Interfaces between Ceramic and Polymer Electrolytes: A Comparison of Oxide and Sulfide Solid Electrolytes for Hybrid Solid-State Batteries
Abstract
:1. Introduction
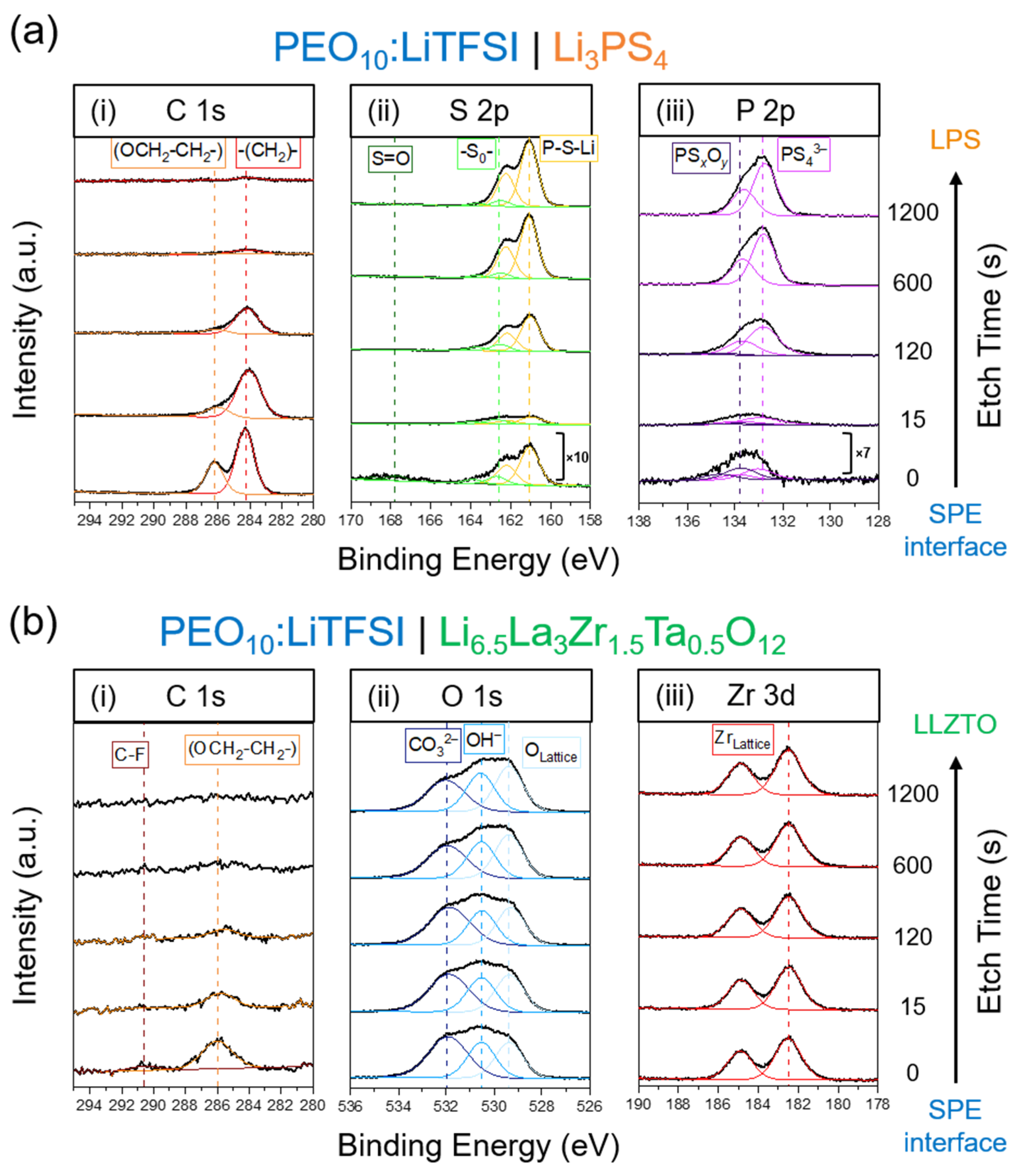
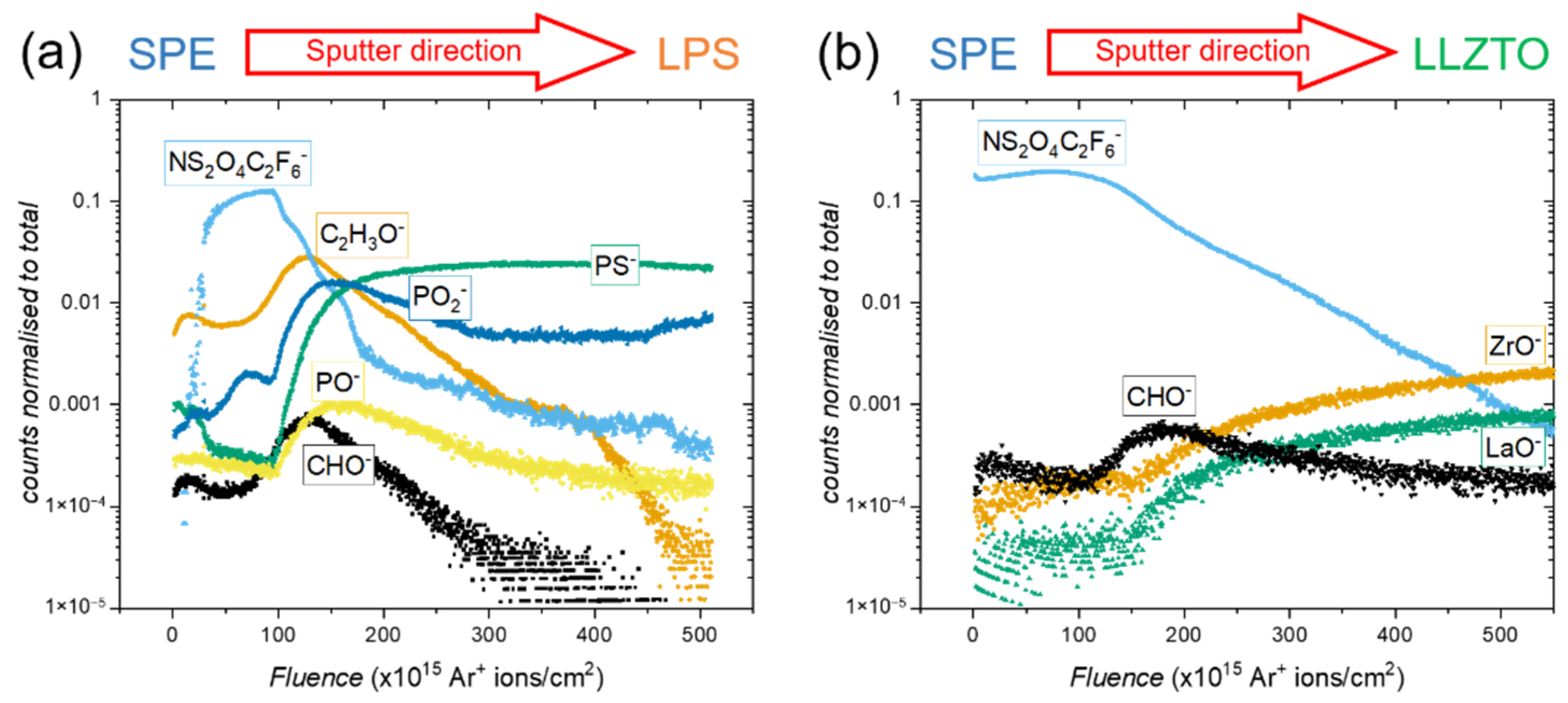
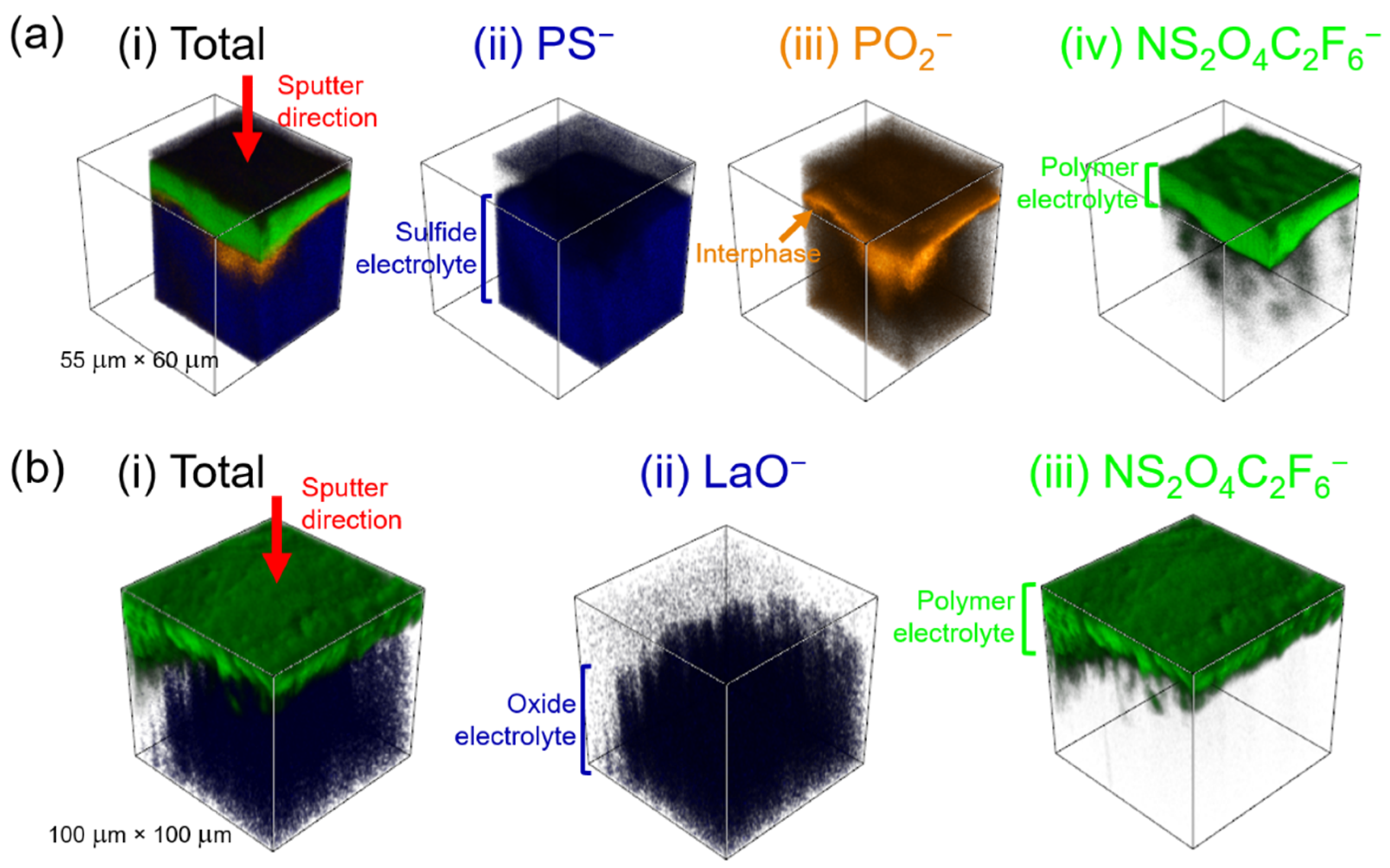
2. Results
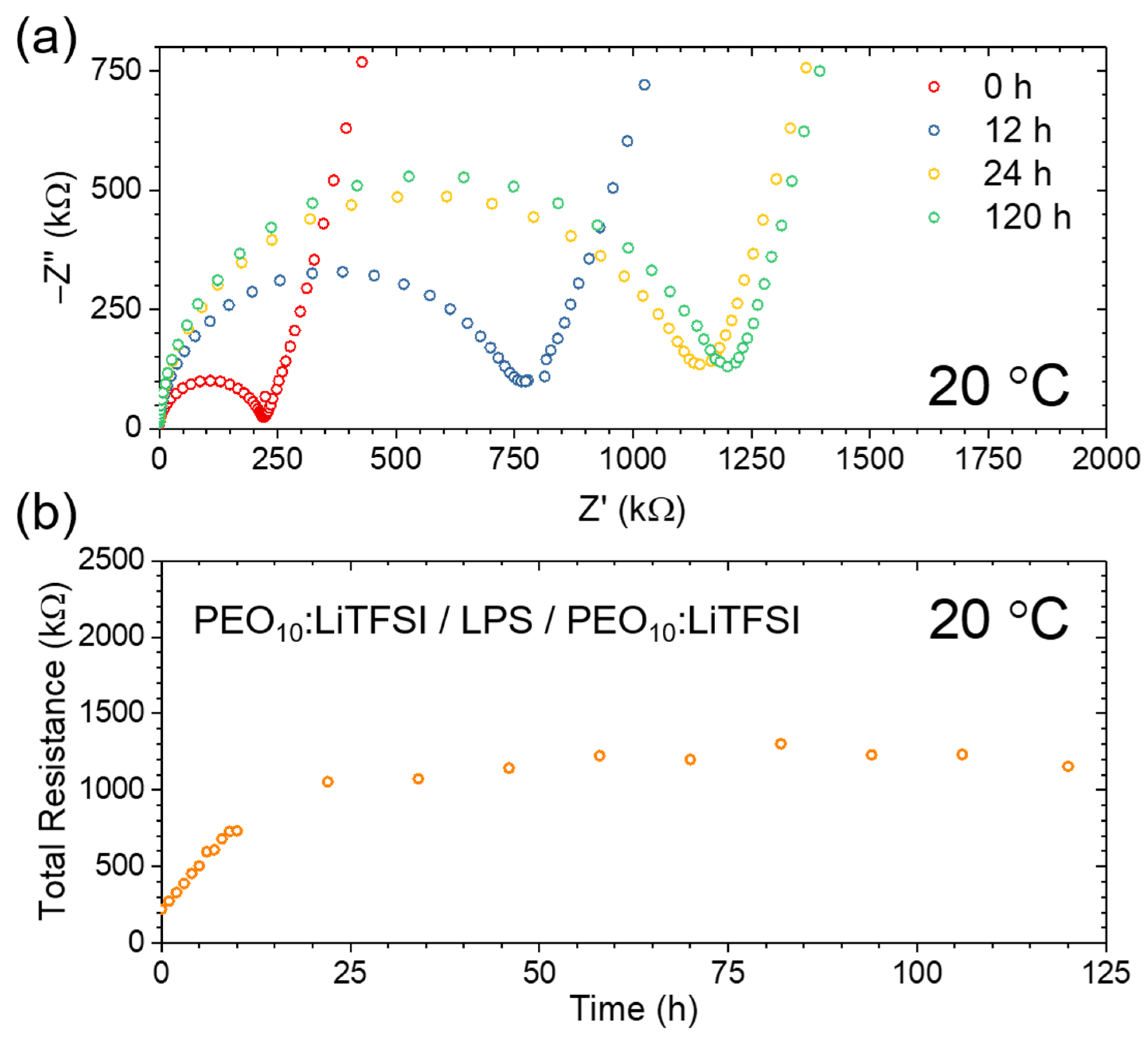
2.1. The LPS/PEO10:LiTFSI Interface
2.2. The LLZTO/PEO10:LiTFSI Interface
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Electrochemical Characterisation
4.3. X-ray Photoelectron Spectroscopy
4.4. Time-of-Flight Secondary Ion Mass Spectrometry
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Janek, J.; Zeier, W.G. A solid future for battery development. Nat. Energy 2016, 1, 16141. [Google Scholar] [CrossRef]
- Pasta, M.; Armstrong, D.; Brown, Z.L.; Bu, J.; Castell, M.R.; Chen, P.; Cocks, A.; Corr, S.A.; Cussen, E.J.; Darnbrough, E.; et al. 2020 roadmap on solid-state batteries. J. Phys. Energy 2020, 2, 032008. [Google Scholar] [CrossRef]
- Kerman, K.; Luntz, A.; Viswanathan, V.; Chiang, Y.-M.; Chen, Z. Review—Practical Challenges Hindering the Development of Solid State Li Ion Batteries. J. Electrochem. Soc. 2017, 164, A1731–A1744. [Google Scholar] [CrossRef]
- Ning, Z.; Jolly, D.S.; Li, G.; De Meyere, R.; Pu, S.D.; Chen, Y.; Kasemchainan, J.; Ihli, J.; Gong, C.; Liu, B.; et al. Visualizing plating-induced cracking in lithium-anode solid-electrolyte cells. Nat. Mater. 2021, 20, 1121–1130. [Google Scholar] [CrossRef] [PubMed]
- Jolly, D.S.; Ning, Z.; Hartley, G.O.; Liu, B.; Melvin, D.L.R.; Adamson, P.; Marrow, J.; Bruce, P.G. Temperature Dependence of Lithium Anode Voiding in Argyrodite Solid-State Batteries. ACS Appl. Mater. Interfaces 2021, 13, 22708–22716. [Google Scholar] [CrossRef] [PubMed]
