The Role of Ultraviolet Radiation in the Ocular System of Mammals
Abstract
:1. Introduction
2. Ocular Effects of UVR
2.1. Eyelid
2.2. Pterygium
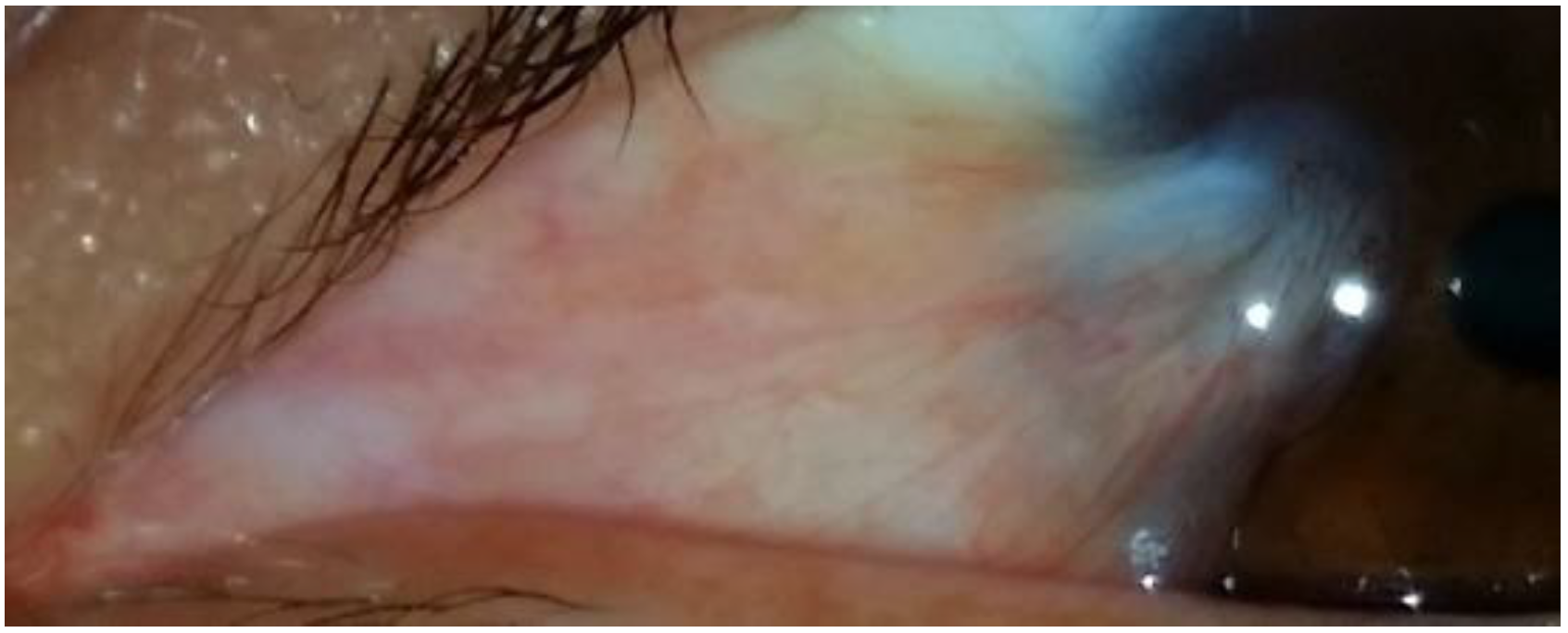
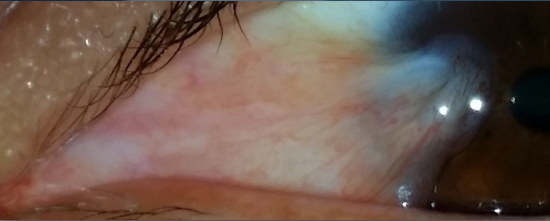
2.3. Pinguecula
2.4. Ocular Surface Squamous Neoplasia (OSSN)
2.5. Cataract
2.6. Climatic Droplet Keratopathy (CDK)
2.7. Age-Related Macular Degeneration (AMD)
2.8. Uveal Melanoma
2.9. Photokeratitis
3. Occupational Exposure
4. Protection Against UVR
4.1. Environmental Photoprotection
4.2. Ocular Photoprotection
4.3. Physical Photoprotection
| Mean UV Transmittance | |||||
|---|---|---|---|---|---|
| UVB | UVA | ||||
| Lens | Luminous transmittance (tv) | Normal use | Prolonged exposure | Normal use | Prolonged exposure |
| Cosmetic lens (light) | >40% | ≤12.5% tv | ≤1% UVB | Tv | ≤50% tv |
| General purpose lens(medium to dark) | 8%–40% | ≤12.5% tv | ≤1% UVB tv | Tv | ≤50% tv |
| Special purpose lens(very dark) | 3%–8% | ≤1% UVB | ≤1% UVB | _50% tv | ≤50% tv |
| Special purpose lens(strongly colored) | >8% | ≤1% UVB | ≤1% UVB | ≤50% tv | |
| UV Light Intensity | Minimal | Low | Moderate | High | Very High |
|---|---|---|---|---|---|
| International color codes | Green | Yellow | Orange | Red | Purple |
| Index | 0–2 | 3–5 | 6–7 | 8–10* | ≥11 |
5. The role of Therapeutic UVR in the Eye
5.1. Increased Corneal Graft Survival
5.2. Antimicrobial Effect
5.3. Collagen Crosslinking in the Cornea
Infectious Keratitis
Corneal Ectasia
5.4. UVR in Prevention of Progression of Myopia
6. Conclusions
Conflict of Interest
References
- World Health Organization, and International Commission on Non-Ionizing Radiation Protection. Global Solar UV Index: A Practical Guide; World Health Organization: Geneva, Switzerland.
