Tuning of Fiber Optic Surface Reflectivity through Graphene Oxide-Based Layer-by-Layer Film Coatings
Abstract
1. Introduction
2. Materials and Methods
2.1. PEI/Graphene Oxide Deposition
Graphene Adsorption Kinetics
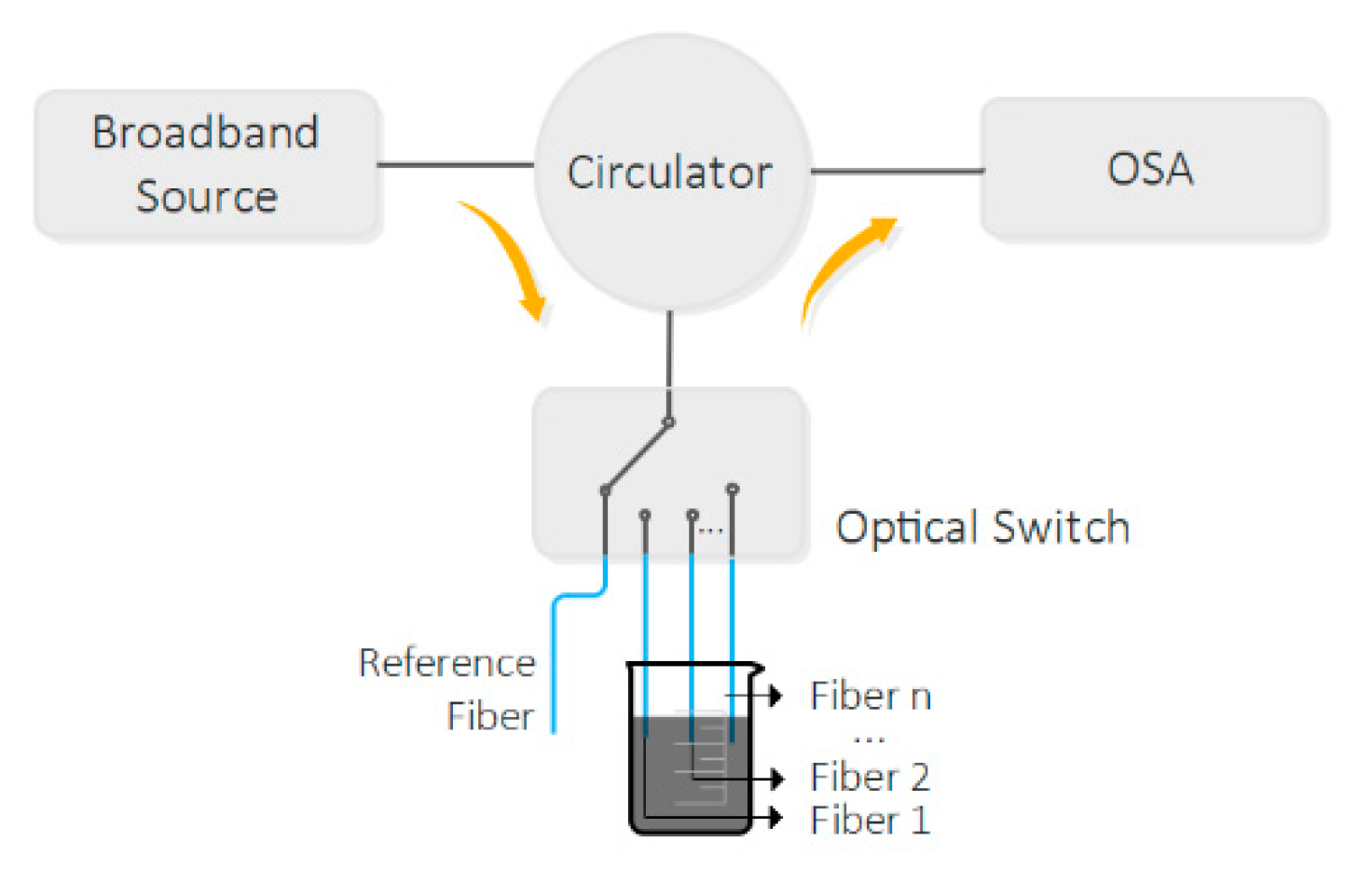
2.2. Hollow Microsphere Fabrication and Operation Principle
3. Results
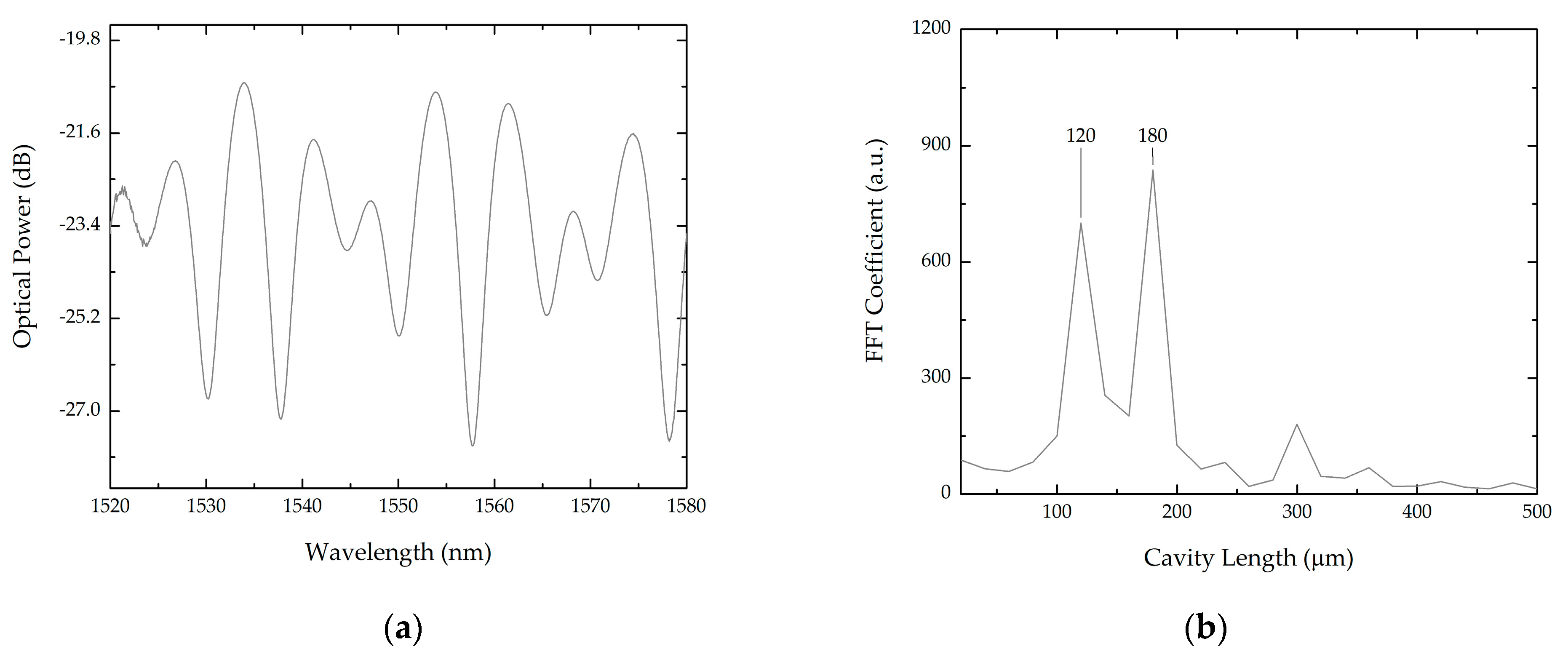
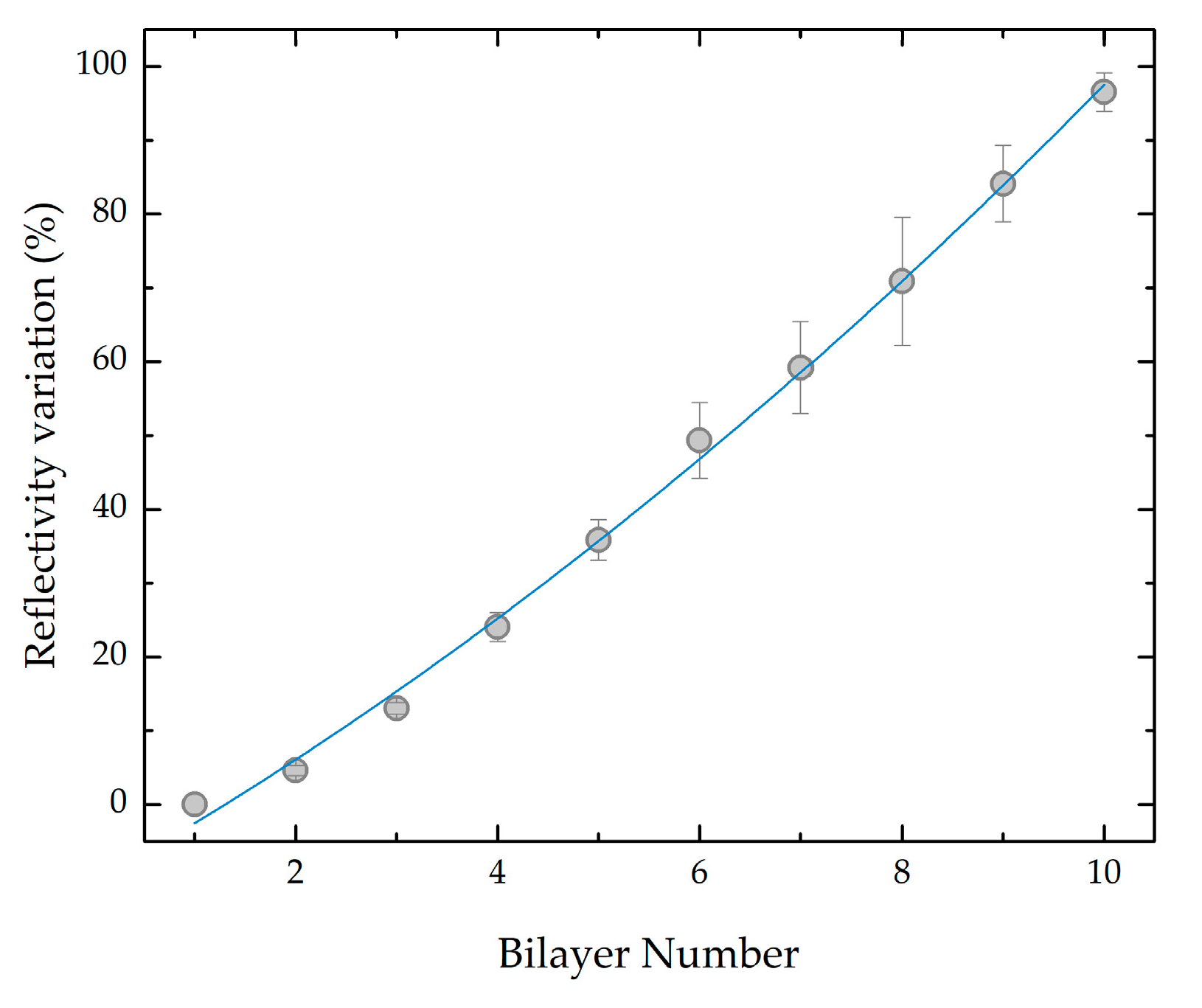
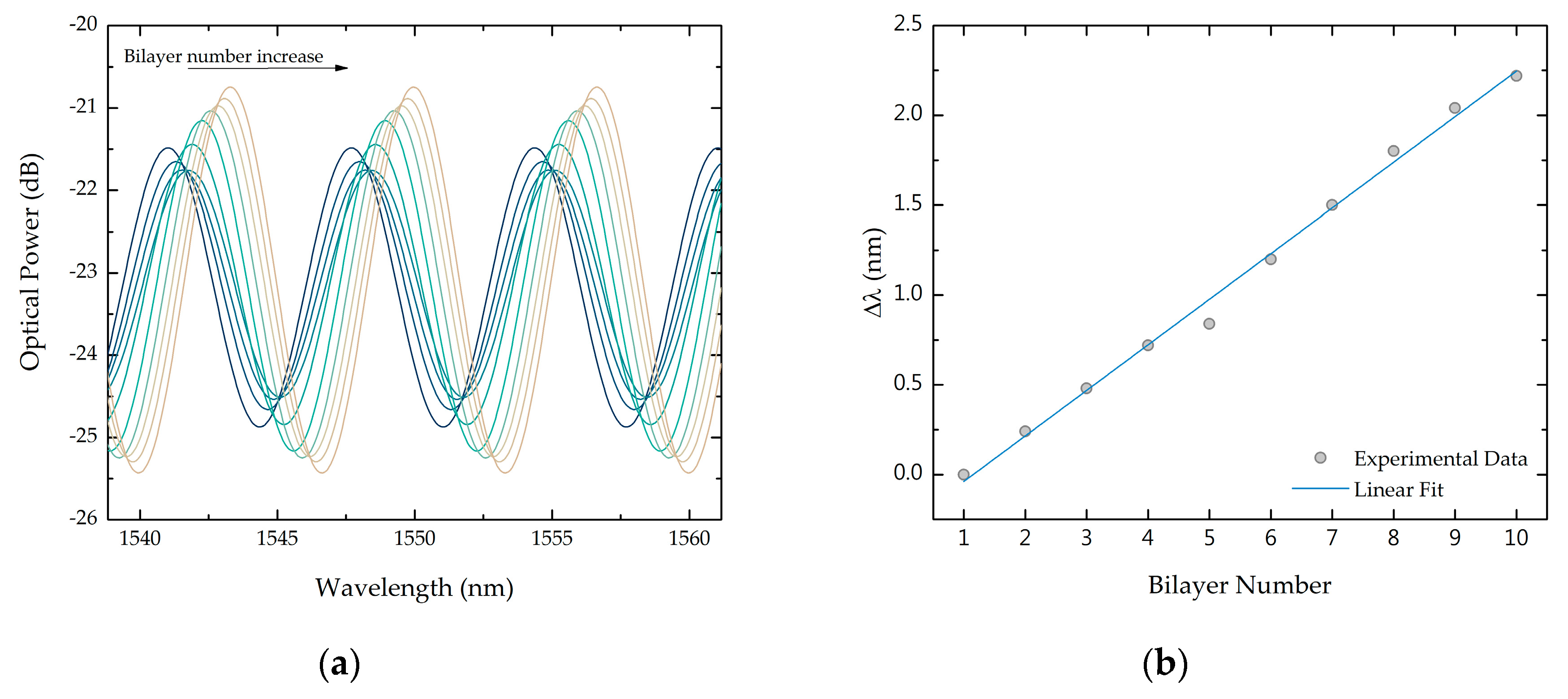
3.1. Bilayer Deposition
3.1.1. Cleaved Fibers
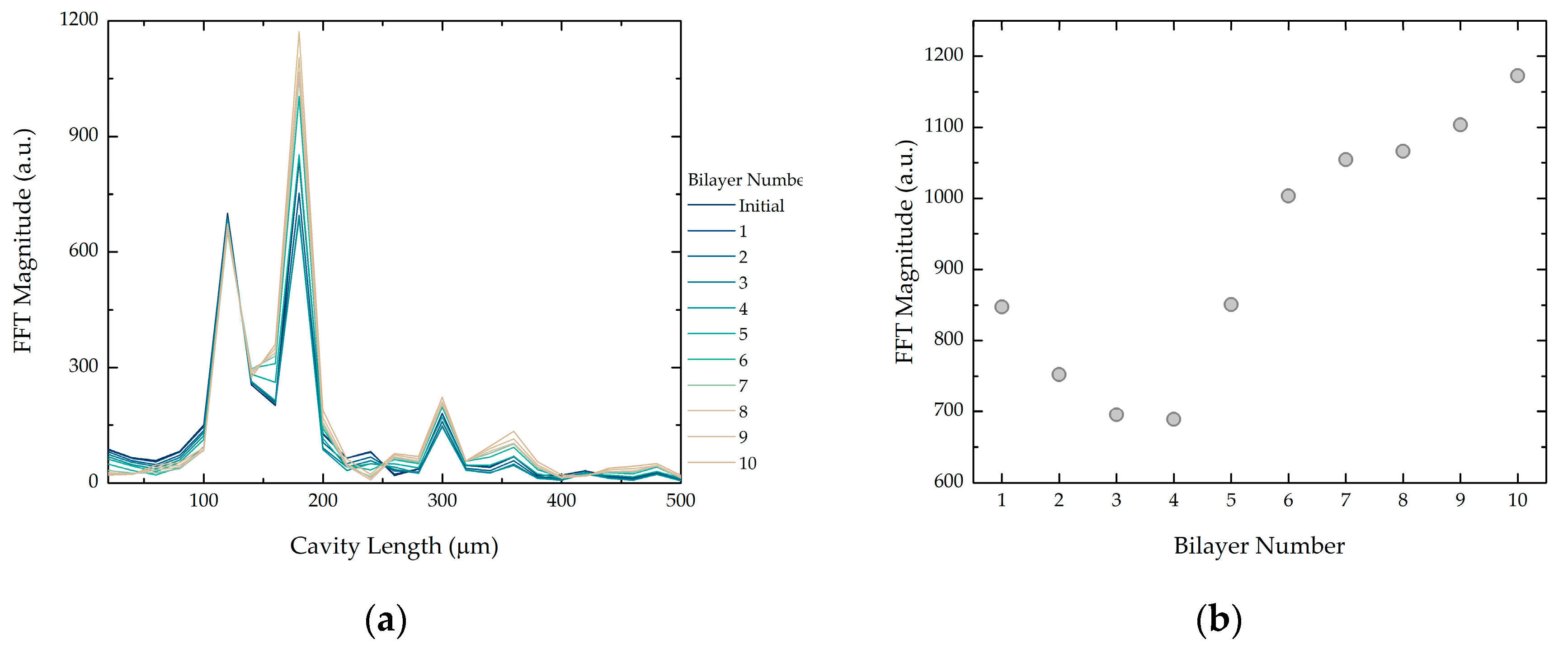
3.1.2. Hollow Microspheres
3.2. Graphene Oxide Adsorption Kinetics
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene and graphene oxide: Synthesis, properties, and applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef]
- Britnell, L.; Gorbachev, R.V.; Jalil, R.; Belle, B.D.; Schedin, F.; Mishchenko, A.; Georgiou, T.; Katsnelson, M.I.; Eaves, L.; Morozov, S.V.; et al. Field-effect tunneling transistor based on vertical graphene heterostructures. Science 2012, 335, 947–950. [Google Scholar] [CrossRef] [PubMed]
