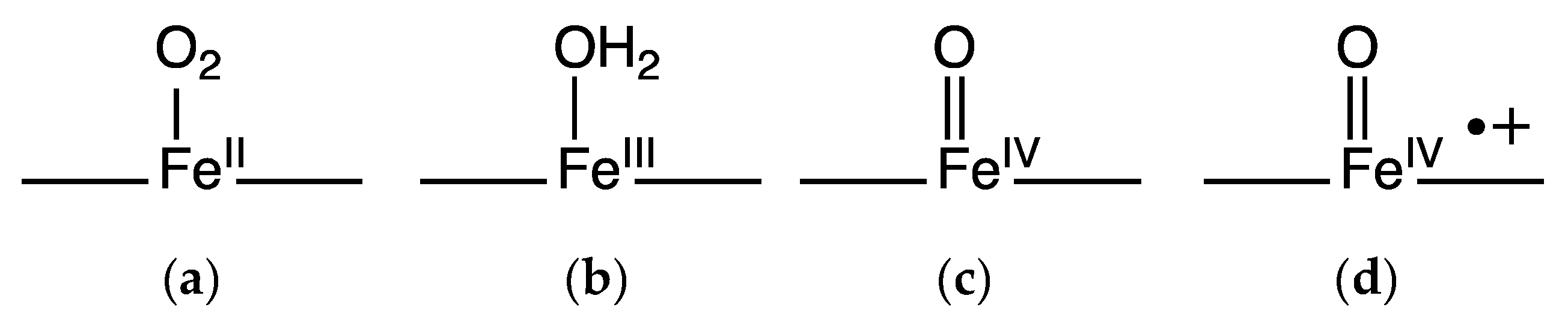Abstract
Some hemoproteins have the structural robustness to withstand extraction of the heme cofactor and replacement with a heme analog. Recent reports have reignited interest and exploration in this field by demonstrating the versatility of these systems. Heme binding proteins can be utilized as protein scaffolds to support heme analogs that can facilitate new reactivity by noncovalent bonding at the heme-binding site utilizing the proximal ligand for support. These substituted hemoproteins have the capability to enhance catalytic reactivity and functionality comparatively to their native forms. This review will focus on progress and recent advances of artificially engineered hemoproteins utilized as a new target for the development of biocatalysts.
1. Introduction
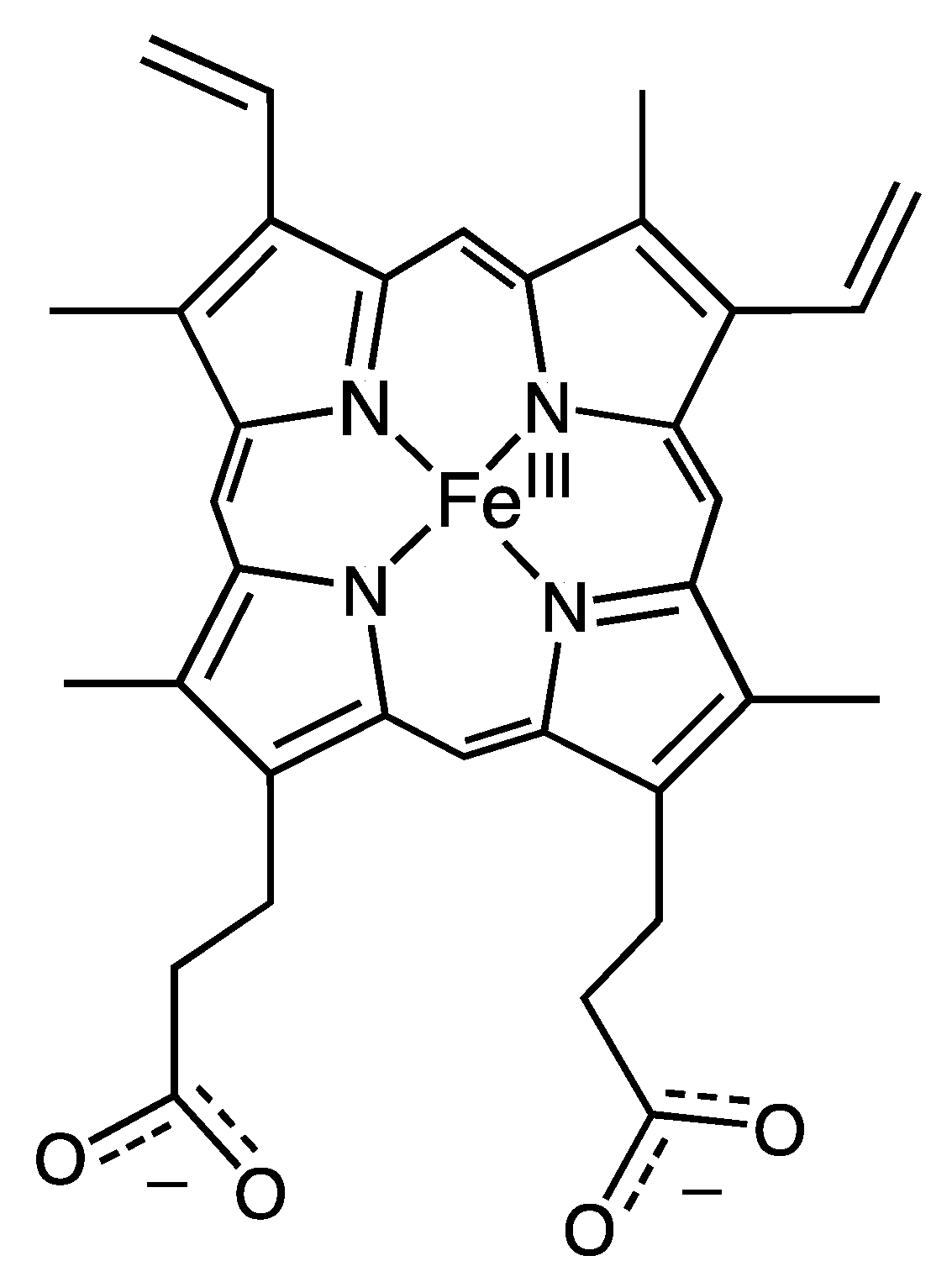
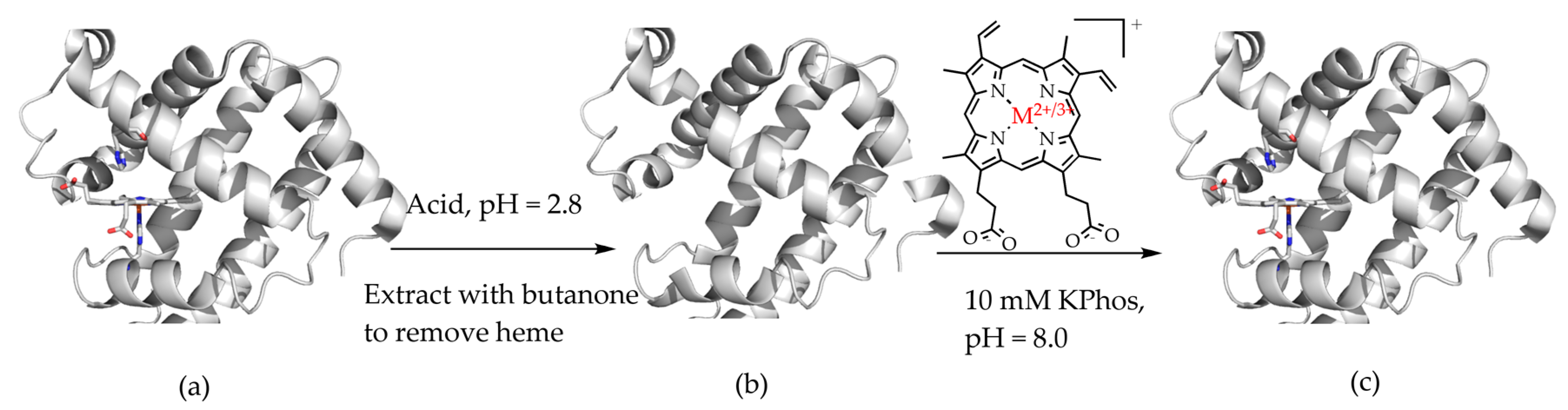
The ability to replace heme cofactors with transition metal complex structural analogs within intrinsically robust hemoproteins has recently met with unique possibilities for enhanced functionalities that are not displayed in their native forms. Since the discovery of the ability to separate the heme cofactor from the protein matrix of hemoproteins in the 1940s, researchers have exploited the versatility of their stabile protein structures in order to explore structure and function relationships by the substitution of new cofactors. The structure of the heme b cofactor (iron protoporphyrin IX) is shown in Figure 1. Teale described a simple acidification and heme extraction technique which allowed for generations of hemoprotein researchers to easily exchange cofactors and explore new possibilities for functionalities as shown in Figure 2 [1]. Many decades later the utility of replacing the native heme cofactor with heme analogs has emerged as a strategy to produce synthetic metalloenzymes to function as robust biocatalysts [2,3,4,5,6,7,8,9,10].
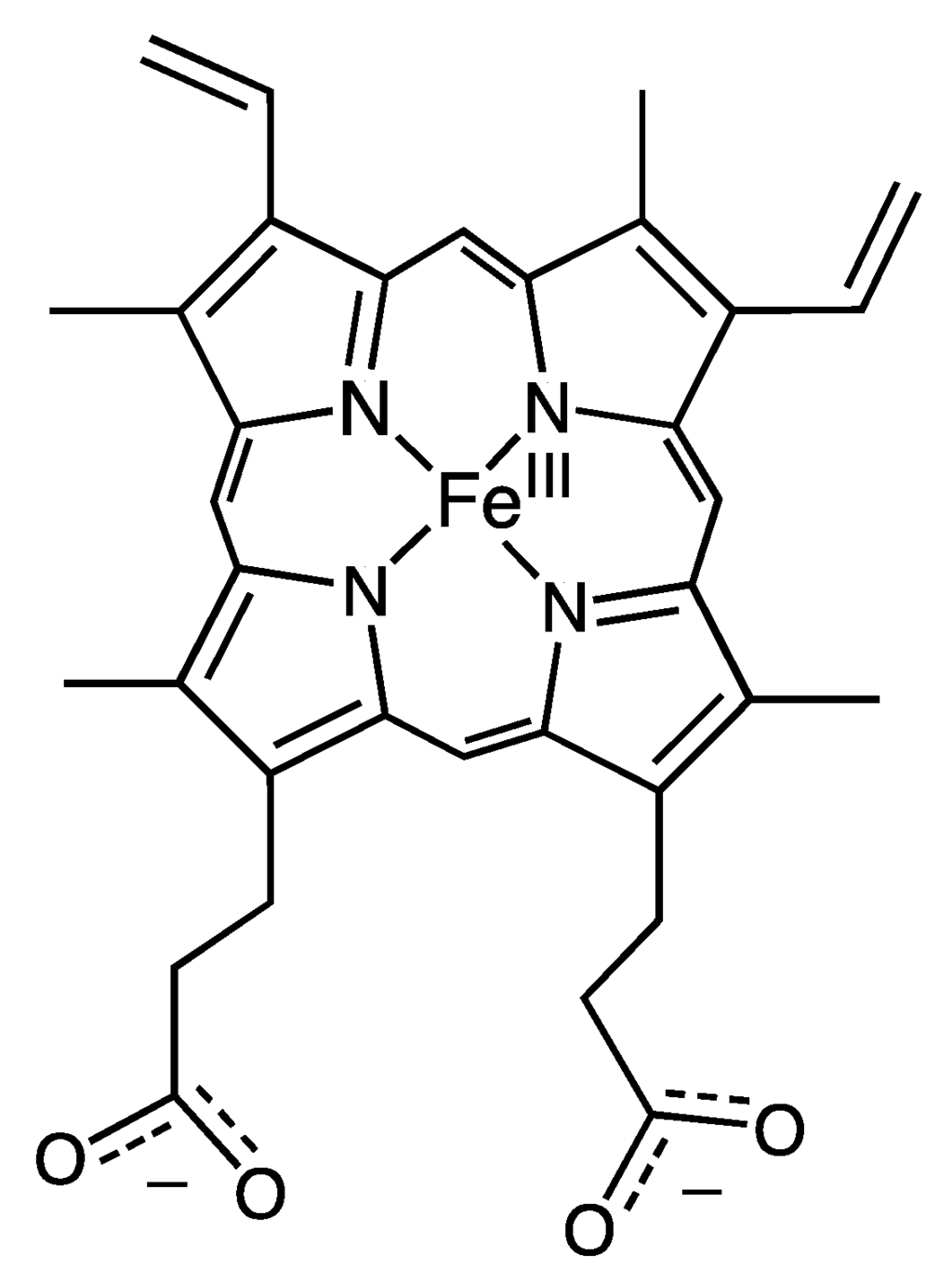
Figure 1.