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Available online: http://monographs.iarc.fr/ENG/Monographs/vol55/mono55.pdf (accessed on 17 October 2014).
- Wargent, J.J.; Jordan, B.R. From ozone depletion to agriculture: understanding the role of UV radiation in sustainable crop production. New Phytolo. 2013, 197, 1058–1076. [Google Scholar] [CrossRef]
- Suparata, W.; Bakar, F.N.A.; Abdullah, M. Remote sensing of Antactcic ozone depletion using GPS meterology. Int. J. Remote Sens. 2013, 34, 2519–2530. [Google Scholar] [CrossRef]
- Parrish, J.A.; Jaenicke, K.F.; Anderson, R.R. Erythema and melanogenesis action spectrum of normal human skin. Photochem. Photobiol. 1982, 36, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Gilchrest, B.A. Actinic injury. Annu. Rev. Med. 1990, 41, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Meeran, S.M.; Punathil, T.; Katiyar, S.K. IL-12 deficiency exacerbates inflammatory responses in UV-irradiated skin and skin tumors. J. Invest. Dermatol. 2008, 128, 2716–2727. [Google Scholar] [CrossRef] [PubMed]
- Farrukh, M.R.; Nissar, U.A.; Afnan, Q.; Rafiq, R.; Sharma, L.; Amin, S.; Kaiser, P.; Sharma, P.; Tasduq, S. Oxidative stress mediated Ca2+ release manifests endoplasmic reticulum stress leading to unfolded protein response in UV-B irradiated human skin cells. J. Dermatol. Sci. 2014, 75, 24–35. [Google Scholar] [CrossRef]
- Osipov, A.N.; Smetanina, N.M.; Pustovalova, M.V.; Arkhangelskaya, E.; Klokov, D. The formation of DNA single-strand breaks and alkali-labile sites in human blood lymphocytes exposed to 365-nm UVA radiation. Free Radic. Biol. Med. 2014, 73, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Balk, S.J. Ultraviolet radiation: a hazard to children and adolescents. Pediatrics 2011, 127, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Wu, S.B.; Hong, C.H.; Yu, H.S.; Wei, Y.H. Molecular Mechanisms of UV-Induced Apoptosis and its Effects on Skin Residential Cells: The Implication in UV-Based Phototherapy. Int. J. Mol. Sci. 2013, 14, 6414–6435. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, C.L.; Ananthaswamy, H.N. p53 and the pathogenesis of skin cancer. Toxicol. Appl. Pharmacol. 2007, 224, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Damian, D.L.; Barnetson, R.S.; Halliday, G.M. Low dose UVA and UVB have different time courses for suppression of contact hypersensitivity to a recall antigen in humans. J. Invest. Dermatol. 1999, 112, 939–944. [Google Scholar] [CrossRef] [PubMed]
- Nghiem, D.X.; Kazimi, N.; Cldesdale, G.; Ananthaswamy, H.N.; Kripke, M.L.; Ullrich, S.E. Ultraviolet A radiation suppresses an established immune response: Implications for sunscreen desigh. J. Invest. Dermatol. 2001, 117, 1193–1199. [Google Scholar] [CrossRef] [PubMed]
- Reme, C.; Reinboth, J.; Clausen, M.; Hafezi, F. Light damage revisited: Converging evidence, diverging views? Graefes. Arch. Clin. Exp. Ophthalmol. 1996, 234, 2–11. [Google Scholar] [CrossRef]
- Oriowo, O.M.; Cullen, A.P.; Sivak, J.G. Impairment of eye lens cell physiology and optics by broadband ultraviolet A-ultraviolet B radiation. Photochem. Photobiol. 2002, 76, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Clement-Lacroix, P.; Michel, L.; Moysan, A.; Morliere, P.; Dubertret, L. UVA-induced immune suppression in human skin: Protective effect of vitamin E in human epidermal cells in vitro. Br. J. Dermatol. 1996, 134, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Zigman, S.L. UVA photobiology. J. Ocul. Pharmacol. Ther. 2000, 16, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Young, S.; Sands, J. Sun and the eye: Prevention and detection of light-induced disease. Clin. Dermatol. 1998, 16, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Kolozsvari, L.; Nogradi, A.; Hopp, B.; Bor, Z. UV absorbance of the human cornea in the 240- to 400-nm range. Invest. Ophthalmol. Vis. Sci. 2002, 43, 2165–2168. [Google Scholar] [PubMed]
- Cullen, A.P. Photokeratitis and other phototoxic effects on the cornea and conjunctiva. Int. J. Toxicol. 2002, 21, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Strickland, P.T.; Vitasa, B.C.; West, S.K.; Rosenthal, F.S.; Emmett, E.A.; Taylor, H.R. Quantitative carcinogenesis in man: Solar ultraviolet B dose dependence of skin cancer in Maryland watermen. J. Natl. Cancer Inst. 1989, 81, 1910–1913. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, R.P.; Hill, G.B.; Bajdik, C.D.; Coldman, A.J.; Fincham, S.; McLean, D.I.; Threlfall, W.J. Sunlight exposure, pigmentation factors, and risk of nonmelanocytic skin cancer. II.Squamous cell carcinoma. Arch. Dermatol. 1995, 131, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Rosso, S.; Zanetti, R.; Martinez, C.; Tormo, M.J.; Schraub, S.; Sancho-Garnier, H.; Franceschi, S.; Gafà, L.; Perea, E.; Navarro, C.; et al. The multicenter south European study “Helios”. II: different sun exposure patterns in the aetiology of basal cell and squamous cell carcinomas of the skin. Br. J. Cancer 1996, 73, 1447–1454. [Google Scholar] [CrossRef] [PubMed]
