Development, Optimization and Applications of Thin Film Solid Phase Microextraction (TF-SPME) Devices for Thermal Desorption: A Comprehensive Review
Abstract
1. Introduction
2. From Fiber to Thin Film Format: Pros and Cons
3. Types of Desorption Modes for TF-SPME
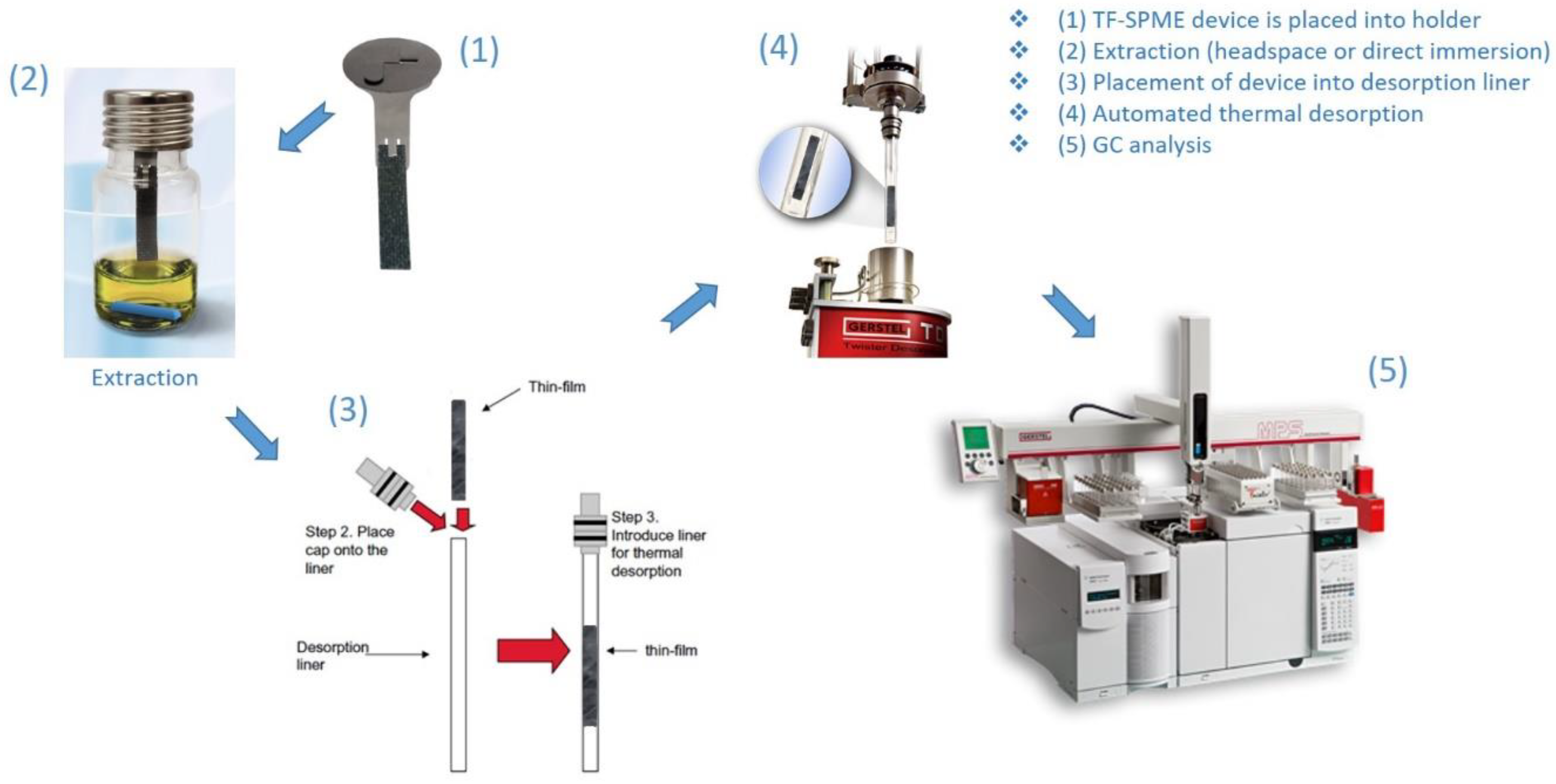
Desorption by Thermal Desorption Unit (TDU)
4. Fundamentals of TF-SPME
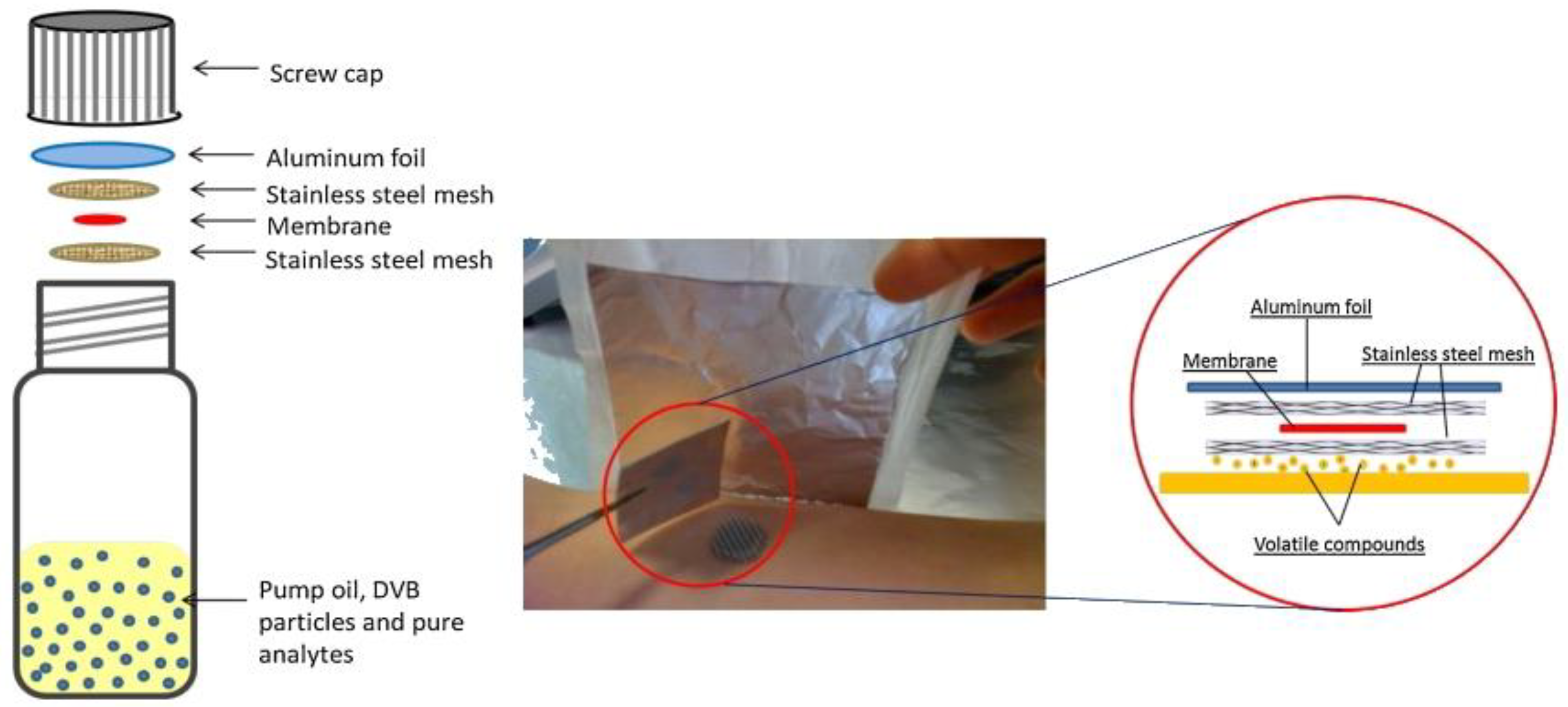
5. Development of the First TF-SPME Device and Improvements up to 2019
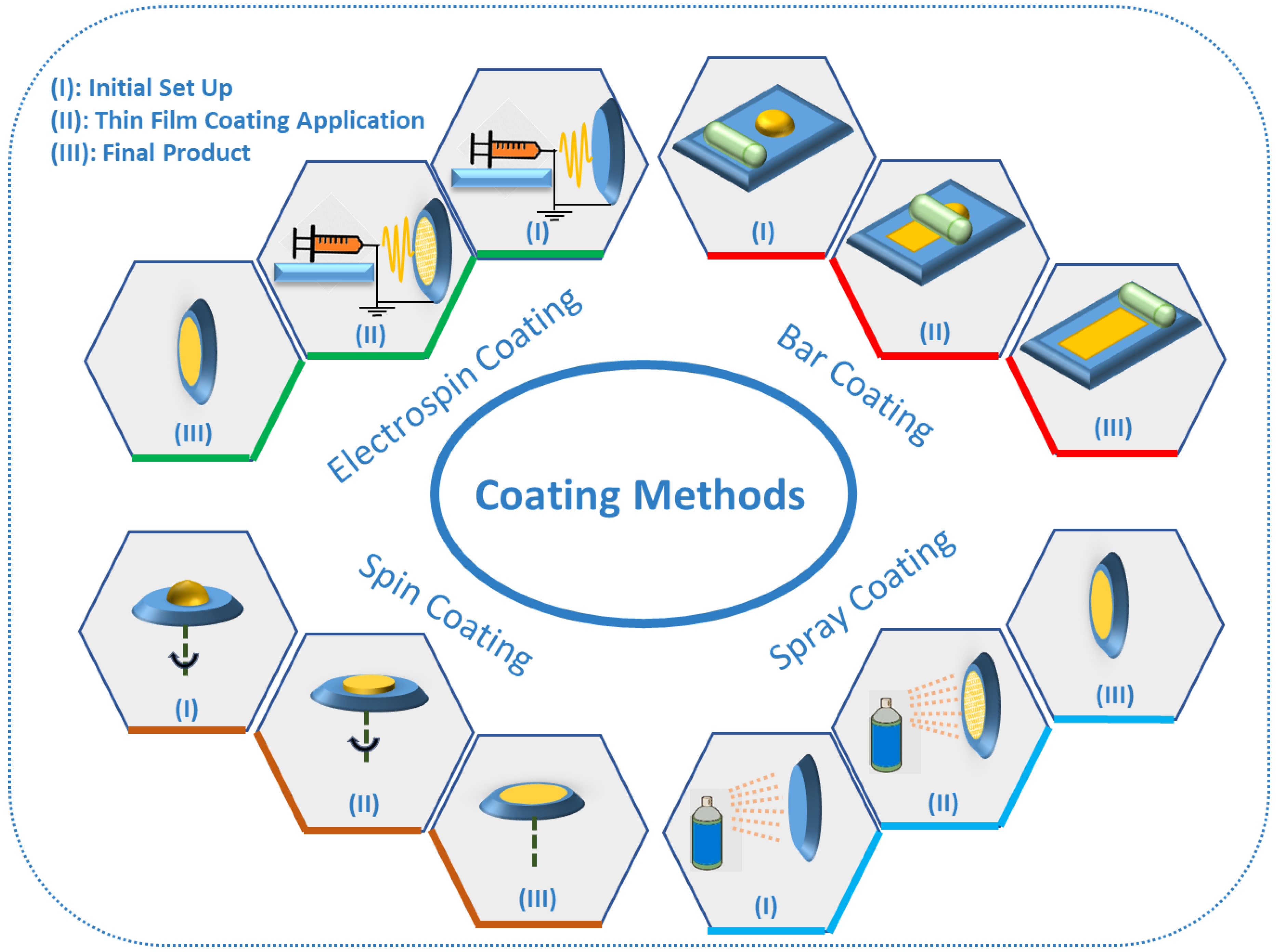
6. TF-SPME Coating Methods
7. Applications
7.1. Environmental Analysis
7.2. Flavors and Fragrance Analysis
7.3. Other Applications of TF-SPME
| Piri-Moghadam et al. [9] | Rodriguez-Lafuente et al. [68] | Piri-Moghadam et al. [55] | Emmons et al. [8] | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Compounds | TF-SPME Carbon Mesh Supported (µg L−1) | TF-SPME Non-Supported (µg L−1) | LLE (µg L−1) LOD | Compounds | Fiber SPME (µg L−1) RDL | Compounds | TF-SPME (ng L−1) | TF-SPME Drill Agitation (ng L−1) | LLE (ng L−1) | Compounds | TF-SPME (µg L−1) | SPME (µg L−1) | SPE (µg L−1) |
| 2,4,6-TCP | 0.050 | 0.025 | 0.50 | 2,4,6-TCP | 0.20 | 2,4,6-TCP | 10 | 100 | 500 | MCHM | 0.10 | 1.0 | 500 |
| 2,3,4,6-TeCP | 0.025 | 0.025 | 0.50 | 2,3,4,6-TeCP | 0.20 | 2,3,4,6-TeCP | 3.0 | 250 | 500 | MMCHC | 0.10 | 2.5 | 250 |
| Trifluralin | 0.050 | 0.050 | 1.0 | Trifluralin | 0.050 | Trifluralin | 3.0 | 50 | 1000 | 4MMCH | 1.0 | 2.5 | 500 |
| Diazinon | 0.050 | 0.050 | 1.0 | Diazinon | 0.50 | Diazinon | 100 | 1000 | 1000 | 1-4CHDM | 0.50 | 0.25 | 5000 |
| Triallate | 0.050 | 0.050 | 1.0 | Triallate | 0.050 | Triallate | 3.0 | 50 | 1000 | DM-1-4CHC | 0.10 | 0.50 | 500 |
| Methyl parathion | 0.50 | 0.50 | 1.0 | Methyl parathion | 0.20 | Methyl parathion | 100 | 1000 | 1000 | MCHCA | 2.0 | 2.50 | 25,000 |
| Alachlor | 0.50 | 0.050 | 0.50 | Alachlor | 0.10 | Alachlor | 10 | 100 | 500 | ||||
| Metalachlor | 0.050 | 0.025 | 5.0 | Metalachlor | 0.10 | Metalachlor | 3.0 | 250 | 500 | ||||
| Chlorpyrifos | 0.25 | 0.10 | 1.0 | Chlorpyrifos | 0.20 | Chlorpyrifos | 10 | 1000 | 1000 | ||||
| Cyanazine | 0.10 | 0.10 | 1.0 | Cyanazine | 1.0 | Cyanazine | 10 | 100 | 1000 | ||||
| 2,4-DCP | 0.10 | 0.050 | 0.25 | 2,4-DCP | 0.10 | ||||||||