- Albertus, P.; Anandan, V.; Ban, C.; Balsara, N.; Belharouak, I.; Buettner-Garrett, J.; Chen, Z.; Daniel, C.; Doeff, M.; Dudney, N.J.; et al. Challenges for and Pathways toward Li-Metal-Based All-Solid-State Batteries. ACS Energy Lett. 2021, 6, 1399–1404. [Google Scholar] [CrossRef]
- Xiao, Y.; Wang, Y.; Bo, S.-H.; Kim, J.C.; Miara, L.J.; Ceder, G. Understanding interface stability in solid-state batteries. Nat. Rev. Mater. 2020, 5, 105–126. [Google Scholar] [CrossRef]
- Zhu, Y.; He, X.; Mo, Y. First principles study on electrochemical and chemical stability of solid electrolyte–electrode interfaces in all-solid-state Li-ion batteries. J. Mater. Chem. A 2016, 4, 3253–3266. [Google Scholar] [CrossRef]
- Richards, W.D.; Miara, L.J.; Wang, Y.; Kim, J.C.; Ceder, G. Interface Stability in Solid-State Batteries. Chem. Mater. 2016, 28, 266–273. [Google Scholar] [CrossRef]
- Zhu, Y.; He, X.; Mo, Y. Origin of Outstanding Stability in the Lithium Solid Electrolyte Materials: Insights from Thermodynamic Analyses Based on First-Principles Calculations. ACS Appl. Mater. Interfaces 2015, 7, 23685–23693. [Google Scholar] [CrossRef]
- Miara, L.; Windmüller, A.; Tsai, C.-L.; Richards, W.D.; Ma, Q.; Uhlenbruck, S.; Guillon, O.; Ceder, G. About the Compatibility between High Voltage Spinel Cathode Materials and Solid Oxide Electrolytes as a Function of Temperature. ACS Appl. Mater. Interfaces 2016, 8, 26842–26850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dück, G.; Naqash, S.; Finsterbusch, M.; Breuer, U.; Guillon, O.; Fattakhova-Rohlfing, D. Co-Sintering Study of Na0.67[Ni0.1Fe0.1Mn0.8]O2 and NaSICON Electrolyte–Paving the way to High Energy Density All-Solid-State Batteries. Front. Energy Res. 2021, 9, 689416. [Google Scholar] [CrossRef]
- Ihrig, M.; Finsterbusch, M.; Tsai, C.-L.; Laptev, A.M.; Tu, C.-H.; Bram, M.; Sohn, Y.J.; Ye, R.; Sevinc, S.; Lin, S.-K.; et al. Low temperature sintering of fully inorganic all-solid-state batteries—Impact of interfaces on full cell performance. J. Power Sources 2021, 482, 228905. [Google Scholar] [CrossRef]