- Corona, R.; Dogliotti, E.; D’Errico, M.; Sera, F.; Iavarone, I.; Baliva, G.; Chinni, L.M.; Gobello, T.; Mazzanti, C.; Puddu, P.; et al. Risk factors for basal cell carcinoma in a Mediterranean population: role of recreational sun exposure early in life. Arch. Dermatol. 2001, 137, 1162–1168. [Google Scholar] [PubMed]
- Kricker, A.; Armstrong, B.K.; English, D.R.; Heenan, P.J. Does intermittent sun exposure cause basal cell carcinoma? A case–control study in Western Australia. Int. J. Cancer 1995, 60, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Naldi, L.; DiLandro, A.; D’Avanzo, B.; Parazzini, F. Host-related and environmental risk factors for cutaneous basal cell carcinoma: evidence from an Italian case–control study. J. Am. Acad. Dermatol. 2000, 42, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Vainio, H.; Miller, A.B.; Bianchini, F. An international evaluation of the cancer-preventive potential of sunsc- reens. Int. J. Cancer 2000, 88, 838–842. [Google Scholar] [CrossRef] [PubMed]
- Brenner, M.; Hearing, V.J. The Protective Role of Melanin against UV Damage in Human Skin. Photochem. Photobiol. 2008, 84, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Fischer, T.W.; Slominski, A.; Zmijewski, M.A.; Reiter, R.J.; Paus, R. Melatonin as a major skin protectant: From free radical scavenging to DNA damage repair. Expl. Dermatol. 2008, 17, 713–730. [Google Scholar]
- Desotelle, J.A.; Wilking, M.J.; Ahmad, N. The Circadian Control of Skin and Cutaneous Photodamage. Photochem. Photobiol. 2013, 88, 1037–1047. [Google Scholar] [CrossRef]
- Makhzoumi, Z.H.; Arron, S.T. Photosensitizing Agents and Risk of Non-Melanoma Skin Cancer: A Population-Based Case-Control Study. J. Invest. Dermatol. 2013, 133, 1922–1923. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.N.; Zens, M.S.; Perry, A.E. Photosensitizing agents and risk of non-melanotic skin cancer: a population based case-control study. J. Invest. Dermatol. 2013, 133, 1950–1955. [Google Scholar] [CrossRef] [PubMed]
- Moran, D.J.; Hollows, F.C. Pterygium and ultraviolet radiation: a positive correlation. Br. J. Ophthalmol. 1984, 68, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.E.; Bergmanson, J.P.G.; Wallace, D.; Saldana, G.; Dempsey, H.; McEvoy, H.; Collum, L.M.T. Quantification of the ultraviolet radiation (UVR) field in the human eye in vivo using novel instrumentation and the potential benefits of UVR blocking hydrogel contact lens. Br. J. Ophthalmol. 2001, 85, 1080–1085. [Google Scholar] [CrossRef] [PubMed]
- Taylor, H.R.; West, S.K.; Rosenthal, F.S.; Munoz, B.; Newland, H.S.; Emmett, E.A. Corneal changes associated with chronic ultraviolet radiation. Arch. Ophthalmol. 1989, 107, 1481–1484. [Google Scholar] [CrossRef] [PubMed]
- Threllfall, T.J.; English, D.R. Sun exposure and pterygium of the eye: A dose–response curve. Am. J. Ophthalmol. 1999, 128, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Mimura, T.; Usui, T.; Mori, M.; Yamamoto, H.; Obata, H.; Yamagami, S.; Funatsu, H.; Noma, H.; Honda, N.; Amano, S. Pinguecula and contact lenses. Eye 2010, 24, 1685–1691. [Google Scholar] [CrossRef] [PubMed]
- Clear, A.S.; Chirambu, M.C.; Hutt, M.S.R. Solar keratosis, pterygium and squamous cell carcinoma of the conjunctiva in Malawi. Br. J. Ophthalmol. 1979, 63, 1902–1909. [Google Scholar] [CrossRef]
- Norn, M.S. Spheroid degeneration, pinguecula, and pterygium among Arabs in the Red Sea territory, Jordan. Acta. Ophthalmol. (Copenh.) 1982, 60, 949–954. [Google Scholar] [CrossRef]
- Klintworth, G.K. Chronic actinic keratopathy—a condition associated with conjunctival elastosis (pingueculae) and typified by characteristic extracellular concretions. Am. J. Pathol. 1972, 67, 327–348. [Google Scholar] [PubMed]
- Johnson, G.J. Aetiology of spheroidal degeneration of the cornea in Labrador. Br. J. Ophthalmol. 1981, 65, 270–283. [Google Scholar] [CrossRef] [PubMed]
- Pe’er, J. Ocular surface squamous neoplasia. Ophthalmol. Clin. North. Am. 2005, 18, 1–13. [Google Scholar]
- Templeton, A.C. Tumors of the eye and adnexa in Africans of Uganda. Cancer 1967, 20, 1689–1698. [Google Scholar] [CrossRef]
- Pola, E.C.; Masanganise, R.; Rusakaniko, S. The trend of ocular surface squamous neoplasia among ocular surface tumour biopsies submitted for histology from Sekuru. Kaguvi Eye Unit, Harare between 1996 and 2000. Cent. Afr. J. Med. 2003, 49, 1–4. [Google Scholar] [PubMed]
