Structural Classification of Quasi-One-Dimensional Ternary Nitrides
Abstract
:1. Introduction
2. Synthesis Routes for Nitrides
3. Crystal Structures of One-Dimensional Nitrides
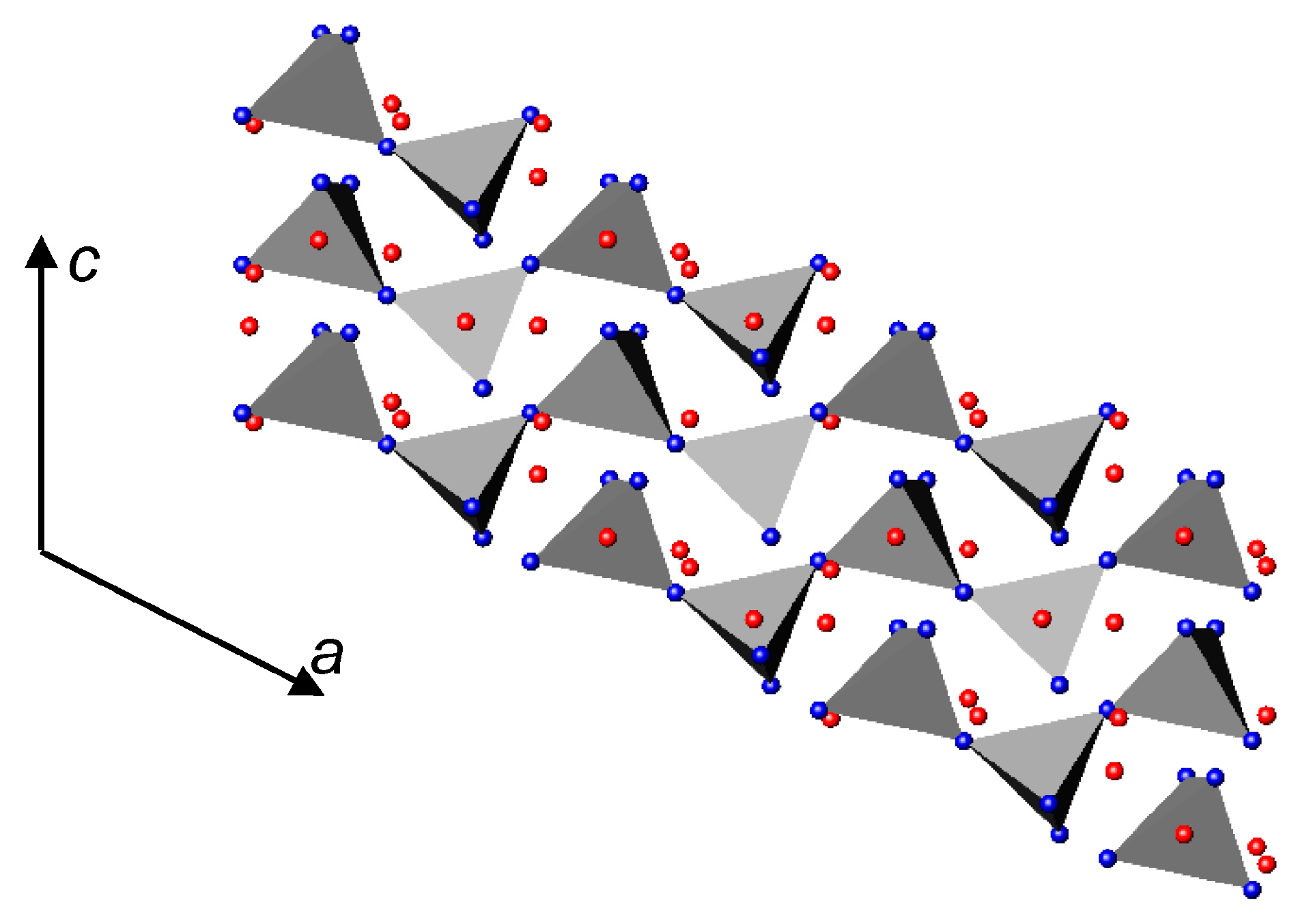
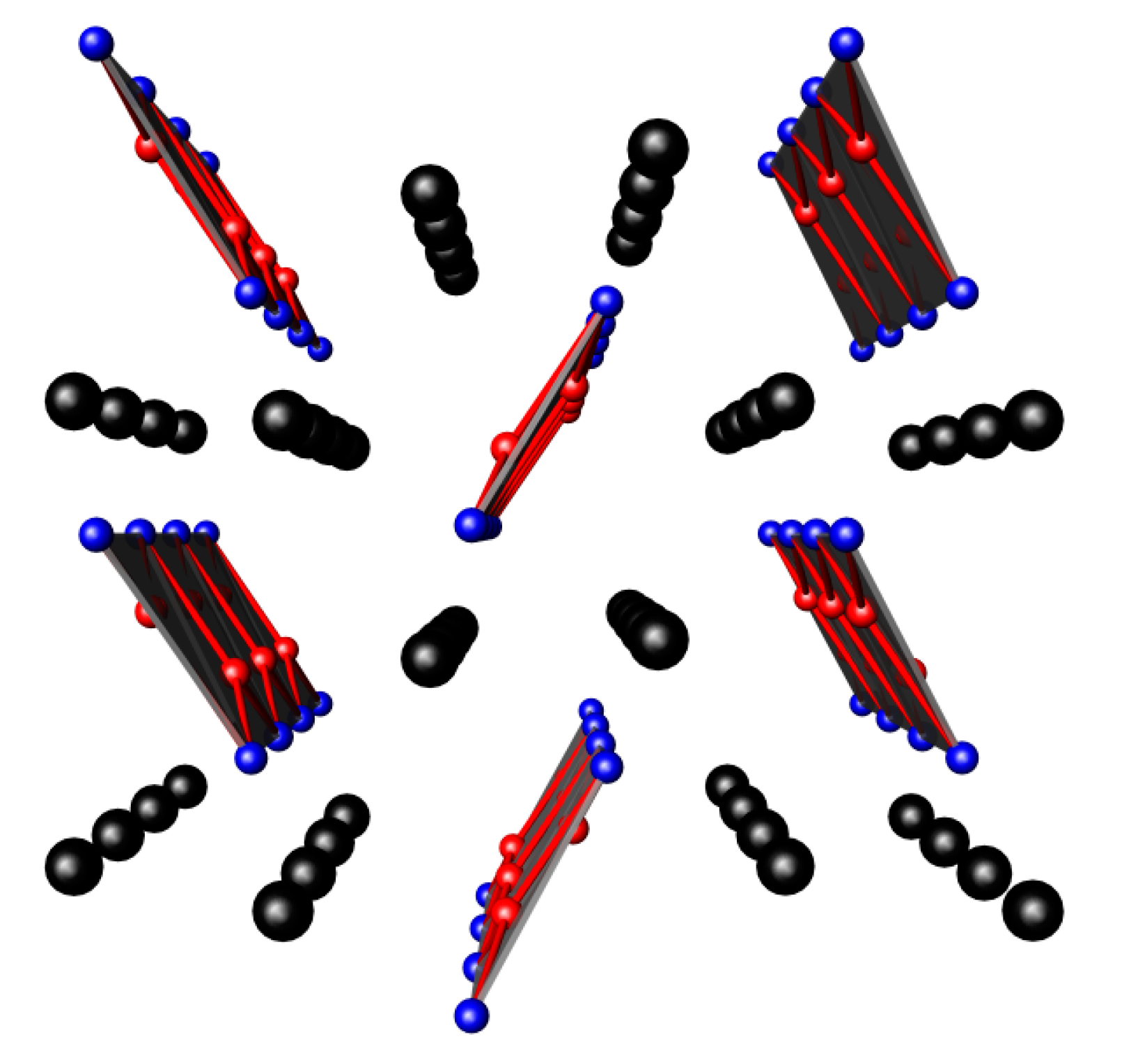
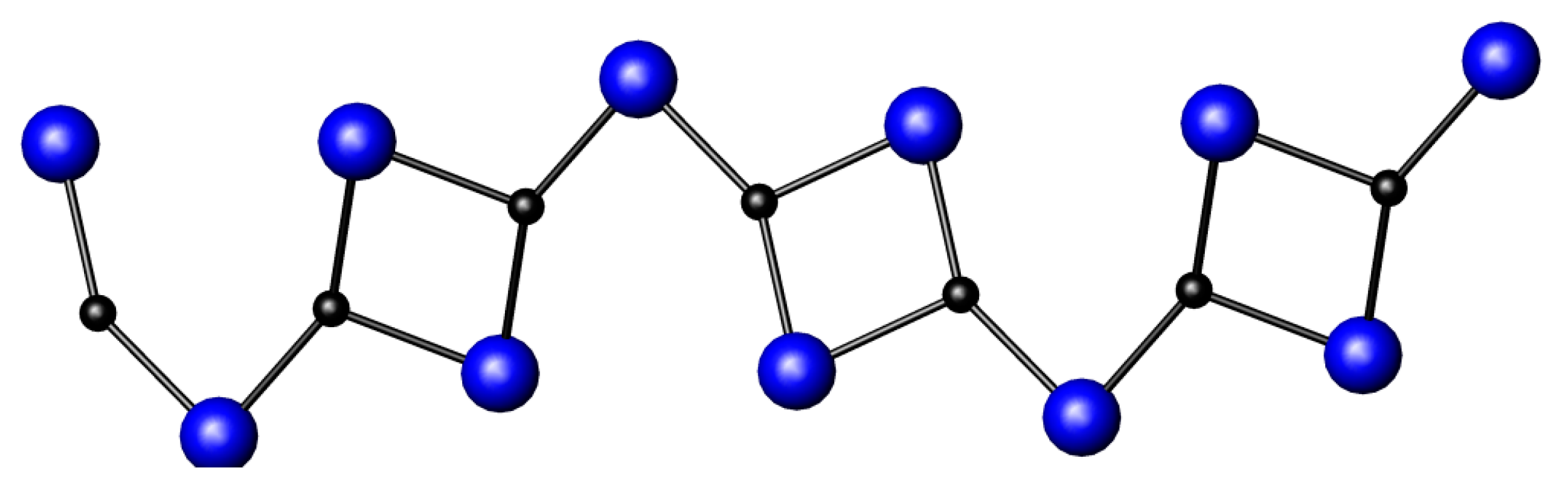
3.1. Nitrides Containing Infinite Chains of Octahedra
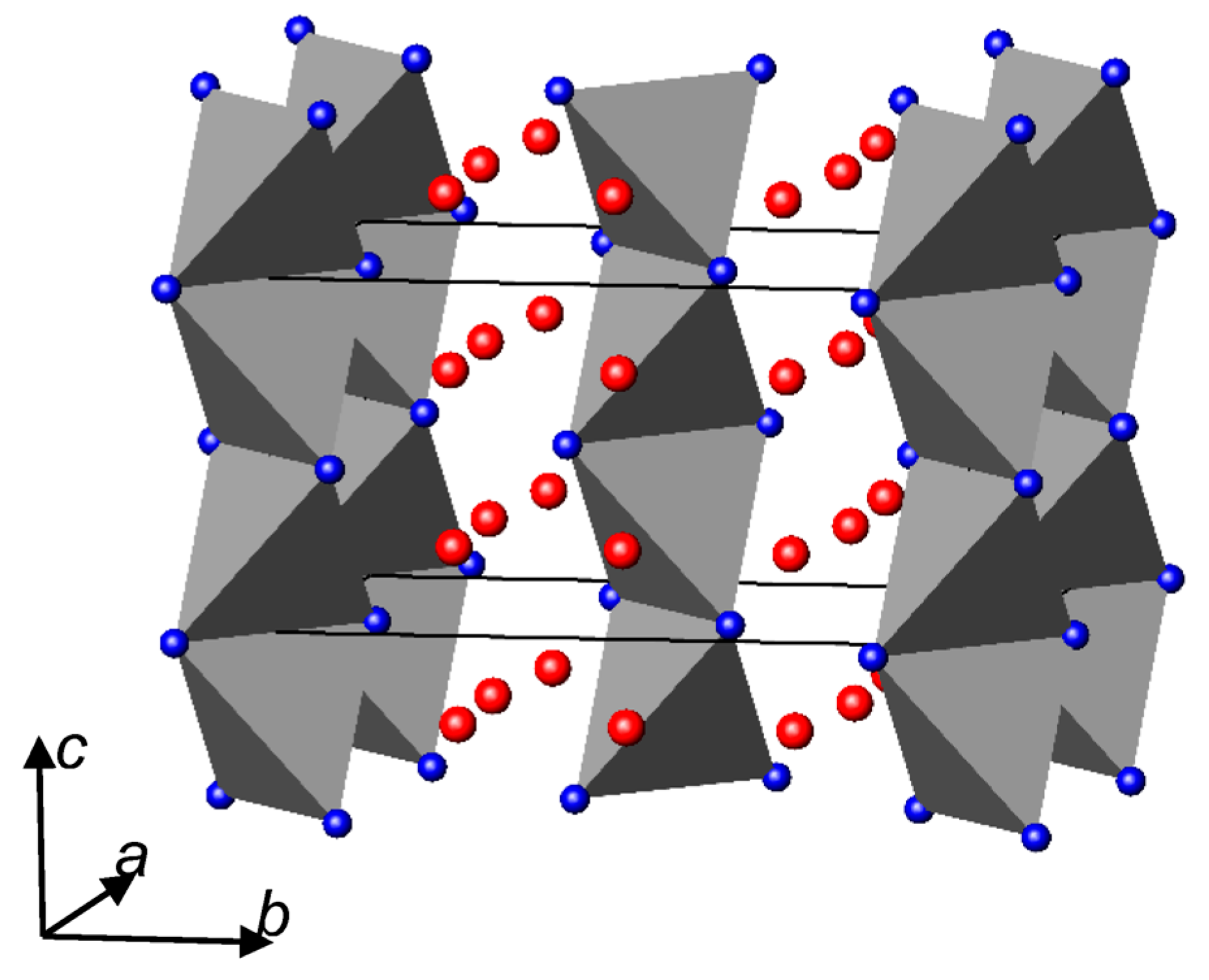
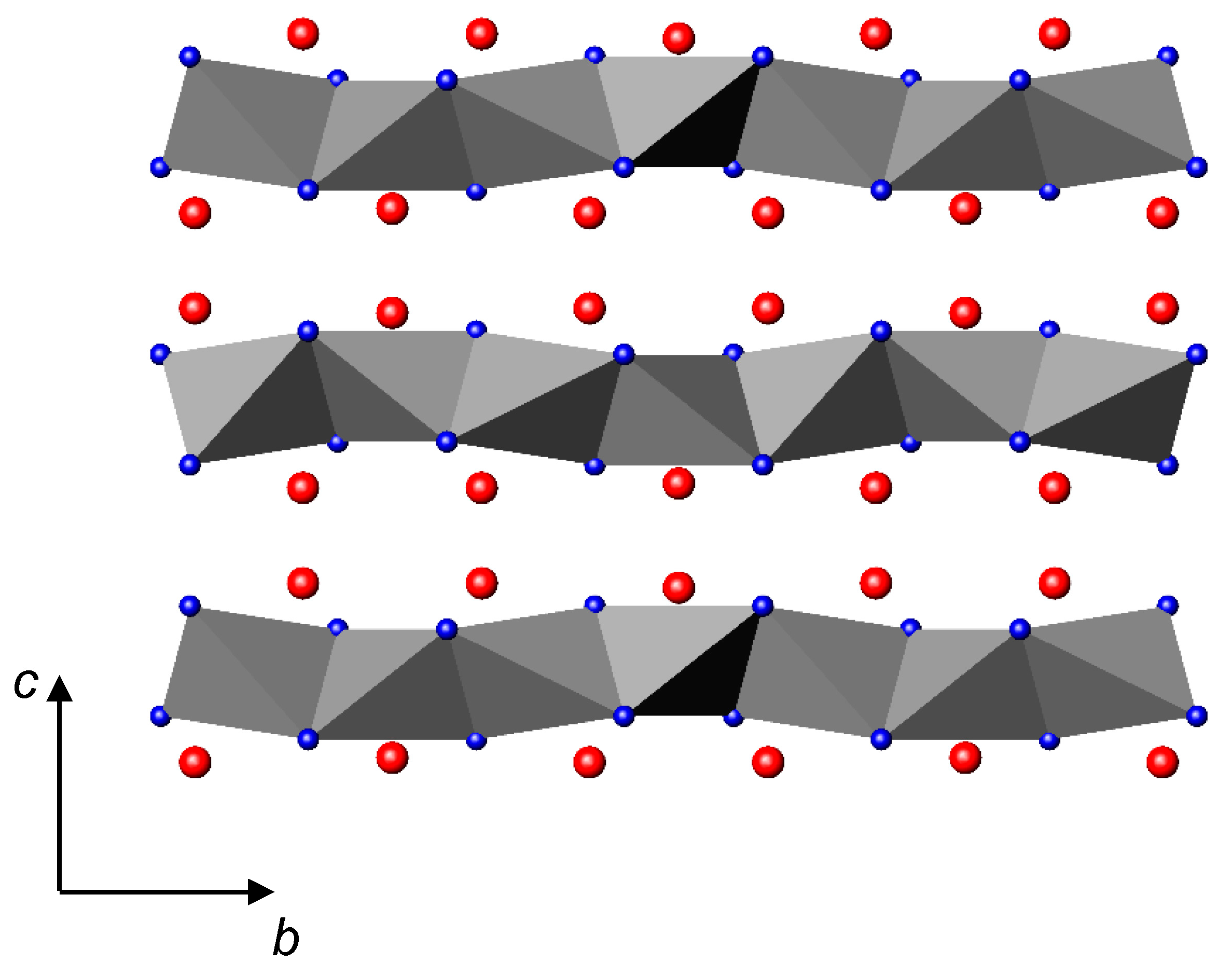
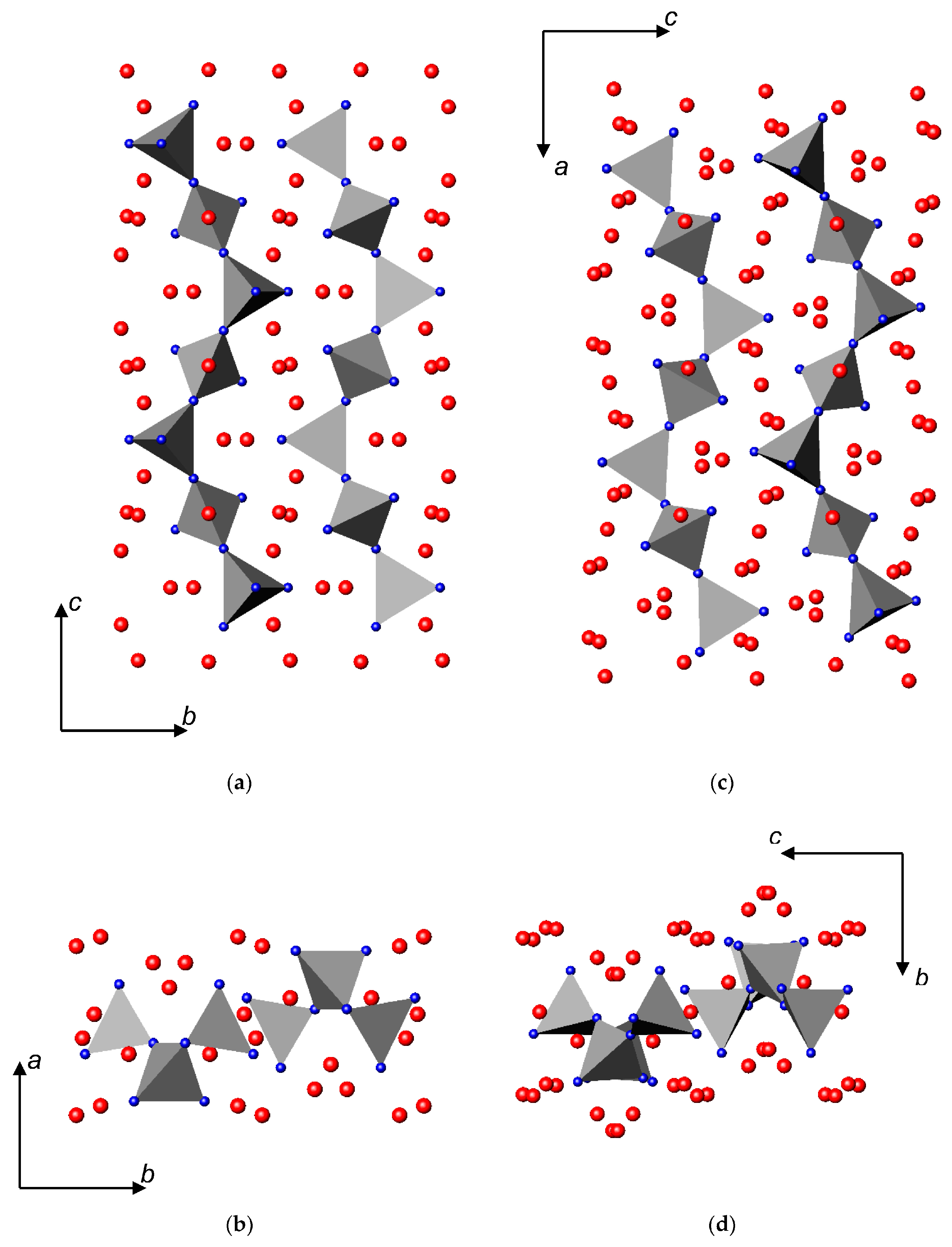
