Dental Imaging Using Mesoscopic Fluorescence Molecular Tomography: An ex Vivo Feasibility Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
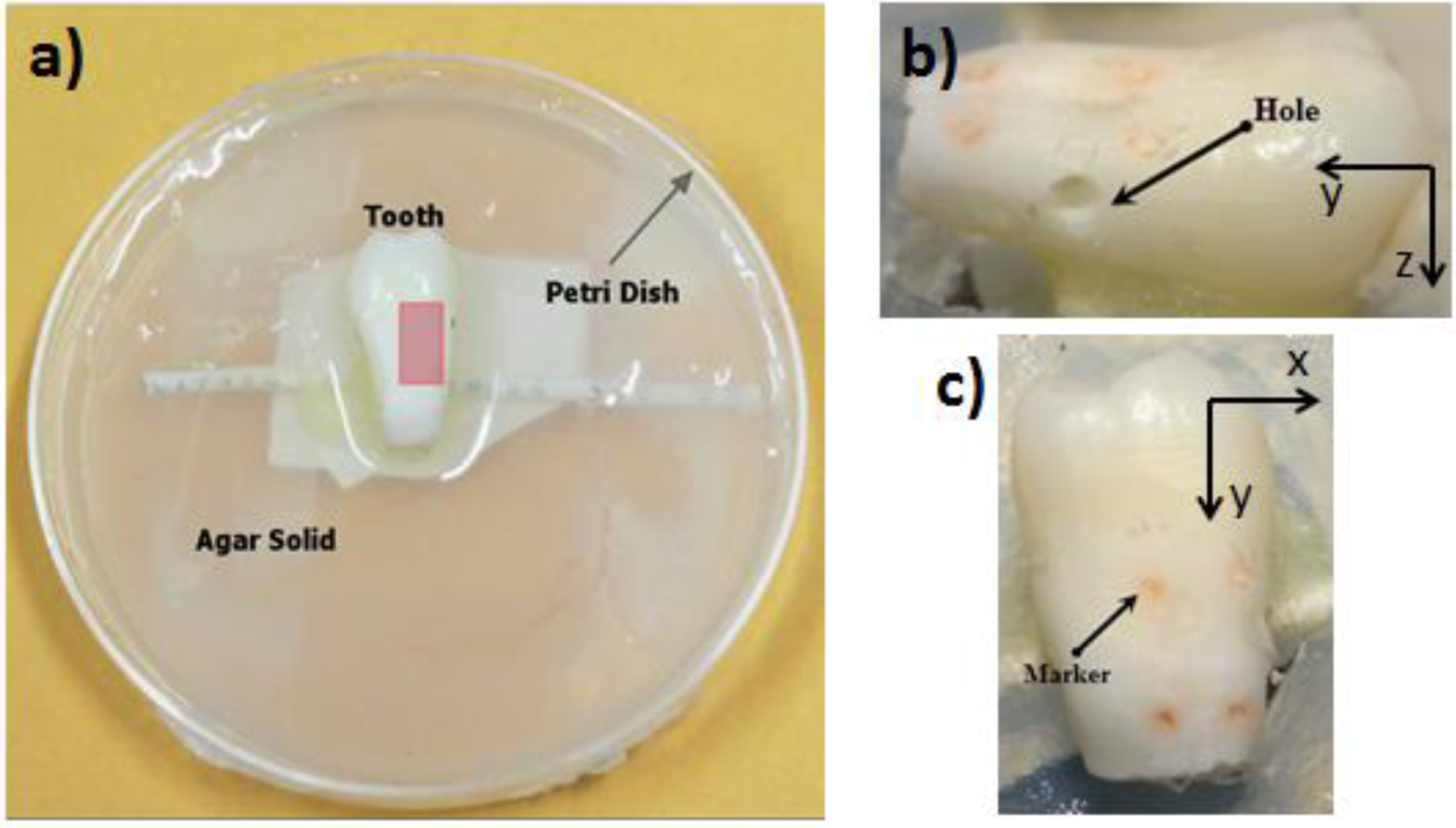
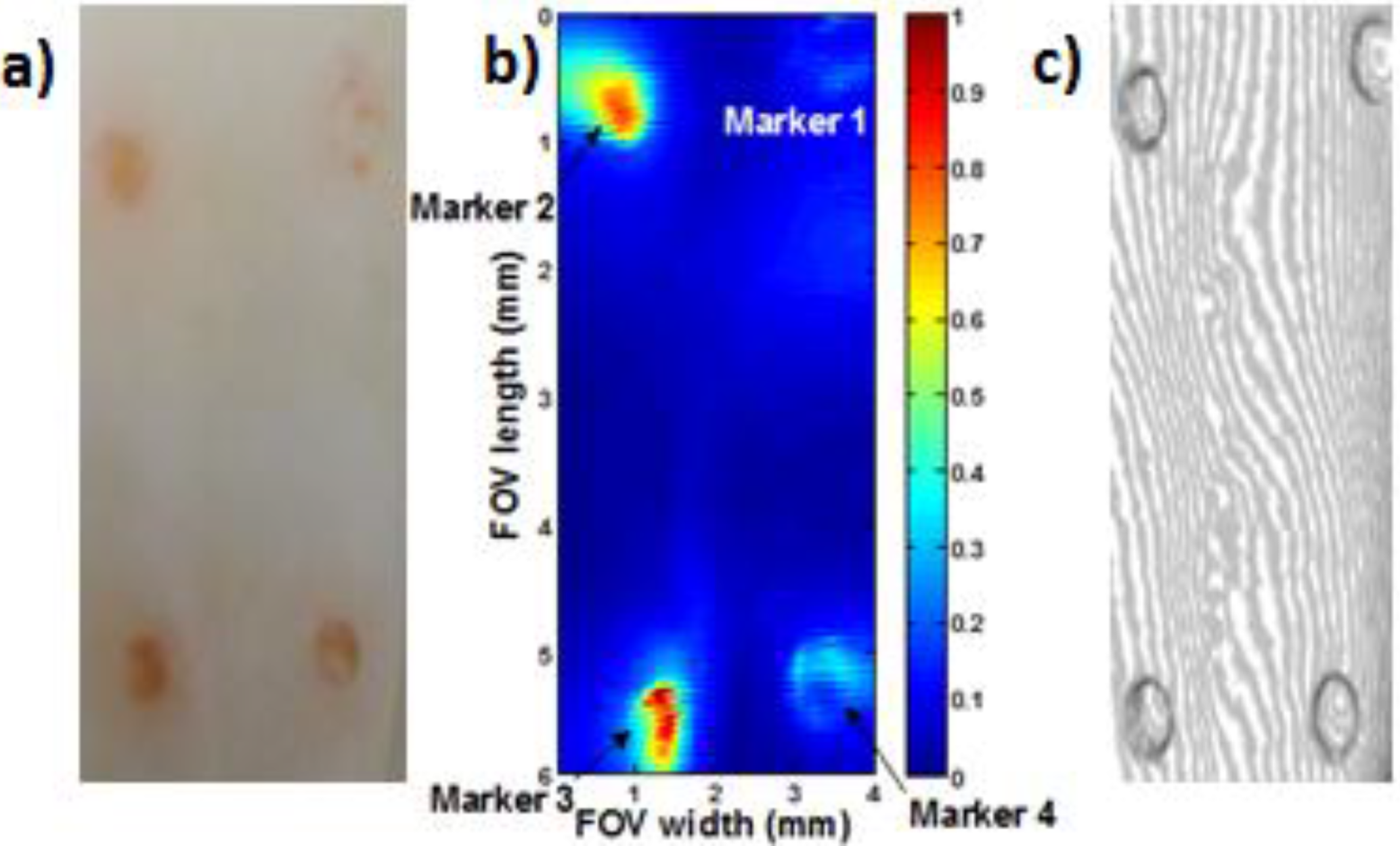
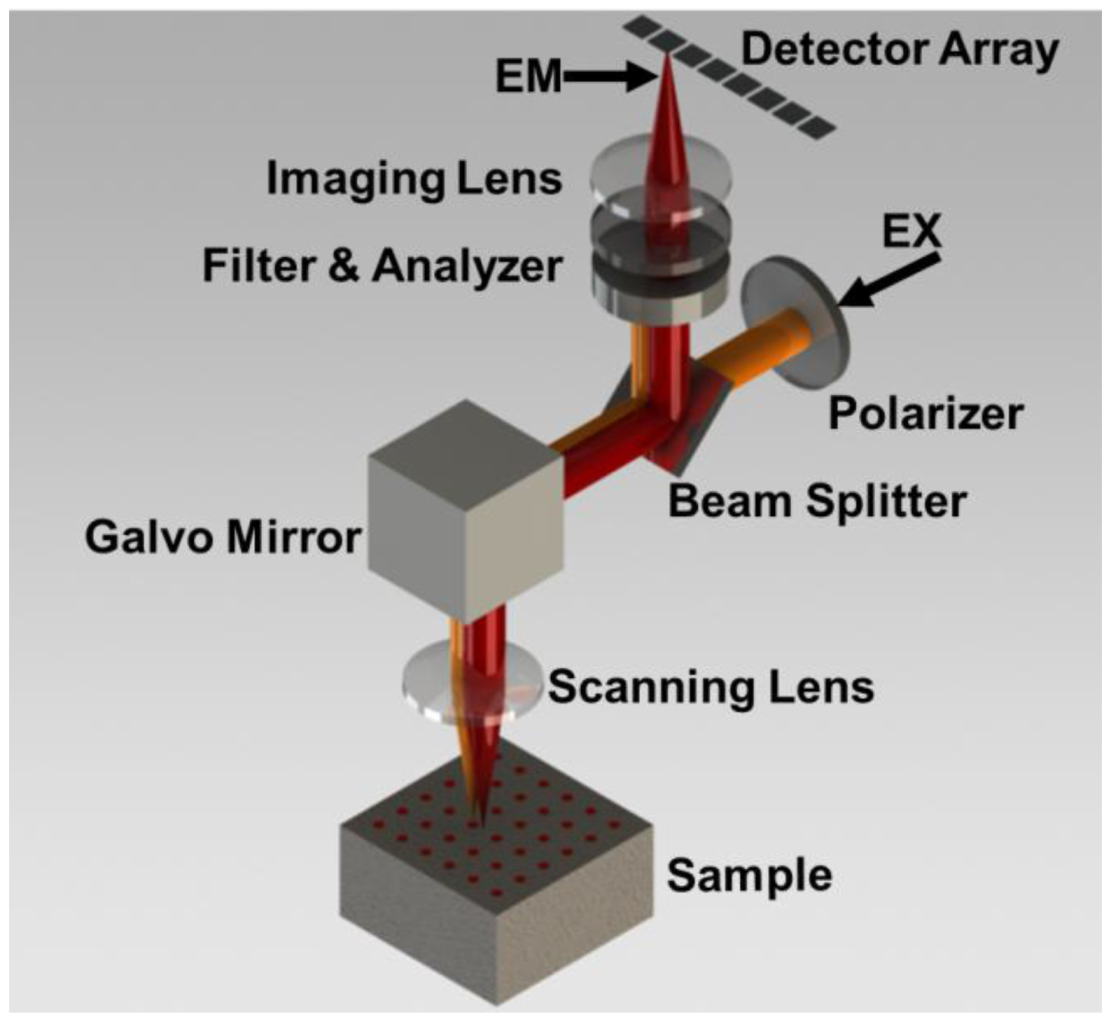
2.2. Optical Imaging System
2.3. Optical Reconstruction
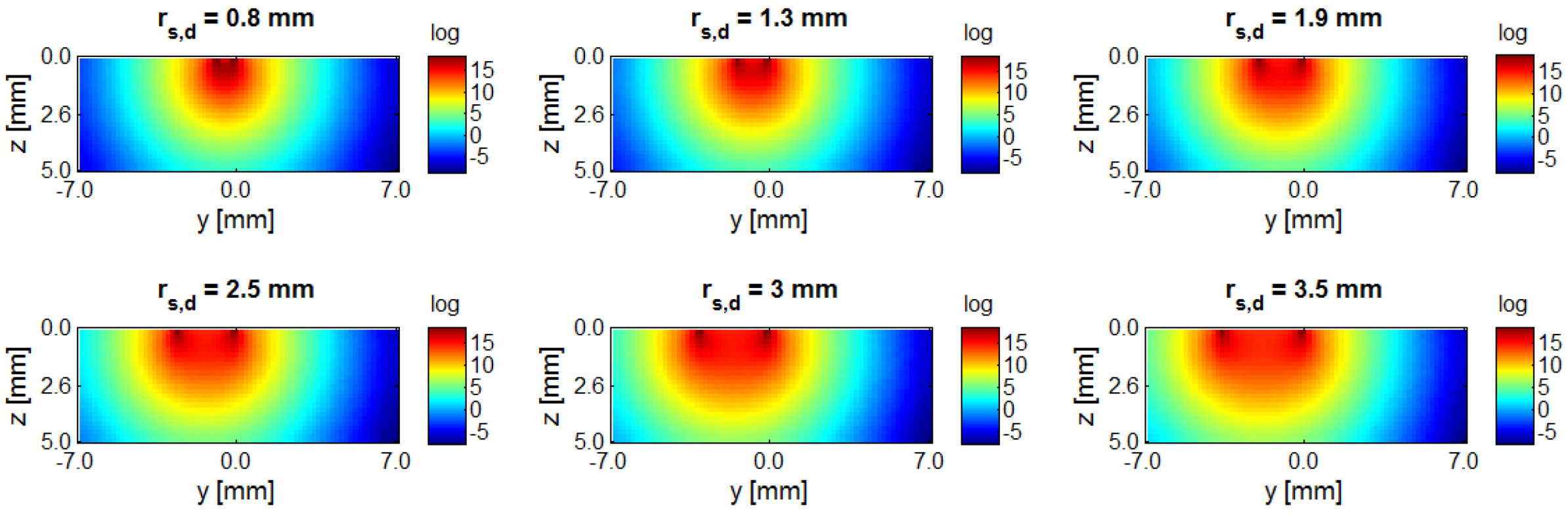
