The Efficacy of Photobiomodulation Therapy in Improving Tissue Resilience and Healing of Radiation Skin Damage
Abstract
1. Introduction
2. Materials and Methods
2.1. Iodine Seeds
2.2. Animal Procedures
2.3. Photobiomodulation (PBM) Therapy
2.4. Wound-Image Analyses
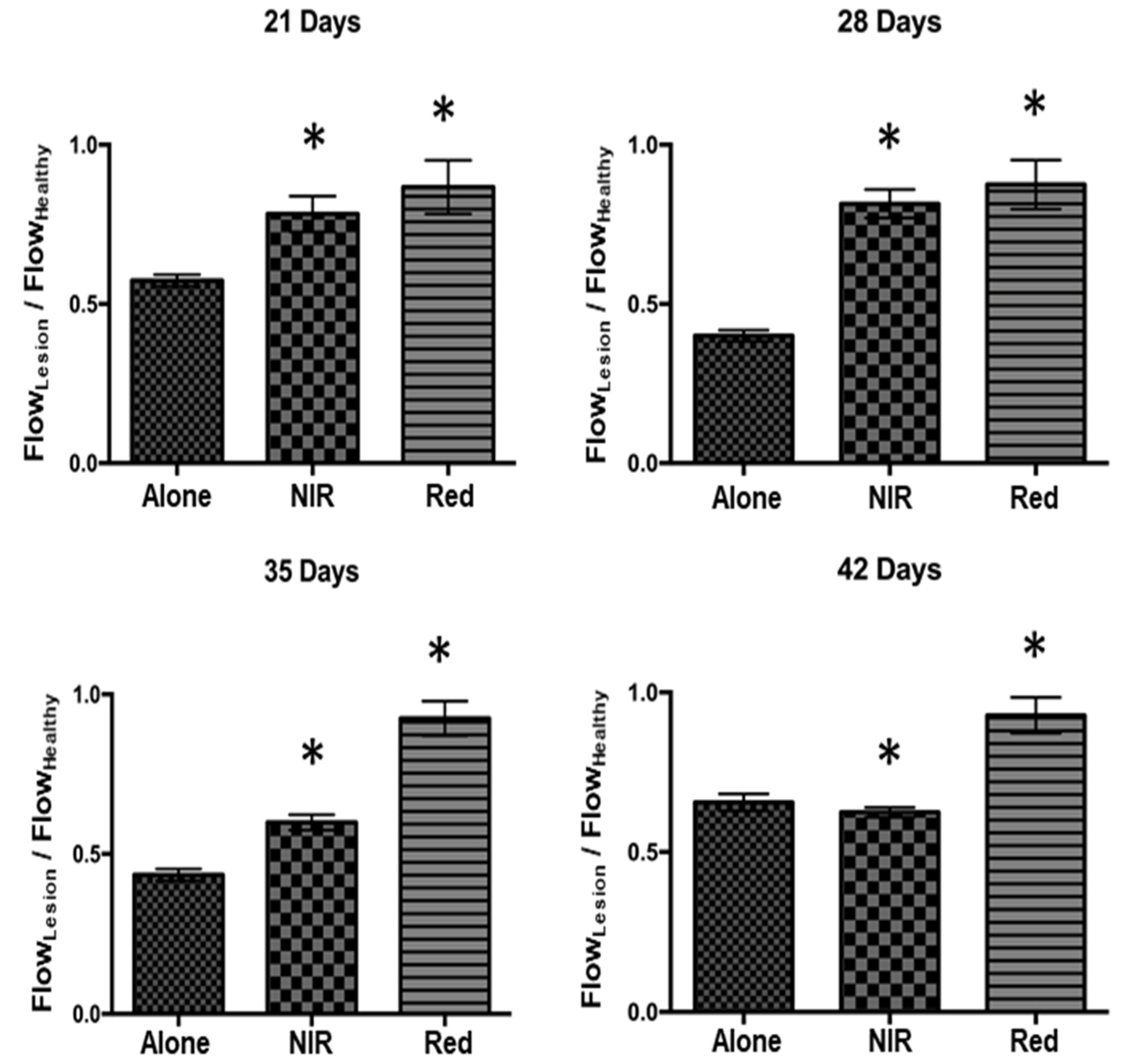
2.5. Tissue Perfusion Analysis
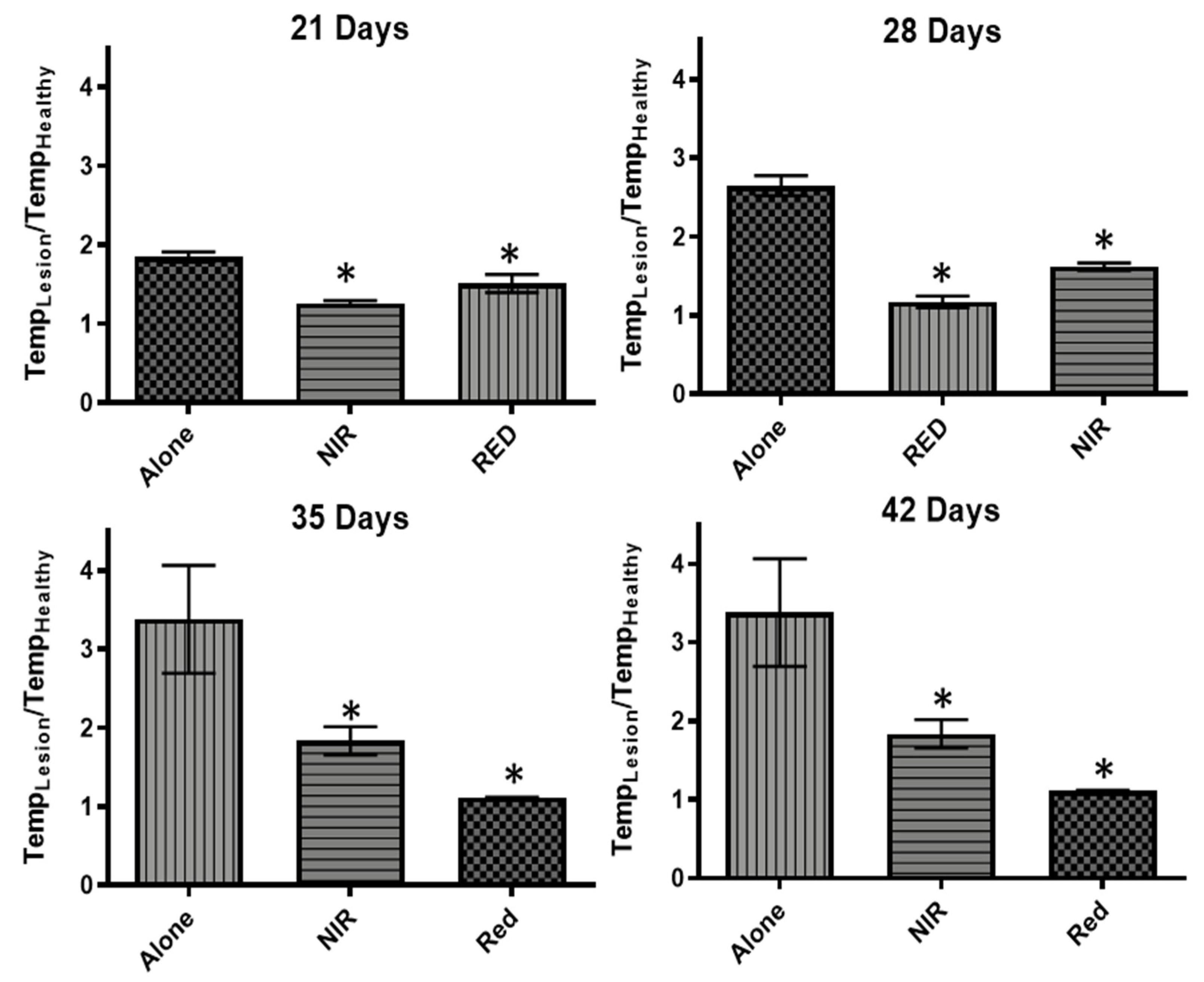
2.6. Tissue Temperature to Assess Inflammation
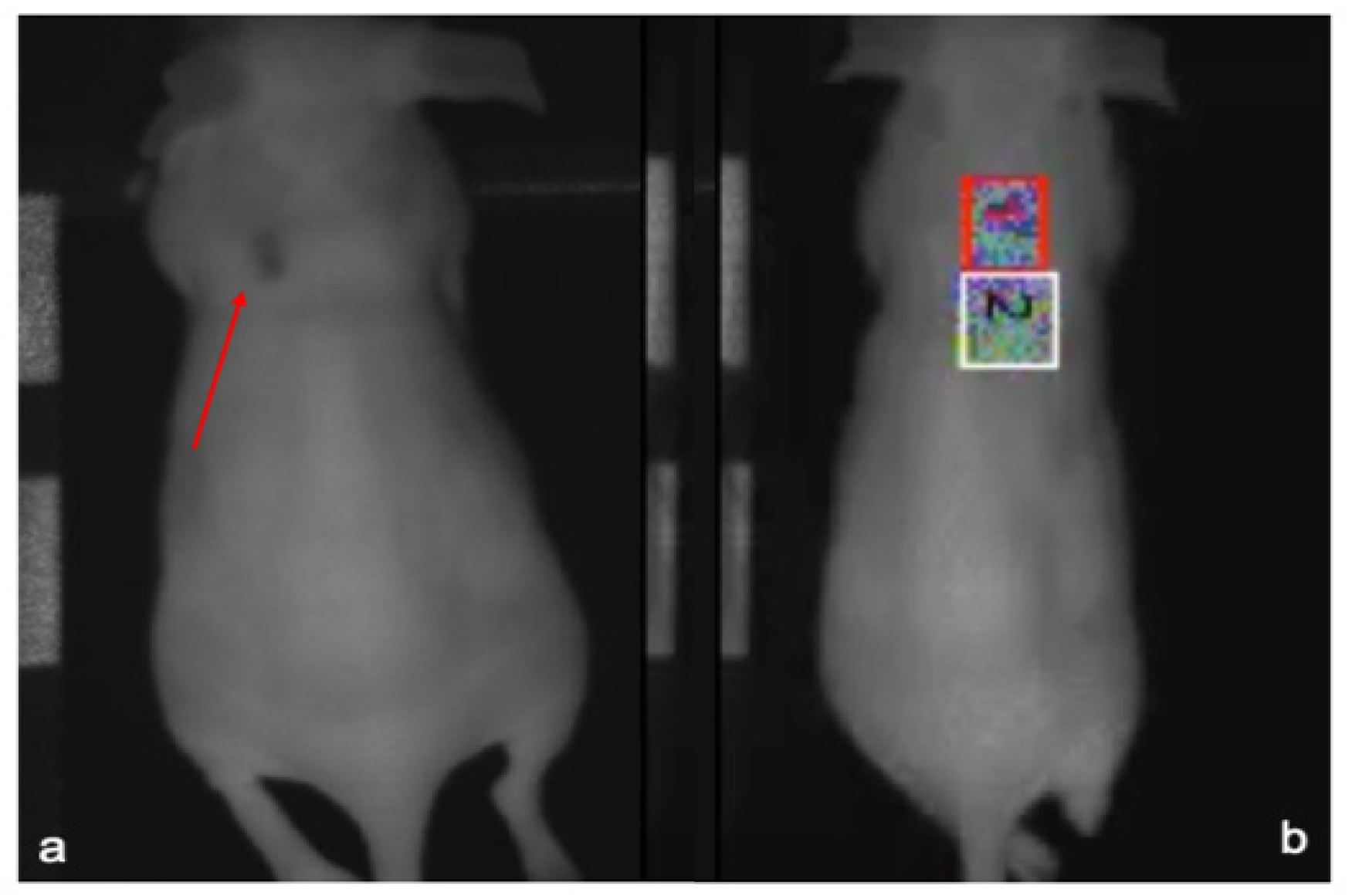
2.7. Metabolic Analysis with Micro-Positron Emission Tomography (µPET-CT) Imaging
2.8. Histology
2.9. Statistical Analysis
3. Results
3.1. Radionecrosis Lesions and PBM Treatments
3.2. Tissue Perfusion Analysis
3.3. Thermal Tissue Imaging for Inflammation
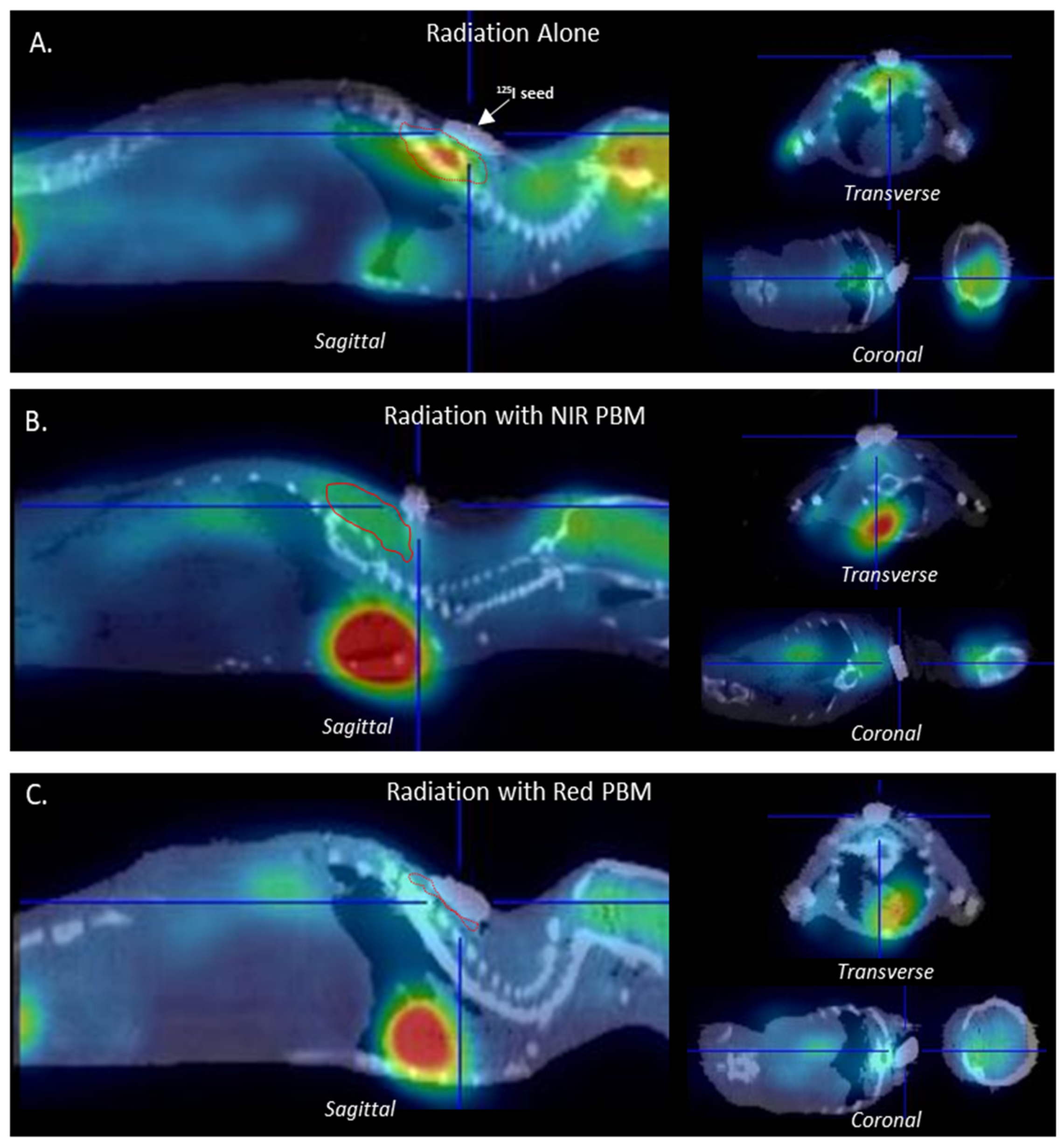
3.4. Metabolic Tissue Analysis Using µPET-CT Imaging
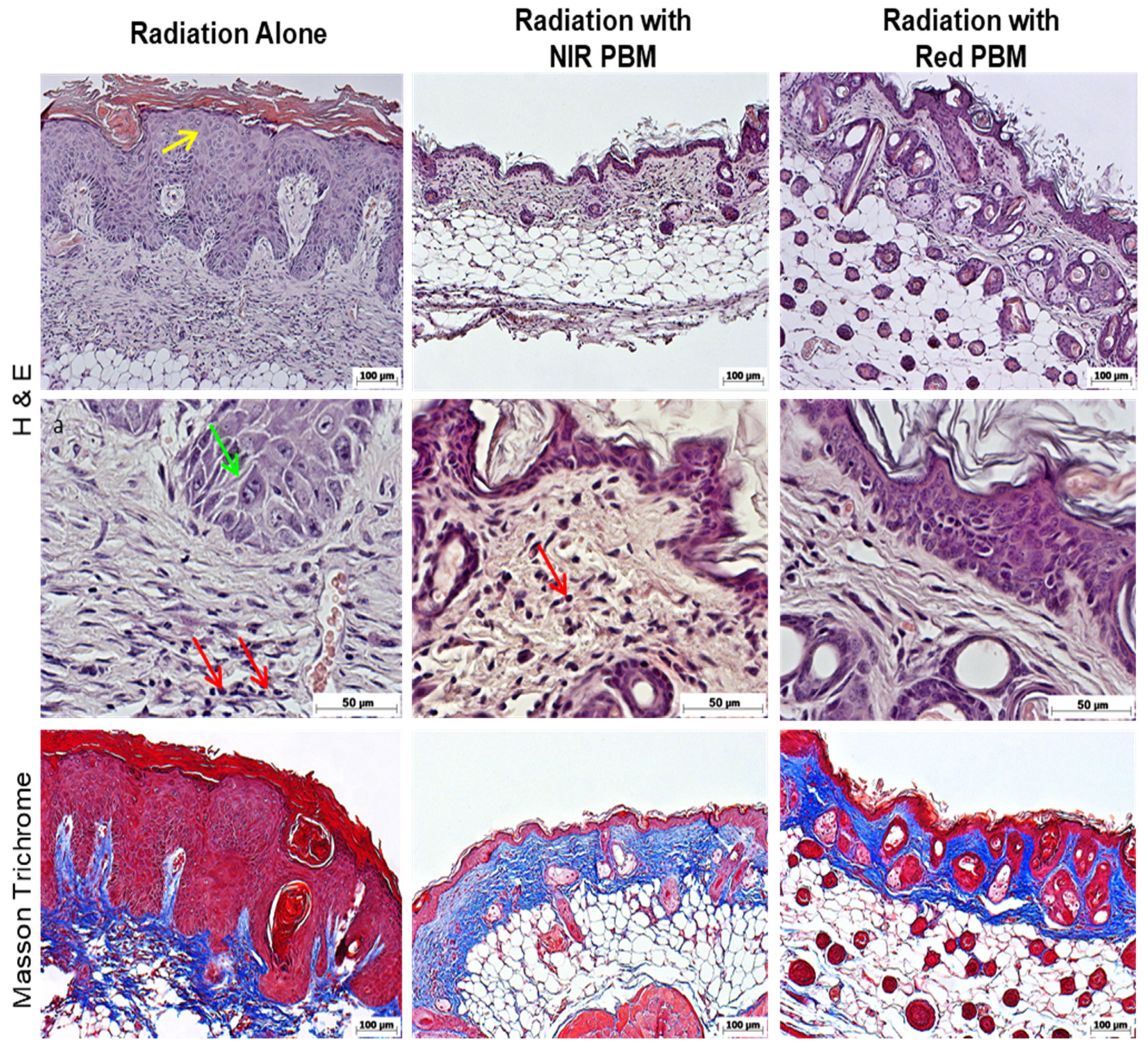
3.5. Histology Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Jham, B.C.; Freire, A.R.S. Oral complications of radiotherapy in the head and neck. Rev. Bras. Otorrinolaringol. 2006, 72, 704–708. [Google Scholar] [CrossRef]
- Cotran, R.S.; Kumar, V.; Collins, T. Robbins Pathologic Basis of Disease, 6th ed.; W.B. Saunders Company: Newberg, OR, USA, 1999; pp. 154–196. [Google Scholar]
- Isser, A.; Vier-Pelisser, F.B.; Fontanella, V.R.C.; De Figueiredo, M.A.Z. Análise Microscópica do Efeito da Radioterapia Fracionada por Cobalto-60 em Mandíbula de Rato. Radiol. Bras. 2007, 40, 113–118. [Google Scholar]