- Lee, G.A.; Hirst, L.W. Incidence of ocular surface epithelial dysplasia in metropolitan Brisbane. A 10-year survey. Arch. Ophthalmol. 1992, 110, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Sun, E.C.; Fears, T.R.; Goedert, J.J. Epidemiology of squamous cell conjunctival cancer. Cancer Epidemiol. Biomarkers Prev. 1997, 6, 73–77. [Google Scholar] [PubMed]
- Lee, G.A.; Williams, G.; Hirst, L.W.; Green, A.C. Risk factors in the development of ocular surface epithelial dysplasia. Ophthalmology 1994, 101, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, F.S.; Phoon, C.; Bakalian, A.E.; Taylor, H.R. The ocular does of ultraviolet radiation to outdoor workers. Invest. Ophthalmol. Vis. Sci. 1988, 29, 649–656. [Google Scholar]
- Erie, J.C.; Campbell, R.J.; Liesegang, T.J. Conjunctival and corneal intraepithelial and invasive neoplasia. Ophthalmology 1986, 93, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Taylor, H.R.; West, S.K.; Rosenthal, F.S.; Muñoz, B.; Newland, H.S.; Abbey, H.; Emmett, E.A. Effect of ultraviolet radiation on cataract formation. N Engl. J. Med. 1988, 319, 1429–1433. [Google Scholar] [CrossRef] [PubMed]
- Dolezal, J.M.; Perkins, E.S.; Wallace, R.B. Sunlight, skin sensitivity, and senile cataract. Am. J. Epidemiol. 1989, 129, 559–568. [Google Scholar] [PubMed]
- Bochow, T.W.; West, S.K.; Azar, A.; Munoz, B.; Sommer, A.; Taylor, H.R. Ultraviolet light exposure and risk of posterior subcapsular cataracts. Arch. Ophthalmol. 1989, 107, 369–372. [Google Scholar] [CrossRef] [PubMed]
- Mohan, M.; Sperduto, R.D.; Angra, S.K.; Milton, R.C.; Mathur, R.L.; Underwood, B.A.; Jaffery, N.; Pandya, C.B.; Chhabra, V.K.; Vajpayee, R.B. India–US case–control study of age-related cataracts India–US Case–Control Study Group. Arch. Ophthalmol. 1989, 107, 670–676. [Google Scholar]
- Leske, M.C.; Chylack, L.T., Jr.; Wu, S.Y. The Lens Opacities Case–Control Study. Risk factors for cataract. Arch. Ophthalmol. 1991, 109, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Cruickshanks, K.J.; Klein, B.E.; Klein, R. Ultraviolet light exposure and lens opacities: the Beaver Dam Eye Study. Am. J. Public Health 1992, 82, 1658–1662. [Google Scholar] [CrossRef] [PubMed]
- Rosmini, F.; Stazi, M.A.; Milton, R.C.; Sperduto, R.D.; Pasquini, P.; Maraini, G. A dose–response effect between a sunlight index and age-related cataracts. Italian-American Cataract Study Group. Ann. Epidemiol. 1994, 4, 266–270. [Google Scholar] [CrossRef] [PubMed]
- Hirvela, H.; Luukinen, H.; Laatikainen, L. Prevalence and risk factors of lens opacities in the elderly in Finland. Apopulation-based study. Ophthalmology 1995, 102, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Javitt, J.C.; Taylor, H.R. Cataract and latitude. Doc. Ophthalmol. 1994, 88, 307–325. [Google Scholar] [CrossRef] [PubMed]
- West, S.K.; Duncan, D.D.; Munoz, B.; Rubin, G.S.; Fried, L.P.; Bandeen-Roche, K.; Schein, O.D. Sunlight exposure and risk of lens opacities in a population-based study: the Salisbury Eye Evaluation project. J. Am. Med. Assoc. 1998, 280, 714–718. [Google Scholar] [CrossRef]
- McCarty, C.A.; Nanjan, M.B.; Taylor, H.R. Attributable risk estimates for cataract to prioritize medical and public health action. Invest. Ophthalmol. Vis. Sci. 2000, 41, 3720–3725. [Google Scholar] [PubMed]
- Katoh, N.; Jonasson, F.; Sasaki, H.; Kojima, M.; Ono, M.; Takahashi, N.; Sasaki, K. Cortical lens opacification in Iceland. Risk factor analysis—Reykjavik Eye Study. Acta Ophthalmol. Scand 2001, 79, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Neale, R.E.; Purdie, J.L.; Hirst, L.W.; Green, A.C. Sun exposure as a risk factor for nuclear cataract. Epidemiology 2003, 14, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Taylor, H.R. The environment and the lens. Br. J. Ophthalmol. 1980, 64, 303–310. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hollows, F.; Moran, D. Cataract—the ultraviolet risk factor. Lancet 1981, 2, 1249–1250. [Google Scholar] [CrossRef] [PubMed]
- Brilliant, L.B.; Grasset, N.C.; Pokhrel, R.P.; Kolstad, A.; Lepkowski, J.M.; Brilliant, G.E.; Hawks, W.N. Associations among cataract prevalence, sunlight hours, and altitude in the Himalayas. Am. J. Epidemiol. 1983, 118, 250–264. [Google Scholar] [PubMed]
