. Environ. Assess. Manag. 2011, 8, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Van der Ent, A.; Baker, A.J.M.; Reeves, R.D.; Pollard, A.J.; Schat, H. Hyperaccumulators of metal and metalloid trace elements: Facts and fiction. Plant Soil 2013, 362, 319–334. [Google Scholar] [CrossRef]
- Meyer, J.S.; Farley, K.J.; Garman, E.R. Metal Mixtures Modeling Evaluation project: 1. Background. Environ. Toxicol. Chem. 2015, 34, 726–740. [Google Scholar] [CrossRef] [PubMed]
- Farley, K.J.; Meyer, J.S. Metal Mixture Modeling Evaluation project: 3. Lessons learned and steps forward. Environ. Toxicol. Chem. 2015, 34, 821–832. [Google Scholar] [CrossRef] [PubMed]
- Nys, C.; Janssen, C.R.; De Schamphelaere, K.A.C. Development and validation of a metal mixture bioavailability model (MMBM) to predict chronic toxicity of Ni–Zn–Pb mixtures to Ceriodaphnia dubia. Environ. Pollut. 2017, 220 Pt B, 1271–1281. [Google Scholar] [CrossRef]
- Nys, C.; Van Regenmortel, T.; Janssen, C.R.; Oorts, K.; Smolders, E.; De Schamphelaere, K.A.C. A framework for ecological risk assessment of metal mixtures in aquatic systems. Environ. Toxicol. Chem. 2018, 37, 623–642. [Google Scholar] [CrossRef] [PubMed]
- Lynch, N.R.; Hoang, T.C.; O’Brien, T.E. Acute toxicity of binary-metal mixtures of copper, zinc, and nickel to Pimephales promelas: Evidence of more-than-additive effect. Environ. Toxicol. Chem. 2015, 35, 446–457. [Google Scholar] [CrossRef]
- Traudt, E.M.; Ranville, J.F.; Meyer, J.S. Effect of age on acute toxicity of cadmium, copper, nickel, and zinc in individual-metal exposures to Daphnia magna neonates. Environ. Toxicol. Chem. 2016, 36, 113–119. [Google Scholar] [CrossRef]
- Traudt, E.M.; Ranville, J.F.; Meyer, J.S. Acute toxicity of ternary Cd–Cu–Ni and Cd–Ni–Zn mixtures to Daphnia magna: Dominant metal pairs change along a concentration gradient. Environ. Sci. Technol. 2017, 51, 4471–4481. [Google Scholar] [CrossRef]
- Crémazy, A.; Brix, K.V.; Wood, C.M. Chronic toxicity of binary mixtures of six metals (Ag, Cu, Cd, Pb, Ni and Zn) to the great pond snail Lymnaea stagnalis. Environ. Sci. Technol. 2018, 52, 5979–5988. [Google Scholar] [CrossRef] [PubMed]
- Crémazy, A.; Brix, K.V.; Wood, C.M. Using the Biotic Ligand Model framework to investigate binary metal interactions on the uptake of Ag, Cd, Cu, Ni, Pb and Zn in the freshwater snail Lymnaea stagnalis. Sci. Total Environ. 2019, 647, 1611–1625. [Google Scholar] [CrossRef] [PubMed]
- Katsnelson, B.A.; Panov, V.G.; Minigaliyeva, I.A.; Varaksin, A.N.; Privalova, L.I.; Slyshkina, T.V.; Grebenkina, S.V. Further development of the theory and mathematical description of combined toxicity: An approach to classifying types of action of three-factorial combinations (a case study of manganese-chromium-nickel subchronic intoxication). Toxicology 2015, 334, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Minigaliyeva, I.A.; Katsnelson, B.A.; Privalova, L.I.; Gurvich, V.B.; Panov, V.G.; Varaksin, A.N.; Makeyev, O.H.; Sutunkova, M.P.; Loginova, N.V.; Kireyeva, E.P.; et al. Toxicodynamic and Toxicokinetic descriptors of combined Chromium (VI) and Nickel toxicity. Int. J. Toxicol. 2014, 33, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Kenston, S.S.F.; Su, H.; Li, Z.; Kong, L.; Wang, Y.; Song, X.; Gu, Y.; Barber, T.; Aldinger, J.; Hua, Q.; et al. The systemic toxicity of heavy metal mixtures in rats. Toxicol. Res. 2018, 7, 396–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- International Agency for Research on Cancer (IARC). Welding, Molybdenum Trioxide, and Indium Tin Oxide. IARC Monogr. Eval. Carcinog. Risks Hum. 2018, 118, 36–265. [Google Scholar]
