Fe Species Intercalation Confined by the Interlayer Environment of V2CTx MXene for Lithium-Ion Storage
Abstract
1. Introduction
2. Results and Discussion
2.1. Microstructure and Morphology
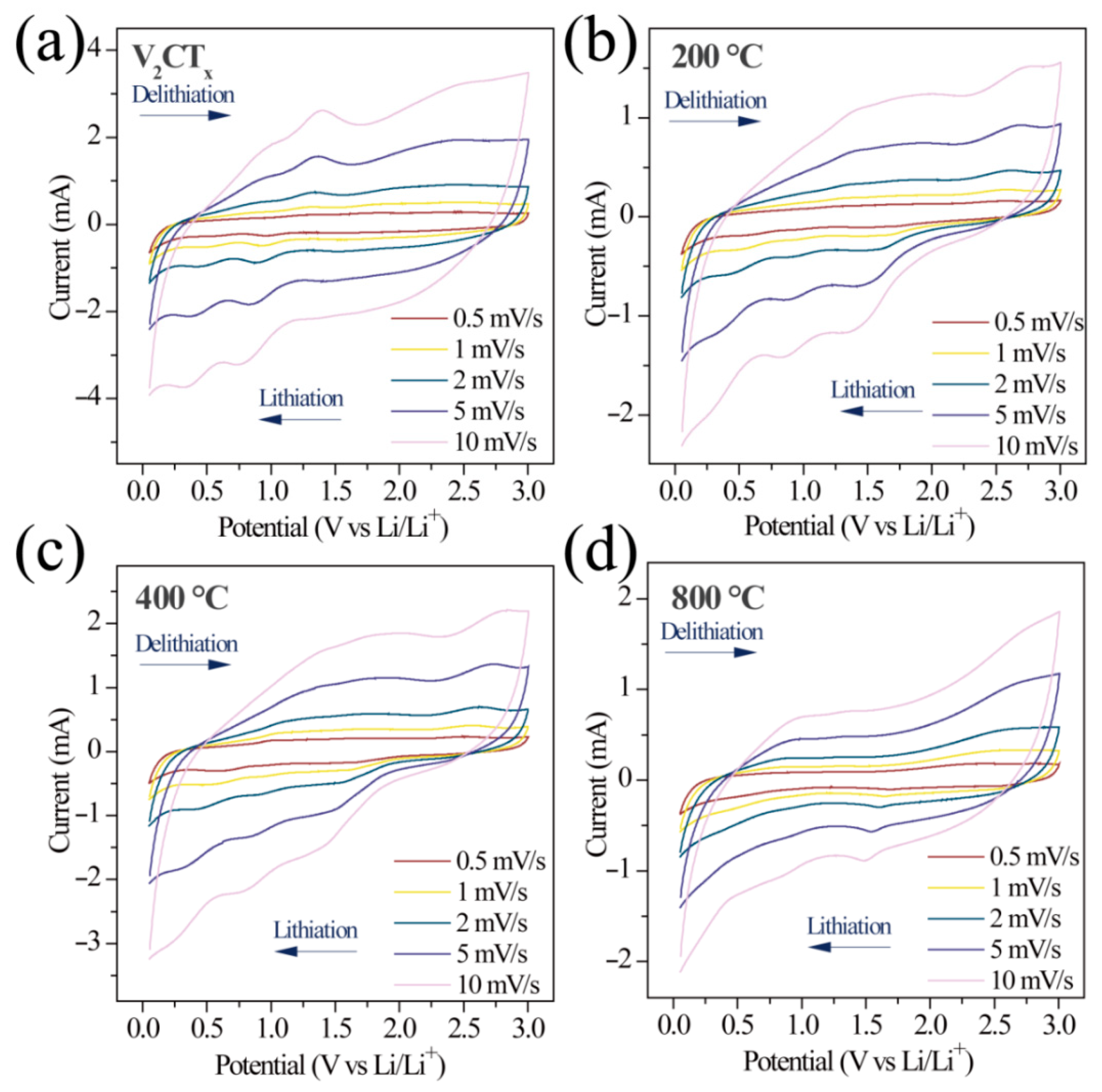
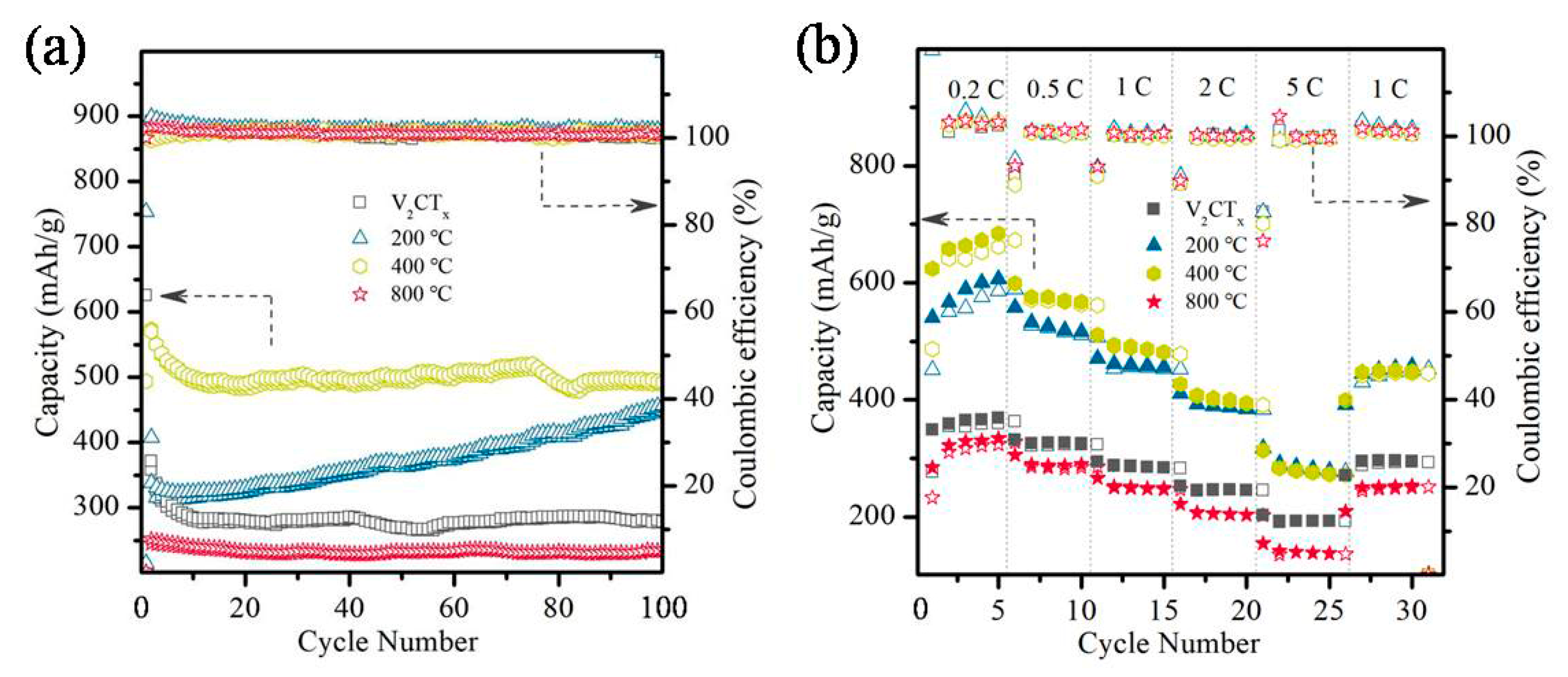
2.2. Electrochemical Properties
3. Experimental Section
3.1. Reagents and Materials
3.2. Preparation of V2CTx MXene
3.3. Preparation of V2CTx MXene Pre-Intercalated with Fe Species with Different Annealing Temperatures
3.4. Microstructural Characterization
3.5. Electrochemical Measurements
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ryu, H.H.; Park, N.Y.; Seo, J.H.; Yu, Y.S.; Sharma, M.; Mücke, R.; Kaghazchi, P.; Yoon, C.S.; Sun, Y.K. A highly stabilized Ni-rich NCA cathode for high-energy lithium-ion batteries. Mater. Today 2020, 36, 73–82. [Google Scholar] [CrossRef]
- Ming, F.-W.; Liang, H.-F.; Zhang, W.-L.; Ming, J.; Lei, Y.-J.; Emwas, A.H.; Alshareef, H.N. Porous MXenes enable high performance potassium ion capacitors. Nano Energy 2019, 62, 853–860. [Google Scholar] [CrossRef]
- Zhang, B.; Zhu, J.-F.; Shi, P.; Wu, W.-L.; Wang, F. Fluoride-free synthesis and microstructure evolution of novel two-dimensional Ti3C2(OH)2 nanoribbons as high-performance anode materials for lithium-ion batteries. Ceram. Int. 2019, 45, 8395–8405. [Google Scholar] [CrossRef]
- Ji, H.-S.; Xu, X.-H.; Li, X.; Li, K.-F.; Yuan, L.; Han, Z.-W.; Tang, K. A low-cost Si@C composite for lithium-ion batteries anode materials synthesized via freeze-drying process using kerf loss Si waste. Ionics 2024, 30, 2585–2599. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Katsnelson, M.I.; Grigorieva, I.V.; Dubonos, S.V.; Firsov, A.A. Two-dimensional gas of massless Dirac fermions in graphene. Nature 2005, 438, 197–200. [Google Scholar] [CrossRef]
