Synthesis and Structure Determination of the Quaternary Zinc Nitride Halides Zn2NX1−yX′y (X, X′ = Cl, Br, I; 0 < y < 1)
Abstract
:1. Introduction
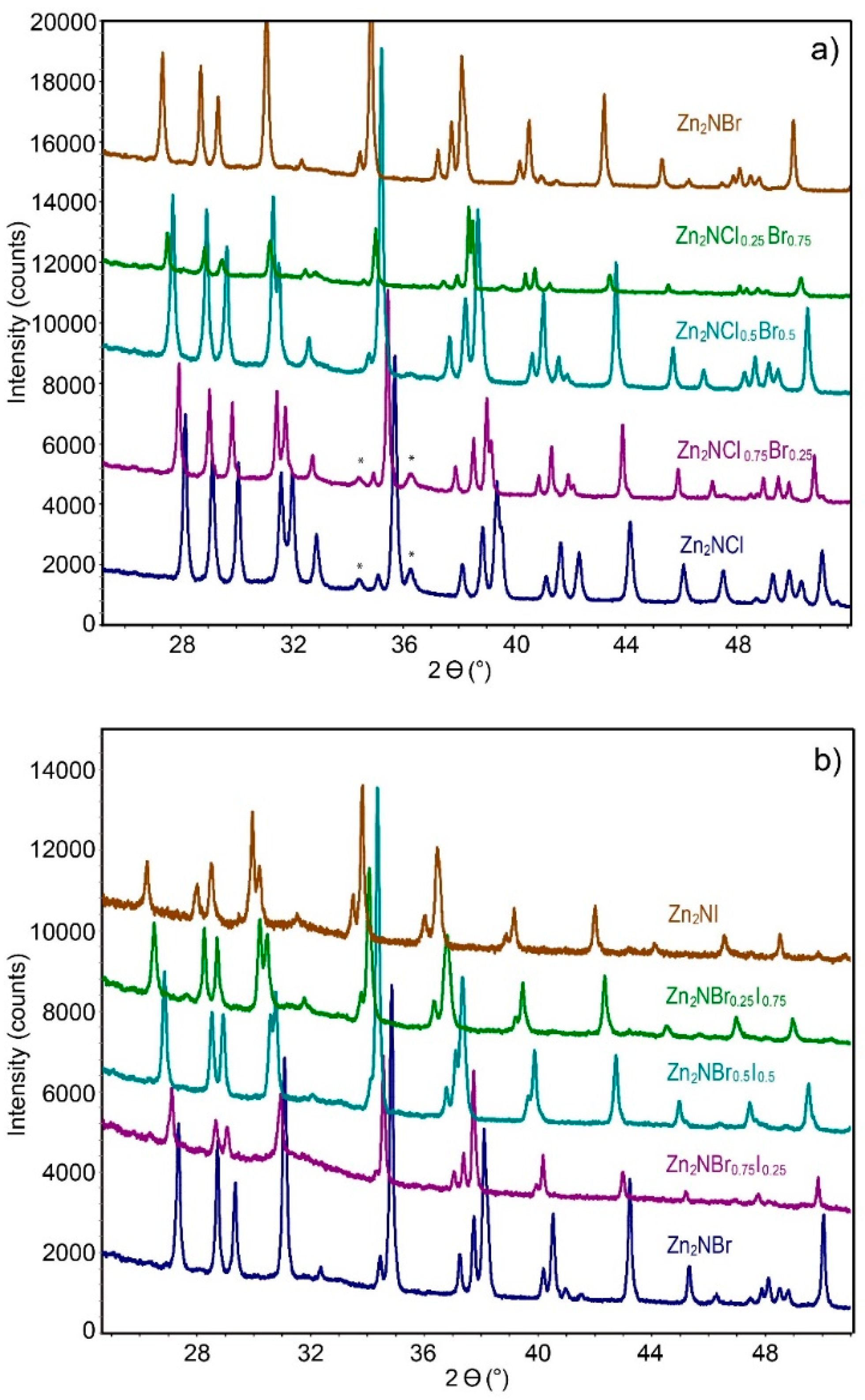
2. Result and Discussion
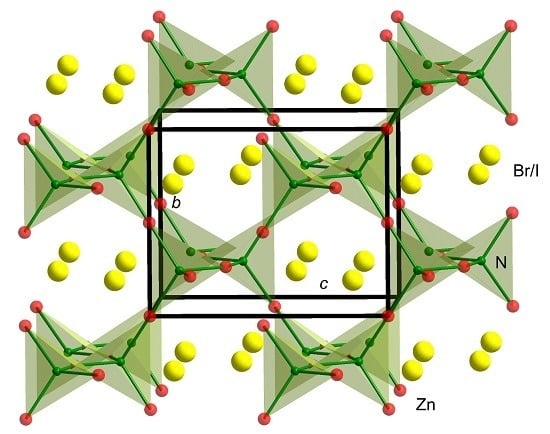
2.1. Crystal Structure
2.1.1. Crystal Structure of Zn2NCl0.47Br0.53
2.1.2. Crystal Structure of Zn2NBr0.62I0.38
2.2. Structure Discussion of Zn2NX1−yX′y (X, X′ = Cl, Br, I; 0 < y < 1)
3. Experimental
3.1. Synthesis of Zn2NX1−yX′y (X, X′ = Cl, Br, I; 0 < y < 1)
3.2. X-ray Crystallography
4. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Marx, R.; Mayer, H.-M. Preparation and Crystal Structure of Ordered and Disordered Lithium Nitride Dichloride, Li5NCl2. J. Solid State Chem. 1997, 130, 90–96. [Google Scholar] [CrossRef]
- Marx, R. Preparation and Crystal Structure of Lithium Nitride Chloride Li4NCl. J. Solid State Chem. 1997, 128, 241–246. [Google Scholar] [CrossRef]
- Andersson, S. Magnesium nitride fluorides. J. Solid State Chem. 1970, 1, 306–309. [Google Scholar] [CrossRef]
- Brogan, M.A.; Hughes, R.W.; Smith, R.I.; Gregory, D.H. Structural studies of magnesium nitride fluorides by powder neutron diffraction. J. Solid State Chem. 2012, 185, 213–218. [Google Scholar] [CrossRef]
- Li, Y.; George, J.; Liu, X.; Dronskowski, R. Synthesis, Structure Determination and Electronic Structure of Magnesium Nitride Chloride, Mg2NCl. Z. Anorg. Allg. Chem. 2015, 641, 266–269. [Google Scholar] [CrossRef]
- Reckeweg, O.; DiSalvo, F.J. Alkaline earth metal nitride compounds with the composition M2NX (M = Ca, Sr, Ba; X = □, H, Cl or Br). Solid State Sci. 2002, 4, 575–584. [Google Scholar] [CrossRef]
- Bowman, A.; Smith, R.I.; Gregory, D.H. Ternary and quaternary layered nitride halides, Ca2N(X, X′) (X, X′ = Cl, Br, I): Evolution of structure with composition. J. Solid State Chem. 2005, 178, 1807–1817. [Google Scholar] [CrossRef]
- Bailey, A.S.; Hughes, R.W.; Hubberstey, P.; Ritter, C.; Smith, R.I.; Gregory, D.H. New Ternary and Quaternary Barium Nitride Halides; Synthesis and Crystal Chemistry. Inorg. Chem. 2011, 50, 9545–9553. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, P.; Linz, W.; Seifert, H.J. Nitridfluoride der schweren Erdalkalimetalle. Naturwissenschaften 1971, 58, 219–220. [Google Scholar] [CrossRef]
