Highly Acidic Macro-Porous Cation Exchange Resin D001 for Efficient Separation of Co(II) from Nd(III) and Dy(III) During Rare Earth Recycling
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Material Pretreatment
2.3. Static Adsorption Experiment
2.4. Analytical Methods
2.5. Column Separation Experiment
3. Results and Discussion
3.1. Characterization
3.2. Batch Adsorption Experiment
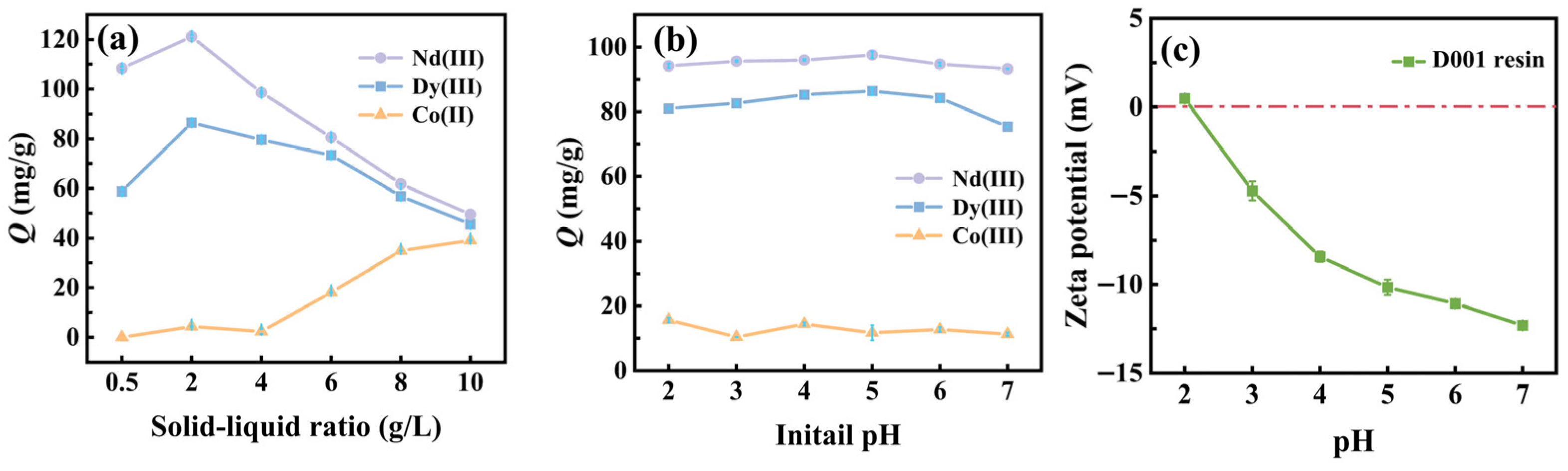
3.2.1. Effect of Solid-Liquid Ratio on Adsorption Capacity
3.2.2. Effect of pH on Adsorption
3.2.3. Adsorption Kinetics Study
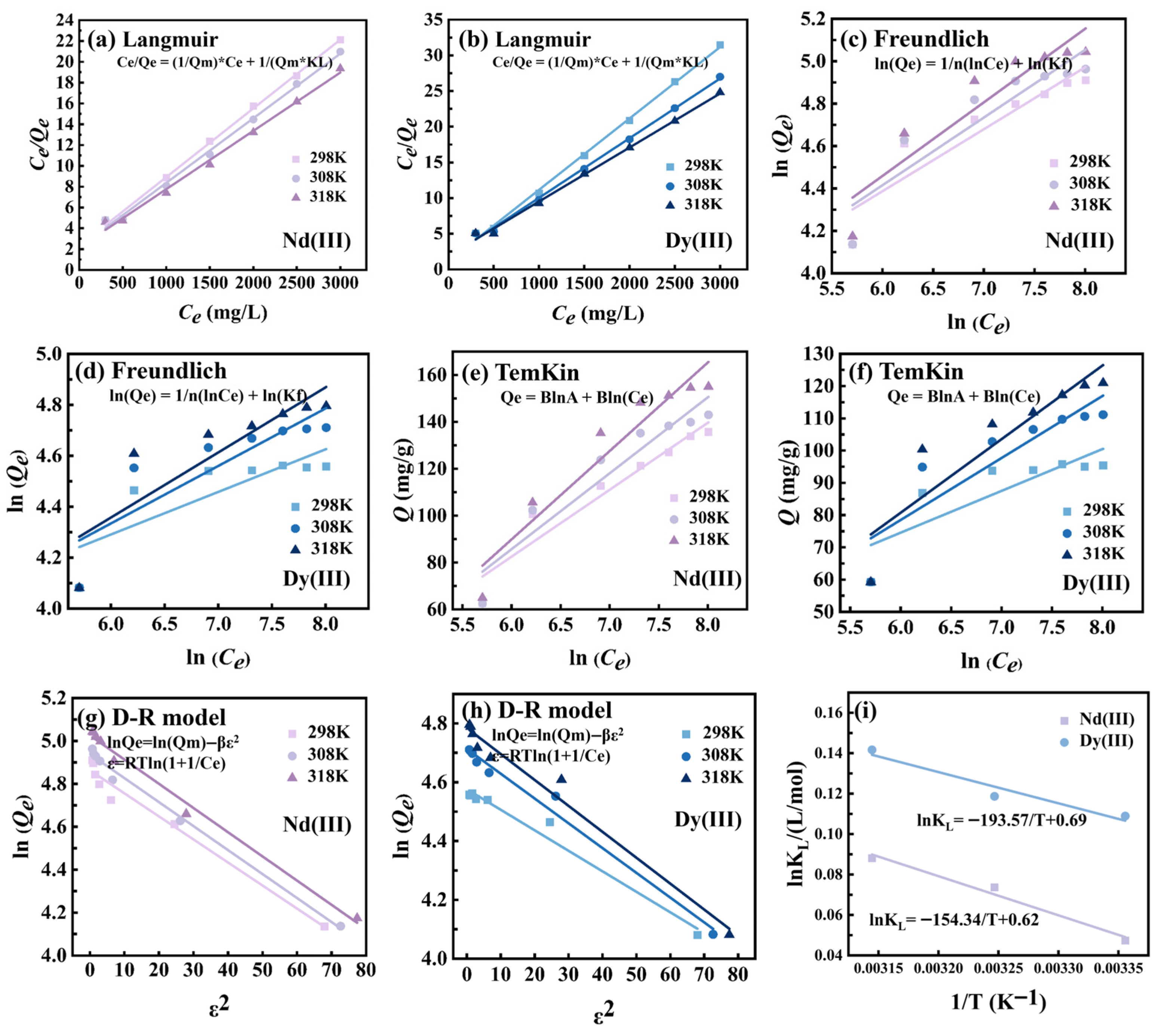
3.2.4. Adsorption Isotherm Study
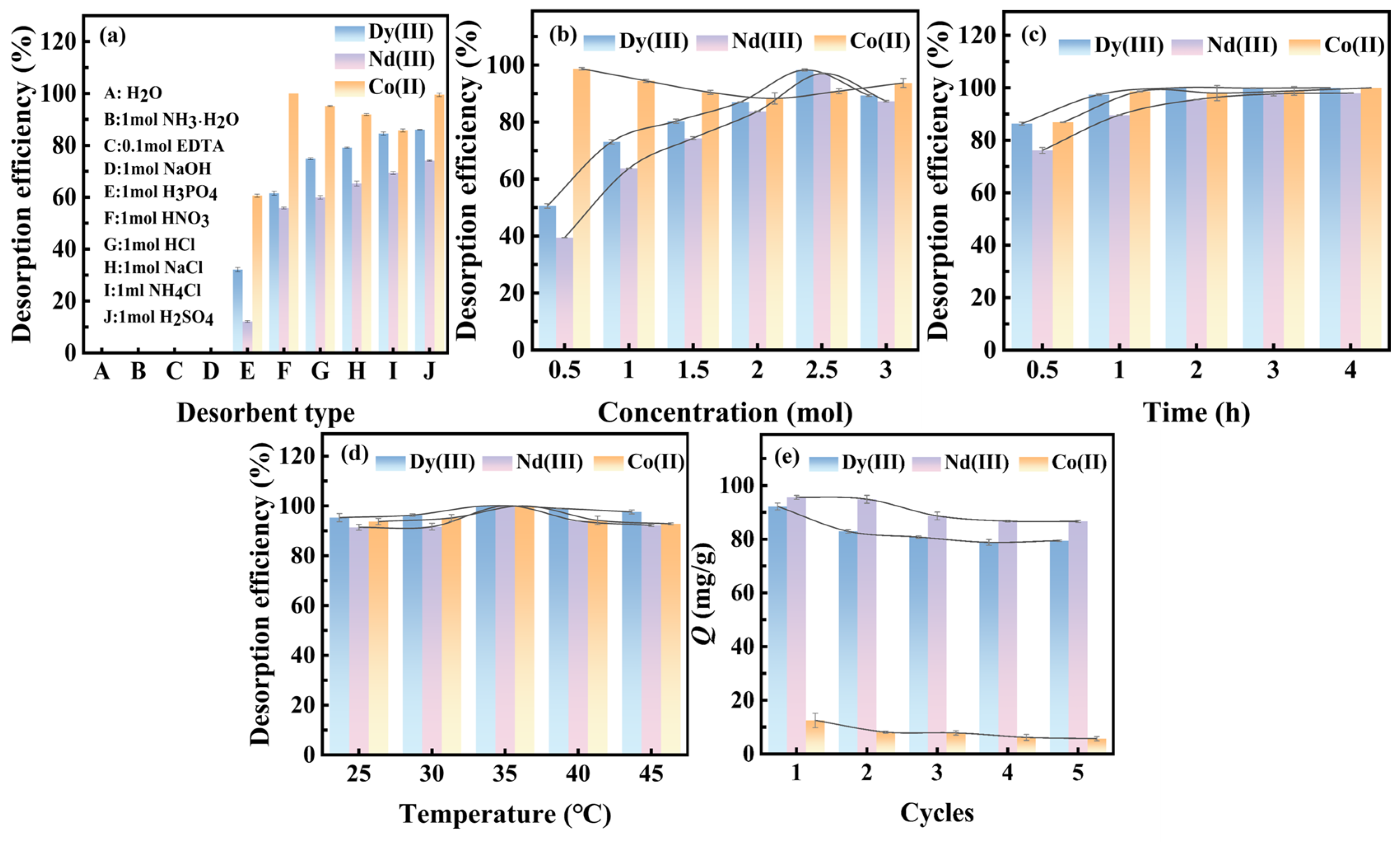
3.2.5. Desorption Performance and Stability of the Resin
3.2.6. Column Experiments
3.2.7. Adsorption Selectivity
3.3. Adsorption Mechanism Study
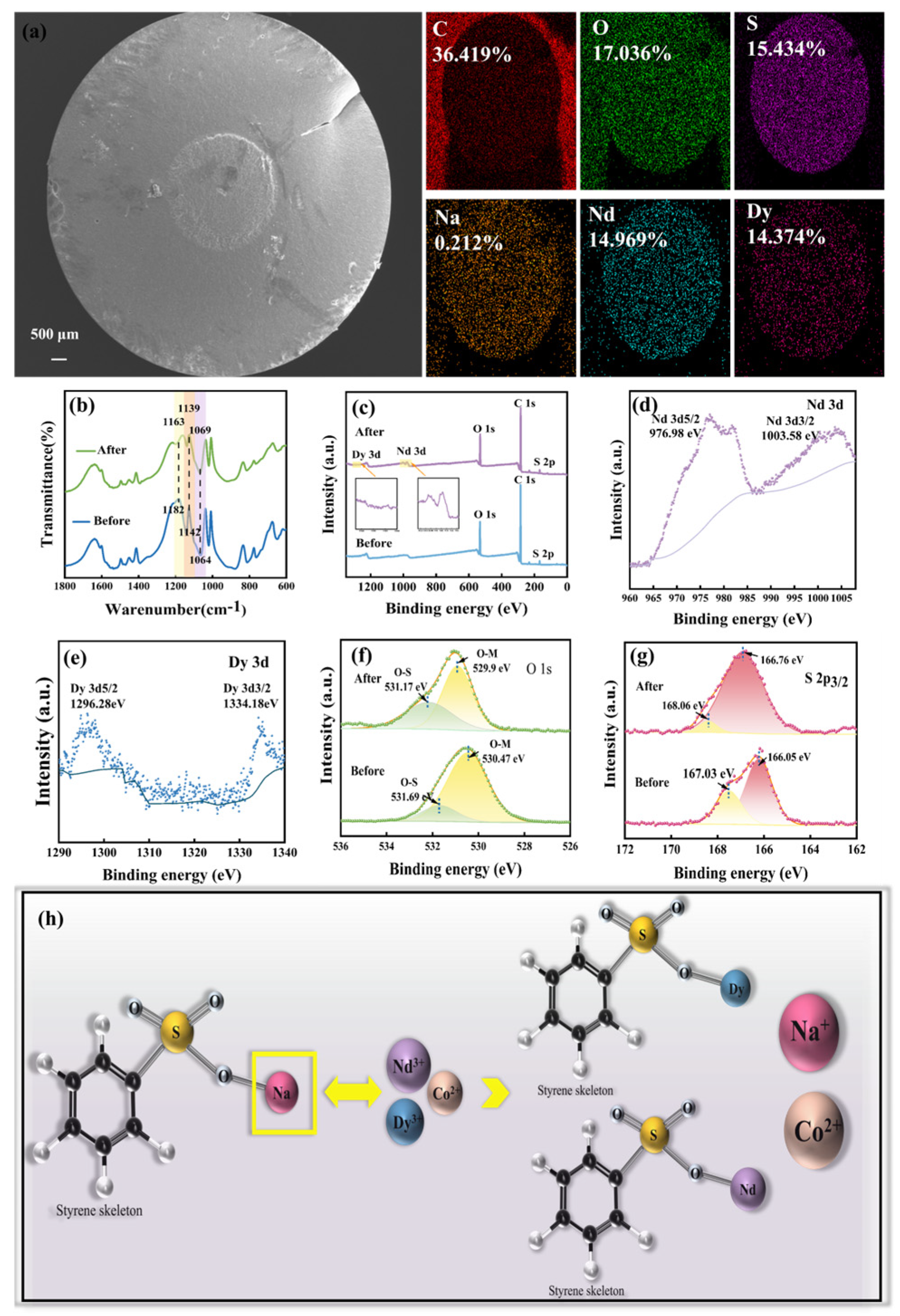
3.3.1. SEM-EDS Analysis
3.3.2. FI-IR Analysis
3.3.3. XPS Analysis
3.3.4. Comparison with Other Reported Adsorbents
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Gutfleisch, O.; Willard, M.A.; Brück, E.; Chen, C.H.; Sankar, S.G.; Liu, J.P. Magnetic materials and devices for the 21st century: Stronger, lighter, and more energy efficient. Adv. Mater. 2011, 23, 821–842. [Google Scholar] [CrossRef]
- Coey, J.M.D. Perspective and prospects for rare earth permanent magnets. Engineering 2020, 6, 119–131. [Google Scholar] [CrossRef]
- München, D.D.; Stein, R.T.; Veit, H.M. Rare earth elements recycling potential estimate based on end-of-life ndfeb permanent magnets from mobile phones and hard disk drives in brazil. Minerals 2021, 11, 1190. [Google Scholar] [CrossRef]
- Li, W.; Yang, L.J.; Zhang, Q.K.; Xu, C.; Zhu, Q.H.; Song, Z.L.; Zheng, B.Z.; Hu, F.Q.; Jiang, J.J. Effect of the grain boundary tb/dy diffused microstructure on the magnetic properties of sintered nd-fe-b magnets. J. Magn. Magn. Mater. 2020, 502, 7. [Google Scholar] [CrossRef]
- Hioki, K. Development of high-performance hot-deformed neodymium–iron–boron magnets without heavy rare-earth elements. Materials 2023, 16, 6581. [Google Scholar] [CrossRef] [PubMed]
