Blue Light and Eye Damage: A Review on the Impact of Digital Device Emissions
Abstract
1. Introduction
1.1. What Is the Blue Light Emission from Digital Devices Impact?
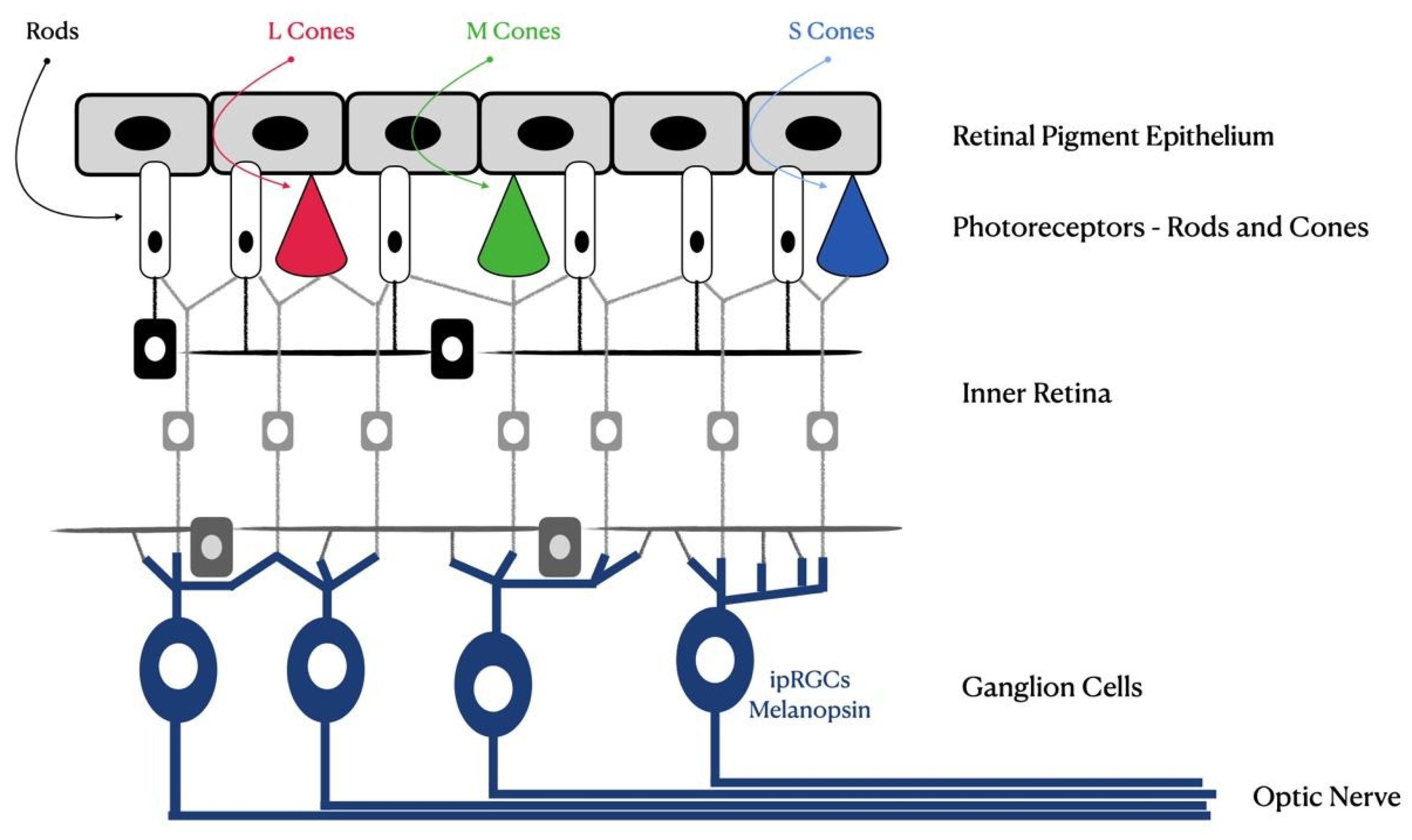
1.1.1. Retinal Impact
1.1.2. Impact on Crystalline Lens
1.1.3. Impact on the Cornea and Ocular Surface
2. Materials and Methods
