Low-Temperature Ordering in the Cluster Compound (Bi8)Tl[AlCl4]3
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis and Role of the Ionic Liquid
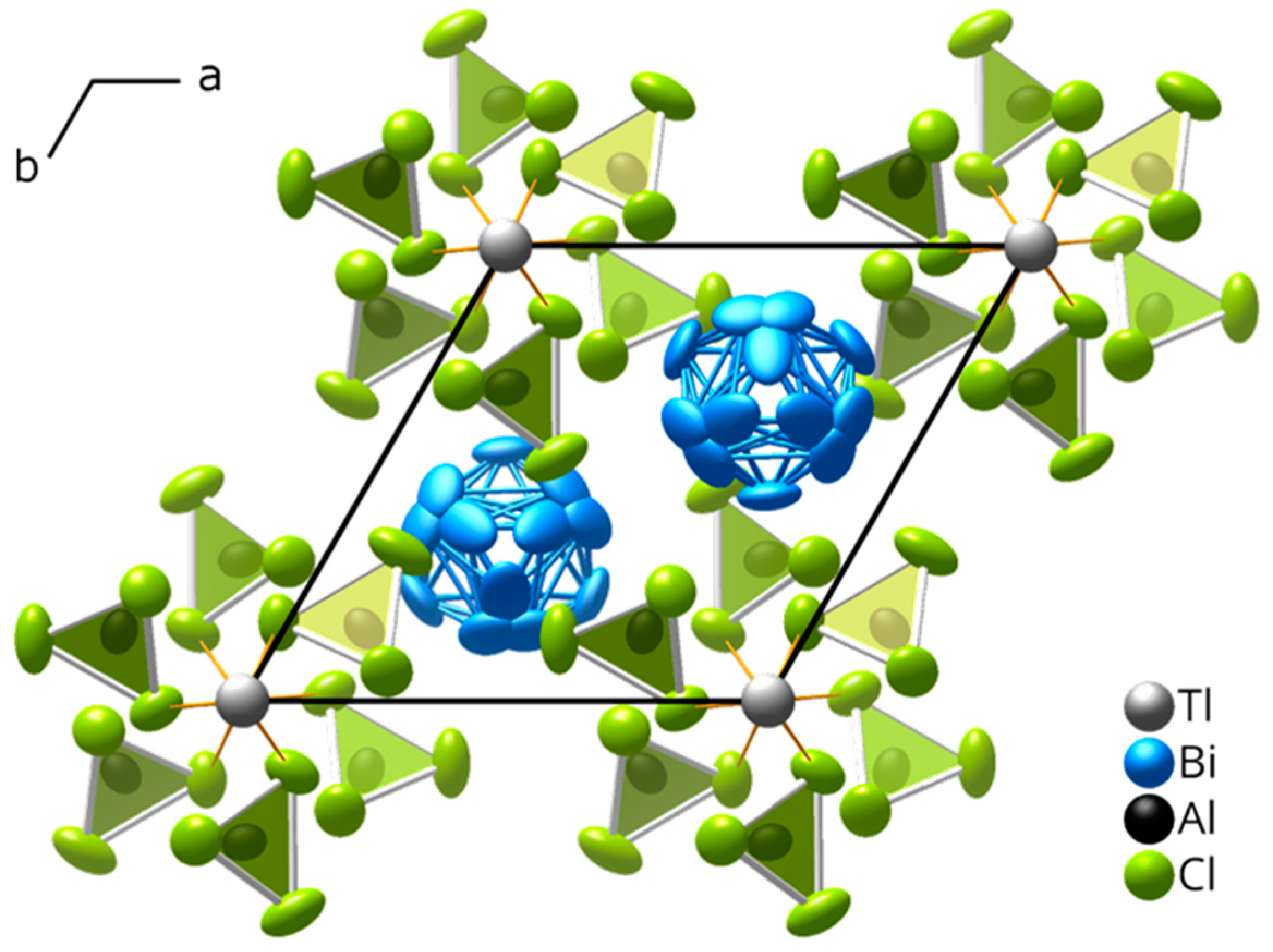
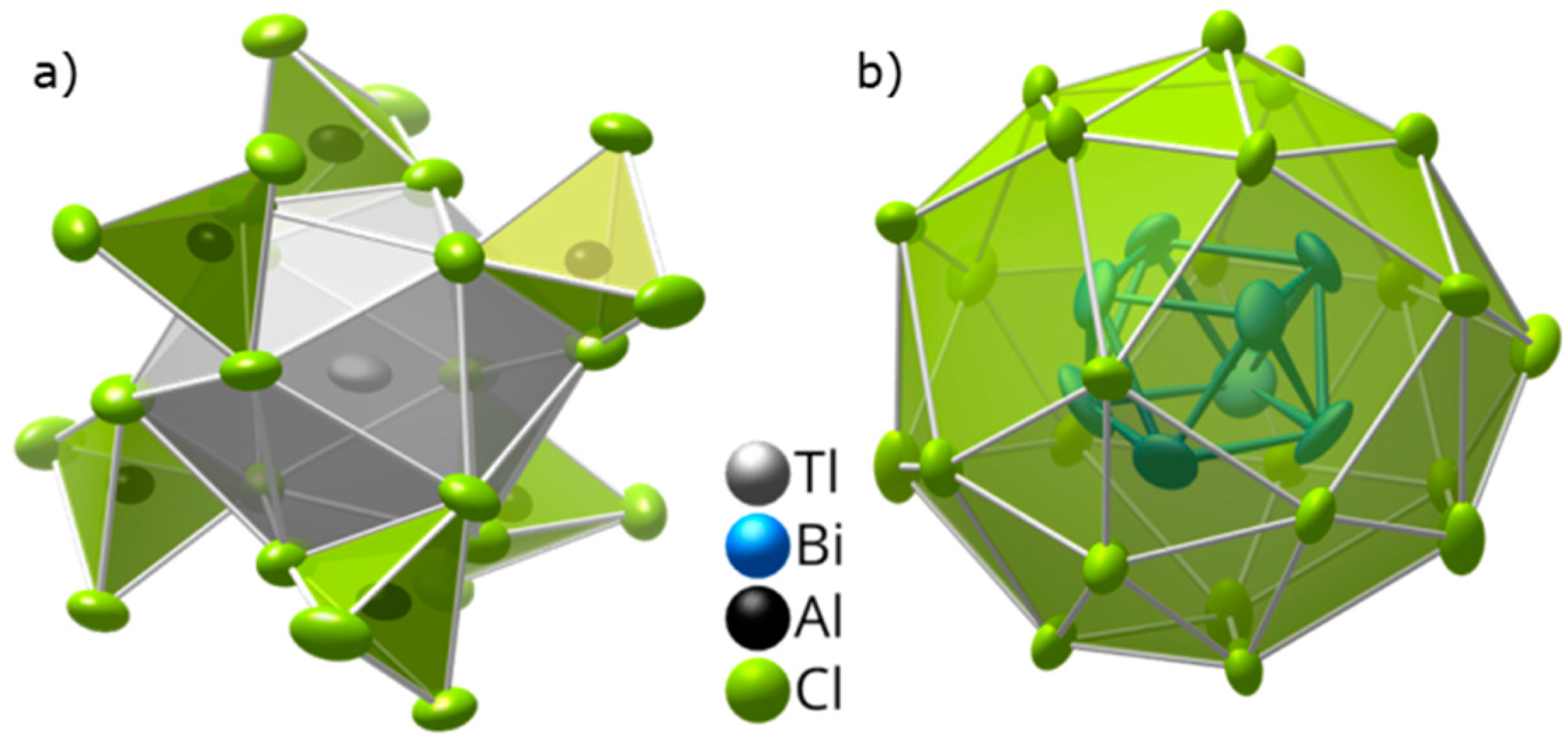
2.2. Crystal Structure of (Bi8)Tl[AlCl4]3 at Room Temperature
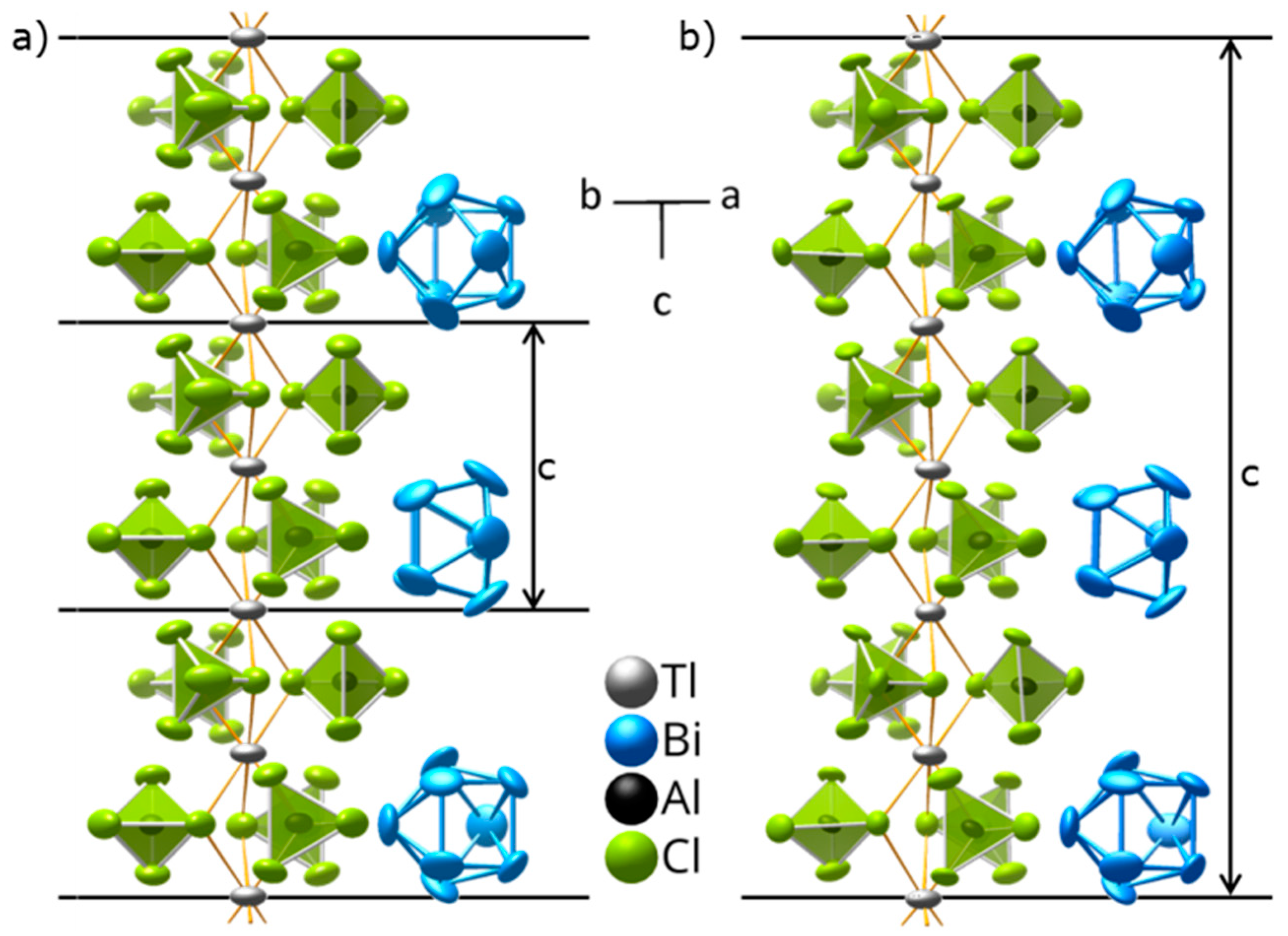
2.3. Low-Temperature Structure of (Bi8)Tl[AlCl4]3
3. Materials and Methods
3.1. Synthesis Results and Role of the Ionic Liquid
3.2. EDS Analysis
3.3. X-ray Crystal Structure Determination
3.3.1. Crystallographic Data for (Bi8)Tl[AlCl4]3 at 296(1) K
3.3.2. Crystallographic Data for (Bi8)Tl[AlCl4]3 at 170(2) K
3.4. Differential Scanning Calorimetry
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hershaft, A.; Corbett, J.D. Crystal Structure of Bismuth Monochloride. J. Chem. Phys. 1962, 36, 551–552. [Google Scholar] [CrossRef]
- Ruck, M.; Steden, F. Bi53+-Polykationen in geordneten und plastischen Kristallen von Bi5[AlI4]3 und Bi5[AlBr4]3. Z. Anorg. Allg. Chem. 2007, 633, 1556–1562. [Google Scholar] [CrossRef]
- Fischer, A.; Lindsjö, M.; Kloo, L. Improvements of and Insights into the Isolation of Bismuth Polycations from Benzene Solution—Single-Crystal Structure Determinations of Bi8[GaCl4]2 and Bi5[GaCl4]3. Eur. J. Inorg. Chem. 2005, 2005, 670–675. [Google Scholar] [CrossRef]
- Corbett, J.D. Homopolyatomic ions of the heavy post-transition elements. Preparation, properties, and bonding of Bi5(AlCl4)3 and Bi4(AlCl4). Inorg. Chem. 1968, 7, 198–208. [Google Scholar] [CrossRef]