- Okuno, E.; Caldas, I.L.; Chow, C. Física Para Ciências Biológicas e Biomédicas; Harper & Row do Brasil: São Paulo, Brazil, 1982. [Google Scholar]
- Thomadsen, B.R.; Erickson, B.A.; Eifel, P.J.; Hsu, I.C.; Patel, R.R.; Petereit, D.G.; Fraass, B.A.; Rivard, M.J. A review of safety, quality management, and practice guidelines for high-dose-rate brachytherapy: Executive summary. Pract. Radiat. Oncol. 2014, 4, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Peschel, R.; Chen, Z.; Roberts, K.; Nath, R. Long-term complications with prostate implants: Iodine-125 vs. Palladium-103. Rad. Oncol. Inv. 1999, 7, 278–288. [Google Scholar] [CrossRef]
- Barros, P.B.; Leal, P.R.A. Ulceras Complexas por Radionecrose-Fisiopatologia, Diagnóstico e Tratamento. Rev. Bras. Cir. Plást. 2003, 18, 17–26. [Google Scholar]
- Akita, S.; Akino, K.; Hirano, A.; Ohtsuru, A.; Yamashita, S. Noncultured autologous adipose-derived stem cellsbarre therapy for chronic radiation injury. Stem. Cells Int. 2010, 2010, 532704. [Google Scholar] [CrossRef]
- Belcher, A.E.; Selekof, J. Skin care for the oncology patient. In Chronic Wound Care: A Clinical Source Book for Healthcare Professionals, 3rd ed.; Krasner, D.L., Rodheaver, G.T., Sibbald, R.G., Eds.; HMP Communications: Wayne, PA, USA, 2001; pp. 711–720. [Google Scholar]
- Rudolph, R. Radiation ulcers. In Chronic Problems in Wounds; Rudolph, R., Noe, J.M., Eds.; Little, Brown & Co.: Boston, MA, USA, 1983; pp. 715–716. [Google Scholar]
