Abstract
Lysergic acid diethylamide (LSD) is a powerful hallucinogen. Its detection is limited by its low dosage; moreover, LSD is rapidly metabolized into 2-oxo-3-hydroxy-LSD (O-H-LSD). In this study we validated two methods for determination of LSD and O-H-LSD in blood. Method #1 consisted in the upgrade of a previously developed procedure for detection of 163 compounds. Method #2 was specific for LSD and O-H-LSD. Analyses were performed through LC-MS/MS by dynamic (#1) and/or MRM mode (#2), in positive ionization. Transitions were: 324→223,208 m/z for LSD; 356→237,222 m/z for O-H-LSD. Validations were performed following the AAFS’s guidelines. Linearity was good for both methods. Sensitivity was in line with previously validated methods with LOQs at 0.0375 (#1) and 0.025 (#2) ng/mL for LSD and 0.01875 (#1) and 0.0125 (#2) ng/mL for O-H-LSD. Bias and %CV always met the acceptance criteria. RRs were >83%, except for O-H-LSD with method #1. The methods were successfully applied to two real cases. Method #1 proved to be useful for screening purposes, while method #2 can represent a sensitive and reliable tool for confirmation procedures.
1. Introduction
Lysergic acid diethylamide (LSD) is a semi-synthetic compound obtained from lysergic acid, a psychoactive substance produced by the Claviceps purpurea, a rye-infesting fungus [1]. LSD was synthesized by Albert Hofmann in 1938, although its psychedelic effects were accidentally discovered just 5 years later. This substance can cause symptoms like visual misperceptions, audiovisual synesthesia, derealization, depersonalization and positive feelings like happiness and trust [2]. There is a limited number of studies concerning pharmacokinetics of LSD in humans, which would be of interest for clinical and forensic toxicology due to widespread consumption of this substance and the current use in therapeutics. LSD is mainly administered orally, but it can be smoked, injected or snorted. Oral ingestion leads to a rapid and complete adsorption in gastrointestinal tract, presenting an oral bioavailability of approximately 71% [3,4]. After an oral application of 100–250 μg of LSD, the effect appears after 30–45 min, reaching its maximum after 1.5–2.5 h and lasts for 9–12 h [5]. However, adsorption of LSD is influenced by the size of the meal, the pH values of the stomach and duodenum, as well as the gastric evacuation rate [6,7]. Intravenous (i.v.) injection is pharmacologically the safest and best reproducible method of administration. The onset is very fast (it takes a few minutes) and the maximum is reached after 1 h (as in peroral administration). Slow i.v. application can take up to 20 min for the first effect to occur. The effects of LSD can last up to 9–10 h. Pharmacokinetic studies in animals showed that 14C-labelled LSD is well absorbed, and there is a quick distribution from plasma into tissues, in particular in the liver, followed by kidney, spleen, brain, muscle and fat tissue. LSD distribution was studied in the mouse, guinea pig, rhesus monkey, rat and cat. In rats, after 3 h, LSD was in a higher percentage in the gut contents and liver, while in the cat, after 90 min, bile, plasma, lung and liver presented the highest percentages. An in vitro study on guinea pigs showed that LSD binds extensively to plasma proteins (65–90%), at plasma concentrations of 0.1 and 20 mg/L [6,7,8,9,10]. Aghajanian and Bing estimated a plasma elimination half-life of 175 min [11].