- Sen, S.; Trevisanello, E.; Niemöller, E.; Shi, B.-X.; Simon, F.J.; Richter, F.H. The role of polymers in lithium solid-state batteries with inorganic solid electrolytes. J. Mater. Chem. A 2021, 9, 18701–18732. [Google Scholar] [CrossRef]
- Liu, J.; Gao, X.; Hartley, G.O.; Rees, G.; Gong, C.; Richter, F.H.; Janek, J.; Xia, Y.; Robertson, A.W.; Johnson, L.R.; et al. The Interface between Li6.5La3Zr1.5Ta0.5O12 and Liquid Electrolyte. Joule 2020, 4, 101–108. [Google Scholar] [CrossRef] [Green Version]
- Busche, M.R.; Drossel, T.; Leichtweiss, T.; Weber, D.A.; Falk, M.; Schneider, M.; Reich, M.-L.; Sommer, H.; Adelhelm, P.; Janek, J. Dynamic formation of a solid-liquid electrolyte interphase and its consequences for hybrid-battery concepts. Nat. Chem. 2016, 8, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Abe, T.; Sagane, F.; Ohtsuka, M.; Iriyama, Y.; Ogumi, Z. Lithium-Ion Transfer at the Interface Between Lithium-Ion Conductive Ceramic Electrolyte and Liquid Electrolyte-A Key to Enhancing the Rate Capability of Lithium-Ion Batteries. J. Electrochem. Soc. 2005, 152, A2151. [Google Scholar] [CrossRef]
- Sagane, F.; Abe, T.; Ogumi, Z. Li+-ion transfer through the interface between Li+-ion conductive ceramic electrolyte and Li+-ion- concentrated propylene carbonate solution. J. Phys. Chem. C 2009, 113, 20135–20138. [Google Scholar] [CrossRef]
- Yamada, Y.; Sagane, F.; Iriyama, Y.; Abe, T.; Ogumi, Z. Kinetics of lithium-ion transfer at the interface between Li0.35La0.55TiO3 and binary electrolytes. J. Phys. Chem. C 2009, 113, 14528–14532. [Google Scholar] [CrossRef]
- Gupta, A.; Kazyak, E.; Dasgupta, N.P.; Sakamoto, J. Electrochemical and Surface Chemistry Analysis of Lithium Lanthanum Zirconium Tantalum Oxide (LLZTO)/Liquid Electrolyte (LE) Interfaces. J. Power Sources 2020, 474, 228598. [Google Scholar] [CrossRef]