- Mosca, R.C.; Ferreira, D.C.; Napolitano, C.M.; Santin, S.P.; Dornelles, L.D.P.; Alvarenga, E.O.; Mathor, M.B. An athymic mouse model to mimic cutaneous radiation injury by Cobalt-60. In Proceedings of the 2013 International Nuclear Atlantic Conference, Associação Brasileira De Energia Nuclear-Aben, Recife, Brazil, 24–29 November 2013; pp. 1–9. [Google Scholar]
- Mosca, R.C.; Steffens, D.; Pelissari, C.; Mantesso, A.; Mathor, M.B. Full-thickness tissue engineered development using human keratinocyte and adipose tissue derived mesenchymal stem cell. In Proceedings of the 9th World Congress on Alternative and Animal Use in the Life Science, Prague, Czech Republic, 24–28 August 2014; Altex Proceedings. Springer: Swiss, 2014; p. 230. [Google Scholar]
- Rifkin, L.H.; Stojadinovic, S.; Stewart, C.H.; Song, K.H.; Maxted, M.C.; Bell, M.H.; Kashefi, N.S.; Speiser, M.P.; Saint-Cyr, M.; Story, M.D.; et al. An athymic rat model of cutaneous radiation injury designed to study human tissue-based wound therapy. Radiat. Oncol. 2012, 8, 68. [Google Scholar] [CrossRef]
- Gupta, A.; Dai, T.; Hamblin, M.R. Effect of red and near-infrared wavelengths on low-level laser (light) therapy-induced healing of partial-thickness dermal abrasion in mice. Lasers Med. Sci. 2014, 29, 257–265. [Google Scholar] [CrossRef]
- Mosca, R.C.; Ribeiro, M.S.; Zeituni, C.A.; Souza, C.D. Low-level laser therapy to treat cutaneous radionecrosis induced by 125I seed in a murine model. In Proceedings of the 15th Congress of the World Federation for Laser Dentistry (WFLD 2016), Nagoya, Japan, 17–19 July 2016; Journal of Japanese Society for Laser Dentistry: Nagoya, Japan, 2016; Volume 27, p. 117. [Google Scholar]