- Schäfer, L.; Skokov, K.; Maccari, F.; Radulov, I.; Koch, D.; Mazilkin, A.; Adabifiroozjaei, E.; Molina-Luna, L.; Gutfleisch, O. A novel magnetic hardening mechanism for nd-fe-b permanent magnets based on solid-state phase transformation. Adv. Funct. Mater. 2023, 33, 2208821. [Google Scholar] [CrossRef]
- Shuai, Z.C.; Zhu, Y.M.; Gao, P.; Han, Y.X. Rare earth elements resources and beneficiation: A review. Miner. Eng. 2024, 218, 17. [Google Scholar] [CrossRef]
- Yang, W.Y.; Wu, K.J.; Chen, H.; Huang, J.; Yu, Z. Emerging role of rare earth elements in biomolecular functions. Isme J. 2025, 19, 16. [Google Scholar] [CrossRef]
- Koese, M.; Parzer, M.; Sprecher, B.; Kleijn, R. Self-sufficiency of the european union in critical raw materials for e-mobility. Resour. Conserv. Recycl. 2025, 212, 10. [Google Scholar] [CrossRef]
- Niskanen, J.; Lahtinen, M.; Perämäki, S. Acetic acid leaching of neodymium magnets and iron separation by simple oxidative precipitation. Cleaner Eng. Technol. 2022, 10, 9. [Google Scholar] [CrossRef]
- Jones, K.G.; David, T.; Loula, M.; Matejková, S.; Blahut, J.; Filimonenko, A.; Litecká, M.; Rohlícek, J.; Böserle, J.; Polasek, M. Macrocyclic chelators for aqueous lanthanide separations via precipitation: Toward sustainable recycling of rare-earths from ndfeb magnets. J. Am. Chem. Soc. 2025, 147, 22666–22676. [Google Scholar] [CrossRef] [PubMed]
- Dudarko, O.; Kobylinska, N.; Kessler, V.; Seisenbaeva, G. Recovery of rare earth elements from ndfeb magnet by mono- and bifunctional mesoporous silica: Waste recycling strategies and perspectives. Hydrometallurgy 2022, 210, 14. [Google Scholar] [CrossRef]
- Jung, Y.; Lee, Y.; Yoon, S.J.; Choi, J.W. Synergistic effect of core/shell-structured composite fibers: Efficient recovery of rare-earth elements from spent ndfeb permanent magnets. Adv. Fiber Mater. 2024, 6, 1729–1745. [Google Scholar] [CrossRef]
- Gergoric, M.; Ravaux, C.; Steenari, B.M.; Espegren, F.; Retegan, T. Leaching and recovery of rare-earth elements from neodymium magnet waste using organic acids. Metals 2018, 8, 721. [Google Scholar] [CrossRef]