| Bendiocarb | 0.050 | 0.025 | 2.0 | Bendiocarb | 0.05 | ||||||||
| Phorate | 0.25 | 0.25 | 0.5 | Phorate | 0.25 | ||||||||
| Carbofurane | 0.10 | 0.10 | 5.0 | Carbofurane | 0.20 | ||||||||
| Simazine | 0.25 | 0.25 | 1.0 | Simazine | 0.050 | ||||||||
| Atrazine | 0.075 | 0.25 | 0.50 | Atrazine | 0.050 | ||||||||
| PCP | 0.075 | 0.10 | 0.50 | PCP | 0.20 | ||||||||
| Terbufos | 0.25 | 0.25 | 0.50 | Terbufos | 0.50 | ||||||||
| Metribuzine | 0.25 | 0.10 | 5.0 | Metribuzine | 0.20 | ||||||||
| Carbaryl | 0.10 | 0.10 | 5.0 | Carbaryl | 0.50 | ||||||||
| Prometryn | 0.075 | 0.075 | 0.25 | Prometryn | 0.20 | ||||||||
| Malathion | 0.50 | 0.50 | 5.0 | Malathion | 0.20 | ||||||||
8. Concluding Remarks and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tobiszewski, M.; Mechlińska, A.; Namieśnik, J. Green analytical chemistry—Theory and practice. Chem. Soc. Rev. 2010, 39, 2869–2878. [Google Scholar] [CrossRef] [PubMed]
- Gałuszka, A.; Migaszewski, Z.M.; Konieczka, P.; Namieśnik, J. Analytical Eco-Scale for assessing the greenness of analytical procedures. TrAC—Trends Anal. Chem. 2012, 37, 61–72. [Google Scholar] [CrossRef]
- Gałuszka, A.; Migaszewski, Z.; Namieśnik, J. The 12 principles of green analytical chemistry and the SIGNIFICANCE mnemonic of green analytical practices. TrAC—Trends Anal. Chem. 2013, 50, 78–84. [Google Scholar] [CrossRef]
- Spietelun, A.; Marcinkowski, Ł.; De La Guardia, M.; Namieśnik, J. Green aspects, developments and perspectives of liquid phase microextraction techniques. Talanta 2014, 119, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Płotka-Wasylka, J. A new tool for the evaluation of the analytical procedure: Green Analytical Procedure Index. Talanta 2018, 181, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Pawliszyn, J. Sample preparation: Quo vadis? Anal. Chem. 2003, 75, 2543–2558. [Google Scholar] [CrossRef] [PubMed]
- Mirnaghi, F.S.; Goryński, K.; Rodriguez-Lafuente, A.; Boyaci, E.; Bojko, B.; Pawliszyn, J. Microextraction versus exhaustive extraction approaches for simultaneous analysis of compounds in wide range of polarity. J. Chromatogr. A 2013, 1316, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Emmons, R.; Devasurendra, A.; Godage, N.; Gionfriddo, E. Exploring the Efficiency of Various Extraction Approaches for Determination of Crude (4-methylcyclohexyl)methanol (MCHM) Constituents in Environmental Samples. LC-GC N. Am. 2019, 37, 27–34. [Google Scholar]
