In Situ Studies and Magnetic Properties of the Cmcm Polymorph of LiCoPO4 with a Hierarchical Dumbbell-Like Morphology Synthesized by Easy Single-Step Polyol Synthesis
Abstract
:1. Introduction
2. Results and Discussion
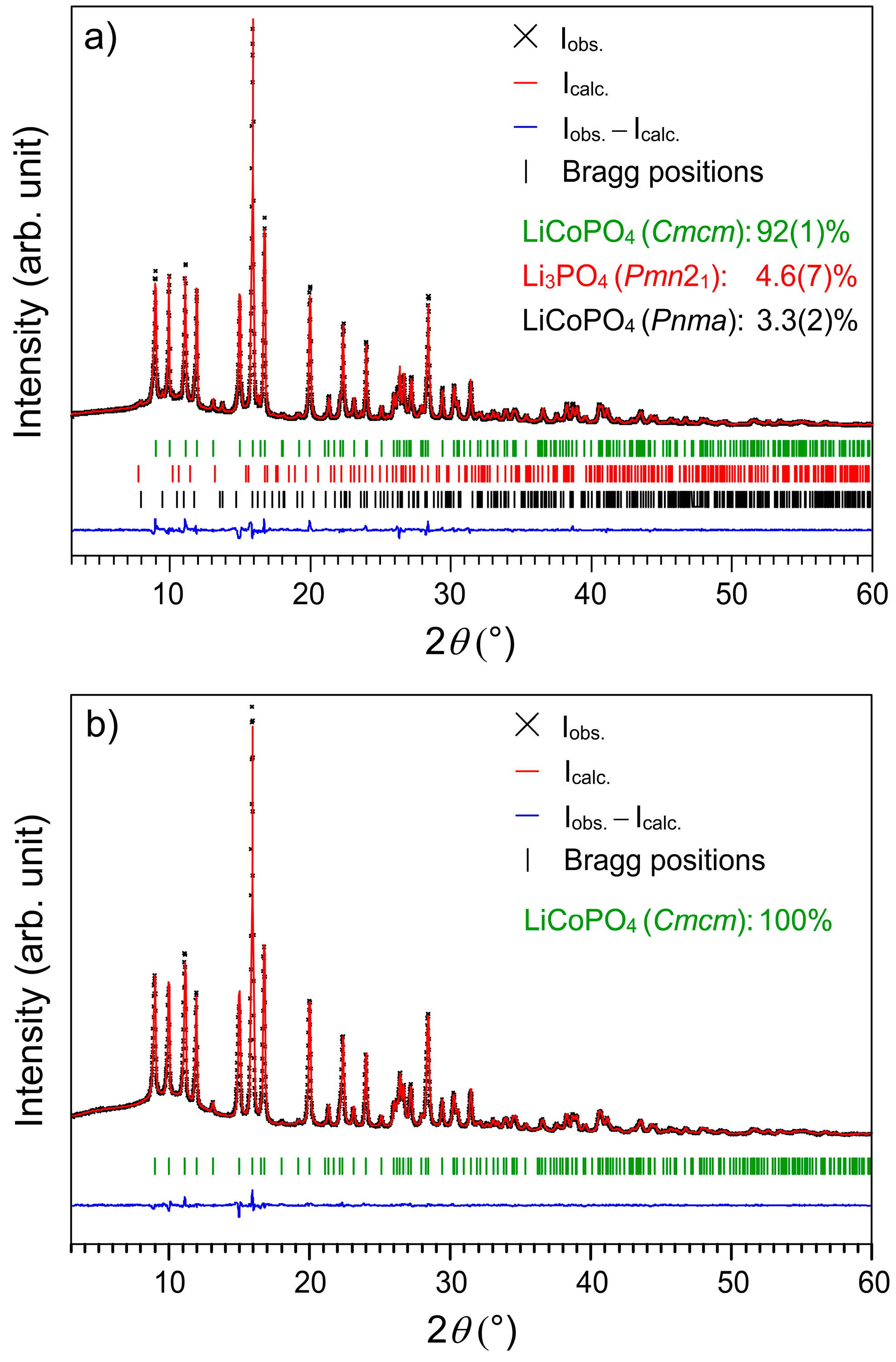
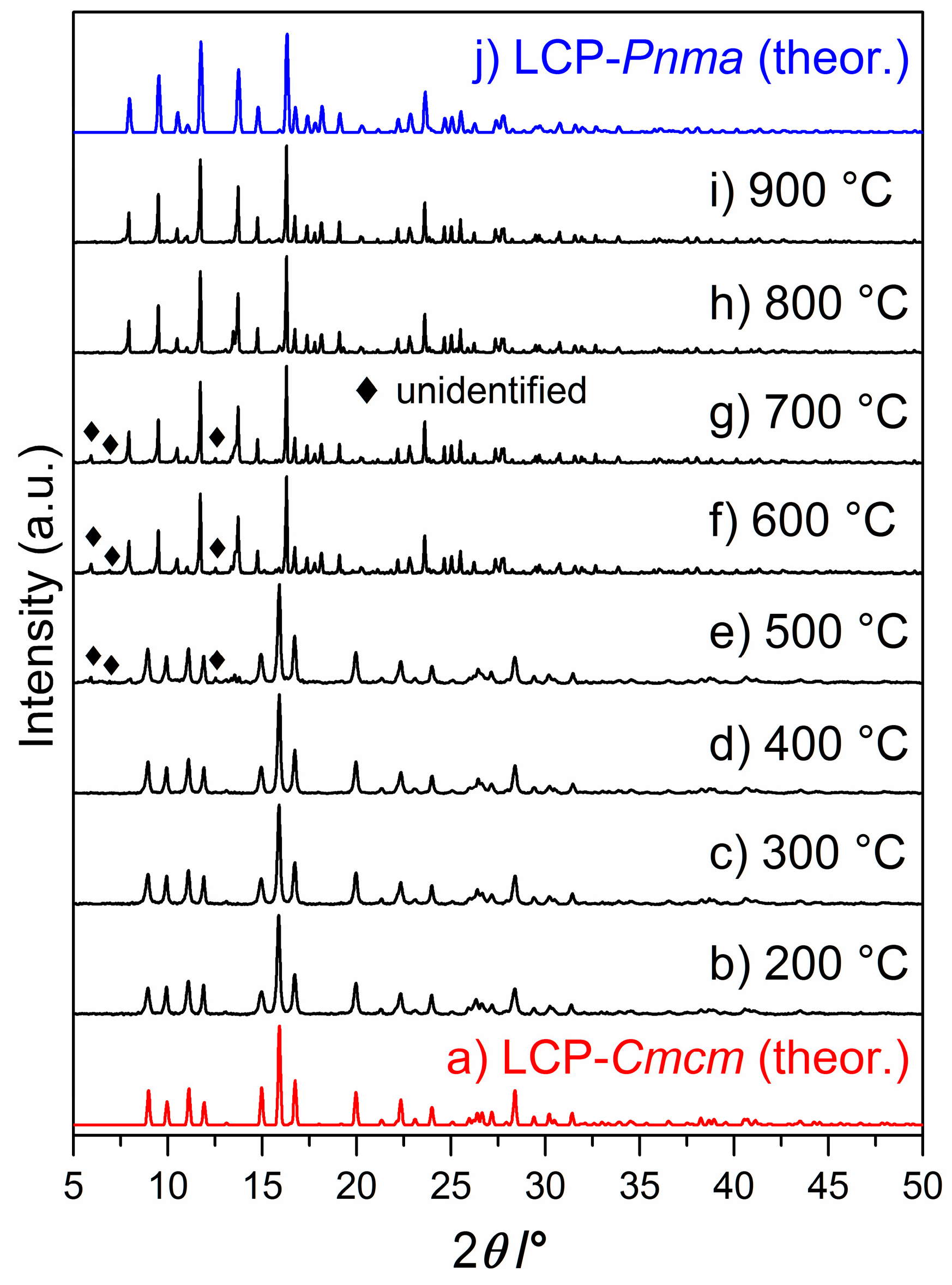
2.1. X-ray Powder Diffraction
2.2. Scanning Electron Microscopy (SEM)
2.3. Elemental Analysis
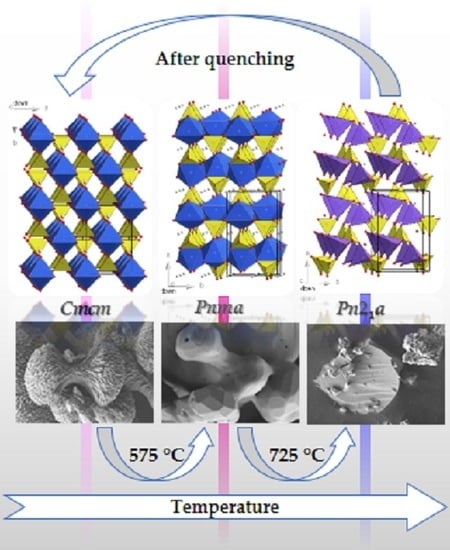
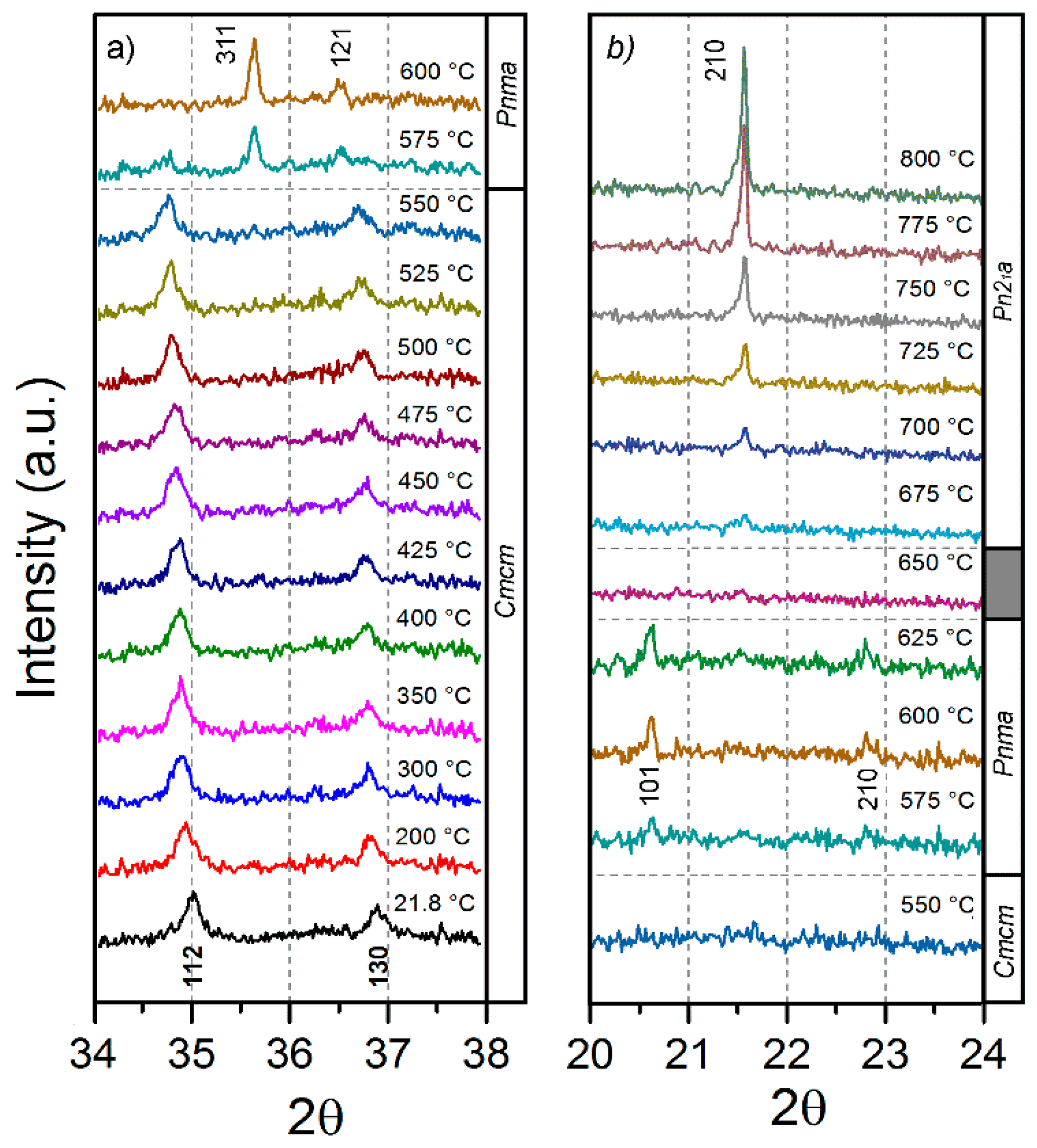
2.4. Thermal Stability and Phase Transformation
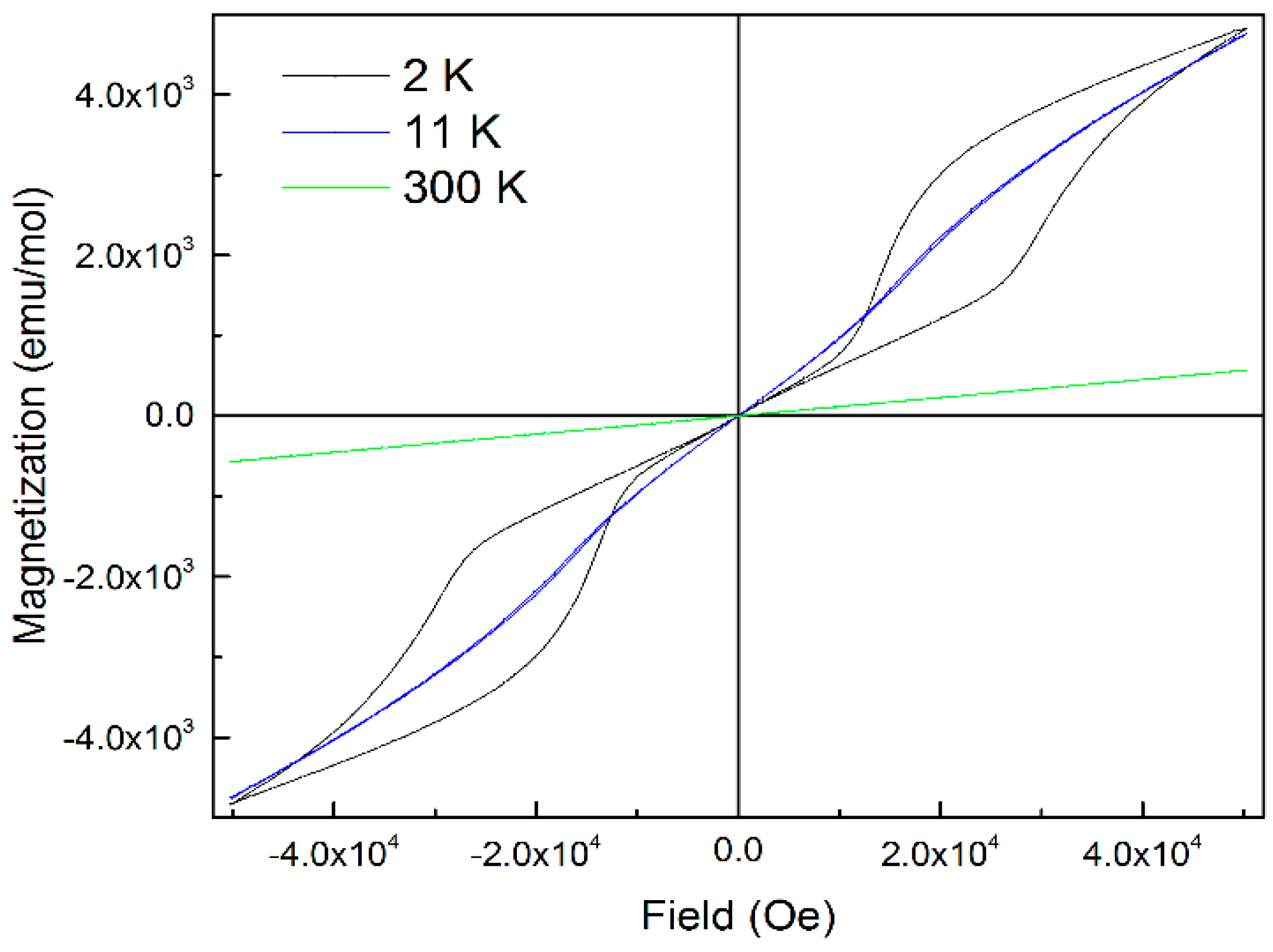
2.5. Magnetic Properties
3. Materials and Methods
3.1. Solvothermal Synthesis
3.2. Polyol Synthesis
3.3. Structural, Physical and Chemical Properties Determination
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Whittingham, M.S. Electrical energy storage and intercalation chemistry. Science 1976, 192, 1126–1127. [Google Scholar] [CrossRef] [PubMed]
- Thackeray, M. Lithium-ion batteries: An unexpected conductor. Nat. Mater. 2002, 1, 81–82. [Google Scholar] [CrossRef] [PubMed]
- Tarascon, J.M.; Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 2001, 414, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Qian, D.; Wang, Z.; Meng, Y.S. Recent progress in cathode materials research for advanced lithium ion batteries. Mater. Sci. Eng. R Rep. 2012, 73, 51–65. [Google Scholar] [CrossRef]
- Padhi, A.K.; Nanjundaswamy, K.S.; Goodenough, J.B. Phospho-olivines as positive-electrode materials for rechargeable lithium batteries. J. Electrochem. Soc. 1997, 144, 1188–1194. [Google Scholar] [CrossRef]
