Coumarins Synthesis and Transformation via C–H Bond Activation—A Review
Abstract
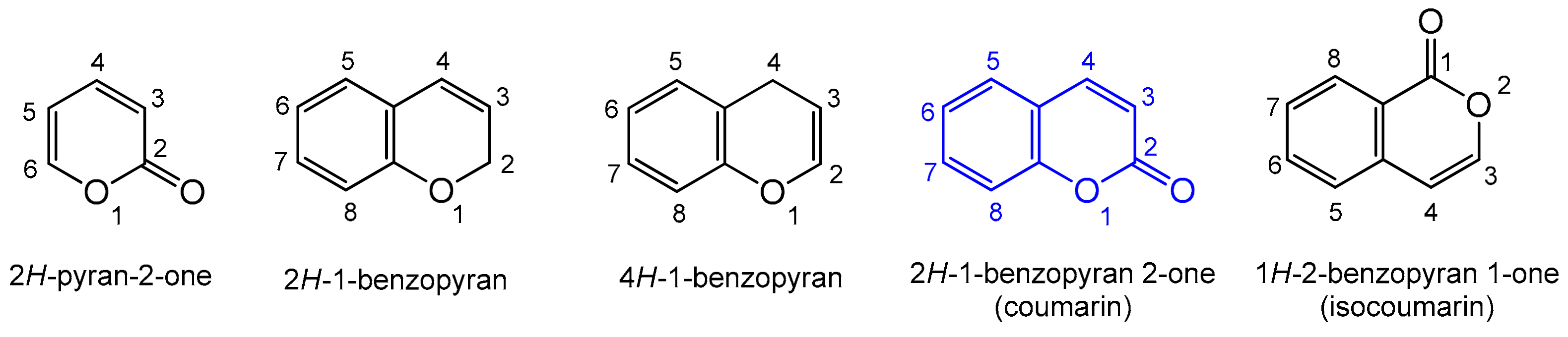
:1. Introduction to Coumarins
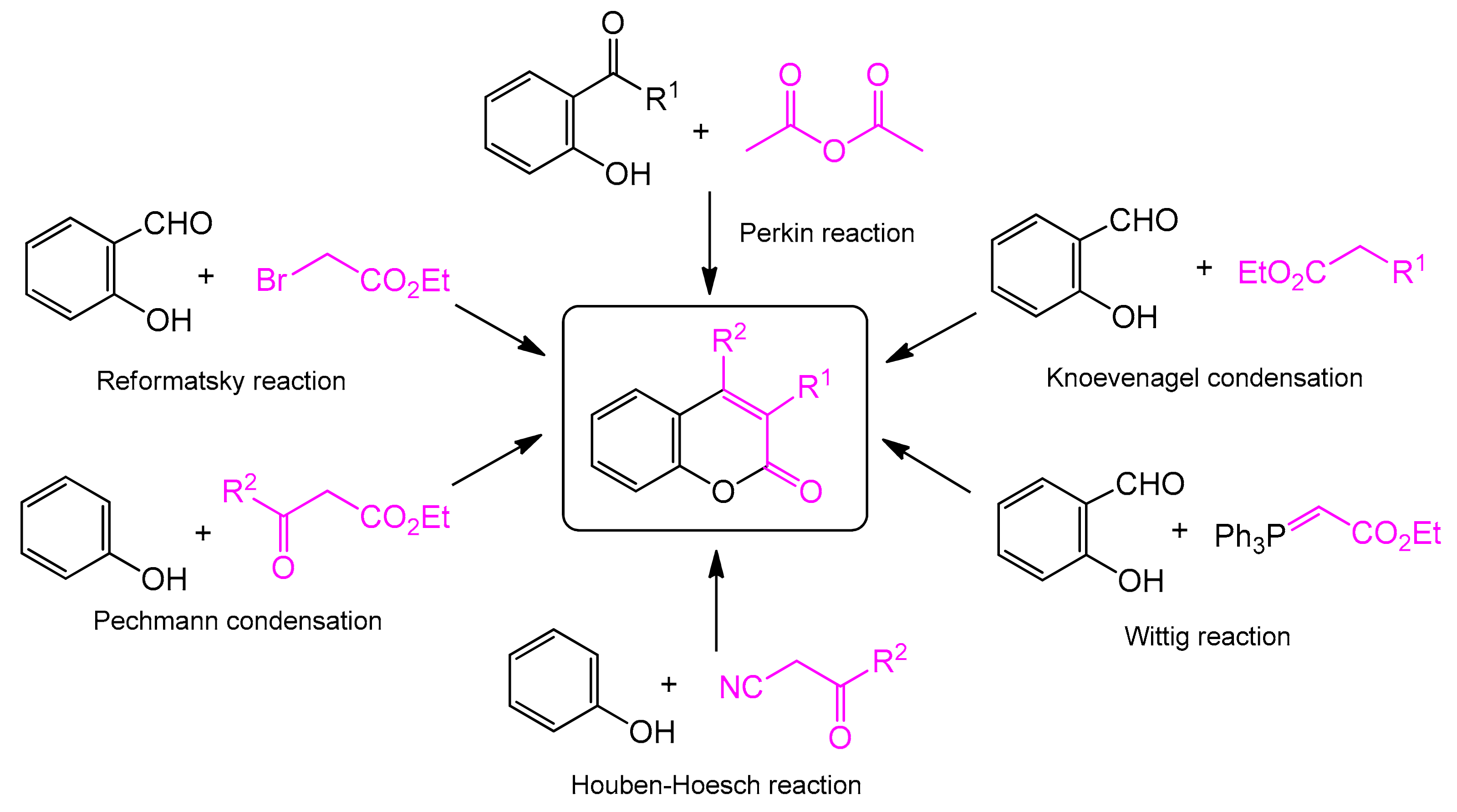
2. Methods for the Synthesis of the Coumarin Core
3. C–H Bond Activation in Coumarin Synthesis
3.1. Direct Synthesis of Coumarins through Intermolecular Hydroarylation of Alkynes
3.2. Direct Synthesis of Coumarins through the Intramolecular Hydroarylation of Alkynes
3.3. Direct Synthesis of Coumarins via the Intermolecular Hydroarylation of Alkenes
3.4. Direct Synthesis of Coumarins via Intramolecular Hydroarylation of Alkenes
3.5. Intramolecular Cyclocarbonylation and Cyclocarboxylation
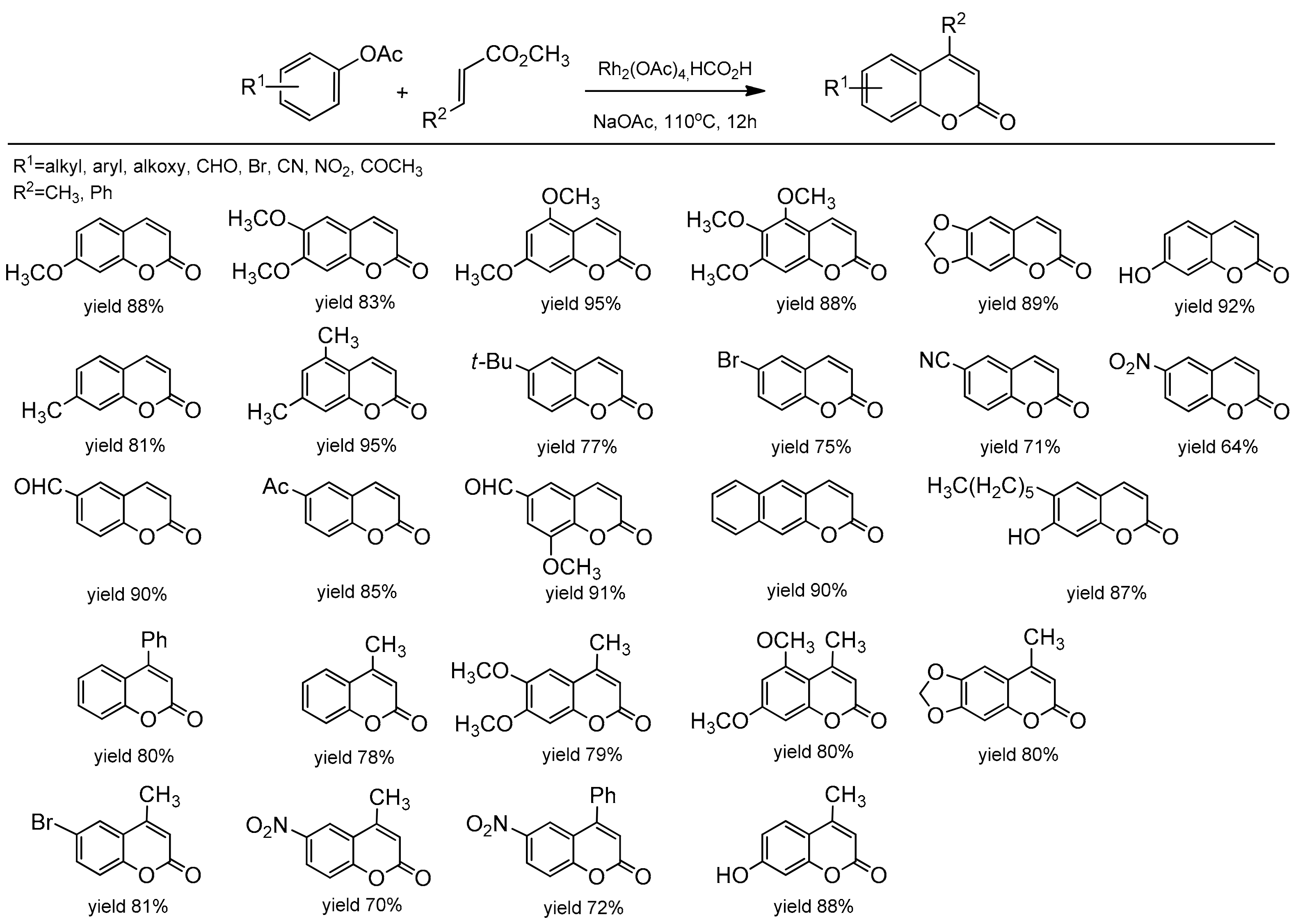
3.6. Other Methods for Direct Synthesis of Coumarins by C–H Bond Activation
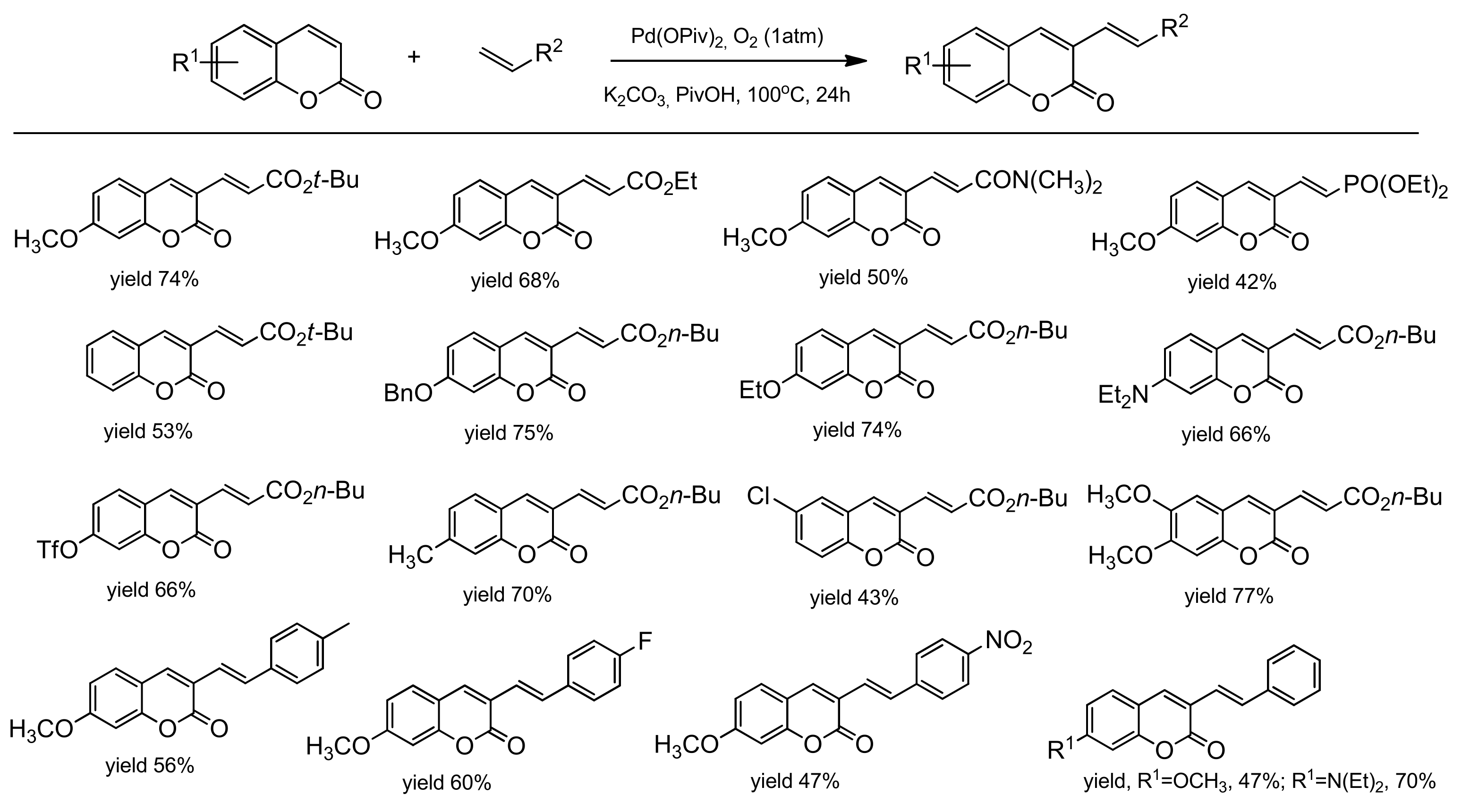
3.7. C–H bond Activation Strategy for the Synthesis of C-3 and C-4 Substituted Coumarins
3.7.1. C-3 Selective Reactions
3.7.2. C-4 Selective Reactions
4. Summary
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Fairlamb, I.J.S.; Marrison, L.R.; Dickinson, J.M.; Lu, F.-J.; Schmidt, J.P. 2-Pyrones possessing antimicrobial and cytotoxic activities. Bioorg. Med. Chem. 2004, 12, 4285–4299. [Google Scholar] [CrossRef] [PubMed]
- McGlacken, G.P.; Fairlamb, I.J.S. 2-Pyrone natural products and mimetics: Isolation, characterisation and biological activity. Nat. Prod. Rep. 2005, 22, 369–385. [Google Scholar] [CrossRef] [PubMed]
- Favre, H.A.; Powell, W.H. Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013; Royal Society of Chemistry Publishing: London, UK, 2014. [Google Scholar] [CrossRef]
- Venugopala, K.N.; Rashmi, V.; Odhav, B. Review on natural coumarin lead compounds for their pharmacological activity. BioMed Res. Int. 2013, 2013, 963248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stefanachi, A.; Leonetti, F.; Pisani, L.; Catto, M.; Carotti, A. Coumarin: A natural, privileged and versatile scaffold for bioactive compounds-literature. Molecules 2018, 23, 250. [Google Scholar] [CrossRef] [Green Version]
- Srikrishna, D.; Godugu, C.; Dubey, P.K. A Review on pharmacological properties of coumarins. Mini Rev. Med. Chem. 2018, 18, 113–141. [Google Scholar] [CrossRef]
- Balewski, Ł.; Szulta, S.; Jalińska, A.; Kornicka, A. A Mini-review: Recent advances in coumarin-metal complexes with biological properties. Front. Chem. 2021, 9. [Google Scholar] [CrossRef]
- Peng, X.-M.; Damu, G.L.V.; Zhou, C.-H. Current developments of coumarin compounds in medicinal chemistry. Curr. Pharm. Des. 2013, 19, 3884–3930. [Google Scholar] [CrossRef]
- El-Naggar, A.M.; Ahmed, F.S.; Abd El-Salam, A.M.; Rady, M.A.; Latif, M.S.A. Synthesis and biological activity of some new 3-and 6-substituted coumarin amino acid derivatives. Part I. J. Heterocycl. Chem. 1981, 18, 1203–1207. [Google Scholar] [CrossRef]
- Grover, J.; Jachak, S.M. Coumarins as privileged scaffold for anti-inflammatory drug development. RSC Adv. 2015, 5, 38892–38905. [Google Scholar] [CrossRef]
- Emami, S.; Dadashpour, S. Current developments of coumarin based anti-cancer agents in medicinal chemistry. Eur. J. Med. Chem. 2015, 102, 611–630. [Google Scholar] [CrossRef]
- Thakur, A.; Singla, R.; Jaitak, V. Coumarins as anticancer agents: A review on synthetic strategies, mechanism of action and SAR studies. Eur. J. Med. Chem. 2015, 101, 476–495. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Liu, Y.; Jia, H.; Zhou, Y.-D.; Nagle, D.G. Benzochromenones from the marine crinoid comantheria rotula inhibit hypoxia-inducible factor-1 (HIF-1) in cell-based reporter assays and differentially suppress the growth of certain tumor cell lines. J. Nat. Prod. 2007, 70, 1462–1466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akkol, E.K.; Genç, Y.; Karpuz, B.; Sobarzo-Sánchez, E.; Capasso, R. Coumarins and coumarin-related compounds in pharmacotherapy of cancer. Cancers 2020, 12, 1959. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.Z.; Osman, H.; Ali, M.A.; Ahsan, M.J. Therapeutic potential of coumarins as antiviral agents. Eur. J. Med. Chem. 2016, 123, 236–255. [Google Scholar] [CrossRef]