- Piri-Moghadam, H.; Gionfriddo, E.; Rodriguez-Lafuente, A.; Grandy, J.J.; Lord, H.L.; Obal, T.; Pawliszyn, J. Inter-laboratory validation of a thin film microextraction technique for determination of pesticides in surface water samples. Anal. Chim. Acta 2017, 964, 74–84. [Google Scholar] [CrossRef]
- Ouyang, G.; Pawliszyn, J. A critical review in calibration methods for solid-phase microextraction. Anal. Chim. Acta 2008, 627, 184–197. [Google Scholar] [CrossRef]
- Reyes-Garcés, N.; Gionfriddo, E.; Gómez-Ríos, G.A.; Alam, M.N.; Boyaci, E.; Bojko, B.; Singh, V.; Grandy, J.; Pawliszyn, J. Advances in Solid Phase Microextraction and Perspective on Future Directions. Anal. Chem. 2018, 90, 302–360. [Google Scholar] [CrossRef]
- Bruheim, I.; Liu, X.; Pawliszyn, J. Thin-film microextraction. Anal. Chem. 2003, 75, 1002–1010. [Google Scholar] [CrossRef]
- Kremser, A.; Jochmann, M.A.; Schmidt, T.C. PAL SPME Arrow—Evaluation of a novel solid-phase microextraction device for freely dissolved PAHs in water. Anal. Bioanal. Chem. 2016, 408, 943–952. [Google Scholar] [CrossRef]
- Yuan, Y.; Lin, X.; Li, T.; Pang, T.; Dong, Y.; Zhuo, R.; Wang, Q.; Cao, Y.; Gan, N. A solid phase microextraction Arrow with zirconium metal–organic framework/molybdenum disulfide coating coupled with gas chromatography–mass spectrometer for the determination of polycyclic aromatic hydrocarbons in fish samples. J. Chromatogr. A 2019, 1592, 9–18. [Google Scholar] [CrossRef]
- Baltussen, E.; Sandra, P.; David, F.; Cramers, C. Stir Bar Sorptive Extraction (SBSE), a Novel Extraction Technique for Aqueous Samples: Theory and Principles. J. Microcolumn Sep. 1999, 737–747. [Google Scholar] [CrossRef]
- Qin, Z.; Bragg, L.; Ouyang, G.; Pawliszyn, J. Comparison of thin-film microextraction and stir bar sorptive extraction for the analysis of polycyclic aromatic hydrocarbons in aqueous samples with controlled agitation conditions. J. Chromatogr. A 2008, 1196–1197, 89–95. [Google Scholar] [CrossRef]
- David, F.; Ochiai, N.; Sandra, P. Two decades of stir bar sorptive extraction: A retrospective and future outlook. TrAC—Trends Anal. Chem. 2019, 112, 102–111. [Google Scholar] [CrossRef]