- Hamblin, M.R.; Zahra, T.; Contag, C.H.; Mcmanus, A.T.; Hasan, T. Optical monitoring and treatment of potentially lethal wound infections in vivo. J. Infect. Dis. 2003, 187, 1717–1725. [Google Scholar] [CrossRef]
- Anders, J.J.; Lanzafame, R.J.; Arany, P.R. Low-level light/laser therapy versus photobiomodulation therapy. Photomed. Laser Surg. 2015, 33, 183–184. [Google Scholar] [CrossRef]
- Mester, E.; Spiry, T.; Szende, B.; Tota, J.G. Effect of laser rays on wound healing. Am. J. Surg. 1971, 122, 532–535. [Google Scholar] [CrossRef]
- Posten, W.; Wrone, D.A.; Dover, J.S.; Arndt, K.A.; Silapunt, S.; Alam, M. Low-level laser therapy for wound healing: Mechanism and efficacy. Dermatol. Surg. 2005, 31, 334–340. [Google Scholar] [CrossRef]
- Mosca, R.C.; Ong, A.A.; Albasha, O.; Bass, K.; Arany, P. Photobiomodulation Therapy for Wound Care: A Potent, Noninvasive, Photoceutical Approach. Adv. Skin Wound Care 2019, 32, 157–167. [Google Scholar] [CrossRef]
- Karu, T.I. Multiple roles of cytochrome c oxidase in mammalian cells under action of red and IR-A radiation. IUBMB Life 2010, 62, 607–610. [Google Scholar] [CrossRef] [PubMed]
- Arany, P.R. Craniofacial Wound Healing with Photobiomodulation Therapy: New Insights and Current Challenges. J. Dent. Res. 2016, 95, 977–984. [Google Scholar] [CrossRef]