- Hautier, G.; Jain, A.; Ong, S.P.; Kang, B.; Moore, C.; Doe, R.; Ceder, G. Phosphates as lithium-ion battery cathodes: An evaluation based on high-throughput ab initio calculations. Chem. Mater. 2011, 23, 3495–3508. [Google Scholar] [CrossRef]
- Strobridge, F.C.; Clement, R.J.; Leskes, M.; Middlemiss, D.S.; Borkiewicz, O.J.; Wiaderek, K.M.; Chapman, K.W.; Chupas, P.J.; Grey, C.P. Identifying the structure of the intermediate, Li2/3CoPO4, formed during electrochemical cycling of LiCoPO4. Chem. Mater. 2014, 26, 6193–6205. [Google Scholar] [CrossRef] [PubMed]
- Sergio, B.; Stefania, P. Recent Advances in the Development of LiCoPO4 as High Voltage Cathode Material for Li-Ion Batteries. In Nanotechnology for Sustainable Energy; American Chemical Society: Washington, DC, USA, 2013; Volume 1140, pp. 67–99. [Google Scholar]
- Howard, W.F.; Spotnitz, R.M. Theoretical evaluation of high-energy lithium metal phosphate cathode materials in Li-ion batteries. J. Power Sources 2007, 165, 887–891. [Google Scholar] [CrossRef]
- Wolfenstine, J.; Allen, J.L.; Jow, T.R.; Thompson, T.; Sakamoto, J.; Jo, H.; Choe, H. LiCoPO4 mechanical properties evaluated by nanoindentation. Ceram. Int. 2014, 40, 13673–13677. [Google Scholar] [CrossRef]
- Chen, H.; Chen, M.; Du, C.; Cui, Y.; Zuo, P.; Cheng, X.; Yin, G. Synthesis and electrochemical performance of hierarchical nanocomposite of carbon coated LiCoPO4 crosslinked by graphene. Mater. Chem. Phys. 2016, 171, 6–10. [Google Scholar] [CrossRef]
- Theil, S.; Fleischhammer, M.; Axmann, P.; Wohlfahrt-Mehrens, M. Experimental investigations on the electrochemical and thermal behaviour of LiCoPO4-based cathode. J. Power Sources 2013, 222, 72–78. [Google Scholar] [CrossRef]