- Lin, L.-X.; Sherrell, P.; Liu, Y.-Q.; Lei, W.; Zhang, S.-W.; Zhang, H.-J.; Wallace, G.G.; Chen, J. Engineered 2D Transition Metal Dichalcogenides-A Vision of Viable Hydrogen Evolution Reaction Catalysis. Adv. Energy Mater. 2020, 10, 1903870. [Google Scholar] [CrossRef]
- Pramanik, A.; Mahapatra, P.L.; Tromer, R.; Xu, J.-N.; Costin, G.; Li, C.-X.; Saju, S.; Alhashim, S.; Pandey, K.; Srivastava, A.; et al. Biotene: Earth-Abundant 2D Material as Sustainable Anode for Li/Na-ion Battery. ACS Appl. Mater. Interfaces 2024, 16, 2417–2427. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.-L.; Fang, Z.-W.; Zhu, Y.; Yan, C.-S.; Yu, G.-H. Holey 2D Nanomaterials for Electrochemical Energy Storage. Adv. Energy Mater. 2018, 8, 1702179. [Google Scholar] [CrossRef]
- Susmi, A.T.; Jayesh, C. Recent advances in synthesis and properties of zirconium-based MXenes for application in rechargeable batteries. Energy Storage 2023, 5, e475. [Google Scholar]
- Zhang, J.; Jia, R.; Tan, K.B.; Li, J.-M.; Xu, S.-C.; Ying, G.-B.; Han, W.-J.; Lu, M. A Review of MAX Series Materials: From Diversity, Synthesis, Prediction, Properties Oriented to Functions. Nano-Micro Lett. 2025, 17, 173. [Google Scholar] [CrossRef]
- Pang, J.-B.; Mendes, R.G.; Bachmatiuk, A.; Zhao, L.; Ta, H.Q.; Gemming, T.; Liu, H.; Liu, Z.-F.; Rummeli, M.H. Applications of 2D MXenes in energy conversion and storage systems. Chem. Soc. Rev. 2019, 48, 72–133. [Google Scholar] [CrossRef]
- Xu, X.-D.; Zhang, Y.-L.; Sun, H.-Y.; Zhou, J.-W.; Yang, F.; Li, H.; Chen, H.; Chen, Y.-C.; Liu, Z.; Qiu, Z.-P.; et al. Progress and Perspective: MXene and MXene-based Nanomaterials for High-performance Energy Storage Devices. Adv. Electron. Mater. 2021, 7, 2000967. [Google Scholar] [CrossRef]
