Abstract
The main objective discussed in this research was to determine the structural characteristics of carob kibble water-soluble polysaccharide (KWSP), extracted from Ceratonia siliqua L., and to assess its in vitro antioxidant activities, as well as its effect on whipped cream. The results obtained through 13C, 1H, and the hetero-nuclear single quantum coherence (HSQC) nuclear magnetic resonance (NMR) and Fourier transform infrared spectroscopy (FT-IR) analysis indicated that KWSP had the characteristic bands of polysaccharides. Thin layer chromatography (TLC) and high performance liquid chromatography (HPLC) suggested that KWSP is a hetero-polysaccharide composed of glucose and fructose, with an average molecular weight (Mw) amounting to 65 KDa. In addition, KWSP showed a good water holding capacity (WHC), a good oil holding capacity (OHC), and an emulsification stability, rated as 3.14 ± 0.05 g/g, 0.87 ± 0.02 g/g, and 71 ± 0.01%, respectively. The antioxidant activity of KWSP was investigated in vitro, demonstrating important DPPH and ABTS⋅+ radical scavenging activities and a good total antioxidant capacity. KWSP exhibited antibacterial activity against Listeria monocytogenes, Staphylococcus aureus, and Salmonella enterica. On the other hand, the incorporation of KWSP in whipped cream was investigated, to enhance its antioxidant capacity and consequently to extend its expiration date. Moreover, KWSP reduces the formation of conjugated dienes and trienes in cream fat.
1. Introduction
Plants have served humans and animals to meet their nutritional and medicinal requirements. Medicinal plants have been used in traditional medicine and ethno-medicine worldwide [1]. One of these medicinal plants is the Ceratonia siliqua L., commonly known as carob.
Ceratonia siliqua L. belongs to the family of Fabaceae trees and has been used in various industries (food, cosmetic products, pharmaceutical sectors, etc.) that require the functional substances extracted from the carob fruit [2]. The carob tree has been grown since antiquity in most countries of the Mediterranean basin. Its value was recognized by the ancient Greeks and by the Arabs, who disseminated it throughout the countries of North Africa and north into Spain and Portugal [3]. It has spread in recent times to many other countries, such as Mexico, Chile, Argentina, etc. Due to its sugary pulp, carob pods have been used as a cocoa substitute in confectionaries or as a fermented syrup. The seeds (∼10% of the pod) are available commercially for carob bean gum extraction (locust bean gum is especially used in the food industry) [4]. Furthermore, extensive research has revealed that carob kibble and seeds exhibit various bioactive molecules, such as phenolic compounds (flavonoids and tannins) and polysaccharides that have antioxidant, anti-inflammatory, and antibacterial activities [3,5,6]. Reactive oxygen species (ROS) are very reactive molecules that can damage macromolecules in organs, cells, and tissues. The abundance of ROS-like superoxide anion, hydrogen peroxide, and hydroxyl radicals with high concentration may play a key role in treating neurodegenerative, neoplasia, and atherosclerosis diseases [7]. Therefore, the immune system can be protected from oxidation by reducing ROS. Numerous researchers have demonstrated that polysaccharides can offer protection to living organisms against free radicals [8]. In this sense, there has been a great deal of interest among researchers in exploring new polysaccharides derived from animal, plant, algal, and microbial sources that can be assembled from monosaccharides joined by O-glycosidic linkages [9]. Thanks to their molecular weight, monosaccharide composition, type of glycosidic bonds, and degree of branching, as well as three-dimensional conformation, these macromoleculares provide a variety of biological activities, such as being anti-oxidation, anti-radiation, immunity enhancing, anti-tumor, and anti-aging, as well as lowering blood sugar [10].
Recently, researchers on natural bioactive substances have been particularly focused on the incorporation of natural compounds that have biological activities, to protect food against viruses and contaminants and reduce lipid oxidation to refine their characteristics/composition [11]. Polysaccharides isolated and characterized from carob pulp and seeds exhibit functional properties that further affect their application in various industrial areas [12]. The endosperm of carob seeds contains a large amount of galactomannan, which is invested in the production of locust gum as a stabilizer added to multiple different foods [13]. It is noteworthy that prior works emphasized that, although the value of carob kibbles polysaccharide is increasingly being incorporated into health foods, their properties and physicochemical characteristics have been little studied. Within this framework, Benković et al. [14] highlighted the abundance of polysaccharides in the carob kibbles and determined their strong antioxidative activities, without their identification. However, a few studies have investigated the polysaccharide derived from carob kibbles.
From this perspective, our research centers on this natural carbohydrate, to characterize the purified polysaccharide extracted from Ceratonia siliqua kibbles to evaluate their functional properties and antibacterial activity and to assess their antioxidant activities in vivo and in vitro, once incorporated into whipped cream.
2. Materials and Methods
2.1. Materials
Cultivated dry Ceratonia siliqua L. kibbles from Sfax, Tunisia were bought from a market from the same area. The material was ground with a Moulinex blender LM 241, and then sieved with a 0.2 mm mesh size. The obtained powder was stored at 4 °C.
2.2. Extraction of Carob Kibble Water-Soluble Polysaccharide (KWSP)
The extraction of carob kibble water-soluble polysaccharide (KWSP) was performed as described in details in [15]. Briefly, carob kibble powder was pre-extracted with 95% ethanol at room temperature, to remove pigments. Subsequently, the dry residue was extracted twice with 20 volumes of deionized water at 90 °C while stirring for 4 h. The extract was combined, filtered, and evaporated under vacuum. The concentrated liquid was precipitated with 95% (v/v) ethanol at 4 °C for 24 h and then centrifuged (4500× g) for 15 min using a refrigerated centrifuge (380R Hettich Zentrifugen, ROTINA, Kirchlengern, Germany). Finally, the precipitate was dried at 50 °C for 24 h to obtain the desired KWSP.
2.3. Physico-Chemical Analysis of KWSP
pH (1% solution at 25 °C) was evaluated using a digital pH meter (Systronics Instruments, New Delhi, India) with an electrode completely immersed into the solution. KWSP color was obtained using a Color Flex spectrocolorimeter (Hunter Associates Laboratory Inc; Reston, VA, USA). This was indicated in terms of L* (whiteness or brightness), a* (redness/greenness), and b* (yellowness/blueness) coordinates. The moisture and ash contents were specified according to the Association of Official Analytical Chemists (AOAC) standard methods [16]. The crude protein content was evaluated after multiplying the total nitrogen content by a factor 6.25. Crude fat was estimated gravimetrically, after Soxhlet extraction of dried samples. Total carbohydrates were determined through the phenol-sulfuric acid method [17].
2.4. Water and Oil Holding Capacities of KWSP
The water holding capacity (WHC) and the oil holding capacity (OHC) were measured by the method reported by Ben Slima et al. [18]. For WHC, KWSP (0.5 g) was mixed with 50 mL of distilled water. The mixture was stirred for 5 s every 15 min for 1 h at room temperature and then centrifuged at 8000 rpm for 20 min. The superior phase was eliminated and the tube was inclined to a 45° angle on a filter paper. WHC of KWSP was calculated according to Equation (1).
For OHC, the samples (0.5 g) and 10 mL of soybean oil were mixed and shaken for 1 h. Then, they were centrifuged at 8000 rpm for 20 min. The supernatant was removed and the centrifuge tube was left for 30 min on Whatman N° 1 filter paper, after inclining to a 45° angle. Their capacities were determined by the weight of the tube contents after elimination, divided by the weight of KWSP and expressed as the weight % of KWSP.
2.5. Emulsifying Properties of KWSP
Emulsifying capacity and emulsion stability were determined as described by Freitas et al. [19]. An amount of 4 mL of KWSP aqueous solution was added to 6 mL of soybean oil (a ratio of 3:2, v/v) and agitated in a vortex for 2 min at room temperature. Emulsification indices of E1, E24, and E168 were calculated after 1, 24, and 168 h, respectively, according to Equation (2).
where he (mm) corresponds to the emulsion layer height and ht (mm) refers to the entire mixture height after t hour.
2.6. Spectroscopic Analysis of KWSP
2.6.1. Nuclear Magnetic Resonance (NMR) Analysis
The nuclear magnetic resonance (NMR) analysis of KWSP was carried out as follows: A quantity of 40 mg of KWSP sample was solubilized in 1mL of D2O. All NMR experiments, including 1H NMR, 13C NMR, and the hetero-nuclear single quantum coherence (HSQC), were recorded using a spectrometer at 600 MHz (BRUKER 600M, Bruker BioSpin, Rheinstetten, Germany).
2.6.2. Fourier Transform Infrared Spectroscopy (FT-IR) Analysis of KWSP
The Fourier transform infrared spectroscopy (FT-IR) analysis of the KWSP was conducted on a Nicolet FT-IR spectrometer equipped with a horizontal attenuated total reflection (ATR) accessory. Internal crystal reflection was performed with zinc selenide and had a 45° angle of incidence to the IR beam. The spectrum was acquired at a 4 cm−1 resolution and recorded between 4000–500 cm−1 (mid-IR region). The data were examined using the OPUS 3.0 data collection software program (Bruker, Ettlingen, Germany).
2.6.3. HPLC and TLC Analysis of KWSP
The monosaccharide composition was analyzed with high performance liquid chromatography (HPLC). This analysis was performed using an Aminex HPX-87H column with a mobile phase of 0.001 N H2SO4, a flow rate of 0.4 mL/min, and a column temperature of 60 °C. The monosaccharide composition experiments were divided into two independent experiments. For the thin layer chromatography (TLC) analysis, sugars were released in a Chloroform/acetic acid/water (6:7:1) (v/v) mobile phase using TLC plates (Merck, Darmstadt, Germany). Glucose, fructose, sucrose, mannose, arabinose, and galactose were used as standards.
2.6.4. Average Molecular Weight (Mw) of KWSP
The average molecular weight was determined using high-pressure gel filtration chromatography, as previously reported by Ben Slima et al. [18].
2.6.5. Differential Scanning Calorimetry (DSC) Measurement
Thermal analysis of KWSP was carried out with a differential scanning calorimeter (Mettler Toledo 181 Star) from −2.77 to 250 °C at a rate of 5 °C/min. Differential scanning calorimetry (DSC) was performed to plot the transition phase.
2.7. Antioxidant Activities
The 1,1-diphenyl-2-picrylhydrazyl (DPPH) and the preformed radical monocation of 2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid): ABTS⋅+ assays are widely used as a spectrophotometric technique for the assessment of the antioxidant capacity of natural products. They display the radical scavenging ability of antioxidants, even when present in a KWSP extract.
2.7.1. DPPH Radical-Scavenging Assay
The DPPH radical-scavenging activity of KWSP was determined according to Bersuder et al. [20]. An amount of 500 μL of the carbohydrate at different concentrations (0.1 to 10 mg/mL) was added to 375 μL of 99.5% ethanol and 125 μL of DPPH (0.02% in ethanol). The reaction was measured after incubation for 1 h at 517 nm using a spectrophotometer. The butylated hydroxytoluene (BHT) was defined as a positive control. The DPPH value was estimated as shown in Equation (3).
2.7.2. ABTS⋅+ Radical Scavenging Activity
The ABTS⋅+ radical scavenging activity of KWSP was estimated according to the method of Braca et al. [21] with minor modifications at different concentrations (0.1 to 5 mg/mL). The absorbance of the mixture was specified spectrophotometrically at 734 nm. Trolox was the standard control. The ABTS⋅+ radical scavenging activity was identified with Equation (4).
With
Acontrol: the absorbance of the control reaction;
Asample: the absorbance of KWSP
2.7.3. Total Antioxidant Activity
The total antioxidant capacity of KWSP was determined by the phosphate molybdate method using BHT as the standard compound [22]. The basis of this test lies in the reduction of Mo (VI) to Mo (V) using the extract and green phosphate/Mo (V) complex at acidic pH formation. The reaction was measured at 695 nm. BHT was the positive control. The antioxidant activity was demonstrated as ascorbic acid equivalents.
2.8. Antibacterial Activity of KWSP
The target bacterial strains were obtained from international culture collections (ATCC). They included two Gram-positive bacteria: Staphylococcus aureus ATCC 6538 and Listeria monocyogenes ATCC 19115; and two Gram-negative bacteria: Salmonella enterica 14028 and Pseudomonas aeruginosa ATCC 4912.
Antibacterial activity was determined by means of agar-well diffusion assay using Luria–Bertani medium. The inhibitory effects of KWSP were assessed by monitoring the ability of the indicator bacteria to grow in the presence of the polysaccharide, according to the Skytta and Mattila Sandholm method [23]. Distilled water was employed as a negative control, while the antibiotic gentamicin (20 µg/well) was used as a positive control.
2.9. Incorporation of KWSP in Whipped Cream
KWSP was added to heavy cream (0.1%) that was subsequently whipped with a domestic blender for 10 min. A remaining fraction of the cream was whipped in the absence of the polysaccharide for comparability purposes. Afterwards, 5 g of each sample was added to 25 mL of ethanol and then incubated for 2 h at 37 °C in a rotatory shaker, before they were filtered, in order to remove undesirable small particles. The filtrate was stored at 4 °C for the various experiments.
2.9.1. Antioxidant Activities of Whipped Cream
The antioxidant activity was evaluated following four different methods, including DPPH and ABTS⋅+ activity procedures, which are reported in Section 2.7. The two other supplementary methods for the antioxidant capacity of KWSP were provided by testing the reducing power and ferrous chelating.
- Reducing power assay: The potential of peptidic fractions of ZBPH-Z to reduce iron (III) was defined, as clarified by the method of Yildirim et al. [9]. An amount of 1 mL aliquot of peptidic fractions at concentrations from 1 to 5 mg/mL was added to 2.5 mL 0.2 M Na phosphate buffer (Sigma Chemical) (pH 6.6) and 2.5 mL of 1% (w/v) potassium ferricyanide solution (Sigma Chemical). These mixtures were incubated for 30 min at 50 °C. Subsequently, they were mixed with 2.5 mL 10% (w/v) of trichloro acetic acid (TCA), and the reaction mixture was then centrifuged for 10 min at 6000× g. Finally, 2.5 mL of the supernatant solution from each sample mixture was added to 2.5 mL distilled water and 0.5 mL 0.1% (w/v) ferric chloride. After 10 min, the absorbance was determined at 700 nm. The higher absorbance of the reaction mixture suggested a higher reducing power. Control was carried out following the same method, but distilled water was used instead of the sample. The butylated hydroxyanisole (BHA) was used as a positive control;
- The ferrous chelating activity of KWSP: 15 mg/mL was determined following the method of Dinis et al. [9]. An amount of 0.5 mL of sample and 1.6 mL of distilled water were added to 0.05 mL of FeCl2 (2 mM), following the addition of 0.1 mL of ferrozine (5 mM) after15 min. After 10 min, the Fe2+-ferrozine complex absorbance was defined at 562 nm. The Ethylenediaminetetraacetic acid (EDTA) was used as a reference. The chelating antioxidant activity for Fe2+ was measured in terms of Equation (5):
2.9.2. Effect of KWSP on K232 and K270 Values
Whipped cream lipids were extracted following the Folch method proposed by Jordi Folch, with some modifications [24]. The extraction solvent was a mixture of chloroform and methanol at a ratio of 2:1 (v/v), where the final volume must be 20 times higher than the 1 g sample. Then, water was added to separate both phases. The lower phase was used for analyzing fats.