- Wong, L.; Ho, S.C.; Coggon, D.; Cruddas, A.M.; Hwang, C.H.; Ho, C.P.; Robertshaw, A.M.; MacDonald, D.M. Sunlight exposure, antioxidant status, and cataract in Hong Kong fishermen. J. Epidemiol. Community Health 1993, 47, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Perkins, E.S. The association between pinguecula, sunlight and cataract. Ophthalmic Res. 1985, 17, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Collman, G.W.; Shore, D.L.; Shy, C.M.; Checkoway, H.; Luria, A.S. Sunlight and other risk factors for cataracts: an epidemiologic study. Am. J. Public Health 1988, 78, 1459–1462. [Google Scholar] [CrossRef] [PubMed]
- Prokofyeva, E.; Wegener, A.; Zrenner, E. Cataract Prevalence and Prevention in Europe: A Literature Review. Acta. Ophthalmologica. 2013, 91, 395–405. [Google Scholar]
- Kronschlager, M.; Lofgren, S.; Yu, Z.; Talebizadeh, N.; Varma, S.; Soderberg, P. Caffeine Eye Drops Protect Against UV-B Cataract. Exp. Eye Res. 2013, 113, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Maddock, J.E.; O’Riordan, D.L.; Lee, T.; Mayer, J.; McKenzie, T.L. Use of Sunglasses in Public Outdoor Recreation Settings in Honolulu, Hawaii. Optom. Vis. Sci. 2009, 86, 165–166. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.J. The Environment and the Eye. Eye 2004, 18, 1235–1250. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Oliva, M.S.; Taylor, H. Ultraviolet radiation and the eye. Int. Ophthalmol. Clin. 2005, 45, 1–17. [Google Scholar] [PubMed]
- Gray, R.H.; Johnson, G.J.; Freedman, A. Climatic Droplet Keratopathy. Surv. Ophthalmol. 1992, 36, 241–253. [Google Scholar]
- Delcourt, C.; Carriere, I.; Ponton-Sanchez, A.; Fourrey, S.; Lacroux, A.; Papoz, L. Light exposure and the risk of age-related macular degeneration. Arch. Ophthalmol. 2001, 119, 1463–1468. [Google Scholar] [CrossRef] [PubMed]
- Cruickshanks, K.J.; Klein, R.; Klein, B.E. Sunlight andage-related macular degeneration. The Beaver Dam EyeStudy. Arch. Ophthalmol. 1993, 111, 514–518. [Google Scholar] [CrossRef] [PubMed]
- West, S.K.; Rosenthal, F.S.; Bressler, N.M.; Bressler, S.B.; Munoz, B.; Fine, S.L.; Taylor, H.R. Exposure to sunlight and other riskfactors for age-related macular degeneration. Arch. Ophthalmol. 1989, 107, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Taylor, H.R.; West, S.; Munoz, B.; Rosenthal, F.S.; Bressler, S.B.; Bressler, N.M. The long-term effects of visible lighton the eye. Arch. Ophthalmol. 1992, 110, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Darzins, P.; Mitchell, P.; Heller, R.F. Sun exposure and age-related macular degeneration. An Australian case–control study. Ophthalmology 1997, 104, 770–776. [Google Scholar] [CrossRef] [PubMed]
- McCarty, C.A.; Mukesh, B.N.; Fu, C.L.; Mitchell, P.; Wang, J.J.; Taylor, H.R. Risk factors for age-related maculopathy: The Visual Impairment Project. Arch. Ophthalmol. 2001, 119, 1455–1462. [Google Scholar] [CrossRef] [PubMed]
- Khan, J.C.; Shahid, H.; Thurlby, D.A.; Bradley, M.; Clayton, D.G.; Moore, A.T.; Bird, A.C.; Yates, J.R. Age related macular degeneration and sun exposure, iris colour, and skin sensitivity to sunlight. Br. J. Ophthalmol. 2006, 90, 29–32. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mitchell, P.; Smith, W.; Wang, J.J. Iris colour skin sun sensitivity and age-related maculopathy. The Blue Mountain Eye Study. Ophthalmology 1998, 105, 1359–1363. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.J.; Jakobsen, K.; Smith, W.; Mitchell, P. Five-year incidence of age-related maculopathy in relation to iris, skin or hair colour and skin sun sensitivity: the Blue Mountain Eye Study. Ophthalmology 2003, 31, 317–321. [Google Scholar]
- De Oliveira Miguel, N.C.; Meyer-Rochow, V.B.; Allodi, S. A structural study of the retinal photoreceptor, plexiform and ganglion cell layers following exposure to UV-B and UV-C radiation in the albino rat. Micron 2003, 34, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Ajani, U.A.; Seddon, J.M.; Hsieh, C.C.; Egan, K.M.; Albert, D.M.; Gragoudas, E.S. Occupation and risk of uveal melanoma. An exploratory study. Cancer 1992, 70, 2891–2900. [Google Scholar] [CrossRef] [PubMed]
- Holly, E.A.; Aston, D.A.; Char, D.H.; Kristiansen, J.J.; Ahn, D.K. Uveal melanoma in relation to ultraviolet light exposure and host factors. Cancer Res. 1990, 50, 5773–5777. [Google Scholar] [PubMed]
- Tucker, M.A.; Shields, J.A.; Hartge, P.; Augsburger, J.; Hoover, R.N.; Fraumeni, J.F., Jr. Sunlight exposure as risk factor for intraocular malignant melanoma. N Engl. J. Med. 1985, 313, 789–792. [Google Scholar] [CrossRef] [PubMed]
- Vajdic, C.M.; Kricker, A.; Giblin, M.; McKenzie, J.; Aitken, J.; Giles, G.G.; Armstrong, B.K. Sun exposure predicts risk of ocular melanoma in Australia. Int. J. Cancer 2002, 101, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.M.; Gragoudas, E.S.; Glynn, R.J.; Egan, K.M.; Albert, D.M.; Blitzer, P.H. Host factors, UV radiation, and risk of uveal melanoma. A case–control study. Arch. Ophthalmol. 1990, 108, 1274–1280. [Google Scholar] [CrossRef]