- Yamada, A.; Hosoya, M.; Chung, S.-C.; Kudo, Y.; Hinokuma, K.; Liu, K.-Y.; Nishi, Y. Olivine-type cathodes: Achievements and problems. J. Power Sources 2003, 119–121, 232–238. [Google Scholar] [CrossRef]
- Ellis, B.L.; Lee, K.T.; Nazar, L.F. Positive electrode materials for Li-ion and Li-batteries. Chem. Mater. 2010, 22, 691–714. [Google Scholar] [CrossRef]
- Truong, Q.D.; Devaraju, M.K.; Ganbe, Y.; Tomai, T.; Honma, I. Controlling the shape of LiCoPO4 nanocrystals by supercritical fluid process for enhanced energy storage properties. Sci. Rep. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D.; van der Ven, A.; Ceder, G. Li conductivity in LixMPO4 (M = Mn, Fe, Co, Ni) olivine materials. Electrochem. Solid State Lett. 2004, 7, A30–A32. [Google Scholar] [CrossRef]
- Tealdi, C.; Spreafico, C.; Mustarelli, P. Lithium diffusion in Li1−xFePO4: The effect of cationic disorder. J. Mater. Chem. 2012, 22, 24870–24876. [Google Scholar] [CrossRef]
- Jaehne, C.; Neef, C.; Koo, C.; Meyer, H.-P.; Klingeler, R. A new LiCoPO4 polymorph via low temperature synthesis. J. Mater. Chem. A 2013, 1, 2856–2862. [Google Scholar] [CrossRef]
- Amador, U.; Gallardo-Amores, J.M.; Heymann, G.; Huppertz, H.; Moran, E.; Arroyo, Y.D.D.M.E. High pressure polymorphs of LiCoPO4 and LiCoAsO4. Solid State Sci. 2009, 11, 343–348. [Google Scholar] [CrossRef]
- Garcia-Moreno, O.; Alvarez-Vega, M.; Garcia-Alvarado, F.; Garcia-Jaca, J.; Gallardo-Amores, J.M.; Sanjuan, M.L.; Amador, U. Influence of the structure on the electrochemical performance of lithium transition metal phosphates as cathodic materials in rechargeable lithium batteries: A new high-pressure form of LixMPO4 (M = Fe and Ni). Chem. Mater. 2001, 13, 2455. [Google Scholar] [CrossRef]