- Gergoric, M.; Ekberg, C.; Foreman, M.R.S.; Steenari, B.M.; Retegan, T. Characterization and leaching of neodymium magnet waste and solvent extraction of the rare-earth elements using todga. J. Sust. Metall. 2017, 3, 638–645. [Google Scholar] [CrossRef]
- Liu, J.; Zhao, Y.X.; Wang, Z.R.; Jia, M.H.; Xia, W.X.; Wu, G.Z.; Guo, W.D.; Chi, R.A.; Huang, K. Review on micro-mechanism of forming emulsification during rare earth extraction by acidic extractants. J. Rare Earths 2025, 43, 9–20. [Google Scholar] [CrossRef]
- Zeng, B.; Zeng, X.R.; Huang, L.J.H.; Huang, W.F.; Shu, R.H. Adsorption properties of a polyamine special ion exchange resin for removing molybdenum from ammonium tungstate solutions. Sustainability 2023, 15, 3837. [Google Scholar] [CrossRef]
- Nogueira, M.; Matos, I.; Bernardo, M.; Pinto, F.; Fonseca, I.; Lapa, N. Recovery of nd3+ and dy3+ from e-waste using adsorbents from spent tyre rubbers: Batch and column dynamic assays. Molecules 2025, 30, 92. [Google Scholar] [CrossRef]
- Zhong, Y.L.; Ning, S.Y.; Wu, K.; Li, Z.Y.; Wang, X.P.; He, C.L.; Fujita, T.; Wang, J.; Chen, L.F.; Yin, X.B.; et al. Novel phosphate functionalized sodium alginate hydrogel for efficient adsorption and separation of nd and dy from co. J. Environ. Manag. 2024, 353, 12. [Google Scholar] [CrossRef]
- Jasim, A.Q.; Ajjam, S.K. Removal of heavy metal ions from wastewater using ion exchange resin in a batch process with kinetic isotherm. S. Afr. J. Chem. Eng. 2024, 49, 43–54. [Google Scholar] [CrossRef]
- Yan, X.F.; Zhen, H.G.; Wang, B.; Yang, L. Selectivity and competitive mechanism of cation exchange resin for fe, mg, and al ions in phosphoric acid. Can. J. Chem. Eng. 2024, 102, 1272–1288. [Google Scholar] [CrossRef]
- El-Din, A.S.M.; Rizk, H.E.; Borai, E.H.; El Afifi, E.M. Selective separation and purification of cerium (iii) from concentrate liquor associated with monazite processing by cationic exchange resin as adsorbent. Chem. Pap. 2023, 77, 2525–2538. [Google Scholar] [CrossRef]
- Hong, H.J.; Yoo, H.; Jeon, J.H.; Kim, H.S.; Lee, J.Y. Differential adsorption of vanadium(v) and tungsten(w) on ion exchange resins: A novel approach for separation and recovery of spent catalyst leachate. J. Clean. Prod. 2023, 426, 11. [Google Scholar] [CrossRef]