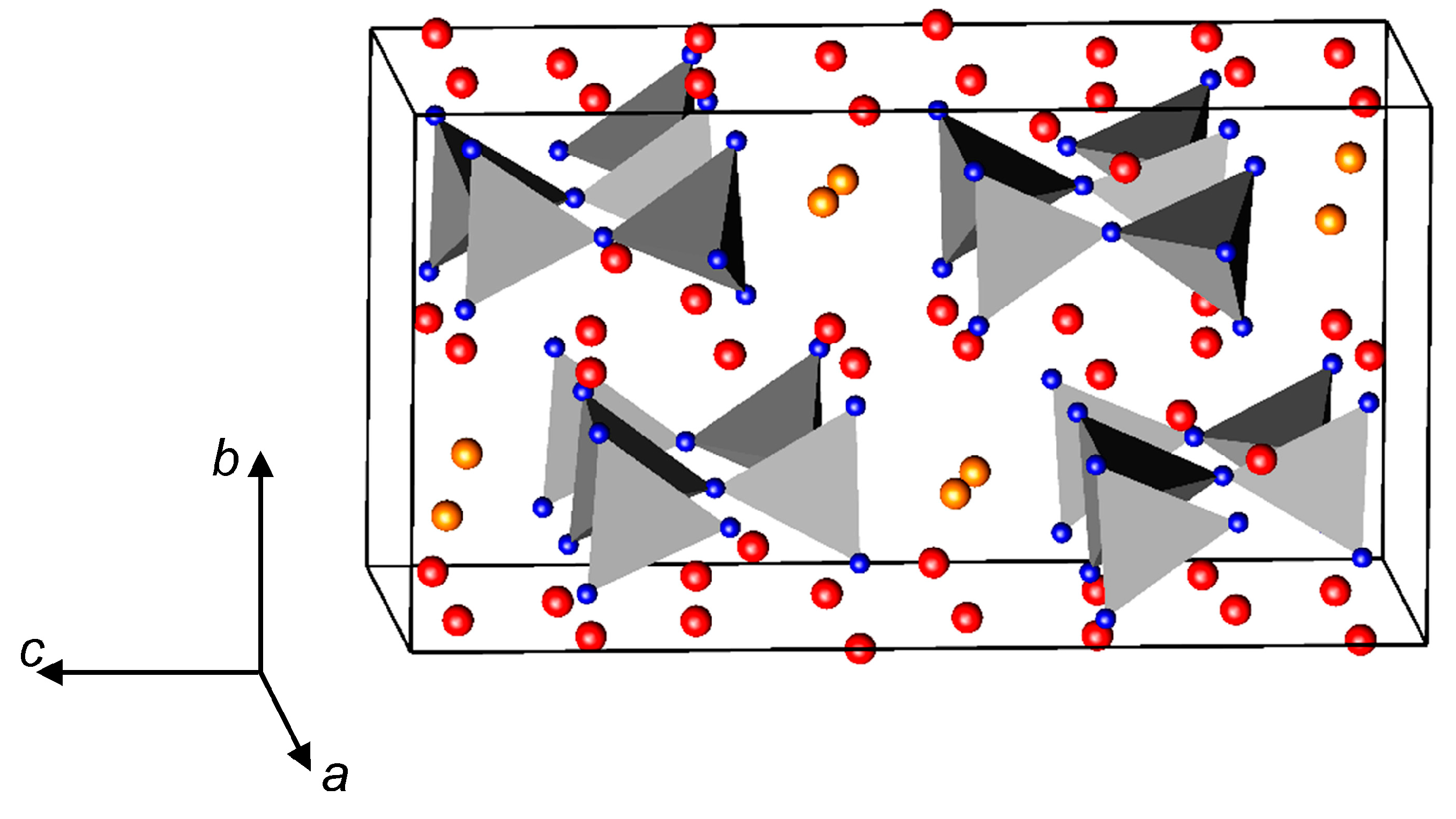
3.2. Nitrides Containing Infinite Chains of Tetrahedra
3.3. Nitrides Containing Infinite Chains of Square Planar Units
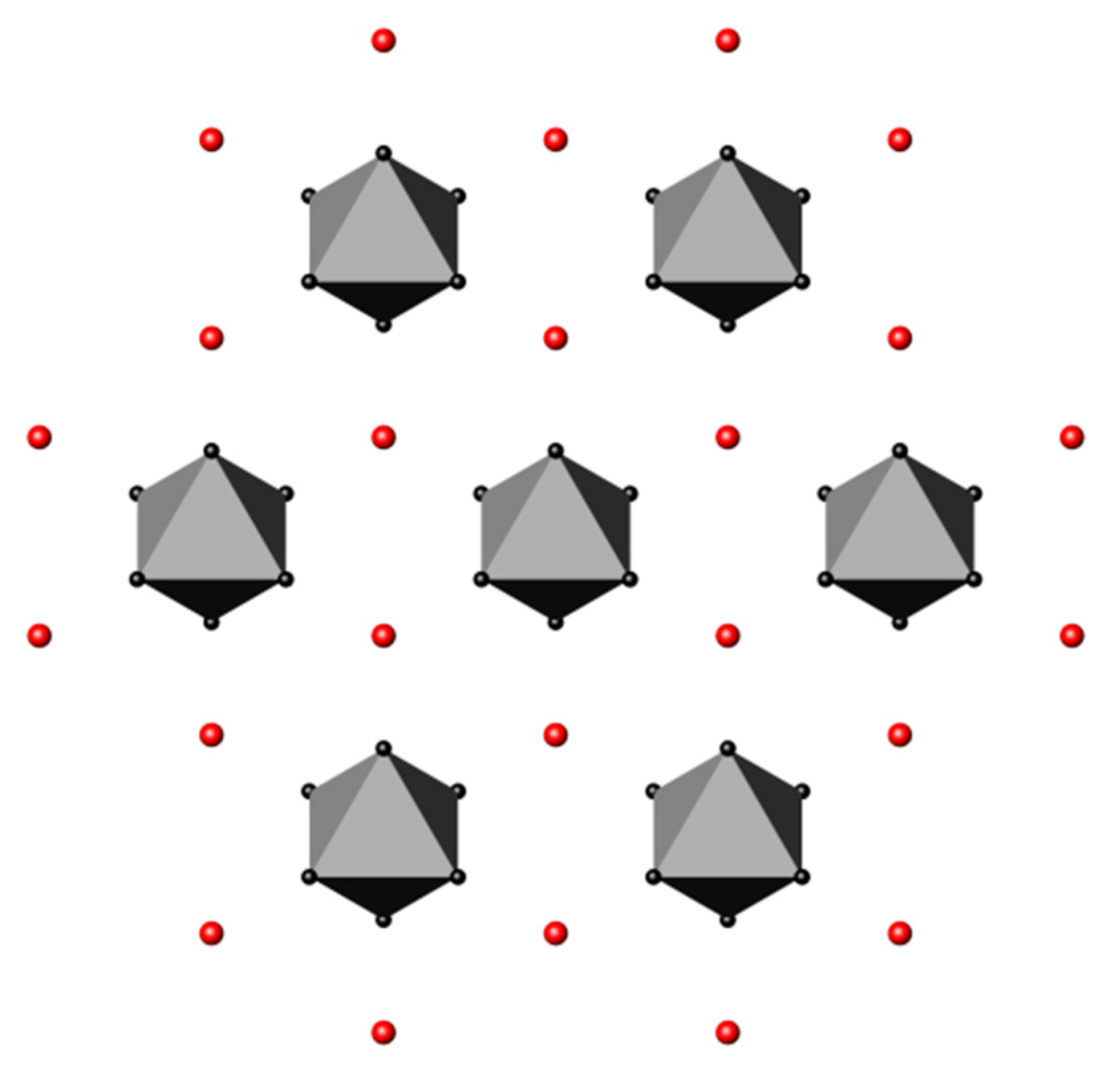
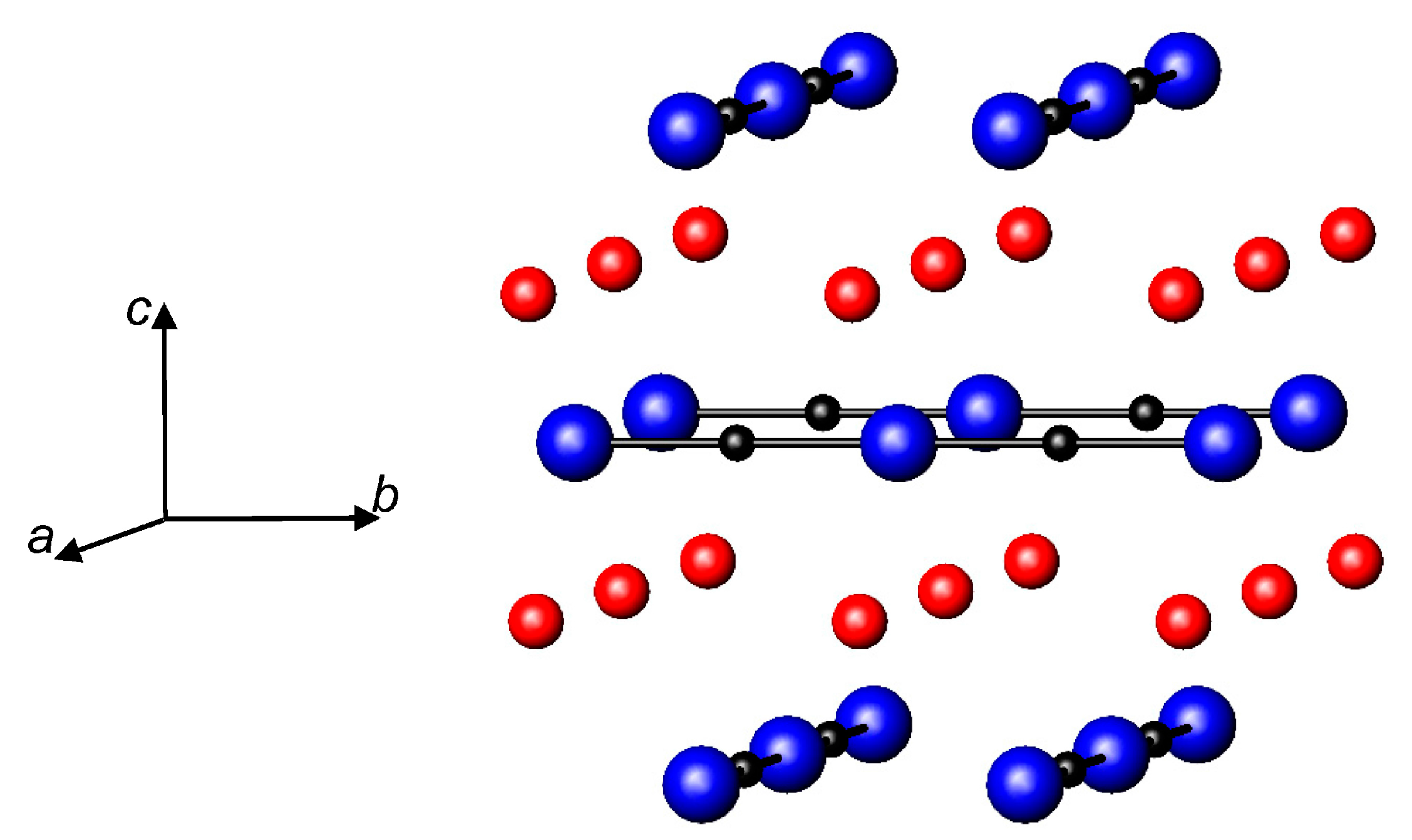
3.4. Nitrides Containing Infinite Chains of Triangular Units
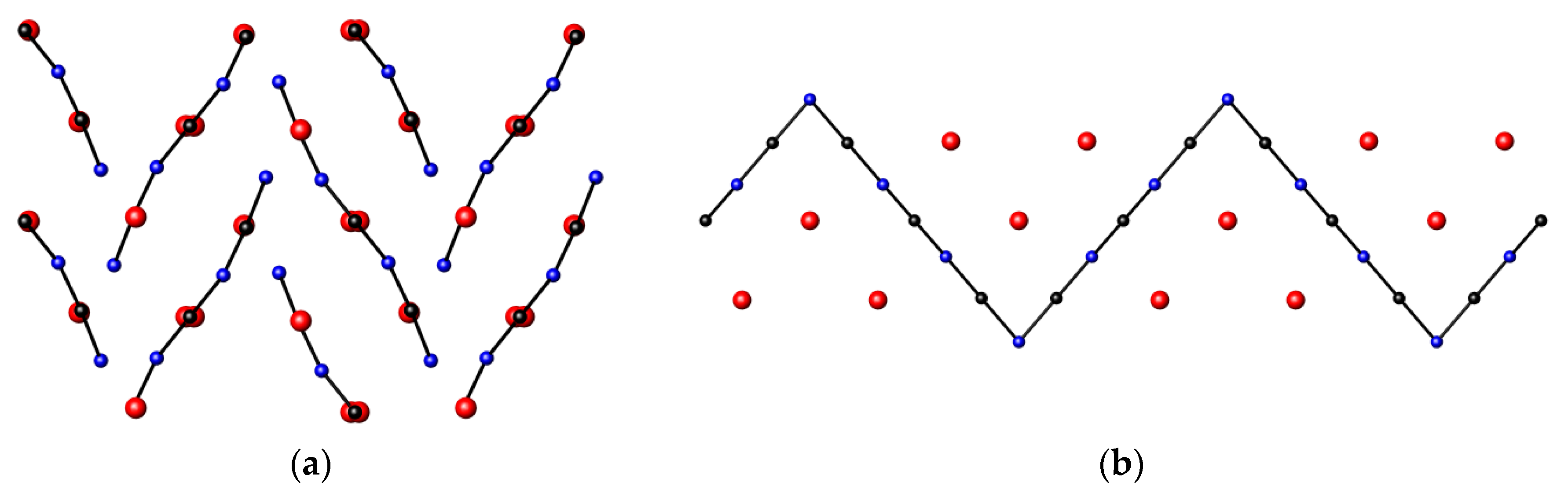
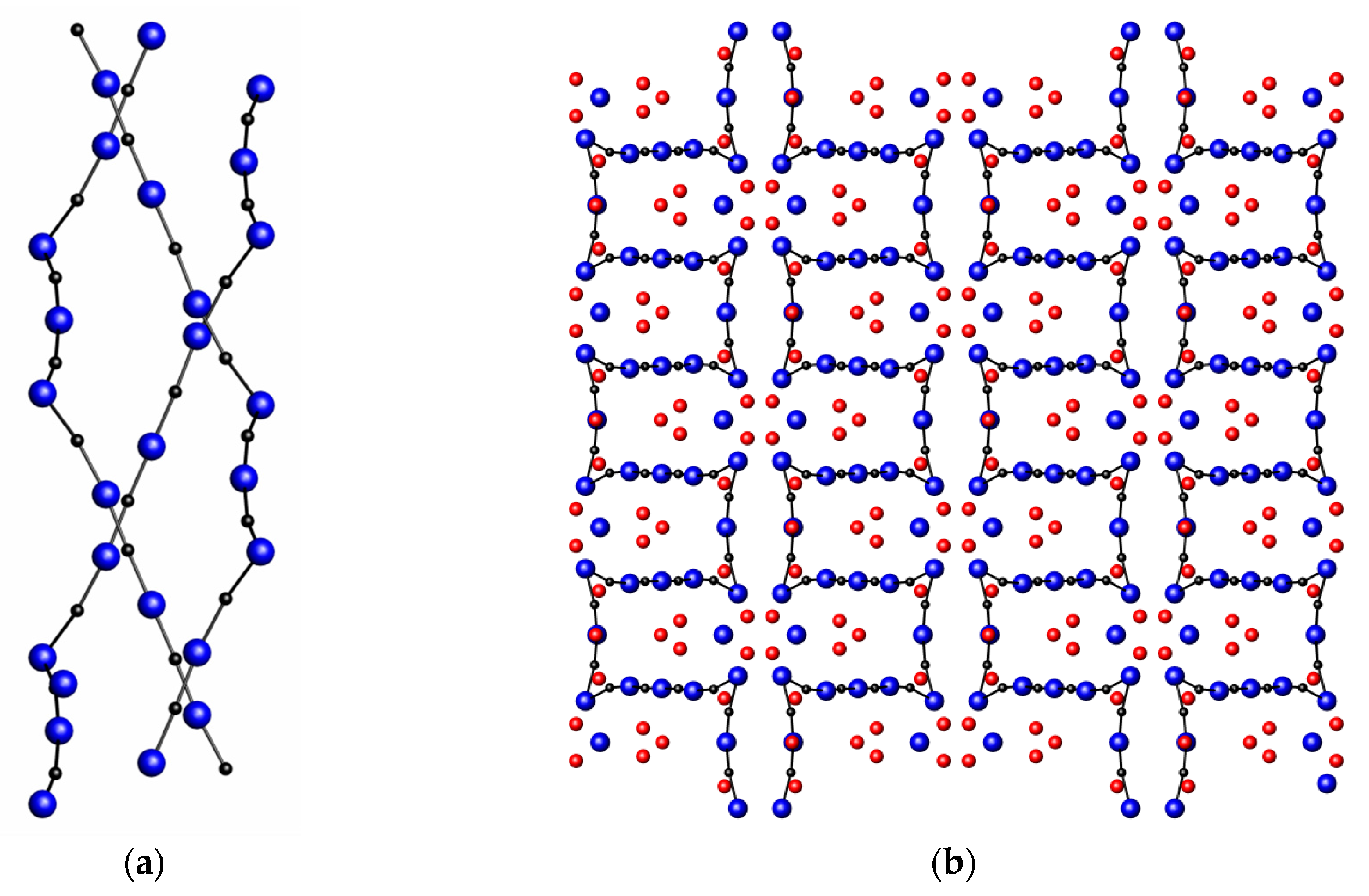
3.5. Nitrides Containing Infinite Linear Chains