- Arany, P.R.; Cho, A.; Hunt, T.D.; Sidhu, G.; Shin, K.; Hahm, E.; Huang, G.X.; Weaver, J.; Chen, A.C.; Padwa, B.L.; et al. Photoactivation of endogenous latent transforming growth factor-β1 directs dental stem cell differentiation for regeneration. Sci. Transl. Med. 2014, 6, 238ra69. [Google Scholar] [CrossRef]
- Zadik, Y.; Arany, P.R.; Fregnani, E.R.; Bossi, P.; Antunes, H.S.; Bensadoun, R.J.; Gueiros, L.A.; Majorana, A.; Nair, R.G.; Ranna, V.; et al. Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO). Systematic review of photobiomodulation for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer 2019, 27, 3969–3983. [Google Scholar] [CrossRef]
- Núñez, S.C.; Nogueira, G.E.; Ribeiro, M.S.; Garcez, A.S.; Lage-Marques, J.L. He-Ne laser effects on blood microcirculation during wound healing: A method of in vivo study through laser Doppler flowmetry. Lasers Surg. Med. 2004, 35, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Chaves, M.E.; Araújo, A.R.; Piancastelli, A.C.; Pinotti, M. Effects of low-power light therapy on wound healing: LASER × LED. An. Bras. Dermatol. 2014, 89, 616–623. [Google Scholar] [CrossRef]
- De Oliveira, R.A.; Boson, L.L.B.; Portela, S.M.M.; Filho, A.L.M.M.; De Oliveira Santiago, D. Low-intensity LED therapy (658 nm) on burn healing: A series of cases. Lasers Med. Sci. 2018, 33, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Brassolatti, P.; De Andrade, A.L.M.; Bossini, P.S.; Otterço, A.N.; Parizotto, N.A. Evaluation of the low-level laser therapy application parameters for skin burn treatment in experimental model: A systematic review. Lasers Med. Sci. 2018, 33, 1159–1169. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Shi, X.; Chen, Y.; Huang, W.; Song, Q.; Lin, X.; Liu, Y.; Chen, K.; Wang, Z. Functional imaging of interstitial brachytherapy in pancreatic carcinoma xenografts using spectral CT: How does iodine concentration correlate with standardized uptake value of (18)FDG-PET-CT? Br. J. Radiol. 2016, 89, 20150573. [Google Scholar] [CrossRef]
- Garduño-Ramón, M.A.; Vega-Mancilla, S.G.; Morales-Henández, L.A.; Osornio-Rios, R.A. Supportive Noninvasive Tool for the Diagnosis of Breast Cancer Using a Thermographic Camera as Sensor. Sensors 2017, 3, 17. [Google Scholar] [CrossRef]
- Khan, I.; Tang, E.; Arany, P. Molecular pathway of near-infrared laser phototoxicity involves ATF-4 orchestrated ER stress. Sci. Rep. 2015, 5, 10581. [Google Scholar] [CrossRef] [PubMed]
- Anders, J.J.; Moges, H.; Wu, X.; Erbele, I.D.; Alberico, S.L.; Saidu, E.K.; Smith, J.T.; Pryor, B.A. In vitro and in vivo optimization of infrared laser treatment for injured peripheral nerves. Lasers Surg. Med. 2014, 46, 34–45. [Google Scholar] [CrossRef]
- Huang, Y.Y.; Sharma, S.K.; Carroll, J.; Hamblin, M.R. Biphasic dose response in low level light therapy—An update. Dose Response 2011, 9, 602–618. [Google Scholar] [CrossRef] [PubMed]
- Karu, T.I.; Kolyakov, S.F. Exact action spectra for cellular responses relevant to phototherapy. Photomed Laser Surg. 2005, 23, 355–361. [Google Scholar] [CrossRef]