- Ahmed, E.; Köhler, D.; Ruck, M. Room-Temperature Synthesis of Bismuth Clusters in Ionic Liquids and Crystal Growth of Bi5(AlCl4)3. Z. Anorg. Allg. Chem. 2009, 635, 297–300. [Google Scholar] [CrossRef]
- Hampel, S.; Schmidt, P.; Ruck, M. Synthese, thermochemische Eigenschaften und Kristallstruktur von Bi7Cl10. Z. Anorg. Allg. Chem. 2005, 631, 272–283. [Google Scholar] [CrossRef]
- Beck, J.; Brendel, C.J.; Bengtsson-Kloo, L.; Krebs, B.; Mummert, M.; Stankowski, A.; Ulvenlund, S. The Crystal Structure of Bi8(AlCl4)2 and the Crystal Structure, Conductivity, and Theoretical Band Structure of Bi6Cl7 and Related Subvalent Bismuth Halides. Chem. Ber. 1996, 129, 1219–1226. [Google Scholar] [CrossRef]
- Ruck, M. Bi24Ru3Br20: Ein pseudo-tetragonales Subbromid mit [RuBi6Br12]-Clustern und [Ru2Bi17Br4]-Gruppen. Z. Anorg. Allg. Chem. 1997, 623, 1591–1598. [Google Scholar] [CrossRef]
- Wahl, B.; Ruck, M. Die molekularen Cluster [Bi10Au2](EBi3X9)2 (E = As, Bi; X = Cl, Br)—Synthese, Kristallstrukturen, Drillingsbildung und chemische Bindung. Z. Anorg. Allg. Chem. 2008, 634, 2267–2275. [Google Scholar] [CrossRef]
- Groh, M.F.; Isaeva, A.; Frey, C.; Ruck, M. [Ru(Bi8)2]6+—A Cluster in a Highly Disordered Crystal Structure is the Key to the Understanding of the Coordination Chemistry of Bismuth Polycations. Z. Anorg. Allg. Chem. 2013, 639, 2401–2405. [Google Scholar] [CrossRef]