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rao, C.N.R.; Cheetham, A.K.; Mahesh, R. Giant Magnetoresistance and Related Properties of Rare-Earth Manganates and Other Oxide Systems. Chem. Mater. 1996, 8, 2421–2432. [Google Scholar] [CrossRef]
- Rao, C.N.R.; Cheetham, A.K. Giant Magnetoresistance, Charge-Ordering and Related Aspects of Manganates and Other Oxide Systems. Adv. Mater. 1997, 9, 1009–1017. [Google Scholar] [CrossRef]
- Bednorz, J.G.; Müller, K.A. Possible High-Tc Superconductivity in the Ba–La–Cu–O System. Z. Phys. B Condens. Matter 1986, 64, 189–193. [Google Scholar] [CrossRef]
- Wu, M.K.; Ashburn, J.R.; Torng, C.J. Superconductivity at 93 K in a New Mixed-Phase Y–Ba–Cu–O Compound System at Ambient Pressure. Phys. Rev. Lett. 1987, 58, 908–910. [Google Scholar] [CrossRef] [PubMed]
- Titova, S.; Bryntse, I.; Irvine, J.; Mitchell, B.; Balakirev, V. Structural Anomalies of 1223 Hg(Tl)–Ba–Ca–Cu–O Superconductors in the Temperature Range 100–300K. J. Superconduct. 1998, 11, 471–479. [Google Scholar] [CrossRef]
- Smart, L.E.; Moore, E.A. Solid State Chemistry: An Introduction, 3rd ed.; CRC Taylor & Francis: London, UK, 2005. [Google Scholar]
- Ningthoujam, R.S.; Gajbhive, N.S. Synthesis, electron transport properties of transition metal nitrides and applications. Prog. Mat. Sci. 2015, 70, 50–154. [Google Scholar] [CrossRef]
- Kroschwitz, J.I.; Howe-Grant, M. (Eds.) Encyclopedia of Chemical Technology, 4th ed.; John Wiley & Sons: Chichester, UK, 1991–1998.