Structure of iron(III) protoporphyrin IX (heme b). Cofactor found in the described heme proteins.
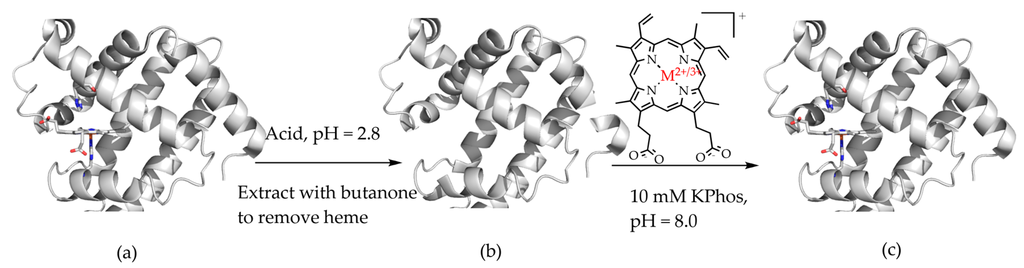
Figure 2.
Teale’s method of cofactor replacement in hemoproteins: (a) acidification of protein to release the heme cofactor; (b) apoprotein followed by alkalination and reconstitution with new cofactor (M = Fe, Mn, Cr, Ru, Rh, or Co); (c) to regenerate new holoprotein [1].
Replacement of the iron ion of the heme with other transition metals gained traction in the 1960s where researchers reported replacement with first and second row transition metals into stabile hemoproteins that could be readily manipulated (e.g., horseradish peroxidase, myoglobin, hemoglobin, and cytochrome c peroxidase) [11,12,13,14,15,16,17]. Yonetani and coworkers reported, in a series of papers, the substitution of horseradish peroxidase, myoglobin, and cytochrome c peroxidase with manganese protoporphyrin IX. They presented detailed analyses of the substituted hemoproteins regarding their spectroscopic properties upon ligand binding and oxidation utilizing UV/vis and electron paramagnetic resonance (EPR) spectroscopies. These types of studies were ongoing through the 1970s with a myriad of literature reports that show many heme proteins replaced with other transition metals and some modified porphyrins.
After a lag in research reports, the topic reemerged in the late 1980s through the 1990s with a few reports of transition metal substituted hemoproteins [18,19,20,21]. A report by Groves et al. on a manganese-substituted horseradish peroxidase was the most thoroughly characterized system showing the reaction with meta-chloroperbenzoic acid (m-CPBA) indicating that a Mn-oxo species was formed analogous to the compound I species formed by its iron ion counterpart [19]. This report was unique in comparison to other reports that were published earlier; for the first time, upon reaction with a two-electron oxidant, an intermediate was produced and fully characterized spectroscopically. The production of a Mn-oxo species lends itself to oxidative reactivity and a possibility emerged that metal-substituted hemoproteins might be a target to construct new types of biocatalysts.
The versatility of robust hemoproteins to provide a structural scaffold was explored by substitution with synthetic metal cofactors that resemble the flat porphyrin macrocycle in structure and by utilizing the proximal histidine residue as an anchor for the metal complex [6,7,22,23]. Watanabe, Lu, and colleagues have spent a considerable effort in exploring the potential of these types of substitutions particularly in myoglobin where enantioselective sulfoxidation of thioanisole is shown with Watanabe’s CrIII(Schiff-base)-myoglobin enhancing the oxidation rates by up to 15-fold comparatively to CrIII(Schiff-base) alone [8,10,22,24,25,26]. These examples did show promising improved functionality in some cases, but others have proven to be difficult to isolate in high yields and/or require protein modification or some form of covalent anchor in order to handle efficiently.
In the last five years, this area of research has started to expand into exploring new types of functionality and catalysis in reference to their native forms. To date, most studies have focused on transition metal substituted heme or iron-containing porphyrin derivatives and have compared these substituted proteins with their native function, which in some studies have resulted in better native function [2,11,12,13,14,18,19,20,21,27,28,29,30,31,32,33,34,35,36,37]. There have been a limited number of studies where enhanced reactivity and functionality is addressed and characterized. A couple of studies have shown much improved reactivity with respect to C–H bond cleavage and can be found with either a manganese porphyrin derivative or an alternative metal substituted in myoglobin [38,39]. Hayashi and coworkers describe a manganese porphycene-substituted myoglobin that catalyzes the H2O2-dependent alkane hydroxylation of C(sp3)–H bonds [38]. The ability of myoglobin and other heme-containing proteins to function as powerful catalysts offers unique opportunities to discover untapped entries into biocatalysis. This review is not meant to be exhaustive but rather to provide a framework of important discoveries in the area and to highlight examples of recent advances of artificially engineered hemoproteins maintaining a particular focus on enhanced functionalities.
2. Metal-Substituted Heme Proteins
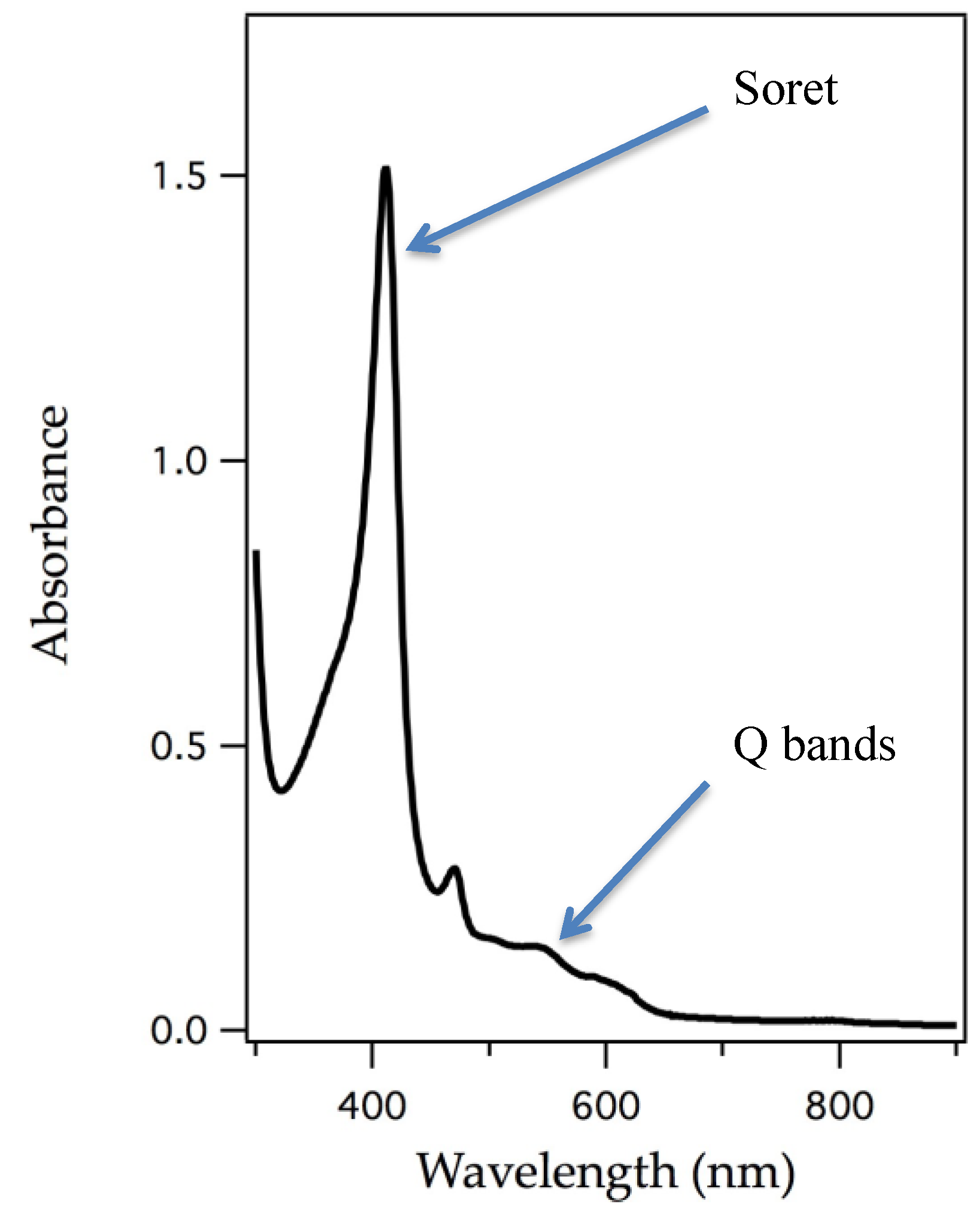
The removal of the heme cofactor from heme-containing proteins and addition of an appropriate metalloporphyrin into the protein matrix has been ongoing for decades. Successful reconstitution is shown by appearance of an intense peak, called the Soret band, which is a signature of a π to π* electronic transition, a characteristic of porphyrin macrocycles [40]. The Soret band and other visible bands shift with change of ligand binding to the central metal, identity of the metal, and metal oxidation state. Figure 3 represents an example of an electronic absorption spectrum of a porphyrin system. UV/vis spectroscopy has been a powerful technique to understand electronic changes specific to the porphyrin macrocycle and it also clearly identifies substitution.
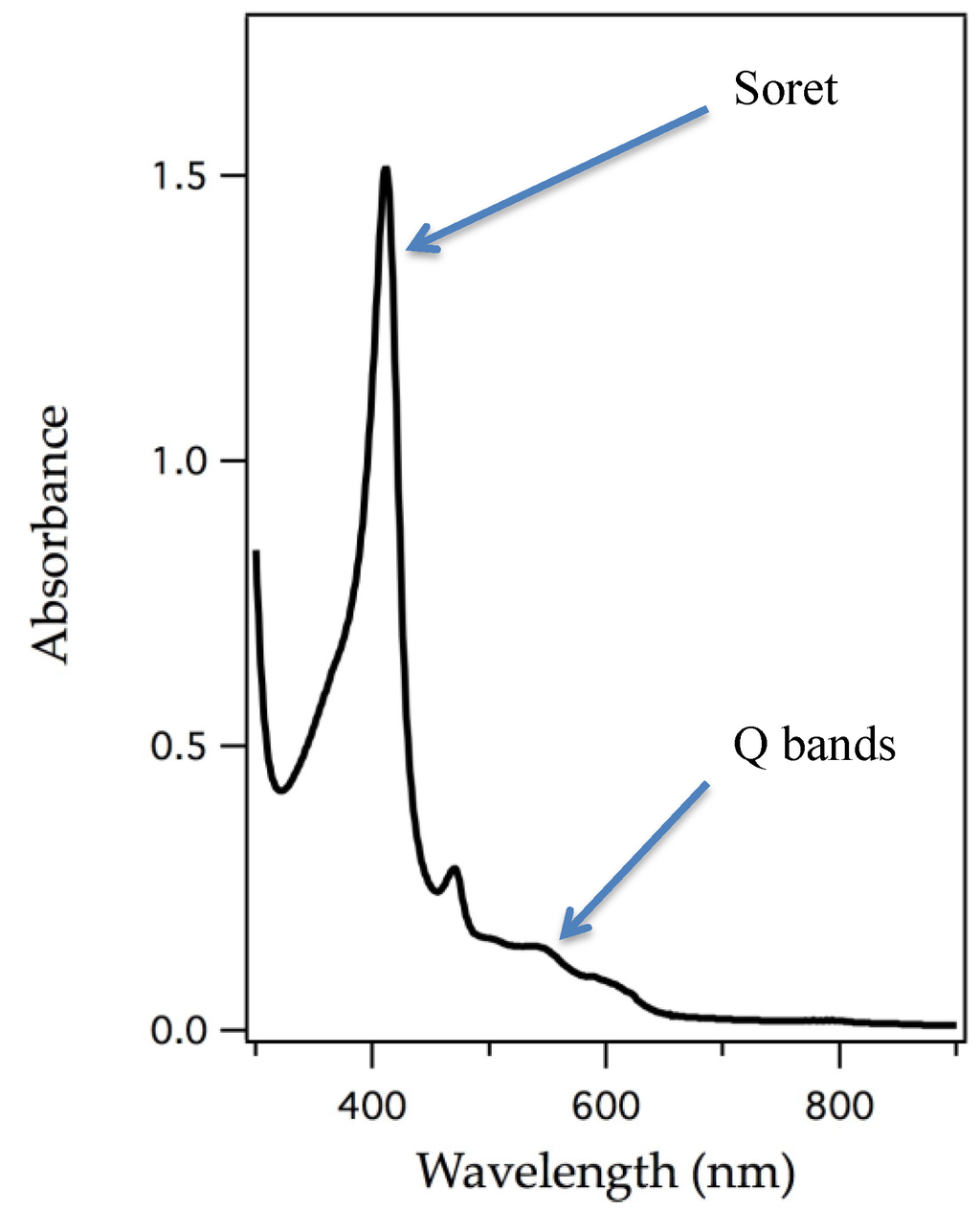
Figure 3.