- Hsieh, H.-P.; Hsu, T.-A.; Yeh, J.-Y.; Horng, J.-T.; Shih, S.-R.; Chang, S.-T.; Chao, Y.-S. Coumarin Compounds and Their Use for Treating Viral Infection. U.S. Patent Application No. 12/481,789, 17 December 2009. [Google Scholar]
- Arora, R.K.; Kaur, N.; Bansal, Y.; Bansal, G. Novel coumarin-benzimidazole derivatives as antioxidants and safer anti-inflammatory agents. Acta Pharm. Sin. B 2014, 4, 368–375. [Google Scholar] [CrossRef] [Green Version]
- Pu, W.; Lin, Y.; Zhang, J.; Wang, F.; Wang, C.; Zhang, G. 3-Arylcoumarins: Synthesis and potent anti-inflammatory activity. Bioorg. Med. Chem. Lett. 2014, 24, 5432–5434. [Google Scholar] [CrossRef]
- Chen, L.Z.; Sun, W.; Bol, W.; Wang, J.Q.; Xiu, C.; Tang, W.J.; Shi, J.B.; Zhou, H.P.; Liu, X.H. New arylpyrazoline-coumarins: Synthesis and anti-inflammatory activity. Eur. J. Med. Chem. 2017, 138, 170–181. [Google Scholar] [CrossRef]
- Kostova, I.; Bhatia, S.; Grigorov, P.; Balkansky, S.; Parmar, V.S.; Prasad, A.K.; Saso, L. Coumarins as antioxidants. Curr. Med. Chem. 2011, 18, 3929–3951. [Google Scholar] [CrossRef]
- Kathiravan, M.K.; Salake, A.B.; Chothe, A.S.; Dudhe, P.B.; Watode, R.P.; Mukta, M.S.; Gadhwe, S. The biology and chemistry of antifungal agents: A review. Bioorg. Med. Chem. 2012, 20, 5678–5698. [Google Scholar] [CrossRef]
- Satish, G. Chapter 8—Anticoagulant agents. In Advances in Structure and Activity Relationship of Coumarin Derivatives; Penta, S., Ed.; Academic Press: Boston, MA, USA, 2016; pp. 151–159. [Google Scholar]
- Abdelhafez, O.M.; Amin, K.M.; Batran, R.Z.; Maher, T.J.; Nada, S.A.; Sethumadhavan, S. Synthesis, anticoagulant and PIVKA-II induced by new 4-hydroxycoumarin derivatives. Bioorg. Med. Chem. 2010, 18, 3371–3378. [Google Scholar] [CrossRef]
- Yu, D.; Suzuki, M.; Xie, L.; Morris-Natschke, S.L.; Lee, K.-H. Recent progress in the development of coumarin derivatives as potent anti-HIV agents. Med. Res. Rev. 2003, 23, 322–345. [Google Scholar] [CrossRef] [PubMed]
- Kostova, I. Coumarins as inhibitors of HIV reverse transcriptase. Curr. HIV Res. 2006, 4, 347–363. [Google Scholar] [CrossRef]
- Wadhwa, P.; Priti, J.; Santosh, R.; Hemant, R.A.J. Quinoline, coumarin and other heterocyclic analogs based HIV-1 integrase inhibitors. Curr. Drug Discov. Technol. 2018, 15, 2–19. [Google Scholar] [CrossRef] [PubMed]
- Dorababu, A. Pharmacological report of recently designed multifunctional coumarin and coumarin-heterocycle derivatives. Arch. Pharm. 2021, 355, e2100345. [Google Scholar] [CrossRef]
- Edmondson, D.E.; Mattevi, A.; Binda, C.; Li, M.; Hubalek, F. Structure and mechanism of monoamine oxidases. Curr. Med. Chem. 2004, 11, 1983–1993. [Google Scholar] [CrossRef] [PubMed]
- Wimbiscus, M.; Kostenko, O.; Malone, D. MAO inhibitors: Risks, benefits, and lore. Clevel. Clin. J. Med. 2010, 77, 859–882. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Liu, Z.; Koo, S.; Jangjili, P.; Kim, J.S.; Lin, W. Coumarin-based small-molecule fluorescent chemosensors. Chem. Rev. 2019, 119, 10403–10519. [Google Scholar] [CrossRef]
- Ansary, I.; Taher, A. One-pot synthesis of coumarin derivatives. In Phytochemicals in Human Health; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef] [Green Version]
- Perkin, W.H. On the artificial production of coumarin and formation of its homologues. J. Chem. Soc. 1868, 21, 53–63. [Google Scholar] [CrossRef] [Green Version]
- Perkin, W.H. On the hydride of aceto-salicyl. J. Chem. Soc. 1868, 21, 181–186. [Google Scholar] [CrossRef] [Green Version]
- Woods, L.L.; Sapp, J. A new one-step synthesis of substituted coumarins. J. Org. Chem. 1962, 27, 3703–3705. [Google Scholar] [CrossRef]
- Sethna, S.M.; Shah, N.M.; Shah, R.C. Aluminium chloride, a new reagent for the condensation of β-ketonic esters with phenols. Part I. The condensations of methyl β-resorcylate, β-resorcylic acid, and resacetophenone with ethyl acetoacetate. J. Chem. Soc. 1938, 228–232. [Google Scholar] [CrossRef]