- Pang, S.-Y.; Wong, Y.-T.; Yuan, S.-G.; Liu, Y.; Tsang, M.K.; Yang, Z.-B.; Huang, H.-T.; Wong, W.T.; Hao, J.-H. Universal Strategy for HF-Free Facile and Rapid Synthesis of Two-Dimensional MXenes as Multifunctional Energy Materials. J. Am. Chem. Soc. 2019, 141, 9610–9616. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.-X.; Du, M.-D.; Li, Y.-N.; Du, S.-J.; Zhao, L.-X.; Zheng, S.-R.; Zhang, J.; Li, H.-B.; Qiao, L.; Tan, K.B.; et al. Solidify Eutectic Electrolytes via the Added MXene as Nucleation Sites for a Solid-State Zinc-Ion Battery with Reconstructed Ion Transport. Nano Lett. 2024, 24, 8818–8825. [Google Scholar] [CrossRef] [PubMed]
- Naguib, M.; Mochalin, V.N.; Barsoum, M.W.; Gogotsi, Y. 25th Anniversary Article: MXenes: A New Family of Two-Dimensional Materials. Adv. Mater. 2014, 26, 992–1005. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.-M.; Qin, J.-D.; Zhu, Y.-X.; He, K.-L.; Chen, H.; Hoh, H.Y.; Batmunkh, M.; Benedetti, T.M.; Zhang, Q.-T.; Su, C.-L.; et al. Green and scalable electrochemical routes for cost-effective mass production of MXenes for supercapacitor electrodes. Carbon Energy 2023, 5, e295. [Google Scholar] [CrossRef]
- Zhang, T.; Shevchuk, K.; Wang, R.J.; Kim, H.; Hourani, J.; Gogotsi, Y. Delamination of Chlorine-Terminated MXene Produced Using Molten Salt Etching. Chem. Mater. 2024, 36, 1998–2006. [Google Scholar] [CrossRef]
- Fan, Y.-X.; Li, L.; Zhang, Y.; Zhang, X.-T.; Geng, D.-C.; Hu, W.-P. Recent Advances in Growth of Transition Metal Carbides and Nitrides (MXenes) Crystals. Adv. Funct. Mater. 2022, 32, 2111357. [Google Scholar] [CrossRef]
- Li, T.-F.; Yao, L.-L.; Liu, Q.-L.; Gu, J.-J.; Luo, R.-C.; Li, J.-H.; Yan, X.-D.; Wang, W.-Q.; Liu, P.; Chen, B.; et al. Fluorine-Free Synthesis of High-Purity Ti3C2Tx (T = OH, O) via Alkali Treatment. Angew. Chem. Int. Edit. 2018, 57, 6115–6119. [Google Scholar] [CrossRef]
- Anasri, B.; Gogotsi, Y. MXenes: Trends, growth, and future directions. Graphene 2D Mater. 2022, 7, 75–79. [Google Scholar] [CrossRef]
- Sun, Y.-J.; Chen, D.-S.; Liang, Z.-Q. Two-dimensional MXenes for energy storage and conversion applications. Mater. Today Energy 2017, 5, 22–36. [Google Scholar] [CrossRef]