- Pane, A.R.; Hirst, L.W. Ultraviolet light exposure as a risk factor for ocular melanoma in Queensland, Australia. Ophthalmic Epidemiol. 2000, 7, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Guenel, P.; Laforest, L.; Cyr, D.; Févotte, J.; Sabroe, S.; Dufour, C.; Lutz, J.M.; Lynge, E. Occupational risk factors, ultraviolet radiation, and ocular melanoma: a case–control study in France. Cancer Causes Control. 2001, 12, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Shah, C.P.; Weis, E.; Lajous, M.; Shields, J.A.; Shields, C.L. Intermittent and chronic ultraviolet light exposure and uveal melanoma: a meta-analysis. Ophthalmology 2005, 112, 1599–1607. [Google Scholar] [CrossRef] [PubMed]
- Vajdic, C.M.; Kricker, A.; Giblin, M.; McKenzie, J.; Aitken, J.; Giles, G.G.; Armstrong, B.K. Eye colour and cutaneous nevi predict risk of ocular melanoma in Australia. Int. J. Cancer 2001, 92, 906–912. [Google Scholar]
- Bachem, A. Ophthalmic ultraviolet action spectra. Am. J. Ophthalmol. 1956, 41, 969–975. [Google Scholar] [CrossRef] [PubMed]
- Tenkate, T.D. Occupational exposure to ultraviolet radiation: a health risk assessment. Rev Environ Health 1999, 14, 187–209. [Google Scholar] [CrossRef] [PubMed]
- Turaka, K.; Shields, C.L.; Shah, C.P.; Say, E.A.; Shields, J.A. Bilateral uveal melanoma in an arc welder. Graefes. Arch. Clin. Exp. Ophthalmol. 2011, 249, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Shao, D.; Ding, X.; Liang, X.; Yang, J.; Li, J. Chronic phototoxic maculopathy caused by welding arc in occupational welders. Can. J. Ophthalmol. 2012, 47, 45–50. [Google Scholar] [CrossRef]
- Ajayi Iyiade, A.; Omotoye Olusola, J. Pattern of eye diseases among welders in a Nigeria community. Afr. Health Sci. 2012, 12, 210–216. [Google Scholar]
- Tenkate, T.D. Optical radiation hazards of welding arcs. Rev. Environ. Health 1998, 13, 131–146. [Google Scholar] [CrossRef] [PubMed]
- Abarca, J.F.; Casiccia, C.C.; Zamorano, F.D. Increase in sunburns and photosensitivity disorders at the edge of the Antarctic ozone hole, southern Chile, 1986–2000. J. Am. Acad. Dermatol. 2002, 46, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Kullavanijaya, P.; Lim, H.W. Photoprotection. J. Am. Acad. Dermatol. 2005, 52, 937–958. [Google Scholar] [CrossRef] [PubMed]
- Longstreth, J. Anticipated public health consequences of global climate change. Environ. Health Perspect. 1991, 96, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Young, S.; Sands, J. Sun and the eye: Prevention and detection of light-induced disease. Clin. Dermatol. 1998, 16, 477–485. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, R.L.; Bjorn, L.O.; Bais, A.; Ilyasad, M. Changes in biologically active ultraviolet radiation reaching the Earth’s surface. Photochem. Photobiol. Sci. 2003, 2, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Babu, V.; Misra, R.B.; Joshi, P.C. Ultraviolet-B effects on ocular tissues. Biochem. Biophys. Res. Commun. 1995, 210, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Snodderly, D.M. Evidence for protection against age-related macular degeneration by carotenoids and antioxidant vitamins. Am. J. Clin. Nutr. 1995, 62, 1448S–1461S. [Google Scholar]
- Ringvold, A. Corneal epithelium and UV-protection of the eye. Acta. Ophthalmol. Scand. 1998, 76, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Reddy, G.B.; Bhat, K.S. Protection against UVB inactivation (in vitro) of rat lens enzymes by natural antioxidants. Mol. Cell. Biochem. 1999, 194, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Reme, C.; Reinboth, J.; Clausen, M.; Hafezi, F. Light damage revisited: converging evidence, diverging views? Graefes. Arch. Clin. Exp. Ophthalmol. 1996, 234, 2–11. [Google Scholar] [CrossRef]
- Dillon, J. The photophysics and photobiology of the eye. J. Photochem. Photobiol. B 1991, 10, 23–40. [Google Scholar]
- Pitts, D.G.; Bergmanson, J.P.G. The UV problem: have the rules changed? J. Am. Optom. Assoc. 1989, 60, 420–424. [Google Scholar]
- Walsh, J.E.; Bergmanson, J.P. Does the eye benefit from wearing ultraviolet-blocking contact lenses? Eye Contact Lens 2011, 37, 267–272. [Google Scholar] [CrossRef]
- Bergmanson, J.P.; Sheldon, T.M. Ultraviolet radiation revisited. CLAO J. 1997, 23, 196–204. [Google Scholar] [PubMed]
- American Academy of Ophthalmology. This summer keep an eye on UV safety. Available online: www.aao.org/newsroom/release/ 20070629.cfm (accessed on 8 August 2013).
- American Optometric Association. UV protection. Available online: www.aoa.org/uvprotection.xml (accessed on 8 August 2013).