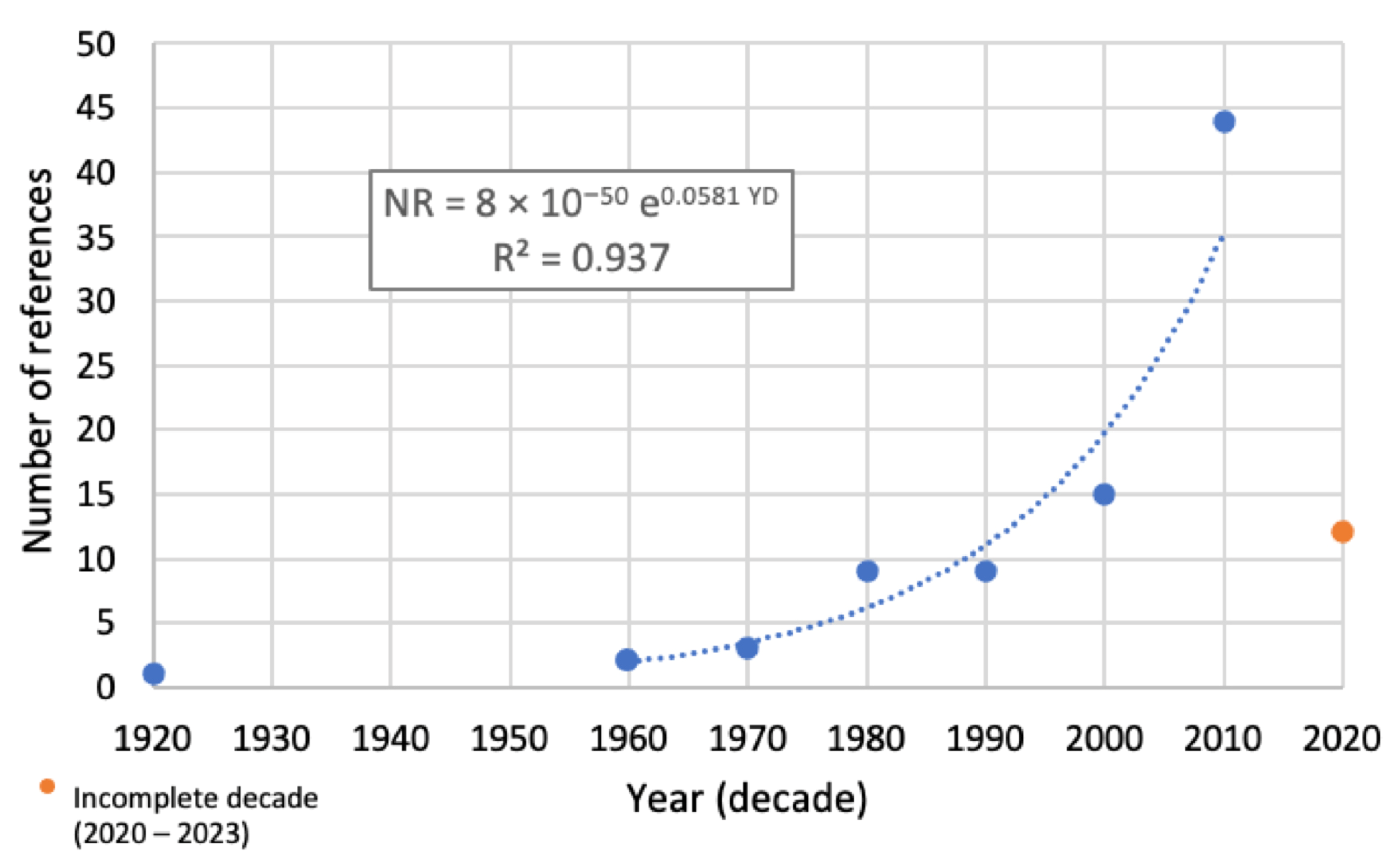
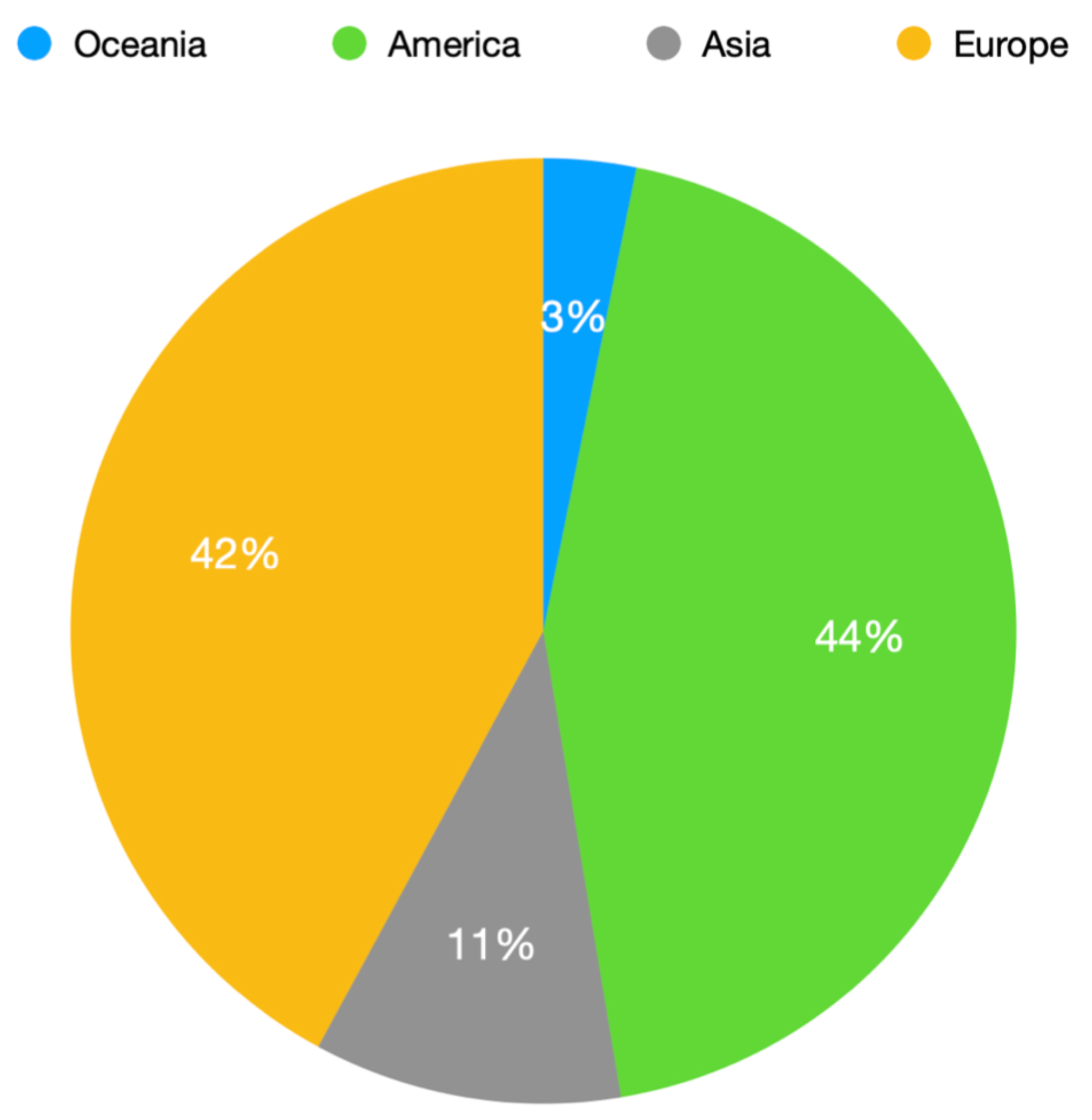
3. Results
4. Discussion
4.1. Retinal Damage from Blue Light
4.2. Blue Light Exposure by Digital Devices
4.3. Relationship between Digital Devices and Ocular Structures
- high daily use of digital devices;
- little or no exposure to digital devices daily.
