Design of a Molecularly Imprinted Stir-Bar for Isolation of Patulin in Apple and LC-MS/MS Detection
Abstract
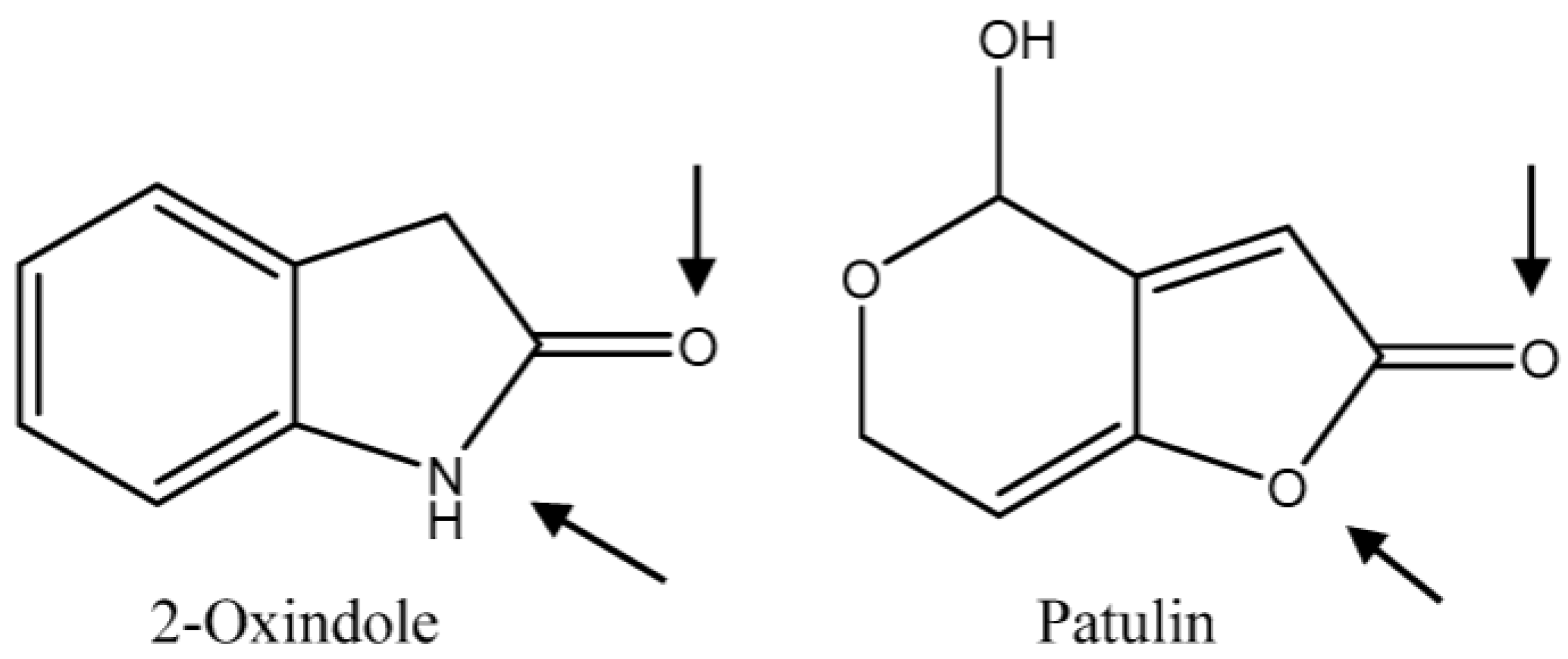
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Apparatus
2.3. Design of Molecularly Imprinted Stir-Bars (MMISB) for Patulin Extraction
2.4. Application to Real Samples
3. Results and Discussion
3.1. Molecularly Imprinted Stir-Bars (MMISB) for Patulin Extraction
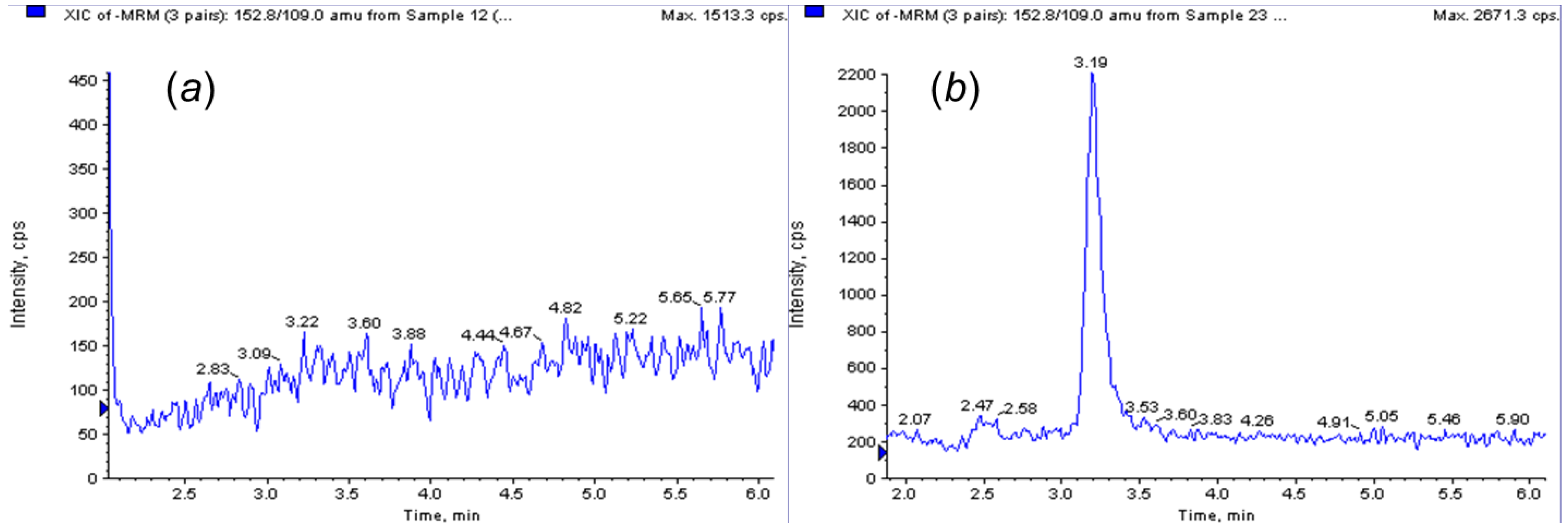
3.2. Analytical Method and Application of MMISB in Apple Samples
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Desmarchelier, A.; Mujahid, C.; Racault, L.; Perring, L.; Lancova, K. Analysis of Patulin in Pear- and Apple-Based Foodstuffs by Liquid Chromatography Electrospray Ionization Tandem Mass Spectrometry. J. Agric. Food Chem. 2011, 59, 7659–7665. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.J.N.D.; Schuch, P.Z.; Bernardi, C.R.; Vainstein, M.H.; Jablonski, A.; Bender, R.J. Patulin in food: State-of-the-art and analytical trends. Rev. Bras. Frutic. 2007, 29, 406–413. [Google Scholar]
- EC Commission Regulation (EC) No. 165/2010 of 26 February 2010 amending Regulation (EC) No. 1881/2006 Setting the Maximum Levels for Certain Contaminants in Foodstuffs as Regards Aflatoxins. Off. J. Eur. Union 2010, 50, 8–11.