- Zimmer, A.T. The influence of metallurgy on the formation of welding aerosols. J. Environ. Monit. 2002, 4, 628–632. [Google Scholar] [CrossRef]
- Zeidler-Erdely, P.C.; Meighan, T.G.; Erdely, A.; Battelli, L.A.; Kashon, M.L.; Keane, M.; Antonini, J.M. Lung tumor promotion by chromium-containing welding particulate matter in a mouse model. Part. Fibre Toxicol. 2013, 10, 45. [Google Scholar] [CrossRef]
- Falcone, L.M.; Erdely, A.; Meighan, T.G.; Battelli, L.A.; Salmen, R.; McKinney, W.; Stone, S.; Cumpston, A.; Cumpston, J.; Andrews, R.N.; et al. Inhalation of gas metal arc-stainless steel welding fume promotes lung tumorigenesis in A/J mice. Arch. Toxicol. 2017, 91, 2953–2962. [Google Scholar] [CrossRef]
- Zeidler-Erdely, P.C.; Falcone, L.M.; Antonini, J.M. Influence of welding fume metal composition on lung toxicity and tumor formation in experimental animal models. J. Occup. Environ. Hyg. 2019, 16, 372–377. [Google Scholar] [CrossRef]
- Future Markets, Inc. The Global Market for Metal and Metal Oxide Nanoparticles, 2010–2027, 4th ed.; Future Markets, Inc.: Edinburgh, UK, 2017. [Google Scholar]
- Biskos, G.; Schmidt-Ott, A. Airborne engineered nanoparticles: Potential risks and monitoring challenges for assessing their impacts on children. Paediatr. Respir. Rev. 2012, 13, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Munoz, A.; Costa, M. Elucidating the mechanisms of nickel compound uptake: A review of particulate and nano-nickel endocytosis and toxicity. Toxicol. Appl. Pharmacol. 2012, 260, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magaye, R.; Zhao, J. Recent progress in studies of metallic nickel and nickel-based nanoparticles’ genotoxicity and carcinogenicity. Environ. Toxicol. Pharmacol. 2012, 34, 644–650. [Google Scholar] [CrossRef] [PubMed]
- Sutunkova, M.P.; Privalova, L.I.; Minigalieva, I.A.; Gurvich, V.B.; Panov, V.G.; Katsnelson, B.A. The most important inferences from the Ekaterinburg nanotoxicology team’s animal experiments assessing adverse health effects of metallic and metal oxide nanoparticles. Toxicol. Rep. 2018, 5, 363–376. [Google Scholar] [CrossRef] [PubMed]
- Sutunkova, M.P.; Solovyeva, S.N.; Minigalieva, I.A.; Gurvich, V.B.; Valamina, I.E.; Makeyev, O.H.; Shur, V.Y.; Shishkina, E.V.; Zubarev, I.V.; Saatkhudinova, R.R.; et al. Toxic effects of low-level long-term inhalation exposures of rats to nickel oxide nanoparticles. Int. J. Mol. Sci. 2019, 20, 1778. [Google Scholar] [CrossRef] [PubMed]
- Horie, M.; Stowe, M.; Tabei, M.; Kuroda, E. Metal ion release of manufactured metal oxide nanoparticles is involved in the allergic response to inhaled ovalbumin in mice. Occup. Dis. Environ. 2016, 4, 17–26. [Google Scholar] [CrossRef]
- Ispas, C.; Andreescu, D.; Patel, A.; Goia, D.V.; Andreescu, S.; Wallace, K.N. Toxicity and developmental defects of different sizes and shape nickel nanoparticles in zebrafish. Environ. Sci. Technol. 2009, 43, 6349–6356. [Google Scholar] [CrossRef]
- Ogami, A.; Morimoto, Y.; Myojo, T.; Oyabu, T.; Murakami, M.; Todoroki, M.; Nishi, K.; Kadoya, C.; Yamamoto, M.; Tanaka, I. Pathological features of different sizes of nickel oxide following intratracheal instillation in rats. Inhal. Toxicol. 2009, 21, 812–818. [Google Scholar] [CrossRef]
- Latvala, S.; Hedberg, J.; Di Bucchianico, S.; Möller, L.; Odnevall Wallinder, I.; Elihn, K.; Karlsson, H.L. Nickel release, ROS generation and toxicity of Ni and NiO micro- and nanoparticles. PLoS ONE 2016, 11, e0159684. [Google Scholar] [CrossRef]