- Xuesong, L.; Yanwu, Z.; Weiwei, C.; Mark, B.; Boyang, H.; David, C.; Richard, P. Transfer of Large-Area Graphene Films for High-Performance Transparent Conductive Electrodes. Nano Lett. 2009, 9, 4359–4363. [Google Scholar]
- Wang, X.; Zhi, L.; Müllen, K. Transparent, conductive graphene electrodes for dye-sensitized solar cells. Nano Lett. 2008, 8, 323–327. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Wu, S.; Yin, Z.; Zhang, H. Graphene-based electronic sensors. Chem. Sci. 2012, 3, 1764. [Google Scholar] [CrossRef]
- Gerasimov, G. Graphene-Based Gas Sensors. J. Mater. Chem. A 2013, 1, 10078–10091. [Google Scholar] [CrossRef]
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved Synthesis of Graphene Oxide. ACS Nano 2010, 4, 4806–4814. [Google Scholar] [CrossRef]
- Brodie, B.C. On the Atomic Weight of Graphite. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 1859, 149, 249–259. [Google Scholar]
- Staudenmaier, L. Procedure for the preparation of graphitic acid. Ber Dtsch. Chem. Ges. 1899, 32, 1394–1399. [Google Scholar] [CrossRef]
- Hummers, W.S.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Mkhoyan, K.A.; Contryman, A.W.; Silcox, J.; Stewart, D.A.; Eda, G.; Mattevi, C.; Miller, S.; Chhowalla, M. Atomic and electronic structure of graphene-oxide. Nano Lett. 2009, 9, 1058–1063. [Google Scholar] [CrossRef] [PubMed]
- Culshaw, B.; Kersey, A. Fiber-optic sensing: A historical perspective. J. Lightwave Technol. 2008, 26, 1064–1078. [Google Scholar] [CrossRef]
- Wu, Y.; Yao, B.; Zhang, A.; Rao, Y.; Wang, Z.; Cheng, Y.; Gong, Y.; Zhang, W.; Chen, Y.; Chiang, K.S. Graphene-coated microfiber Bragg grating for high-sensitivity gas sensing. Opt. Lett. 2014, 39, 1235. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Wu, Y.; Liu, X.; Fu, F.; Gong, Y.; Rao, Y.-J.; Chen, Y. Miniature fiber-optic NH3 gas sensor based on Pt nanoparticle-incorporated graphene oxide. Sens. Actuators B Chem. 2017, 244, 107–113. [Google Scholar] [CrossRef]
- Zhang, H.; Kulkarni, A.; Kim, H.; Woo, D.; Kim, Y.-J.; Hong, B.H.; Choi, J.-B.; Kim, T. Detection of Acetone Vapor Using Graphene on Polymer Optical Fiber. J. Nanosci. Nanotechnol. 2011, 11, 5939–5943. [Google Scholar] [CrossRef]