- We also propose to create the following guidelines:
- duration of daily exposure administration set to at least 1 year if no blue light damage occurs earlier;
- the lowest light intensity set to approximately the average of that emitted by personal digital devices;
- if a relationship is found between blue light exposure and RPE cell layer damage at each intensity tested, this relationship can be extrapolated to the light levels of all digital devices;
- after extrapolation, if the duration of exposure required to damage the RPE cell layers is within the range that a person might experience over a lifetime using these devices, this may indicate that their use may lead to retinal damage;
- otherwise, it suggests that the use of such devices is not harmful to the retina.
5. Conclusions
- investigate and relate the impacts on the eye health of shift workers more exposed to artificial lighting;
- the effects of blue light on circadian rhythms and ocular health at different stages of life;
- interventions in the investigation of light exposure and the inhibition of myopia in children may also be of great interest;
- the impact of the retina overexposure to blue wavelengths from common white light sources;
- how to mitigate health and environmental lighting impacts while maintaining the obvious benefits of using artificial light at night;
- impact of long-term use of digital devices on the cornea, lens and retina;
- a multidisciplinary approach that develops progress in research on safety in the use of artificial blue light and digital devices and their implications for users.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Watson, A. In-Home Media Consumption Due to the Coronavirus Outbreak among Internet Users Worldwide as of March 2020, by Country. Available online: https://www.statista.com/statistics/1106498/home-media-consumption-coronavirus-worldwide-by-country/ (accessed on 31 January 2023).
- Bahkir, F.A.; Grandee, S.S. Impact of the COVID-19 lockdown on digital device-related ocular health. Indian J. Ophthalmol. 2020, 68, 2378–2383. [Google Scholar] [CrossRef] [PubMed]
- Digital 2023: Global Overview Report. Available online: https://datareportal.com/reports/digital-2023-global-overview-report (accessed on 5 April 2023).
- CIE 2019. CIE Position Statement on the Blue Light Hazard. 2019. Available online: http://cie.co.at/publications/position-statement-blue-light-hazard-april-23-2019 (accessed on 5 April 2023).
- Renard, G.; Leid, J. Les dangers de la lumière bleue: La vérité! [The dangers of blue light: True story!]. J. Fr. Ophtalmol. 2016, 39, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Nasrallah, M. Why is the sky blue? Sci. Am. 2003, 289, 103. [Google Scholar]
- Sanchez-Ramos, C.; Bonnin-Arias, C.; Blázquez-Sánchez, V.; Aguirre-Vilacoro, V.; Cobo, T.; García-Suarez, O.; Perez-Carrasco, M.J.; Alvarez-Peregrina, C.; Vega, J.A. Retinal Protection from LED-Backlit Screen Lights by Short Wavelength Absorption Filters. Cells 2021, 10, 3248. [Google Scholar] [CrossRef]
- Jaadane, I.; Villalpando Rodriguez, G.E.; Boulenguez, P.; Chahory, S.; Carré, S.; Savoldelli, M.; Jonet, L.; Behar-Cohen, F.; Martinsons, C.; Torriglia, A. Effects of white light-emitting diode (LED) exposure on retinal pigment epithelium in vivo. J. Cell Mol. Med. 2017, 21, 3453–3466. [Google Scholar] [CrossRef] [PubMed]