- Kong, L.; Tang, M.; Zhang, T.; Wang, D.; Hu, K.; Lu, W.; Wei, C.; Liang, G.; Pu, Y. Nickel nanoparticles exposure and reproductive toxicity in healthy adult rats. Int. J. Mol. Sci. 2014, 15, 21253–21269. [Google Scholar] [CrossRef]
- Gallo, A.; Boni, R.; Buttino, I.; Tosti, E. Spermiotoxicity of nickel nanoparticles in the marine invertebrate Ciona intestinalis (Ascidians). Nanotoxicology 2016, 10, 1096–1104. [Google Scholar] [CrossRef] [PubMed]
- Griffitt, R.J.; Luo, J.; Gao, J.; Bonzongo, J.-C.; Barber, D.S. Effects of particle composition and species on toxicity of metallic nanomaterials in aquatic organisms. Environ. Toxicol. Chem. 2008, 27, 1972–1978. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Vitiello, V.; Casals, E.; Puntes, V.F.; Iamunno, F.; Pellegrini, D.; Changwen, W.; Benvenuto, G.; Buttino, I. Toxicity of nickel in the marine calanoid copepod Acartia tonsa: Nickel chloride versus nanoparticles. Aquat. Toxicol. 2016, 170, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Oukarroum, A.; Barhoumi, L.; Samadani, M.; Dewez, D. Toxic effects of nickel oxide bulk and nanoparticles on the aquatic plant Lemna gibba L. BioMed. Res. Int. 2015, 2015, 501326. [Google Scholar] [CrossRef]
- Minigalieva, I.A.; Katsnelson, B.A.; Privalova, L.I.; Sutunkova, M.P.; Gurvich, V.B.; Shur, V.Y.; Shishkina, E.V.; Valamina, I.E.; Makeyev, O.H.; Panov, V.G.; et al. Attenuation of combined nickel(II) oxide and manganese(II, III) oxide nanoparticles’ adverse effects with a complex of bioprotectors. Int. J. Mol. Sci. 2015, 16, 22555–22583. [Google Scholar] [CrossRef] [PubMed]
- Minigalieva, I.; Bushueva, T.; Fröhlich, E.; Meindl, C.; Öhlinger, K.; Panov, V.; Varaksin, A.; Shur, V.; Shishkina, E.; Gurvich, V.; et al. Are in vivo and in vitro assessments of comparative and combined toxicity of the same metallic nanoparticles compatible, or contradictory, or both? A juxtaposition of data obtained in respective experiments with NiO and Mn3O4 nanoparticles. Food Chem. Toxicol. 2017, 109 Pt 1, 393–404. [Google Scholar] [CrossRef]
- Katsnelson, B.A.; Minigaliyeva, I.A.; Panov, V.G.; Privalova, L.I.; Varaksin, A.N.; Gurvich, V.B.; Sutunkova, M.P.; Shur, V.Y.; Shishkina, E.V.; Valamina, I.E.; et al. Some patterns of metallic nanoparticles’ combined subchronic toxicity as exemplified by a combination of nickel and manganese oxide nanoparticles. Food Chem. Toxicol. 2015, 86, 351–364. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.A. Evaluation of some biological, biochemical, and hematological aspects in male albino rats after acute exposure to the nano-structured oxides of nickel and cobalt. Environ. Sci. Pollut. Res. Int. 2019, 26, 17407–17417. [Google Scholar] [CrossRef]
- Fischer, L.A.; Menné, T.; Johansen, J.D. Experimental nickel elicitation thresholds—A review focusing on occluded nickel exposure. Contact Dermat. 2005, 52, 57–64. [Google Scholar] [CrossRef]
- Buekers, J.; De Brouwere, K.; Lefebvre, W.; Willems, H.; Vendenbroele, M.; Van Sprang, P.; Eliat-Eliat, M.; Hicks, K.; Schlekat, C.E.; Oller, A.R. Assessment of human exposure to environmental sources of nickel in Europe: Inhalation exposure. Sci. Total Environ. 2015, 521–522, 359–371. [Google Scholar] [CrossRef]

| Genotoxicity/Carcinogenicity | In Vitro Studies | In Vivo Animal Studies | Human Studies |
|---|---|---|---|
| DNA damage | Positive, indirect damage; e.g., single strand breaks, DNA fragmentation [78,79,80] | Positive [81,82,83] | Positive in welders but unassignable to Ni [84,85]; positive in mucosa of orthodontic patients but unassignable to Ni [86,87,88] |
