A Novel Microalgae Harvesting Method Using Laser Micromachined Glass Fiber Reinforced Polymers
Abstract
1. Introduction
2. Methods
2.1. Materials
2.2. UV Laser Micromachining
2.3. Algal Cultivation
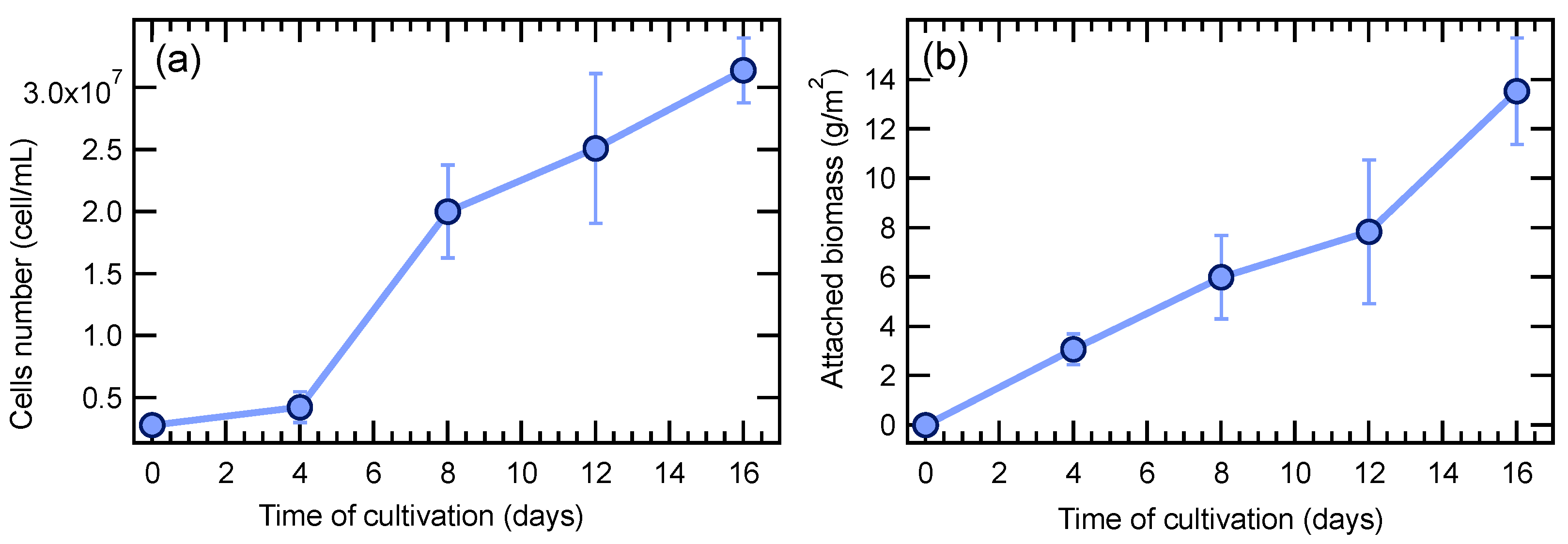
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hattab, M.A. Microalgae Harvesting Methods for Industrial Production of Biodiesel: Critical Review and Comparative Analysis. J. Fundam. Renew. Energy Appl. 2015, 5. [Google Scholar] [CrossRef]
- Mirhashemi, F.S.; Sadrnia, H. NOX emissions of compression ignition engines fueled with various biodiesel blends: A review. J. Energy Inst. 2020, 93, 129–151. [Google Scholar] [CrossRef]
- Dincer, K. Lower Emissions from Biodiesel Combustion. Energy Sources Part A Recover. Util. Environ. Eff. 2008, 30, 963–968. [Google Scholar] [CrossRef]
- Tsavatopoulou, V.D.; Aravantinou, A.F.; Manariotis, I.D. Biofuel conversion of Chlorococcum sp. and Scenedesmus sp. biomass by one- and two-step transesterification. Biomass Convers. Biorefinery. 2019. [Google Scholar] [CrossRef]
- Kadir, W.N.A.; Lam, M.K.; Uemura, Y.; Lim, J.W.; Lee, K.T. Harvesting and pre-treatment of microalgae cultivated in wastewater for biodiesel production: A review. Energy Convers. Manag. 2018, 171, 1416–1429. [Google Scholar] [CrossRef]
- Aravantinou, A.F.; Theodorakopoulos, M.A.; Manariotis, I.D. Selection of microalgae for wastewater treatment and potential lipids production. Bioresour. Technol. 2013, 147, 130–134. [Google Scholar] [CrossRef]
- Tsavatopoulou, V.D.; Vakros, J.; Manariotis, I.D. Lipid conversion of Scenedesmus rubescens biomass into biodiesel using biochar catalysts from malt spent rootlets. J. Chem. Technol. Biotechnol. 2020. [Google Scholar] [CrossRef]
- Lv, J.; Guo, B.; Feng, J.; Liu, Q.; Nan, F.; Liu, X.; Xie, S. Integration of wastewater treatment and flocculation for harvesting biomass for lipid production by a newly isolated self-flocculating microalga Scenedesmus rubescens SX. J. Clean. Prod. 2019, 240, 118211. [Google Scholar] [CrossRef]
- Kumar, L.; Roy, A.; Saxena, G.; Kundu, K.; Bharadvaja, N. Isolation, Identification and biomass productivity analysis of microalga Scenedesmus rubescens from DTU Lake. J. Algal Biomass Util. 2017, 8, 56–67. [Google Scholar]
- Rinanti, A.; Purwadi, R. Harvesting of freshwater microalgae biomass by Scenedesmus sp. as bioflocculant. IOP Conf. Ser. Earth Environ. Sci. 2018, 106, 012087. [Google Scholar] [CrossRef]
- Vergini, S.; Aravantinou, A.F.; Manariotis, I.D. Harvesting of freshwater and marine microalgae by common flocculants and magnetic microparticles. J. Appl. Phycol. 2016, 28, 1041–1049. [Google Scholar] [CrossRef]