2.4. Image Registration of Multimodal Data Sets

3. Results
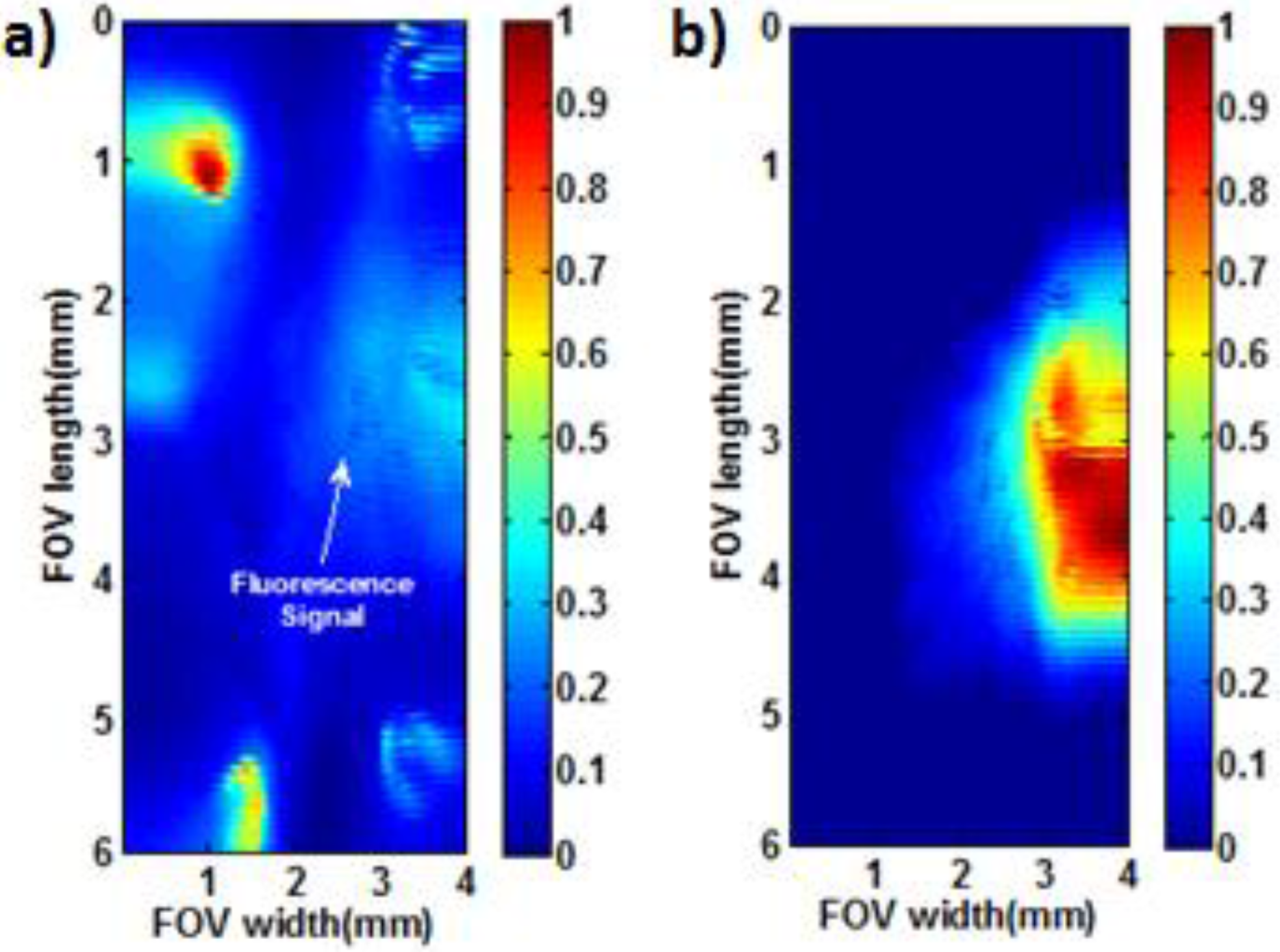
| Conc. (µM) | 1 mm | 2 mm | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | |
| 6.5 | <0 | <0 | <0 | <0 | <0 | <0 | <0 | 0.4 | 1.2 | <0 | <0 | <0 | <0 | <0 |
| 13 | 13.5 | 15.4 | 12.9 | 13.7 | 12.2 | 11.5 | 1.9 | 8.0 | 9.3 | 7.8 | 7.5 | 7.1 | 4.3 | <0 |