- Abe, T.; Ohtsuka, M.; Sagane, F.; Iriyama, Y.; Ogumi, Z. Lithium Ion Transfer at the Interface between Lithium-Ion-Conductive Solid Crystalline Electrolyte and Polymer Electrolyte. J. Electrochem. Soc. 2004, 151, A1950. [Google Scholar] [CrossRef]
- Sagane, F.; Abe, T.; Iriyama, Y.; Ogumi, Z. Li+ and Na+ transfer through interfaces between inorganic solid electrolytes and polymer or liquid electrolytes. J. Power Sources 2005, 146, 749–752. [Google Scholar] [CrossRef]
- Tenhaeff, W.; Yu, X.; Hong, K.; Perry, K.A.; Dudney, N.J. Ionic Transport Across Interfaces of Solid Glass and Polymer Electrolytes for Lithium Ion Batteries. J. Electrochem. Soc. 2011, 158, A1143. [Google Scholar] [CrossRef]
- Chen, X.C.; Liu, X.; Pandian, A.S.; Lou, K.; Delnick, F.M.; Dudney, N.J. Determining and Minimizing Resistance for Ion Transport at the Polymer/Ceramic Electrolyte Interface. ACS Energy Lett. 2019, 4, 1080–1085. [Google Scholar] [CrossRef]
- Gupta, A.; Sakamoto, J. Controlling ionic transport through the PEO-LITFSi/LLZTO interface. Electrochem. Soc. Interface 2019, 28, 63–69. [Google Scholar] [CrossRef]
- Sagane, F.; Abe, T.; Ogumi, Z. Sodium-ion transfer at the interface between ceramic and organic electrolytes. J. Power Sources 2010, 195, 7466–7470. [Google Scholar] [CrossRef]
- Langer, F.; Palagonia, M.S.; Bardenhagen, I.; Glenneberg, J.; La Mantia, F.; Kun, R. Impedance Spectroscopy Analysis of the Lithium Ion Transport through the Li7La3Zr2O12/P(EO)20Li Interface. J. Electrochem. Soc. 2017, 164, A2298–A2303. [Google Scholar] [CrossRef]
- Tenhaeff, W.E.; Perry, K.A.; Dudney, N.J. Impedance Characterization of Li Ion Transport at the Interface between Laminated Ceramic and Polymeric Electrolytes. J. Electrochem. Soc. 2012, 159, A2118–A2123. [Google Scholar] [CrossRef]