- Hernaez, M.; Mayes, A.G.; Melendi-Espina, S. Graphene oxide in lossy mode resonance-based optical fiber sensors for ethanol detection. Sensors 2018, 18, 58. [Google Scholar] [CrossRef]
- Sharma, A.K.; Gupta, J. Graphene based chalcogenide fiber-optic evanescent wave sensor for detection of hemoglobin in human blood. Opt. Fiber Technol. 2018, 41, 125–130. [Google Scholar] [CrossRef]
- Divagar, M.; Gowri, A.; John, S.; Sai, V.V.R. Graphene oxide coated U-bent plastic optical fiber based chemical sensor for organic solvents. Sens. Actuators B Chem. 2018, 262, 1006–1012. [Google Scholar] [CrossRef]
- Wang, Y.; Shen, C.; Lou, W.; Shentu, F.; Zhong, C.; Dong, X.; Tong, L. Fiber optic relative humidity sensor based on the tilted fiber Bragg grating coated with graphene oxide. Appl. Phys. Lett. 2016, 109, 1–6. [Google Scholar] [CrossRef]
- Ma, J.; Jin, W.; Ho, H.L.; Dai, J.Y. High-sensitivity fiber-tip pressure sensor with graphene diaphragm. Opt. Lett. 2012, 37, 2493. [Google Scholar] [CrossRef]