- Kreder, K.J.; Assat, G.; Manthiram, A. Microwave-assisted solvothermal synthesis of three polymorphs of LiCoPO4 and their electrochemical properties. Chem. Mater. 2015, 27, 5543–5549. [Google Scholar] [CrossRef]
- Assat, G.; Manthiram, A. Rapid microwave-assisted solvothermal synthesis of non-olivine Cmcm polymorphs of LiMPO4 (M = Mn, Fe, Co, and Ni) at low temperature and pressure. Inorg. Chem. 2015, 54, 10015–10022. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Zhao, M.; Song, X. Influence of the preparation conditions on the morphology and electrochemical performance of nano-sized Cu–Sn alloy anodes. J. Alloy. Compd. 2007, 439, 249–253. [Google Scholar] [CrossRef]
- Pan, Q.; Jin, H.; Wang, H.; Yin, G. Flower-like cuo film-electrode for lithium ion batteries and the effect of surface morphology on electrochemical performance. Electrochim. Acta 2007, 53, 951–956. [Google Scholar] [CrossRef]
- Tang, K.; Sun, J.; Yu, X.; Li, H.; Huang, X. Electrochemical performance of LiFePO4 thin films with different morphology and crystallinity. Electrochim. Acta 2009, 54, 6565–6569. [Google Scholar] [CrossRef]
- Dinh, H.-C.; Mho, S.-I.; Yeo, I.-H. Electrochemical analysis of conductive polymer-coated LiFePO4 nanocrystalline cathodes with controlled morphology. Electroanalysis 2011, 23, 2079–2086. [Google Scholar] [CrossRef]
- Ryu, W.-H.; Lim, S.-J.; Kim, W.-K.; Kwon, H. 3-D dumbbell-like LiNi1/3Mn1/3Co1/3O2 cathode materials assembled with nano-building blocks for lithium-ion batteries. J. Power Sources 2014, 257, 186–191. [Google Scholar] [CrossRef]