- Lanzafame, R.J.; Stadler, I.; Kurtz, A.F.; Connelly, R.; Peter, T.A.; Brondon, P.; Olson, D. Reciprocity of exposure time and irradiance on energy density during photoradiation on wound healing in a murine pressure ulcer model. Lasers Surg. Med. 2007, 39, 534–542. [Google Scholar] [CrossRef]
- Oron, U.; Yaakobi, T.; Oron, A.; Hayam, G.; Gepstein, L.; Rubin, O.; Wolf, T.; Haim, S.B. Attenuation of infarct size in rats and dogs after myocardial infarction by low-energy laser irradiation. Lasers Surg. Med. 2001, 28, 204–211. [Google Scholar] [CrossRef]
- Li, J.; Xiao, Y. Application of FDG-PET/CT in Radiation Oncology. Front. Oncol. 2013, 11, 80. [Google Scholar] [CrossRef]
- Tang, T.T.; Rendon, D.A.; Zawaski, J.A.; Afshar, S.F.; Kaffes, C.K.; Sabek, O.M.; Gaber, M.W. Imaging Radiation-Induced Gastrointestinal, Bone Marrow Injury and Recovery Kinetics Using 18F-FDG PET. PLoS ONE 2017, 12, e0169082. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.; Visweswaran, S.; Kanagaraj, K.; Raavi, V.; Arunan, M.; Venkatachalapathy, E.; Paneerselvam, S.; Jose, M.T.; Ozhimuthu, A.; Perumal, V. 18F-FDG PET/CT scanning: Biological effects on patients: Entrance surface dose, DNA damage, and chromosome aberrations in lymphocytes. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2019, 838, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Arany, P.R. Healing Tumors with Light: Science Fiction or the Future of Medicine? Photomed. Laser Surg. 2018, 36, 227–229. [Google Scholar] [CrossRef] [PubMed]
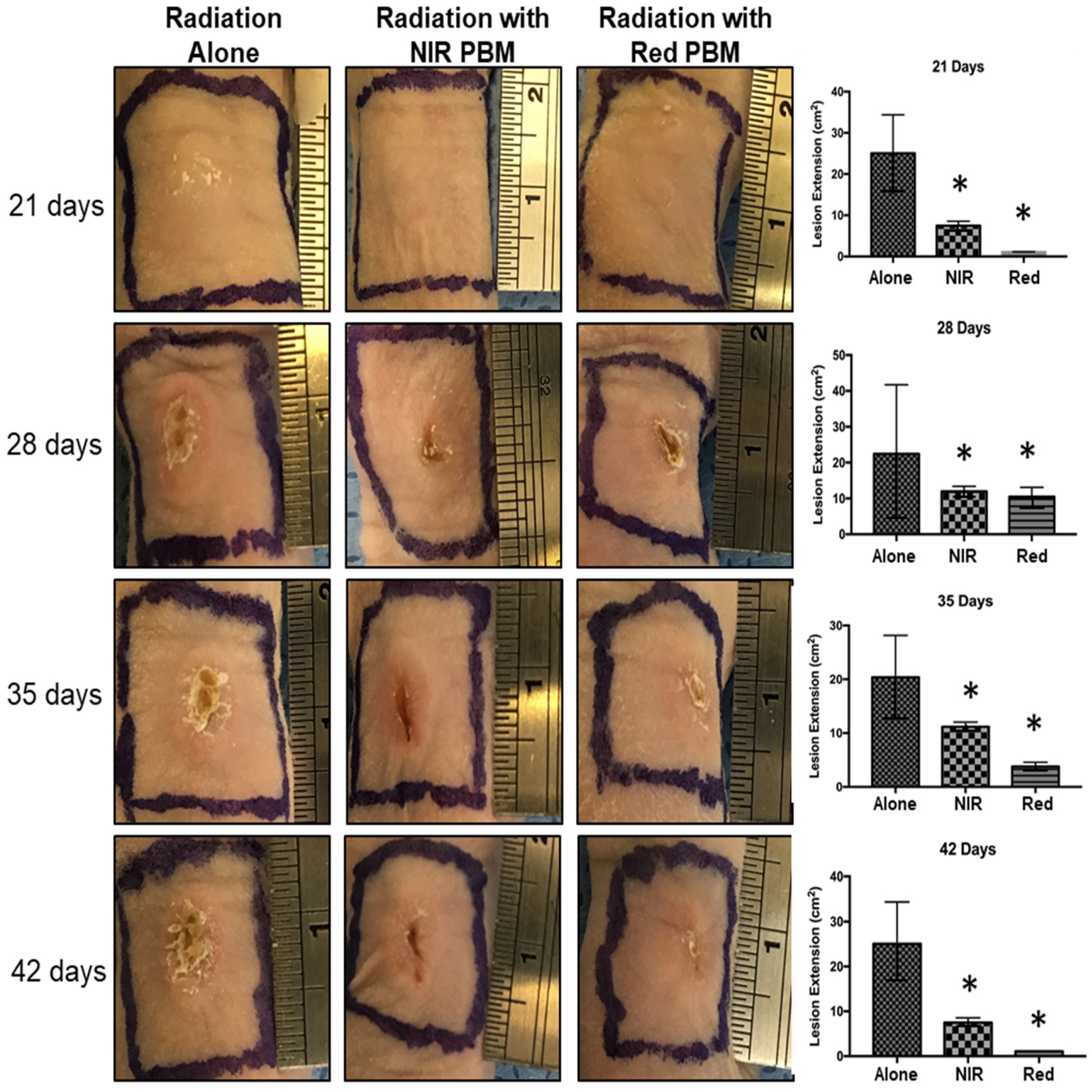