- Corrie, J.E.T. A convenient synthesis of N-(7-dimethylamino-4-methylcoumarin-3-yl)-maleimide incorporating a novel variant of the Pechmann reaction. J. Chem. Soc. Perkin Trans. 1990, 1, 2151–2152. [Google Scholar] [CrossRef]
- Smitha, G.; Reddy, S.C. ZrCl4- catalyzed Pechmann reaction: Synthesis of coumarins under solvent-free conditions. Synth. Commun. 2004, 34, 3997–4003. [Google Scholar] [CrossRef]
- Valizadeh, H.; Shockravi, A. An efficient procedure for the synthesis of coumarin derivatives using TiCl4 as catalyst under solvent-free conditions. Tetrahedron Lett. 2005, 46, 3501–3503. [Google Scholar] [CrossRef]
- John, E.; Israelstam, S. Notes. Use of cation exchange resins in organic reactions. I. The Von Pechmann reaction. J. Org. Chem. 1961, 26, 240–242. [Google Scholar] [CrossRef]
- Laufer, M.C.; Hausmann, H.; Hölderich, W.F. Synthesis of 7-hydroxycoumarins by Pechmann reaction using nafion resin/silica nanocomposites as catalysts. J. Catal. 2003, 218, 315–320. [Google Scholar] [CrossRef]
- Hoefnagel, A.J.; Gunnewegh, E.A.; Downing, R.S.; van Bekkum, H. Synthesis of 7-hydroxycoumarins catalysed by solid acid catalysts. J. Chem. Soc. Chem. Commun. 1995, 225–226. [Google Scholar] [CrossRef]
- Salem, M.A.; Helal, M.H.; Gouda, M.A.; Ammar, Y.A.; El-Gaby, M. An overview on synthetic strategies to coumarins. Synth. Commun. 2018, 48, 1534–1550. [Google Scholar] [CrossRef]
- Vekariya, R.H.; Patel, H.D. Recent advances in the synthesis of coumarin derivatives via Knoevenagel condensation: A review. Synth. Commun. 2014, 44, 2756–2788. [Google Scholar] [CrossRef]
- Borges, F.; Roleira, F.; Milhazes, N.; Santana, L.; Uriarte, E. Simple coumarins and analogues in medicinal chemistry: Occurrence, synthesis and biological activity. Curr. Med. Chem. 2005, 12, 887–916. [Google Scholar] [CrossRef]
- Heravi, M.M.; Khaghaninejad, S.; Mostofi, M. Pechmann reaction in the synthesis of coumarin derivatives. Adv. Heterocycl. Chem. 2014, 112, 1–50. [Google Scholar] [CrossRef]
- Lanman, B.A. Pechmann coumarin synthesis. In Name Reactions in Heterocyclic Chemistry II; Li, J.J., Ed.; John Wiley & Sons: New York, NY, USA, 2011. [Google Scholar]
- Vekariya, R.H.; Patel, H.D. Synthesis of bromocarbonyl compounds: Recent advances. Tetrahedron 2014, 70, 3949–3961. [Google Scholar] [CrossRef]
- Ibrahem, I.; Sundén, H.; Rios, R.; Zhao, G.-L.; Córdova, A. One-pot pyrrolidine-catalyzed synthesis of benzopyrans, benzothiopyranes, and dihydroquinolidines. Chimia 2007, 61, 219–223. [Google Scholar] [CrossRef]
- Bouhaoui, A.; Eddahmi, M.; Dib, M.; Khouili, M.; Aires, A.; Catto, M.; Bouissane, L. Synthesis and biological properties of coumarin derivatives. A review. Chem. Select 2021, 6, 5848–5870. [Google Scholar] [CrossRef]
- Choi, H.; Kim, J.; Lee, K. Metal-free, Brønsted acid-mediated synthesis of coumarin derivatives from phenols and propiolic acids. Tetrahedron Lett. 2016, 57, 3600–3603. [Google Scholar] [CrossRef]
- Gouda, M.A.; Hussein, B.H.M.; El-Demerdash, A.; Ibrahim, M.E.; Salem, M.A.; Helal, M.H.; Hamama, W.S. A Review: Synthesis and medicinal importance of coumarins and their analogues. Curr. Bioact. Compd. 2020, 16, 993–1008. [Google Scholar] [CrossRef]
- Shilov, E.; Shul’pin, G.B. Activation of C−H bonds by metal complexes. Chem. Rev. 1997, 97, 2879–2932. [Google Scholar] [CrossRef]
- Kuhl, N.; Hopkinson, M.N.; Wencel-Delord, J.; Glorius, F. Beyond directing groups: Transition-metal-catalyzed C-H activation of simple arenes. Angew. Chem. Int. Ed. 2012, 51, 10236–10254. [Google Scholar] [CrossRef]
- Pratap, R.; Ram, V.J. Natural and synthetic chromenes, fused chromenes, and versatility of dihydrobenzo[h]chromenes in organic synthesis. Chem. Rev. 2014, 114, 10476–10526. [Google Scholar] [CrossRef]
- Tasior, M.; Kim, D.; Singha, S.; Krzeszewski, M.; Ahn, K.H.; Gryko, D.T. π-Expanded coumarins: Synthesis, optical properties and applications. J. Mater. Chem. C 2015, 3, 1421–1446. [Google Scholar] [CrossRef]