In order to infer the influence of KWSP on cream fats, we calculated the coefficients of specific extinction at 232 nm (K232) and 270 nm (K270) with a spectrophotometer (model 35, Beckman Instruments, Inc., Fullerton, CA, USA), adopting Ben Hamouda et al. method [25]. A quantity of 0.05 g of fat was added to 25 mL of cyclohexane, so as to measure K232, and 0.25 g, to determine K270.
2.10. Statistical Analysis
In this study, three analyses of each sample were made, and each experiment was carried out in triplicate (n = 3). The arithmetic mean value and standard deviation (SD) were calculated from the data obtained. For comparative studies, a two-sample t-test for difference of means using Minitab 14 software was used. All tests were performed at a 0.05 level of significance.
3. Results and Discussion
3.1. Physicochemical Characterization of KWSP
The application of polysaccharides basically relies on their physicochemical properties. The obtained results from the physicochemical analysis, as well as biochemical composition of KWSP, are summarized in Table 1.

Table 1.
Physicochemical and functional properties of KWSP (g water or oil/g wet sample).
Multiple factors can influence the polysaccharide yield during the extraction procedure, including duration, temperature, and water/raw material ratio. As a result, the extraction yield of KWSP was equal to 8.30 ± 0.86%, with a pH value of 6.59 at 25 °C. In fact, pH is one of the prominent factors that can exert an effect by modifying the associative molecular structure of polysaccharide in an aqueous environment [26]. In particular, color is an attractive characteristic that can define the susceptibility of KWSP to an appropriate implementation. L* corresponds to the lightness value, from 0 (black) to 100 (white), which was equal to (L* = 45.40), while a* and b* values amounted to 7.32 and 11.71, respectively, which can be attributed to the dark color of this polysaccharide.
In addition, the results obtained from the phenol–sulfuric acid test revealed that the carbohydrate content of the purified polysaccharide was about 86.68%. Previous studies reported similar values from potato peels (88%) [27] and Sorghum bicolor (L.) seeds (78.84%) [18]. The percentages of moisture and ash were 12% and 3.66%, respectively. The analysis suggested that the KWSP was free of fat, and a relatively important fraction of protein (9.13%) was recorded. The low value of water activity (Wa = 0.43) of KWSP (Table 1) plays an intrinsic role in the prevention of bacterial growth [28].
3.2. Functional Properties of KWSP
3.2.1. Water-Holding and Oil-Binding Capacities of KWSP
The data obtained in this study demonstrated that the WHC of KWSP polysaccharide was around 3.14 g/g ± 0.5. This finding agrees well with the one found in the case of the WHC values of Bicolor sorghum [18] and was considerably higher than the ones obtained after polysaccharide extraction from Carob seeds (1.4 g/g) [29], almond (1.95%) [9], and pistachio (1.46%) [10]. Nevertheless, the WHC value was noticeably lower than the one obtained from galactomannans of Gleditsia triacanthos gum (15.20 g/g) [30]. The difference in WHC values was mainly assigned to the chemical structure of the polysaccharide. Other factors may also interfere, such as the porosity, ionic strength, extraction conditions, pH, temperature, type of ions in solution, and vegetable families [31]. The OHC of KWSP was approximately 0.87 ± 0.02% (Table 1). This capacity was lower than that of the polysaccharides extracted from foenum-graecum seeds (22 g/g) [32], and galactomannan extracted from endosperm of Gleditsia japonica var. delavayi seeds (3.56 g/g) [33]. The OHC and WHC values of KWSP may be associated with the hydrophobic nature of the polysaccharide, which can provide data on its use as a low-calorie stabilizer [34].
3.2.2. Emulsification Capacity of KWSP
The results presented in Table 2 show that after 1 h, KWSP had a high EC percentage, (up to 94.73%) which decreased and reached 72% after 24 h. Basically, the emulsions stabilized with KWSP were stable up to 168 h, with an emulsification index of 71%. The results were higher than those of pumpkin pectin (62.50 to 52.50) and pomegranate peel pectin (68.63 to 59.14) [35]. Therefore, the emulsifying capacity of polysaccharide can be considered from two perspectives: (1) the ability to fix to the oil–water interface; (2) the formation of a protective thickness around emulsified oil droplets. The hydrophobic content groups, molecular conformation, and surface tension can affect the emulsifying capacity of polysaccharides [26].

Table 2.
Emulsion properties of KWSP (%).
3.3. Spectroscopic Analysis Results of KWSP
3.3.1. Nuclear Magnetic Resonance (NMR) Spectroscopy
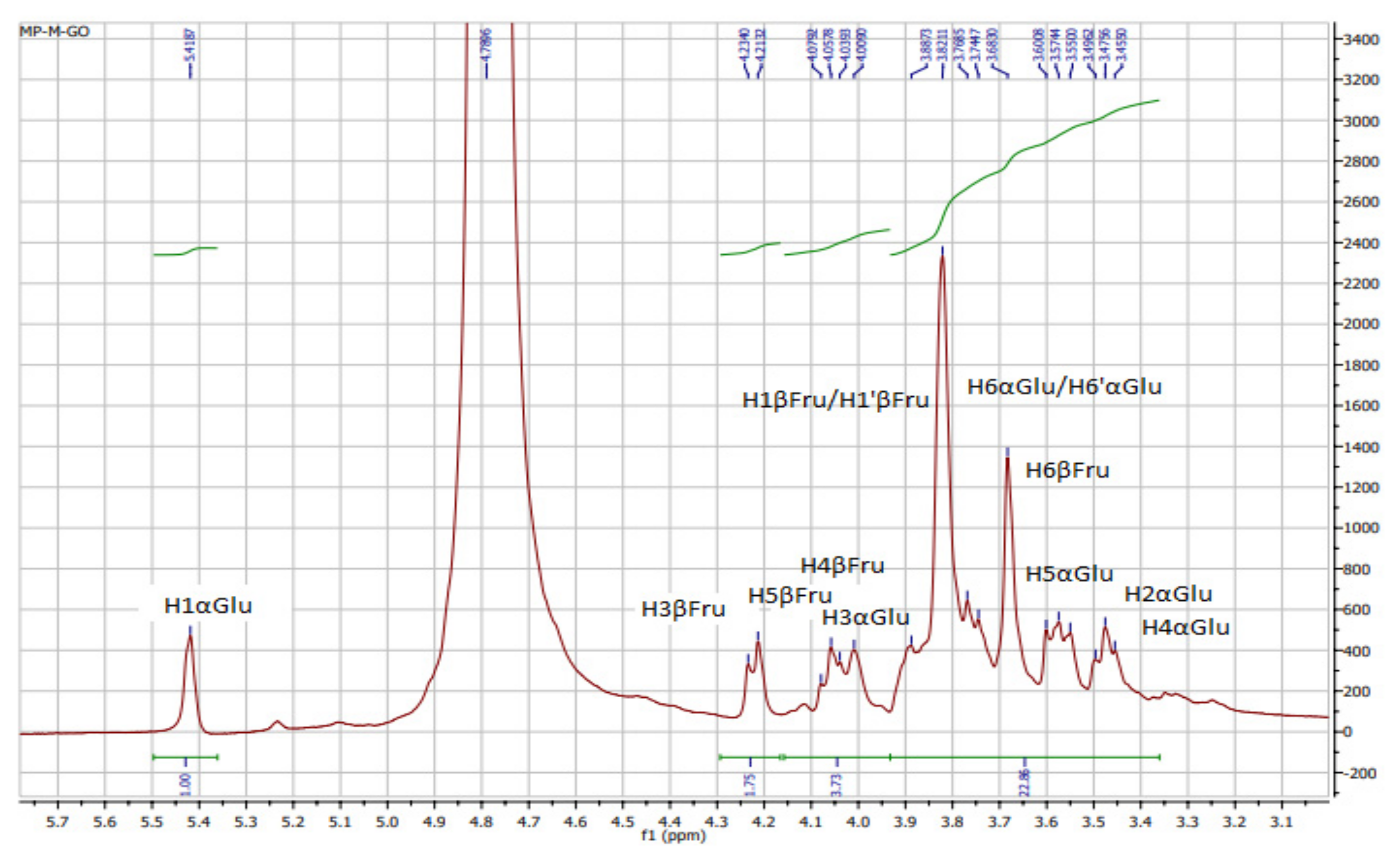
The 1H, 13C, and HSQC NMR of KWSP are plotted in Figure 1, Figure 2 and Figure 3, respectively. Characteristic absorption peaks of polysaccharide were found within the range of 3.4 and 4 ppm, including the anomeric proton (4.4–5.5 ppm) [36]. As can be seen from Figure 1, compared with the spectrum of OP%US by Medlej et al. [37], 3.82–3.88, 4.22, 4.13, 4.08, and 3.74 ppm were the signal areas of fructose protons related to H1, H3, H4, H5, and H6, respectively [37]. Consequently, the presence of anomeric proton α H1 was indicated by the NMR shift at 5.42 ppm, which belongs to glucose, while the signals at 3.47, 4, 3.45, 3.60, and 3.74–3.76 ppm were assigned to the other glucose proton rings from H2 to H6.
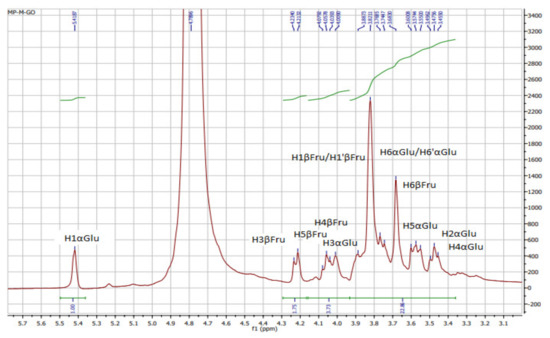
Figure 1.
1H NMR spectra of polysaccharides from Ceratonia siliqua kibbles.
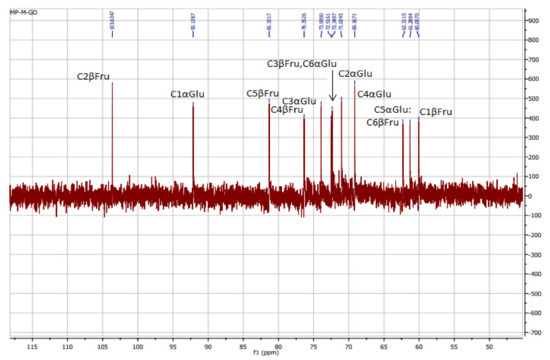
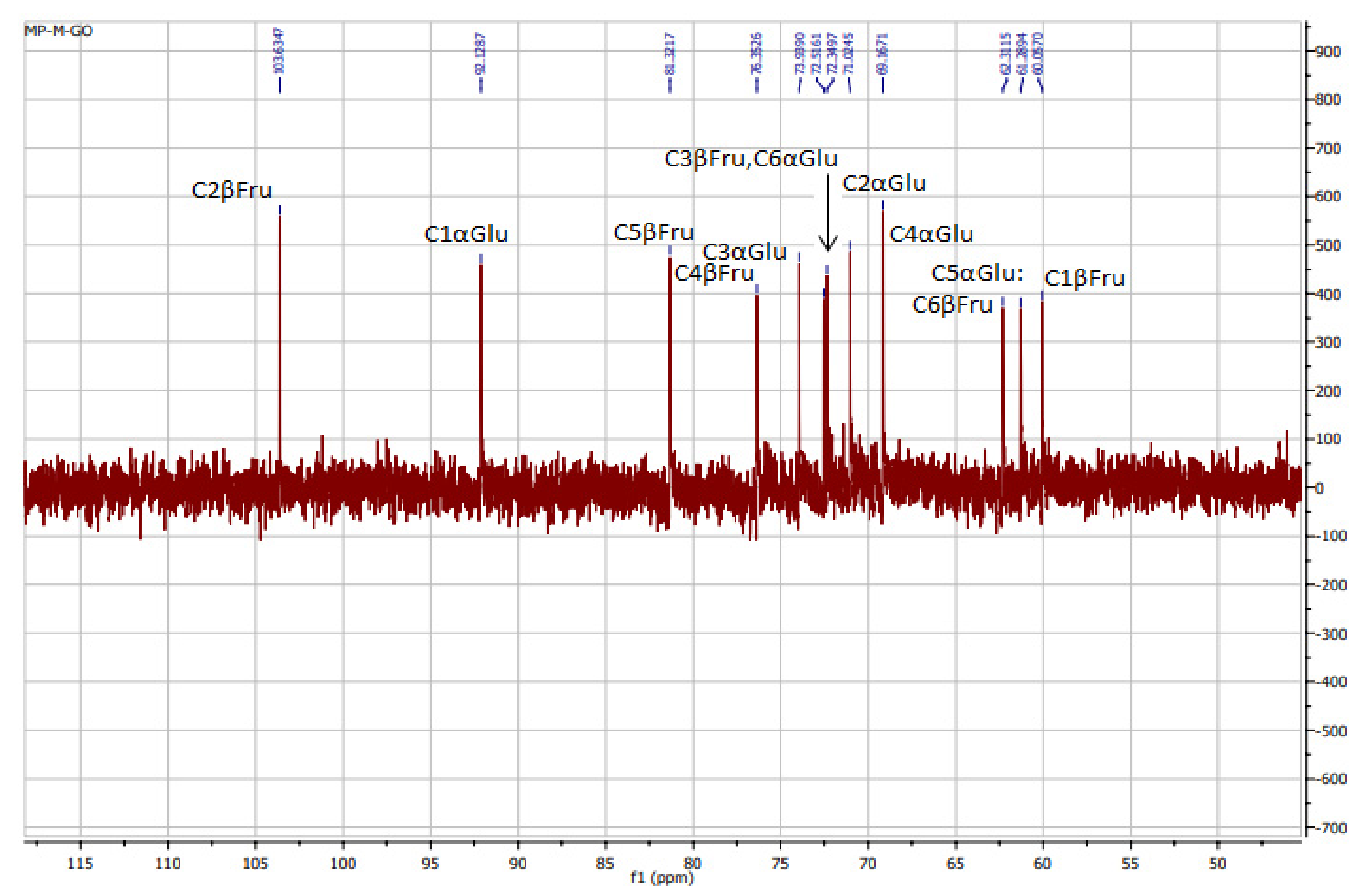
Figure 2.