- Jameson, D.; Hurvich, L.M. Handbook of Sensory Physiology: Visual Psychophysics; Springer: Berlin/Heidelberg, Germany, 1973; Volume 7/4. [Google Scholar]
- Widomska, J.; Witold, K.S. Why has Nature Chosen Lutein and Zeaxanthin to Protect the Retina? J. Clin. Exp. Ophthalmol. 2014, 5, 326. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Ren, Y.; Reinach, P.S.; Xiao, B.; Lu, H.; Zhu, Y.; Qu, J.; Chen, W. Reactive oxygen species activated NLRP3 inflammasomes initiate inflammation in hyperosmolarity stressed human corneal epithelial cells and environment-induced dry eye patients. Exp. Eye Res. 2015, 134, 133–140. [Google Scholar] [CrossRef]
- Boettner, E.A.; Wolter, J.R. Transmission of the Ocular Media. Investig. Ophthalmol. Vis. Sci. 1962, 1, 776–783. [Google Scholar]
- Norren, D.V.; Vos, J.J. Spectral transmission of the human ocular media. Vis. Res. 1974, 14, 1237–1244. [Google Scholar] [CrossRef]
- Mellerio, J. Yellowing of the human lens: Nuclear and cortical contributions. Vis. Res. 1987, 27, 1581–1587. [Google Scholar] [CrossRef]
- Babizhayev, M.A. Mitochondria induce oxidative stress, generation of reactive oxygen species and redox state unbalance of the eye lens leading to human cataract formation: Disruption of redox lens organization by phospholipid hydroperoxides as a common basis for cataract disease. Cell Biochem. Funct. 2011, 29, 183–206. [Google Scholar] [CrossRef] [PubMed]
- Norton, T.T.; Siegwart, J.T., Jr. Light levels, refractive development, and myopia–a speculative review. Exp. Eye Res. 2013, 114, 48–57. [Google Scholar] [CrossRef]
- Ramamurthy, D.; Chua, S.Y.L.; Saw, S. A review of environmental risk factors for myopia during early life, childhood and adolescence. Clin. Exp. Optom. 2015, 98, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Rucker, F.J.; Britton, S.; Spatcher, M.; Hanowsky, S. Blue Light Protects Against Temporal Frequency Sensitive Refractive Changes. Investig. Ophthalmol. Vis. Sci. 2015, 56, 6121–6131. [Google Scholar] [CrossRef]
- Fulk, G.W.; Cyert, L.A.; Parker, D.A. Seasonal Variation in Myopia Progression and Ocular Elongation. Optom. Vis. Sci. 2002, 79, 46–51. [Google Scholar] [CrossRef]
- Ashby, R.; Ohlendorf, A.; Schaeffel, F. The Effect of Ambient Illuminance on the Development of Deprivation Myopia in Chicks. Investig. Ophthalmol. Vis. Sci. 2009, 50, 5348–5354. [Google Scholar] [CrossRef] [PubMed]
- Stone, R.A.; Cohen, Y.; McGlinn, A.M.; Davison, S.; Casavant, S.; Shaffer, J.; Khurana, T.S.; Pardue, M.T.; Iuvone, P.M. Development of Experimental Myopia in Chicks in a Natural Environment. Investig. Ophthalmol. Vis. Sci. 2016, 57, 4779–4789. [Google Scholar] [CrossRef]
- McClean, A.; Derbyshire, L. Understanding Blue Light. Available online: https://www.aop.org.uk/our-voice/policy/position-statements/2023/01/03/visible-blue-light (accessed on 25 March 2023).
- Arnault, E.; Barrau, C.; Nanteau, C.; Gondouin, P.; Bigot, K.; Viénot, F.; Gutman, E.; Fontaine, V.; Villette, T.; Cohen-Tannoudji, D.; et al. Phototoxic Action Spectrum on a Retinal Pigment Epithelium Model of Age-Related Macular Degeneration Exposed to Sunlight Normalized Conditions. PLoS ONE 2013, 8, e71398. [Google Scholar] [CrossRef]
- Wahl, S.; Engelhardt, M.; Schaupp, P.; Lappe, C.; Ivanov, I.V. The inner clock—Blue light sets the human rhythm. J. Biophotonics 2019, 12, e201900102. [Google Scholar] [CrossRef]