- Jiang, X.Z.; Luo, Y.H.; Mu, S.; Meng, B.J.; Wang, W.; Yu, G.; Deng, S.B. Structure-selectivity relationship of anion exchange resins with different quaternary amine functional groups for highly selective removal of pfas from chromium-plating wastewater. Water Res. 2025, 268, 10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.W.; Wu, W.L.; Gao, Y.C.; Zhao, S.H.; Zong, Y.M.; Xue, G.; Song, Y.H.; Ding, L. Efficient removal of nitrophenol by magnetic ion exchange resins: Role of nitrophenol functional groups based on characterisation, dft calculations and site energy distributions. Sep. Purif. Technol. 2025, 354, 15. [Google Scholar] [CrossRef]
- Kovalenko, O.V.; Baulin, V.E.; Shulga, Y.M.; Baulin, D.; Tsivadze, A.Y. Recovery and separation of pr, nd, and dy using solvent impregnated resin from e-wastes. J. Chromatogr. A 2026, 1765, 10. [Google Scholar] [CrossRef]
- Hermassi, M.; Granados, M.; Valderrama, C.; Skoglund, N.; Ayora, C.; Cortina, J.L. Impact of functional group types in ion exchange resins on rare earth element recovery from treated acid mine waters. J. Clean. Prod. 2022, 379, 13. [Google Scholar] [CrossRef]
- Cui, Y.; Gao, S.; Zhang, R.; Cheng, L.; Yu, J. Study on the moisture absorption and thermal properties of hygroscopic exothermic fibers and related interactions with water molecules. Polymers 2020, 12, 98. [Google Scholar] [CrossRef]
- Wang, X.Z.; Zheng, Y.H.; Xue, Y.; Yan, Y.D.; Ma, F.Q.; Zhang, M.L.; Bai, H.Y.; Kou, Z.Q.; Liu, J.P. Study on the destruction process of cationic exchange resins treated by Li2CO3-Na2CO3-K2CO3 molten salt. J. Environ. Chem. Eng. 2021, 9, 10. [Google Scholar] [CrossRef]
- Jankovic, B.; Dodevski, V.; Veljkovic, F.; Jankovic, M.; Manic, N. Application of model-free and model-based kinetic methods in evaluation of reactions complexity during thermo-oxidative degradation process: Case study of 4-(hydroxymethyl)phenoxymethyl polystyrene resin. Fire 2024, 7, 165. [Google Scholar] [CrossRef]
- Shah, K.H.; Shah, N.S.; Khan, G.A.; Sarfraz, S.; Iqbal, J.; Batool, A.; Jwuiyad, A.; Shahida, S.; Han, C.; Wawrzkiewicz, M. The cr(iii) exchange mechanism of macroporous resins: The effect of functionality and chemical matrix, and the statistical verification of ion exchange data. Water 2023, 15, 3655. [Google Scholar] [CrossRef]