- Zhou, N.; Uchaker, E.; Wang, H.-Y.; Zhang, M.; Liu, S.-Q.; Liu, Y.-N.; Wu, X.; Cao, G.; Li, H. Additive-free solvothermal synthesis of hierarchical flower-like LiFePO4/C mesocrystal and its electrochemical performance. RSC Adv. 2013, 3, 19366–19374. [Google Scholar] [CrossRef]
- Cao, J.; Zhu, Y.; Bao, K.; Shi, L.; Liu, S.; Qian, Y. Microscale Mn2O3 hollow structures: Sphere, cube, ellipsoid, dumbbell, and their phenol adsorption properties. J. Phys. Chem. C 2009, 113, 17755–17760. [Google Scholar] [CrossRef]
- Zeng, G.; Caputo, R.; Carriazo, D.; Luo, L.; Niederberger, M. Tailoring two polymorphs of LiFePO4 by efficient microwave-assisted synthesis: A combined experimental and theoretical study. Chem. Mater. 2013, 25, 3399–3407. [Google Scholar] [CrossRef]
- Eftekhari, A. Surface modification of thin-film based LiCoPO4 5 V cathode with metal oxide. J. Electrochem. Soc. 2004, 151, A1456–A1460. [Google Scholar] [CrossRef]
- Choi, D.; Li, X.; Henderson, W.A.; Huang, Q.; Nune, S.K.; Lemmon, J.P.; Sprenkle, V.L. LiCoPO4 cathode from a CoHPO4·xH2O nanoplate precursor for high voltage Li-ion batteries. Heliyon 2016, 2, e00081. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, J.; Marino, C.; Haering, D.; Stinner, C.; Nordlund, D.; Doeff, M.M.; Gasteiger, H.A.; Nilges, T. Facile, ethylene glycol-promoted microwave-assisted solvothermal synthesis of high-performance LiCoPO4 as a high-voltage cathode material for lithium-ion batteries. RSC Adv. 2016, 6, 82984–82994. [Google Scholar] [CrossRef]