| Radionecrosis (Days Post 125I Seeding) | Radiation Alone Group | Radiation NIR-PBM Group | Radiation Red-PBM Group |
|---|---|---|---|
| First Sign | 21 | 21 | 21 |
| Maximum Severity | 42 | 35 | 28 |
| Healing | 61 | 49 | 42 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mosca, R.C.; Santos, S.N.; Nogueira, G.E.C.; Pereira, D.L.; Costa, F.C.; Pereira, J.X.; Zeituni, C.A.; Arany, P.R. The Efficacy of Photobiomodulation Therapy in Improving Tissue Resilience and Healing of Radiation Skin Damage. Photonics 2022, 9, 10. https://doi.org/10.3390/photonics9010010
Mosca RC, Santos SN, Nogueira GEC, Pereira DL, Costa FC, Pereira JX, Zeituni CA, Arany PR. The Efficacy of Photobiomodulation Therapy in Improving Tissue Resilience and Healing of Radiation Skin Damage. Photonics. 2022; 9(1):10. https://doi.org/10.3390/photonics9010010
Chicago/Turabian StyleMosca, Rodrigo Crespo, Sofia Nascimento Santos, Gesse Eduardo Calvo Nogueira, Daisa Lima Pereira, Francielli Campos Costa, Jonathas Xavier Pereira, Carlos Alberto Zeituni, and Praveen Ravindra Arany. 2022. "The Efficacy of Photobiomodulation Therapy in Improving Tissue Resilience and Healing of Radiation Skin Damage" Photonics 9, no. 1: 10. https://doi.org/10.3390/photonics9010010
APA StyleMosca, R. C., Santos, S. N., Nogueira, G. E. C., Pereira, D. L., Costa, F. C., Pereira, J. X., Zeituni, C. A., & Arany, P. R. (2022). The Efficacy of Photobiomodulation Therapy in Improving Tissue Resilience and Healing of Radiation Skin Damage. Photonics, 9(1), 10. https://doi.org/10.3390/photonics9010010