13C NMR spectra of polysaccharides from Ceratonia siliqua kibbles.
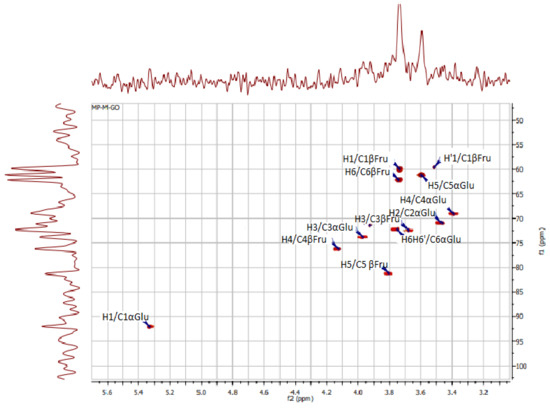
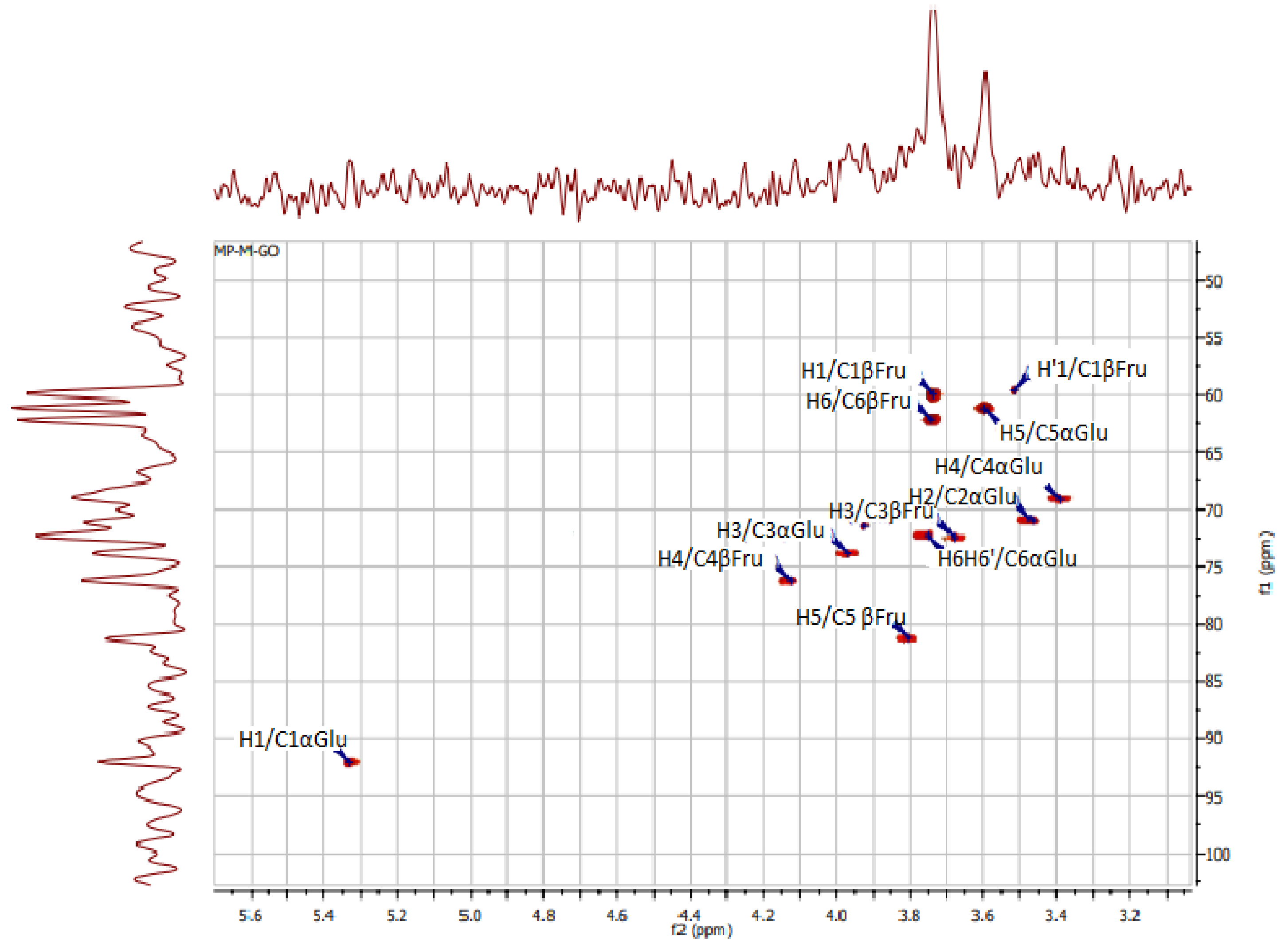
Figure 3.
HSQC spectra of polysaccharides from Ceratonia siliqua kibbles.
It is obvious from Figure 2 that 103.63 was the signal area of C2, and this was confirmed by the chemical shift of an anomeric carbon in β linkage (103–105 ppm) [36]. However, the signals at 60.05, 72.51, 76.24, 81.19, and 62.17 ppm were attributed to C1, C3, C4, C5, and C6 of fructose rings, respectively.
In addition, the signal at 92.13 ppm is suggestive of the α glycosidic configuration of glucose, which is in good accordance with what was found by Medlej et al. [37]. For a further structural clarification of KWSP, the HSQC spectrum is presented in Figure 3, where the correlations between 13C and 1H signals are illustrated. The structure of the produced polysaccharide proved to be composed of fructose and glucose, which is set forward in Table 3.

Table 3.
HSQC spectrum interpretation of the KWSP.
3.3.2. FT-IR Spectrometric Analysis of KWSP
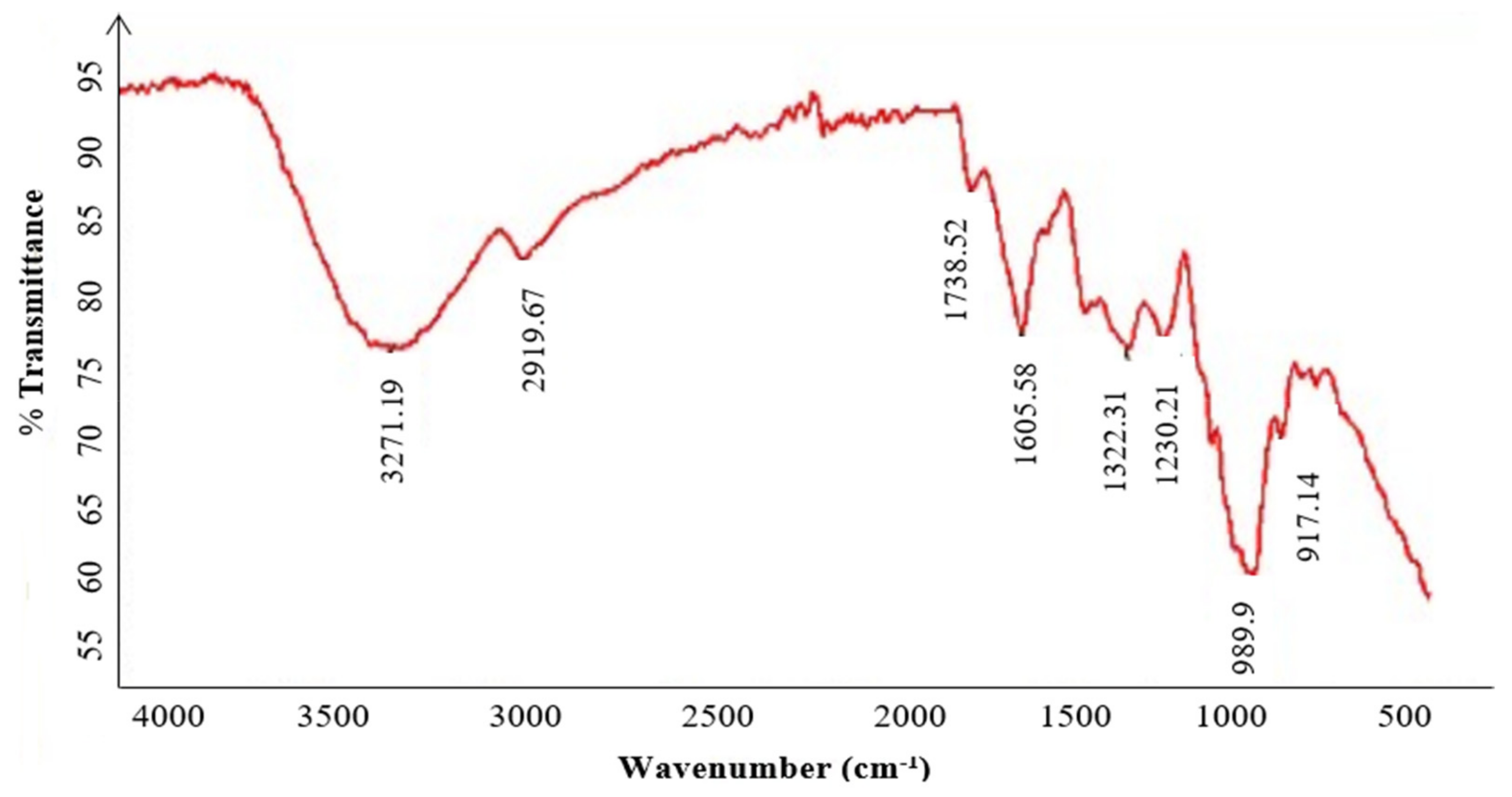
The FT-IR spectra of the purified polysaccharide (Figure 4) highlight the characteristics peaks of KWSP at 3271, 2919, 1738, 1605, 1322, 1230, 989, and 917cm−1. The peak at 3271 cm−1 indicates the stretching vibration of the OH groups due to inter- and intra-molecular hydrogen bands [38]. The peak at 2919 cm−1 corresponds to the stretching of the CH groups of free sugars [39]. Furthermore, the absorption observed at 1738 cm−1 determines the existence of the carboxylate CO bonds in the acylamino group, corroborating the presence of proteins [38]. In addition, the FT-IR spectrum suggests that the region between 1605 and 1500 cm−1 reflects bound water [40]. The most important peaks refer to those observed at 1322 cm−1 and 1230 cm−1 obtained from the bending vibration of the stretching vibration of the ester sulfate groups (S=O) of sulfate, as previously elaborated in the literature [41]. The absorption at 980 cm−1 can be assigned to the (C-O-C) group [38].
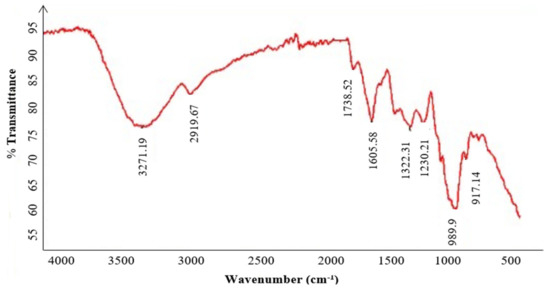
Figure 4.
FT-IR spectrometric analysis of KWSP.
3.3.3. HPLC Analysis and TLC Analysis of KWSP
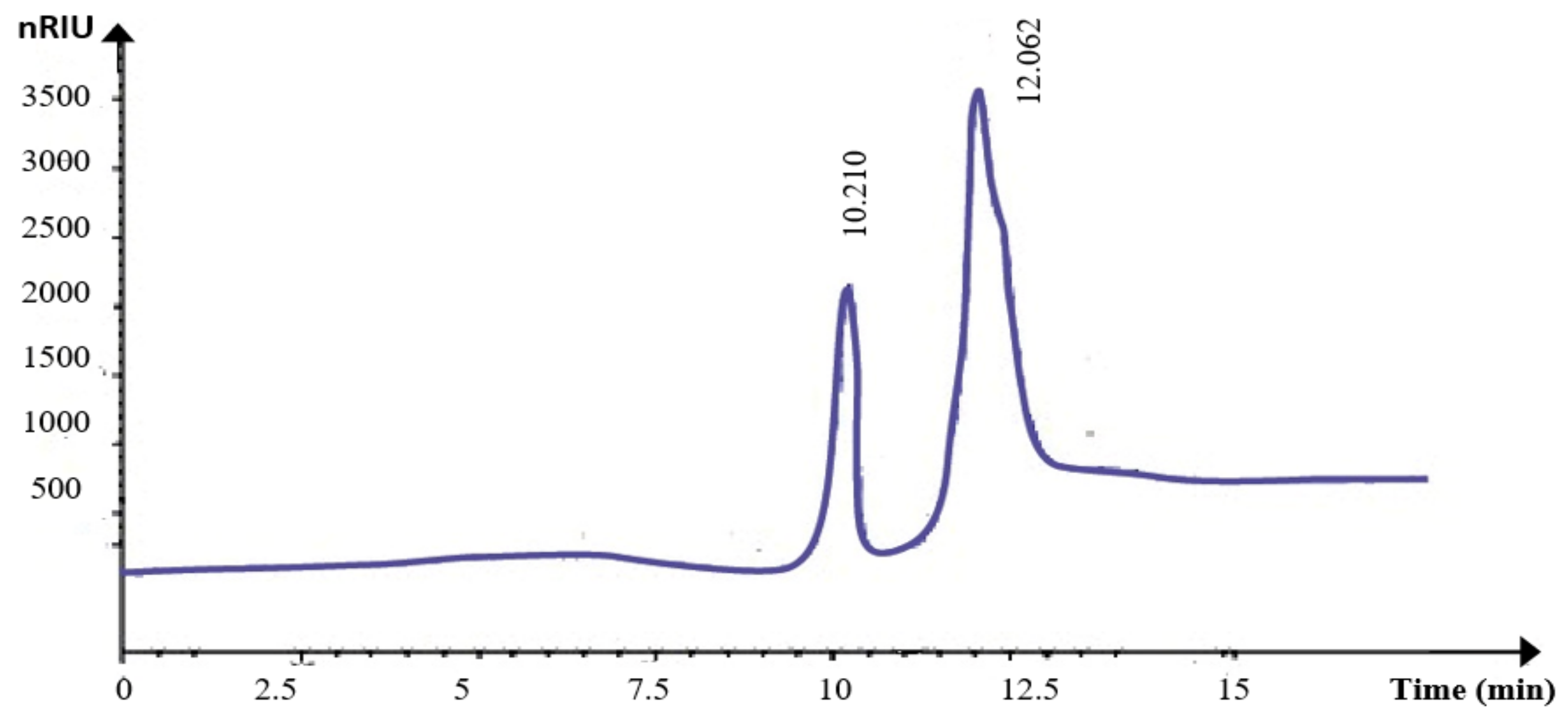
The monosaccharide composition of KWSP was explored through HPLC analysis. The chromatograms revealed two peaks at 10.21 min and 12.05 min for hydrolyzed KWSP (Figure 5), corresponding to those of the glucose and fructose, respectively, used as standards. The hydrolysis of KWSP contributes to the appearance of two plugs with retention factors of 0.7 and 0.75.