- Yamaguchi, M.S.; McCartney, M.M.; Linderholm, A.L.; Ebeler, S.E.; Schivo, M.; Davis, C.E. Headspace sorptive extraction-gas chromatography–mass spectrometry method to measure volatile emissions from human airway cell cultures. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2018, 1090, 36–42. [Google Scholar] [CrossRef]
- Grandy, J.J.; Boyaci, E.; Pawliszyn, J. Development of a Carbon Mesh Supported Thin Film Microextraction Membrane As a Means to Lower the Detection Limits of Benchtop and Portable GC/MS Instrumentation. Anal. Chem. 2016, 88, 1760–1767. [Google Scholar] [CrossRef]
- Mukhtar, N.H.; See, H.H. Carbonaceous nanomaterials immobilised mixed matrix membrane microextraction for the determination of polycyclic aromatic hydrocarbons in sewage pond water samples. Anal. Chim. Acta 2016, 931, 57–63. [Google Scholar] [CrossRef]
- Boyaci, E.; Goryński, K.; Viteri, C.R.; Pawliszyn, J. A study of thin film solid phase microextraction methods for analysis of fluorinated benzoic acids in seawater. J. Chromatogr. A 2016, 1436, 51–58. [Google Scholar] [CrossRef]
- Gionfriddo, E.; Boyacl, E.; Pawliszyn, J. New Generation of Solid-Phase Microextraction Coatings for Complementary Separation Approaches: A Step toward Comprehensive Metabolomics and Multiresidue Analyses in Complex Matrices. Anal. Chem. 2017, 89, 4046–4054. [Google Scholar] [CrossRef]
- Ghani, M.; Ghoreishi, S.M.; Salehinia, S.; Mousavi, N.; Ansarinejad, H. Electrochemically decorated network-like cobalt oxide nanosheets on nickel oxide nanoworms substrate as a sorbent for the thin film microextraction of diclofenac. Microchem. J. 2019, 146, 149–156. [Google Scholar] [CrossRef]
- Olcer, Y.A.; Tascon, M.; Eroglu, A.E.; Boyaci, E. Thin film microextraction: Towards faster and more sensitive microextraction. TrAC Trends Anal. Chem. 2019. [Google Scholar] [CrossRef]
- Walles, M.; Gu, Y.; Dartiguenave, C.; Musteata, F.M.; Waldron, K.; Lubda, D.; Pawliszyn, J. Approaches for coupling solid-phase microextraction to nanospray. J. Chromatogr. A 2005, 1067, 197–205. [Google Scholar] [CrossRef]
- Gómez-Ríos, G.A.; Reyes-Garcés, N.; Bojko, B.; Pawliszyn, J. Biocompatible Solid-Phase Microextraction Nanoelectrospray Ionization: An Unexploited Tool in Bioanalysis. Anal. Chem. 2016, 88, 1259–1265. [Google Scholar] [CrossRef]