- Brogioli, D.; Langer, F.; Kun, R.; La Mantia, F. Space-Charge Effects at the Li7La3Zr2O12/Poly(ethylene oxide) Interface. ACS Appl. Mater. Interfaces 2019, 11, 11999–12007. [Google Scholar] [CrossRef]
- Zhang, T.; Imanishi, N.; Hasegawa, S.; Hirano, A.; Xie, J.; Takeda, Y.; Yamamoto, O.; Sammes, N. Li/Polymer Electrolyte/Water Stable Lithium-Conducting Glass Ceramics Composite for Lithium–Air Secondary Batteries with an Aqueous Electrolyte. J. Electrochem. Soc. 2008, 155, A965. [Google Scholar] [CrossRef] [Green Version]
- Zhang, T.; Imanishi, N.; Hasegawa, S.; Hirano, A.; Xie, J.; Takeda, Y.; Yamamoto, O.; Sammes, N. Water-Stable Lithium Anode with the Three-Layer Construction for Aqueous Lithium–Air Secondary Batteries. Electrochem. Solid-State Lett. 2009, 12, 3–7. [Google Scholar] [CrossRef]
- Wang, H.; Im, D.; Lee, D.J.; Matsui, M.; Takeda, Y.; Yamamoto, O.; Imanishi, N. A Composite Polymer Electrolyte Protect Layer between Lithium and Water Stable Ceramics for Aqueous Lithium-Air Batteries. J. Electrochem. Soc. 2013, 160, A728–A733. [Google Scholar] [CrossRef]
- Liu, W.; Milcarek, R.J.; Falkenstein-Smith, R.L.; Ahn, J. Interfacial impedance studies of multilayer structured electrolyte fabricated with solvent-casted PEO10-LiN(CF3SO2)2 and Ceramic Li1.3Al0.3Ti1.7(PO4)3 and its application in all-solid-state lithium ion batteries. J. Electrochem. Energy Convers. Storage 2016, 13, 021008. [Google Scholar] [CrossRef]
- Riphaus, N.; Stiaszny, B.; Sedlmaier, S.J.; Beyer, H.; Gasteiger, H.A. Understanding Chemical Stability Issues between Different Solid Electrolytes in All-Solid-State Batteries. J. Electrochem. Soc. 2019, 166, A975–A983. [Google Scholar] [CrossRef]