The origin of intensely colored porphyrins arises from its π-conjugated system. A typical UV/vis spectrum of a porphyrin highlighting the π to π* electronic transition called the Soret band and the weaker, Q bands [40].
Table 1 highlights some metal-substituted hemoproteins and interesting results obtained from these studies. As shown from the table, there has been a plethora of metal-substituted hemoproteins reports going back many decades. While there are many more examples, the table serves to highlight the most impactful studies and only a few examples will be discussed in detail.

Table 1.
Transition metal substituted hemoproteins and their chemical and spectroscopic properties. EPR refers to electron paramagnetic resonance.
2.1. Manganese
Yonetani and coworkers first described the replacement of the iron ion with manganese in hemoproteins such as horseradish peroxidase, myoglobin, hemoglobin, and cytochrome c peroxidase [11,12]. Manganese substituted globins do not bind dioxygen which is typical for manganese complexes [12]. One study utilized this aspect of manganese coordination chemistry to determine the extent of cooperativity of oxygenation of hemoglobin subunits. They also reported hydrogen peroxide oxidations of manganese substituted myoglobin, horseradish peroxidase, and cytochrome c peroxidase. Only the peroxidases seemed to exhibit Mn(IV) character upon reaction with hydrogen peroxidase in these early reports, along with catalyzing peroxidatic oxidation of known reductants [18,21].
Groves describes the spectroscopic characterization of an intermediate produced by the reaction of manganese-substituted horseradish peroxidase (Mn-HRP) with meta-chloroperbenzoic acid. This detailed study showed the presence of a Mn(IV) species by electron paramagnetic resonance. Resonance Raman of 18O isotopic substitution revealed the presence of a Mn–O moiety as the active intermediate while a redox titration showed that one equivalent of reductant restored the Mn(III) state and yet another equivalent regenerated the resting state protein. This oxidized intermediate was described as Mn-HRP-I which is an analog of the prototypical compound I of the iron-heme horseradish peroxidase. Figure 4 shows the structures and names given to these oxygen species of iron heme systems [19]. Although manganese-substituted HRP displays a compound I like species, it is not clear if reactivity studies were conducted on this manganese complex.
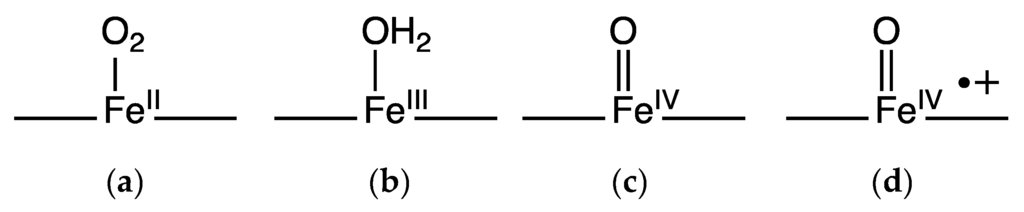
Figure 4.
Oxygen species in hemoproteins: dioxygen bound (a); met form in globins or resting enzyme state in peroxidases (b); compound II (c) and compound I (d).
Mn(II)–NO adducts of manganese substituted hemoproteins are known and they have their own interesting properties. They have been investigated as potential nitric oxide sensors due to their preferential binding of NO over dioxygen. Mn(II)–NO exhibits a uniquely bent NO adduct representing the first known case shown in either biological or synthetic Mn-porphyrin systems [43].
2.2. Cobalt
Myoglobin and hemoglobin have been targets of cobalt substitution since cobalt has dioxygen bonding properties [2,13,14,15,16,17]. These globins have been substituted with cobalt and their dioxygen association and dissociation rate constants have been measured in comparison with their native forms. Co hemoglobin binds dioxygen cooperatively but less cooperative than the iron version and binds O2 with less affinity [14].
Cobalt substituted myoglobin has also been shown to provide enhanced C–H amination activity. Although less efficient than its manganese substituted myoglobin counterpart, substitution with Co(II) exhibits enhanced catalytic activity in comparison to its iron counterpart showing 64 TTN (total turnover numbers) also representing a case where the protein matrix provides significant activity enhancement since free Co(II)PPIX shows a TTN of five [41].
2.3. Chromium
Chromium substituted myoglobin has not shown reactivity upon reaction with two-electron oxidants. One study utilized a unique approach to generate a high valent nitrogen species after it was determined that photolysis of Cr-azido species in myoglobin and cytochrome c peroxidase did not yield the expected nitride species [42]. A CrN(PPIX) was produced synthetically by photolysis of the azide complex and was used for direct incorporation into myoglobin and cytochrome c peroxidase. The species was purified and the UV/vis spectrum suggested incorporation of the Cr(N) species into both myoglobin and cytochrome c peroxidase, although the electronic spectra were similar enough to free CrN(PPIX) that adherence of this species and not direct binding at the active site could not be ruled out. Further electron paramagnetic resonance (EPR) confirmed a low spin Cr(V) complex, although direct evidence of incorporation of the Cr(N)PPIX moiety could not be confirmed due to similarities of their EPR spectra.
3. Rational Design of Heme Proteins that Promotes New Function
One strategy of engineering function into a hemoprotein that does not naturally perform a particular function of interest is to alter amino acids involved in reactivity and substrate binding to modify noncovalent interactions or to minimize steric constraints for substrates to bind. Several examples show that the successful engineering of hemoproteins can be used to afford enhanced desired reactivity. For clarity, Figure 4 shows the oxygen species that may exist in globins and peroxidases during oxidative processes that are referenced in the following text. The examples provided in this section maintain the native iron heme cofactor while changing amino acids around the active site to impart new functionality.
3.1. Myoglobin
Myoglobin is a dioxygen storage protein and its active site structure is quite amenable to positioning dioxygen bound to the ferrous ion rather than participating in oxidative chemistry; in fact, it is quite stable with respect to autoxidation to metmyoglobin. In its native form, peroxidative reactivity of substrates is not very efficient even though oxidative intermediates are presumably formed from the reaction with peroxides. Lack of reactivity of the native form of myoglobin has been examined and many research studies have concluded that the active site of myoglobin lacks a substrate-binding site, which would prohibit oxidative intermediates to productively react with substrates [4,8,24,25,44,45,46,47,48]. To this end, many studies have employed the use of myoglobin mutants to affect a change of reactivity by (a) opening up the active site to afford substrate binding or (b) changing amino acids at the active site to mimic structural aspects of other enzymes and their functions.
3.1.1. Peroxygenase and Peroxidase Activity
A series of reports have described the engineering of the myoglobin active site to induce peroxygenase (sulfoxidation and epoxidation) and peroxidase reactivity [4,9,44,45,46,47,48]. Many mutants have been described, and some have showed increased reactivity. The exchange of a leucine residue (L29) with a hydrogen bonding histidine residue resulted in much enhanced peroxygenase activity as shown by the increased rate and enantioselectivity of thioanisole oxidation, presumably by adding a hydrogen bond between the histidine residue and the ferryl (FeIV=O) species. Watanabe proposed that myoglobin’s inherent lack of native reactivity is due to the absence of an appropriate substrate-binding site. A double mutant (F43W/H64L) of horse heart myoglobin served to open up the active site and hydroxylation of an internal tryptophan residue occurred [46]. For clarity, Figure 5 shows the mentioned amino acid mutations. This report represented the first case of myoglobin acting to oxidize aromatic carbons. From these reports, some researchers have concluded that the active site of myoglobin is not amenable to substrate binding and therefore not able to serve as a useful target to discover new types of reactivity. The distal histidine residue in myoglobin is in close proximity to the heme and it serves as a hydrogen bond acceptor. The H64D mutant of myoglobin is shown to react with m-chloroperbenzoic acid (mCPBA) to afford a compound I species that promotes peroxidase activity showing reactivity towards guaiacol (2-methoxyphenol) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonate) (ABTS), which are common target substrates to assay for peroxidase activity [44,47].