- Molina Grima, E.; Belarbi, E.-H.; Acién Fernández, F.G.; Robles Medina, A.; Chisti, Y. Recovery of microalgal biomass and metabolites: Process options and economics. Biotechnol. Adv. 2003, 20, 491–515. [Google Scholar] [CrossRef]
- Barros, A.I.; Gonçalves, A.L.; Simões, M.; Pires, J.C.M. Harvesting techniques applied to microalgae: A review. Renew. Sustain. Energy Rev. 2015, 41, 1489–1500. [Google Scholar] [CrossRef]
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Fasaei, F.; Bitter, J.H.; Slegers, P.M.; van Boxtel, A.J.B. Techno-economic evaluation of microalgae harvesting and dewatering systems. Algal Res. 2018, 31, 347–362. [Google Scholar] [CrossRef]
- Carbone, D.A.; Gargano, I.; Pinto, G.; De Natale, A.; Pollio, A. Evaluating microalgal attachment to surfaces: A first approach towards a laboratory integrated assessment. Chem. Eng. Trans. 2017, 57, 73–78. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Yeh, K.-L.; Aisyah, R.; Lee, D.-J.; Chang, J.-S. Cultivation, photobioreactor design and harvesting of microalgae for biodiesel production: A critical review. Bioresour. Technol. 2011, 102, 71–81. [Google Scholar] [CrossRef]
- Beach, E.S.; Eckelman, M.J.; Cui, Z.; Brentner, L.; Zimmerman, J.B. Preferential technological and life cycle environmental performance of chitosan flocculation for harvesting of the green algae Neochloris oleoabundans. Bioresour. Technol. 2012, 121, 445–449. [Google Scholar] [CrossRef]
- Wang, J.-H.; Zhuang, L.-L.; Xu, X.-Q.; Deantes-Espinosa, V.M.; Wang, X.-X.; Hu, H.-Y. Microalgal attachment and attached systems for biomass production and wastewater treatment. Renew. Sustain. Energy Rev. 2018, 92, 331–342. [Google Scholar] [CrossRef]
- Roostaei, J.; Zhang, Y.; Gopalakrishnan, K.; Ochocki, A.J. Mixotrophic Microalgae Biofilm: A Novel Algae Cultivation Strategy for Improved Productivity and Cost-efficiency of Biofuel Feedstock Production. Sci. Rep. 2018, 8, 12528. [Google Scholar] [CrossRef]
- Zhang, Q.; Yu, Z.; Zhu, L.; Ye, T.; Zuo, J.; Li, X.; Xiao, B.; Jin, S. Vertical-algal-biofilm enhanced raceway pond for cost-effective wastewater treatment and value-added products production. Water Res. 2018, 139, 144–157. [Google Scholar] [CrossRef]
- Thakur, P.; Teli, S.N.; Lad, S. Multiobjective Optimization in Drilling of Composites. In Proceedings of the International Conference on Intelligent Manufacturing and Automation, Wuhan, China, 16–18 October 2020; Vasudevan, H., Kottur, V.K.N., Raina, A.A., Eds.; Springer: Singapore, 2019; pp. 279–286, ISBN 9789811324895. [Google Scholar]
- Podola, B.; Li, T.; Melkonian, M. Porous Substrate Bioreactors: A Paradigm Shift in Microalgal Biotechnology? Trends Biotechnol. 2017, 35, 121–132. [Google Scholar] [CrossRef]
- Lien, N.T. The Cultivation of Spirulina Platensis on Vertical Aeroponic Substrates. JST 2018, 54, 307. [Google Scholar] [CrossRef]