- Simon, F.J.; Hanauer, M.; Henss, A.; Richter, F.H.; Janek, J. Properties of the Interphase Formed between Argyrodite-Type Li6PS5Cl and Polymer-Based PEO10:LiTFSI. ACS Appl. Mater. Interfaces 2019, 11, 42186–42196. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Fu, W.; Payzant, E.A.; Yu, X.; Wu, Z.; Dudney, N.J.; Kiggans, J.; Hong, K.; Rondinone, A.J.; Liang, C. Anomalous high ionic conductivity of nanoporous β-Li3PS4. J. Am. Chem. Soc. 2013, 135, 975–978. [Google Scholar] [CrossRef]
- Hayashi, A.; Hama, S.; Morimoto, H.; Tatsumisago, M.; Minami, T. Preparation of Li2S-P2S5 Amorphous Solid Electrolytes by Mechanical Milling. J. Am. Chem. Soc. 2001, 84, 477–479. [Google Scholar] [CrossRef]
- Phuc, N.H.H.; Totani, M.; Morikawa, K.; Muto, H.; Matsuda, A. Preparation of Li3PS4 solid electrolyte using ethyl acetate as synthetic medium. Solid State Ionics 2016, 288, 240–243. [Google Scholar] [CrossRef] [Green Version]
- Simon, F.J.; Hanauer, M.; Richter, F.H.; Janek, J. Interphase Formation of PEO20:LiTFSI-Li6PS5Cl Composite Electrolytes with Lithium Metal. ACS Appl. Mater. Interfaces 2020, 12, 11713–11723. [Google Scholar] [CrossRef]
- Xu, C.; Sun, B.; Gustafsson, T.; Edström, K.; Brandell, D.; Hahlin, M. Interface layer formation in solid polymer electrolyte lithium batteries: An XPS study. J. Mater. Chem. A 2014, 2, 7256–7264. [Google Scholar] [CrossRef]
- Auvergniot, J.; Cassel, A.; Foix, D.; Viallet, V.; Seznec, V.; Dedryvère, R. Redox activity of argyrodite Li6PS5Cl electrolyte in all-solid-state Li-ion battery: An XPS study. Solid State Ionics 2017, 300, 78–85. [Google Scholar] [CrossRef]
- Connell, J.G.; Fuchs, T.; Hartmann, H.; Krauskopf, T.; Zhu, Y.; Sann, J.; Garcia-Mendez, R.; Sakamoto, J.; Tepavcevic, S.; Janek, J. Kinetic versus Thermodynamic Stability of LLZO in Contact with Lithium Metal. Chem. Mater. 2020, 32, 10207–10215. [Google Scholar] [CrossRef]
- Allen, J.L.; Wolfenstine, J.; Rangasamy, E.; Sakamoto, J. Effect of substitution (Ta, Al, Ga) on the conductivity of Li7La3Zr2O12. J. Power Sources 2012, 206, 315–319. [Google Scholar] [CrossRef]