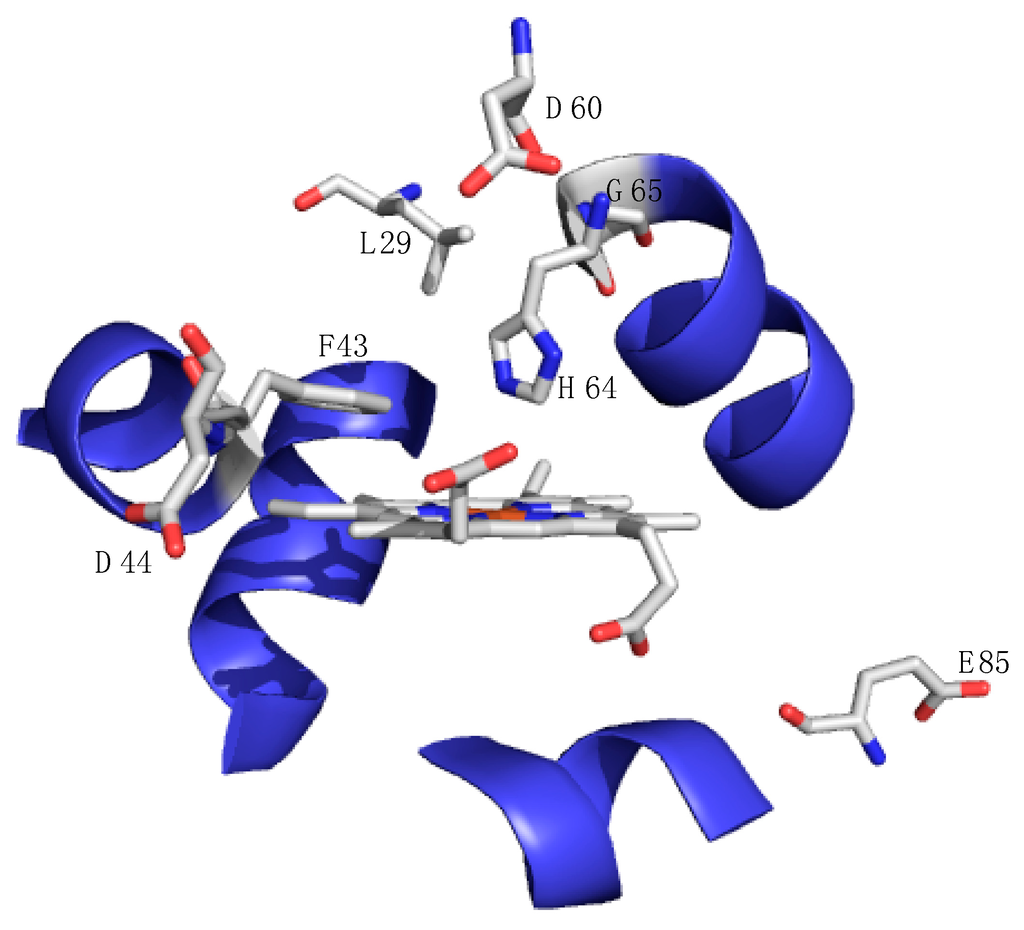
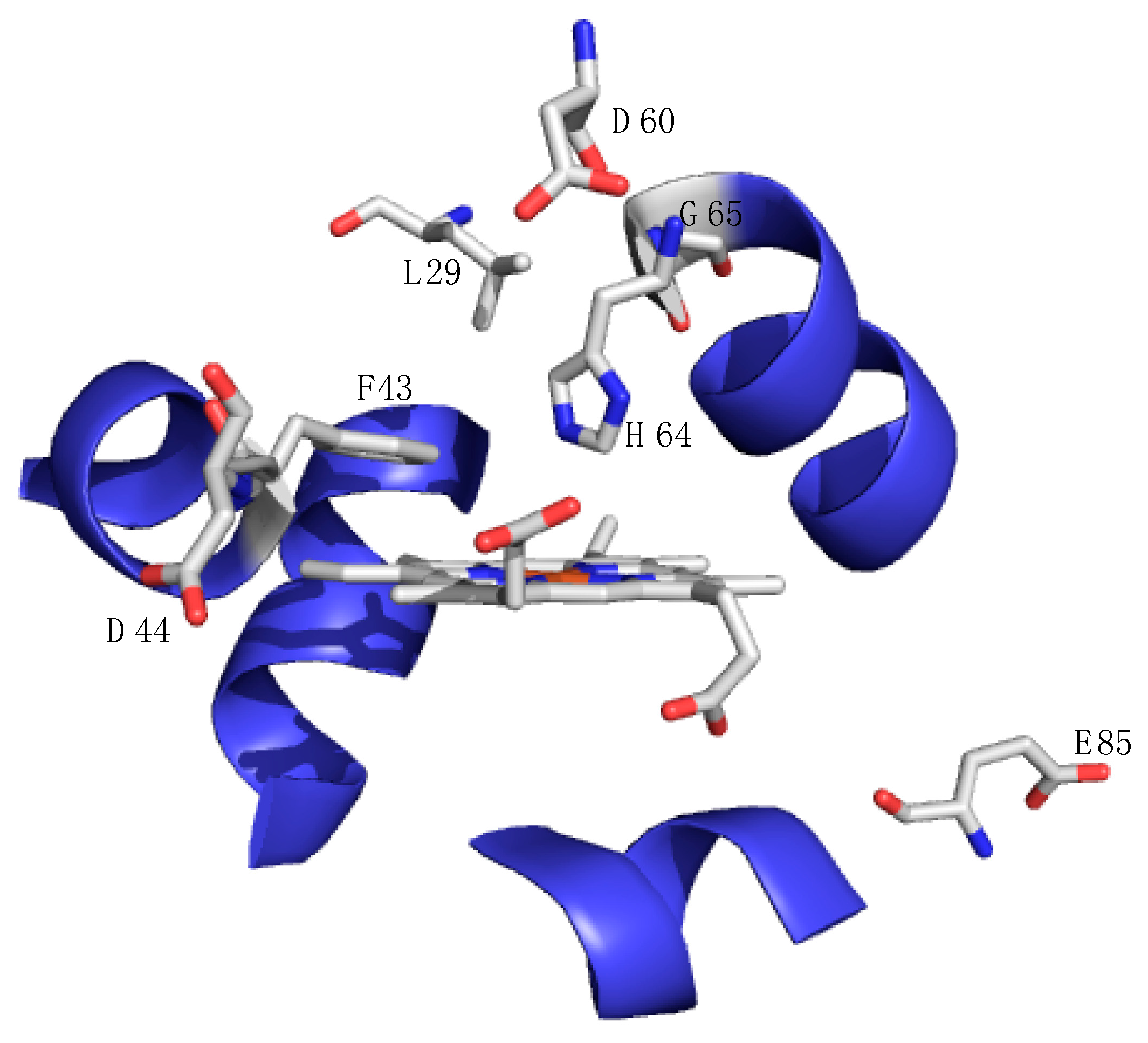
Figure 5.
X-ray crystal structure of the active site of sperm whale myoglobin (pdb accession number: 1JP6) depicting amino acids that have been modified to facilitate desired function as described in the text: carbon atoms are depicted in gray, oxygen atoms are red, and nitrogen atoms are blue while the helical structure about the active site is shown in blue. All amino acid mutations are less than 8 Å from the active site heme [49].
3.1.2. Dehalogenase Activity
Much work has focused on increasing myoglobin’s peroxidase activity and the same mutants were also applied to study dehalogenation activity [50]. Studies by Dawson and coworkers have shown that the differing types of reactivity, peroxidase vs. dehalogenase, arose from changes at the proximal and distal histidine positions, thus also demonstrating how the role of hydrogen bonding interactions can directly influence reactivity and change types of functionality overall, thereby adding to our knowledge of structure-function relationships of globins.
3.1.3. Oxidase Activity
Yi Lu and coworkers have been exploring the versatility of myoglobin to impart function via well-designed myoglobin mutants. By incorporating a copper binding site and changing the electrostatics of the solvent accessible sites, the L29H/F43H/G65Y and D44K/D60K/E85K mutant of myoglobin transformed the oxygen storage protein into a robust oxidase with dramatic enhanced functionality. A few such studies incorporated a CuB site in myoglobin in order to provide insight into the mechanism of reduction of dioxygen by heme-copper oxidases [8,51,52,53,54]. In this way, myoglobin serves as a small model protein that mimics the large membrane-bound heme-copper oxidase. The CuB site was engineered into myoglobin by substitution of two amino acids that are close to the active site: F43H and L29H serve as two histidine ligands while H64 provides the third histidine ligand (see Figure 5 for a reference to these point changes). This new copper-binding site is in close proximity to the heme active site, thus setting up a cooperative network of copper and heme just as in heme-copper oxidases. Results suggest that coupling of the two metal ion sites can have dramatic effects on reduction potential at the heme site.
Another study utilizing the same system as the heme-copper oxidase model showed the versatility of such an approach by demonstrating how myoglobin can also function as a small working model of bacterial NO reductases [53].
3.1.4. Some Recent Examples of the Versatility of Myoglobin Mutants
Fasan and coworkers have developed a library of myoglobin mutants and explored chemistries with alternative metal centers showing some interesting capabilities in reactivity that both native myoglobin and free hemin do not exhibit. They have developed several target chemical transformations that are synthetically valuable to organic synthesis methodology. In the last two years, their group has shown significantly enhanced reactivity towards olefin cyclopropanation [55], provided the first biocatalytic example of the conversion of primary azides to aldehydes [56], carbene N–H and S–H insertion [57,58], and olefination of aldehydes by fine-tuning the active site with larger and smaller apolar residues within the protein scaffold of myoglobin [59].
3.2. Horseradish Peroxidase
Thioanisole sulfoxidation by horseradish peroxidase proceeds via a peroxygenase mechanism by a two-step electron transfer from the same substrate. Expression of two mutants of horseradish peroxidase, F41L, and F41T, in a baculovirus/insect cell system resulted in enhanced reactivity of sulfoxidation compared with native horseradish peroxidase. Although impressive to impart greater functional performance by a single point mutation, the change was not effective at turning the peroxygenase activity into a greater epoxidation catalyst [60].
4. Insertion of Schiff-Base Cofactors into Myoglobin
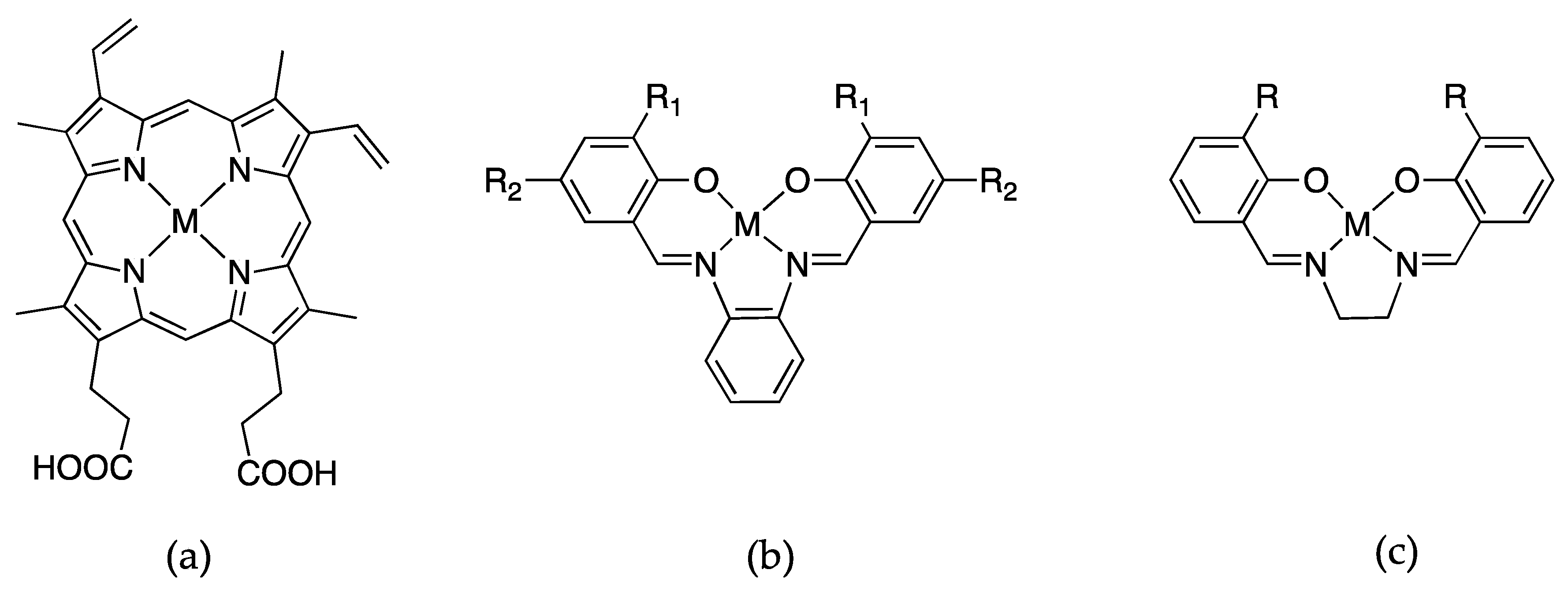
Another entry into the field of designer enzymes is the substitution of Schiff-base metal complexes into myoglobin. Metal-salen complexes are known to be powerful and robust catalysts and their planar structures make them an ideal choice to mimic the structure of porphyrins so that the active sites of the hemoproteins stay relatively unchanged [61,62,63,64,65]. Figure 6 shows the comparison of protoporphyrin IX and two salen metal complexes that have been utilized as surrogates for the heme cofactor.
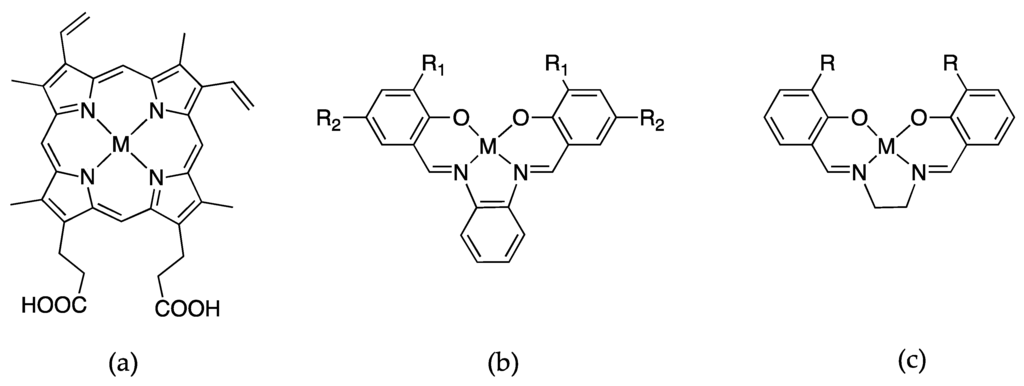
Figure 6.
Schiff-base metal complexes that are substituted into apomyoglobin, where M = Fe, Mn, Cr, or Co. (a) protoporphyrin IX; (b) salophen, where R1 = R2 = H or R1 = Me and R2 = H or R1 =H and R2 = tBu and (c) salen where R = Me, Et, or n-Pr.