- Medina, F.G.; Marrero, J.G.; Macías-Alonso, M.; González, M.C.; Córdova-Guerrero, I.; Teissier García, A.G.; Osegueda-Robles, S. Coumarin heterocyclic derivatives: Chemical synthesis and biological activity. Nat. Prod. Rep. 2015, 32, 1472–1507. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.-W.; Kim, N.-J.; Yun, H.; Han, Y.T. Recent advances in synthesis of 4-arylcoumarins. Molecules 2018, 23, 2417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdou, M.M.; Abu-Rayyanb, A.; Bedira, A.G.; Abdel-Fattaha, S.; Omara, A.M.A.; Ahmedc, A.A.; El-Desoky, E.-S.I.; Ghaithd, E.A. 3-(Bromoacetyl)coumarins: Unraveling their synthesis, chemistry, and applications. RSC Adv. 2021, 11, 38391–38433. [Google Scholar] [CrossRef]
- Koleva, A.I.; Petkova-Yankova, N.I.; Nikolova, R.D. Synthesis and chemical properties of 3-phosphono-coumarins and 1,2-benzoxaphosphorins as precursors for bioactive compounds. Molecules 2019, 24, 2030. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Lan, J.; You, J. Oxidative C−H/C−H coupling reactions between two (hetero)arenes. Chem. Rev. 2017, 117, 8787–8863. [Google Scholar] [CrossRef]
- Bhatia, R.; Pathania, S.; Singh, V.; Rawal, R.K. Metal-catalyzed synthetic strategies toward coumarin derivatives. Chem. Hetero. Comp. 2018, 54, 280–291. [Google Scholar] [CrossRef]
- Kanchana, U.S.; Diana, E.J.; Mathew, T.V.; Anilkumar, G. Palladium-catalyzed cross-coupling reactions of coumarin derivatives: An overview. Appl. Organomet. Chem. 2020, 34, e5983. [Google Scholar] [CrossRef]
- Sharma, R.K.; Katiyar, D. Recent advances in transition-metal-catalyzed synthesis of coumarins. Synthesis 2016, 48, 2303–2322. [Google Scholar] [CrossRef]
- Choi, H.; Min, M.; Peng, Q.; Kang, D.; Paton, R.S.; Hong, S. Unraveling innate substrate control in site-selective palladium-catalyzed C–H heterocycle functionalization. Chem. Sci. 2016, 7, 3900–3909. [Google Scholar] [CrossRef] [Green Version]
- Trost, B.M.; Toste, F.D. A new palladium-catalyzed addition: A mild method for the synthesis of coumarins. J. Am. Chem. Soc. 1996, 118, 6305–6306. [Google Scholar] [CrossRef]
- Trost, B.M.; Toste, F.D.; Greenman, K. Atom economy. Palladium-catalyzed formation of coumarins by addition of phenols and alkynoates via a net C-H insertion. J. Am. Chem. Soc. 2003, 125, 4518–4526. [Google Scholar] [CrossRef]
- Jia, C.; Lu, W.; Oyamada, J.; Kitamura, T.; Matsuda, K.; Irie, M.; Fujiwara, Y. Novel Pd(II)- and Pt(II)-catalyzed regio- and stereoselective trans-hydroarylation of alkynes by simple arenes. J. Am. Chem. Soc. 2000, 122, 7252–7263. [Google Scholar] [CrossRef]
- Juzo, O.; Chengguo, J.; Yuzo, F.; Tsugio, K. Direct synthesis of coumarins by Pd(II)-catalyzed reaction of alkoxyphenols and alkynoates. Chem. Lett. 2002, 380–381. [Google Scholar] [CrossRef]
- Kotani, M.; Yamamoto, K.; Oyamada, J.; Fujiwara, Y.; Kitamura, T. A Convenient synthesis of coumarins by palladium(II)-catalyzed reaction of phenols with propiolic acids. Synthesis 2004, 1466–1470. [Google Scholar] [CrossRef]
- Oyamada, J.; Kitamura, T. Synthesis of coumarins by Pt-catalyzed hydroarylation of propiolic acids with phenols. Tetrahedron 2006, 62, 6918–6925. [Google Scholar] [CrossRef]
- Kutubi, S.; Hashimoto, T.; Kitamura, T. Improved synthesis of coumarins by iron(III)-catalyzed cascade reaction of propiolic acids and phenols. Synthesis 2011, 8, 1283–1289. [Google Scholar] [CrossRef]
- Jia, C.; Piao, D.; Kitamura, T.; Fujiwara, Y. New method for preparation of coumarins and quinolinones via Pd-catalyzed intramolecular hydroarylation of C-C triple bonds. J. Org. Chem. 2000, 65, 7516–7522. [Google Scholar] [CrossRef]
- Jia, C.; Piao, D.; Oyamada, J.; Lu, W.; Kitamura, T.; Fujiwara, Y. Efficient activation of aromatic C–H bonds for addition to C–C multiple bonds. Science 2000, 287, 1992–1995. [Google Scholar] [CrossRef]
- Li, R.; Wang, S.R.; Lu, W. FeCl3-Catalyzed alkenylation of simple arenes with aryl-substituted alkynes. Org. Lett. 2007, 9, 2219–2222. [Google Scholar] [CrossRef]
- Pastine, S.J.; Youn, S.W.; Sames, D. Pt-IV-Catalyzed cyclization of arene-alkyne substrates via intramolecular electrophilic hydroarylation. Org. Lett. 2003, 7, 1055–1058. [Google Scholar] [CrossRef]