- Chen, X.-F.; Zhu, Y.-Z.; Zhang, M.; Sui, J.-Y.; Peng, W.-C.; Li, Y.; Zhang, G.-L.; Zhang, F.-B.; Fan, X.-B. N-Butyllithium-Treated Ti3C2Tx MXene with Excellent Pseudocapacitor Performance. ACS Nano 2019, 13, 9449–9456. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Zhang, B.-S.; Zhang, W.; Zheng, W.-T. Breaking the lithium storage limit via independent bilayer units within 2D layer materials. J. Energy Chem. 2020, 41, 1–2. [Google Scholar] [CrossRef]
- Sun, N.; Yang, B.-Y.; Zheng, J.-C.; He, Z.-J.; Tong, H.; Tang, L.-B.; An, C.-S.; Xiao, B. Effect of synthesis temperature on the phase structure, morphology and electrochemical performance of Ti3C2 as an anode material for Li-ion batteries. Ceram. Int. 2018, 44, 16214–16218. [Google Scholar] [CrossRef]
- Cui, Y.; Xie, X.; Yang, R.; Qin, J.-W.; Zheng, L.-R.; Cao, M.-H. Cold pressing-built microreactors to thermally manipulate microstructure of MXene film as an anode for high-performance lithium-ion batteries. Electrochim. Acta 2019, 305, 11–23. [Google Scholar] [CrossRef]
- Li, X.-L.; Li, M.; Yang, Q.; Li, H.-F.; Xu, H.-L.; Chai, Z.-F.; Chen, K.; Liu, Z.-X.; Tang, Z.-J.; Ma, L.-T.; et al. Phase transition induced unusual electrochemical performance of V2CTx MXene for aqueous zinc hybrid-ion battery. ACS Nano 2020, 1, 541–551. [Google Scholar] [CrossRef]
- Tang, Q.; Zhou, Z.; Shen, P.-W. Are MXenes Promising Anode Materials for Li Ion Batteries? Computational Studies on Electronic Properties and Li Storage Capability of Ti3C2 and Ti3C2X2 (X=F, OH) Monolayer. J. Am. Chem. Soc. 2012, 134, 16909–16916. [Google Scholar] [CrossRef]
- Luo, J.-M.; Tao, X.-Y.; Zhang, J.; Xia, Y.; Huang, H.; Zhang, L.-Y.; Gan, Y.-P.; Liang, C.; Zhang, W.-K. Sn4+ Ion Decorated Highly Conductive Ti3C2 MXene: Promising Lithium-Ion Anodes with Enhanced Volumetric Capacity and Cyclic Performance. ACS Nano 2016, 10, 2491–2499. [Google Scholar] [CrossRef]