- Li, C.; Liu, Q.; Peng, X.; Fan, S. Analyzing the temperature sensitivity of Fabry-Perot sensor using multilayer graphene diaphragm. Opt. Express 2015, 23, 27494. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Xuan, H.; Ho, H.L.; Jin, W.; Yang, Y.; Fan, S. Fiber-optic fabry-pérot acoustic sensor with multilayer graphene diaphragm. IEEE Photonics Technol. Lett. 2013, 25, 932–935. [Google Scholar] [CrossRef]
- Xu, M.G.; Dakin, J.P. Novel hollow-glass microsphere sensor for monitoring high hydrostatic pressure. In Fiber Optic and Laser Sensors X; SPIE: Boston, MA, USA, 1993; Volume 1795, pp. 2–7. [Google Scholar]
- Martins, J.; Diaz, C.A.R.; Domingues, M.F.; Ferreira, R.A.S.; Antunes, P.; Andre, P.S. Low-Cost and High-Performance Optical Fiber-Based Sensor for Liquid Level Monitoring. IEEE Sens. J. 2019, 19, 4882–4888. [Google Scholar] [CrossRef]
- Novais, S.; Ferreira, M.S.; Pinto, J.L. Lateral Load Sensing with an Optical Fiber Inline Microcavity. IEEE Photonics Technol. Lett. 2017, 29, 1502–1505. [Google Scholar] [CrossRef]
- Liu, S.; Yang, K.; Wang, Y.; Qu, J.; Liao, C.; He, J.; Li, Z.; Yin, G.; Sun, B.; Zhou, J.; et al. High-sensitivity strain sensor based on in-fiber rectangular air bubble. Sci. Rep. 2015, 5, 7624. [Google Scholar] [CrossRef]