- Pastine, S.J.; Youn, S.W.; Sames, D. Pt(IV)-catalyzed cyclization of arene–alkyne substrates via C–H bond functionalization. Tetrahedron 2003, 59, 8859–8868. [Google Scholar] [CrossRef]
- Menon, R.S.; Findlay, A.D.; Bissember, A.C.; Banwell, M.G. The Au(I)-catalyzed intramolecular hydroarylation of terminal alkynes under mild conditions: Application to the synthesis of 2H-chromenes, coumarins, benzofurans, and dihydroquinolines. J. Org. Chem. 2009, 74, 8901–8903. [Google Scholar] [CrossRef] [PubMed]
- Aparece, M.D.; Vadola, P.A. Gold-catalyzed dearomative spirocyclization of aryl alkynoate esters. Org. Lett. 2014, 16, 6008–6011. [Google Scholar] [CrossRef] [PubMed]
- Vadola, P.A.; Sames, D. Catalytic coupling of arene C–H bonds and alkynes for the synthesis of coumarins: Substrate scope and application to the development of neuroimaging agents. J. Org. Chem. 2012, 77, 7804–7814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Do, J.H.; Kim, H.N.; Yoon, J.; Kim, J.S.; Kim, H.-J. A rationally designed fluorescence turn-on probe for the gold(III) ion. Org. Lett. 2010, 12, 932–934. [Google Scholar] [CrossRef]
- Shi, Z.; He, C. Efficient functionalization of aromatic C-H bonds catalyzed by gold(III) under mild and solvent-free conditions. J. Org. Chem. 2004, 69, 3669–3671. [Google Scholar] [CrossRef] [PubMed]
- Aoki, S.; Oyamada, J.; Kitamura, T. Formation of coumarins by palladium(II)-catalyzed reaction of phenols with ethyl acrylates. Bull. Chem. Soc. Jpn. 2005, 78, 468–472. [Google Scholar] [CrossRef]
- Sharma, U.; Naveen, T.; Maji, A.; Manna, S.; Maiti, D. Angew. Palladium-catalyzed synthesis of benzofurans and coumarins from phenols and olefins. Chem. Int. Ed. 2013, 52, 12669–12673. [Google Scholar] [CrossRef]
- Zhang, X.-S.; Li, Z.-W.; Shi, Z.-J. Palladium-catalyzed base-accelerated direct C–H bond alkenylation of phenols to synthesize coumarin derivatives. Org. Chem. Front. 2014, 1, 44–49. [Google Scholar] [CrossRef]
- Gadakh, S.K.; Dey, S.; Sudalai, A. Rh-Catalyzed synthesis of coumarin derivatives from phenolic acetates and acrylates via C–H bond activation. J. Org. Chem. 2015, 80, 11544–11550. [Google Scholar] [CrossRef]
- Carral-Menoyo, A.; Misol, A.; Gomez-Redondo, M.; Sotomayor, N.; Lete, E. Palladium(II)-catalyzed intramolecular C-H alkenylation for the synthesis of chromanes. J. Org. Chem. 2019, 84, 2048–2060. [Google Scholar] [CrossRef]
- Ortiz-de-Elguea, V.; Carral-Menoyo, A.; Simón-Vidal, L.; Martinez-Nunes, M.; Barbolla, I.; Lete, M.G.; Sotomayor, N.; Lete, E. Pd(II)-catalyzed Fujiwara−Moritani reactions for the synthesis and functionalization of substituted coumarins. ACS Omega 2021, 6, 29483–29494. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.-F.; Neumann, H.; Beller, M. Synthesis of heterocycles via palladium-catalyzed carbonylations. Chem. Rev. 2012, 113, 1–35. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, J.; Zeng, F.; Alper, H. Synthesis of coumarins via Pd-catalyzed oxidative cyclocarbonylation of 2-vinylphenols. Org. Lett. 2012, 14, 5602–5605. [Google Scholar] [CrossRef] [PubMed]
- Seoane, A.; Casanova, N.; Quiñones, N.; Mascareñas, J.L.; Gulías, M. Straightforward assembly of benzoxepines by means of a rhodium(III)-catalyzed C–H functionalization of o-vinylphenols. J. Am. Chem. Soc. 2014, 136, 834–837. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.-G.; Zhang, S.-S.; Jiang, C.-Y.; Wu, J.-Q.; Li, Q.; Wang, H. Cp*Co(III)-Catalyzed annulations of 2-alkenylphenols with CO: Mild access to coumarin derivatives. Org. Lett. 2015, 17, 5404–5407. [Google Scholar] [CrossRef]
- Sasano, K.; Takaya, J.; Iwasawa, N. Palladium(II)-catalyzed direct carboxylation of alkenyl C−H Bonds with CO2. J. Am. Chem. Soc. 2013, 135, 10954–10957. [Google Scholar] [CrossRef]
- Kim, D.; Min, M.; Hong, S. One-pot catalysis of dehydrogenation of cyclohexanones to phenols and oxidative Heck coupling: Expedient synthesis of coumarins. Chem. Commun. 2013, 49, 4021–4022. [Google Scholar] [CrossRef]