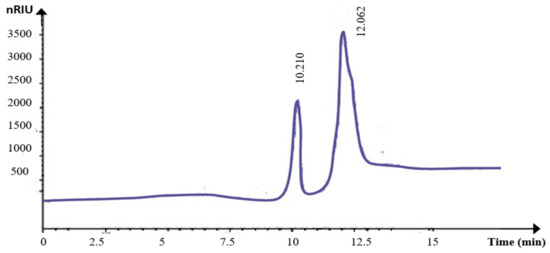
Figure 5.
HPLC analysis of KWSP.
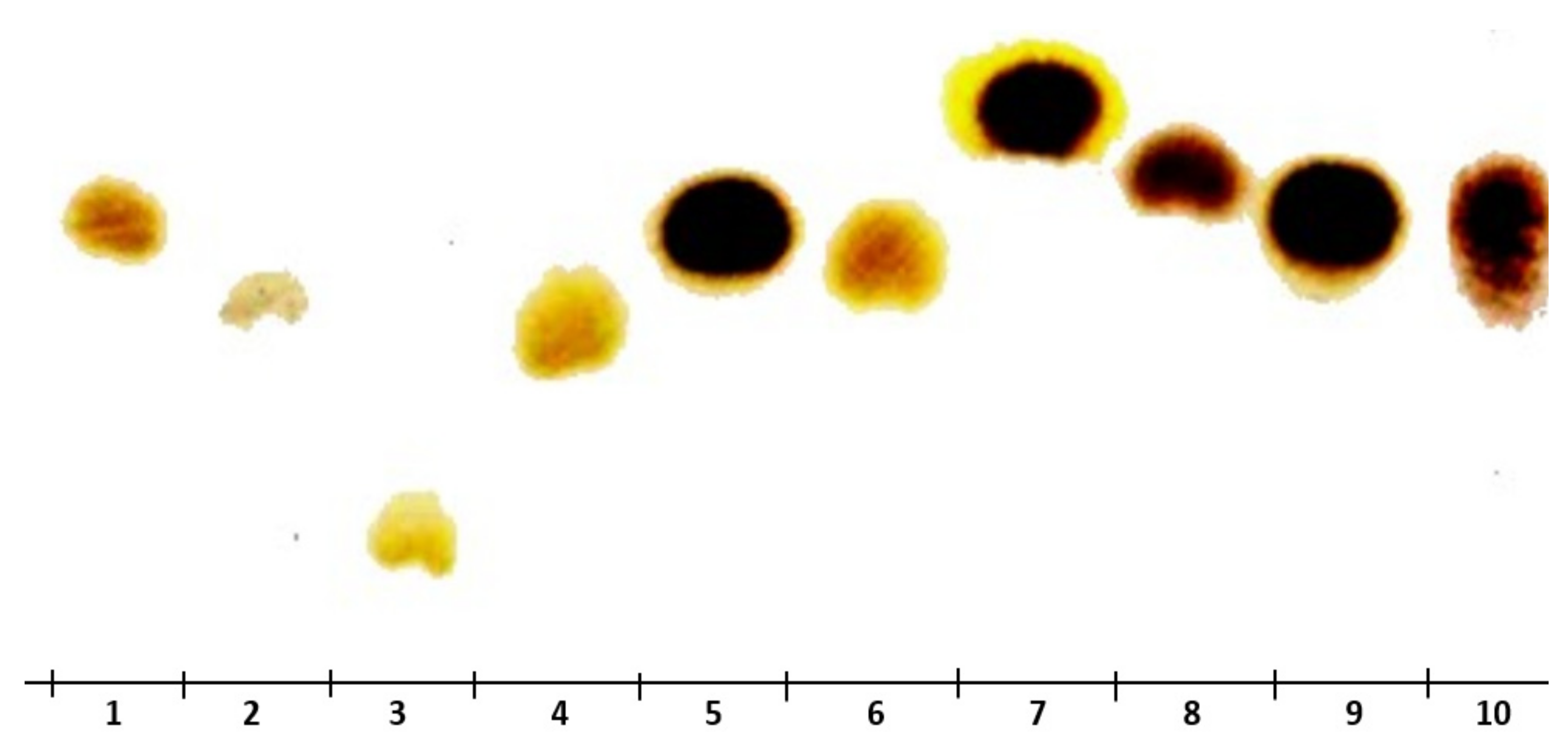
The results outlined in Figure 6 suggest that these factors were the same for glucose and fructose (used as standards). These findings corroborate that KWSP is a hetero-polysaccharide composed of a chain of glucose and fructose molecules.
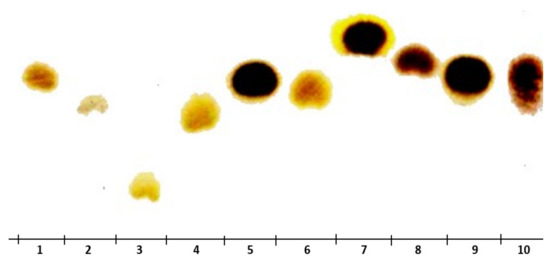
Figure 6.
TLC analysis of KWSP, 1 arabinose, 2 glucose, 3 lactose, 4 galactose, 5 mannose, 6 fructose, 7 rhamnose, 8 xylose, 9 glutamic acid, 10 KWSP.
3.3.4. Average Molecular Weight (Mw) of KWSP
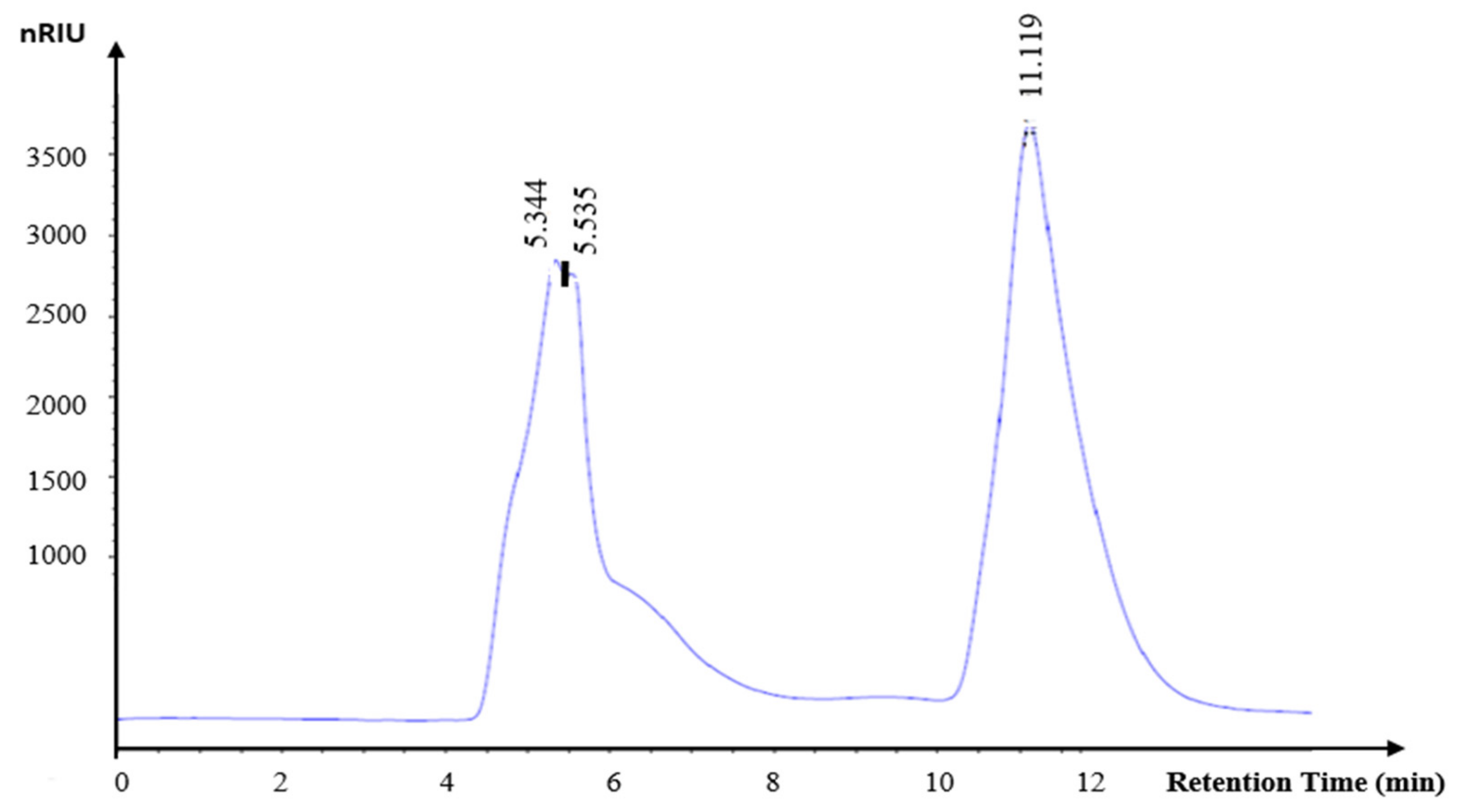
As demonstrated in Figure 7, KWSP exhibits a molecular weight of 65 kDa. The origin of polysaccharide fruits plays an important role in the average molecular weight. Additionally, the harvesting and manufacturing practices, among other factors, may influence their molecular weight.
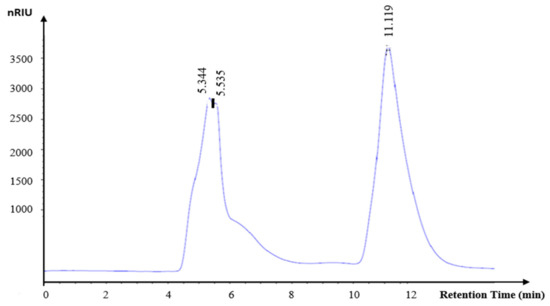
Figure 7.
Gel filtration chromatographs of KWSP.
3.3.5. DSC Measurement
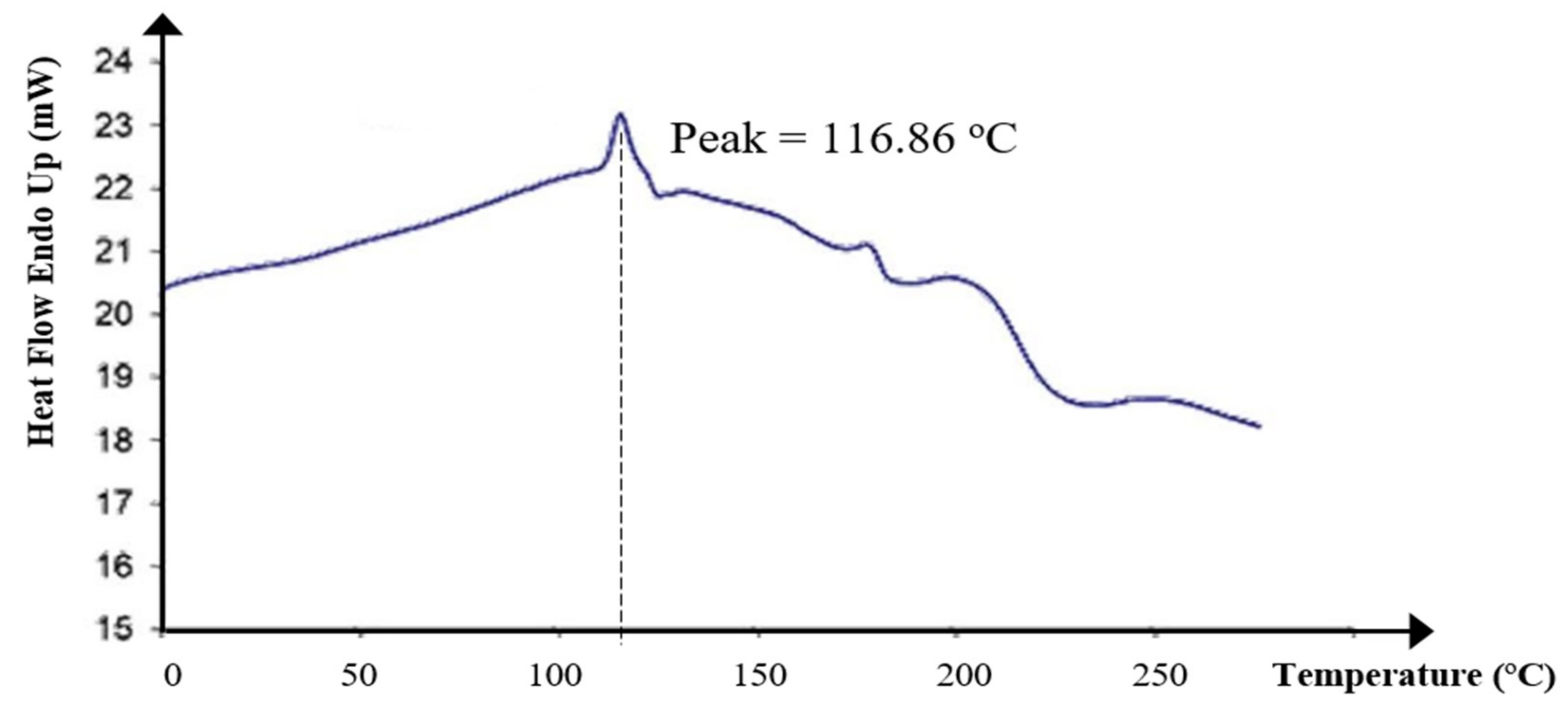
The glass transition temperature (Tg) of KWSP was shown by a peak at 116.86 °C, as portrayed in Figure 8. The heat of glass transition was 22.96 J/g. Basically, the heat of glass transition and exothermic peak corresponds to the capacity of the biopolymer to lose water. Furthermore, the polymer nature, as well as the temperature, may affect the Tg value.