- Romero, V.; Costas-Mora, I.; Lavilla, I.; Bendicho, C. Headspace thin-film microextraction onto graphene membranes for specific detection of methyl(cyclopentadienyl)-tricarbonyl manganese in water samples by total reflection X-ray fluorescence. Spectrochim. Acta—Part B At. Spectrosc. 2016, 126, 65–70. [Google Scholar] [CrossRef]
- De la Calle, I.; Ruibal, T.; Lavilla, I.; Bendicho, C. Direct immersion thin-film microextraction method based on the sorption of pyrrolidine dithiocarbamate metal chelates onto graphene membranes followed by total reflection X-ray fluorescence analysis. Spectrochim. Acta—Part B At. Spectrosc. 2019, 152, 14–24. [Google Scholar] [CrossRef]
- Riazi Kermani, F.; Pawliszyn, J. Sorbent coated glass wool fabric as a thin film microextraction device. Anal. Chem. 2012, 84, 8990–8995. [Google Scholar] [CrossRef]
- Grandy, J.J.; Singh, V.; Lashgari, M.; Gauthier, M.; Pawliszyn, J. Development of a Hydrophilic Lipophilic Balanced Thin Film Solid Phase Microextraction Device for Balanced Determination of Volatile Organic Compounds. Anal. Chem. 2018, 90, 14072–14080. [Google Scholar] [CrossRef]
- Wilcockson, J.B.; Gobas, F.A.P.C. Thin-film solid-phase extraction to measure fugacities of organic chemicals with low volatility in biological samples. Environ. Sci. Technol. 2001, 35, 1425–1431. [Google Scholar] [CrossRef]
- Rodil, R.; von Sonntag, J.; Montero, L.; Popp, P.; Buchmeiser, M.R. Glass-fiber reinforced poly(acrylate)-based sorptive materials for the enrichment of organic micropollutants from aqueous samples. J. Chromatogr. A 2007, 1138, 1–9. [Google Scholar] [CrossRef]
- Boggia, L.; Sgorbini, B.; Bertea, C.M.; Cagliero, C.; Bicchi, C.; Maffei, M.E.; Rubiolo, P. Direct Contact—Sorptive Tape Extraction coupled with Gas Chromatography—Mass Spectrometry to reveal volatile topographical dynamics of lima bean (Phaseolus lunatus L.) upon herbivory by Spodoptera littoralis Boisd. BMC Plant Biol. 2015, 15. [Google Scholar] [CrossRef]
- Vernarelli, L.; Whitecavage, J.A.; Stuff, J. Analysis of Food Samples using Thin Film Solid Phase Microextraction (TF-SPME) and Thermal Desorption GC/MS. 2019, 1–7. Available online: http://www.gerstel.com/pdf/AppNote-202.pdf (accessed on 30 July 2019).