| 26 | 21.5 | 23.2 | 23.5 | 23.5 | 20.7 | 12.8 | 7.7 | 9.9 | 11.3 | 12.8 | 13.1 | 11.4 | 11.9 | 7.8 |
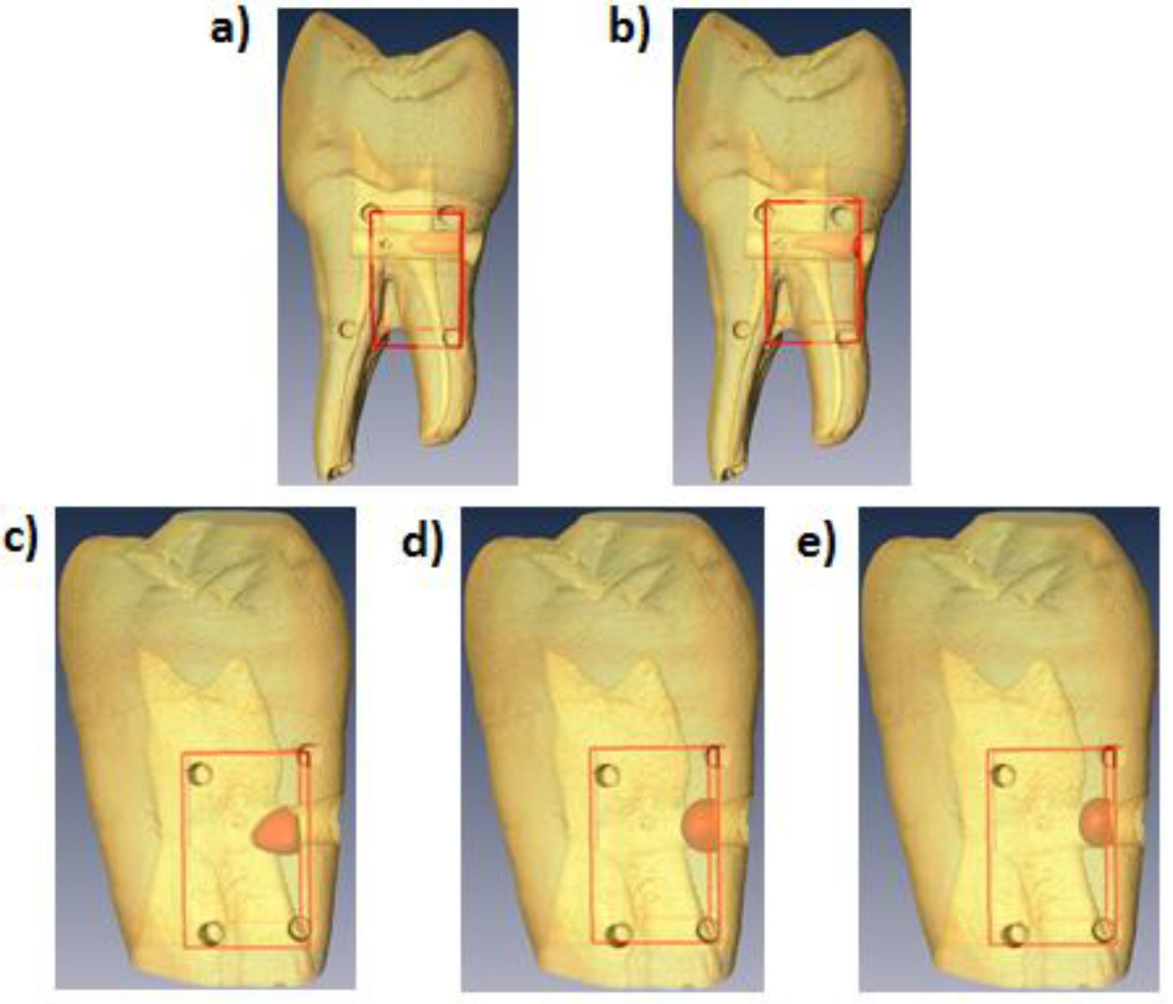
| Dye Concentration (µM) | MFMT Vol. (mm3) | Micro-CT Vol. (mm3) | Vol. Error | MFMT Centroid (mm) | Micro-CT Centroid (mm) | Centroid Error | |
|---|---|---|---|---|---|---|---|
| 1 mm | 13 | 2.96 | 2.67 | 11% | (2.1, 2.3, –1.0) | (1.7, 2.0, –1.0) | 24% |
| 26 | 3.18 | 2.86 | 11% | (1.6, 2.2, –1.1) | (1.8, 2.0, –1.0) | 11% | |
| 2 mm | 6.5 | 2.34 | 3.03 | 23% | (1.9, 2.5,–1.8) | (1.9, 2.4, –2.0) | 10% |
| 13 | 2.04 | 2.67 | 24% | (1.9, 2.5,–1.8) | (1.7, 2.4, –2.0) | 12% | |
| 26 | 2.90 | 2.86 | 1% | (2.1, 2.1,–1.8) | (1.8, 2.4, –2.0) | 17% | |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflict of Interest
References
- Pereira, A.C.; Verdonschot, E.H.; Huysmans, M.C. Caries detection methods: Can they aid decision making for invasive sealant treatment? Caries Res. 2001, 35, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Dental Caries (Tooth Decay) in Adults (Age 20 to 64). Available online: http://www.nidcr.nih.gov/DataStatistics/FindDataByTopic/DentalCaries/DentalCariesAdults20to64.htm (accessed on 1 May 2014).