- Brause, A.R.; Trucksess, M.W.; Thomas, F.S.; Page, S.W. Determination of patulin in apple juice by liquid chromatography: Collaborative study. J. AOAC Int. 1996, 79, 451–455. [Google Scholar] [PubMed]
- Barreira, M.J.; Alvito, P.C.; Almeida, C.M. Occurrence of patulin in apple-based-foods in Portugal. Food Chem. 2010, 121, 653–658. [Google Scholar] [CrossRef]
- Regal, P.; Diaz-Bao, M.; Barreiro, R.; Cepeda, A.; Fente, C. Application of molecularly imprinted polymers in food analysis: Clean-up and chromatographic improvements. Cent. Eur. J. Chem. 2012, 10, 766–784. [Google Scholar] [CrossRef]
- Díaz-Bao, M.; Regal, P.; Barreiro, R.; Fente, C.A.; Cepeda, A. A facile method for the fabrication of magnetic molecularly imprinted stir-bars: A practical example with aflatoxins in baby foods. J. Chromatogr. A 2016, 1471, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wen, Y.; Ling, Y. Graphene oxide-based magnetic solid phase extraction combined with high performance liquid chromatography for determination of Patulin in Apple Juice. Food Anal. Method 2017, 10, 210–218. [Google Scholar] [CrossRef]
- Khorrami, A.; Taherkhani, M. Synthesis and Evaluation of a Molecularly Imprinted Polymer for Pre-concentration of Patulin from Apple Juice. Chromatographia 2011, 73, 151–156. [Google Scholar] [CrossRef]
- Turiel, E.; Martín-Esteban, A. Molecularly imprinted stir bars for selective extraction of thiabendazole in citrus samples. J. Sep. Sci. 2012, 35, 2962–2969. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Bao, M.; Regal, P.; Barreiro, R.; Miranda, J.; Cepeda, A. Magnetic molecularly imprinted stirring bar for isolation of patulin using grafting technique. Int. Electron. Conf. Synth. Org. Chem. 2015, 19, d002. [Google Scholar] [CrossRef]
- Lucci, P.; Moret, S.; Bettin, S.; Conte, L. Selective solid-phase extraction using a molecularly imprinted polymer for the analysis of patulin in apple-based foods. J. Sep. Sci. 2017, 40, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Kong, W.; Zhou, S.; Yin, L.; Wan, L.; Yang, M. Molecularly imprinted polymer-based solid phase clean-up for analysis of ochratoxin A in beer, red wine, and grape juice. J. Sep. Sci. 2013, 36, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Razali, M.; Kim, J.F.; Attfield, M.; Budd, P.M.; Drioli, E.; Lee, Y.M.; Szekely, G. Sustainable wastewater treatment and recycling in membrane manufacturing. Green Chem. 2015, 17, 5196–5205. [Google Scholar] [CrossRef]
- Vasapollo, G.; Del Sole, R.; Mergola, L.; Lazzoi, M.R.; Scardino, A.; Scorrano, S.; Mele, G. Molecularly imprinted polymers: Present and future prospective. Int. J. Mol. Sci. 2011, 12, 5908–5945. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, X.; Lu, W.; Wu, X.; Li, J. Molecular imprinting: Perspectives and applications. Chem. Soc. Rev. 2016, 45, 2137–2211. [Google Scholar] [CrossRef] [PubMed]
- Kupai, J.; Razali, M.; Buyuktiryaki, S.; Kecili, R.; Szekely, G. Long-term stability and reusability of molecularly imprinted polymers. Polym. Chem. 2017, 8, 666–673. [Google Scholar] [CrossRef]
- Şengül, Ü. Comparing determination methods of detection and quantification limits for aflatoxin analysis in hazelnut. J. Food Drug Anal. 2016, 24, 56–62. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Regal, P.; Díaz-Bao, M.; Barreiro, R.; Fente, C.; Cepeda, A. Design of a Molecularly Imprinted Stir-Bar for Isolation of Patulin in Apple and LC-MS/MS Detection. Separations 2017, 4, 11. https://doi.org/10.3390/separations4020011
Regal P, Díaz-Bao M, Barreiro R, Fente C, Cepeda A. Design of a Molecularly Imprinted Stir-Bar for Isolation of Patulin in Apple and LC-MS/MS Detection. Separations. 2017; 4(2):11. https://doi.org/10.3390/separations4020011
Chicago/Turabian StyleRegal, Patricia, Mónica Díaz-Bao, Rocío Barreiro, Cristina Fente, and Alberto Cepeda. 2017. "Design of a Molecularly Imprinted Stir-Bar for Isolation of Patulin in Apple and LC-MS/MS Detection" Separations 4, no. 2: 11. https://doi.org/10.3390/separations4020011
APA StyleRegal, P., Díaz-Bao, M., Barreiro, R., Fente, C., & Cepeda, A. (2017). Design of a Molecularly Imprinted Stir-Bar for Isolation of Patulin in Apple and LC-MS/MS Detection. Separations, 4(2), 11. https://doi.org/10.3390/separations4020011








