Gateway of Landfilled Plastic Waste Towards Circular Economy in Europe
Abstract
1. Introduction: Plastic as a Material and Waste
2. Concerns, Challenges and Solutions in Plastic Waste Treatment
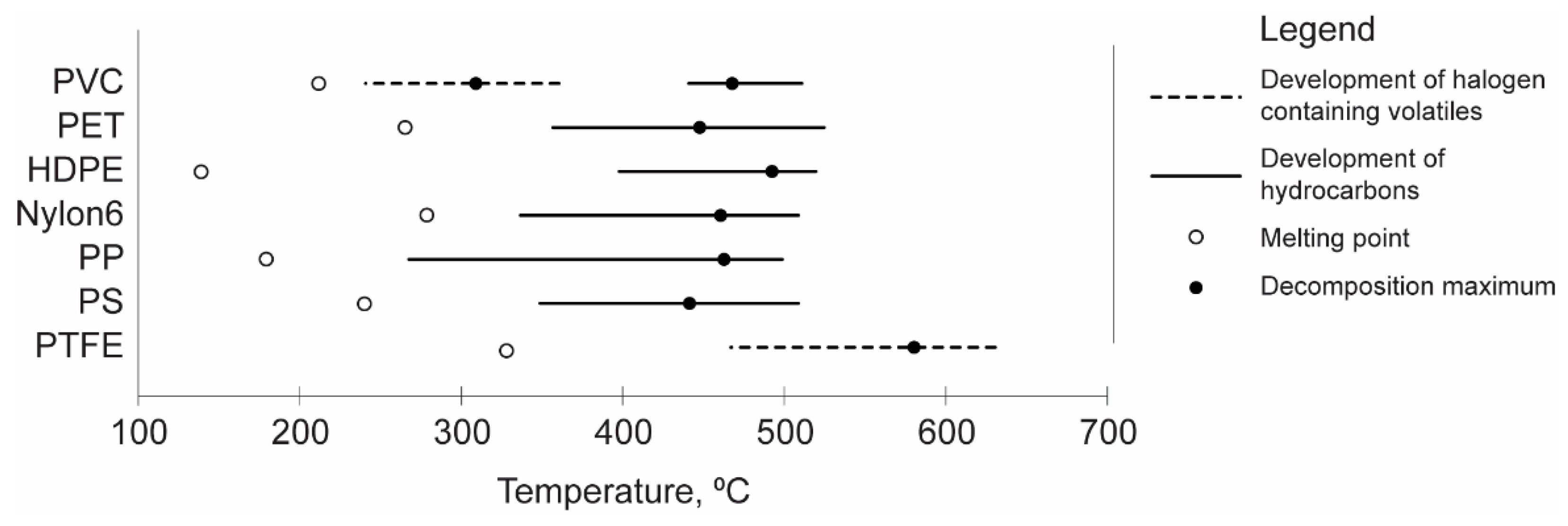
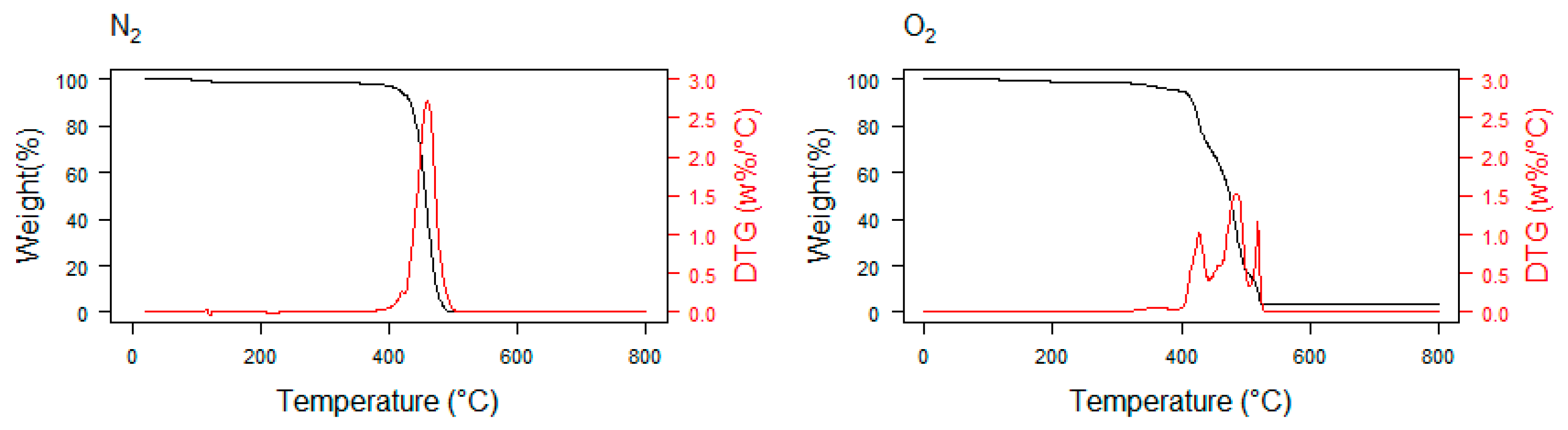
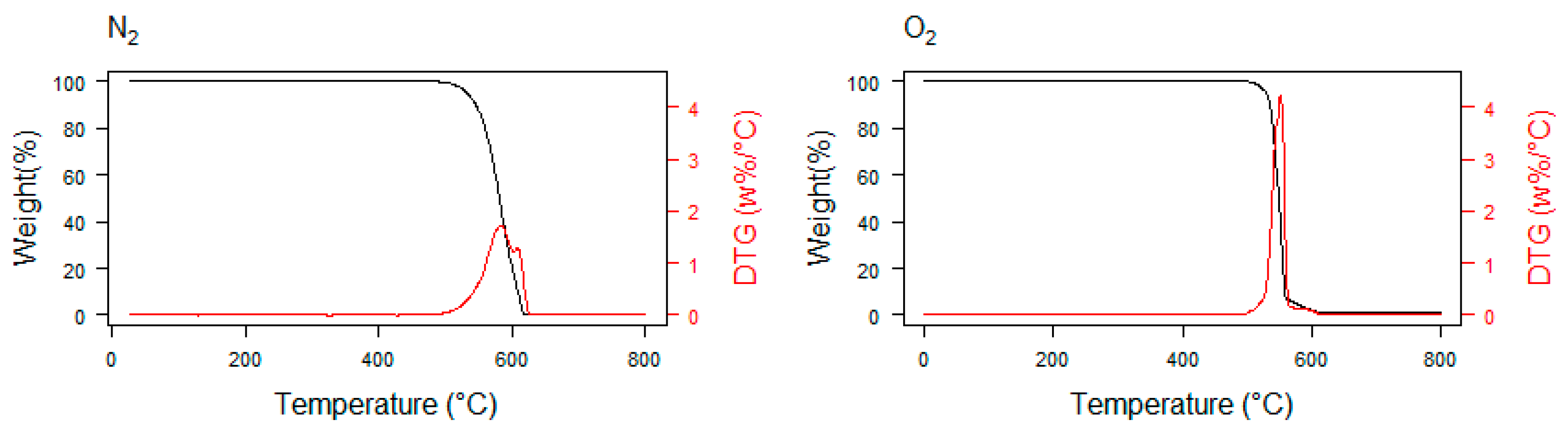
3. Thermogravimetry as a Promising Method in Plastic Waste Decomposition
4. Discussion on Economic Background
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bottero, J.Y.; Auffan, M.; Borschnek, D.; Chaurand, P.; Labille, J.; Levard, C.; Masion, A.; Tella, M.; Rose, J.; Wiesner, M.R. Nanotechnology, global development in the frame of environmental risk forecasting. A necessity of interdisciplinary researches. C. R. Geosci. 2015, 347, 35–42. [Google Scholar] [CrossRef]
- New Scientists. The Daily Newspaper. 28 January 2015. Plastic Age: How It’s Reshaping Rocks, Oceans and Life. By Christina Reed. Available online: https://www.newscientist.com/article/mg22530060-200-plastic-age-how-its-reshaping-rocks-oceans-and-life/ (accessed on 21 January 2019).