- Shimizu, S.; Matubayasi, N. Sorption hysteresis: A statistical thermodynamic fluctuation theory. Langmuir 2024, 40, 11504–11515. [Google Scholar] [CrossRef] [PubMed]
- Otter, D.; Mondal, S.S.; Alrefai, A.; Krätz, L.; Holdt, H.J.; Bart, H.J. Characterization of an isostructural mof series of imidazolate frameworks potsdam by means of sorption experiments with water vapor. Nanomaterials 2021, 11, 1400. [Google Scholar] [CrossRef] [PubMed]
- Babazadeh, M.; Irannezhad, M.; Abolghasemi, H.; Hosseiniyan, S.B.; Ehsani, A. 3d mathematical modeling of external mass transfer effect in high-rate adsorption process. Surf. Interfaces 2022, 29, 11. [Google Scholar] [CrossRef]
- Chiu, H.-H.; Ho, M.-K.; Hsu, T.-E.; Yu, S.-L.; Manjunatha, K.; Cheng, C.-L.; Li, T.-Y.; Chang, C.-K.; Tummala, S.; Ho, Y.-P.; et al. Manipulating and investigating the room-temperature magnetic memory phenomenon: The impact of rare-earth ion doping on nickel oxide nanoparticles. Mater. Today Chem. 2024, 39, 102190. [Google Scholar] [CrossRef]
- Chen, X.; Wang, C.; Li, M.; Zhang, D.; Wei, Y. Preparation of polydentate phosphonate-functionalized adsorbent for selective extraction of rare earth ions in harsh acidic solution. Microchim. Acta 2025, 192, 304. [Google Scholar] [CrossRef]
- Vareda, J.P. On validity, physical meaning, mechanism insights and regression of adsorption kinetic models. J. Mol. Liq. 2023, 376, 14. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Wang, J.L.; Guo, X. Rethinking of the intraparticle diffusion adsorption kinetics model: Interpretation, solving methods and applications. Chemosphere 2022, 309, 14. [Google Scholar] [CrossRef]
- Wei, F.; Jin, S.L.; Yao, C.Y.; Wang, T.H.; Zhu, S.P.; Ma, Y.B.; Qiao, H.; Shan, L.X.; Wang, R.C.; Lian, X.X.; et al. Revealing the combined effect of active sites and intra-particle diffusion on adsorption mechanism of methylene blue on activated red-pulp pomelo peel biochar. Molecules 2023, 28, 4426. [Google Scholar] [CrossRef]