- Verhoeven, G.J. The reflection of two fields—Electromagnetic radiation and its role in (aerial). AARGnews 2017, 55, 13–18. [Google Scholar] [CrossRef]
- Youssef, P.N.; Sheibani, N.; Albert, D.M. Retinal light toxicity. Eye 2011, 25, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Behar-Cohen, F.; Martinsons, C.; Viénot, F.; Zissis, G.; Barlier-Salsi, A.; Cesarini, J.; Enouf, O.; Garcia, M.; Picaud, S.; Attia, D. Light-emitting diodes (LED) for domestic lighting: Any risks for the eye? Prog. Retin. Eye Res. 2011, 30, 239–257. [Google Scholar] [CrossRef] [PubMed]
- Valiente-Soriano, F.J.; Ortín-Martínez, A.; Di Pierdomenico, J.; García-Ayuso, D.; Gallego-Ortega, A.; de Imperial-Ollero, J.A.M.; Jiménez-López, M.; Villegas-Pérez, M.P.; Wheeler, L.A.; Vidal-Sanz, M. Topical Brimonidine or Intravitreal BDNF, CNTF, or bFGF Protect Cones Against Phototoxicity. Transl. Vis. Sci. Technol. 2019, 8, 36. [Google Scholar] [CrossRef] [PubMed]
- Jaadane, I.; Boulenguez, P.; Chahory, S.; Carré, S.; Savoldelli, M.; Jonet, L.; Behar-Cohen, F.; Martinsons, C.; Torriglia, A. Retinal damage induced by commercial light emitting diodes (LEDs). Free Radic. Biol. Med. 2015, 84, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-H.; Wu, M.-R.; Huang, W.-J.; Chow, D.S.-L.; Hsiao, G.; Cheng, Y.-W. Low-Luminance Blue Light-Enhanced Phototoxicity in A2E-Laden RPE Cell Cultures and Rats. Int. J. Mol. Sci. 2019, 20, 1799. [Google Scholar] [CrossRef] [PubMed]
- Krigel, A.; Berdugo, M.; Picard, E.; Levy-Boukris, R.; Jaadane, I.; Jonet, L.; Dernigoghossian, M.; Andrieu-Soler, C.; Torriglia, A.; Behar-Cohen, F. Light-induced retinal damage using different light sources, protocols and rat strains reveals LED phototoxicity. Neuroscience 2016, 339, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Hu, Q.; Li, L.; Tang, X.; Zou, J.; Huang, L.; Li, X. Protective effects of autophagy against blue light-induced retinal degeneration in aged mice. Sci. China Life Sci. 2019, 62, 244–256. [Google Scholar] [CrossRef]
- Nakamura, M.; Yako, T.; Kuse, Y.; Inoue, Y.; Nishinaka, A.; Nakamura, S.; Shimazawa, M.; Hara, H. Exposure to excessive blue LED light damages retinal pigment epithelium and photoreceptors of pigmented mice. Exp. Eye Res. 2018, 177, 1–11. [Google Scholar] [CrossRef]
- Feeney-Burns, L.; Berman, E.R.; Rothman, H. Lipofuscin of Human Retinal Pigment Epithelium. Am. J. Ophthalmol. 1980, 90, 783–791. [Google Scholar] [CrossRef]
- Weiter, J.J.; Delori, F.C.; Wing, G.L.; Fitch, K.A. Retinal pigment epithelial lipofuscin and melanin and choroidal melanin in human eyes. Investig. Ophthalmol. Vis. Sci. 1986, 27, 145–152. [Google Scholar]
- Feeney-Burns, L.; Hilderbrand, E.S.; Eldridge, S. Aging human RPE: Morphometric analysis of macular, equatorial, and peripheral cells. Investig. Ophthalmol. Vis. Sci. 1984, 25, 195–200. [Google Scholar]
- Taylor, H.R.; West, S.; Muñoz, B.; Rosenthal, F.S.; Bressler, S.B.; Bressler, N.M. The Long-term Effects of Visible Light on the Eye. Arch. Ophthalmol. 1992, 110, 99–104. [Google Scholar] [CrossRef] [PubMed]
- The Eye Disease Case-Control Study Group. Risk Factors for Neovascular Age-Related Macular Degeneration. Arch. Ophthalmol. 1992, 110, 1701–1708. [Google Scholar] [CrossRef]