- Gregory, D.H. Structural Families in Nitride Chemistry. J. Chem. Soc. Dalton Trans. 1999, 259–270. [Google Scholar] [CrossRef]
- Rouxel, J. (Ed.) Chrystal Chemistry and Properties of Materials with Quasi-One-Dimensional Structures; Reidel: Boston, MA, USA, 1986.
- Lieber, C.M. One-dimensional nanostructures: Chemistry, physics & applications. Solid State Commun. 1998, 107, 606–616. [Google Scholar]
- Jiantao, H.U.; Odom, T.W.; Lieber, C.M. Chemistry and Physics in One Dimension: Synthesis and Properties of Nanowires and Nanotubes. Acc. Chem. Res. 1999, 32, 435–445. [Google Scholar]
- Shi, W.; Hughes, R.W.; Denholme, S.J.; Gregory, D.H. Synthesis design strategies to anisotropic chalcogenide nanostructures. Cryst. Eng. Commun. 2010, 12, 641–659. [Google Scholar] [CrossRef]
- Hahm, J.-I. Zinc Oxide Nanomaterials for Biomedical Fluorescence Detection. J. Nanosci. Nanotechnol. 2014, 14, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Sagawa, T.; Yoshikawa, S.; Imahori, H. One-Dimensional Nanostructured Semiconducting Materials for Organic Photovoltaics. J. Phys. Chem. Lett. 2010, 1, 1020–1025. [Google Scholar] [CrossRef]
- DiSalvo, F.J.; Clarke, S.J. Ternary Nitrides: A Rapidly Growing Class of New Materials. Curr. Opin. Solid State Mater. Sci. 1996, 1, 241–249. [Google Scholar] [CrossRef]
- Zur Loye, H.-C.; Houmes, J.D.; Bem, D.S. Recent developments in ternary nitride chemistry. In The Chemistry of Transition Metal Carbides and Nitrides; Oyama, S.T., Ed.; Blackie Academic & Professional: Glasgow, UK, 1996. [Google Scholar]
- Brese, N.E.; O’Keeffe, M. Crystal-Chemistry of Inorganic Nitrides. Struct. Bond. 1992, 79, 307–378. [Google Scholar]
- Niewa, R.; Jacobs, H. Group V and VI Alkali Nitridometalates: A Growing Class of Compounds With Structures Related to Silicate Chemistry. Chem. Rev. 1996, 96, 2053–2062. [Google Scholar] [CrossRef] [PubMed]
- Emeléus, H.J.; Sharpe, A.G. (Eds.) Advances in Inorganic Chemistry and Radiochemistry, 9th ed.; Academic Press: London, UK, 1966.
- Gregory, D.H. Nitride Chemistry of the s-block Elements. Coord. Chem. Rev. 2001, 215, 301–345. [Google Scholar] [CrossRef]
- Weast, R.C. (Ed.) Handbook of Chemistry and Physics, 51st ed.; The Chemical Rubber Company: Cleveland, OH, USA, 1970.
- Juza, R.; Langer, K.; von Benda, K. Ternary Nitrides, Phosphides, and Arsenides of Lithium. Angew. Chem. Int. Ed. Engl. 1968, 7, 360–370. [Google Scholar] [CrossRef]
- Addison, C.C.; Pulham, R.J.; Trevillion, E.A. Reaction between Barium and Nitrogen in Liquid Sodium—Solubility Studies. J. Chem. Soc. Dalton Trans. 1975, 2082–2085. [Google Scholar] [CrossRef]
- Brese, N.E.; O’Keeffe, M. Synthesis, Crystal-Structure and Physical-Properties of Sr2N. J. Solid State Chem. 1990, 87, 134–140. [Google Scholar] [CrossRef]
- Vajenine, G.V.; Grzechnik, A.; Syassen, K.; Loa, I.; Hanfland, M.; Simon, A. Interplay of Metallic and Ionic Bonding in Layered Subnitrides AE2N (AE = Ca, Sr or Ba) under High Pressure. C. R. Chim. 2005, 8, 1897–1905. [Google Scholar] [CrossRef]
- Beilby, G.T.; Henderson, G.G. The Action of Ammonia on Metals at High Temperature. J. Chem. Soc. Trans. 1901, 1245–1265. [Google Scholar] [CrossRef]
- Gomathi, A. Ternary Metal Nitrides by the Urea Route. Mater. Res. Bull. 2007, 42, 870–874. [Google Scholar] [CrossRef]
- Zhao, H.; Lei, M.; Chen, X.; Tang, W. Facile Route to Metal Nitrides through Melamine and Metal Oxides. J. Mater. Chem. 2006, 16, 4407–4412. [Google Scholar] [CrossRef]
- Weil, K.S.; Kumta, P.N. Synthesis of Ternary Transition Metal Nitrides Using Chemically Complexed Precursors. Mater. Sci. Eng. B 1996, 38, 109–117. [Google Scholar] [CrossRef]
- Herle, P.S.; Hegde, M.S.; Sooryanarayana, K.; Row, T.N.G.; Subbanna, G.N. Ni2Mo3N: A New Ternary Interstitial Nitride with a Filled Beta-Manganese Structure. Inorg. Chem. 1998, 37, 4128–4130. [Google Scholar] [CrossRef]
- Bem, D.S.; Gibson, C.P.; zur Loye, H.-C. Synthesis of Intermetallic Nitrides by Solid-State Pecursor Reduction. Chem. Mater. 1993, 5, 397–399. [Google Scholar] [CrossRef]
- Prior, T.J.; Battle, P.D. Facile Synthesis of Interstitial Metal Nitrides with the Filled Beta-Manganese Structure. J. Solid State Chem. 2003, 172, 138–147. [Google Scholar] [CrossRef]
- Prior, T.J.; Oldham, S.E.; Couper, V.J.; Battle, P.D. Ferromagnetic Nitrides with the Filled Beta-Mn Structure Fe2−xMxMo3N (M = Ni, Pd, Pt). Chem. Mater. 2005, 17, 1867–1873. [Google Scholar] [CrossRef]
- Prior, T.J.; Battle, P.D. Superparamagnetism and Metal-Site Ordering in Quaternary Nitrides with the η-Carbide Structure. J. Mater. Chem. 2004, 14, 3001–3007. [Google Scholar] [CrossRef]