- Zhao, Y.; Han, F.; Yang, L.; Xia, C. Access to coumarins by rhodium-catalyzed oxidative annulation of aryl thiocarbamates with internal alkynes. Org. Lett. 2015, 17, 1477–1480. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, J.; Mao, F.; Kwong, F.Y. Palladium-catalyzed cross-dehydrogenative functionalization of C (sp2)-H bonds. Chem. Asian J. 2014, 9, 26–47. [Google Scholar] [CrossRef]
- Min, M.; Kim, Y.; Hong, S. Regioselective palladium-catalyzed olefination of coumarins via aerobic oxidative Heck reactions. Chem. Commun. 2013, 49, 196–198. [Google Scholar] [CrossRef]
- Wang, X.; Pan, S.; Li, Y.; Wang, H.; Chen, Z.; Huang, K. Regioselective palladium-catalyzed decarboxylative cross-coupling reaction of alkenyl acids with coumarins: Synthesis of 3-styrylcoumarin compounds. J. Org. Chem. 2015, 80, 2407–2411. [Google Scholar] [CrossRef] [PubMed]
- Jafarpour, F.; Zarei, S.; Olia, M.B.A.; Jalalimanesh, N.; Rahiminejadan, S. Palladium-catalyzed decarboxylative cross-coupling reactions: A route for regioselective functionalization of coumarins. J. Org. Chem. 2013, 78, 2957–2964. [Google Scholar] [CrossRef] [PubMed]
- Jafarpour, F.; Olia, M.B.A.; Hazrati, H. Highly regioselective α-arylation of coumarins via palladium- catalyzed C-H activation/desulfitative coupling. Adv. Synth. Catal. 2013, 355, 3407–3412. [Google Scholar] [CrossRef]
- Mi, X.; Huang, M.; Zhang, J.; Wang, C.; Wu, Y. Regioselective palladium-catalyzed phosphonation of coumarins with dialkyl H-phosphonates via C-H functionalization. Org. Lett. 2013, 15, 6266–6269. [Google Scholar] [CrossRef]
- Pashazadeh, R.; Rajai-Daryasarei, S.; Mirzaei, S.; Soheilizad, M.; Ansari, S.; Shabanian, M. A regioselective approach to C3-aroylcoumarins via cobalt catalyzed C(sp2)–H activation carbonylation of coumarin. Synthesis 2019, 51, 3014–3020. [Google Scholar] [CrossRef]
- Min, M.; Hong, S. Regioselective palladium-catalyzed direct cross-coupling of coumarins with simple arenes. Chem. Commun. 2012, 48, 9613–9615. [Google Scholar] [CrossRef]
- Li, Y.; Qi, Z.; Wang, H.; Fu, X.; Duan, C. Palladium-catalyzed oxidative Heck coupling reaction for direct synthesis of 4-arylcoumarins using coumarins and arylboronic acids. J. Org. Chem. 2012, 77, 2053–2057. [Google Scholar] [CrossRef]
- Khoobi, M.; Alipour, M.; Zarei, S.; Jafarpour, F.; Abbas Shafiee, A. A facile route to flavone and neoflavone backbones via a regioselective palladium catalysed oxidative Heck reaction. Chem. Comm. 2012, 48, 2985–2987. [Google Scholar] [CrossRef]
- Wang, Y.-J.; Wang, T.-T.; Yao, L.; Wang, Q.-L.; Zhao, L.-M. Access to 4-alkenylated coumarins via ruthenium-catalyzed olefinic C−H alkenylation of coumarins with modifiable and removable directing groups. J. Org. Chem. 2020, 85, 9514–9524. [Google Scholar] [CrossRef]
- Wu, X.; Yang, Y.; Han, J.; Wang, L. Palladium catalyzed C–I and vicinal C–H dual activation of diaryliodonium salts for diarylation: Synthesis of 4,5-benzocoumarins. Org. Lett. 2015, 17, 5654–5657. [Google Scholar] [CrossRef] [PubMed]




































| Entry (References) | R1 of Phenol | R2 | R3 | Metal Source | Base | Solvent | Yield (%) |
|---|---|---|---|---|---|---|---|
| 1 [66] | 3-OCH3 | H | H | Pd(OAc)2 | - | TFA | 19 |
| 2 [70] | 3-OCH3 | H | H | K2PtCl4/AgO Tf | - | TFA | 48 a |
| 3 [71] | 3-OCH3 | H | Ph | FeCl3/AgOTf | - | TFA/DCE | 75 |
| 4 [69] | 3-OCH3 | H | Ph | Pd(OAc)2 | - | TFA | 65 |
| 5 [70] | 3-OCH3 | H | Ph | K2PtCl4/AgOTf | - | TFA | 82 |
| 6 [68] | 3-OCH3 | Et | Ph | Pd(OAc)2 | - | TFA | 85 |
| 7 [70] | 3-OCH3 | H | n-C5H11 | PtCl2/AgO Tf | - | TFA | 93 |
| 8 [70] | 4-OCH3 | H | H | K2PtCl4/AgO Tf | - | TFA | 22 |
| 9 [71] | 3,5-di-OCH3 | H | Ph | FeCl3/AgOTf | - | TFA/DCE | 62 |
| 10 [70] | 3,5-di-OCH3 | H | Ph | K2PtCl4/AgOAc | - | TFA | 69 |
| 11 [70] | 3,5-di-OCH3 | Et | H | AgBF4 | - | THF | 92 |
| 12 [70] | 3,5-di-OCH3 | Et | H | PtCl2 | NaOAc | HCOOH | 49 |
| 13 [68] | 3,5-di-OCH3 | Et | Ph | Pd(OAc)2 | - | TFA | 56 |
| 14 [65] | 3,5-di-OCH3 | Et | CH3 | Pd2(dba)3 | NaOAc | HCOOH | 63 |
| 15 [68] | 3,5-di-OCH3 | Et | CH3 | Pd(OAc)2 | - | TFA | 97 |
| 16 [65] | 3,5-di-OCH3 | Et | (CH2)2OCO2Et | Pd2(dba)3 | NaOAc | HCOOH | 58 |
| 17 [65] | 3,5-di-OCH3 | Et | (CH2)3CN | Pd2(dba)3 | NaOAc | HCOOH | 67 |