As with other hallucinogens, LSD interacts with brain 5-HT receptors to produce agonist or partial antagonist effects on serotonin activity. This entails both the presynaptic 5-HT1A and 5-HT1B receptors, as well as the postsynaptic 5-HT2 receptor. The perceived effects of the influence appear after 30–60 min, peak after 1–6 h and last up to 8–12 h. In the 1970s, it became very popular as a recreational drug; however, even if its consumption has decreased, LSD still represents one of the most powerful and dangerous illicit drugs [7,12]. Many studies have focused on its use in psychotherapy [13,14,15]. Indeed, there is some evidence supporting the use of LSD for the treatment of several psychological disorders, such as alcoholism, anxiety and depression, autism spectrum disorder and nicotine addiction [16,17,18,19]. LSD has a short half-life (2.4–3.0 h), it is extensively metabolized, and only 1% of the dose is present as parent drug in the urine [7,20]. The major metabolite is the 2-oxo-3-hydroxy-LSD (O-H-LSD) which can be used as a marker of LSD consumption for its higher concentrations (16–43 times than LSD) and for its wider detection windows (day/hour) [4,7,21]. Different analytical approaches have been described since 1970s, including radioimmunoassay (RIA), high-performance liquid chromatography (HPLC) with fluorescent detection (FLD) following immunoaffinity extraction (IAE) and capillary electrophoresis (CE) with laser-induced fluorescence detection (LIF) [22,23,24]. Currently, several methods employing HPLC or gas chromatography (GC) coupled to mass spectrometry (single, MS, or tandem, MS/MS) are commonly used [25,26,27,28,29,30]. However, LSD detection by GC–MS is limited by the non-volatile nature of LSD and its low thermostability. Moreover, GC-MS procedures are hampered by long sample treatments, often including a derivatization step. Thus, LC-MS/MS may be considered as the method of choice for LSD determination in biological samples [29,30,31,32,33,34,35]. However, these procedures are focused only on LSD and its main metabolites, and often require the analysis of high sample volumes (>500 µL). To the best of our knowledge, in the literature there is only a multi-analyte method that allows the simultaneous detection of new psychoactive substances (NPS) and LSD [36]. This could represent a limitation of LSD detection since modern forensic toxicology is increasingly focused toward wide-broad analysis in order to detect, and quantify, a high number and variety of compounds. Indeed, many LC-MS/MS screening methods have been published in last years, especially when the new psychoactive substances (NPS) phenomenon arose. These methods allow one to perform highly specific screening, overcoming the limits of classical screening techniques (i.e., immunoassays). In this work, two methods for LSD and O-H-LSD detection in whole blood were validated. The first one is a multi-analytes procedure allowing one to screen for almost 200 substances of forensic toxicological interest (i.e., benzodiazepines, amphetamines, synthetic cannabinoids, synthetic cathinones, opioids, fentanyl derivatives and other NPS). In this way, we aim to match the modern analytical approach in forensic toxicology. The second procedure is highly specific and can be used for targeted analyses or for confirmation in the case of positive results of screening analysis. The new procedure is more sensitive and less sample-consuming than previously published ones.
2. Materials and Methods
2.1. Chemicals and Reagents
Acetonitrile (ACN) for protein precipitation (PP) was purchased from Panreac Quimica S.L.U. (purity > 99.9%, Castellar del Vallès, Spain). ACN and water (H2O) for LC-MS/MS were acquired from Biosolve Chimie SARL (ULC/MS-CC/SFC purity grade, Dieuze, France). Dichloromethane (DCM) and iso-propanol (i-Pro) used for liquid–liquid extraction (LLE) were obtained from Sigma-Aldrich Co. (purity > 99.9%, St. Louis, MO, USA). Potassium dihydrogen phosphate and potassium hydrogen phosphate used for the buffer solution (pH 8, 0.1 M) were acquired from Carlo Erba (purity > 90.0%, Milan, Italy) and J.T. Baker (Denver, The Netherlands), respectively. Certified reference standards of LSD, LSD-D3 (internal standard, IS) and O-H-LSD were obtained from LGC standards (Milan, Italy). Analyte-to-IS ratio was used for quantitative analysis. All standards were diluted to the appropriate concentration with ACN. For standard and IS solutions included in method #1, see the previously published article [37]. Blank blood samples, used for method development and validation, were collected from laboratory personnel and volunteer non-consumers of any drug.
2.2. Sample Treatments
2.2.1. Method #1: PP
Sample treatment was based on a previously published procedure for the detection of 120 NSP and 43 Drugs in blood [37]. Briefly, 200 µL of whole blood was added with 700 µL of cold ACN (0 °C) and 10 µL of IS (5 ng/mL). After vortex mixing and centrifugation (2500× g, 5 min), the supernatant was dried under a nitrogen stream at 40 °C and then reconstituted with 100 µL of H2O. 7 µL were injected into the LC-MS/MS system.