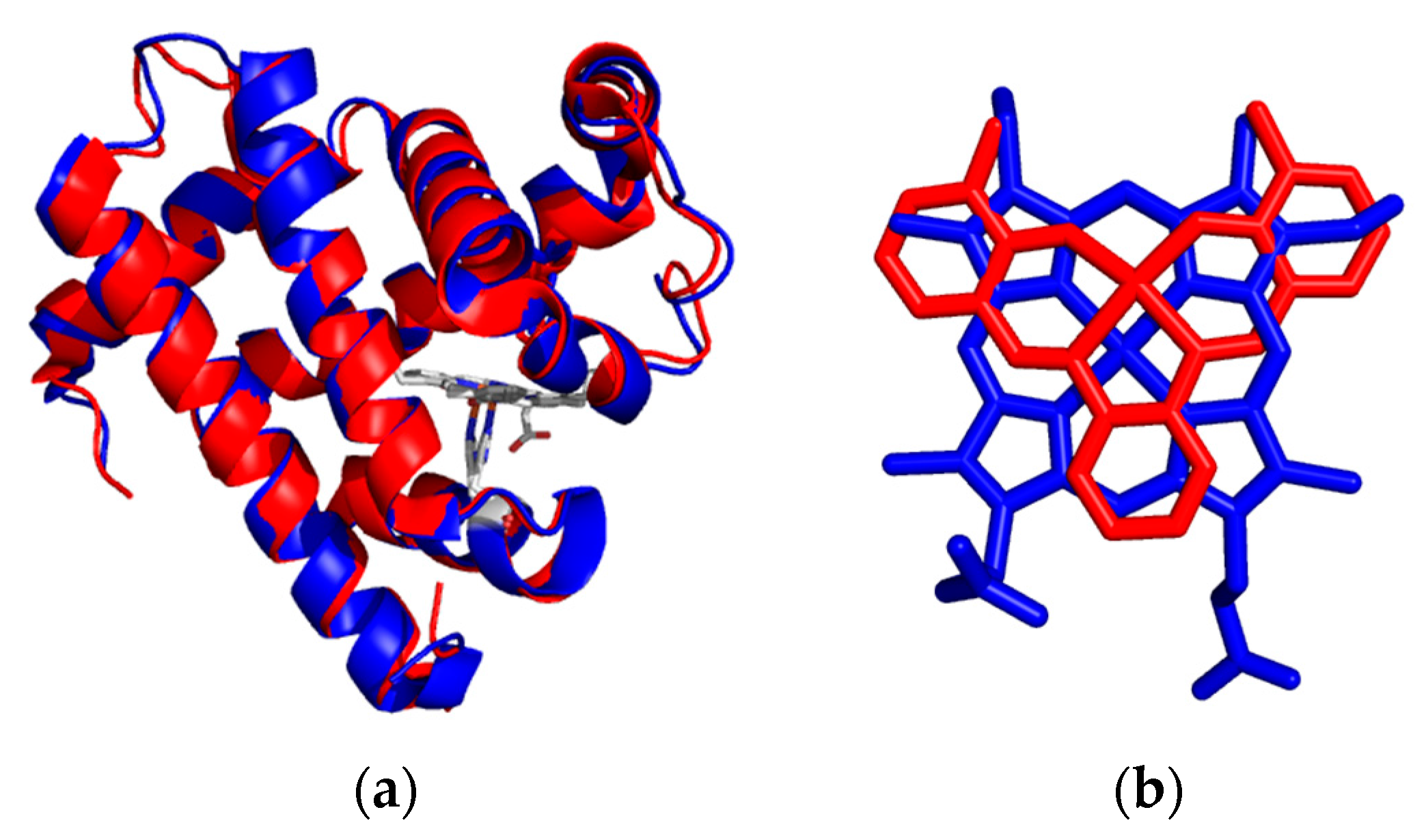
The protein matrix of myoglobin acts to anchor the metal complex and provides regio-selectivity of the sulfoxidation of thioanisole due to the amino acids in the protein scaffold. Several studies emerged from this idea that metal complexes, not just porphyrin or porphyrin-like structures, could serve as the active site moiety. By a simple comparison of their structures with respect to heme b, it is observed that the relatively planar Schiff-base ligands could simulate a porphyrin structure at the active site of hemoproteins. The crystal structure, shown in Figure 7, reinforces this idea that the similarity in structure between the porphyrin and the Schiff-base ligands allows the protein to have similar non-covalent interactions with the Schiff-base ligand as the native porphyrin cofactor and to ligate the metal ion to the proximal histidine residue serving as an anchor.
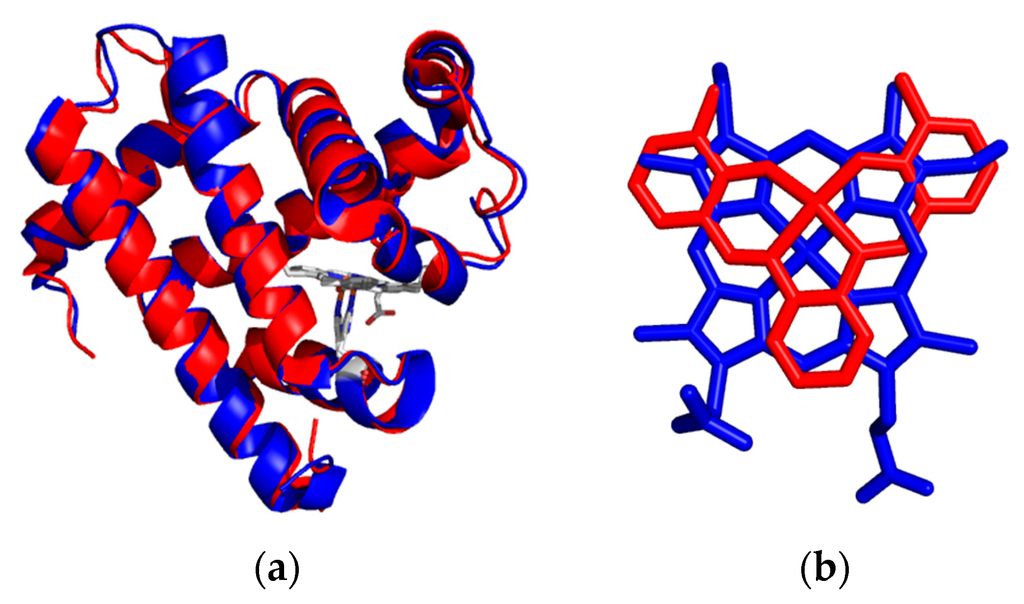
Figure 7.
Overlay of the X-ray crystal structures of metmyoglobin (blue) and iron salophen substituted apoA71Gmyoglobin (red) (a); Active site comparison of the heme and iron salophen cofactors (b).
The development of inserting metal complexes into hemoproteins opens up exciting possibilities for the design of synthetic metalloenzymes. A particularly important distinction between the substitution of the salen-like complexes and porphyrins and porphyrin-derivatives was the low yields of substitution of the Schiff-base metal complexes into apomyoglobin [7,25,26]. The redesign of the active site structure of myoglobin with a few amino acid changes in order to improve substrate accessibility and steric hindrance of the Schiff-base ligand offered additional success. Upon analysis of the active site structure of myoglobin determination of an A71G substitution enforced accommodation of substrates in the active site by reducing steric constraints. Figure 7 shows the overlay of the X-ray crystal structures of metmyoglobin and A71Gmyoglobin substituted with iron salophen [22]. As mentioned previously, the structure of salophen mimics the planar structure of porphyrin very well as they are directly overlaid on each other. By reducing steric constraints, the active site substituted with chromium salophen supports enantioselective sulfoxidation of thioanisole.
Lu and coworkers used dual covalent anchors by introducing cysteines in the heme pocket to provide a covalent anchor via methane thiosulfonate groups. The dual anchoring system provided an enhanced rate and enantioselectivity of sulfoxidation of thioanisole [24].
5. Porphyrin-Derivative Substitution
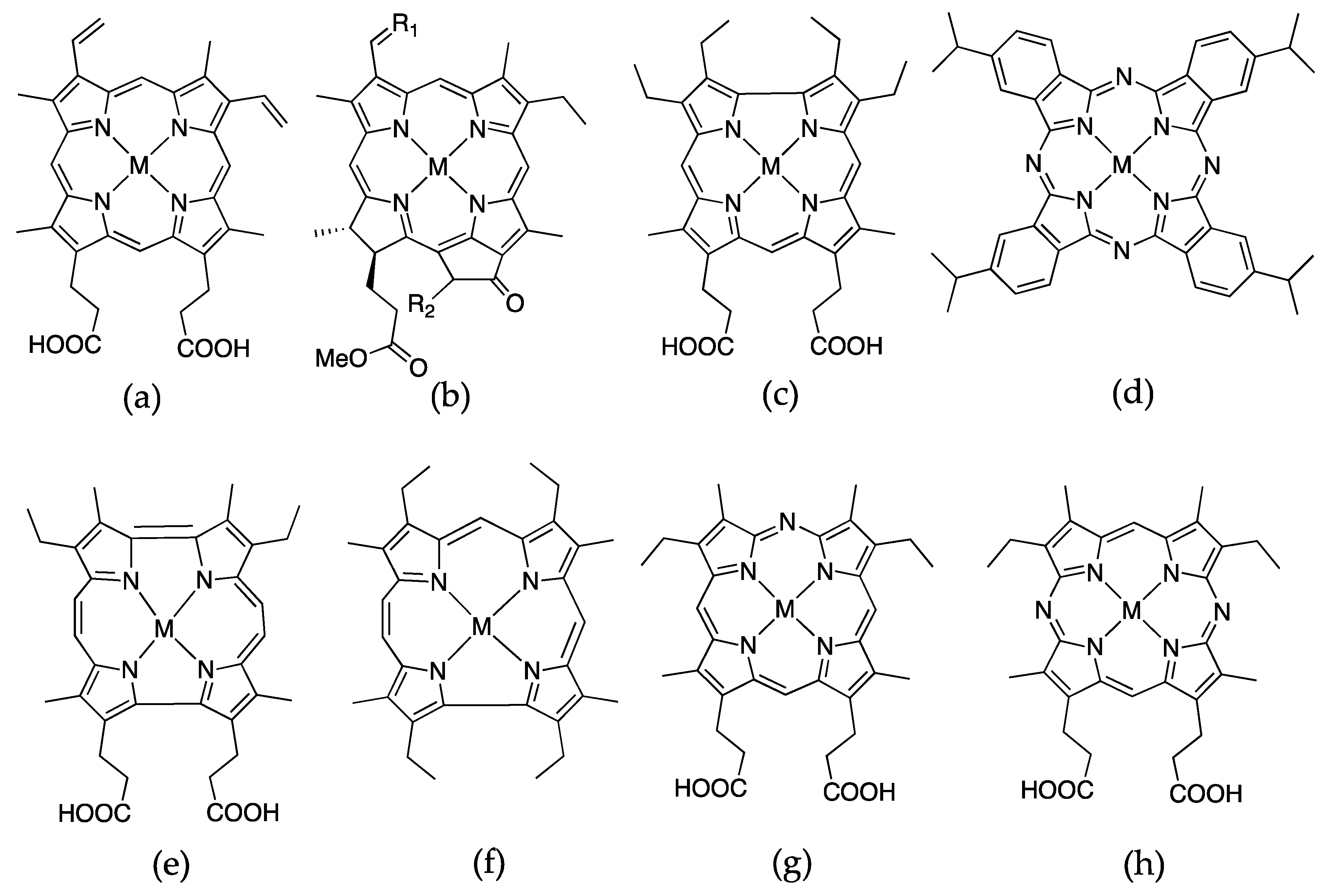
Another entry into the area of engineered hemoproteins comes from the insertion of porphyrin-derivatives into globins and peroxidases. Each type of structure as outlined below offers interesting exploration into structure function relationships since their electronic structures are so similar yet they impart different properties upon substitution. Many of these examples utilize myoglobin due to its exceptional versatility to serve as a protein scaffold for incorporation of non-native cofactors. Figure 8 shows the porphyrin-derivatives that have been successfully incorporated into hemoproteins.