- Hirvelä, H.; Luukinen, H.; Läärä, E.; Laatikainen, L. Risk Factors of Age-related Maculopathy in a Population 70 Years of Age or Older. Ophthalmology 1996, 103, 871–877. [Google Scholar] [CrossRef] [PubMed]
- Darzins, P.; Mitchell, P.; Heller, R. Sun Exposure and Age-related Macular Degeneration. An Australian case-control study. Ophthalmology 1997, 104, 770–776. [Google Scholar] [CrossRef]
- McCarty, C.A.; Mukesh, B.N.; Fu, C.L.; Mitchell, P.; Wang, J.J.; Taylor, H.R. Risk Factors for Age-Related Maculopathy: The Visual Impairment Project. Arch. Ophthalmol. 2001, 119, 1455–1462. [Google Scholar] [CrossRef]
- Delcourt, C.; Carrière, I.; Ponton-Sanchez, A.; Fourrey, S.; Lacroux, A.; Papoz, L.; POLA Study Group. Light Exposure and the Risk of Age-Related Macular Degeneration: The Pathologies Oculaires Liées à l’Age (POLA) study. Arch. Ophthalmol. 2001, 119, 1463–1468. [Google Scholar] [CrossRef]
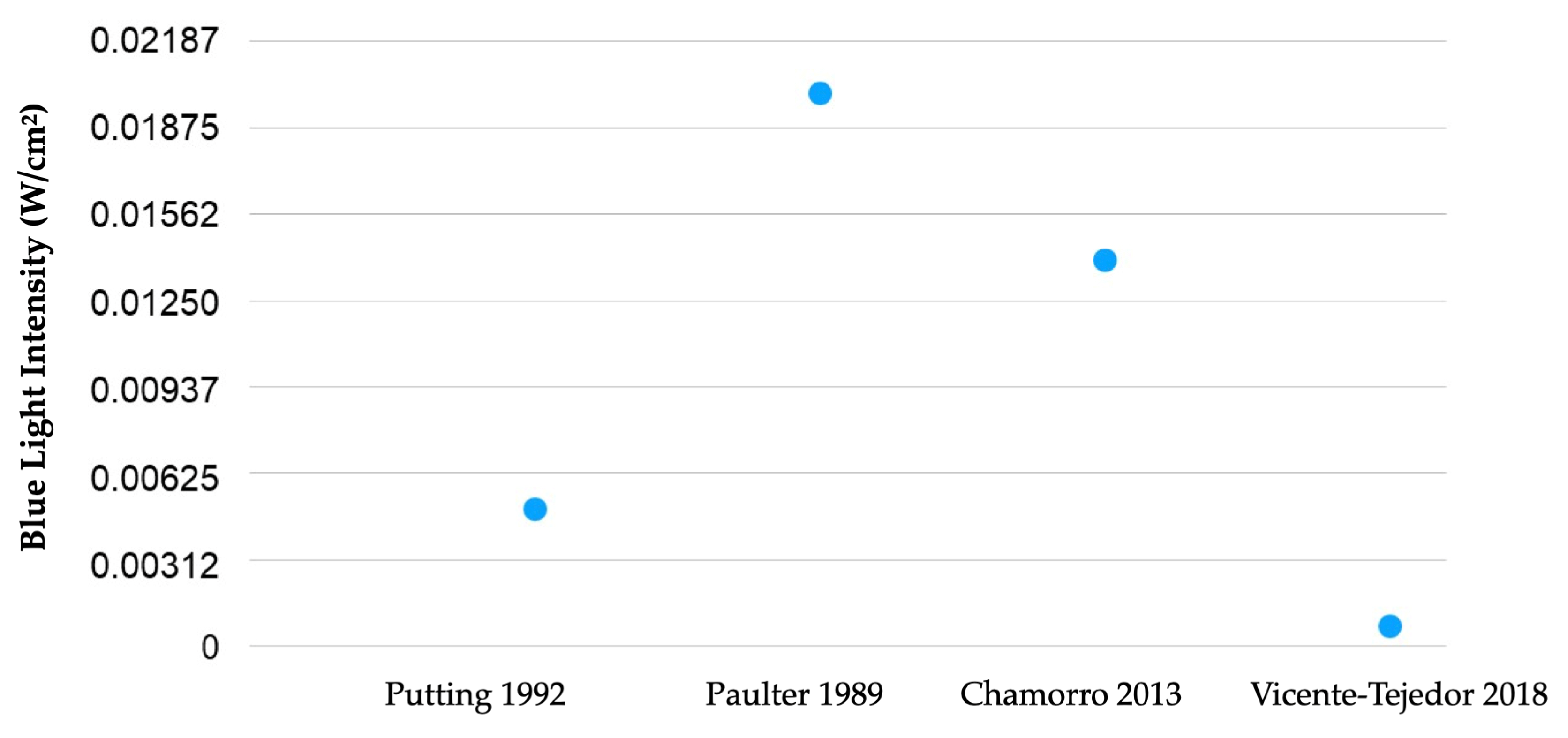
- Putting, B.J.; Zweypfenning, R.C.; Vrensen, G.F.; Oosterhuis, J.A.; Van Best, J.A. Blood-retinal barrier dysfunction at the pigment epithelium induced by blue light. Investig. Ophthalmol. Vis. Sci. 1992, 33, 3385–3393. [Google Scholar]
- Pautler, E.L.; Morita, M.; Beezley, D. Reversible and irreversible blue light damage to the isolated, mammalian pigment epithelium. Prog. Clin. Biol. Res. 1989, 314, 555–567. [Google Scholar]
- Chamorro, E.; Bonnin-Arias, C.; Pérez-Carrasco, M.J.; de Luna, J.M.; Vázquez, D.; Sánchez-Ramos, C. Effects of Light-emitting Diode Radiations on Human Retinal Pigment Epithelial Cells In Vitro. Photochem. Photobiol. 2013, 89, 468–473. [Google Scholar] [CrossRef]
- Vicente-Tejedor, J.; Marchena, M.; Ramírez, L.; García-Ayuso, D.; Gómez-Vicente, V.; Sánchez-Ramos, C.; De La Villa, P.; Germain, F. Removal of the blue component of light significantly decreases retinal damage after high intensity exposure. PLoS ONE 2018, 13, e0194218. [Google Scholar] [CrossRef] [PubMed]
- Egringras, P.; Emiddleton, B.; Skene, D.J.; Revell, V.L. Bigger, Brighter, Bluer-Better? Current Light-Emitting Devices—Adverse Sleep Properties and Preventative Strategies. Front. Public Health 2015, 3, 233. [Google Scholar] [CrossRef]
- Pazikadin, A.R.; Rifai, D.; Ali, K.; Mamat, N.H.; Khamsah, N. Design and Implementation of Fuzzy Compensation Scheme for Temperature and Solar Irradiance Wireless Sensor Network (WSN) on Solar Photovoltaic (PV) System. Sensors 2020, 20, 6744. [Google Scholar] [CrossRef]
- Udovicic, L.; Janßen, M. Photobiological Safety of Common Office Light Sources. In Proceedings of the 29th CIE Session, Washington, DC, USA, 14–22 June 2019; CIE: Vienna, Austria, 2019; pp. 1256–1261. [Google Scholar] [CrossRef]
- Tomany, S.C.; Cruickshanks, K.J.; Klein, R.; Klein, B.E.K.; Knudtson, M.D. Sunlight and the 10-Year Incidence of Age-Related Maculopathy: The Beaver Dam Eye Study. Arch. Ophthalmol. 2004, 122, 750–757, Erratum in Arch. Ophthalmol. 2005, 123, 362. [Google Scholar] [CrossRef]
- Rozanowska, M.B. Light-Induced Damage to the Retina: Current Understanding of the Mechanisms and Unresolved Questions: A Symposium-in-Print. Photochem. Photobiol. 2012, 88, 1303–1308. [Google Scholar] [CrossRef] [PubMed]
- Sciences Online: American Society for Photobiology. Available online: http://photobiology.info/Rozanowska.html (accessed on 20 March 2022).