- Groh, M.F.; Isaeva, A.; Ruck, M. [Ru2Bi14Br4](AlCl4)4 by Mobilization and Reorganization of Complex Clusters in Ionic Liquids. Chem. Eur. J. 2012, 18, 10886–10891. [Google Scholar] [CrossRef] [PubMed]
- Knies, M.; Kaiser, M.; Isaeva, A.; Müller, U.; Doert, T.; Ruck, M. The Intermetalloid Cluster Cation (CuBi8)3+. Chem. Eur. J. 2018, 24, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Müller, U.; Isaeva, A.; Richter, J.; Knies, M.; Ruck, M. Polyhedral Bismuth Polycations Coordinating Gold(I) with Varied Hapticity in a Homoleptic Heavy-Metal Cluster. Eur. J. Inorg. Chem. 2016, 2016, 3580–3584. [Google Scholar] [CrossRef]
- Ruck, M.; Locherer, F. Coordination chemistry of homoatomic ligands of bismuth, selenium and tellurium. Coord. Chem. Rev. 2015, 285, 1–10. [Google Scholar] [CrossRef]
- Groh, M.F.; Müller, U.; Isaeva, A.; Ruck, M. Ionothermal Syntheses, Crystal Structures, and Chemical Bonding of the Rhodium-Centered Clusters [RhBi9]4+ and [(RhBi7)I8]. Z. Anorg. Allg. Chem. 2017, 643, 1482–1490. [Google Scholar] [CrossRef]
- Groh, M.F.; Wolff, A.; Wahl, B.; Rasche, B.; Gebauer, P.; Ruck, M. Pentagonal Bismuth Antiprisms with Endohedral Palladium or Platinum Atoms by Low-Temperature Syntheses. Z. Anorg. Allg. Chem. 2017, 643, 69–80. [Google Scholar] [CrossRef]
- Wosylus, A.; Dubenskyy, V.; Schwarz, U.; Ruck, M. Dynamic Disorder of Bi82+ Clusters in the Plastic Phase (Bi8)3Bi[InI4]9. Z. Anorg. Allg. Chem. 2009, 635, 1030–1035. [Google Scholar] [CrossRef]