- Zhang, Y.-Q.; Guo, B.-S.; Hu, L.-Y.; Xu, Q.-J.; Li, Y.; Liu, D.-Y.; Xu, M.-W. Synthesis of SnS nanoparticle-modified MXene (Ti3C2Tx) composites for enhanced sodium storage. J. Alloys Compd. 2018, 732, 448–453. [Google Scholar] [CrossRef]
- Duan, X.; Lv, F.-S.; Liu, J.-Q.; Liu, T.; Yuan, S. Synergistic effect of MXene and Co3O4 nanoneedles: Constructing high-performance lithium-ion capacitor anodes. J. Solid State Chem. 2025, 350, 125519. [Google Scholar] [CrossRef]
- Wang, Z.-T.; Wang, R.-C.; Tang, L.-B.; Li, Y.-J.; Mao, J.; Dai, K.-H.; He, Z.-J.; Zheng, J.-C. A sandwich-like Ti3C2@VO2 composite synthesized by a hydrothermal method for lithium storage. Solid State Ionics 2021, 369, 115714. [Google Scholar] [CrossRef]
- Lu, M.; Zhang, Y.-P.; Chen, J.-N.; Han, W.-J.; Zhang, W.; Li, H.-B.; Zhang, X.; Zhang, B.-S. K+ alkalization promoted Ca2+ intercalation in V2CTx MXene for enhanced Li storage. J. Energy Chem. 2020, 49, 358–364. [Google Scholar] [CrossRef]
- VahidMohammadi, A.; Mojtabavi, M.; Caffrey, N.M.; Wanunu, M.; Beidaghi, M. Assembling 2D MXenes into Highly Stable Pseudocapacitive Electrodes with High Power and Energy Densities. Adv. Mater. 2019, 31, 1806931. [Google Scholar] [CrossRef]
- Anasori, B.; Lukatskaya, M.R.; Gogotsi, Y. 2D metal carbides and nitrides (MXenes) for energy storage. Nat. Rev. Mater. 2017, 2, 16098. [Google Scholar] [CrossRef]
- Hu, J.-P.; Xu, B.; Ouyang, C.-Y.; Yang, S.-Y.-A.; Yao, Y.-G. Investigations on V2C and V2CX2 (X=F, OH) Monolayer as a Promising Anode Material for Li Ion Batteries from First-Principles Calculations. J. Phys. Chem. C 2014, 118, 24274–24281. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, L.-B.; Shen, C.-J.; Qin, G.; Hu, Q.-K.; Zhou, A.-G. Synthesis of NaV6O15 nanorods via thermal oxidation of sodium-intercalated 2D V2CTx and their electrochemical properties as anode for lithium-ion batteries. Electrochim. Acta 2017, 248, 178–187. [Google Scholar] [CrossRef]