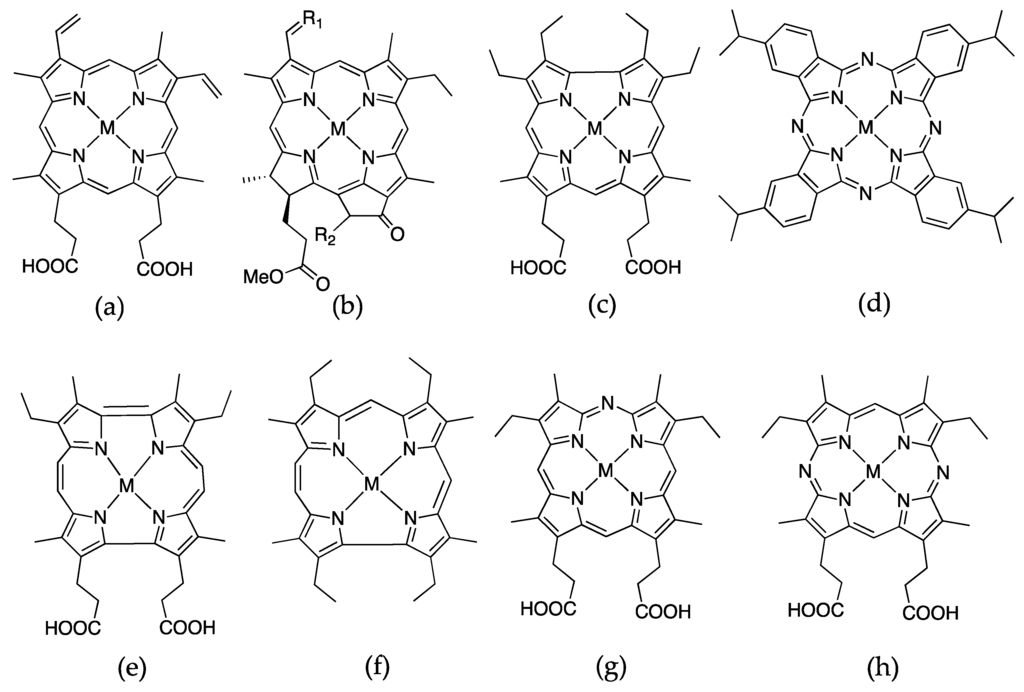
Figure 8.
Porphyrin and porphyrin derivatives that have been successfully substituted into apohemoproteins, where M = Fe, Co, Cr, or Mn: (a) protopophyrin IX; (b) chlorin; (c) corrole; (d) phthalocyanine; (e) porphycene; (f) hemiporphycene; (g) monoazahemin; (h) diazahemin.
5.1. Chlorin
The magnesium complexes of chlorins are derived from the photosynthetic pigment of green leaves. Iron chlorins are found naturally in several enzymes as many forms where each structure is two less π electrons in comparison to iron protoporphyrin IX (Figure 8b). These are collectively referred as green hemes since they are green in color rather than red as in other globins and enzymes that have heme b as the native cofactor. Ligand binding studies of CO and O2 represent faster association of these small ligands but lower affinity for dioxygen due to decreased electron density in the iron d-orbitals weakening the Fe–O2 bond [66,67].
5.2. Corrole
The structure of iron corrole resembles porphyrin but lacks one meso carbon (Figure 8c). Hayashi and coworkers have substituted the iron corrole macrocycle into myoglobin and horseradish peroxidase and the oxidation of guaiacol was monitored [68]. It was shown that incorporation of the iron corrole into myoglobin enhanced its peroxidase functionality, while incorporation into horseradish peroxidase showed a decrease in its native peroxidatic function. This difference in reactivity can be attributed to the trianionic character of the macrocycle in comparison to the porphyrin.
5.3. Azahemins and Phthalocyanines
Phthalocyanines are the tetraza derivatives of porphyrins, the structure is shown in Figure 8d. Reconstitution of a phthalocyanine in myoglobin results in the binding of the distal histidine to facilitate a 6-coordinate iron ion which results in a substantial structural change to accommodate binding [29]. The strong proclivity to be 6-coordinate is not unusual for phthalocyanines since there are few examples of a coordination number of five [69,70]. Azahemins are a group of porphyrin derivatives that are porphyrin and phthalocyanine hybrids where either one or two nitrogen atoms replace carbons at the meso positions as shown in Figure 8g,h, respectively. Both of these groups have been incorporated into myoglobin and their ligand binding capabilities investigated [30,31]. In contrast to native myoglobin, each binds nitrogenous ligands to the ferrous versions, although binding is stronger in the diazahemin representing electronic similarities with phthalocyanines.
5.4. Porphycene
Hayashi and coworkers have several examples of utilizing a new type of porphyrin derivative, porphycene (Figure 8e). It is an entirely synthetic porphyrin with a difficult ten-step synthesis yielding only a milligram of the porphycene for reconstitution into apomyoglobin. Iron porphycene substituted myoglobin yielded interesting functionality where the new myoglobin has an extremely high affinity for dioxygen and selectivity against CO binding without autoxidation of the Fe(II) in comparison to the native myoglobin [32,34,35]. Another study incorporated iron hemiporphycene (Figure 8f) into myoglobin also showed the new myoglobin exhibiting high dioxygen affinity in comparison with native myoglobin [33]. Cobalt porphycene myoglobin also demonstrated increased O2 affinity [71]. Most notably, no amino acids were changed to afford the enhanced functionality. These studies represented a largely untapped field of study where engineering function into proteins did not necessarily involve mutation of amino acid residues.
6. New Catalytic Functions Identified
Replacement of the heme cofactor within hemoproteins has been studied for over seventy years, but investigations into the potential of new functionality and reactivity has been limited. Only recently have reports emerged that explore the diverse chemistries that are possible. It has been shown recently that minor changes in electronic structure of the substituted cofactor or with the replacement with alternative metals has profound effects on the ability of relatively non-reactive proteins (myoglobin) into functionally interesting enzymes. Impressive results have emerged that have shown that replacement of the heme cofactor with other metals or with metal porphyrin derivative complexes can in itself be a useful method of producing synthetic metalloenzymes that have diverse functionalities.
6.1. Peroxidase Activity
Replacement of the heme cofactor in myoglobin with porphycene, where M = Fe, resulted in a myoglobin that had enhanced peroxidase activity toward guaiacol by 11-fold [35]. This enhanced reactivity is displayed only by the slightly different electronic structure of porphycene in comparison with heme b. The same substitution into horseradish peroxidase dramatically increased the rate of sulfoxidation of thioanisole, a reaction HRP is known to perform.
6.2. C–H Bond Cleavage Activity
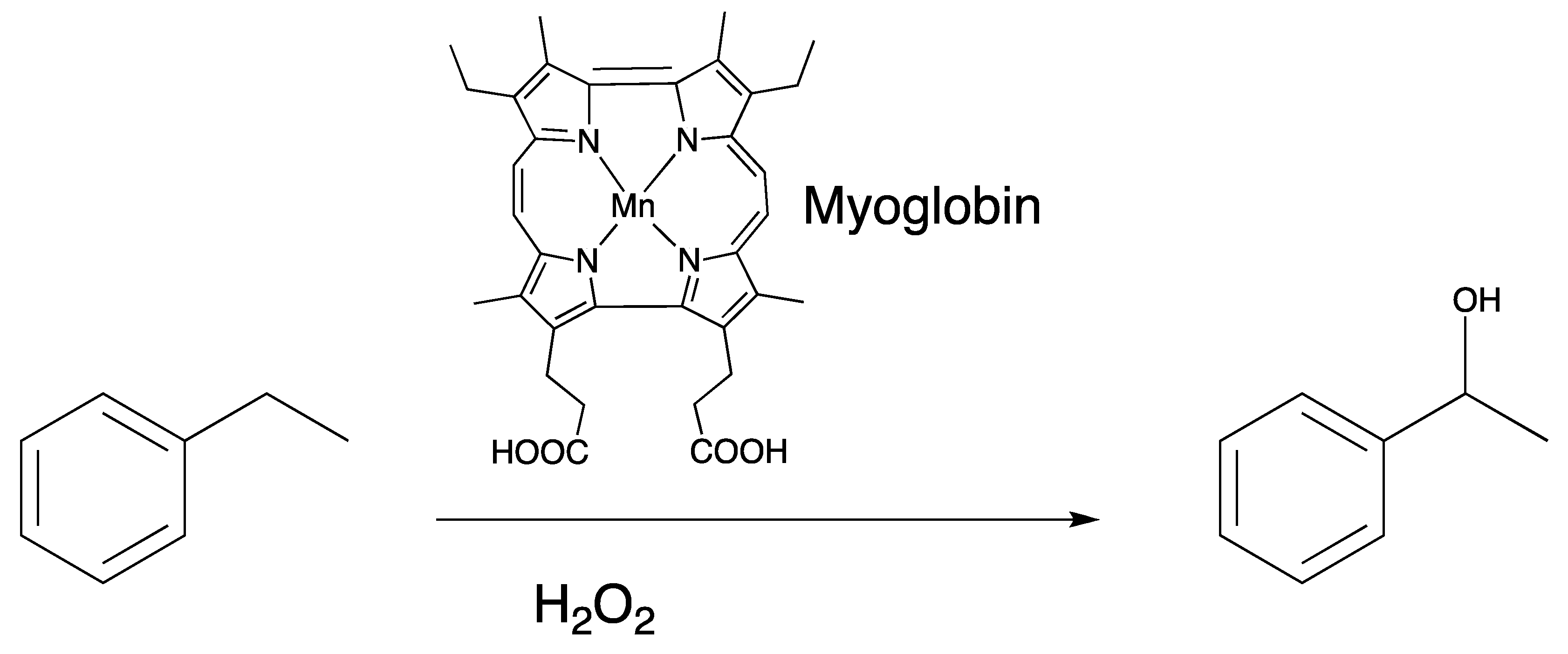
The heme in myoglobin was substituted with manganese protoporphyrin IX and was shown to exhibit oxidative reactivity that is not displayed with the native iron heme in myoglobin, which is known to be a very weak peroxygenase. The presence (and stability) of an oxidizing intermediate lends itself to afford greater oxidative capabilities than what is displayed in native myoglobin [39]. Substitution with the manganese ion at the heme center results in C–H bond cleavage of the weak C–H bonds of cyclohexadiene (77 kcal/mol). The rate of C–H bond cleavage shows a linear dependence of kobs with the concentration of C–H bonds of substrate. The linear dependence with substrate concentration indicates that C–H bond cleavage is rate-limiting, although the mechanism of a stepwise proton-electron transfer or a concerted hydrogen atom abstraction mechanism cannot be discerned.
6.3. Hydroxylase Activity
Cytochromes P450 are the only heme enzymes that perform monooxygenation of unactivated alkanes [72,73,74,75,76]. They are able to insert oxygen atoms into C–H bonds, which comprise some of the most difficult chemical transformations that are performed by enzymes. Interestingly, P450cam substituted with manganese protoporphyin IX does not hydroxylate its native substrate even though the spectral evidence suggests the presence of a Mn(IV)-oxo species upon reaction with iodosobenzene [75]. Comparatively to P450 enzymes, myoglobin functions as a dioxygen storage protein and does not participate in oxidative reactivity even though both proteins comprise the same heme cofactor. Difference in reactivity stems from (a) different proximal ligand (thiolate vs. imidozolate) and (b) promiscuous active site of most P450s to allow access from a variety of substrates [77]. Prior studies of substituted myoglobins have concluded that myoglobin cannot support reactivity of substrates since its active site structure does not have a clearly defined substrate binding site [46]. The last five years of exploring alternative chemistries of myoglobin has in some ways disproven this hypothesis and that myoglobin in particular may be more conducive to engineering function than originally thought. Substitution of the native heme cofactor in myoglobin with manganese porphycene had surprisingly enhanced reactivity as shown in Figure 9.
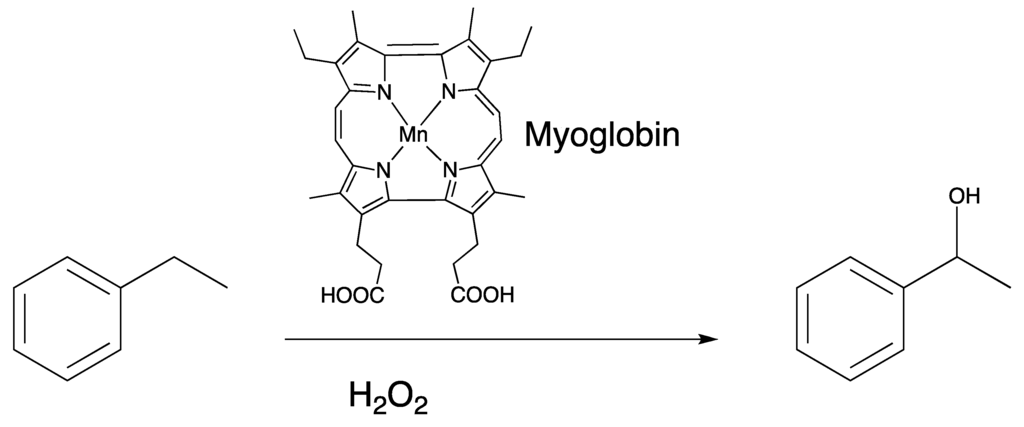
Figure 9.