2.2.2. Method #2: LLE
A 200-µL aliquot of whole blood was mixed with 10 µL of IS (5 ng/mL) and then extracted twice with 2 mL of a mixture of DCM/i-Pro (9:1, v/v) at pH 8 (500 µL of phosphate buffer). The mixture was then vortexed and centrifuged (2500× g, 5 min), and the organic layer was collected and dried under a gentle stream of nitrogen (40 °C). The residue was reconstituted with 100 µL of H2O, and 9 µL were injected into the chromatographic system.
2.3. LC-MS/MS
Analysis was conducted using an HPLC Agilent 1290 Infinity system (Agilent Technologies, Palo Alto, CA, USA) interfaced with an Agilent 6460 Triple Quad MS (Agilent Technologies), equipped with an electrospray ion source (ESI) operating in positive mode. The ESI configuration was gas temperature 325 °C; gas flow rate 10 L/min; nebulizer 20 psi. Multiple reaction monitoring (MRM) transitions, data acquisition and elaboration were performed using the Agilent Mass Hunter Workstation software package.
2.3.1. Method #1
Chromatographic separation was performed through a Zorbax Eclipse Plus C18 (2.1 × 100 mm, 1.8 μm, Agilent Technologies). The mobile phase initially consisted of 5 mM aqueous formic acid (A) and ACN (B) 99:1. The gradient of elution was carried out as follows: from 0 to 5 min, linear ramp from 0 to 5%B; from 5 to 7 min, ramp to 10%B; isocratic hold from 7 to 10 min; from 10 to 15 min, ramp to 20%B; from 15 to 20 min, ramp to 30%B; isocratic hold up to 22 min; from 22 to 25 min, ramp to 40%B; from 25 to 28 min, ramp to 50%B; from 28 to 30 min, ramp to 70%B; from 30 to 35 min to 100%B and isocratic hold to 37 min. Post-time was set at 2 min. The flow rate was 0.6 mL/min. Besides the 163 compounds included in the previously published paper, LSD, O-H-LSD and IS (Table 1) and 7 other substances (6-monoacetylmorphine, codeine, dextromethorphan, fentanyl-D5, morphine, oxycodone and tramadol) were added [37,38]. Currently, 338 transitions are monitored in dynamic MRM mode (max concurrent MRM: 38; Min/Max Dwell: 9.66).

Table 1.
MRM transitions and LC-MS/MS conditions. Quantitative transitions are highlighted in bold.
2.3.2. Method #2
A shorter Zorbax Eclipse Plus C18 (2.1 × 50 mm, 1.8 μm, Agilent Technologies) column was used for this method, given the very lower number of analytes (3). The initial mobile phase was A/B 99:1. The gradient was programmed as follows: 50%B in 3 min; 100%B in 2 min; isocratic hold to 6 min. Post-time was set at 1.5 min. The injection volume was 9 µL and the flow rate was 0.5 mL/min. Capillary and nozzle were optimized at 1000 and 300 V, respectively.
2.4. Validation Parameters
Validation was performed following the American Academy of Forensic Sciences’ (AAFS) standard practices for method validation in forensic toxicology [39].
2.4.1. Interferences Studies
Ten different blank whole blood samples were investigated for potential endogenous interfering peaks. Exogenous interferences were estimated by spiking 10 different blank blood samples with 200 ng/mL of common drugs and their main metabolites (including barbiturates, cannabinoids, cocaine, opioids).
2.4.2. Limit of Detection (LOD) and Lower Limit of Quantification (LLOQ)
LOD is the lowest concentration producing a signal-to-noise ratio (SNR) ≥ 3 and meets the identification criteria. It was calculated by analyzing three replicates of blank specimens spiked with decreasing quantities of each substance.
LLOQ estimation was achieved by using the same methodological approach, but its value had to provide a SNR ≥ 10 and had to meet identification, bias and precision criteria.