- Nicklow, R.A.; Wagner, T.R.; Raymond, C.C. Preparation and Single-Crystal Structure Analysis of Ca2NF. J. Solid State Chem. 2001, 160, 134–138. [Google Scholar] [CrossRef]
- Jack, D.R.; Zeller, M.; Wagner, T.R. Doubled-cubic Ca2NF. Acta Crystallogr. 2005, C61, i6–i8. [Google Scholar]
- Wagner, T.R. Preparation and single-crystal structure analysis of Sr2NF. J. Solid State Chem. 2002, 169, 13–18. [Google Scholar] [CrossRef]
- Seibel, H.; Wagner, T.R. Preparation and crystal structure of Ba2NF. J. Solid State Chem. 2004, 177, 2772–2776. [Google Scholar] [CrossRef]
- Hadenfeldt, C.; Herdejürgen, H. Darstellung und Kristallstruktur der Calciumnitridhalogenide Ca2NCl und Ca2NBr. Z. Anorg. Allg. Chem. 1987, 545, 177–183. [Google Scholar] [CrossRef]
- Hadenfeldt, C.; Herdejürgen, H. Darstellung und Kristallstruktur der Calciumpnictidiodide Ca2NI, Ca2PI und Ca2AsI. Z. Anorg. Allg. Chem. 1988, 558, 35–40. [Google Scholar] [CrossRef]
- Bowman, A.; Smith, R.I.; Gregory, D.H. Synthesis and structure of the ternary and quaternary strontium nitride halides, Sr2N(X, X′) (X, X′ = Cl, Br, I). J. Solid State Chem. 2006, 179, 130–139. [Google Scholar] [CrossRef]
- Ehrlich, G.M.; Badding, M.E.; Brese, N.E.; Trail, S.S.; DiSalvo, F.J. New cerium nitride chlorides: Ce6Cl12N2 and CeNCl. J. Alloys Compds. 1996, 235, 133–134. [Google Scholar] [CrossRef]
- Meyer, H.J.; Jones, N.L.; Corbett, J.D. A new yttrium sesquichloride nitride, β-Y2Cl3N, that is isostructural with the binary yttrium sesquichloride. Inorg. Chem. 1989, 28, 2635–2637. [Google Scholar] [CrossRef]
- Schwanitz-Schüller, U.; Simon, A. Synthese und Kristallstruktur von Gd2NCl3. Z. Naturforsch. 1985, 40b, 705–709. [Google Scholar]
- Schurz, C.M.; Schleid, T. Chains of trans-edge connected [ZM4] tetrahedra (Z = N and O) in the lanthanide nitride chlorides M2NCl3 and Na2M4ONCl9 (M = La–Nd). J. Alloys Compds. 2009, 485, 110–118. [Google Scholar] [CrossRef]
- Yamanaka, S.; Yasunaga, T.; Yamaguchi, K.; Tagawa, M. Structure and superconductivity of the intercalation compounds of TiNCl with pyridine and alkali metals as intercalants. J. Mater. Chem. 2009, 19, 2573–2582. [Google Scholar] [CrossRef]
- Istomin, S.Y.; Köhler, J.; Simon, A. Crystal structure of β-ZrNCl refined from X-ray powder diffraction data, electronic band structures of β-ZrNCl and superconducting LixZrNCl. Physica C 1999, 319, 219–228. [Google Scholar] [CrossRef]