MnPorphycene (MnPc) substituted myoglobin and its reactivity towards a C(sp3)–H bond. Catalytic hydroxylation of ethylbenzene by MnPc reconstituted myoglobin displays a TON (turnover number) of 13 and kinetic isotope effect of 2.4.
Given that myoglobin, apart from well-designed mutant versions and substitution with alternative cofactors, does not participate in oxidative chemistry, it is surprising that a simple substitution with manganese porphycene invokes much improved reactivity accessing substrates that are only equaled in P450 enzymes. The reaction was determined to be a catalytic with a turnover number of 13 after completion of the reaction with 1-phenylethanol isolated as the only product. Importantly, manganese porphycene alone does not hydroxylate ethylbenzene, thus showing the importance of the protein matrix of myoglobin. The kinetic isotope effect utilizing rate for reaction with ethylbenzene/rate for reaction with deuterated ethylbenzene is 2.4. A linear relationship with a negative slope was observed in the logarithmic initial rates plotted against BDE (bond dissociation energy), thus providing support that the rate-determining step of the reaction involves C–H bond activation by a Mn(V)-oxo species similar to the consensus mechanism for the hydroxylation by P450 enzymes.
7. Outlook and Conclusions
A target to access new types of biocatalysis is engineering structurally robust hemoproteins to perform desired reactivity. It has recently been discovered that simple substitution of the heme with a metal porphyrin or porphyrin-derivative can impart dramatically enhanced reactivity with respect to native function of hemoproteins without the requirement of recombinant protein or covalent anchors. These studies will allow for the understanding of the synergistic relationship between structure and function and how structural features can be tuned and modulated to afford anticipated chemical reactivity.
It is truly an exciting time in the field of metalloprotein design. The long-term goal of engineering synthetic metalloenzymes is to afford desirable chemical transformations. The use of small molecule inorganic complexes to catalyze required reactions has its limitations due to the reliance on precious metals, while metalloenzymes are able to catalyze chemical transformations with a significantly higher degree of efficiency and selectivity even with relatively abundant transition metals. The design of metalloenzymes to afford desirable reactivity has the potential to offer significant advantages for industrial and biotechnology applications.
Acknowledgments
The authors would like to thank Benedictine University for financial support.
Author Contributions
Kari L. Stone wrote the manuscript and Syeda M. Ahmed worked on the literature.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Teale, F.W.J. Cleavage of the haem-protein link by acid methylethylketone. Biochim. Biophys. Acta 1959, 35. [Google Scholar] [CrossRef]
- Dickinson, L.C. Metal replaced hemoproteins. J. Chem. Educ. 1976, 53, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Roncone, R.; Monzani, E.; Labo, S.; Sanangelantoni, A.M.; Casella, L. Catalytic activity, stability, unfolding, and degradation pathways of engineered and reconstituted myoglobin. J. Biol. Inorg. Chem. 2005, 10, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Roncone, R.; Monzani, E.; Murtas, M.; Battaini, G.; Pennati, A.; Sanangelantoni, A.M.; Zuccotti, S.; Bolognesi, M.; Casella, L. Engineering peroxidase activity in myoglobin: The haem cavity structure and peroxide activation in the T67R/S92D mutant and its derivative reconstituted with protohaemin-l-histidine. Biochem. J. 2005, 377, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.M.; Ward, T.R. Design of artificial metalloenzymes. Appl. Organomet. Chem. 2005, 19, 35–39. [Google Scholar] [CrossRef]
- Ueno, T.; Koshiyama, T.; Ohashi, M.; Kondo, K.; Kono, M.; Suzuki, A.; Yamane, T.; Watanabe, Y. Coordinated design of cofactor and active site structures in development of new protein catalysts. J. Am. Chem. Soc. 2005, 127, 6556–6562. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, N.; Ueno, T.; Unno, M.; Matsui, T.; Ikeda-Saito, M.; Watanabe, Y. Ligand design for the improvement of stability of metal complex: Protein hybrids. Chem. Commun. 2008, 229–231. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yeung, N.; Sieracki, N.; Marshall, N.M. Design of functional metalloproteins. Nature 2009, 460, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, T.; Fukumoto, K.; Watanabe, T.; Hayashi, T. Precise design of artificial cofactors for enhancing peroxidase activity of myoglobin: Myoglobin mutant H64D reconstituted with a “single-winged cofactor” is equivalent to native horseradish peroxidase in oxidation activity. Chemistry 2011, 6, 2491–2499. [Google Scholar]
- Hayashi, T.; Sano, Y.; Onoda, A. Generation of new artificial metalloproteins by cofactor modification of native hemoproteins. Isr. J. Chem. 2015, 55, 76–84. [Google Scholar] [CrossRef]
- Yonetani, T.; Asakura, T. Studies on cytochrome c peroxidase: XV. Comparison of manganese porphyrin-containing cytochrome c peroxidase, horseradish peroxidase, and myoglobin. J. Biol. Chem. 1969, 244, 4580–4588. [Google Scholar] [PubMed]
- Waterman, M.R.; Yonetani, T. Studies on modified hemoglobins: I. Properties of hybrid hemoglobins containing manganese protoporphyrin IX. J. Biol. Chem. 1970, 245, 5847–5852. [Google Scholar] [CrossRef]
- Yamamoto, H.; Kayne, F.J.; Yonetani, T. Studies on cobalt myoglobins and hemoglobins, II. Kinetic studies of reversible oxygenation of cobalt myoglobins and hemoglobins by the temperature jump relaxation method. J. Biol. Chem. 1974, 249, 691–698. [Google Scholar] [PubMed]
- Dickinson, L.C.; Chien, J.C.W. Comparative biological chemistry of cobalt hemoglobin. J. Biol. Chem. 1973, 248, 5005–5011. [Google Scholar] [PubMed]
- Gupta, R.K.; Mildvan, A.S.; Yonetani, T.; Srivastava, T.S. EPR study of 17O nuclear hyperfine interaction in cobalt-oxyhemoglobin: Conformation of bound oxygen. Biochem. Biophys. Res. Commun. 1975, 67, 1005–1012. [Google Scholar] [CrossRef]
- Woodruff, W.H.; Adams, D.H.; Spiro, T.G.; Yonetani, T. Resonance raman spectra of cobalt myoglobins and cobalt porphyrins. Evaluation of protein effects on porphyrin structure. J. Am. Chem. Soc. 1975, 97, 1695–1698. [Google Scholar] [CrossRef] [PubMed]
- Ikeda-Saito, M.; Yamamoto, H.; Imai, K.; Kayne, F.J.; Yonetani, T. Studies of cobalt myoglobins and hemoglobins. Preparation of isolated chains containg cobaltous protoporphrin IX and characterization of their equilibrium and kinetic properties of oxygenation and EPR spectra. J. Biol. Chem. 1977, 252, 620–624. [Google Scholar] [PubMed]
- Hori, H.; Ikeda-Saito, M.; Yonetani, T. Electron paramagnetic resonance and spectrophotometeric studies of the peroxide compounds of manganese-substituted horseradish peroxidase, cytochrome-c peroxidase and manganese-porphyrin model complexes. Biochim. Biophys. Acta 1987, 912, 74–81. [Google Scholar] [CrossRef]
- Nick, R.J.; Ray, G.B.; Fish, K.M.; Spiro, T.G.; Groves, J.T. Evidence for a weak Mn=O bond and a non-porphyrin radical in manganese-substituted horseradish peroxidase compound I. J. Am. Chem. Soc. 1991, 113, 1838–1840. [Google Scholar] [CrossRef]
- Mondal, M.S.; Mazumdar, S.; Mitra, S. Binding of cyanide and thiocyanate to manganese reconstituted myoglobin and formation of peroxide compound: Optical spectral, multinuclear NMR, and kinetic studies. Inorg. Chem. 1993, 32, 5362–5367. [Google Scholar] [CrossRef]
- Mondal, M.S.; Mitra, S. Kinetic studies of the two-step reactions of H2O2 with manganese-reconstituted myoglobin. Biochim. Biophys. Acta 1996, 1296, 174–180. [Google Scholar] [CrossRef]
- Ueno, T.; Ohashi, M.; Kono, M.; Kondo, K.; Suzuki, A.; Yamane, T.; Watanabe, Y. Crystal structures of artificial metalloproteins: Tight binding of FeIII(schiff-base) by mutation of Ala71 to Gly in apo-myoglobin. Inorg. Chem. 2004, 43, 2852–2858. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Hayashi, T.; Ando, T.; Hisaeda, Y.; Ueno, T.; Watanabe, Y. Hybridization of modified-heme reconstitution and distal histidine mutation to functionalize sperm whale myoglobin. J. Am. Chem. Soc. 2004, 126, 436–437. [Google Scholar] [CrossRef] [PubMed]
- Carey, J.R.; Ma, S.K.; Pfister, T.D.; Garner, D.K.; Kim, H.K.; Abramite, J.A.; Wang, Z.; Guo, Z.; Lu, Y. A site-selective dual anchoring strategy for artificial metalloprotein design. J. Am. Chem. Soc. 2004, 126, 10812–10813. [Google Scholar] [CrossRef] [PubMed]
- Garner, D.K.; Liang, L.; Barrios, D.A.; Zhang, J.-L.; Lu, Y. The important role of covalent anchor positions in tuning catalytic properties of a rationally designed mnsalen-containing metalloenzyme. ACS Catal. 2011, 1, 1083–1089. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-L.; Garner, D.K.; Liang, L.; Barrios, D.A.; Lu, Y. Noncovalent modulation of pH-dependent reactivity of a Mn-salen cofactor in myoglobin with hydrogen peroxide. Chemistry 2009, 15, 7481–7489. [Google Scholar] [CrossRef] [PubMed]
- Paulson, D.R.; Addison, A.W.; Dolphin, D.; James, B.R. Preparation of ruthenium(II) and ruthenium(III) myoglobin and the reaction of dioxygen, and carbon monoxide, with ruthenium(II) myoglobin. J. Biol. Chem. 1979, 254, 7002–7006. [Google Scholar] [PubMed]
- Aoyama, Y.; Aoyagi, K.; Toi, H.; Ogoshi, H. Reconstituted myoglobins with rhodium(III) complexes of mesoporphyrin and deuteroporphryin IX. Inorg. Chem. 1983, 22, 3046–3050. [Google Scholar] [CrossRef]
- Stynes, D.V.; Liu, S.; Marcus, H. Distal histidine coordination to iron in phthalocyanine-reconstituted myoglobin. Inorg. Chem. 1985, 24, 4335–4338. [Google Scholar] [CrossRef]
- Neya, S.; Kaku, T.; Funasaki, N.; Shiro, Y.; Iizuka, T.; Imai, K.; Hori, H. Novel ligand binding properties of the myoglobin substituted with monoazahemin. J. Biol. Chem. 1995, 270, 13118–13123. [Google Scholar] [CrossRef] [PubMed]
- Neya, S.; Hori, H.; Imai, K.; Kawamura-Konishi, Y.; Suzuki, H.; Shiro, Y.; Iizuka, T.; Funasaki, N. Remarkable functional aspects of myoglobin induced by diazaheme prosthetic group. J. Biochem. 1997, 121, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Dejima, H.; Matsuo, T.; Sato, H.; Murata, D.; Hisaeda, Y. Blue myoglobin reconstituted with an iron porphycene shows extremely high oxygen affinity. J. Am. Chem. Soc. 2002, 124, 11226–11227. [Google Scholar] [CrossRef] [PubMed]