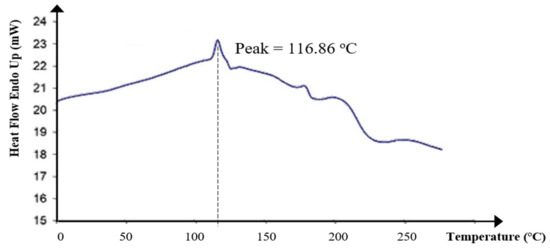
Figure 8.
Thermal behavior from differential scanning calorimetry of KWSP.
3.4. Antioxidant Activities Results
3.4.1. DPPH Radical-Scavenging Experiment
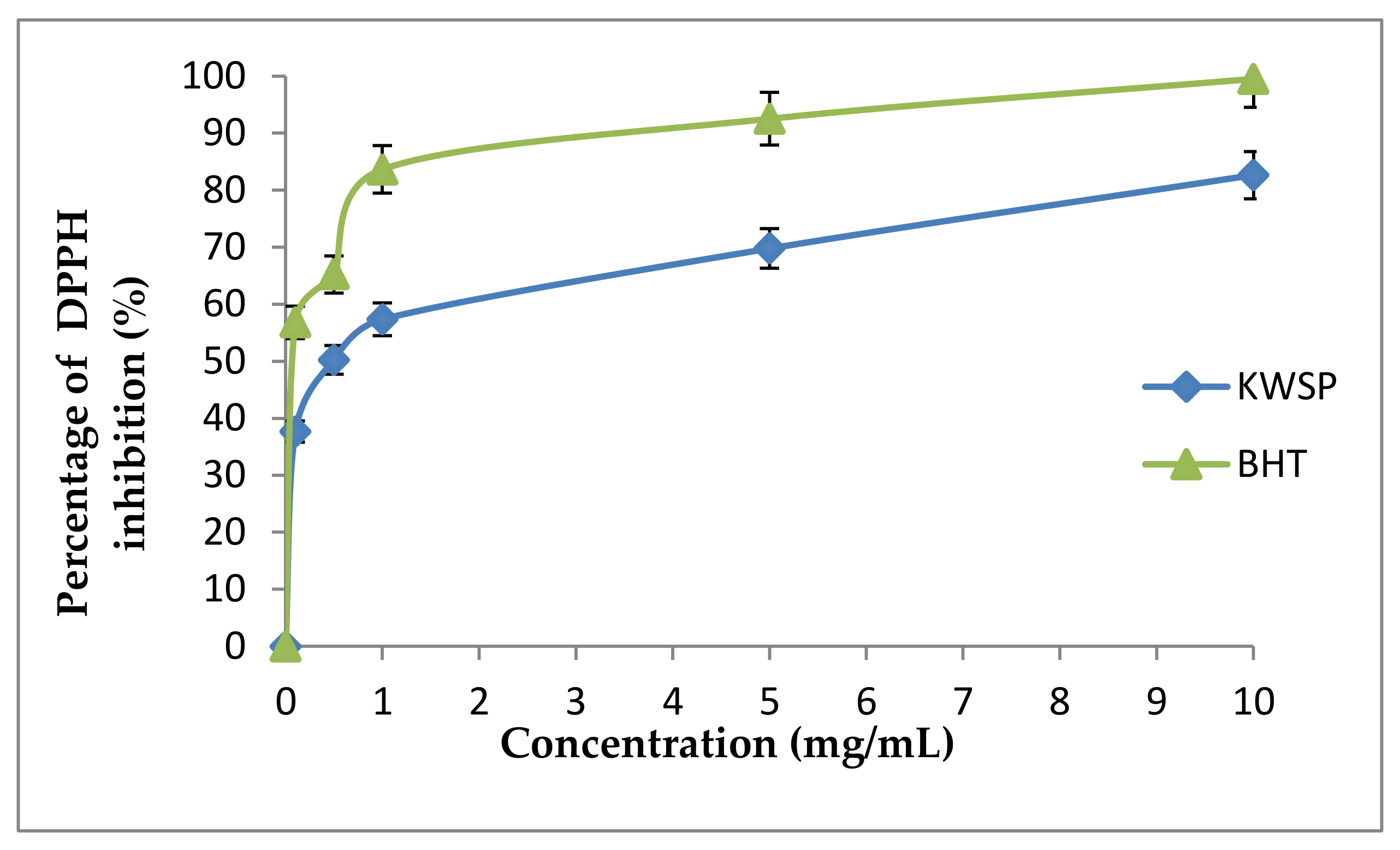
Notably, the DPPH radical assay represents one of the most rapid, sensitive, simple, and widely used assays to examine the antioxidant potential of polysaccharides, through estimating their radical scavenging power [42]. Within this framework, the scavenging ability of KWSP from Ceratonia siliqua L. on DPPH radical was investigated by comparison with BHT, which is a synthetic antioxidant. The DPPH radical-scavenging activity of KWSP at varying concentrations (0.1–10 mg/mL) is illustrated in Figure 9, reflecting that the DPPH radical scavenging activity of KWSP increased with the increase in the concentration of polysaccharide, reaching a maximum 82.63% at the concentration of 10 mg/mL. KWSP possessed an antioxidant activity on DPPH radical, with an IC50 of 0.4 mg/mL when IC50 of BHT was 0.08 mg/mL. With respect to state of the art works, it has been shown that the molecules are potentially antioxidant for an IC50 < 500 μg/mL; with the best candidate showing IC50 < 100 μg/mL [43]. In previous research performed by Bouala et al. [43], the IC50 of Astragalus armatus Lam (Fabaceae) was 0.33 mg/mL, which is nearly the same as our finding [43]. Hence, the yielded results indicate that KWSP may be used as a natural source of antioxidant agent.
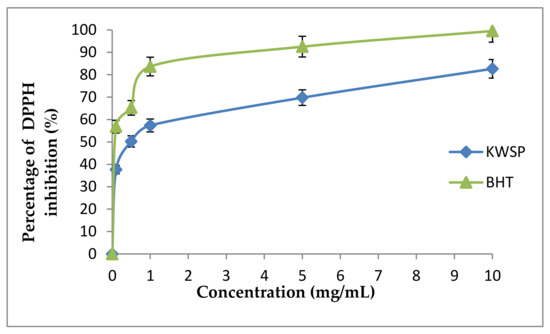
Figure 9.
DPPH radical scavenging activity according to the concentration of polysaccharide. Each point represents the mean of three independent experiments (SD < 10% of the mean).
3.4.2. ABTS⋅+ Radical Scavenging Activity
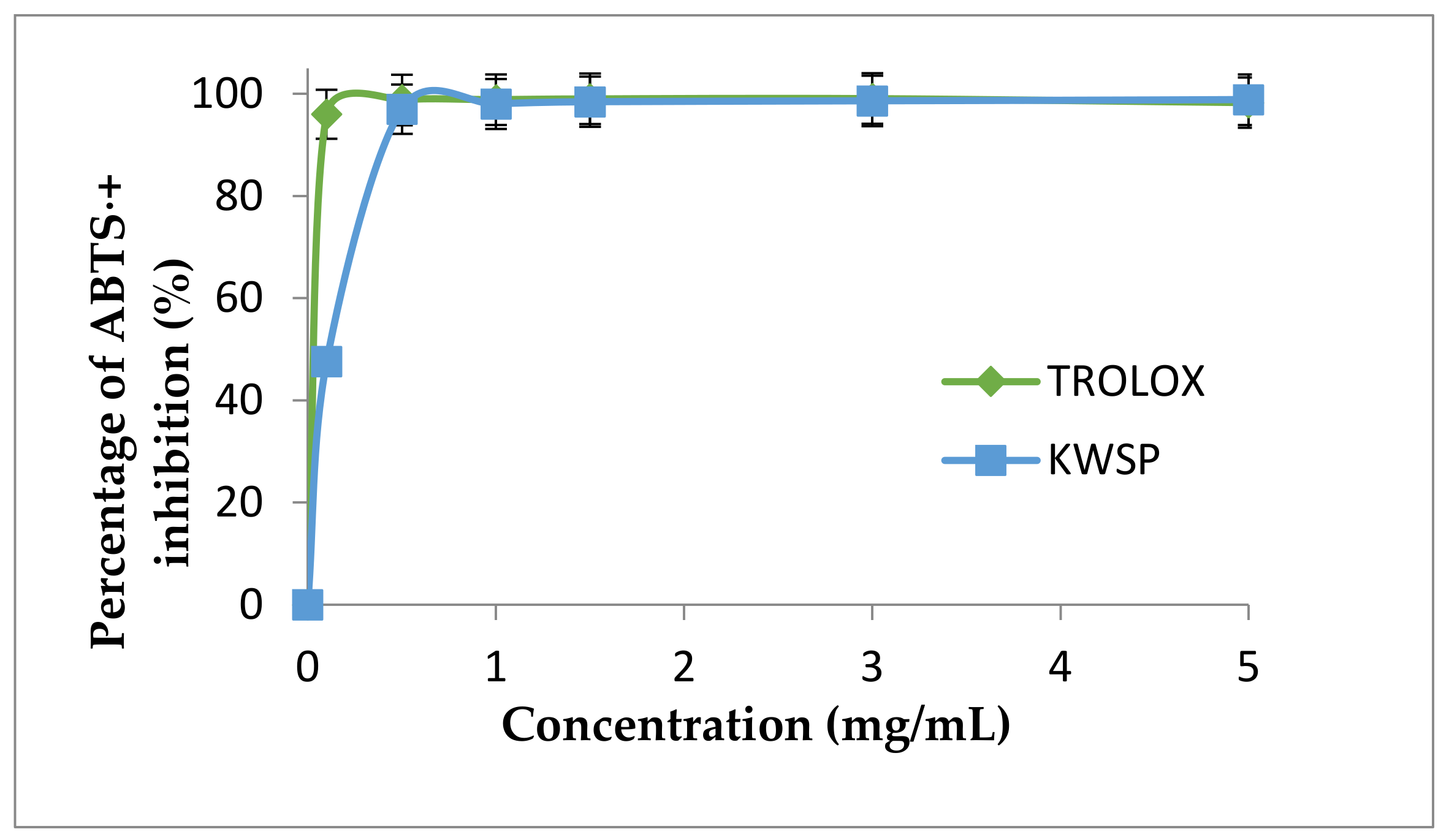
As mentioned in materials and methods section, ABTS⋅+ is another test utilized to estimate the antioxidant capacities of foods and natural products [44]. The ABTS⋅+ scavenging potential of KWSP is presented in Figure 10. The scavenging activities increased with the increase in concentration, from 0.1 to 5 mg/mL. Trolox was used as a positive control in this experiment, displaying in turn a dose-dependent increase of ABTS⋅+ scavenging activity, reaching the maximum level at 5 mg/mL 99.75%, and with the IC50 of BHT amounting to 0.01 mg/mL. The scavenging activity of KWSP is expressed in terms of the percentage of the maximum activity of the positive control. At the highest concentration, the investigated KWSP exhibited 92.84% at 5 mg/mL and the corresponding IC50 value was 0.15 mg/mL. Our results proved to be higher than those obtained by Mzoughi et al. [45], who asserted that the IC50 value of polysaccharide extracted from quinoa was 1.1 mg/mL [45].
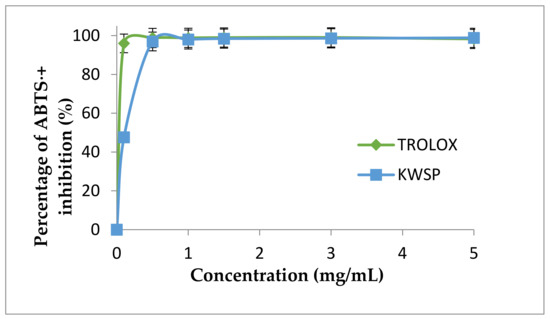
Figure 10.
ABTS⋅+ radical scavenging activity according to the concentration of polysaccharide. Each point represents the mean of three independent experiments (SD < 10% of the mean).
3.4.3. Total Antioxidant Activity
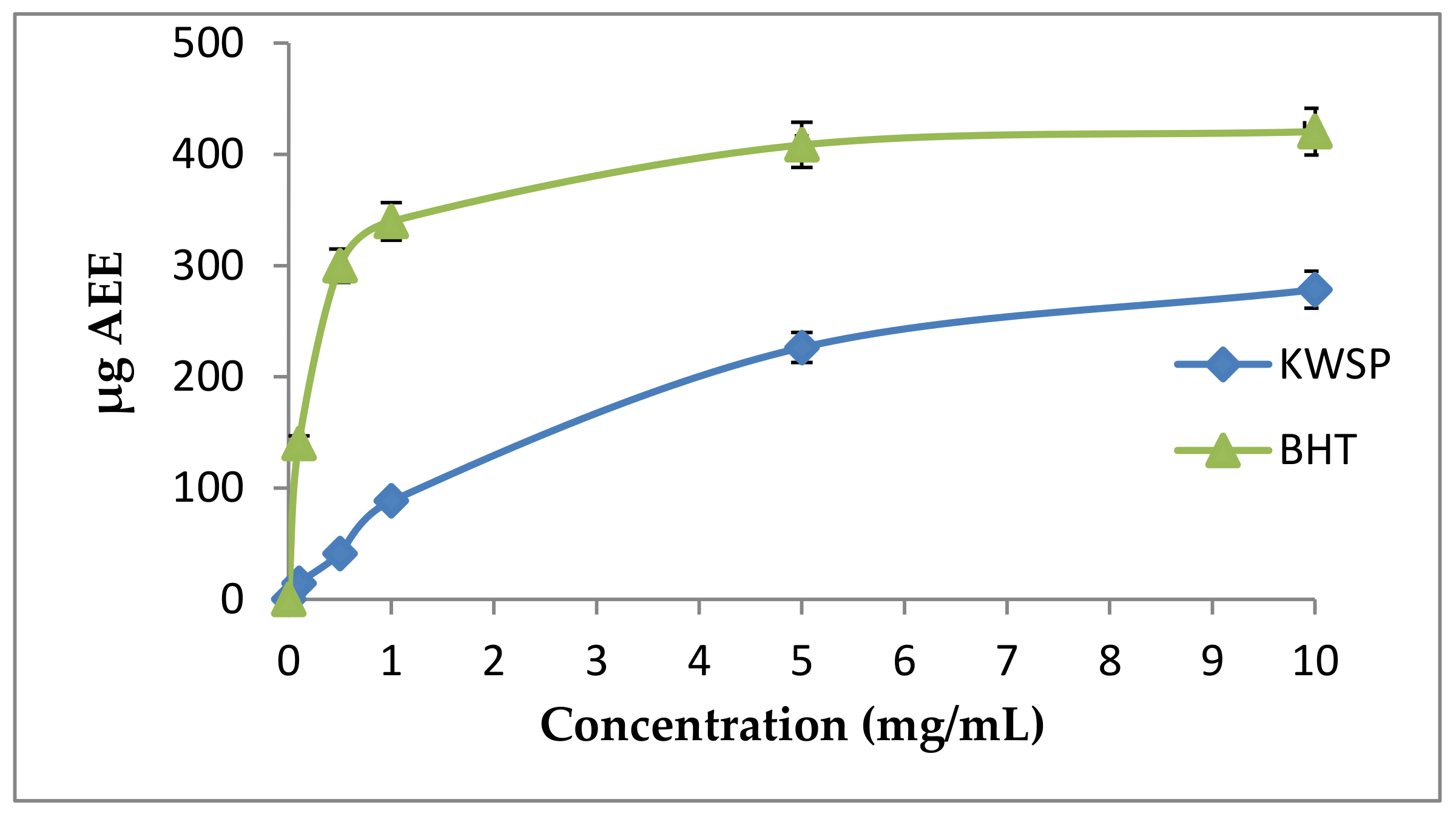
The phosphomolybdate method is also a quantitative test. The total antioxidant capacity is expressed as ascorbic acid equivalents (AAE). The total antioxidant capacity of the carbohydrate was determined to be 10 mg of KWSP, equivalent to 20 μg ascorbic acid. The obtained results were lower than BHT (Figure 11). Furthermore, our findings were similar to that of a previous work elaborated by Ben Slima et al. [18].