- Jiang, R.; Cudjoe, E.; Bojko, B.; Abaffy, T.; Pawliszyn, J. A non-invasive method for in vivo skin volatile compounds sampling. Anal. Chim. Acta 2013, 804, 111–119. [Google Scholar] [CrossRef]
- Jiang, R.; Pawliszyn, J. Preparation of a particle-loaded membrane for trace gas sampling. Anal. Chem. 2014, 86, 403–410. [Google Scholar] [CrossRef]
- Gómez-Ríos, G.A.; Gionfriddo, E.; Poole, J.; Pawliszyn, J. Ultrafast Screening and Quantitation of Pesticides in Food and Environmental Matrices by Solid-Phase Microextraction-Transmission Mode (SPME-TM) and Direct Analysis in Real Time (DART). Anal. Chem. 2017, 89, 7240–7248. [Google Scholar] [CrossRef]
- Jastrzembski, J.A.; Sacks, G.L. Solid Phase Mesh Enhanced Sorption from Headspace (SPMESH) Coupled to DART-MS for Rapid Quantification of Trace-Level Volatiles. Anal. Chem. 2016, 88, 8617–8623. [Google Scholar] [CrossRef]
- Mirabelli, M.F.; Gionfriddo, E.; Pawliszyn, J.; Zenobi, R. Fast screening of illicit drugs in beverages and biological fluids by direct coupling of thin film microextraction to dielectric barrier discharge ionization-mass spectrometry. Analyst 2019, 144, 2788–2796. [Google Scholar] [CrossRef]
- Cho, D.S.; Gibson, S.C.; Bhandari, D.; McNally, M.E.; Hoffman, R.M.; Cook, K.D.; Song, L. Evaluation of direct analysis in real time mass spectrometry for onsite monitoring of batch slurry reactions. Rapid Commun. Mass Spectrom. 2011, 25, 3575–3580. [Google Scholar] [CrossRef]
- Piri-Moghadam, H.; Alam, M.N.; Pawliszyn, J. Review of geometries and coating materials in solid phase microextraction: Opportunities, limitations, and future perspectives. Anal. Chim. Acta 2017, 984, 42–65. [Google Scholar] [CrossRef]
- Jiang, R.; Pawliszyn, J. Thin-film microextraction offers another geometry for solid-phase microextraction. TrAC—Trends Anal. Chem. 2012, 39, 245–253. [Google Scholar] [CrossRef]
- Semenov, S.N.; Koziel, J.A.; Pawliszyn, J. Kinetics of solid-phase extraction and solid-phase microextraction in thin adsorbent layer with saturation sorption isotherm. J. Chromatogr. A 2000, 873, 39–51. [Google Scholar] [CrossRef]
- Harner, T.; Farrar, N.J.; Shoeib, M.; Jones, K.C.; Gobas, F.A.P.C. Characterization of polymer-coated glass as a passive air sampler for persistent organic pollutants. Environ. Sci. Technol. 2003, 37, 2486–2493. [Google Scholar] [CrossRef]
- Kennedy, K.E.; Hawker, D.W.; Müller, J.F.; Bartkow, M.E.; Truss, R.W. A field comparison of ethylene vinyl acetate and low-density polyethylene thin films for equilibrium phase passive air sampling of polycyclic aromatic hydrocarbons. Atmos. Environ. 2007, 41, 5778–5787. [Google Scholar] [CrossRef]
- Reichenberg, F.; Smedes, F.; Jönsson, J.A.; Mayer, P. Determining the chemical activity of hydrophobic organic compounds in soil using polymer coated vials. Chem. Cent. J. 2008, 2, 1–10. [Google Scholar] [CrossRef]
- Victoria Otton, S.; deBruyn, A.M.H.; Meloche, L.M.; Gobas, F.A.P.C.; Ikonomou, M.G. Assessing Exposure of Sediment Biota To Organic Contaminants By Thin-Film Solid Phase Extraction. Environ. Toxicol. Chem. 2008, 28, 247. [Google Scholar]
- St. George, T.; Vlahos, P.; Harner, T.; Helm, P.; Wilford, B. A rapidly equilibrating, thin film, passive water sampler for organic contaminants; Characterization and field testing. Environ. Pollut. 2011, 159, 481–486. [Google Scholar] [CrossRef]
- Bragg, L.; Qin, Z.; Alaee, M.; Pawliszyn, J. Field sampling with a polydimethylsiloxane thin-film. J. Chromatogr. Sci. 2006, 44, 317–323. [Google Scholar] [CrossRef]
- Godage, N.H.; Gionfriddo, E. A critical outlook on recent developments and applications of matrix compatible coatings for solid phase microextraction. TrAC—Trends Anal. Chem. 2019, 111, 220–228. [Google Scholar] [CrossRef]
- Guerra, P.; Lai, H.; Almirall, J.R. Analysis of the volatile chemical markers of explosives using novel solid phase microextraction coupled to ion mobility spectrometry. J. Sep. Sci. 2008, 31, 2891–2898. [Google Scholar] [CrossRef]
- Bessonneau, V.; Boyaci, E.; Maciazek-Jurczyk, M.; Pawliszyn, J. In vivo solid phase microextraction sampling of human saliva for non-invasive and on-site monitoring. Anal. Chim. Acta 2015, 856, 35–45. [Google Scholar] [CrossRef]
- Li, S.; Wu, D.; Yan, X.; Guan, Y. Acetone-activated polyimide electrospun nanofiber membrane for thin-film microextraction and thermal desorption-gas chromatography-mass spectrometric analysis of phenols in environmental water. J. Chromatogr. A 2015, 1411, 1–8. [Google Scholar] [CrossRef]