- Noell, W.K.; Walker, V.S.; Kang, B.S.; Berman, S. Retinal damage by light in rats. Investig. Ophthalmol. 1966, 5, 450–473. [Google Scholar]
- O’Hagan, J.B.; Khazova, M.; Price, L.L.A. Low-energy light bulbs, computers, tablets and the blue light hazard. Eye 2016, 30, 230–233. [Google Scholar] [CrossRef]
- Tosini, G.; Ferguson, I.; Tsubota, K. Effects of blue light on the circadian system and eye physiology. Mol. Vis. 2016, 22, 61–72. [Google Scholar]
- Nash, T.R.; Chow, E.S.; Law, A.D.; Fu, S.D.; Fuszara, E.; Bilska, A.; Bebas, P.; Kretzschmar, D.; Giebultowicz, J.M. Daily blue-light exposure shortens lifespan and causes brain neurodegeneration in Drosophila. NPJ Aging Mech. Dis. 2019, 5, 8. [Google Scholar] [CrossRef]
- Xie, C.; Li, X.; Tong, J.; Gu, Y.; Shen, Y. Effects of white light-emitting diode (LED) light exposure with different Correlated Color Temperatures (CCTs) on human lens epithelial cells in culture. Photochem. Photobiol. 2014, 90, 853–859. [Google Scholar] [CrossRef]
- Weale, R.A. Age and the transmittance of the human crystalline lens. J. Physiol. 1988, 395, 577–587. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Pokorny, J.; Smith, V.C. Optical density of the human lens. J. Opt. Soc. Am. A 1997, 14, 953–960. [Google Scholar] [CrossRef] [PubMed]
- Resnikoff, S.; Pascolini, D.; Etya’Ale, D.; Kocur, I.; Pararajasegaram, R.; Pokharel, G.P.; Mariotti, S.P. Global data on visual impairment in the year 2002. Bull. World Health Organ. 2004, 82, 844–851. [Google Scholar]
- Mainster, M.A. The Spectra, Classification, and Rationale of Ultraviolet-Protective Intraocular Lenses. Am. J. Ophthalmol. 1986, 102, 727–732. [Google Scholar] [CrossRef]
- Van Der Hoeve, J. Eye Lesions Produced by Light Rich in Ultraviolet Rays. Senile Cataract, Senile Degeneration of Macula. Am. J. Ophthalmol. 1920, 3, 178–194. [Google Scholar] [CrossRef]
- Tso, M.O.; La Piana, F.G. The human fovea after sungazing. Trans. Sect. Ophthalmol. Am. Acad. Ophthalmol. Otolaryngol. 1975, 79, OP788-95. [Google Scholar] [PubMed]
- Mainster, M.A.; Findl, O.; Dick, H.B.; Desmettre, T.; Ledesma-Gil, G.; Curcio, C.A.; Turner, P.L. The Blue Light Hazard Versus Blue Light Hype. Am. J. Ophthalmol. 2022, 240, 51–57. [Google Scholar] [CrossRef]
- Tso, M.O. Pathogenetic Factors of Aging Macular Degeneration. Ophthalmology 1985, 92, 628–635. [Google Scholar] [CrossRef]
- Marshall, J. Radiation and the ageing eye. Ophthalmic Physiol. Opt. 1985, 5, 241–263. [Google Scholar] [CrossRef]
- Mainster, M.A. Light and macular degeneration: A biophysical and clinical perspective. Eye 1987, 1, 304–310. [Google Scholar] [CrossRef]
- Mainster, M.A. Violet and blue light blocking intraocular lenses: Photoprotection versus photoreception. Br. J. Ophthalmol. 2006, 90, 784–792. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.D. The ageing retina: Physiology or pathology. Eye 1987, 1, 282–295. [Google Scholar] [CrossRef] [PubMed]
- Remé, C.; Reinboth, J.; Clausen, M.; Hafezi, F. Light damage revisited: Converging evidence, diverging views? Graefes Arch. Clin. Exp. Ophthalmol. 1996, 234, 2–11. [Google Scholar] [CrossRef]
- Boulton, M.; Różanowska, M.; Różanowski, B. Retinal photodamage. J. Photochem. Photobiol. B Biol. 2001, 64, 144–161. [Google Scholar] [CrossRef] [PubMed]
- Margrain, T.H.; Boulton, M.; Marshall, J.; Sliney, D.H. Do blue light filters confer protection against age-related macular degeneration? Prog. Retin. Eye Res. 2004, 23, 523–531. [Google Scholar] [CrossRef]
- Bernstein, P.S.; Khachik, F.; Carvalho, L.S.; Muir, G.J.; Zhao, D.-Y.; Katz, N.B. Identification and Quantitation of Carotenoids and their Metabolites in the Tissues of the Human Eye. Exp. Eye Res. 2001, 72, 215–223. [Google Scholar] [CrossRef]
- Gao, S.; Qin, T.; Liu, Z.; Caceres, M.A.; Ronchi, C.F.; Chen, C.-Y.O.; Yeum, K.-J.; Taylor, A.; Blumberg, J.B.; Liu, Y.; et al. Lutein and zeaxanthin supplementation reduces H2O2-induced oxidative damage in human lens epithelial cells. Mol. Vis. 2011, 17, 3180–3190. [Google Scholar]
- Lee, H.S.; Cui, L.; Li, Y.; Choi, J.S.; Choi, J.-H.; Li, Z.; Kim, G.E.; Choi, W.; Yoon, K.C. Correction: Influence of Light Emitting Diode-Derived Blue Light Overexposure on Mouse Ocular Surface. PLoS ONE 2016, 11, e0167671. [Google Scholar] [CrossRef]
- Yam, J.C.S.; Kwok, A.K.H. Ultraviolet light and ocular diseases. Int. Ophthalmol. 2014, 34, 383–400. [Google Scholar] [CrossRef] [PubMed]
- Vera, J.; Redondo, B.; Ortega-Sanchez, A.; Molina-Molina, A.; Molina, R.; Rosenfield, M.; Jiménez, R. Blue-blocking filters do not alleviate signs and symptoms of digital eye strain. Clin. Exp. Optom. 2023, 106, 85–90. [Google Scholar] [CrossRef]