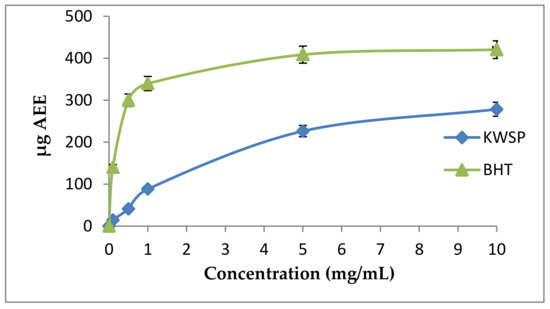
Figure 11.
Total antioxidant activity according to the concentration of polysaccharide. Each point represents the mean of three independent experiments (SD < 10% of the mean).
3.5. Antibacterial Activity Results
The antibacterial activity of KWSP against S. enterica, L. monocytogenes, and S. aureus was demonstrated at a concentration of 20 mg/mL, in terms of inhibition zone diameter (mm). The negative control, distilled water, did not show any inhibition zone against all the test strains. The inhibition zone for Gentamicin, which was used as a positive control for bacteria, ranged from 15 to 21 mm. KWSP exhibited an antibacterial activity against S. enterica (10.00 ± 0.46 mm), and its inhibition zones against L. monocytogenes and S. aureus were 8.90 ± 0.63 mm and 8.20 ± 0.25 mm, respectively (Table 4). Moreover, as mentioned in Table 4, an inhibition zone for P. aeruginosa was not obtained, even though the test was applied three times. Monomer of polysaccharide, carbohydrate percentage, and conformational structure can affect antibacterial capacity [46]. Polysaccharide extracted from plants presented a better antibacterial effect, which showed a different antibacterial effect than previous studies [23,47]. The difference in terms of antibacterial activity is probably related to the sources and concentration of polysaccharide. The antibacterial activity can be assigned to the change of cell wall and membrane permeability, resulting in structural damage and the release of cell components [47].

Table 4.
Antibacterial activities of KWSP.
3.6. Incorporation of KWSP in Whipped Cream Results
3.6.1. Antioxidant Properties of KWSP in Whipped Cream
To study the effect of the incorporation of KWSP in whipped cream, three samples for control and three samples with the incorporation of KWSP were evaluated according to the following four antioxidant proprieties: DPPH, ABTS+, reducing power, and chelating power. All results obtained are shown in Table 5.

Table 5.
Comparative statistical results between control sample and sample with KWSP.
To validate the superiority of the antioxidant properties of KWSP in whipped cream, a two-sample t-test was used to determine whether the mean with KWSP was higher than the mean control (one-sided statistical test).
For each antioxidant propriety, the null hypothesis stated that the mean result of whipped cream would be higher than the mean of control results. As all p-values in Table 5 are less than the significance level of 0.05, each null hypothesis was rejected and we concluded the superiority of the incorporation of KWSP for all the studied antioxidant proprieties.
Polysaccharides involve numerous polar groups, such as hydroxyl and carboxyl groups, which can build up hydrogen bonding with cream molecules to ensure the nutritional quality [48]. So far, numerous polysaccharides have been successfully integrated to enhance the quality of food. The usage levels are very different, depending on the used polysaccharide types, usually ranging from 0.20 to 8 g/100 g (polysaccharide/food) [26].
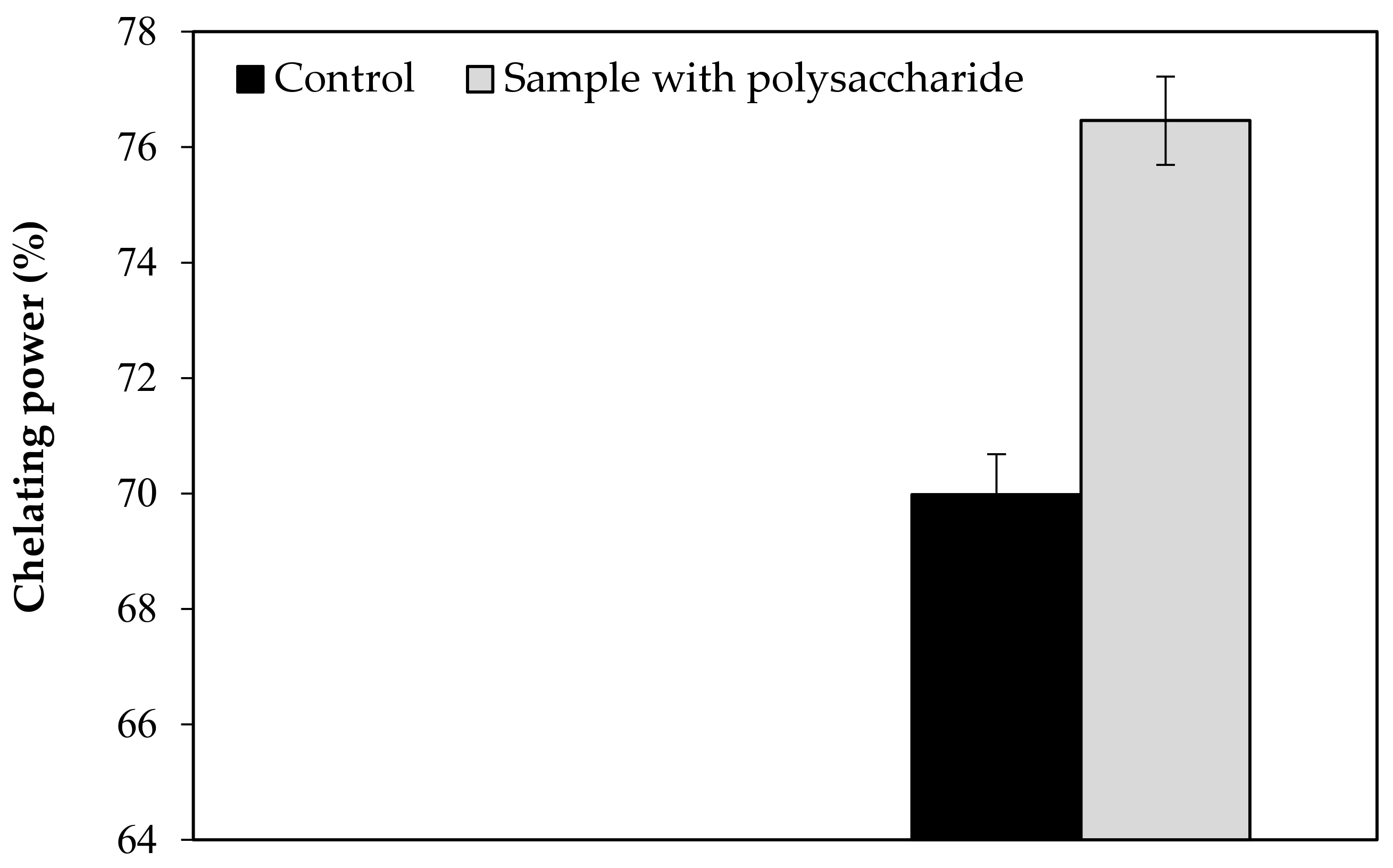
Our findings revealed that the KWSP extracted from Ceratonia siliqua L. can effectively indicate a good antioxidant activity in vitro. KWSP polysaccharide can be safely added as a natural antioxidant compound to whipped cream, which was exemplified by the antioxidant report, including DPPH, ABTS+, reducing power, and chelating power. The general observations outlined above show the expected result, confirming the influence of KWSP on cream oxidation. The obtained values of samples are recorded in Table 5 and presented in Figure A1, Figure A2, Figure A3 and Figure A4.
As shown in Figure A1, Figure A2, Figure A3 and Figure A4, the inhibition capacity values increased in the presence of different KWSP contents compared to the control. Accordingly, a very low percentage (0.1%) of our polysaccharide greatly improved the antioxidant impact of the cream. Our findings revealed that KWSP is an efficient natural antioxidant, based on four methods. These results are consistent with the previous work undertaken by Puangmanee et al. [49], who reported that ice cream with glycated whey protein produces ice cream with a good antioxidant activity. It seems that the potent antioxidant activity is the byproduct of the relationship between the structure of the conjugated sugar and the antioxidant activity [49].
3.6.2. Amelioration of K232 and K270
As the reduction or elimination of fat droplets induces a change in terms of the physicochemical properties of the food (optical properties, texture, and taste), it is necessary to optimize or reduce the fat in foods, to maintain their textural properties [23]. From this perspective, many substances are used to substitute or enhance the value of fat droplets in food. Generally, polysaccharides have been increasingly utilized as a substitute for a lower fat content and to provide a greasy texture and a highly appealing sensory quality. The K232 and K270 values indicate the formation of conjugated dienes of polyunsaturated fatty acids and the presence of primary and secondary oxidation products, respectively [25]. Owing to the incorporation of KWSP, a sharp decrease in K232 and K270 was detected for the enriched extracted fat samples. Indeed, the primary and secondary oxidation products dropped from 14.35 to 10.45 for K232 and from 2.44 to 1.3 for K270. Therefore, we can assert that KWSP displays a good antioxidant capacity, to reduce the formation of conjugated dienes and trienes in cream fat. The reduction in the extinction coefficient in whipped cream with polysaccharides can be assigned to the inactivation in terms of lipid free radicals or the prevention of the hydroperoxides from becoming free radicals [50].
4. Conclusions
Food polysaccharides refer to the polysaccharides that have been approved for industrial use in the formulation of food products. At this stage of analysis, it is worth noting that polysaccharides have been extensively used in the food industry to prepare food products with improved functionality, where their applications are dependent on their physicochemical properties. From this perspective, researchers are always searching for novel properties. In this regard, the current study started by discussing the physicochemical properties and identifying the polysaccharides extracted from carob kibbles (KWSP), through hot water techniques, as well as investigating their antioxidant, functional, and antibacterial activities.
The results obtained in this study revealed that KWSP exhibits a good WHC, OHC, and emulsification stability. The spectroscopic analyses yielded that KWSP is a heteropolysaccharide, corresponding to a glucose and fructose polymer. The average molecular weight of KWSP is around 65 kDa. Furthermore, KWSP has an antibacterial activity against S. enterica, L. monocytogenes, and S. aureus. KWSP also exhibits antioxidant activities. Thus, it could be utilized as a natural antioxidant when incorporated into whipped cream. Accordingly, KWSP are being considered as a prospective antioxidant from a natural source. Their enrichment in whipped cream may not only enhance its antioxidant profile, but also may remarkably restrain the formation of harmful products that might have negative effects on consumers’ health.
This research may provide some reasonable insights into the antioxidant and antibacterial activities of a purified polysaccharide extracted from Ceratonia siliqua L. and its enhancement of performance in the food industry, such as in whipped cream. Some future research perspectives should be addressed. First, a full understanding of the structure–property relationship of the polysaccharide will be studied, for its incorporation in other dairy products, such as butter, cheese, or milk. Second, it seems that their interesting characteristics may further suggest the application of KWSP in the cosmetics industry.
Author Contributions
Conceptualization, A.D. and M.B.; methodology, A.D., S.B.S. and M.B. software, A.D., S.B.S., M.D.R.G.d.S. and R.B.S.; validation, A.D., M.B. and M.D.R.G.d.S.; formal analysis, A.D., S.B.S., M.D.R.G.d.S. and R.B.S.; investigation, A.D., W.H. and A.M.A.; resources, A.D., S.B.S., M.D.R.G.d.S., R.B.S. and M.B.; data curation, A.D. and M.B.; writing—original draft preparation, A.D. and M.B.; writing—review and editing, A.D., A.M.A. and W.H.; visualization, M.B., A.M.A. and W.H.; supervision, R.B.S. and M.B.; project administration, M.B., A.M.A. and W.H.; funding acquisition, A.M.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Tunisian Ministry of Higher Education and Scientific Research, grant number LR14ES08 and the APC was funded by Taif University Researchers Supporting Project number (TURSP-2020/229), Taif University, Taif, Saudi Arabia.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Acknowledgments
This research was supported by (1) the Tunisian Ministry of Higher Education and Scientific Research, Contract program number LR14ES08 and by (2) Taif University Researchers Supporting Project number (TURSP-2020/229), Taif University, Taif, Saudi Arabia. First, the authors are grateful for this financial support. Second, the authors would like to thank the editors and the anonymous referees for their valuable and constructive comments on the first draft of this manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix A

Figure A1.
Effect of KWSP on DPPH.