- Lee, G.A.; Hirst, L.W.; Sheehan, M. Knowledge of sunlight effects on the eyes and protective behaviors in the general community. Ophthalmic Epidemiol. 1994, 1, 67–84. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.A.; Hirst, L.W.; Sheehan, M. Knowledge of sunlight effects on the eyes and protective behaviors in adolescents. Ophthalmic Epidemiol. 1999, 6, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Tuchinda, C.; Srivannaboon, S.; Lim, H.W. Photoprotection by window glass, automobile glass, and sunglasses. J. Am. Acad. Dermatol. 2006, 54, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Q.; Balagula, Y.; Osterwalder, U. Photoprotection: A review of the current and future technologies. Dermatol. Ther. 2010, 23, 31–47. [Google Scholar] [CrossRef]
- Leow, Y.H.; Tham, S.N. UV-protective sunglasses for UVA irradiation protection. Int. J. Dermatol. 1995, 34, 808–810. [Google Scholar] [CrossRef] [PubMed]
- Semes, L. UV-A absorbing characteristics of commercial sunglasses intended for recreational and general use. J. Am. Optom. Assoc. 1991, 62, 754–758. [Google Scholar] [PubMed]
- Dain, S.J. Sunglasses and sunglass standards. Clin. Exp. Optom. 2003, 86, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Sliney, D.H. Epidemiological studies of sunlight and cataract: The critical factor of ultraviolet exposure geometry. Ophthalmic Epidemiol. 1994, 1, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Coroneo, M.T.; Muller-Stolzenburg, N.W.; Ho, A. Peripheral light focusing by the anterior eye and the ophthalmohelioses. Ophthalmic Surg. 1991, 22, 705–711. [Google Scholar] [PubMed]
- American Optometric Association. Shopping guide for sunglasses. Available online: http://aoa.org/documents/SunglassShopping Guide0810.pdf (accessed on 2 August 2011).
- Carolyn, B.; Lyde, R.; Bergstresser, P.R. Ultraviolet protection from sun avoidance. Dermatol. Ther. 1997, 4, 72–78. [Google Scholar]
- Anstey, A.; Taylor, D.; Chalmers, I.; Ansari, E. Ultraviolet radiation blocking characteristics of contact lenses: Relevance to eye protection for psoralen-sensitised patients. Photodermatol. Photoimmunol. Photomed. 1999, 15, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.A.; Fusaro, R.M. Broad-spectrum photoprotection: the roles of tinted auto windows, sunscreens and browning agents in the diagnosis and treatment of photosensitivity. Dermatol. 1992, 185, 237–241. [Google Scholar] [CrossRef]
- Anstey, A.; Taylor, D.; Chalmers, I.; Ansari, E. Ultraviolet radiation-blocking characteristics of contact lenses: relevance to eye protection for psoralen-sensitised patients. Photodermatol. Photoimmunol. Photomed. 1999, 15, 193–197. [Google Scholar] [CrossRef] [PubMed]
- National Weather Service Climate Prediction Center. UV index: Information. Available online: www.cpc.ncep.noaa.gov/products/stratosphere/uv_index/uv_what.shtml (accessed on 11 August 2013).
- Guymer, R.H.; Mandel, T.E. UV-B irradiation of donor skin and cornea prior to allotransplantation in mice. Transplant. Proc. 1989, 21, 3771–3771. [Google Scholar] [PubMed]
- Dana, M.R.; Olkowski, S.T.; Ahmadian, H.; Stark, W.J.; Young, E.M. Low-dose ultraviolet-B irradiation of donor corneal endothelium and graft survival. Invest. Ophthalmol. Vis. Sci. 1990, 31, 2261–2268. [Google Scholar] [PubMed]
- Norval, M.; Halliday, G.M. The consequences of UV-Induced Immunosuppression for Human Health. Photochem. Photobiol. 2011, 87, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.C.; Sarvan, J.; Maske, R.; Els, W.J. Evidence that UV-B irradiation decreases corneal Langerhans cells and improves corneal graft in the rabbit. Transplantation. 1994, 57, 1281–1284. [Google Scholar] [CrossRef] [PubMed]
- Tsugita, A.; Okada, Y.; Uehara, K. Photosensitized inactivation of ribonucleic acids in the presence of riboflavin. Biochim. Biophys. Acta. 1965, 103, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Cardo, L.J.; Salata, J.; Mendez, J.; Reddy, H.; Goodrich, R. Pathogen inactivation of Trypanosoma cruzi in plasma and platelet concentrates using riboflavin and ultraviolet light. Transfus. Apher. Sci. 2007, 37, 131–137. [Google Scholar] [CrossRef]
- Martins, S.A.; Combs, J.C.; Noguera, G.; Camacho, W.; Wittmann, P.; Walther, R.; Cano, M.; Dick, J.; Behrens, A. Antimicrobial efficacy of riboflavin/UVA combination (365 nm) in vitro for bacterial and fungal isolates: A potential new treatment for infectious keratitis. Invest. Ophthalmol. Vis. Sci. 2008, 49, 3402–3408. [Google Scholar] [CrossRef] [PubMed]
- Makdoumi, K.; Backman, A.; Mortensen, J.; Crafoord, S. Evaluation of antibacterial efficacy of photo-activated riboflavin using ultraviolet light (UVA). Graefes. Arch. Clin. Exp. Ophthalmol. 2010, 248, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Anwar, H.M.; El-Danasoury, A.M.; Hashem, A.N. Corneal collagen crosslinking in the treatment of infectious keratitis. Clin. Ophthalmol. 2011, 5, 1277–1280. [Google Scholar] [CrossRef] [PubMed]