- Cardona, G.; García, C.; Serés, C.; Vilaseca, M.; Gispets, J. Blink Rate, Blink Amplitude, and Tear Film Integrity during Dynamic Visual Display Terminal Tasks. Curr. Eye Res. 2011, 36, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Portello, J.K.; Rosenfield, M.; Bababekova, Y.; Estrada, J.M.; Leon, A. Computer-related visual symptoms in office workers. Ophthalmic Physiol. Opt. 2012, 32, 375–382. [Google Scholar] [CrossRef]
- Akkaya, S.; Atakan, T.; Acikalin, B.; Aksoy, S.; Ozkurt, Y. Effects of long-term computer use on eye dryness. North. Clin. Istanb. 2018, 5, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Rosenfield, M. Computer vision syndrome: A review of ocular causes and potential treatments. Ophthalmic Physiol. Opt. 2011, 31, 502–515. [Google Scholar] [CrossRef] [PubMed]
- Talens-Estarelles, C.; Sanchis-Jurado, V.; Esteve-Taboada, J.J.; Pons, M.; García-Lázaro, S. How Do Different Digital Displays Affect the Ocular Surface? Optom. Vis. Sci. 2020, 97, 1070–1079. [Google Scholar] [CrossRef]
- Busch, E.M.; Gorgels, T.G.; van Norren, D. Temporal sequence of changes in rat retina after UV-A and blue light exposure. Vis. Res. 1999, 39, 1233–1247. [Google Scholar] [CrossRef]
- Dorey, C.K.; Delori, F.C.; Akeo, K. Growth of cultured RPE and endothelial cells is inhibited by blue light but not green or red light. Curr. Eye Res. 1990, 9, 549–559. [Google Scholar] [CrossRef]
- Hunter, J.J.; Morgan, J.I.; Merigan, W.H.; Sliney, D.H.; Sparrow, J.R.; Williams, D.R. The susceptibility of the retina to photochemical damage from visible light. Prog. Retin. Eye Res. 2012, 31, 28–42. [Google Scholar] [CrossRef]
- Schick, T.; Ersoy, L.; Lechanteur, Y.T.E.; Saksens, N.T.M.; Hoyng, C.B.; den Hollander, A.I.; Kirchhof, B.; Fauser, S. History of Sunlight Exposure is a Risk Factor for Age-related Macular Degeneration. Retina 2016, 36, 787–790. [Google Scholar] [CrossRef]
- Fletcher, A.E.; Bentham, G.C.; Agnew, M.; Young, I.S.; Augood, C.; Chakravarthy, U.; de Jong, P.T.; Rahu, M.; Seland, J.; Soubrane, G.; et al. Sunlight Exposure, Antioxidants, and Age-Related Macular Degeneration. Arch. Ophthalmol. 2008, 126, 1396–1403. [Google Scholar] [CrossRef]
- Salvatori, A.; Menon, S.; Zwysen, W. The Effect of Computer Use on Job Quality; OECD Social, Employment and Migration Working Papers Paris; OECD: Paris, France, 2018; No. 200. [Google Scholar] [CrossRef]
- Nagata, J.M.; Magid, H.S.A.; Gabriel, K.P. Screen Time for Children and Adolescents During the Coronavirus Disease 2019 Pandemic. Obesity 2020, 28, 1582–1583. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, P.; Biswas, A.; Sahu, S. COVID-19 pandemic and lockdown: Cause of sleep disruption, depression, somatic pain, and increased screen exposure of office workers and students of India. Chronobiol. Int. 2020, 37, 1191–1200. [Google Scholar] [CrossRef] [PubMed]
- Hefner, B. Warding off the blues. Rev. Optom. 2018, 155, 71–78. [Google Scholar]
- Spaide, R. Etiology of late-age-related macular disease. In Age-Related Macular Degeneration: A Comprehensive Textbook; Alfaro, D.V., Quiroz-Mercado, H., Liggett, P.E., Mieler, W.F., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006; pp. 23–39. [Google Scholar]
- Kozlowski, M.R. The ARPE-19 Cell Line: Mortality Status and Utility in Macular Degeneration Research. Curr. Eye Res. 2015, 40, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Abdouh, M.; Lu, M.; Chen, Y.; Goyeneche, A.; Burnier, J.V.; Burnier, M.N. Filtering blue light mitigates the deleterious effects induced by the oxidative stress in human retinal pigment epithelial cells. Exp. Eye Res. 2022, 217, 108978. [Google Scholar] [CrossRef]
- Gordon, A. Digital eye strain, blue light, and contact lens wear. Contact Lens Spectr. 2018, 1, 27–32. [Google Scholar]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hipólito, V.; Coelho, J.M.P. Blue Light and Eye Damage: A Review on the Impact of Digital Device Emissions. Photonics 2023, 10, 560. https://doi.org/10.3390/photonics10050560
Hipólito V, Coelho JMP. Blue Light and Eye Damage: A Review on the Impact of Digital Device Emissions. Photonics. 2023; 10(5):560. https://doi.org/10.3390/photonics10050560
Chicago/Turabian StyleHipólito, Vladimiro, and João M. P. Coelho. 2023. "Blue Light and Eye Damage: A Review on the Impact of Digital Device Emissions" Photonics 10, no. 5: 560. https://doi.org/10.3390/photonics10050560
APA StyleHipólito, V., & Coelho, J. M. P. (2023). Blue Light and Eye Damage: A Review on the Impact of Digital Device Emissions. Photonics, 10(5), 560. https://doi.org/10.3390/photonics10050560