- Haak, R.; Wicht, M.J.; Noack, M.J. Conventional digital and contrast-enhanced bitewing radiograph in the decision to restore proximal carious lesions. Caries Res. 2001, 35, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Møystad, A.; Svanaes, D.B.; Risnes, S.; Larheim, T.A.; Gröndahl, H.G. Detection of approximal caries with a storage phosphor system. A comparison of enhanced digital images with dental X-ray film. Dentomaxillofac Radiol. 1996, 25, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Tam, L.E.; McComb, D. Diagnosis of occlusal caries: Part II. Recent diagnostic technologies. J. Can. Dent. Assoc. 2001, 67, 459–463. [Google Scholar] [PubMed]
- Park, Y.-S.; Ahn, J.-S.; Kwon, H.-B.; Lee, S.-P. Current status of dental caries diagnosis using cone beam computed tomography. Imag. Sci. Dent. 2011, 41, 43–51. [Google Scholar] [CrossRef]
- Hintze, H.; Wenze, A.; Danielsen, B.; Nyvad, B. Reliability of visual examination, Fibre-Optic transillumination, and Bite-wing radiography, and Reproducibility of direct visual examination following tooth separation for the identification of cavitated carious lesions in contacting approximal surfaces. Caries Res. 1998, 32, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, A. Bitewing and digital bitewing radiography for detection of caries lesions. J. Dent. Res. 2004, 83, C72–C75. [Google Scholar] [CrossRef] [PubMed]
- Tyndall, D.A.; Rathore, S. Cone-beam CT diagnostic applications: Caries, periodontal bone assessment, and endodontic applications. Dent. Clin. North Am. 2008, 52, 825–841. [Google Scholar] [CrossRef] [PubMed]
- Kamburoğlu, K.; Kurt, H.; Kolsuz, E.; Öztaş, B.; Tatar, I.; Çelik, H.H. Occlusal caries depth measurements obtained by five different imaging modalities. J. Digit. Imag. 2011, 24, 804–813. [Google Scholar] [CrossRef]
- Pauwels, R.; Beinsberger, J.; Collaert, B.; Theodorakou, C.; Rogers, J.; Walker, A.; Cockmartin, L.; Bosmans, H.; Jacobs, R.; Bogaerts, R.; et al. Effective dose range for dental cone beam computed tomography scanners. Eur. J. Radiol. 2012, 81, 267–271. [Google Scholar] [CrossRef]
- David, C.; Christopher, L.; Vincent, P.W.; Bryan, C.; Don, A. Michael pepper three-dimensional terahertz pulse imaging of dental tissue. J. Biomed. Opt. 2003, 8, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Smirnov, S.V.; Ya, V.G.; Tsypkin, A.N.; Bespalov, V.G. Experimental studies of the possibilities of diagnosing caries in the solid tissues of a tooth by means of terahertz radiation. J. Opt. Technol. 2014, 81, 464–467. [Google Scholar] [CrossRef]
- Karsten, K.; Herbert, S.; Joerg, H.; Bruce, J.T.; Rudolf, W. Steiner In-vivo fluorescence detection and imaging of porphyrin-producing bacteria in the human skin and in the oral cavity for diagnosis of acne vulgaris, caries, and squamous cell carcinoma. Proc. SPIE 1994, 2135, 129–138. [Google Scholar]
- Lussi, A.; Megert, B.; Longbottom, C.; Reich, E.; Francescut, P. Clinical performance of a laser fluorescence device for detection of occlusal caries lesions. Eur. J. Oral Sci. 2001, 109, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.Q.; Welander, U.; Angmar-Månsson, B. Occlusal caries detection with KaVo DIAGNOdent and radiography: An in vitro comparison. Caries Res. 2000, 34, 151–158. [Google Scholar] [CrossRef]
- Spitzer, D.; Ten Bosch, J.J. Luminescence quantum yields of sound and carious dental enamel. Calcif. Tissue Res. 1997, 24, 249–251. [Google Scholar] [CrossRef]
- Ferreira Zandoná, A.; Ando, M.; Eggertssonn, H.; Kelly, S.A.; Katz, B.; Eckert, G.; Ofner, S.; Sa, L.; Lukantsova, L.; Wefel, J.S.; et al. Clinical Validation of Caries Detection Methodologies: Preliminary Results. J. Dent. Res. 2004, 83, C84–C88. [Google Scholar] [CrossRef] [PubMed]
- Sofia, T.; Susan, A.-K.; Stein, B.; Svante, T.; Birgit, A.-M. Application of quantitative light-induced fluorescence to monitor incipient lesions in caries-active children. A comparative study of remineralisation by fluoride varnish and professional cleaning. Eur. J. Oral Sci. 2001, 109, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Stookey, G.K. Quantitative light fluorescence: A technology for early monitoring of the caries process. Dent. Clin. North Am. 2005, 49, 753–770. [Google Scholar] [CrossRef] [PubMed]