- Burlakovs, J.; Jani, Y.; Kriipsalu, M.; Vincevica-Gaile, Z.; Kaczala, F.; Celma, G.; Ozola, R.; Rozina, L.; Rudovica, V.; Hogland, M.; et al. On the way to ‘Zero Waste’ management: Recovery potential of elements, including rare earth elements, from fine fraction of waste. J. Clean. Prod. 2018, 186, 81–90. [Google Scholar] [CrossRef]
- Horvath, B.; Mallinguh, E.; Fogarassy, C. Designing business solutions for plastic waste management to enhance circular transitions in Kenya. Sustainability 2018, 10, 1664. [Google Scholar] [CrossRef]
- Santibañez-Aguilar, J.E.; Ponce-Ortega, J.M.; Betzabe González-Campos, J.; Serna- González, M.; El-Halwagi, M.M. Optimal planning for the sustainable utilization of municipal solid waste. Waste Manag. 2013, 33, 2607–2622. [Google Scholar] [CrossRef] [PubMed]
- Brunner, P.H.; Recberger, H. Waste to energy—Key element for sustainable waste management. Waste Manag. 2015, 37, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Giannis, A.; Lam, W.Y.; Lin, S.X.; Yin, K.; Yuan, G.A.; Wang, J.Y. Characterization of Singapore RDF resources and analysis of their heating value. Sustain. Environ. Res. 2016, 26, 51–54. [Google Scholar] [CrossRef]
- Gug, J.; Cacciola, D.; Sobkowicz, M.J. Processing and properties of a solid energy fuel from municipal solid waste (MSW) and recycled plastics. Waste Manag. 2015, 35, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Othman, S.N.; Noor, Z.Z.; Abba, A.H.; Yusuf, R.O.; Hassan, M.A.A. Review on life cycle assessment of integrated solid waste management in some Asian countries. J. Clean. Prod. 2013, 41, 251–262. [Google Scholar] [CrossRef]
- Buekens, A. Introduction to Feedstock Recycling of Plastics; Feedstock Recycling and Pyrolysis of Waste Plastics; Wiley Series: Hoboken, NJ, USA, 2006; pp. 3–41. [Google Scholar]
- Andrady, A.L. The plastic in microplastics: A review. Mar. Pollut. Bull. 2017, 119, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Al-Salem, S.M.; Evangelisti, S.; Lettieri, P. Life cycle assessment of alternative technologies for municipal solid waste and plastic solid waste management in the Greater London area. Chem. Eng. J. 2014, 244, 391–402. [Google Scholar] [CrossRef]
- Shonfield, P. LCA of Management Options for Mixed Waste Plastics; WRAP: Banbury, UK, 2008. [Google Scholar]
- Klavins, M.; Bisters, V.; Burlakovs, J. Small scale gasification application and perspectives in circular economy. Environ. Clim. Technol. 2018, 22, 42–54. [Google Scholar] [CrossRef]
- Porshnov, D.; Ozols, V.; Ansone-Bertina, L.; Burlakovs, J.; Klavins, M. Thermal decomposition study of major refuse derived fuel components. Energy Procedia 2018, 147, 48–53. [Google Scholar] [CrossRef]
- Porshnov, D.; Ansone-Bertina, L.; Ozols, V.; Burlakovs, J.; Klavins, M. Thermogravimetric study of municipal waste plastics considering perspectives of waste pyro-gasification. SGEM Renew. Energy Sources Clean Technol. 2018. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Kaczala, F.; Burlakovs, J.; Kriipsalu, M.; Hogland, M.; Hogland, W. Hunting for valuables from landfills and assessing their market opportunities—A case study with Kudjape landfill in Estonia. Waste Manag. Res. 2017, 35, 627–635. [Google Scholar] [CrossRef]
- Hogland, M.; Arina, D.; Kriipsalu, M.; Jani, Y.; Kaczala, F.; Salomão, A.L.; Orupõld, K.; Pehme, K.M.; Rudovica, V.; Denafas, G.; et al. Remarks on four novel landfill mining case studies in Estonia and Sweden. J. Mater. Cycles Waste 2018, 20, 1355–1363. [Google Scholar] [CrossRef]