- Chen, X.; Koiwasaki, T.; Yamanaka, S. High-Pressure Synthesis and Crystal Structures of β-MNCl (M = Zr and Hf). J. Solid State Chem. 2001, 159, 80–86. [Google Scholar] [CrossRef]
- Yamanaka, S.; Hotehama, K.-I.; Kawaji, H. Superconductivity at 25.5 K in electron-doped layered hafnium nitride. Nature 1998, 392, 580–582. [Google Scholar] [CrossRef]
- Weiser, H.B. The Luminescence of the Iodide of Millon’s Base. J. Phys. Chem. 1917, 21, 37–47. [Google Scholar] [CrossRef]
- Lipscomb, W.N. The structure of mercuric amidochloride, HgNH2Cl. Acta Crystallogr. 1951, 4, 266–268. [Google Scholar] [CrossRef]
- Lipscomb, W.N. The structure of Millon’s base and its salts. Acta Crystallogr. 1951, 4, 156–158. [Google Scholar] [CrossRef]
- Nijssen, L.; Lipscomb, W.N. A hexagonal modification of a salt of Millon’s base. Acta Crystallogr. 1954, 7, 103–106. [Google Scholar] [CrossRef]
- Holleman, A.F.; Wiberg, E. Inorganic chemistry; Academic Press: San Diego, CA, USA, 2001. [Google Scholar]
- Liu, X.; Wessel, C.; Pan, F.; Dronskowski, R. Synthesis and single-crystal structure determination of the zinc nitride halides Zn2NX (X = Cl, Br, I). J. Solid State Chem. 2013, 203, 31–36. [Google Scholar] [CrossRef]
- Marchand, R.; Lang, J. Sur la préparation d'halogénonitrures de zinc. C. R. Hebd. Seances Acad. Sci. 1970, 270, 540–542. [Google Scholar]
- Grey, I.E.; Hill, R.J.; Hewat, A.W. A neutron powder diffraction study of the β to γ phase transformation in NaFeO2. Z. Kristallogr. 1990, 193, 51–69. [Google Scholar] [CrossRef]
- Partin, D.E.; Williams, D.J.; O’Keeffe, M. The Crystal Structures of Mg3N2 and Zn3N2. J. Solid State Chem. 1997, 132, 56–59. [Google Scholar] [CrossRef]
- Yakel, H.L.; Brynestad, J. Refinement of the crystal structure of orthorhombic zinc chloride. Inorg. Chem. 1978, 17, 3294–3296. [Google Scholar] [CrossRef]
- Chieh, C.; White, M.A. Crystal structure of anhydrous zinc bromide. Z. Kristallogr. 1984, 166, 189–197. [Google Scholar]
- Fourcroy, P.H.; Carre, D.; Rivet, J. Structure cristalline de l’iodure de zinc ZnI2. Acta Crystallogr. 1978, B34, 3160–3162. [Google Scholar] [CrossRef]
- Bruker APEX2, version 1.08; Bruker AXS Inc.: Madison, WI, USA, 2005.