- Houmes, J.D.; zur Loye, H.-C. Plasma Nitridation of Metal Oxides. Chem. Mater. 1996, 8, 2551–2553. [Google Scholar] [CrossRef]
- Kroke, E. High-Pressure Synthesis of Novel Binary Nitrogen Compounds of Main Group Elements. Angew. Chem. Int. Ed. Engl. 2002, 41, 77–82. [Google Scholar] [CrossRef]
- Niewa, R.; DiSalvo, F.J. Recent Developments in Nitride Chemistry. Chem. Mater. 1998, 10, 2733–2752. [Google Scholar] [CrossRef]
- Rauch, P.E.; Simon, A. The New Subnitride NaBa3N—An Extension of Alkali-Metal Suboxide Chemistry. Angew. Chem. Int. Ed. Engl. 1992, 31, 1519–1521. [Google Scholar] [CrossRef]
- Snyder, G.J.; Simon, A. The Infinite Chain Nitride Na5Ba3N—A One-Dimensional Void Metal. J. Am. Chem. Soc. 1995, 117, 1996–1999. [Google Scholar] [CrossRef] [PubMed]
- Simon, A. Group 1 and 2 Suboxides and Subnitrides—Metals with Atomic Size Holes and Tunnels. Coord. Chem. Rev. 1997, 163, 253–270. [Google Scholar] [CrossRef]
- Gudat, A.; Kniep, R.; Rabenau, A.; Bronger, W.; Ruschewitz, U. Li3FeN2, a Ternary Nitride with Chains—Crystal Structure and Magnetic Properties. J. Less Common Met. 1990, 161, 31–36. [Google Scholar] [CrossRef]
- Cordier, G.; Gudat, A.; Kniep, R.; Rabenau, A. LiCaN and Li4SrN2 Derivatives of the Fluorite and Lithium Nitride Structures. Angew. Chem. Int. Ed. Engl. 1989, 28, 1702–1703. [Google Scholar] [CrossRef]
- Shannon, R.D.; Prewitt, C.T. Effective Ionic Radii in Oxides and Fluorides. Acta Crystallogr. B 1969, 25, 925–946. [Google Scholar] [CrossRef]
- Baur, W.H. Effective Ionic Radii in Nitrides. Cryst. Rev. 1987, 1, 59–83. [Google Scholar] [CrossRef]
- Peters, J.; Krebs, B. Silicon Disulfide and Silicon Diselenide—A Reinvestigation. Acta Crystallogr. B 1982, 38, 1270–1272. [Google Scholar] [CrossRef]
- Makovicky, E. Crystal Structures of Sulfides and Other Chalcogenides. Rev. Miner. Geochem. 2006, 61, 7–125. [Google Scholar] [CrossRef]
- Yamane, H.; DiSalvo, F.J. Ba3Ga2N4. Acta Crystallogr. C 1996, 52, 760–761. [Google Scholar] [CrossRef]
- Blasé, W.; Cordier, G.; Ludwig, M.; Kniep, R. Sr3[Al2N4]: A Nitridoaluminate with Corrugated Tetrahedral Chains . Z. Naturforsch. B 1994, 49, 501. [Google Scholar]
- Clarke, S.J.; DiSalvo, F.J. Synthesis and Structure of One-, Two- and Three-Dimensional Alkaline Earth Metal Gallium Nitrides: Sr3Ga2N4, Ca3Ga2N4 and Sr3Ga3N5. Inorg. Chem. 1997, 36, 1143–1148. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, M.; Niewa, R.; Kniep, R. Dimers [Al2N6]12– and Chains in the Crystal Structures of Ca6[Al2N6] and Ba3[Al2N4]. Z. Naturforsch. B 1999, 54, 461–465. [Google Scholar] [CrossRef]
- Niewa, R.; Zherebtsov, D.A.; Borrmann, H.; Kniep, R. Preparation and Crystal Structure of Li4TaN3. Z. Anorg. Allg. Chem. 2002, 628, 2505–2508. [Google Scholar] [CrossRef]
- Jacobs, H.; Niewa, R. Synthesis and Crystal Structure of a Sodium Nitrido Tungstate (VI), Na3WN3. Eur. J. Solid State Inorg. Chem. 1994, 31, 105–113. [Google Scholar]
- Rauch, P.E.; DiSalvo, F.J.; Brese, N.E.; Partin, D.E.; O’Keeffe, M. Synthesis and Neutron Diffraction Study of Na3WN3 and Na3MoN3. J. Solid State Chem. 1994, 110, 162–166. [Google Scholar] [CrossRef]
- Niewa, R.; Jacobs, H. Na5Rb(WN3)2 and Na5Cs(WN3)2, Two New Sodium Nitrido Tungstates(VI) with the Heavier Alkali Metals Rubidium and Cesium. J. Alloys Comp. 1996, 234, 171–177. [Google Scholar] [CrossRef]
- Niewa, R.; Jacobs, H. Na2KWN3 and Na11Rb(WN3)4, Two New Alkali Metal Nitrido Tungstates(VI) with Closely Related Structures. J. Alloys Comp. 1996, 233, 61–68. [Google Scholar] [CrossRef]
- Bowman, A.; Gregory, D.H. Synthesis and Characterisation of the Ternary Nitride Sr2TaN3. J. Alloys Comp. 2003, 348, 80–87. [Google Scholar] [CrossRef]
- Zherebtsov, D.A.; Aksel’rud, L.G.; Niewa, R. Crystal Structure of Dicalcium Trinitrido Monovanadate(V), Ca2VN3. Z. Kristallogr. New Cryst. Struct. 2002, 217, 469. [Google Scholar]
- Seeger, O.; Hofmann, M.; Strähle, J.; Laval, J.P.; Frit, B. Synthesis and Structure of BaZrN2 and Ba2NbN3. Z. Anorg. Allg. Chem. 1994, 620, 2008–2013. [Google Scholar] [CrossRef]
- Benz, R.; Zachariasen, W.H. Crystal Structures of Th2CrN3, Th2MnN3, U2CrN3 and U2MnN3. J. Nucl. Mater. 1970, 37, 109–113. [Google Scholar] [CrossRef]
- Broll, S.; Jeitschko, W. The Ternary Rare-Earth Chromium Nitrides Ce2CrN3 and Ln3Cr10−xN11 with Ln = La, Ce, Pr. Z. Naturforsch. B 1995, 50, 905–912. [Google Scholar] [CrossRef]
- Niewa, R.; Vajenine, G.V.; DiSalvo, F.J.; Luo, H.; Yelon, W.B. Unusual Bonding in Ternary Nitrides: Preparation, Structure and Properties of Ce2MnN3. Z. Naturforsch. B 1998, 53, 63–74. [Google Scholar] [CrossRef]