- Burlakovs, J.; Kriipsalu, M.; Klavins, M.; Bhatnagar, A.; Vincevica-Gaile, Z.; Stenis, J.; Jani, Y.; Mykhaylenko, V.; Denafas, G.; Turkadze, T.; et al. Paradigms on landfill mining: From dump site scavenging to ecosystem services revitalization. Resour. Conserv. Recycl. 2016, 123, 73–84. [Google Scholar] [CrossRef]
- Jani, Y.; Kaczala, F.; Marchand, C.; Hogland, M.; Kriipsalu, M.; Hogland, W.; Kihl, A. Characterization of mined fine fraction and waste composition from a Swedish landfill. Waste Manag. Res. 2016, 34, 1292–1299. [Google Scholar] [CrossRef]
- Hogland, W. Remediation of an old landfill site—soil analysis, leachate quality and gas production. Environ. Sci. Pollut. Res. 2002, 1, 49–54. [Google Scholar] [CrossRef]
- Kaartinen, T.; Sormunen, K.; Rintala, J. Case study on sampling, processing and characterization of landfilled municipal solid waste in the view of landfill mining. J. Clean. Prod. 2013, 55, 56–66. [Google Scholar] [CrossRef]
- Hull, R.M.; Krogmann, U.; Asce, M.; Storm, F. Composition and characteristics of excavated materials from New Jersey landfill. J. Environ. Eng. 2005, 3, 478–490. [Google Scholar] [CrossRef]
- Sormunen, K.; Ettala, M.; Rintala, J. Detailed internal characterization of twoFinish landfills by waste sampling. Waste Manag. 2008, 28, 151–163. [Google Scholar] [CrossRef]
- Denafas, G.; Bucinskas, A. Feasibilities and perspectives for landfill mining in Lithuania. In Proceedings of the Linnaeus Eco-Tech’2014, Sweden, Kalmar, 24–26 November 2015. [Google Scholar]
- Hogland, W.; Marques, M.; Nimmermark, S. Landfill mining and waste characterization: A strategy for remediation of contaminated areas. J. Mater. Cycles Waste 2004, 6, 119–124. [Google Scholar] [CrossRef]
- Zuberi, M.S.; Ali, S.F. Greenhouse effect reduction by recovering energy from waste landfills in Pakistan. Renew. Sustain. Energy Rev. 2015, 44, 117–131. [Google Scholar] [CrossRef]
- Burlakovs, J.; Pehme, K.-M.; Anne, O.; Kriipsalu, M.; Hogland, W. Remarks on novel case studies for integrated pollution prevention in Baltic Sea Region. SGEM Mar. Ocean Ecosyst. 2018. [Google Scholar] [CrossRef]
- Klaine, S.J.; Koelmans, A.A.; Horne, N.; Carley, S.; Handy, R.D.; Kapustka, L.; Nowack, B.; von der Kammer, F. Paradigms to assess the environmental impact of manufactured nanomaterials. Environ. Toxicol. Chem. 2012, 31, 3–14. [Google Scholar] [CrossRef]
- Tolaymat, T.; El Badawy, A.; Sequeira, R.; Genaidy, A. An integrated science-based methodology to assess potential risks and implications of engineered nanomaterials. J. Hazard. Mater. 2015, 298, 270–281. [Google Scholar] [CrossRef]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sc. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef]
- Lopez-Urionabarrenechea, A.; de Marco, I.; Caballero, B.M.; Laresgoiti, M.F.; Adrados, A. Upgrading of chlorinated oils coming from pyrolysis of plastic waste. Fuel Proc. Technol. 2015, 137, 229–239. [Google Scholar] [CrossRef]
- Buekens, A.; Cen, K. Waste incineration, PVC, and dioxins. J. Mater. Cycles Waste 2011, 13, 190–197. [Google Scholar] [CrossRef]
- Lim, X. Tainted water: The scientists tracing thousands of fluorinated chemicals in our environment. Nature 2019, 566, 26–29. [Google Scholar] [CrossRef]