- Kohlhaas, M.; Spoerl, E.; Schilde, T.; Unger, G.; Wittig, C.; Pillunat, L.E. Biomechanical evidence of the distribution of cross-links in corneas treated with riboflavin/ultraviolet a light. J. Cataract Refract. Surg. 2006, 32, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Spoerl, E.; Mrochen, M.; Sliney, D.; Trokel, S.; Seiler, T. Safety of UVA–Riboflavin Cross-Linking of the Cornea and Theo Seiler. Cornea 2007, 26, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Ashwin, P.T.; McDonnell, P.J. Collagen cross-linkage: a comprehensive review and directions for future research. Br. J. Ophthalmol. 2010, 94, 965–970. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, H.E. Strengthening the cornea. Cornea 2004, 23, 432. [Google Scholar] [CrossRef]
- Kozobolis, V.; Labiris, G.; Gkika, M.; Sideroudi, H.; Kaloghianni, E.; Papadopoulou, D.; Toufexis, G. UV-A Collagen Cross-Linking Treatment of Bullous Keratopathy Combined With Corneal Ulcer. Cornea 2010, 29, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Spoerl, E.; Huhle, M.; Seiler, T. Induction of cross-links in corneal tissue. Exp. Eye Res. 1998, 66, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Wollensak, G.; Spoerl, E.; Seiler, T. Riboflavin/ultraviolet-Ainduced collagen crosslinking for the treatment of keratoconus. Am. J. Ophthalmol. 2003, 135, 620–627. [Google Scholar] [CrossRef] [PubMed]
- Spoerl, E.; Wollensak, G.; Seiler, T. Increased resistance of crosslinked cornea against enzymatic digestion. Curr. Eye Res. 2004, 29, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Iseli, H.P.; Thiel, M.A.; Hafezi, F.; Kampmeier, J.; Seiler, T. Ultraviolet A/riboflavin corneal cross-linking for infectious keratitis associated with corneal melts. Cornea 2008, 27, 590–594. [Google Scholar] [CrossRef] [PubMed]
- Moren, H.; Malmsjo, M.; Mortensen, J.; Ohrström, A. Riboflavin and ultraviolet a collagen crosslinking of the cornea for the treatment of keratitis. Cornea 2010, 29, 102–104. [Google Scholar] [CrossRef] [PubMed]
- Khan, Y.A.; Kashiwabuchi, R.T.; Martins, S.A.; Castro-Combs, J.M.; Kalyani, S.; Stanley, P.; Flikier, D.; Behrens, A. Riboflavin and ultraviolet light a therapy as an adjuvant treatment for medically refractive Acanthamoeba keratitis: Report of 3 cases. Ophthalmology 2011, 118, 324–331. [Google Scholar] [CrossRef]
- Makdoumi, K.; Mortensen, J.; Sorkhabi, O.; Malmvall, B.E.; Crafoord, S. UVA-riboflavin photochemical therapy of bacterial keratitis: A pilot study. Arch. Clin. Exp. Ophthalmol. 2012, 250, 95–102. [Google Scholar] [CrossRef]
- Butler, T.K.; Males, J.J.; Robinson, L.P.; Wechsler, A.W.; Sutton, G.L.; Cheng, J.; Taylor, P.; McClellan, K. Six-year review of Acanthamoeba keratitis in New South Wales, Australia: 1997–2002. Clin. Exp. Ophthalmol. 2005, 33, 41–46. [Google Scholar] [CrossRef]
- Lee, S.J.; Jeong, H.J.; Lee, J.E.; Xuan, Y.H.; Kong, H.H.; Chung, D.I.; Ock, M.S.; Yu, H.S. Molecular characterization of Acanthamoeba isolated from amebic keratitis related to orthokeratology lens overnight wear. Korean J. Parasitol. 2006, 44, 313–320. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ehlers, N.; Hjortdal, J. Riboflavin-ultraviolet light induced cross-linking in endothelial decompensation. Acta. Ophthalmol. 2008, 86, 549–551. [Google Scholar]
- Wollensak, G. Crosslinking treatment of progressive keratoconus: new hope. Curr. Opin. Ophthalmol. 2006, 17, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Wollensak, G.; Aurich, H.; Wirbelauer, C.; Pham, D.T. Potential use of riboflavin/UVA cross-linking in bullous keratopathy. Ophthalmic Res. 2009, 41, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Schnitzler, E.; Sporl, E.; Seiler, T. Irradiation of cornea with ultraviolet light and riboflavin administration as a new treatment for erosive corneal processes, preliminary results in four patients. Klin. Monatsbl. Augenheilkd. 2000, 217, 190–193. [Google Scholar] [CrossRef] [PubMed]
- Ngo, C.; Saw, S.M.; Dharani, R.; Flitcroft, I. Point-Counterpoint. Does sunlight (bright lights) explain the protective effects of outdoor activity against myopia? Ophthalmic Physiol. Opt. 2013, 33, 368–372. [Google Scholar]
- Rose, K.A.; Morgan, I.G.; Ip, J.; Kifley, A.; Huynh, S.; Smith, W.; Mitchell, P. Outdoor activity reduces the prevalence of myopia in children. Ophthalmology 2008, 115, 1279–1285. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Majdi, M.; Milani, B.Y.; Movahedan, A.; Wasielewski, L.; Djalilian, A.R. The Role of Ultraviolet Radiation in the Ocular System of Mammals. Photonics 2014, 1, 347-368. https://doi.org/10.3390/photonics1040347
Majdi M, Milani BY, Movahedan A, Wasielewski L, Djalilian AR. The Role of Ultraviolet Radiation in the Ocular System of Mammals. Photonics. 2014; 1(4):347-368. https://doi.org/10.3390/photonics1040347
Chicago/Turabian StyleMajdi, Mercede, Behrad Y. Milani, Asadolah Movahedan, Lisa Wasielewski, and Ali R. Djalilian. 2014. "The Role of Ultraviolet Radiation in the Ocular System of Mammals" Photonics 1, no. 4: 347-368. https://doi.org/10.3390/photonics1040347
APA StyleMajdi, M., Milani, B. Y., Movahedan, A., Wasielewski, L., & Djalilian, A. R. (2014). The Role of Ultraviolet Radiation in the Ocular System of Mammals. Photonics, 1(4), 347-368. https://doi.org/10.3390/photonics1040347