- Liu, M.; Qu, X.; Wang, Y.-X.; Zhang, Y.-K.; Zhao, C.-M.; Liu, B.; Xue, X.-X.; Zhang, J.-K.; Wang, Z.; Li, J.-M.; et al. Inspired by philosophizing of “point to area”: Ion pre-intercalation induces the reconstitution of interlayer environment of MXene for lithium storage. Chem. Eng. J. 2023, 451, 139015. [Google Scholar] [CrossRef]
- Persson, I.; Näslund, L.; Halim, J.; Barsoum, M.; Darakchieva, V.; Palisaitis, J.; Rosen, J.; Persson, P. On the organization and thermal behavior of functional groups on Ti3C2 MXene surfaces in vacuum. 2D Mater. 2018, 5, 015002–051035. [Google Scholar] [CrossRef]
- Zhao, X.-F.; Holta, D.E.; Tan, Z.-Y.; Oh, J.H.; Echols, I.J.; Anas, M.; Cao, H.-X.; Lutkenhaus, J.L.; Radovic, M.; Green, M.J. Annealed Ti3C2Tz MXene films for oxidation resistant functional coatings. ACS Appl. Nano Mater. 2020, 3, 10578–10585. [Google Scholar] [CrossRef]
- Kong, F.-Y.; He, X.-D.; Liu, Q.-Q.; Qi, X.-X.; Zheng, Y.-T.; Wang, R.-G.; Bai, Y.-L. Improving the electrochemical properties of MXene Ti3C2 multilayer for Li-ion batteries by vacuum calcination. Electrochim. Acta 2018, 265, 140–150. [Google Scholar] [CrossRef]
- Wang, C.-D.; Xie, H.; Chen, S.-M.; Ge, B.-H.; Liu, D.-B.; Wu, C.-Q.; Xu, W.-J.; Chu, W.-S.; Babu, G.; Ajayan, P.M.; et al. Atomic Cobalt Covalently Engineered Interlayers for Superior Lithium-Ion Storage. Adv. Mater. 2018, 30, 1802525. [Google Scholar] [CrossRef]
- Wang, C.-D.; Chen, S.-M.; Xie, H.; Wei, S.-Q.; Wu, C.-Q.; Song, L. Atomic Sn4+ Decorated into Vanadium Carbide MXene Interlayers for Superior Lithium Storage. Adv. Energy Mater. 2019, 9, 1802977. [Google Scholar] [CrossRef]
- Xie, Y.; Naguib, M.; Mochalin, V.; Barsoum, M.; Gogotsi, Y.; Yu, X.-Q.; Nam, K.; Yang, X.-Q.; Kolesnikov, A.; Kent, P. Role of surface structure on Li-ion energy storage capacity of two-dimensional transition-metal carbides. J. Am. Chem. Soc. 2014, 136, 6385–6394. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.-D.; Wang, M.-S.; Li, Z.-Y.; Fan, G.-X.; Fan, L.-Z.; Zhou, A.-G. Two-dimensional Ti3C2 as anode material for Liionbatteries. Electrochem. Commun. 2014, 47, 80–83. [Google Scholar] [CrossRef]