| 18 [65] | 3,5-di-OCH3 | Et | CH2OH | Pd2(dba)3 | NaOAc | HCOOH | 0 |
| 19 [68] | 3,5-di-OCH3 | CH3 | n-C5H11 | Pd(OAc)2 | - | TFA | 96 |
| 20 [71] | 3,5-di-OCH3 | H | n-C5H11 | FeCl3/AgOTf | - | TFA/DCE | 68 |
| 21 [70] | 3,5-di-OCH3 | Et | n-C5H11 | K2PtCl4/AgOTf | - | TFA | 87 |
| 22 [65] | 3,4,5-tri-OCH3 | Et | H | Pd2(dba)3 | NaOAc | HCOOH | 46 |
| 23 [68] | 3,4,5-tri-OCH3 | Et | H | Pd(OAc)2 | - | TFA | 91 |
| 24 [65] | 3-OCH3-5-CH3 | Et | H | Pd2(dba)3 | NaOAc | HCOOH | 72 |
| 25 [65] | 3,5-di-OH | Et | CH3 | Pd2(dba)3 | NaOAc | HCOOH | 83 |
| 26 [71] | 3,5-di-OH | H | H | FeCl3/AgOTf | - | TFA/DCE | 59 |
| 27 [71] | 1,3,5-tri-OH | H | H | FeCl3/AgOTf | - | TFA/DCE | 95 |
| 28 [71] | 3,4-methylenedioxy | H | H | FeCl3/AgOTf | - | TFA/DCE | 60 |
| 29 [69] | 3,4-methylenedioxy | H | H | Pd(OAc)2 | - | TFA | 53 |
| 30 [70] | 3,4-methylenedioxy | H | H | K2PtCl4/AgOTf | - | TFA | 38 |
| 31 [70] | 3,4-methylenedioxy | H | Ph | K2PtCl4/AgOTf | - | TFA | 77 |
| 32 [70] | 3,4-methylenedioxy | H | n-C5H11 | K2PtCl4/AgOAc | - | TFA | 72 |
| 33 [68] | 3,4-methylenedioxy | Et | Ph | Pd(OAc)2 | - | TFA | 93 |
| 34 [71] | H | H | H | FeCl3/AgOTf | - | TFA/DCE | 33 |
| 35 [70] | H | Et | H | K2PtCl4/AgOTf | - | TFA | 33 |
| 36 [70] | 3-CH3 | Et | H | K2PtCl4/AgOTf | - | TFA | 35 a |
| 37 [70] | 4-CH3 | Et | Et | PtCl2/AgOTf | - | TFA | 35 |
| 38 [70] | 4-CH3 | Et | H | K2PtCl4/AgOTf | - | TFA | 51 |
| 39 [69] | 3,5-di-CH3 | H | H | Pd(OAc)2 | - | TFA | 31 |
| 40 [71] | 3,5-di-CH3 | H | H | FeCl3/AgOTf | - | TFA/DCE | 85 |
| 41 [69] | 3,5-di-CH3 | H | H | Pd(OAc)2 | - | TFA | 49 |
| 42 [70] | 3,5-di-CH3 | H | Ph | K2PtCl4/AgOTf | - | TFA | 50 |
| 43 [70] | 3,5-di-CH3 | Et | Et | PtCl2/AgOTf | - | TFA | 37 |
| 44 [70] | 3,4-di-CH3 | H | Ph | K2PtCl4/AgOTf | - | TFA | 58 |
| 45 [70] | 3,4-di-CH3 | Et | Et | PtCl2/AgOTf | - | TFA | 45 |
| 46 [71] | 3,4,5-tri-CH3 | H | H | FeCl3/AgOTf | - | TFA/DCE | 88 |
| 47 [71] | 4-t-Bu | H | H | FeCl3/AgOTf | - | TFA/DCE | 62 |
| 48 [67] | 4-t-Bu | H | Ph | Pd(OAc)2 | - | TFA/DCM | 60 |
| 49 [67] | 4-t-Bu | H | CH3 | Pd(OAc)2 | - | TFA/DCM | 50 |
| 50 [67] | 4-t-Bu | H | CH3 | Pd(OAc)2 | - | TFA/DCM | 12 |
| 51 [67] | 4-t-Bu | Et | H | Pd(OAc)2 | - | TFA/DCM | 71 |
| 52 [67] | 4-t-Bu | Et | H | Pd(OAc)2 | - | TFA/DCM | 51 |
| 53 [72] | 4-t-Bu | Et | H | Pd(OAc)2 | - | TFA/DCM | 62 |
| 54 [70] | 1-naphthol | Et | n-C5H11 | K2PtCl4/AgOTf | - | TFA | 77 |
| 55 [70] | 1-naphthol | H | n-C5H11 | K2PtCl4/AgOTf | - | TFA | 28 |
| 56 [71] | 1-naphthol | H | n-C5H11 | FeCl3/AgOTf | - | TFA/DCE | 87 |
| 57 [70] | 4-Br | Et | H | K2PtCl4/AgOTf | - | TFA | 7 |

| Reaction Product | R1 | R2 | Metal Source | Temperature/Time | Yield (%) | References |
|---|---|---|---|---|---|---|
 | 6-t-Bu | Ph | Pd(OAc)2 | rt/0.5 h | 70 | [72] |
| 6-t-Bu | Ph | AuCl3/3AgOTf | 50 °C/48 h | 92 | [81] | |
| 6-t-Bu | CH3 | Pd(OAc)2 | rt/0.5 h | 90 | [72] | |
| 6-t-Bu | C6H11 | Pd(OAc)2 | rt/0.5 h | 71 | [72] | |
| 6-t-Bu | H | Pd(OAc)2 | rt/0.5 h | 60 | [72] | |
| 6-t-Bu | H | AuCl3/3AgOTf | 50 °C/48 h | 99 | [81] | |
| 6-t-Bu | Ph | Pd(PPh3)4 | rt/1.0 h | 90 | [72] | |
| 6-t-Bu | Ph | FeCl3 | 80 °C/72 h | 53 | [74] | |
| 5,7-di-CH3 | Ph | Pd(OAc)2 | rt/0.5 h | 87 | [72] | |
| 5,6,7-tri-OCH3 | Ph | Pd(OAc)2 | rt/0.5 h | 91 | [72] | |
| 6-Br-5,7-di-CH3 | Ph | Pd(OAc)2 | rt/0.5 h | 85 | [72] | |
| 7- CH3 | Ph | Pd(OAc)2 | rt/0.5 h | 78 a | [72] | |
| 6- CH3 | Ph | Pd(OAc)2 | rt/10 h | 50 | [72] | |
| 5- CH3-8-i-Pr | Ph | Pd(OAc)2 | rt/1.0 h | 75 | [72] | |
| 8- CH3 | Ph | Pd(OAc)2 | rt/5.0 h | 79 a | [72] | |
| 8- CHO-5-OCH3 | Ph | Pd(OAc)2 | rt/5.0 h | 70 | [73] | |
| 5,7-di-CH3 | CH3 | PtCl4 | 70 °C/36h | 73 | [75] | |
| 5,7-di-CH3 | CH3 | PtCl2 | 70 °C/36h | 28 | [75] | |
| 7- OCH3 | Ph | PtCl4 | 70 °C/24 h | 50 | [76] | |
| 6- CH3 | H | Au(I) b | 18 °C/1.0 h | 60 | [77] | |
| 6- OCH3 | H | Au(I) b | 18 °C/1.0 h | 94 | [77] | |
| 6- OCH3 | CH3 | Au(PPh3)Cl/AgSbF6 | rt/20 h | 48 | [78] | |
| 6- OCH3 | CH3 | Au(PPh3)Cl/AgOTf | rt/20 h | 85 | [78] | |
| 6- OCH3 | Ph | AuCl3/3AgOTf | 50 °C/48 h | 98 | [81] | |