- Wilkes, J.S. Ionic Liquids in Synthesis, 2nd ed.; Wasserscheid, P., Welton, T., Eds.; Wiley-VCH: Weinheim, Germany, 2007; pp. 1–6. [Google Scholar]
- Estager, J.; Holbrey, J.D.; Swadźba-Kwaśny, M. Halometallate ionic liquids—Revisited. Chem. Soc. Rev. 2014, 43, 847–886. [Google Scholar] [CrossRef] [PubMed]
- Timofte, T.; Mudring, A.-V. A Systematic Study on the Crystal Structures of TlMX4 (M = Al, Ga; X = Cl, Br, I). Z. Anorg. Allg. Chem. 2009, 635, 840–847. [Google Scholar] [CrossRef]
- Wade, K. Structural and Bonding Patterns in Cluster Chemistry. Adv. Inorg. Chem. Radiochem. 1976, 18, 1–66. [Google Scholar] [CrossRef]
- Wenninger, M.J. Polyhedron Models; Cambridge University Press: Cambridge, UK, 1971; pp. 20–32. [Google Scholar] [CrossRef]
- MacGillivray, L.R.; Atwood, J.L. A chiral spherical molecular assembly held together by 60 hydrogen bonds. Nature 1997, 389, 469–472. [Google Scholar] [CrossRef]
- Shoemaker, D.P.; Marsh, R.E.; Ewing, F.J.; Pauling, L. Interatomic distances and atomic valences in NaZn13. Acta Crystallogr. 1952, 5, 637–644. [Google Scholar] [CrossRef]
- Xie, W.; Cava, R.J.; Miller, G.J. Packing of Russian doll clusters to form a nanometer-scale CsCl-type compound in a Cr–Zn–Sn complex metallic alloy. J. Mater. Chem. C 2017, 5, 7215–7221. [Google Scholar] [CrossRef]
- McElfresh, M.W.; Hall, J.H.; Ryan, R.R.; Smith, J.L.; Fisk, Z. Structure of the heavy-fermion superconductor UBe13. Acta Crystallogr. Sect. C 1990, 46, 1579–1580. [Google Scholar] [CrossRef]