2.4.3. Calibration Model
The calibration model was investigated for these working ranges: LLOQ-10 ng/mL for LSD (0.025 and/or 0.0375, 0.25, 0.375, 0.5, 1, 5 and 10 ng/mL) and LLOQ-10 ng/mL for O-H-LSD (0.0125 and/or 0.0187, 0.125, 0.187, 0.25, 0.5, 5 and 10 ng/mL). Five replicates of blank blood spiked at the proper concentrations were analyzed and the least-squares regression procedure was applied to the data. Linearity was evaluated by means of the coefficient of determination (R2, acceptance criterium: ≥0.9900).
2.4.4. Bias and Precision
Five replicates of three separated blank blood samples spiked at three different concentrations (quality control, QC) were analyzed: 0.075 (≤3 times the first calibration level), 0.75 and 7.5 (~80% of the highest calibrator) ng/mL for LSD; 0.0375 (≤3 times the LLOQ), 0.375 and 7.5 (~80% of the highest calibrator) ng/mL for O-H-LSD. Bias must not exceed ±20% at each concentration.
Precision was expressed as coefficient of variation (%CV). Three replicates at QC levels were analyzed five times. Within-run precisions were calculated for each QC separately for each of the five runs. For between-run precision, evaluation for each concentration was performed over the five runs. %CV was accepted if <20%.
2.4.5. Recovery Rate (RR), Matrix Effect (ME) and Carry-Over
RRs were estimated by the comparison of analytes slopes from QC1 and QC3 spiked before and after the extraction over 6 replicates.
The post-extraction addition approach was adopted for ME estimation. Ionization suppression (IoS) or enhancement (IoE) was calculated by comparison of areas from two set of samples as follows: set 1 consisted of two neat standards at QC1 and QC3 concentrations; set 2 consisted of ten different blood samples extracted in duplicate and then spiked at QC1 and QC3 levels. Each neat standard was injected six times to establish the mean area of set 1. IoS or IoE should not exceed ±25%.
Carry-over estimation was achieved by injecting the extracted blank samples into the LC-MS/MS system immediately after the highest calibrator over five runs.
3. Results
3.1. MRM Transitions and Chromatographic Separation
The two most intense MRM transitions were used as qualifier and quantifier (Table 1). [M + H]+ parent ions for LSD, LSD-D3 and O-H-LSD were first recognized in SCAN-mode analysis of diluted standard solutions. The specific fragments by collision-induced dissociation (CID) were obtained through the Agilent Mass Hunter Optimizer software and then finely optimized manually. Quantitative vs. qualitative transition ratios were also studied, and their values were: 3.191 for LSD; 2.840 for O-H-LSD; 4.982 for LSD-D3.
3.1.1. Method #1
The retention times of LSD, O-H-LSD and LSD-D3 were obtained by injecting diluted neat standards in the LC-MS/MS set with the previous validated gradient of elution. LSD and LSD-D3 were eluted at 15.270 min and O-H-LSD at 8.810 min. These new retention times were added to the dynamic MRM method with a delta time of 1 min. Ten compounds were concurrent both for the couple LSD/IS and for O-H-LSD, with an average dwell of 20.45 and 21.73, respectively. At the specific detection windows, none of the MRM interfered with the new analyte’s ones.
3.1.2. Method #2
Several gradients of elution were tested in order to obtain the best chromatographic performances in terms of peaks’ shapes, separation and time run. The best results were obtained with the above-described elution program. Elution times are reported in Table 1. Due to the low number of transitions, acquisition was not performed in dynamic MRM mode.
3.2. Optimization of Sample Treatments
PP for method #1 did not require any optimization as LSD and O-H-LSD were added to a fully validated procedure, commonly used in our routinary analysis.
Regarding method #2, several LLE procedures were tested and evaluated in terms of RR and ME at QC levels for various organic solvents (ethyl acetate, diethyl ether, DCM) and their mixtures. The best outcomes were observed with a double LLE with a 9:1 (v/v) mixture of DCM/i-Pro (RR > 85%; ME < 19%).