- Altman, A.; Colwell, R.R. Agricultural Biotechnology; Books in Soils, Plants, and the Environment; Marcel Dekker: New York, NY, USA, 1998; ISBN 978-0-8247-9439-2. [Google Scholar]
- Orfanos, A.G.; Manariotis, I.D. Algal biofilm ponds for polishing secondary effluent and resource recovery. J. Appl. Phycol. 2019, 31, 1765–1772. [Google Scholar] [CrossRef]
- Sirmerova, M.; Prochazkova, G.; Siristova, L.; Kolska, Z.; Branyik, T. Adhesion of Chlorella vulgaris to solid surfaces, as mediated by physicochemical interactions. J. Appl. Phycol. 2013, 25, 1687–1695. [Google Scholar] [CrossRef]
- Li, Z.L.; Zheng, H.Y.; Lim, G.C.; Chu, P.L.; Li, L. Study on UV laser machining quality of carbon fibre reinforced composites. Compos. Part A Appl. Sci. Manuf. 2010, 41, 1403–1408. [Google Scholar] [CrossRef]
- Takahashi, K.; Tsukamoto, M.; Masuno, S.; Sato, Y. Heat conduction analysis of laser CFRP processing with IR and UV laser light. Compos. Part A Appl. Sci. Manuf. 2016, 84, 114–122. [Google Scholar] [CrossRef]
- Rice, E.W. (Ed.) Standard Methods for the Examination of Water and Wastewater, 22th ed.; American Public Health Association: Washington, DC, USA, 2012; ISBN 978-0-87553-013-0. [Google Scholar]
- Wang, S.-K.; Stiles, A.R.; Guo, C.; Liu, C.-Z. Microalgae cultivation in photobioreactors: An overview of light characteristics. Eng. Life Sci. 2014, 14, 550–559. [Google Scholar] [CrossRef]



| Time of Cultivation (Days) | Algae Biomass (g/m2) | UV Micromachined GFRP |
|---|---|---|
| 0 | 0 |  |
| 4 | 3.08 |  |
| 8 | 6.00 |  |
| 12 | 7.84 |  |
| 16 | 13.54 |  |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tourlouki, K.; Tsavatopoulou, V.; Alexandropoulos, D.; Manariotis, I.D.; Mazzucato, S. A Novel Microalgae Harvesting Method Using Laser Micromachined Glass Fiber Reinforced Polymers. Photonics 2020, 7, 42. https://doi.org/10.3390/photonics7020042
Tourlouki K, Tsavatopoulou V, Alexandropoulos D, Manariotis ID, Mazzucato S. A Novel Microalgae Harvesting Method Using Laser Micromachined Glass Fiber Reinforced Polymers. Photonics. 2020; 7(2):42. https://doi.org/10.3390/photonics7020042
Chicago/Turabian StyleTourlouki, Konstantina, Vasiliki Tsavatopoulou, Dimitris Alexandropoulos, Ioannis D. Manariotis, and Simone Mazzucato. 2020. "A Novel Microalgae Harvesting Method Using Laser Micromachined Glass Fiber Reinforced Polymers" Photonics 7, no. 2: 42. https://doi.org/10.3390/photonics7020042
APA StyleTourlouki, K., Tsavatopoulou, V., Alexandropoulos, D., Manariotis, I. D., & Mazzucato, S. (2020). A Novel Microalgae Harvesting Method Using Laser Micromachined Glass Fiber Reinforced Polymers. Photonics, 7(2), 42. https://doi.org/10.3390/photonics7020042