- Kuhnert, E.; Ladenstein, L.; Jodlbauer, A.; Slugovc, C.; Trimmel, G.; Wilkening, H.M.R.; Rettenwander, D. Lowering the Interfacial Resistance in Li6.4La3Zr1.4Ta0.6O12|Poly(Ethylene Oxide) Composite Electrolytes. Cell Rep. Phys. Sci. 2020, 1, 100214. [Google Scholar] [CrossRef]
- Wurster, V.; Engel, C.; Graebe, H.; Ferber, T.; Jaegermann, W.; Hausbrand, R. Characterization of the Interfaces in LiFePO4/PEO-LiTFSI Composite Cathodes and to the Adjacent Layers. J. Electrochem. Soc. 2019, 166, A5410–A5420. [Google Scholar] [CrossRef]
- Sharafi, A.; Kazyak, E.; Davis, A.L.; Yu, S.; Thompson, T.; Siegel, D.J.; Dasgupta, N.P.; Sakamoto, J. Surface Chemistry Mechanism of Ultra-Low Interfacial Resistance in the Solid-State Electrolyte Li7La3Zr2O12. Chem. Mater. 2017, 29, 7961–7968. [Google Scholar] [CrossRef]
- Brugge, R.H.; Hekselman, A.K.O.; Cavallaro, A.; Pesci, F.M.; Chater, R.J.; Kilner, J.A.; Aguadero, A. Garnet Electrolytes for Solid State Batteries: Visualization of Moisture-Induced Chemical Degradation and Revealing Its Impact on the Li-Ion Dynamics. Chem. Mater. 2018, 30, 3704–3713. [Google Scholar] [CrossRef]
- Rosen, M.; Ye, R.; Mann, M.; Lobe, S.; Finsterbusch, M.; Guillon, O.; Fattakhova-Rohlfing, D. Controlling the lithium proton exchange of LLZO to enable reproducible processing and performance optimization. J. Mater. Chem. A 2021, 9, 4831–4840. [Google Scholar] [CrossRef]
- Brugge, R.H.; Pesci, F.M.; Cavallaro, A.; Sole, C.; Isaacs, M.A.; Kerherve, G.; Weatherup, R.S.; Aguadero, A.; Issacs, M. The origin of chemical inhomogeneity in garnet electrolytes and its impact on the electrochemical performance. J. Mater. Chem. A 2020, 8, 14265–14276. [Google Scholar] [CrossRef]
- Huang, Z.; Pang, W.; Liang, P.; Jin, Z.; Grundish, N.; Li, Y.; Wang, C.A. A dopamine modified Li6.4La3Zr1.4Ta0.6O12/PEO solid-state electrolyte: Enhanced thermal and electrochemical properties. J. Mater. Chem. A 2019, 7, 16425–16436. [Google Scholar] [CrossRef]