Figure A1.
Effect of KWSP on DPPH.
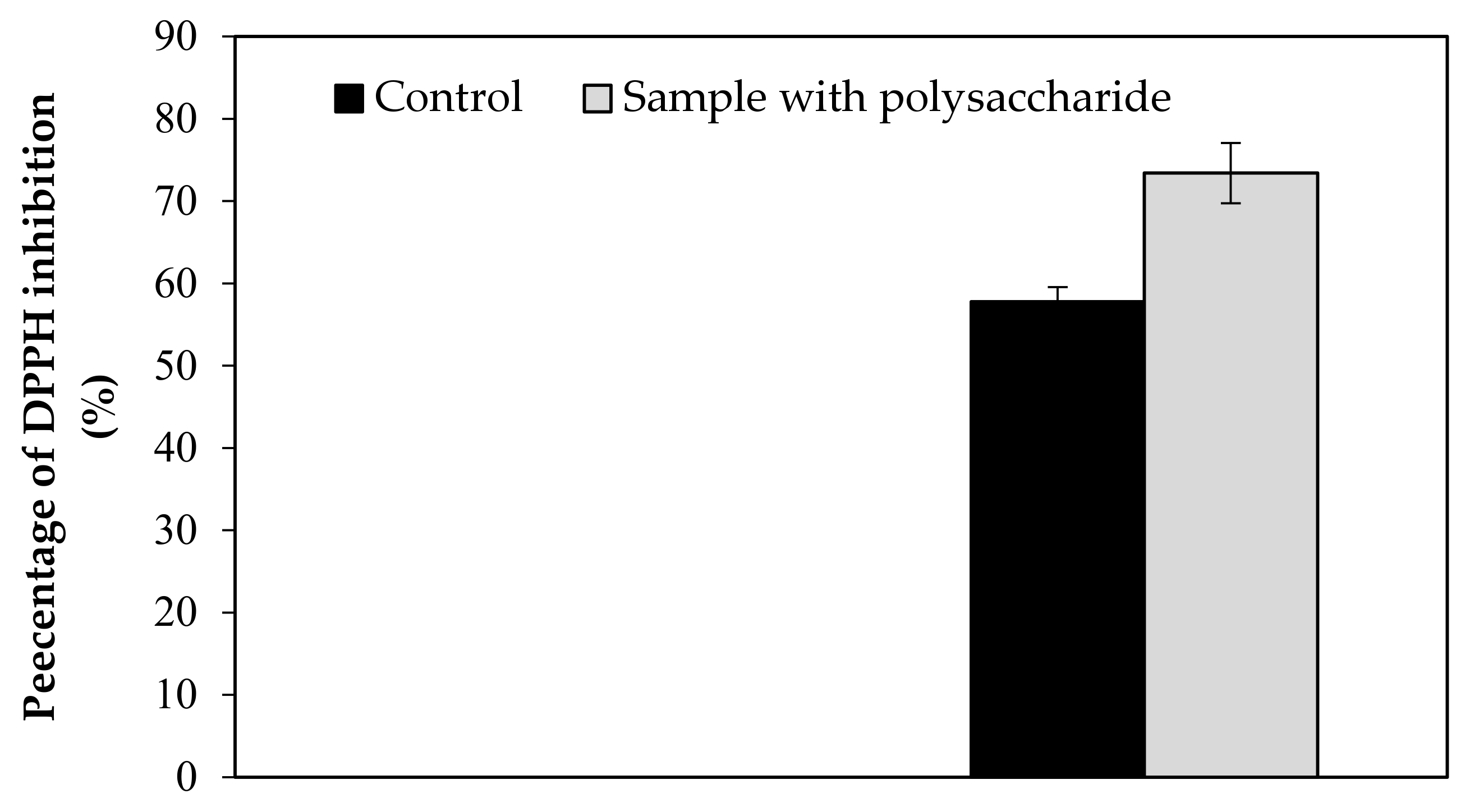
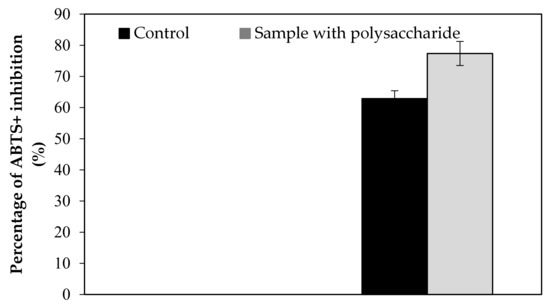
Figure A2.
Effect of KWSP on ABTS+.
Figure A2.
Effect of KWSP on ABTS+.
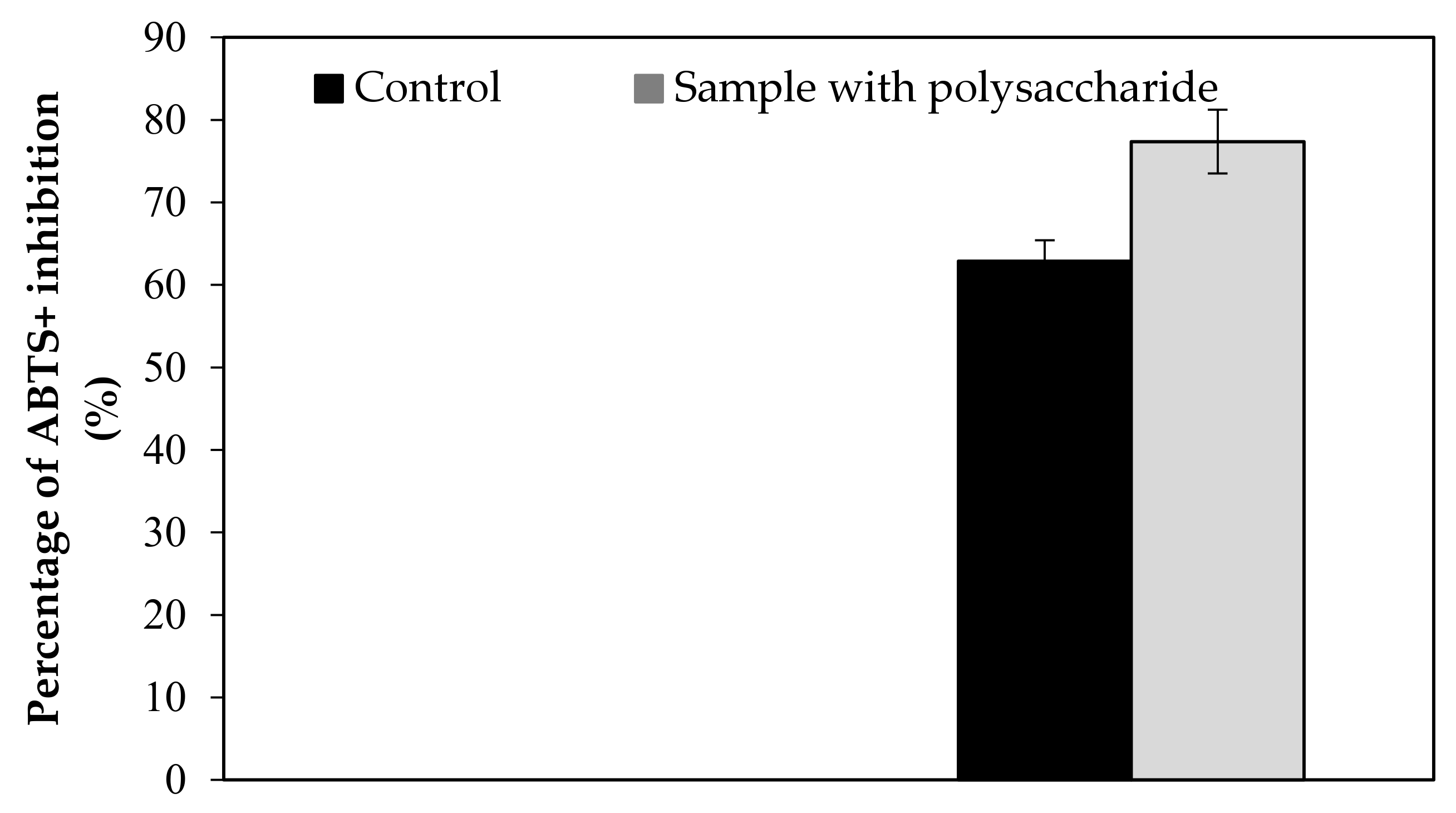
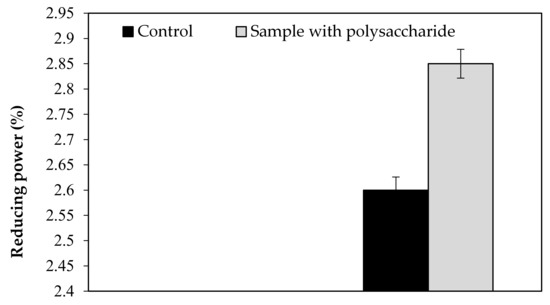
Figure A3.
Effect of KWSP on reducing power.
Figure A3.
Effect of KWSP on reducing power.
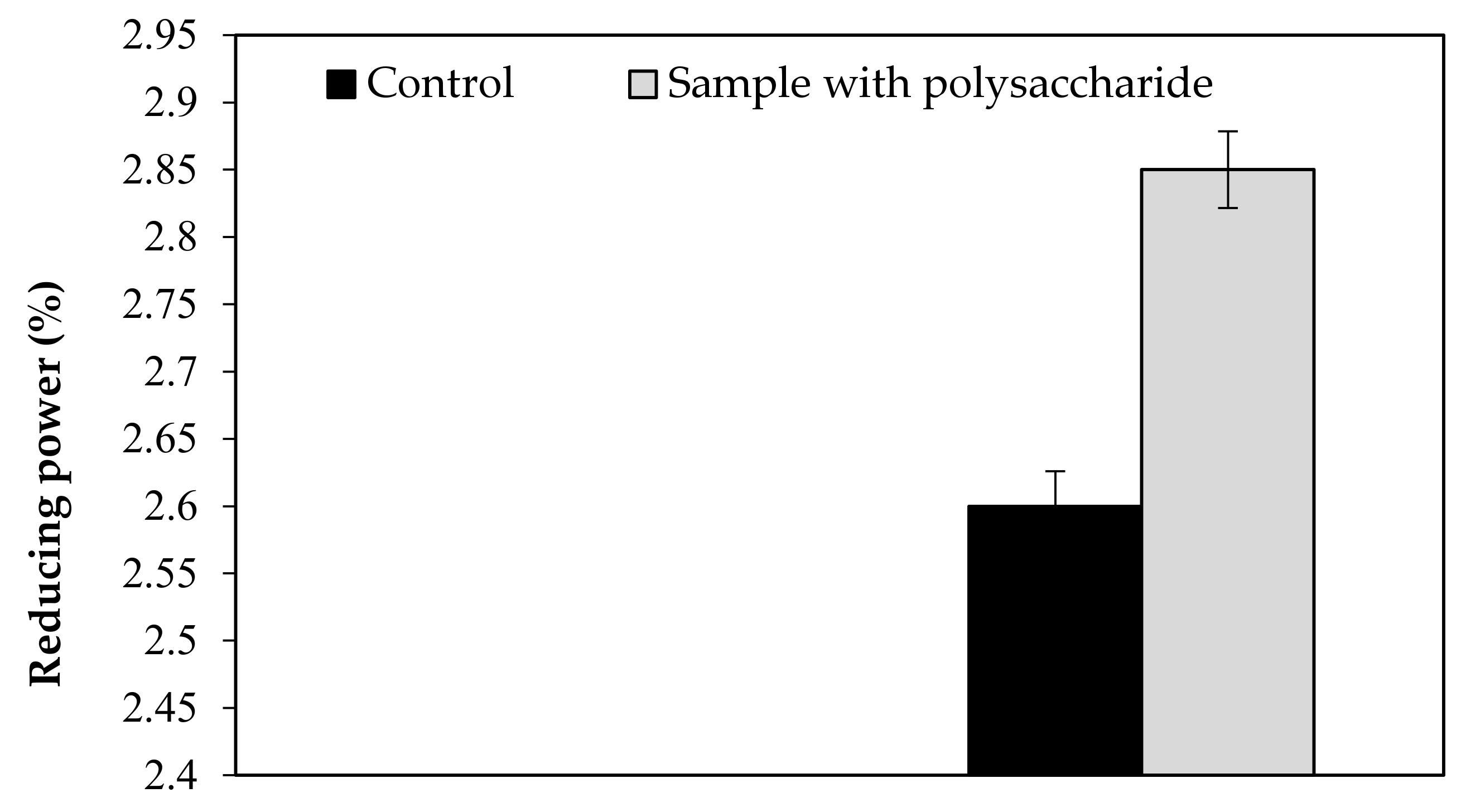
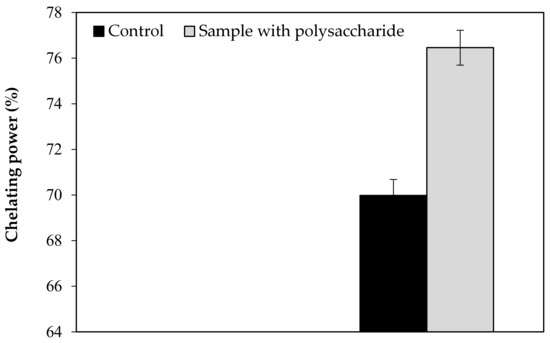
Figure A4.
Effect of KWSP on Chelating power.
Figure A4.
Effect of KWSP on Chelating power.
References
- Bourhia, M.; Bouothmany, K.; Bakrim, H.; Hadrach, S.; Salamatullah, A.M.; Alzahrani, A.; Khalil Alyahya, H.; Albadr, N.A.; Gmouh, S.; Laglaoui, A.; et al. Chemical Profiling, Antioxidant, Antiproliferative, and Antibacterial Potentials of Chemically Characterized Extract of Citrullus colocynthis L. Seeds. Separations 2021, 8, 114. [Google Scholar] [CrossRef]
- Rtibi, K.; Selmi, S.; Gramia, D.; Amri, M.; Etod, B.; El-benna, J.; Sebai, H.; Marzouki, L. Chemical constituents and pharmacological actions of carob pods and leaves (Ceratonia siliqua L.) on the gastrointestinal tract. J. Biomed. Pharmacother. 2017, 93, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Batlle, I.; Tous, J. Carob Tree. Ceratonia siliqua L.; Promoting the Conservation and Use of Underutilized and Neglected Crops; International Plant Genetic Resources Institute: Rome, Italy, 1997; Available online: https://www.bioversityinternational.org/fileadmin/_migrated/uploads/tx_news/Carob_tree_Ceratonia_siliqua_L._347.pdf (accessed on 16 April 2022).