- Oliva, J.M.; Vegas, Á. Merging boron solid state and molecular chemistry: Energy landscapes in the exo/endo closo-borane complex Sc[B24H24]+. Chem. Phys. Lett. 2012, 533, 50–55. [Google Scholar] [CrossRef]
- Perenthaler, E.; Schulz, H.; Rabenau, A. Die Strukturen von LiAlCl4 und NaAlCl4 als Funktion der Temperatur. Z. Anorg. Allg. Chem. 1982, 491, 259–265. [Google Scholar] [CrossRef]
- Baenziger, N.C. The crystal structure of NaAlCl4. Acta Crystallogr. 1951, 4, 216–219. [Google Scholar] [CrossRef]
- Mairesse, G.; Barbier, P.; Wignacourt, J.-P. Potassium tetrachloroaluminate. Acta Crystallogr. Sect. B 1978, 34, 1328–1330. [Google Scholar] [CrossRef]
- Takeda, Y.; Kanamura, F.; Shimada, M.; Koizumi, M. The crystal structure of BaNiO3. Acta Crystallogr. Sect. B 1976, 32, 2464–2466. [Google Scholar] [CrossRef]
- Lander, J.J. The crystal structures of NiO·3BaO, NiO·BaO, BaNiO3 and intermediate phases with composition near Ba2Ni2O5; with a note on NiO. Acta Crystallogr. 1951, 4, 148–156. [Google Scholar] [CrossRef]
- Goldschmidt, V.M. Die Gesetze der Krystallochemie. Naturwissenschaften 1926, 14, 477–485. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr. Sect. A 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Hahn, F. X-Shape, Crystal Optimization for Numerical Absorption Correction Program; Stoe & Cie GmbH: Darmstadt, Germany, 2008. [Google Scholar]