| Impair DNA repair | Positive [74,89,90,91] | ||
| Gene Mutations | Low-to-none [92,93,94,95] | Low mutations [23,92,96] | |
| Chromosomal alterations | a Mixed results [23,96,97,98] | a Mixed results [99,100,101] | a Mixed results [102,103,104,105,106,107] |
| HIF-1α-dependent changes | Stabilization of HIF-1α, increased HIF-1α-dependent gene expressions, e.g., Cap43 [108,109,110,111] | Increased HIF-1α-dependent Cap43 expression [108], hematocrit and/or hemoglobin [51] | |
| Apoptosis/Autophagy | Increase [109,112,113,114,115] | Increase via increased expression of caspase 8 in workers [116] | |
| Histone modifications | |||
| *Hypermethylation | Positive at H3K9 [117,118] | In workers at H3K4 [119,120,121] | |
| *Hyperphosphorylation | Positive at H3S10 [122] | ||
| *Hyperubiquitination | Positive, inhibiting deubiquitinating enzyme activity [123] | ||
| *Hypo/hyperacetylation | Hypoacetylation via inhibiting histone acetyltransferase activity [124,125] | Increased H3K9 acetylation in steel workers [121] | |
| Oxidative stress, ROS production | Positive [78,124,126,127] | Positive in rats and mice [80,128,129] | Oxidative DNA lesions linked to nickel levels [130] |
| Human Health Effects | ||
| Endpoint (Underlying Studies and Data) | Threshold Values a | Reference |
| NACD (human) | Dermal-NOAEL = 0.44 µg Ni/cm2 skin/day a | [267] |
| Oral-BMDL0.1 = 4.3 µg Ni/kg bw/day adult (in addition to diet) | [145] | |
| Respiratory cancer (human) | Inhalation—practical threshold = 0.1 mg Ni/m3 inhalable aerosol | [13] |
| Inhalation—practical threshold = 0.5 µg Ni/m3 ng/m3 PM10 aerosol | [268] | |
| Respiratory non cancer (animal) | Inhalation-NOAEC = 0.03 mg/m3 (Ni sulfate) respirable aerosol | [13] |
| Inhalation-NOAEC = 9.4 µg/m3 (Ni sulfate) PM10 aerosol | [268] | |
| Reproductive (animal) | Oral-BMDL0.05 = 1.8 mg Ni/kg bw/day adult | [145] |
| Environmental Health Effects | ||
| Compartments | Threshold Values a | Reference |
| Freshwater | Bioavailability-based EQS = 4 µg Ni/L | [208] |
| HC5 = 7.1 to 43.6 µg Ni/L for EU waters representing the 10th to 90th percentile of bioavailability conditions | [196] | |
| Marine water | Temperate HC5 = 8.6 µg Ni/L | [209] |
| Tropical aggregated HC5 = 8.2 µg Ni/L | [212] | |
| Sediment | HC5 = 136–437 mg Ni/kg (AVS: 0.77–38.4 µM/g dw) | [202] |
| Soil | HC5 = 8.6–194.3 mg Ni/kg (eCEC: 10.4–36.0 cmol/kg) | [194] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buxton, S.; Garman, E.; Heim, K.E.; Lyons-Darden, T.; Schlekat, C.E.; Taylor, M.D.; Oller, A.R. Concise Review of Nickel Human Health Toxicology and Ecotoxicology. Inorganics 2019, 7, 89. https://doi.org/10.3390/inorganics7070089
Buxton S, Garman E, Heim KE, Lyons-Darden T, Schlekat CE, Taylor MD, Oller AR. Concise Review of Nickel Human Health Toxicology and Ecotoxicology. Inorganics. 2019; 7(7):89. https://doi.org/10.3390/inorganics7070089
Chicago/Turabian StyleBuxton, Samuel, Emily Garman, Katherine E. Heim, Tara Lyons-Darden, Christian E. Schlekat, Michael D. Taylor, and Adriana R. Oller. 2019. "Concise Review of Nickel Human Health Toxicology and Ecotoxicology" Inorganics 7, no. 7: 89. https://doi.org/10.3390/inorganics7070089
APA StyleBuxton, S., Garman, E., Heim, K. E., Lyons-Darden, T., Schlekat, C. E., Taylor, M. D., & Oller, A. R. (2019). Concise Review of Nickel Human Health Toxicology and Ecotoxicology. Inorganics, 7(7), 89. https://doi.org/10.3390/inorganics7070089