- Chesters, R.K.; Pitts, N.B.; Matuliene, G.; Kvedariene, A.; Huntington, E.; Bendinskaite, R.; Balciuniene, I.; Matheson, J.R.; Nicholson, J.A.; Gendvilyte, A.; et al. An abbreviated caries clinical trial design validated over 24 months. J. Dent. Res. 2002, 81, 637–640. [Google Scholar] [CrossRef]
- Curnow, M.M.T.; Pine, C.M.; Burnside, G.; Nicholson, J.A.; Chesters, R.K.; Huntington, E. A randomised controlled trial of the efficacy of supervised toothbrushing in high-caries-risk children. Caries Res. 2002, 36, 294–300. [Google Scholar] [CrossRef]
- Featherstone, J.D.B. The science and practice of caries prevention. J. Am. Dent. Assoc. 2000, 131, 887–899. [Google Scholar] [CrossRef] [PubMed]
- Watson, T.F. Fact and artefact in confocal microscopy. Adv. Dent. Res. 1997, 11, 433–441. [Google Scholar] [CrossRef] [PubMed]
- McConnell, G.; Girkin, J.M.; Ameer-Beg, S.M.; Baber, P.R.; Vojnovic, B.; Ng, T.; Banerjee, A.; Waston, T.F.; Cook, R.J. Time-correlated single-photon counting fluorescence lifetime confocal imaging of decayed and sound dental structures with a white-light supercontinuum source. J. Microsc. 2007, 225, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.Y.; Lyu, H.-C.; Hsu Stephen, C.-Y.; Chang, C.-S.; Kao, F.-J. Imaging carious dental tissues with multiphoton fluorescence lifetime imaging microscopy. Biomed. Opt. Express 2010, 2, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Holtzman, J.S.; Osann, K.; Pharar, J.; Lee, K.; Ahn, Y.C.; Tucker, T.; Sabet, S.; Chen, Z.P.; Gukasyan, R.; Smith, P.W. Ability of optical coherence tomography to detect caries beneath commonly used dental sealant. Lasers Surg. Med. 2010, 42, 752–759. [Google Scholar] [CrossRef] [PubMed]
- Douglas, S.M.; Fried, D.; Darling, C.L. Imaging Natural Occlusal Caries Lesions with Optical Coherence Tomography. Proc. Soc. Photo Opt. Instrum. Eng. 2010, 7549, 75490N. [Google Scholar] [PubMed]
- Dunn, A.; Boas, D. Transport-based image reconstruction in turbid media with small source-detector separations. Opt. Lett. 2000, 25, 1777–1779. [Google Scholar] [CrossRef] [PubMed]
- Hillman, E.M.C.; Boas, D.A.; Dale, A.M.; Dunn, A.K. Laminar optical tomography: demonstration of millimeter-scale depth-resolved imaging in turbid media. Opt. Lett. 2004, 29, 1650–1652. [Google Scholar] [CrossRef] [PubMed]
- Ouakli, N.; Guevara, E.; Dubeau, S.; Beaumont, E.; Lesage, F. Laminar optical tomography of the hemodynamic response in the lumbar spinal cord of rats. Opt. Express 2010, 18, 10068–10077. [Google Scholar] [CrossRef] [PubMed]
- Hillman, E.M.C.; Burgess, S.A. Sub-millimeter resolution 3D optical imaging of living tissue using laminar optical tomography. Laser Photon. Rev. 2009, 3, 159–179. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.L.; Lee, V.K.; Yoo, S.S.; Dai, G.H.; Intes, X. The integration of 3-D cell printing and mesoscopic fluorescence molecular tomography of vascular constructs within thick hydrogel scaffolds. Biomaterials 2012, 33, 5325–5332. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, M.S.; Lee, V.K.; Zhao, L.L.; Dai, G.H.; Intes, X. Mesoscopic fluorescence molecular tomography of reporter genes in bioprinted thick tissue. J. Biomed. Opt. 2013, 18, 100501. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Roney, C.A.; Wierwille, J.; Chen, C.W.; Xu, B.Y.; Griffiths, G.; Jiang, J.; Ma, H.Z.; Cable, A.; Summers, R.M.; Chen, Y. Co-registered optical coherence tomography and fluorescence molecular imaging for simultaneous morphological and molecular imaging. Phys. Med. Biol. 2010, 55, 191–206. [Google Scholar] [CrossRef]
- Ozturk, M.S.; Rohrbach, D.; Sunar, U.; Intes, X. Mesoscopic Fluorescence Tomography of a photosensitizer (HPPH) 3D Bio-Distribution in Skin Cancer. Acad. Radiol. 2014, 21, 271–280. [Google Scholar] [CrossRef]
- Björn, S.; Englmeier, K.H.; Ntziachristos, V.; Schulz, R. Reconstruction of fluorescence distribution hidden in biological tissue using mesoscopic epifluorescence tomography. J. Biomed. Opt. 2011, 16, 046005. [Google Scholar] [CrossRef] [PubMed]