| 7-N(Et)2 | Ph | HAuCl4 | rt/5 h | 70 | [80] | |
| H | H | AuCl3/3AgOTf | 50 °C/48 h | 84 | [81] | |
| H | CH3 | AuCl3/3AgOTf | 50 °C/48 h | 99 | [81] | |
| H | Ph | AuCl3/3AgOTf | 50 °C/48 h | 73 | [81] | |
| 6-Ph | Ph | AuCl3/3AgOTf | 70 °C/48 h | 82 | [81] | |
| 6-Br | Ph | AuCl3/3AgOTf | 70 °C/48 h | 44 | [81] | |
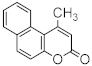 | Pd(OAc)2 | rt/0.5 h | 65 | [72] | ||
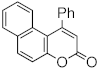 | AuCl3/3AgOTf | 50 °C/48 h | 99 | [81] | ||
 | Pd(OAc)2 | rt/0.5 h | 75 a | [72] | ||
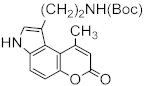 | Au(PPh3)Cl/AgSbF6 | rt/20 h | 93 | [79] | ||
| PtCl4 | 80 °C/20 h | 54 | [79] | |||

| Reaction Product | R1 | R2 | Metal Source/Ligand | Oxidant | Yield (%) | References |
|---|---|---|---|---|---|---|
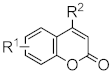 | H | CH3 | Pd(OAc)2/dppb | 1,4-BQ | 75 | [89] |
| H | CH3 | Pd(OAc)2/TMEDA | 1,4-BQ | 61 | [89] | |
| H | CH3 | [Cp*RhCl2]2 | Cu(OAc)2 | 84 | [90] | |
| H | Ph | Pd(OAc)2/dppb | 1,4-BQ | 85 | [89] | |
| H | Ph | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 75 | [91] | |
| H | p-CNC6H4 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 87 | [91] | |
| H | p-CF3C6H4 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 67 | [91] | |
| H | H | Pd(OAc)2/dppb | 1,4-BQ | 58 | [89] | |
| H | H | [Cp*RhCl2]2 | Cu(OAc)2 | 69 | [90] | |
| H | H | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 74 | [91] | |
| H | C2H5 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 67 | [91] | |
| 6-OCH3 | H | [Cp*RhCl2]2 | Cu(OAc)2 | 85 | [90] | |
| 6-OCH3 | H | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 31 | [91] | |
| 6-CO2CH3 | H | [Cp*RhCl2]2 | Cu(OAc)2 | 78 | [90] | |
| 6-CH3 | H | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 35 | [91] | |
| 5- CH3 | H | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 33 | [91] | |
| 6-Cl | H | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 35 | [91] | |
| 6-Br | H | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 47 | [91] | |
| 6-NO2 | H | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 64 | [91] | |
| 7-OCH3 | H | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 61 | [91] | |
| 7-Br | H | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 71 | [91] | |
| 6-Cl | CH3 | Pd(OAc)2/dppb | 1,4-BQ | 56 | [89] | |
| 6-Cl | CH3 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 83 | [91] | |
| 6-Br | CH3 | Pd(OAc)2/dppb | 1,4-BQ | 49 | [89] | |
| 6-F | CH3 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 69 | [91] | |
| 7-F | CH3 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 77 | [91] | |
| 6-NO2 | CH3 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 60 | [91] | |
| 7- CH3 | CH3 | Pd(OAc)2/dppb | 1,4-BQ | 67 | [89] | |
| 6- CH3 | CH3 | Pd(OAc)2/dppb | 1,4-BQ | 70 | [89] | |
| 6- CH3 | CH3 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 53 | [91] | |
| 5-OC2H5 | CH3 | Pd(OAc)2/dppb | 1,4-BQ | 52 | [89] | |
| 6-Cl,7-CH3 | CH3 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 73 | [91] | |
| 6,7-diCH3 | CH3 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 60 | [91] | |
| 7-OC2H5, 8-CH3 | CH3 | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 55 | [91] | |
| 7-OCH3 | Ph | Cp*Co(CO)I2 | Ag2CO3/Cu(OAc)2 | 79 | [91] | |
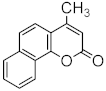 | Pd(OAc)2/dppb | 1,4-BQ | 34 | [89] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szwaczko, K. Coumarins Synthesis and Transformation via C–H Bond Activation—A Review. Inorganics 2022, 10, 23. https://doi.org/10.3390/inorganics10020023
Szwaczko K. Coumarins Synthesis and Transformation via C–H Bond Activation—A Review. Inorganics. 2022; 10(2):23. https://doi.org/10.3390/inorganics10020023
Chicago/Turabian StyleSzwaczko, Katarzyna. 2022. "Coumarins Synthesis and Transformation via C–H Bond Activation—A Review" Inorganics 10, no. 2: 23. https://doi.org/10.3390/inorganics10020023
APA StyleSzwaczko, K. (2022). Coumarins Synthesis and Transformation via C–H Bond Activation—A Review. Inorganics, 10(2), 23. https://doi.org/10.3390/inorganics10020023