- Roseiro, L.B.; Duarte, L.C.; Oliveira, D.L.; Roque, R.; Bernardo-Gil, M.G.; Martins, A.I.; Sepúlveda, C.; Almeida, J.; Meireles, M.; Gírio, F.M.A. Pilar Rauter, Supercritical, ultrasound and conventional extracts from carob (Ceratonia siliqua L.) biomass: Effect on the phenolic profile and antiproliferative activity. J. Ind. Crops Prod. 2013, 47, 132–138. [Google Scholar] [CrossRef]
- De la Fuente-Fernández, M.; González-Hedström, D.; Amor, S.; Tejera-Muñoz, A.; Fernández, N.; Monge, L.; Almodóvar, P.; Andrés-Delgado, L.; Santamaría, L.; Prodanov, M.; et al. Supplementation with a Carob (Ceratonia siliqua L.) Fruit Extract Attenuates the Cardiometabolic Alterations Associated with Metabolic Syndrome in Mice. Antioxidants 2020, 9, 339. [Google Scholar]
- Emilia Brassesco, M.; Brandão, T.R.S.; Silva, C.L.M.; Pintado, M. Carob bean (Ceratonia siliqua L.): A new perspective for functional food. J. Trends Food Sci. Technol. 2021, 114, 310–322. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices, Antioxidant activity and health effects. J. Funct. Foods 2015, 78, 820–897. [Google Scholar] [CrossRef]
- Abidar, S.; Stefan Boiangiu, R.; Stefan Boiangiu, G.; Todirascu-Ciornea, E.; Amakran, A.; Cioanca, O.; Cioanca, L.; Nhiri, M. The Aqueous Extract from Ceratonia siliqua Leaves Protects against 6-Hydroxydopamine in Zebrafish: Understanding the Underlying Mechanism. Antioxidants 2020, 9, 304. [Google Scholar] [CrossRef]
- Hamzaoui, A.; Ghariani, M.; Sellem, I.; Hamdi, M.; Feki, A.; Jaballi, I.; Nasri, M.; Ben Amara, I. Extraction, characterization and biological properties of polysaccharide derived from green seaweed “Chaetomorpha linum” and its potential application in Tunisian beef sausages. Int. J. Biol. Macromol. 2020, 148, 1156–1168. [Google Scholar] [CrossRef]
- Sila, A.; Bayar, N.; Ghazala, I.; Bougatef, A.; Ellouze-Ghorbel, R.; Ellouze-Chaabouni, S. Water-soluble polysaccharides from agro-industrial by-products: Functional and biological Properties. Int. J. Biol. Macromol. 2014, 69, 236–243. [Google Scholar] [CrossRef]
- Lakkab, I.; El Hajaji, H.; Lachkar, N.; Lefter, R.; Ciobica, A.; El Balia, B.; Lachkar, M. Ceratonia siliqua L. seed peels: Phytochemical profile, antioxidant activity, and effect on mood disorders. J. Funct. Foods 2019, 54, 457–465. [Google Scholar] [CrossRef]
- Su, Y.; Li, L. Structural characterization and antioxidant activity of polysaccharide from four auriculariales. Carbohydr. Polym. 2020, 229, 115407. [Google Scholar] [CrossRef]
- Carbas, B.; Salinas, M.V.; Serrano, C.; Passarinho, J.A.; Puppo, M.C.; Ricardo, C.P.; Brites, C. Chemical composition and antioxidant activity of commercial flours from Ceratonia siliqua and Prosopis spp. J. Food Meas. Charact. 2019, 13, 305–311. [Google Scholar] [CrossRef]
- Benković, M.; Belščak-Cvitanović, A.; Bauman, I.; Komes, D.; Srečec, S. Flow properties and chemical composition of carob (Ceratonia siliqua L.) flours as related to particle size and seed presence. Food Res. Int. 2017, 100, 211–218. [Google Scholar] [CrossRef]
- Liu, G.; Xu, S.; Chen, L. Chemical composition and bioactivities of a water-soluble polysaccharide from the endodermis of shaddock. Int. J. Biol. Macromol. 2012, 51, 763–766. [Google Scholar] [CrossRef]
- AOAC (Association of Official Analytical Chemists). Method of Analysis Gaithersburg; AOAC International: Gaithersburg, MD, USA, 2000. [Google Scholar]
- Khemakhem, I.; Abdelhedi, O.; Trigui, I.; Ayadi, M.A.; Bouaziz, M. Structural, antioxidant and antibacterial activities of polysaccharides extracted from olive leaves. Int. J. Biol. Macromol. 2018, 106, 425–432. [Google Scholar] [CrossRef]
- Slima, S.B.; Ktari, N.; Trabelsi, I.; Moussa, H.; Makni, I.; Salah, R.B. Purification, characterization and antioxidant properties of a novel polysaccharide extracted from Sorghum bicolor (L.) seeds in sausage. Int. J. Biol. Macromol. 2018, 106, 168–178. [Google Scholar] [CrossRef]
- Freitas, F.; Alves, V.D.; Torres, C.A.; Cruz, M.; Sousa, I.; Melo, M.J.; Reis, M.A. Fucose-containing exopolysaccharide produced by the newly isolated Enterobacter strain A47 DSM 23139. Carbohydr. Polym. 2011, 83, 159–165. [Google Scholar] [CrossRef]
- Bersuder, P.; Hole, M.; Smith, G. Antioxidants from a heated histidine-glucose model system. I: Investigation of the antioxidant role of histidine and isolation of antioxidants by high-performance liquid chromatography. J. Am. Oil Chem. Soc. 1998, 75, 181–187. [Google Scholar] [CrossRef]
- Braca, A.; De Tommasi, N.; Di Bari, L.; Pizza, C.; Politi, M.; Morelli, I. Antioxidant principles from Bauhinia tarapotensis. J. Nat. Prod. 2001, 64, 892–895. [Google Scholar] [CrossRef]
- Prieto, P.; Pineda, M.; Aguilar, M. Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: Specific application to the determination of Vitamin E. Anal. Biochem. 1999, 269, 337–341. [Google Scholar] [CrossRef]
- Ktari, N.; Bkhairia, I.; Nasri, M.; Salah, R.B. Structure and biological activities of polysaccharide purified from Senegrain seed. Int. J. Biol. Macromol. 2020, 144, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Ezernieks, V.; Rochfort, S.; Cocks, B. Comparison of methylation methods for fatty acid analysis of milk fat. Food Chem. 2018, 261, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Hammouda, I.B.; Márquez-Ruiz, G.; Holgado, F.; Sonda, A.; Skalicka-Wozniak, K.; Bouaziz, M. RP-UHPLC–DAD-QTOF-MS as a Powerful Tool of Oleuropein and Ligstroside Characterization in Olive-Leaf Extract and Their Contribution to the Improved Performance of Refined Olive-Pomace Oil during Heating. J. Agric. Food Chem. 2020, 68, 12039–12047. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Li, A.; Li, X.; Sun, L.; Guo, Y. An overview of classifications, properties of food polysaccharides and their links to applications in improving food textures. Trends Food Sci. Technol. 2020, 102, 1–15. [Google Scholar] [CrossRef]
- Jeddou, K.B.; Chaari, F.; Maktouf, S.; Nouri-Ellouz, O.; Helbert, C.B.; Ghorbel, R.E. Structural, functional, and antioxidant properties of water-soluble polysaccharides from potatoes peels. Food Chem. 2016, 205, 97–105. [Google Scholar] [CrossRef]
- El Gerssifi, M. Les défauts des produits de pâtisserie et biscuiterie au cours du stockage: La prévention par la formulation. Ind. Aliment. Agric. 1998, 115, 82–88. [Google Scholar]
- Petkova, N.; Petrova, I.; Ivanov, I.; Mihov, R.; Hadjikinova, R.; Ognyanov, M.; Nikolova, V. Nutritional and antioxidant potential of carob (Ceratonia siliqua) flour and evaluation of functional properties of its polysaccharide fraction. J. Pharm. Sci. Res. 2017, 9, 2189–2195. [Google Scholar]
- Sciarini, L.S.; Maldonado, F.; Ribotta, P.D.; Pérez, G.T.; León, A.E. Chemical composition and functional properties of Gleditsia triacanthos gum. Food Hydrocoll. 2009, 23, 306–313. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Barber, X.; Pérez-Álvarez, J.A.; Fernández-López, J. Assessment of chemical, physico-chemical, techno-functional and antioxidant properties of fig (Ficus carica L.) powder co-products. Ind. Crops Prod. 2015, 69, 472–479. [Google Scholar] [CrossRef]
- Ktari, N.; Feki, A.; Trabelsi, I.; Triki, M.; Maalej, H.; Slima, S.B.; Salah, R.B. Structure, functional and antioxidant properties in Tunisian beef sausage of a novel polysaccharide from Trigonella foenum-graecum seeds. Int. J. Biol. Macromol. 2017, 98, 169–181. [Google Scholar] [CrossRef]
- Sun, M.; Sun, Y.; Li, Y.; Liu, Y.; Liang, J.; Zhang, Z. Physical properties and antidiabetic potential of a novel galactomannan from seeds of Gleditsia japonica var delavayi. J. Funct. Foods 2018, 46, 546–555. [Google Scholar] [CrossRef]
- De Andrade Vieira, É.; Alcântara, M.A.; Dos Santos, N.A.; Gondim, A.D.; Iacomini, M.; Mellinger, C.; de Magalhães Cordeiro, A.M.T. Mucilages of cacti from Brazilian biodiversity: Extraction, physicochemical and technological properties. Food Chem. 2021, 346, 128892. [Google Scholar] [CrossRef]
- Zhang, W.; Fan, X.; Gu, X.; Gong, S.; Wu, J.; Wang, Z.; Wang, S. Emulsifying properties of pectic polysaccharides obtained by sequential extraction from black tomato pomace. Food Hydrocoll. 2020, 100, 105454. [Google Scholar] [CrossRef]
- Bubb, W.A. NMR spectroscopy in the study of carbohydrates: Characterizing the structural complexity. Concepts Magn. Reson. Part A Educ. J. 2003, 19, 1–19. [Google Scholar] [CrossRef]
- Medlej, M.K.; Cherri, B.; Nasser, G.; Zaviska, F.; Hijazi, A.; Li, S.; Pochat-Bohatier, C. Optimization of polysaccharides extraction from a wild species of Ornithogalum combining ultrasound and maceration and their anti-oxidant properties. Int. J. Biol. Macromol. 2020, 161, 958–968. [Google Scholar] [CrossRef]
- Wang, Y.M.; Wu, F.J.; Du, L.; Li, G.Y.; Takahashi, K.; Xue, Y.; Xue, C.H. Effects of polysaccharides from abalone (Haliotis discus hannai Ino) on HepG2 cell proliferation. Int. J. Biol. Macromol. 2014, 66, 354–361. [Google Scholar] [CrossRef]
- Zhu, J.; Liu, W.; Yu, J.; Zou, S.; Wang, J.; Yao, W.; Gao, X. Characterization and hypoglycemic effect of a polysaccharide extracted from the fruit of Lycium barbarum L. Carbohydr. Polym. 2013, 98, 8–16. [Google Scholar] [CrossRef]
- Park, F.S. Application of IR Spectroscopy in Biochemistry: Biology and Medicine; Plenum Press: New York, NY, USA, 1971; pp. 100–140. [Google Scholar]
- Krichen, F.; Karoud, W.; Sila, A.; Abdelmalek, B.E.; Ghorbel, R.; Ellouz-Chaabouni, S.; Bougatef, A. Extraction, characterization and antimicrobial activity of sulfated polysaccharides from fish skins. Int. J. Biol. Macromol. 2015, 75, 283–289. [Google Scholar] [CrossRef]
- Ahmad, M.M. Characterization and antioxidant activities of polysaccharides extracted from flageolet bean pods waste. Curr. Res. Green Sustain. Chem. 2021, 4, 100154. [Google Scholar] [CrossRef]
- Boual, Z.; Pierre, G.; Delattre, C.; Benaoun, F.; Petit, E.; Gardarin, C.; El Hadj, M.D.O. Mediterranean semi-arid plant Astragalus armatus as a source of bioactive galactomannan. Bioact. Carbohydr. Diet. Fibre 2015, 5, 10–18. [Google Scholar] [CrossRef]
- Mutaillifu, P.; Bobakulov, K.; Abuduwaili, A.; Huojiaaihemaiti, H.; Nuerxiati, R.; Aisa, H.A.; Yili, A. Structural characterization and antioxidant activities of a water soluble polysaccharide isolated from Glycyrrhiza glabra. Int. J. Biol. Macromol. 2020, 144, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Mzoughi, Z.; Majdoub, H. Pectic polysaccharides from edible halophytes: Insight on extraction processes, structural characterizations and immunomodulatory potentials. Int. J. Biol. Macromol. 2021, 173, 554–579. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Wang, W.; Wen, P.; Shen, M.; Li, H.; Ren, Y.; Xie, J. Two water-soluble polysaccharides from mung bean skin: Physicochemical characterization, antioxidant and antibacterial activities. Food Hydrocoll. 2020, 100, 105412. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, Y.T.; Zheng, W.; Han, X.X.; Jiang, Y.H.; Hu, P.L.; Shi, L.E. The antibacterial activity and antibacterial mechanism of a polysaccharide from Cordyceps cicadae. J. Funct. Foods 2017, 38, 273–279. [Google Scholar] [CrossRef]
- Walayat, N.; Liu, J.; Nawaz, A.; Aadil, R.M.; López-Pedrouso, M.; Lorenzo, J.M. Role of Food Hydrocolloids as Antioxidants along with Modern Processing Techniques on the Surimi Protein Gel Textural Properties, Developments, Limitation and Future Perspectives. Antioxidants 2022, 11, 486. [Google Scholar] [CrossRef]
- Puangmanee, S.; Hayakawa, S.; Sun, Y.; Ogawa, M. Application of whey protein isolate glycated with rare sugars to ice cream. Food Sci. Technol. Res. 2008, 14, 457. [Google Scholar] [CrossRef][Green Version]
- Bali, O.; Ammar, I.; Ennouri, M.; Attia, H. Physicochemical characteristics and storage stability of clarified butter fat «smen» produced from pasteurized and non-pasteurized milk. J. Pharm. Health Sci. 2017, 5, 195–205. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).