- Yang, H.; Wu, X.-L.; Cao, M.-H.; Guo, Y.-G. Solvothermal synthesis of LiFePO4 hierarchically dumbbell-like microstructures by nanoplate self-assembly and their application as a cathode material in lithium-ion batteries. J. Phys. Chem. C 2009, 113, 3345–3351. [Google Scholar] [CrossRef]
- Mathew, V.; Alfaruqi, M.H.; Gim, J.; Song, J.; Kim, S.; Ahn, D.; Kim, J. Morphology-controlled LiFePO4 cathodes by a simple polyol reaction for Li-ion batteries. Mater. Charact. 2014, 89, 93–101. [Google Scholar] [CrossRef]
- Ni, J.; Wang, H.; Gao, L.; Lu, L. A high-performance LiCoPO4/C core/shell composite for Li-ion batteries. Electrochim. Acta 2012, 70, 349–354. [Google Scholar] [CrossRef]
- Huang, X.; Ma, J.; Wu, P.; Hu, Y.; Dai, J.; Zhu, Z.; Chen, H.; Wang, H. Hydrothermal synthesis of LiCoPO4 cathode materials for rechargeable lithium ion batteries. Mater. Lett. 2005, 59, 578–582. [Google Scholar] [CrossRef]
- Nicholls, D. 41-Cobalt. In The Chemistry of Iron, Cobalt and Nickel; Pergamon Press: Oxford, UK, 1973; pp. 1053–1107. [Google Scholar]
- Szewczyk, A.; Gutowska, M.U.; Wieckowski, J.; Wisniewski, A.; Puzniak, R.; Diduszko, R.; Kharchenko, Y.; Kharchenko, M.F.; Schmid, H. Phase transitions in single-crystalline magnetoelectric LiCoPO4. Phys. Rev. B 2011, 84, 104419. [Google Scholar] [CrossRef]
- Tian, W.; Li, J.; Lynn, J.W.; Zarestky, J.L.; Vaknin, D. Spin dynamics in the magnetoelectric effect compound LiCoPO4. Phys. Rev. B 2008, 78, 6. [Google Scholar] [CrossRef]
- Rhee, C.H.; Kim, S.J.; Kim, C.S. Crystallographic and magnetic properties of LiCoPO4 at low temperature. J. Korean Phys. Soc. 2010, 56, 611–614. [Google Scholar]
- Petricek, V.; Dusek, M.; Palatinus, L. Crystallographic computing system Jana2006: General features. Z. Kristallogr. 2014, 229, 345–352. [Google Scholar]