- Mikolajczyk, A.P.; Fortela, D.L.B.; Berry, J.C.; Chirdon, W.M.; Hernandez, R.A.; Gang, D.D.; Zappi, M.E. Evaluating the suitability of linear and nonlinear regression approaches for the langmuir adsorption model as applied toward biomass-based adsorbents: Testing residuals and assessing model validity. Langmuir 2024, 40, 20428–20442. [Google Scholar] [CrossRef]
- Debnath, S.; Das, R. Strong adsorption of cv dye by ni ferrite nanoparticles for waste water purification: Fits well the pseudo second order kinetic and freundlich isotherm model. Ceram. Int. 2023, 49, 16199–16215. [Google Scholar] [CrossRef]
- Mudhoo, A. Unveiling new insights: Revised temkin adsorption isotherm parameters from fresh curve fits in adsorption studies. Chem. Eng. Sci. 2025, 311, 14. [Google Scholar] [CrossRef]
- Hu, Q.L.; Zhang, Z.Y. Application of dubinin-radushkevich isotherm model at the solid/solution interface: A theoretical analysis. J. Mol. Liq. 2019, 277, 646–648. [Google Scholar] [CrossRef]
- Zhou, J.; Zhang, X.Y.; Deng, B.C.; Huang, Y.B.; Liu, X.J.; Ning, S.Y.; Kuang, S.T.; Liao, W.P. Separation and purification of heavy rare earth elements by a silica/polymer-based β-aminophosphonic acid resin from chloride media. Sep. Purif. Technol. 2025, 354, 9. [Google Scholar] [CrossRef]
- Salvestrini, S.; Bollinger, J.C. Revisiting the extended van’t hoff equation: Comments on “highly-efficient nitrogen self-doped biochar for versatile dyes’ removal prepared from soybean cake via a simple dual-templating approach and associated thermodynamics”. J. Clean Prod. 2022, 373, 4. [Google Scholar] [CrossRef]
- Li, B.; Deng, Z.Y.; Lin, X.Y.; Chen, W.T.; Li, P.; Wu, J.H. Improved pb(ii) removal by d001 resin with a facile nanoscale α-feooh modification. J. Environ. Chem. Eng. 2024, 12, 9. [Google Scholar] [CrossRef]
- Tang, L.; Lu, Y.Y.; Yao, L.L.; Cui, P. A highly hydrophilic benzenesulfonic-grafted graphene oxide-based hybrid membrane for ethanol dehydration. RSC Adv. 2020, 10, 20358–20367. [Google Scholar] [CrossRef]
- Dyussembayeva, G.; Jumadilov, T.; Grazulevicius, J.; Mukatayeva, Z.; Khimersen, K.; Totkhuskyzy, B. Selective gold ion sorption from iron-containing solution using an interpolymer system of industrial ion exchangers. Polymers 2025, 17, 2808. [Google Scholar] [CrossRef]