- Neya, S.; Imai, K.; Hori, H.; Ishikawa, H.; Ishimori, K.; Okuno, D.; Nagatomo, S.; Hoshino, T.; Hata, M.; Funasaki, N. Iron hemiporphycene as a functional prosthetic group for myoglobin. Inorg. Chem. 2003, 42, 1456–1461. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, T.; Dejima, H.; Hirota, S.; Murata, D.; Sato, H.; Ikegami, T.; Hori, H.; Hisaeda, Y.; Hayashi, T. Ligand binding properties of myoglobin reconstituted with iron porphycene: Unusual O2 binding selectivity against CO binding. J. Am. Chem. Soc. 2004, 126, 16007–16017. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Murata, D.; Makino, M.; Sugimoto, H.; Matsuo, T.; Sato, H.; Shiro, Y.; Hisaeda, Y. Crystal structure and peroxidase activity of myoglobin reconstituted with iron porphycene. Inorg. Chem. 2006, 45, 10530–10536. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-z.; Taniguchi, I.; Mulchandani, A. Redox properties of engineered ruthenium myoglobin. Bioelectrochemistry 2009, 75, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Heinecke, J.L.; Yi, J.; Pereira, J.C.M.; Richter-Addo, G.B.; Ford, P.C. Nitrite reduction by CoII and MnII substituted myoglobins. Towards understanding necessary components of Mb nitrite reductase activity. J. Inorg. Biochem. 2012, 107, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Oohora, K.; Kihira, Y.; Mizohata, E.; Inoue, T.; Hayashi, T. C(sp3)–H bond hydroxylation catalyzed by myoglobin reconstituted with manganese porphycene. J. Am. Chem. Soc. 2013, 135, 17282–17285. [Google Scholar] [CrossRef] [PubMed]
- Stone, K.L.; Hua, J.; Choudhry, H. Manganese-substituted myoglobin: Characterization and reactivity of an oxidizing intermediate towards a weak C–H bond. Inorganics 2015, 3, 219–229. [Google Scholar] [CrossRef]
- Dolphin, D. The Porphyrins: Structure and Synthesis; Academic Press: New York, NY, USA, 1978; Volume 2. [Google Scholar]
- Bordeaux, M.; Singh, R.; Fasan, R. Intramolecular C(sp3)–H amination of arylsulfonyl azides with engineered and artificial myoglobin-based catalysts. Bioorg. Med. Chem. 2014, 22, 5697–5704. [Google Scholar] [CrossRef] [PubMed]
- Hori, H. Light absorption, electron paramagnetic resonance and resonance raman characteristics of nitridochromium(V) protoporphyrin-IX and its reconstituted hemoproteins. Biochim. Biophys. Acta 1991, 1077, 392–399. [Google Scholar] [CrossRef]
- Zahran, Z.N.; Chooback, L.; Copeland, D.M.; West, A.H.; Richter-Addo, G.B. Crystal structures of manganese- and cobalt-substituted myoglobin in complex with NO and nitrite reveal unusual ligand conformations. J. Inorg. Biochem. 2008, 102, 216–233. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, S.-I.; Matsui, T.; Watanabe, Y. Conversion of myoglobin into a highly stereo-specific peroxygenase by the L29H/H64L mutation. J. Am. Chem. Soc. 1996, 118, 9784–9785. [Google Scholar] [CrossRef]
- Matsui, T.; Ozaki, S.-I.; Watanabe, Y. On the formation and reactivity of compund I of the His-64 myoglobin mutants. J. Biol. Chem. 1997, 272, 32735–32738. [Google Scholar] [CrossRef] [PubMed]
- Hara, I.; Ueno, T.; Shin-ichi, O.; Itoh, S.; Lee, K.; Ueyama, N.; Watanabe, Y. Oxidative modification of tryptophan 43 in the heme vicinity of the F43W/H64L myoglobin mutant. J. Biol. Chem. 2001, 276, 36067–36070. [Google Scholar] [CrossRef] [PubMed]
- Pfister, T.D.; Ohki, T.; Ueno, T.; Hara, I.; Adachi, S.; Makino, Y.; Ueyama, N.; Lu, Y.; Watanabe, Y. Monooxygenation of an aromatic ring by F43W/H64D/V68I myoglobin mutant and hydrogen peroxide. J. Biol. Chem. 2005, 280, 12858–12866. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Nakajima, H.; Ueno, T. Reactivities of oxo and peroxo intermediates studied by hemoproteins mutants. Acc. Chem. Res. 2007, 40, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Urayama, P.; Phillips, G.N., Jr.; Gruner, S.M. Probing substates in sperm whale myoglobin using high-pressure crystallography. Structure 2002, 10, 51–60. [Google Scholar] [CrossRef]
- Du, J.; Huang, X.; Sun, S.; Wang, C.; Lebioda, L.; Dawson, J.H. Amphitrite ornata dehaloperoxidase (DHP): Investigations of structural factors that influence the mechanism of halophenol dehalogenation using “peroxidase-like” myoglobin mutants and “myoglobin-like” DHP mutants. Biochemistry 2011, 50, 8172–8180. [Google Scholar] [CrossRef] [PubMed]
- Sigman, J.A.; Kwok, B.C.; Lu, Y. From myoglobin to heme-copper oxidase: Design and engineering of a CuB center into sperm whale myoglobin. J. Am. Chem. Soc. 2000, 122, 8192–8196. [Google Scholar] [CrossRef]
- Zhao, X.; Yeung, N.; Wang, Z.; Guo, Z.; Lu, Y. Effects of metal ions in the CuB center on the redox properties of heme in heme-copper oxidases: Spectroelectrochemical studies of an engineered heme-copper center in myoglobin. Biochemistry 2005, 44, 1210–1214. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Yeung, N.; Russell, B.S.; Garner, D.K.; Lu, Y. Catalytic reduction of NO to N2O by a designed heme copper center in myoglobin: Implications for the role of metal ions. J. Am. Chem. Soc. 2006, 128, 6766–6767. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Cui, C.; Liu, X.; Petrik, I.D.; Wang, J.; Lu, Y. A designed metalloenzyme achieving the catalytic rate of a native enzyme. J. Am. Chem. Soc. 2015, 137, 11570–11573. [Google Scholar] [CrossRef] [PubMed]
- Bordeaux, M.; Tyagi, V.; Fasan, R. Highly diastereoselective and enantioselective olefin cyclopropanation using engineered myoglobin-based catalysts. Angew. Chem. Int. Ed. 2015, 54, 1744–1748. [Google Scholar] [CrossRef] [PubMed]
- Giovani, S.; Singh, R.; Fasan, R. Efficient conversion of primary azides to aldehydes catalyzed by active site variants of myoglobin. Chem. Sci. 2016, 7, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Sreenilayam, G.; Fasan, R. Myoglobin-catalyzed intermolecular carbene N–H insertion with arylamine substrates. Chem. Commun. 2015, 51, 1532–1534. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, V.; Bonn, R.B.; Fasan, R. Intermolecular carbene S–H insertion catalysed by engineered myoglobin-based catalysts. Chem. Sci. 2015, 6, 2488–2494. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, V.; Fasan, R. Myoglobin-catalyzed olefination of aldehydes. Angew. Chem. Int. Ed. 2016, 55, 2512–2516. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, S.-I.; Ortiz de Montellano, P.R. Molecular engineering of horseradish peroxidase: Thioether sulfoxidation and styrene epoxidation by Phe-141 leucine and threonine mutants. J. Am. Chem. Soc. 1995, 117, 7056–7064. [Google Scholar] [CrossRef]
- Gupta, K.C.; Sutar, A.K. Catalytic activities of Schiff base transition metal complexes. Coord. Chem. Rev. 2008, 252, 1420–1450. [Google Scholar] [CrossRef]
- Cozzi, P.G. Metal-salen Schiff base complexes in catalysis: Practical aspects. Chem. Soc. Rev. 2004, 33, 410–421. [Google Scholar] [CrossRef] [PubMed]
- Bois, J.D.; Tomooka, C.S.; Hong, J.; Carreira, E.M. Nitridomanganese(V) complexes: Design, preparation, and use as nitrogen atom-transfer reagents. Acc. Chem. Res. 1997, 30, 364–372. [Google Scholar] [CrossRef]
- Sun, W.; Herdtweck, E.; Kuhn, F.E. Catalytic aziridinations with copper(I) salen complexes. New J. Chem. 2005, 29, 1577–1580. [Google Scholar] [CrossRef]
- Wang, C.; Kurahashi, T.; Inomata, K.; Hada, M.; Fujii, H. Oxygen-atom transfer from iodosylarene adducts of a manganese(IV) salen complex: Effect of arenes and anions on I(III) of the coordinated iodosylarene. Inorg. Chem. 2013, 52, 9557–9566. [Google Scholar] [CrossRef] [PubMed]
- Coulter, E.D.; Sono, M.; Chang, C.K.; Lopez, O.; Dawson, J.H. Electron paramagnetic resonance spectroscopy as a probe of coordination structure in green heme systems: Iron chlorins and iron formylporphyrins reconstituted into myoglobin. Inorg. Chim. Acta 1995, 240, 603–608. [Google Scholar] [CrossRef]
- Sotiriou-Leventis, C.; Chang, C.K. Kinetic study of CO and O2 binding to horse heart myoglobin reconstituted with synthetic iron chlorin green hemes. Inorg. Chim. Acta 2000, 311, 113–118. [Google Scholar] [CrossRef]
- Matsuo, T.; Hayashi, A.; Abe, M.; Matsuda, T.; Hisaeda, Y.; Hayashi, T. Meso-unsubstituted iron corrole in hemoproteins: Remarkable differences in effects on peroxidase activities between myoglobin and horseradish peroxidase. J. Am. Chem. Soc. 2009, 131, 15124–15125. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.H.; Busch, D.H. Complexes derived from strong field ligands. XIX. Magnetic properties of transition metal derivatives of 4,4′,4″,4′′′-tetrasulfophthalocyanine. Inorg. Chem. 1965, 4, 469–471. [Google Scholar] [CrossRef]
- Dolotova, O.; Yuzhakova, O.; Solovyova, L.; Shevchenko, E.; Negrimovsky, V.; Lukyanets, E.; Kaliya, O. Water-soluble manganese phthalocyanines. J. Porphyr. Phthalocya. 2013, 17, 881–888. [Google Scholar] [CrossRef]
- Matsuo, T.; Tsuruta, T.; Maehara, K.; Sato, H.; Hisaeda, Y.; Hayashi, T. Preparation and O2 binding study of myoglobin having a cobalt porphycene. Inorg. Chem. 2005, 44, 9391–9396. [Google Scholar] [CrossRef] [PubMed]
- Ortiz de Montellano, P.R. Cytochrome P450: Structure, Mechanism, and Biochemistry; Springer: New York, NY, USA, 1995; Volume 2. [Google Scholar]
- Davydov, R.; Makris, T.M.; Kofman, V.; Werst, D.E.; Sligar, S.G.; Hoffman, B.M. Hydroxylation of camphor by reduced oxy-cytochrome P450cam: Mechanistic implications of EPR and ENDOR studies of catalytic intermediates in native and mutant enzymes. J. Am. Chem. Soc. 2001, 123, 1403–1415. [Google Scholar] [CrossRef] [PubMed]
- Shaik, S.; Kumar, D.; Visser, S.P.D.; Altun, A.; Thiel, W. Theoretical perspective on the structure and mechanism of cytochrome P450 enzymes. Chem. Rev. 2005, 105, 2279–2328. [Google Scholar] [CrossRef] [PubMed]
- Gelb, M.H.; William, A.; Toscano, J.; Sligar, S.G. Chemical mechanisms for cytochrome P-450 oxidation: Spectral and catalytic properties of a manganese-substituted protein. Proc. Natl. Acad. Sci. USA 1982, 79, 5758–5762. [Google Scholar] [CrossRef] [PubMed]
- Schlichting, I.; Berendzen, J.; Chu, K.; Stock, A.M.; Maves, S.A.; Benson, D.E.; Sweet, R.M.; Ringe, D.; Petsko, G.A.; Sligar, S.G. The catalytic pathway of cytochrome P450cam at atomic resolution. Science 2000, 287, 1615–1622. [Google Scholar] [CrossRef] [PubMed]
- Baylon, J.L.; Lenov, I.L.; Sligar, S.G.; Tajkhorshid, E. Characterizing the membrane-bound state of cytochrome P450 3A4: Structure, depth of insertion, and orientation. J. Am. Chem. Soc. 2013, 135, 8542–8551. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).