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Carvajal, J. Recent advances in magnetic structure determination by neutron powder diffraction. Physica B 1993, 192, 55–69. [Google Scholar] [CrossRef]



| Formula | Zn2NCl0.47Br0.53 | Zn2NBr0.62I0.38 |
|---|---|---|
| Formula weight (g/mol) | 203.87 | 242.55 |
| Color and form | colorless block | colorless block |
| Temperature (K) | 293(2) | 293(2) |
| Crystal system | orthorhombic | orthorhombic |
| Space group; Z | Pna21 (No. 33); 4 | Pnma (No. 62); 4 |
| a (Å) | 6.168(2) | 6.249(4) |
| b (Å) | 7.538(3) | 6.164(4) |
| c (Å) | 6.026(2) | 7.824(5) |
| Cell volume (Å3) | 280.17(18) | 301.4(3) |
| Calculated density (g/cm3) | 4.833 | 5.345 |
| Crystal size (mm3) | 0.03 × 0.03 × 0.01 | 0.05 × 0.04 × 0.01 |
| θ range (deg) | 5.45–32.83 | 5.21–33.36 |
| Index ranges | −8 ≤ h ≤ 9 | −7 ≤ h ≤ 9 |
| −8 ≤ k ≤ 11 | −9 ≤ k ≤ 9 | |
| −8 ≤ l ≤ 9 | −12 ≤ l ≤ 6 | |
| Reflections collected | 2455 | 2215 |
| Indep. reflections, Rint | 824, 0.0366 | 585, 0.0326 |
| Restraints; parameters | 1; 38 | 0; 27 |
| Goodness-of-fit | 1.097 | 1.177 |
| R1[I > 2σ(I)], wR(I) | 0.0371, 0.0867 | 0.0428, 0.1147 |
| largest diff. peak; hole (e/Å3) | 1.136; −1.147 | 2.896; −2.869 |
| Lattice Parameter | Zn2NCl [30] | Zn2NCl0.47Br0.53 | Zn2NBr [30] | Zn2NBr0.62I0.38 | Zn2NI [30] |
|---|---|---|---|---|---|
| a | 6.1241(9) | 6.168(2) | 6.2149(9) | 6.249(4) | 6.3590(13) |
| b | 7.3885(11) | 7.538(3) | 7.6529(11) | 6.164(4) | 6.2592(12) |
| c | 5.9362(9) | 6.026(2) | 6.0859(8) | 7.824(5) | 7.9549(16) |
| V | 268.60(7) | 280.17(18) | 289.46(7) | 301.4(3) | 316.30(11) |
| Atom | Wyckoff Site | x | y | z | Ueq (Å2) | Occ (X, X′) |
|---|---|---|---|---|---|---|
| Zn2NCl0.47Br0.53 | ||||||
| Zn1 | 4a | 0.87415(12) | 0.82228(12) | 0.64994(16) | 0.0204(2) | |
| Zn2 | 4a | 0.97750(14) | 0.46071(15) | 0.47667(16) | 0.0244(2) | |
| N | 4a | 0.9284(7) | 0.3733(7) | 0.1907(10) | 0.0112(8) | |
| Cl | 4a | 0.8107(13) | 0.12201(12) | 0.68891(14) | 0.0157(3) | 0.467(8) |
| Br | 4a | - | - | - | - | 0.533(8) |
| Zn2NBr0.62I0.38 | ||||||
| Zn1 | 4c | 0.13822(12) | 3/4 | 0.31289(12) | 0.0222(3) | |
| Zn2 | 4a | 0 | 0 | 0 | 0.0318(4) | |
| N | 4c | 0.0644(8) | 1/4 | 0.8716(7) | 0.0108(9) | |
| I | 4c | 0.92075(8) | 3/4 | 0.61964(7) | 0.0173(3) | 0.380(12) |
| Br | 4c | - | - | - | - | 0.620(12) |
| Atom | U11 | U22 | U33 | U12 | U13 | U23 |
|---|---|---|---|---|---|---|
| Zn2NCl0.47Br0.53 | ||||||
| Zn1 | 0.0123(3) | 0.0198(4) | 0.0291(4) | 0.0049(3) | 0.0010(3) | 0.0006(3) |