- Landrum, G.A.; Dronskowski, R.; Niewa, R.; DiSalvo, F.J. Electronic Structure and Bonding in Cerium Nitride Compounds: Trivalent Versus Tetravalent Cerium. Chem. Eur. J. 1999, 5, 515–522. [Google Scholar] [CrossRef]
- Niewa, R.; Hu, Z.; Grazioli, C.; Rößler, U.; Golden, M.S.; Knupfer, M.; Fink, J.; Giefers, H.; Wortmann, G.; de Groot, F.M.F.; et al. XAS Spectra of Ce2[MnN3] at the Ce–M4,5, Ce–L3, Mn–L2,3 and N–K threshholds. J. Alloys Comp. 2002, 346, 129–133. [Google Scholar] [CrossRef]
- Höhn, P.; Haag, S.; Milius, W.; Kniep, R. Sr2LiFe2N3 and Ba2LiFe2N3—Nitridoferrate(II) Isotypes with Anions. Angew. Chem. Int. Ed. Engl. 1991, 30, 831–832. [Google Scholar] [CrossRef]
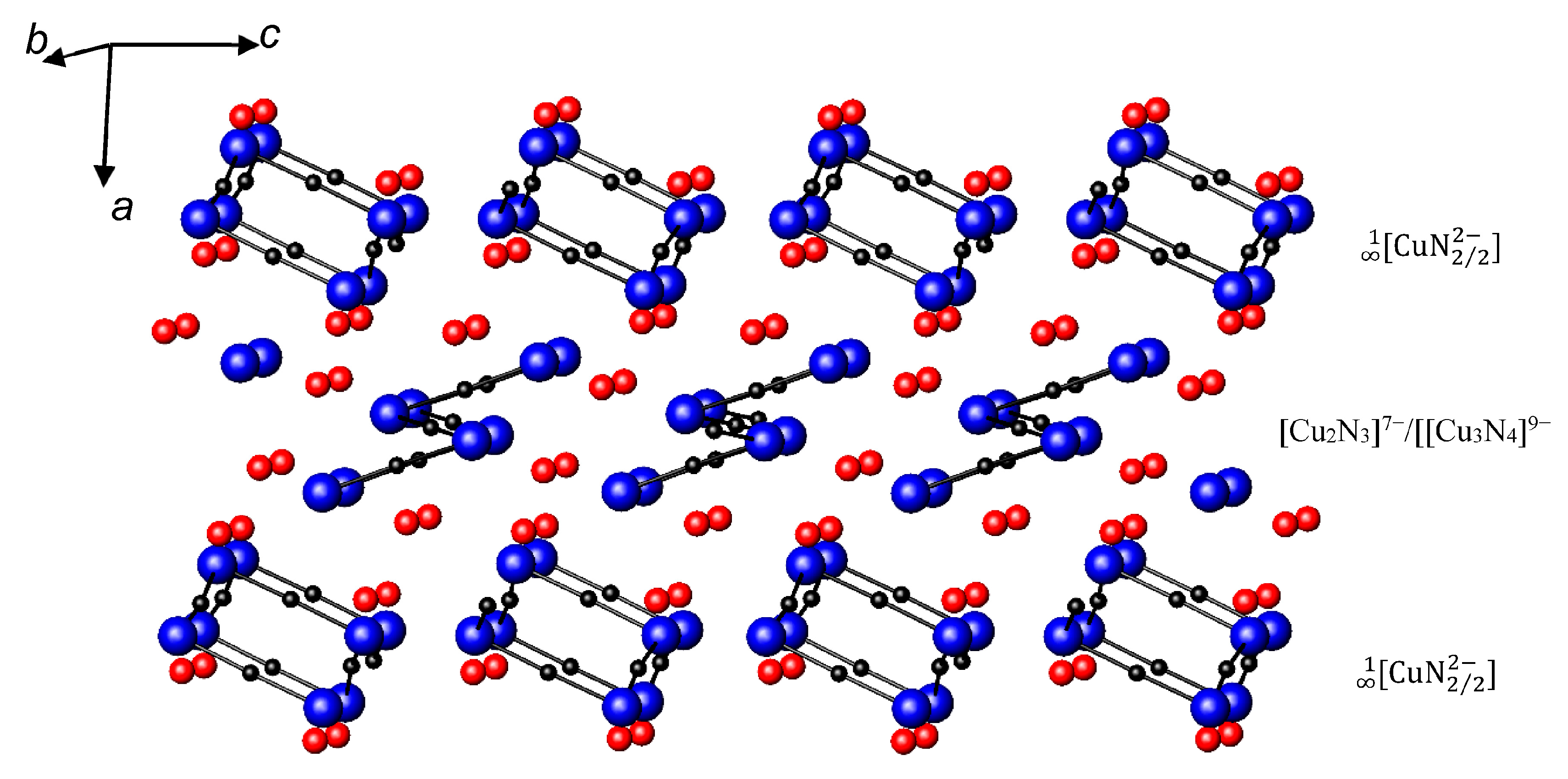
- Niewa, R.; DiSalvo, F.J. Breaking up Chains: The Nitridocuprates(I) BaCuN Ba16[(CuN)8][Cu2N3][Cu3N4] and Ca4Ba[CuN2]2. J. Alloys Comp. 1998, 279, 153–160. [Google Scholar] [CrossRef]
- Gudat, A.; Kniep, R.; Maier, J. Ca[(Ni1−xLix)N]: Limited solid solutions (0 ≤ x ≤ 0.58) in the system Ca[NiN] (Y[CoC]-type structure)-Ca[LiN] (modified fluorite-type structure). J. Alloys Comp. 1992, 186, 339–345. [Google Scholar] [CrossRef]
- Chern, M.Y.; DiSalvo, F.J. Synthesis, Structure, Electric and Magnetic Properties of CaNiN. J. Solid State Chem. 1990, 88, 459–464. [Google Scholar] [CrossRef]
- Yamamoto, T.; Kikkawa, S.; Kanamaru, F. Preparation of New Ternary Nitrides CaMN (M = Co, Ni). Solid State Ionics 1993, 63–65, 148–153. [Google Scholar] [CrossRef]
- Yamamoto, T.; Kikkawa, S.; Kanamaru, F. Preparation, Crystal Structure and Properties of a New Double Metal Nitride SrNiN and of Ca1−xSrxNiN (0 ≤ x ≤ 0.5). J. Solid State Chem. 1995, 115, 353–359. [Google Scholar] [CrossRef]
- Cordier, G.; Gudat, A.; Kniep, R.; Rabenau, A. LiSrN—A 3-Dimensional Structural Arrangement of Corner and Edge Sharing Orthorhombic Bipyramids Ni2/2Sr4/4. Angew. Chem. Int. Ed. Engl. 1989, 28, 201–202. [Google Scholar] [CrossRef]
- Gudat, A.; Haag, S.; Kniep, R.; Rabenau, A. On the Crystal Structure of BaNiN a Low Valency Nitridoniccolate(I). J. Less Common Met. 1990, 159, L29–L31. [Google Scholar] [CrossRef]
- DiSalvo, F.J.; Trail, S.S.; Yamane, H.; Brese, N.E. The Crystal Structure of Sr6Cu3N5 with Isolated, Bent (Cu2N3)7− anions and the Single Crystal Structure Determination of SrCuN. J. Alloys Comp. 1997, 255, 122–129. [Google Scholar] [CrossRef]
- Tennstedt, A.; Kniep, R. BaCoN—A Low Valency Nitridocobaltate with Angled Chains . Z. Anorg. Allg. Chem. 1994, 620, 1781–1785. [Google Scholar] [CrossRef]
- Gudat, A.; Milius, W.; Haag, S.; Kniep, R.; Rabenau, A. On the Crystal-Structure of Ba8Ni6N7, A Low Valency Nitridoniccolate with Infinite Helical [Ni–N] Zigzag Chains. J. Less Common Met. 1991, 168, 305–312. [Google Scholar] [CrossRef]

| Compound | a (Å) | b (Å) | c (Å) |
|---|---|---|---|
| Sr3Al2N4 | 5.901(3) | 10.005(5) | 9.580(4) |
| Sr2Ga2N4 | 5.9552(6) | 10.2753(8) | 9.5595(9) |
| Ba2Al2N4 | 6.179(2) | 10.052(4) | 10.230(4) |
| Ba2Ga2N4 | 6.2010(12) | 10.511(2) | 10.070(2) |
| Compound | Space Group | a (Å) | b (Å) | c (Å) | β (°) |
|---|---|---|---|---|---|
| Ca2VN3 | C2/c | 5.59538(5) | 10.41027(1) | 11.62243(1) | 92.342(1) |
| Sr2VN3 | C2/c | 5.71036(9) | 10.9521(2) | 12.2881(2) | 90.986(1) |
| Sr2NbN3 | C2/c | 5.9864(2) | 11.2271(3) | 12.5465(4) | 92.587(2) |
| Sr2TaN3 | C2/c | 5.9832(2) | 11.2832(2) | 12.5814(2) | 92.26(1) |
| Ba2VN3 | Cmca | 5.8614(4) | 11.4682(7) | 12.9121(9) | 90 |
| Ba2NbN3 | C2/c | 6.132(3) | 11.768(3) | 13.229(4) | 91.65(2) |
| Ba2TaN3 | C2/c | 6.130(3) | 11.815(4) | 13.263(5) | 91.2(1) |
| Compound | a (Å) | b (Å) | c (Å) |
|---|---|---|---|
| Th2CrN3 | 3.8654(5) | 3.5154(2) | 12.8446(19) |
| U2CrN3 | 3.7397(3) | 3.3082(3) | 12.3335(7) |
| Ce2CrN3 | 3.7900(10) | 3.4040(10) | 12.5170(2) |
| Th2MnN3 | 3.7919(3) | 3.5482(2) | 12.8321(4) |
| U2MnN3 | 3.7216(6) | 3.3274(2) | 12.2137(8) |
| Ce2MnN3 | 3.74994(6) | 3.44450(6) | 12.4601(2) |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Headspith, D.A.; Francesconi, M.G. Structural Classification of Quasi-One-Dimensional Ternary Nitrides. Inorganics 2016, 4, 37. https://doi.org/10.3390/inorganics4040037
Headspith DA, Francesconi MG. Structural Classification of Quasi-One-Dimensional Ternary Nitrides. Inorganics. 2016; 4(4):37. https://doi.org/10.3390/inorganics4040037
Chicago/Turabian StyleHeadspith, David Andrew, and Maria Grazia Francesconi. 2016. "Structural Classification of Quasi-One-Dimensional Ternary Nitrides" Inorganics 4, no. 4: 37. https://doi.org/10.3390/inorganics4040037
APA StyleHeadspith, D. A., & Francesconi, M. G. (2016). Structural Classification of Quasi-One-Dimensional Ternary Nitrides. Inorganics, 4(4), 37. https://doi.org/10.3390/inorganics4040037