- Bai, Y.; Meng, Y.Y.; Yang, M.; Tian, R.J.; Wang, J.N.; Jiao, B.X.; Pan, H.B.; Gao, J.W.; Wang, Y.H.; Sun, K.X.; et al. Lattice stabilization and strain homogenization in sn-pb bottom subcells enable stable all-perovskite tandems solar cells. Nat. Commun. 2025, 16, 15. [Google Scholar] [CrossRef]
- Zhang, P.; Lo, I.; O’Connor, D.; Pehkonen, S.; Cheng, H.F.; Hou, D.Y. High efficiency removal of methylene blue using sds surface-modified znfe2o4 nanoparticles. J. Colloid Interface Sci. 2017, 508, 39–48. [Google Scholar] [CrossRef]
- Eltaweil, A.S.; Abdelfatah, A.M.; Hosny, M.; Fawzy, M. Novel biogenic synthesis of a ag@biochar nanocomposite as an antimicrobial agent and photocatalyst for methylene blue degradation. ACS Omega 2022, 7, 8046–8059. [Google Scholar] [CrossRef]
- Henning, L.M.; Simon, U.; Abdullayev, A.; Schmidt, B.; Pohl, C.; Guitar, T.N.; Vakifahmetoglu, C.; Meyer, V.; Bekheet, M.F.; Gurlo, A. Effect of fomes fomentarius cultivation conditions on its adsorption performance for anionic and cationic dyes. ACS Omega 2022, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.T.; Wang, W.Q.; Lin, L.T.; Shan, J.; Zhu, J.Y.; Ning, S.P.; Li, H.M.; Wang, X.W.; Lu, D.C. Mnga with multiple enzyme-like properties for acute wound healing by reducing oxidative stress and modulating signaling pathways. Mater. Today Bio 2025, 30, 11. [Google Scholar] [CrossRef] [PubMed]
- Sentürk, M.; Inan, S. Sorption and separation studies of nd(iii) and dy(iii) using amorphous tin(iv) phosphate. Chem. Pap. 2023, 77, 3835–3845. [Google Scholar] [CrossRef]
- Virtanen, E.J.; Kukkonen, E.; Yliharju, J.; Tuomisto, M.; Frimodig, J.; Kinnunen, K.; Lahtinen, E.; Hännien, M.M.; Väisänen, A.; Haukka, M.; et al. Recovery of rare earth elements from mining wastewater with aminomethylphosphonic acid functionalized 3d-printed filters. Sep. Purif. Technol. 2025, 353, 12. [Google Scholar] [CrossRef]





| Element | T (K) | ΔG0 (J/mol) | ΔH0 (J/mol) | ΔS0 (J/mol K) |
|---|---|---|---|---|
| Nd | 298 | −125 | 160.9 | 5.82 |
| 308 | −184 | |||
| 318 | −241 | |||
| Dy | 298 | −253 | 128.3 | 5.14 |