- Porshnov, D.; Ozols, V.; Klavins, M. Thermogravimetric analysis as express tool for quality assessment of refuse derived fuels used for pyro-gasification. Environ. Technol. 2019. [Google Scholar] [CrossRef]
- Kannan, P.; Al Shoaibi, A.; Srinivasakannan, C. Energy recovery from co-gasification of waste polyethylene and polyethylene terephthalate blends. Comput. Fluids 2013, 88, 38–42. [Google Scholar] [CrossRef]
- Wu, C.; Williams, P.T. Pyrolysis-gasification of post-consumer municipal solid plastic waste for hydrogen production. Int. J. Hydrogen Energy 2010, 35, 949–957. [Google Scholar] [CrossRef]
- Ahmed, I.I.; Gupta, A.K. Hydrogen production from polystyrene pyrolysis and gasification: Characteristics and kinetics. Int. J. Hydrogen Energy 2009, 34, 6253–6264. [Google Scholar] [CrossRef]
- He, M.; Xiao, B.; Hu, Z.; Liu, S.; Guo, X.; Luo, S. Syngas production from catalytic gasification of waste polyethylene: Influence of temperature on gas yield and composition. Int. J. Hydrogen Energy 2009, 34, 1342–1348. [Google Scholar] [CrossRef]
- Williams, P.T.; Slaney, E. Analysis of products from the pyrolysis and liquefaction of single plastics and waste plastic mixtures. Resour. Conserv. Recycl. 2007, 51, 754–769. [Google Scholar] [CrossRef]
- Dunnu, G.; Maier, J.; Scheffknecht, G. Ash fusibility and compositional data of solid recovered fuels. Fuel 2010, 89, 1534–1540. [Google Scholar] [CrossRef]
- Sever, A.S.; Atimtay, A.; Sanin, F.D. Comparison of fuel value and combustion characteristics of two different RDF samples. Waste Manag. 2016, 46, 217–224. [Google Scholar] [CrossRef]
- Medic-Pejic, L.; Fernandes-Anez, N.; Rubio-Arrieta, L.; Garcia-Torrent, J. Thermal behaviour of organic solid recovered fuels (SRF). Int. J. Hydrogen Energy 2016, 41, 16556–16565. [Google Scholar] [CrossRef]
- Beyler, C.L.; Hirschler, M.M. Thermal Decomposition of Polymers; SFPE Handbook of Fire Protection Engineering; Elsevier: Amsterdam, The Netherlands, 2002; pp. 110–131. [Google Scholar]
- Zhou, C.; Fang, W.; Xu, W.; Cao, A.; Wang, R. Characteristics and the recovery potential of plastic wastes obtained from landfill mining. J. Clean. Prod. 2014, 80, 80–86. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burlakovs, J.; Kriipsalu, M.; Porshnov, D.; Jani, Y.; Ozols, V.; Pehme, K.-M.; Rudovica, V.; Grinfelde, I.; Pilecka, J.; Vincevica-Gaile, Z.; et al. Gateway of Landfilled Plastic Waste Towards Circular Economy in Europe. Separations 2019, 6, 25. https://doi.org/10.3390/separations6020025
Burlakovs J, Kriipsalu M, Porshnov D, Jani Y, Ozols V, Pehme K-M, Rudovica V, Grinfelde I, Pilecka J, Vincevica-Gaile Z, et al. Gateway of Landfilled Plastic Waste Towards Circular Economy in Europe. Separations. 2019; 6(2):25. https://doi.org/10.3390/separations6020025
Chicago/Turabian StyleBurlakovs, Juris, Mait Kriipsalu, Dmitry Porshnov, Yahya Jani, Viesturs Ozols, Kaur-Mikk Pehme, Vita Rudovica, Inga Grinfelde, Jovita Pilecka, Zane Vincevica-Gaile, and et al. 2019. "Gateway of Landfilled Plastic Waste Towards Circular Economy in Europe" Separations 6, no. 2: 25. https://doi.org/10.3390/separations6020025
APA StyleBurlakovs, J., Kriipsalu, M., Porshnov, D., Jani, Y., Ozols, V., Pehme, K.-M., Rudovica, V., Grinfelde, I., Pilecka, J., Vincevica-Gaile, Z., Turkadze, T., Hogland, W., & Klavins, M. (2019). Gateway of Landfilled Plastic Waste Towards Circular Economy in Europe. Separations, 6(2), 25. https://doi.org/10.3390/separations6020025