- Monteiro, C.; Silva, S.; Frazao, O. Hollow Microsphere Fabry–Perot Cavity for Sensing Applications. IEEE Photonics Technol. Lett. 2017, 29, 1229–1232. [Google Scholar] [CrossRef]
- Oliveira, O.N., Jr.; Raposo, M.; Dhanabalan, A. Langmuir-Blodgett (LB) and Self-assembled (SA) polymeric films. In Handbook of Surfaces and Interfaces of Materials; Nalwa, S., Ed.; Academic Press: New York, NY, USA, 2001; pp. 1–63. [Google Scholar]
- Keeney, M.; Jiang, X.Y.; Yamane, M.; Lee, M.; Goodman, S.; Yang, F. Nanocoating for biomolecule delivery using layer-by-layer self-assembly. J. Mater. Chem. B 2015, 3, 8757–8770. [Google Scholar] [CrossRef]
- Monteiro, C.S.; Kobelke, J.; Schuster, K.; Bierlich, J.; Silva, S.O.; Frazão, O. High sensitivity strain sensor based on twin hollow microspheres. Microw. Opt. Technol. Lett. 2019, 61, 454–458. [Google Scholar] [CrossRef]
- Liao, C.; Liu, S.; Xu, L.; Wang, C.; Wang, Y.; Li, Z.; Wang, Q.; Wang, D.N. Sub-micron silica diaphragm-based fiber-tip Fabry–Perot interferometer for pressure measurement. Opt. Lett. 2014, 39, 2827. [Google Scholar] [CrossRef]