- Ahmadi, F.; Sparham, C.; Boyacl, E.; Pawliszyn, J. Time Weighted Average Concentration Monitoring Based on Thin Film Solid Phase Microextraction. Environ. Sci. Technol. 2017, 51, 3929–3937. [Google Scholar] [CrossRef]
- Piri-Moghadam, H.; Gionfriddo, E.; Grandy, J.J.; Alam, M.N.; Pawliszyn, J. Development and validation of eco-friendly strategies based on thin film microextraction for water analysis. J. Chromatogr. A 2018, 1579, 20–30. [Google Scholar] [CrossRef]
- Mirnaghi, F.S.; Pawliszyn, J. Development of coatings for automated 96-blade solid phase microextraction-liquid chromatography-tandem mass spectrometry system, capable of extracting a wide polarity range of analytes from biological fluids. J. Chromatogr. A 2012, 1261, 91–98. [Google Scholar] [CrossRef]
- Kim, K.H.; Jahan, S.A.; Kabir, E.; Brown, R.J.C. A review of airborne polycyclic aromatic hydrocarbons (PAHs) and their human health effects. Environ. Int. 2013, 60, 71–80. [Google Scholar] [CrossRef]
- Qin, Z.; Mok, S.; Ouyang, G.; Dixon, D.G.; Pawliszyn, J. Partitioning and accumulation rates of polycyclic aromatic hydrocarbons into polydimethylsiloxane thin films and black worms from aqueous samples. Anal. Chim. Acta 2010, 667, 71–76. [Google Scholar] [CrossRef]
- Qin, Z.; Bragg, L.; Ouyang, G.; Niri, V.H.; Pawliszyn, J. Solid-phase microextraction under controlled agitation conditions for rapid on-site sampling of organic pollutants in water. J. Chromatogr. A 2009, 1216, 6979–6985. [Google Scholar] [CrossRef]
- Xu, C.; Wang, J.; Richards, J.; Xu, T.; Liu, W.; Gan, J. Development of film-based passive samplers for in situ monitoring of trace levels of pyrethroids in sediment. Environ. Pollut. 2018, 242, 1684–1692. [Google Scholar] [CrossRef]
- Sisalli, S.; Adao, A.; Lebel, M.; Le Fur, I.; Sandra, P. Sorptive Tape Extraction—A Novel Sampling Method for the in vivo Study of Skin. LC-GC Eur. 2006, 19, 33–39. [Google Scholar]
- Bicchi, C.; Cordero, C.; Liberto, E.; Rubiolo, P.; Sgorbini, B.; Sandra, P. Sorptive tape extraction in the analysis of the volatile fraction emitted from biological solid matrices. J. Chromatogr. A 2007, 1148, 137–144. [Google Scholar] [CrossRef]
- Mohammadi, V.; Jafari, M.T.; Saraji, M. Flexible/self-supported zeolitic imidazolate framework-67 film as an adsorbent for thin-film microextraction. Microchem. J. 2019, 146, 98–105. [Google Scholar] [CrossRef]
- Wei, F.; Zhang, F.F.; Liao, H.; Dong, X.Y.; Li, Y.H.; Chen, H. Preparation of novel polydimethylsiloxane solid-phase microextraction film and its application in liquid sample pretreatment. J. Sep. Sci. 2011, 34, 331–339. [Google Scholar] [CrossRef]
- Jahnke, A.; Mayer, P.; Broman, D.; McLachlan, M.S. Possibilities and limitations of equilibrium sampling using polydimethylsiloxane in fish tissue. Chemosphere 2009, 77, 764–770. [Google Scholar] [CrossRef]
- Stuff, J.R.; Whitecavage, J.A.; Grandy, J.J.; Pawliszyn, J. Analysis of Beverage Samples using Thin Film Solid Phase Microextraction (TF-SPME) and Thermal Desorption GC/MS. 2018, 1–10. Available online: http://www.gerstel.com/pdf/AppNote-200.pdf (accessed on 30 July 2019).
- Shigeyama, H.; Wang, T.; Ichinose, M.; Ansai, T.; Lee, S.W. Identification of volatile metabolites in human saliva from patients with oral squamous cell carcinoma via zeolite-based thin-film microextraction coupled with GC–MS. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2019, 1104, 49–58. [Google Scholar] [CrossRef]
- Rodriguez-Lafuente, A.; Piri-Moghadam, H.; Lord, H.L.; Obal, T.; Pawliszyn, J. Inter-laboratory validation of automated SPME-GC/MS for determination of pesticides in surface and ground water samples: Sensitive and green alternative to liquid–liquid extraction. Water Qual. Res. J. Canada 2016, 51, 331–343. [Google Scholar] [CrossRef]
- Jastrzembski, J.A.; Bee, M.Y.; Sacks, G.L. Trace-Level Volatile Quantitation by Direct Analysis in Real Time Mass Spectrometry following Headspace Extraction: Optimization and Validation in Grapes. J. Agric. Food Chem. 2017, 65, 9353–9359. [Google Scholar] [CrossRef]
- Armenta, S.; Garrigues, S.; de la Guardia, M. Green Analytical Chemistry. TrAC Trends Anal. Chem. 2008, 27, 497–511. [Google Scholar] [CrossRef]


| Application | TDU Split | Cryo-Trap Split | Rationale |
|---|---|---|---|
| Ultra-trace analysis | Splitless | Solvent-vent 1 | Maximum amount of analyte injected |