| Zn2 | 0.0234(4) | 0.0287(5) | 0.0211(4) | 0.0012(4) | −0.0058(3) | −0.0120(4) |
| N | 0.0077(16) | 0.015(2) | 0.0108(19) | −0.0010(16) | 0.0012(16) | 0.0016(19) |
| Cl | 0.0152(4) | 0.0158(4) | 0.0162(4) | −0.0004(3) | 0.0004(3) | −0.0002(3) |
| Br | 0.0152(4) | 0.0158(4) | 0.0162(4) | −0.0004(3) | 0.0004(3) | −0.0002(3) |
| Zn2NBr0.62I0.38 | ||||||
| Zn1 | 0.0113(5) | 0.0327(5) | 0.0226(5) | 0 | −0.0053(3) | 0 |
| Zn2 | 0.0284(5) | 0.0290(5) | 0.0380(6) | −0.0137(4) | −0.0087(4) | 0.0218(4) |
| N | 0.0078(17) | 0.012(2) | 0.013(2) | 0 | 0.0002(15) | 0 |
| I | 0.0148(4) | 0.0170(4) | 0.0200(4) | 0 | 0.00094(17) | 0 |
| Br | 0.0148(4) | 0.0170(4) | 0.0200(4) | 0 | 0.00094(17) | 0 |
| Zn2NCl0.47Br0.53 | Distance/Angle | Zn2NBr0.62I0.38 | Distance/Angle |
|---|---|---|---|
| Zn1–N | 1.921(5) | Zn1–N | 1.914(5) |
| Zn1–N | 1.928(5) | Zn1–N | 1.920(5) |
| Zn1–Cl/Br | 2.6055(15) | Zn1–Br/I | 2.7579(17) |
| Zn1–Cl/Br | 2.8230(17) | Zn1–Br/I | 3.1487(19) |
| Zn2–N | 1.870(6) | Zn2–N | 1.883(3) |
| Zn2–N | 1.888(6) | Zn2–N | 1.883(3) |
| Zn2–Cl/Br | 2.8292(15) | Zn2–Br/I | 3.1881(15) |
| Zn2–Cl/Br | 2.9261(17) | Zn2–Br/I | 3.1881(15) |
| N–Zn1–N | 138.6(2) | N–Zn1–N | 145.14(16) |
| N–Zn2–N | 155.9(2) | N–Zn2–N | 180 |
| Zn2–N–Zn2 | 110.2(3) | Zn2–N–Zn2 | 109.8(3) |
| Zn2–N–Zn1 | 110.2(3) | Zn2–N–Zn1 | 109.60(17) |
| Zn2–N–Zn1 | 110.1(3) | Zn2–N–Zn1 | 109.60(17) |
| Zn2–N–Zn1 | 106.5(3) | Zn2–N–Zn1 | 105.08(17) |
| Zn2–N–Zn1 | 103.0(3) | Zn2–N–Zn1 | 105.08(17) |
| Zn1–N–Zn1 | 116.4(3) | Zn1–N–Zn1 | 117.4(3) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Liu, X.; Dronskowski, R. Synthesis and Structure Determination of the Quaternary Zinc Nitride Halides Zn2NX1−yX′y (X, X′ = Cl, Br, I; 0 < y < 1). Inorganics 2016, 4, 29. https://doi.org/10.3390/inorganics4040029
Li Y, Liu X, Dronskowski R. Synthesis and Structure Determination of the Quaternary Zinc Nitride Halides Zn2NX1−yX′y (X, X′ = Cl, Br, I; 0 < y < 1). Inorganics. 2016; 4(4):29. https://doi.org/10.3390/inorganics4040029
Chicago/Turabian StyleLi, Yanqing, Xiaohui Liu, and Richard Dronskowski. 2016. "Synthesis and Structure Determination of the Quaternary Zinc Nitride Halides Zn2NX1−yX′y (X, X′ = Cl, Br, I; 0 < y < 1)" Inorganics 4, no. 4: 29. https://doi.org/10.3390/inorganics4040029
APA StyleLi, Y., Liu, X., & Dronskowski, R. (2016). Synthesis and Structure Determination of the Quaternary Zinc Nitride Halides Zn2NX1−yX′y (X, X′ = Cl, Br, I; 0 < y < 1). Inorganics, 4(4), 29. https://doi.org/10.3390/inorganics4040029