- Warren-Smith, S.C.; André, R.M.; Perrella, C.; Dellith, J.; Bartelt, H. Direct core structuring of microstructured optical fibers using focused ion beam milling. Opt. Express 2016, 24, 378. [Google Scholar] [CrossRef] [PubMed]
- Raposo, M.; Pontes, R.S.; Mattoso, L.H.C.; Oliveira, O.N. Kinetics of adsorption of poly(o-methoxyaniline) self-assembled films. Macromolecules 1997, 30, 6095–6101. [Google Scholar] [CrossRef]
- Duarte, A.A.; Filipe, S.L.; Abegão, L.M.G.; Gomes, P.J.; Ribeiro, P.A.; Raposo, M. Adsorption kinetics of DPPG liposome layers: A quantitative analysis of surface roughness. Microsc. Microanal. 2013, 19, 867–875. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, Q.; António Ribeiro, P.; Raposo, M. Villain’s fractal growth of poly[1-[4-(3-carboxy-4-hydroxyphenylazo) benzenesulfonamido]-1,2-ethanediyl, sodium salt] J-aggregates onto layer-by-layer films and its effect on film absorbance spectrum. J. Appl. Phys. 2013, 113. [Google Scholar] [CrossRef]

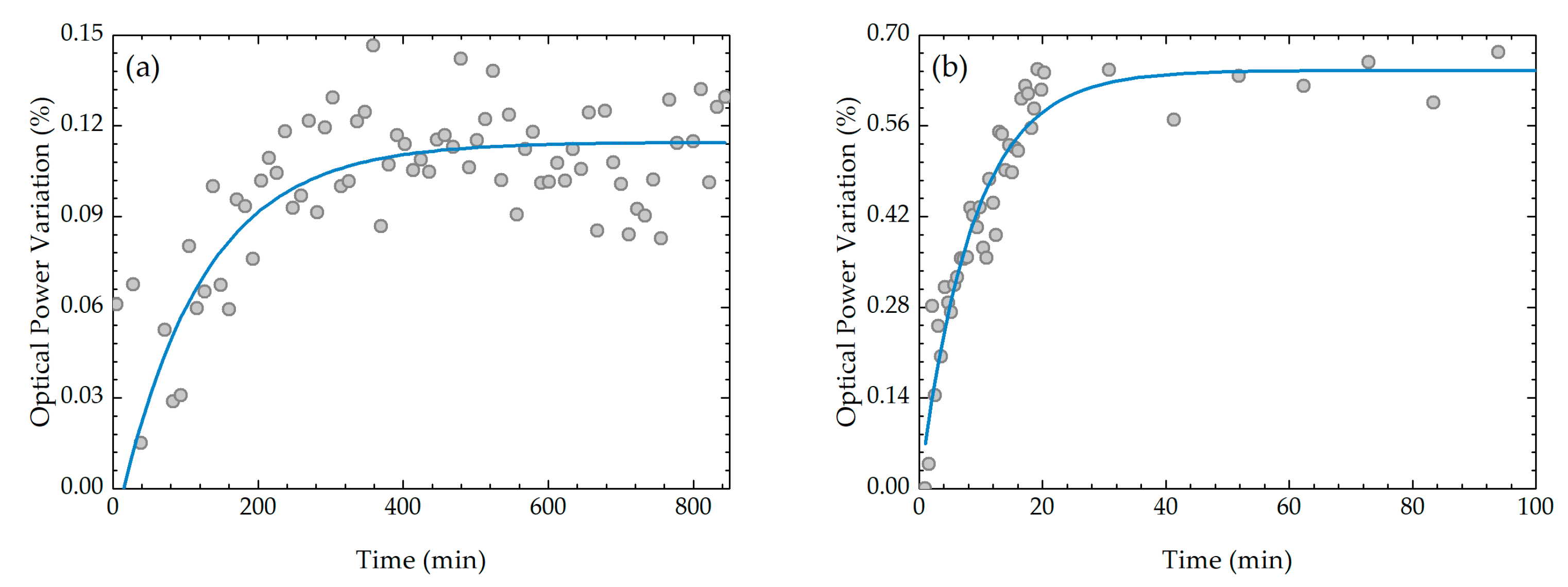
| First GO Layer | Tenth GO Layer | |
|---|---|---|
| (%) | 0.0013 ± 0.0005 | 0.0055 ± 0.0005 |