- Fried, D.; Glena, R.E.; Featherstone, J.D.B.; Seka, W. Nature of light scattering in dental enamel and dentin at visible and near-infrared wavelengths. Appl. Opt. 1995, 34, 1278–1285. [Google Scholar] [CrossRef] [PubMed]
- Oancea, R.; Podariu, A.C.; Vasile, L.; Rosianu, R.S.; Folescu, R. In vitro evaluation of laser fluorescence devices for caries detection through stereomicroscopic imaging. Rom. J. Morphol. Embryol. 2013, 54, 333–341. [Google Scholar]
- Schneiderman, A.; Elbaum, M.; Shultz, T.; Keem, S.; Greenebaum, M.; Driller, J. Assessment of dental caries with digital imaging fiber-optic Transillumination (DIFOTITM): In vitro study. Caries Res. 1997, 31, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Darling, C.L.; Fried, D. In vitro near-infrared imaging of occlusal dental caries using germanium enhanced CMOS camera. Proc. Soc. Photo. Opt. Instrum. Eng. 2010, 7549, 1–7. [Google Scholar]
- Ko, A.C.-T.; Choo-Smith, L.P.; Hewko, M.; Leonardi, L.; Sowa, M.G.; Dong, C.C.S.; Williams, P.; Cleghorn, B. Ex vivo detection and characterization of early dental caries by optical coherence tomography and Raman spectroscopy. J. Biomed. Opt. 2005, 10, 031118. [Google Scholar] [CrossRef] [PubMed]
- Choo-Smith, L.P.; Hewko, M.D.; Dufour, M.L.; Fulton, C.; Qiu, P.L.; Gauthier, B.; Padioleau, C.; Bisaillon, C.E.; Dong, C.; Cleghorn, B.M.; et al. Ex vivo imaging of early dental caries within the interproximal space. Proc. SPIE 2009. [Google Scholar] [CrossRef]
- Shi, X.Q.; Welander, U.; Angmar-Mansson, B. Occlusal caries detection with KaVo DIAGNOdent and radiography: An in vitro comparison. Caries Res. 2000, 34, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Intes, X. Comparison of Monte Carlo methods for fluorescence molecular tomography-computational efficiency. Med. Phy. 2011, 38, 5788–5798. [Google Scholar] [CrossRef]
- Chen, J.; Venugopal, V.; Intes, X. Monte Carlo based method for fluorescence tomographic imaging with lifetime multiplexing using time gates. Biomed. Opt. Express 2011, 2, 871–886. [Google Scholar] [CrossRef] [PubMed]
- Intes, X.; Ripoll, J.; Chen, Y.; Nioka, S.; Yodh, A.G.; Chance, B. In vivo continuous-wave optical breast imaging enhanced with Indocyanine Green. Med. Phys. 2003, 30, 1039. [Google Scholar] [CrossRef] [PubMed]
- Muldoon, T.J.; Burgess, S.A.; Chen, B.R.; Ratner, D.R.; Hillman, E.M.C. Analysis of skin lesions using laminar optical tomography. Biomed. Opt. Express 2012, 3, 1701–1712. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Kurnar, C.; Chance, B. Experimental study of migration depth for the photons measured at sample surface. Proc. SPIE 1991, 1431, 180–191. [Google Scholar]
- Chen, J.; Fang, Q.; Intes, X. Mesh-based Monte Carlo method in time-domain widefield fluorescence molecular tomography. J. Biomed. Opt. 2012, 17, 106009. [Google Scholar] [PubMed]
- Burgess, S.A.; Ratner, D.; Chen, B.R.; Hillman, E.M.C. Fiber-optic and articulating arm implementations of laminar optical tomography for clinical applications. Biomed. Opt. Express 2010, 1, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Long, F.; Mehmet, S.O.; Xavier, I.; Shiva, K. Dental imaging using laminar optical tomography and micro CT. Proc. SPIE 2014. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Long, F.; Ozturk, M.S.; Wolff, M.S.; Intes, X.; Kotha, S.P. Dental Imaging Using Mesoscopic Fluorescence Molecular Tomography: An ex Vivo Feasibility Study. Photonics 2014, 1, 488-502. https://doi.org/10.3390/photonics1040488
Long F, Ozturk MS, Wolff MS, Intes X, Kotha SP. Dental Imaging Using Mesoscopic Fluorescence Molecular Tomography: An ex Vivo Feasibility Study. Photonics. 2014; 1(4):488-502. https://doi.org/10.3390/photonics1040488
Chicago/Turabian StyleLong, Feixiao, Mehmet S. Ozturk, Mark S. Wolff, Xavier Intes, and Shiva P. Kotha. 2014. "Dental Imaging Using Mesoscopic Fluorescence Molecular Tomography: An ex Vivo Feasibility Study" Photonics 1, no. 4: 488-502. https://doi.org/10.3390/photonics1040488
APA StyleLong, F., Ozturk, M. S., Wolff, M. S., Intes, X., & Kotha, S. P. (2014). Dental Imaging Using Mesoscopic Fluorescence Molecular Tomography: An ex Vivo Feasibility Study. Photonics, 1(4), 488-502. https://doi.org/10.3390/photonics1040488