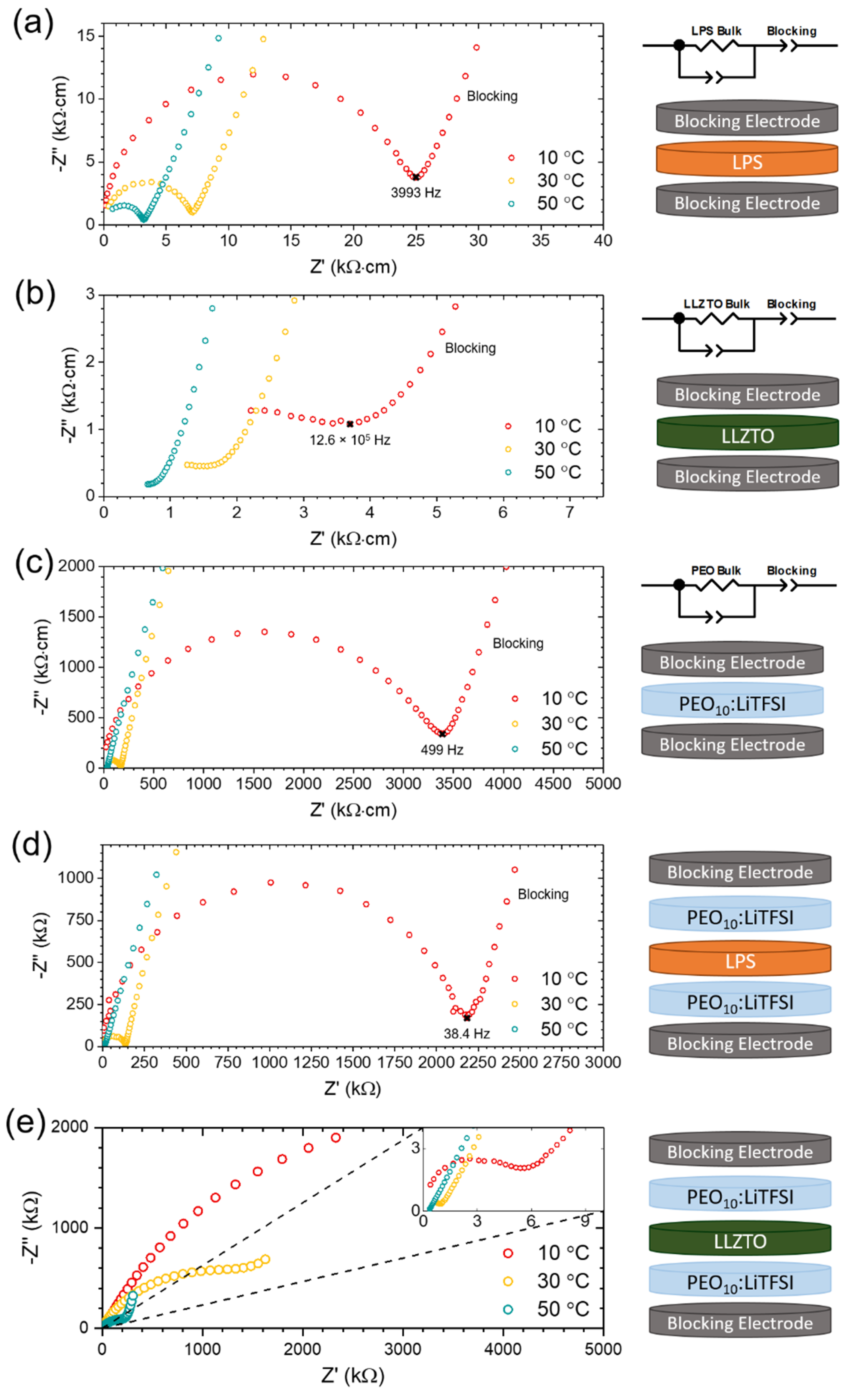
| Temperature/°C | Interfacial Resistance/kΩ·cm2 | |
|---|---|---|
| LLZTO | LPS | |
| 10 | 1540 | 37 |
| 30 | 496 | 2 |
| 50 | 79 | negligible |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spencer Jolly, D.; Melvin, D.L.R.; Stephens, I.D.R.; Brugge, R.H.; Pu, S.D.; Bu, J.; Ning, Z.; Hartley, G.O.; Adamson, P.; Grant, P.S.; et al. Interfaces between Ceramic and Polymer Electrolytes: A Comparison of Oxide and Sulfide Solid Electrolytes for Hybrid Solid-State Batteries. Inorganics 2022, 10, 60. https://doi.org/10.3390/inorganics10050060
Spencer Jolly D, Melvin DLR, Stephens IDR, Brugge RH, Pu SD, Bu J, Ning Z, Hartley GO, Adamson P, Grant PS, et al. Interfaces between Ceramic and Polymer Electrolytes: A Comparison of Oxide and Sulfide Solid Electrolytes for Hybrid Solid-State Batteries. Inorganics. 2022; 10(5):60. https://doi.org/10.3390/inorganics10050060
Chicago/Turabian StyleSpencer Jolly, Dominic, Dominic L. R. Melvin, Isabella D. R. Stephens, Rowena H. Brugge, Shengda D. Pu, Junfu Bu, Ziyang Ning, Gareth O. Hartley, Paul Adamson, Patrick S. Grant, and et al. 2022. "Interfaces between Ceramic and Polymer Electrolytes: A Comparison of Oxide and Sulfide Solid Electrolytes for Hybrid Solid-State Batteries" Inorganics 10, no. 5: 60. https://doi.org/10.3390/inorganics10050060
APA StyleSpencer Jolly, D., Melvin, D. L. R., Stephens, I. D. R., Brugge, R. H., Pu, S. D., Bu, J., Ning, Z., Hartley, G. O., Adamson, P., Grant, P. S., Aguadero, A., & Bruce, P. G. (2022). Interfaces between Ceramic and Polymer Electrolytes: A Comparison of Oxide and Sulfide Solid Electrolytes for Hybrid Solid-State Batteries. Inorganics, 10(5), 60. https://doi.org/10.3390/inorganics10050060