| High concentrations | Splitless | Solvent-vent | Sensitivity/less column load |
| Unknown analysis | Split | Split | Clean cryo-trap/column |
| Watery/fouled devices | Split | Solvent-vent 1 | High sensitivity/clean cryo-trap |
| Extraction Phase Chemistry | Sample | Targeted Analytes | Instrumentation | Coating Method | Ref. |
|---|---|---|---|---|---|
| Environmental Analysis | |||||
| PDMS | Environmental waters | PAHs | GC-MS | N/A | [12] |
| PDMS | Environmental waters | PAHs | GC-MS | N/A | [16] |
| PDMS | Environmental waters | PAHs | GC-MS | N/A | [49] |
| PDMS | Deionized water | PAHs | GC-MS | N/A | [58] |
| PDMS | Environmental waters | PAHs | GC-MS | N/A | [59] |
| Carbonaceous Nanomaterials | Wastewater | PAHs | GC-MS | Evaporation | [20] |
| EVA and LDPE | Air | PAHs | GC-MS | Dipping | [45] |
| Polyacrylate | Environmental waters | PAHs | GC-MS | Dispersion | [32] |
| PDMS | Soil | PAHs | HPLC-FLD | Dispersion | [46] |
| EVA | Environmental waters | Pesticides and PCBs | GC-MS | Dip coating | [48] |
| EVA | Air | PCBs | GC-MS | Dispersion | [44] |
| Various monophasic polymers | Soil | Pyrethroids | GC-MS | N/A | [60] |
| PDMS | Human skin | Sebum constituents (squalene/fatty acids) | GC-MS | N/A | [61] |
| PDMS | Plant Tissue | Plant volatiles | GC-MS | N/A | [33] |
| PDMS | Plants/Perfumes | Plant volatiles/perfumes | GC-MS | N/A | [62] |
| DVB/PDMS | Air | Benzene | GC-MS | Bar coating | [36] |
| Polyacrylonitrile | Environmental waters | UV filters/biocides | LC-MS | Spray coating | [54] |
| DVB/PDMS | Environmental waters | Toluene/xylene/pesticides | GC-MS | Bar coating | [19] |
| DVB/PDMS | Environmental waters | Pesticides | GC-MS | Bar coating | [55] |
| Car/PDMS | Environmental waters | Crude MCHM constituents | GC-MS | Bar coating | [8] |
| HLB/PDMS | Hot tub water | Chlorination byproducts | GC-MS | Bar coating | [30] |
| Graphene | Various aqueous samples | Trace metals | TXRF | Drop-casting | [28] |
| Graphene | Aqueous | Methylcyclopentadienyl manganese tricarbonyl | TXRF | Drop-casting | [27] |
| Zeolitic imidazolate framework | Agricultural Wastewater | Ethion (insecticide) | IMS | Electrospinning | [63] |
| PDMS | Lake water | Organochlorine pesticides | GC-MS | Bar coating | [64] |
| (HLB, C18, SAX, PS-DVB-WAX, HLB-SAX) | Synthetic Seawater | Fluorinated benzoic acids | LC-MS, GC-MS | Spray coating | [21] |
| PDMS | Fish tissue | Polychlorinated biphenyls | GC-MS | N/A | [65] |
| Flavors and fragrance | |||||
| DVB/PDMS | Beverage | Aroma and flavor components | GC-MS | Bar coating | [66] |
| DVB/PDMS | Grapes | Linalool/IBMP | DART-MS | Dip coating | [69] |
| PDMS | Perfumes and foodstuffs | Volatile compounds | GC-MS | N/A | [62] |
| DVB/PDMS | Food | Aroma and flavor components | GC-MS | Bar coating | [34] |
| PDMS | Grapes | Linalool/IBMP | DART-MS | Dip coating | [38] |
| Other | |||||
| PDMS | Human Skin | Volatile organic compounds | GC-MS | N/A | [35] |
| PDMS | Human Skin | Sebum | GC-MS | N/A | [61] |
| PDMS | Human Saliva | Prohibited substances and endogenous steroids | GC-MS, LC-MS | Bar coating | [52] |
| PDMS/DVB | Beverages and biological fluids | Drugs | GC-MS | Dip coating | [39] |
| ZSM-5/PDMS | Human saliva | Volatile metabolites | GC-MS | Deposition | [67] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Emmons, R.V.; Tajali, R.; Gionfriddo, E. Development, Optimization and Applications of Thin Film Solid Phase Microextraction (TF-SPME) Devices for Thermal Desorption: A Comprehensive Review. Separations 2019, 6, 39. https://doi.org/10.3390/separations6030039
Emmons RV, Tajali R, Gionfriddo E. Development, Optimization and Applications of Thin Film Solid Phase Microextraction (TF-SPME) Devices for Thermal Desorption: A Comprehensive Review. Separations. 2019; 6(3):39. https://doi.org/10.3390/separations6030039
Chicago/Turabian StyleEmmons, Ronald V., Ramin Tajali, and Emanuela Gionfriddo. 2019. "Development, Optimization and Applications of Thin Film Solid Phase Microextraction (TF-SPME) Devices for Thermal Desorption: A Comprehensive Review" Separations 6, no. 3: 39. https://doi.org/10.3390/separations6030039
APA StyleEmmons, R. V., Tajali, R., & Gionfriddo, E. (2019). Development, Optimization and Applications of Thin Film Solid Phase Microextraction (TF-SPME) Devices for Thermal Desorption: A Comprehensive Review. Separations, 6(3), 39. https://doi.org/10.3390/separations6030039