| (min) | 120 ± 20 | 10 ± 3 |
| R | 0.87 | 0.93 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monteiro, C.S.; Raposo, M.; Ribeiro, P.A.; Silva, S.O.; Frazão, O. Tuning of Fiber Optic Surface Reflectivity through Graphene Oxide-Based Layer-by-Layer Film Coatings. Photonics 2020, 7, 11. https://doi.org/10.3390/photonics7010011
Monteiro CS, Raposo M, Ribeiro PA, Silva SO, Frazão O. Tuning of Fiber Optic Surface Reflectivity through Graphene Oxide-Based Layer-by-Layer Film Coatings. Photonics. 2020; 7(1):11. https://doi.org/10.3390/photonics7010011
Chicago/Turabian StyleMonteiro, Catarina S., Maria Raposo, Paulo A. Ribeiro, Susana O. Silva, and Orlando Frazão. 2020. "Tuning of Fiber Optic Surface Reflectivity through Graphene Oxide-Based Layer-by-Layer Film Coatings" Photonics 7, no. 1: 11. https://doi.org/10.3390/photonics7010011
APA StyleMonteiro, C. S., Raposo, M., Ribeiro, P. A., Silva, S. O., & Frazão, O. (2020). Tuning of Fiber Optic Surface Reflectivity through Graphene Oxide-Based Layer-by-Layer Film Coatings. Photonics, 7(1), 11. https://doi.org/10.3390/photonics7010011