- Finger, L.W.; Cox, D.E.; Jephcoat, A.P. A correction for powder diffraction peak asymmetry due to axial divergence. J. Appl. Crystallogr. 1994, 27, 892–900. [Google Scholar] [CrossRef]
- Cromer, D.T.; Liberman, D.A. Anomalous dispersion calculations near to and on the long-wavelength side of an absorption edge. Acta Crystallogr. Sect. A 1981, A37, 267–268. [Google Scholar] [CrossRef]
- Berar, J.F.; Lelann, P. E.S.D.’s and estimated probable error obtained in rietveld refinements with local correlations. J. Appl. Crystallogr. 1991, 24, 1–5. [Google Scholar] [CrossRef]
- Steger, E.; Herzog, K. Zum schwingungsspektrum der phosphorsäure. I. Infrarot- und raman-spektren von phosphatlösungen. Z. Anorg. Allg. Chem. 1964, 331, 169–182. [Google Scholar] [CrossRef]
- Jastrzębski, W.; Sitarz, M.; Rokita, M.; Bułat, K. Infrared spectroscopy of different phosphates structures. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2011, 79, 722–727. [Google Scholar] [CrossRef] [PubMed]
- Poovizhi, P.N.; Selladurai, S. Study of pristine and carbon-coated LiCoPO4 olivine material synthesized by modified sol-gel method. Ionics 2011, 17, 13–19. [Google Scholar] [CrossRef]
- Burba, C.M.; Frech, R. Vibrational spectroscopic investigation of structurally-related LiFePO4, NaFePO4, and FePO4 compounds. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2006, 65, 44–50. [Google Scholar] [CrossRef] [PubMed]





| Element | (a) LCP(ST) | (b) LCP(PO) | Theoretical |
|---|---|---|---|
| C (wt %) | 0.7(3) | 0.8(3) | 0 |
| H (wt %) | 0.2(3) | 0.4(3) | 0 |
| Li (wt %) | 4.2(2) | 4.1(2) | 4.3 |
| Co (wt %) | 35(1) | 34(1) | 36.6 |