- Petricek, V.; Dusek, M.; Palatinus, L. JANA2006, The Crystallographic Computing System; Institute of Physics: Praha, Czech Republic, 2011. [Google Scholar]
- Palatinus, L.; Chapuis, G. SUPERFLIP—A computer program for the solution of crystal structures by charge flipping in arbitrary dimensions. J. Appl. Cryst. 2007, 40, 786–790. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXL, Program for Crystal Structure Refinement—Multi-CPU; Georg-August-Universität Göttingen: Göttingen, Germany, 2014. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Knies, M.; Kaiser, M.; Lê Anh, M.; Efimova, A.; Doert, T.; Ruck, M. Low-Temperature Ordering in the Cluster Compound (Bi8)Tl[AlCl4]3. Inorganics 2019, 7, 45. https://doi.org/10.3390/inorganics7040045
Knies M, Kaiser M, Lê Anh M, Efimova A, Doert T, Ruck M. Low-Temperature Ordering in the Cluster Compound (Bi8)Tl[AlCl4]3. Inorganics. 2019; 7(4):45. https://doi.org/10.3390/inorganics7040045
Chicago/Turabian StyleKnies, Maximilian, Martin Kaiser, Mai Lê Anh, Anastasia Efimova, Thomas Doert, and Michael Ruck. 2019. "Low-Temperature Ordering in the Cluster Compound (Bi8)Tl[AlCl4]3" Inorganics 7, no. 4: 45. https://doi.org/10.3390/inorganics7040045
APA StyleKnies, M., Kaiser, M., Lê Anh, M., Efimova, A., Doert, T., & Ruck, M. (2019). Low-Temperature Ordering in the Cluster Compound (Bi8)Tl[AlCl4]3. Inorganics, 7(4), 45. https://doi.org/10.3390/inorganics7040045