| 308 | −304 | |||
| 318 | −356 |
| Adsorbent | Saturated Adsorption Capacity (mg/g) | Adsorption Kinetics (Equilibrium Time) | Reusability | Applicable Acidity/pH | Selectivity for Co |
|---|---|---|---|---|---|
| D001 resin (This work) | Nd:154.98 (318 K) Dy:120.96 (318 K) | 90 min | Nd: 86.6% Dy: 80% after 5 cycles | pH 2–7 (Wide pH range) | Adsorption efficiency: Nd 99.3% Dy 98.3% Co 26.6% |
| Amorphous tin(IV) phosphate (am-SnP) [55] | Nd: 68.03 Dy: 58.14 | Nd: 180 min Dy: 360 min | Significant decline after 2 cycles | pH 2–6 (optimum: Nd 4, Dy 3) | High selectivity (βNd/Co up to 22.8, βDy/Co up to 18.76) |
| DPPA/CaALG hydrogel [19] | Nd: 162.5 (1 M HCl) Dy: 183.5 (1 M HCl) | 6 h | Dropped sharply to ~50% after 3 cycles | 1 M HCl (strong acidic) | Almost no Co adsorption (SFREE/Co ≥ 13,671) |
| 3D-printed aminophosphonate filter [56] | Nd: 34.6 Dy: 37.8 Y: 14.7 | 40 mL @ 90 mL/h (multi-step sequential) | Affected by Sc/Fe/U accumulation pre-treatment needed | pH 2 aqueous (mining wastewater) | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Li, Y.; An, H.; Jiang, Z.; Tan, H.; He, C. Highly Acidic Macro-Porous Cation Exchange Resin D001 for Efficient Separation of Co(II) from Nd(III) and Dy(III) During Rare Earth Recycling. Separations 2026, 13, 77. https://doi.org/10.3390/separations13030077
Li Y, An H, Jiang Z, Tan H, He C. Highly Acidic Macro-Porous Cation Exchange Resin D001 for Efficient Separation of Co(II) from Nd(III) and Dy(III) During Rare Earth Recycling. Separations. 2026; 13(3):77. https://doi.org/10.3390/separations13030077
Chicago/Turabian StyleLi, Yao, Huilin An, Zezuo Jiang, Haixiang Tan, and Chunlin He. 2026. "Highly Acidic Macro-Porous Cation Exchange Resin D001 for Efficient Separation of Co(II) from Nd(III) and Dy(III) During Rare Earth Recycling" Separations 13, no. 3: 77. https://doi.org/10.3390/separations13030077
APA StyleLi, Y., An, H., Jiang, Z., Tan, H., & He, C. (2026). Highly Acidic Macro-Porous Cation Exchange Resin D001 for Efficient Separation of Co(II) from Nd(III) and Dy(III) During Rare Earth Recycling. Separations, 13(3), 77. https://doi.org/10.3390/separations13030077