| P (wt %) | 18.9(3) | 19.6(3) | 19.3 |
| n(Li):n(P) | 0.99(5) | 0.93(5) | 1 |
| n(Co):n(P) | 0.97(3) | 0.91(3) | 1 |
| Variable | 1 kOe | 10 kOe | 25 kOe |
|---|---|---|---|
| TN (K) | 12.7 | 12.7 | 9.1 |
| (K) | −27.6 | −27.7 | −26.2 |
| (cm3/mol) | 1.35 × 10−1 | 1.73 × 10−1 | 1.48 × 10−1 |
| (µB) | 5.46 | 5.42 | 5.56 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alarcón-Suesca, C.; Ludwig, J.; Hlukhyy, V.; Stinner, C.; Nilges, T. In Situ Studies and Magnetic Properties of the Cmcm Polymorph of LiCoPO4 with a Hierarchical Dumbbell-Like Morphology Synthesized by Easy Single-Step Polyol Synthesis. Inorganics 2016, 4, 35. https://doi.org/10.3390/inorganics4040035
Alarcón-Suesca C, Ludwig J, Hlukhyy V, Stinner C, Nilges T. In Situ Studies and Magnetic Properties of the Cmcm Polymorph of LiCoPO4 with a Hierarchical Dumbbell-Like Morphology Synthesized by Easy Single-Step Polyol Synthesis. Inorganics. 2016; 4(4):35. https://doi.org/10.3390/inorganics4040035
Chicago/Turabian StyleAlarcón-Suesca, Carlos, Jennifer Ludwig, Viktor Hlukhyy, Christoph Stinner, and Tom Nilges. 2016. "In Situ Studies and Magnetic Properties of the Cmcm Polymorph of LiCoPO4 with a Hierarchical Dumbbell-Like Morphology Synthesized by Easy Single-Step Polyol Synthesis" Inorganics 4, no. 4: 35. https://doi.org/10.3390/inorganics4040035
APA StyleAlarcón-Suesca, C., Ludwig, J., Hlukhyy, V., Stinner, C., & Nilges, T. (2016). In Situ Studies and Magnetic Properties of the Cmcm Polymorph of LiCoPO4 with a Hierarchical Dumbbell-Like Morphology Synthesized by Easy Single-Step Polyol Synthesis. Inorganics, 4(4), 35. https://doi.org/10.3390/inorganics4040035