- Naguib, M.; Halim, J.; Lu, J.; Cook, K.M.; Hultman, L.; Gogotsi, Y.; Barsoum, M.W. New Two-Dimensional Niobium and Vanadium Carbides as Promising Materials for Li-Ion Batteries. J. Am. Chem. Soc. 2013, 135, 15966–15969. [Google Scholar] [CrossRef]
- Liu, F.; Zhou, J.; Wang, S.; Wang, B.; Zhou, A. Preparation of High-Purity V2C MXene and Electrochemical Properties as Li-Ion Batteries. J. Electrochem. Soc. 2017, 164, A709–A713. [Google Scholar] [CrossRef]
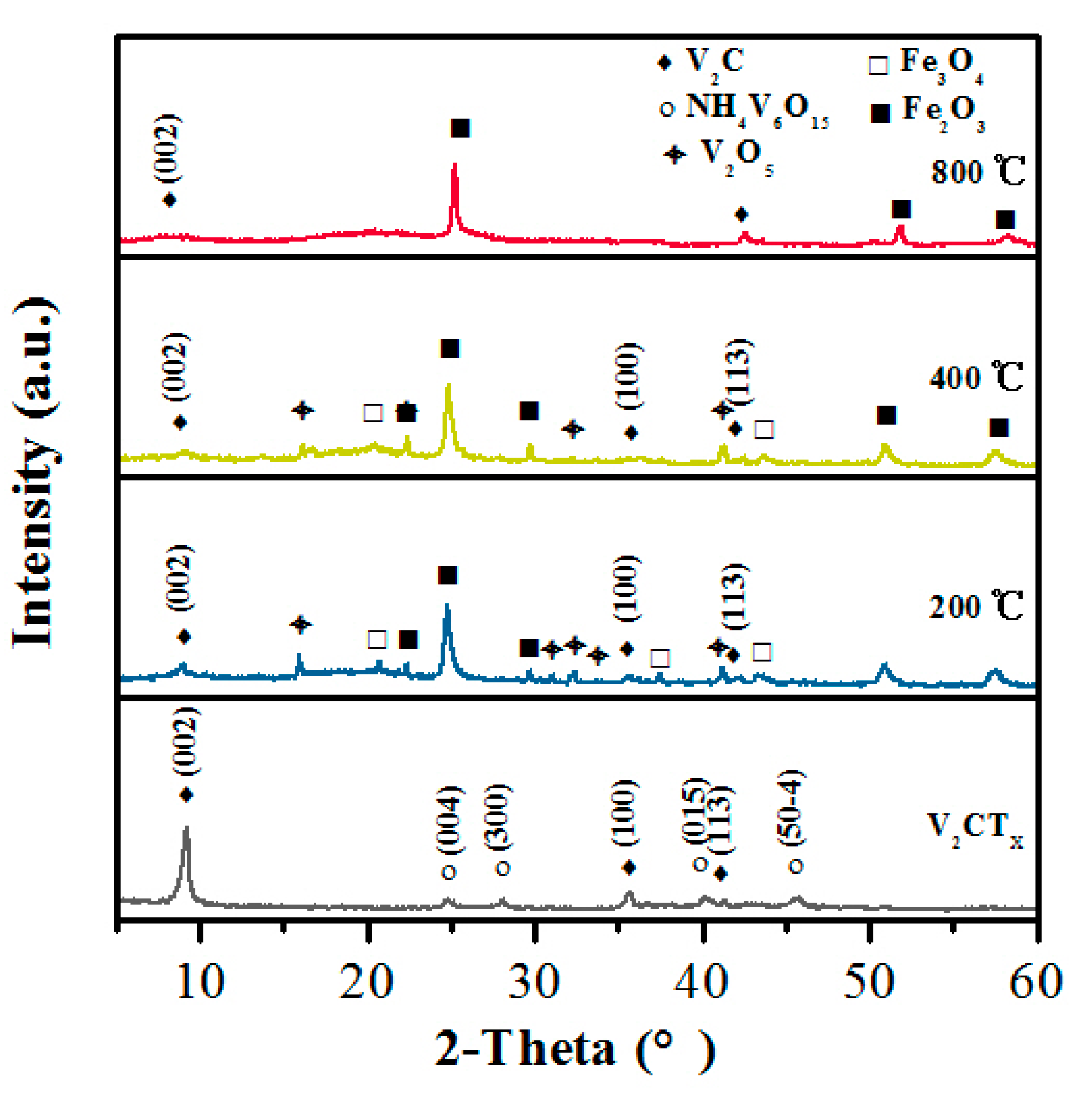



| (002) | (°) | Interlayer Spacing (Å) |
|---|---|---|
| V2CTx | 9.200 | 9.6044 |
| 200 °C | 9.007 | 9.8096 |
| 400 °C | 8.987 | 9.8314 |
| 800 °C | 8.135 | 10.8589 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.; Liu, M.; Li, J.; Han, W.; Xu, S.; Li, H.; Lu, M. Fe Species Intercalation Confined by the Interlayer Environment of V2CTx MXene for Lithium-Ion Storage. Inorganics 2025, 13, 290. https://doi.org/10.3390/inorganics13090290
Li J, Liu M, Li J, Han W, Xu S, Li H, Lu M. Fe Species Intercalation Confined by the Interlayer Environment of V2CTx MXene for Lithium-Ion Storage. Inorganics. 2025; 13(9):290. https://doi.org/10.3390/inorganics13090290
Chicago/Turabian StyleLi, Jiaxin, Miao Liu, Jiaming Li, Wenjuan Han, Shichong Xu, Haibo Li, and Ming Lu. 2025. "Fe Species Intercalation Confined by the Interlayer Environment of V2CTx MXene for Lithium-Ion Storage" Inorganics 13, no. 9: 290. https://doi.org/10.3390/inorganics13090290
APA StyleLi, J., Liu, M., Li, J., Han, W., Xu, S., Li, H., & Lu, M. (2025). Fe Species Intercalation Confined by the Interlayer Environment of V2CTx MXene for Lithium-Ion Storage. Inorganics, 13(9), 290. https://doi.org/10.3390/inorganics13090290