3.3. Methods Validation
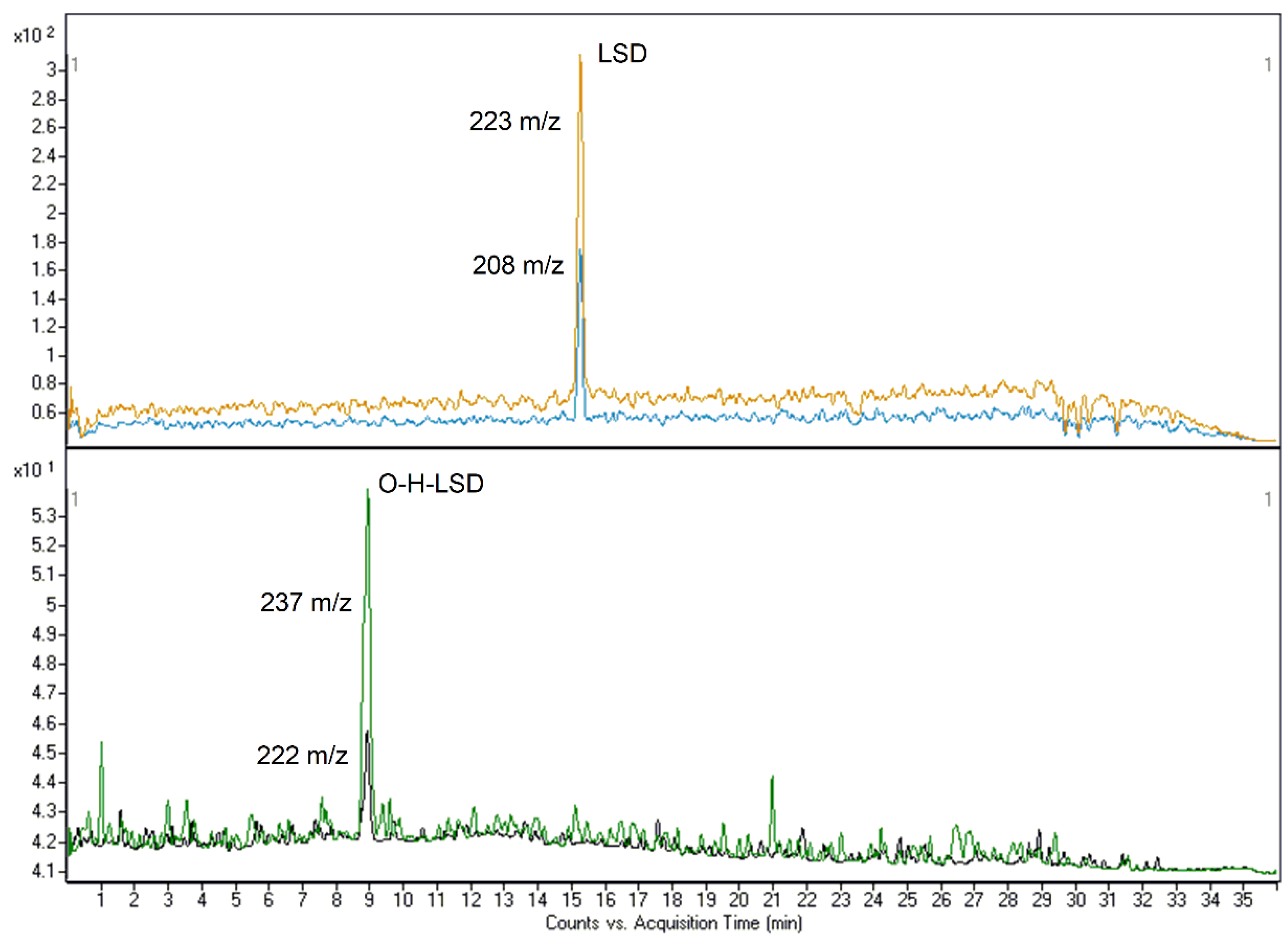
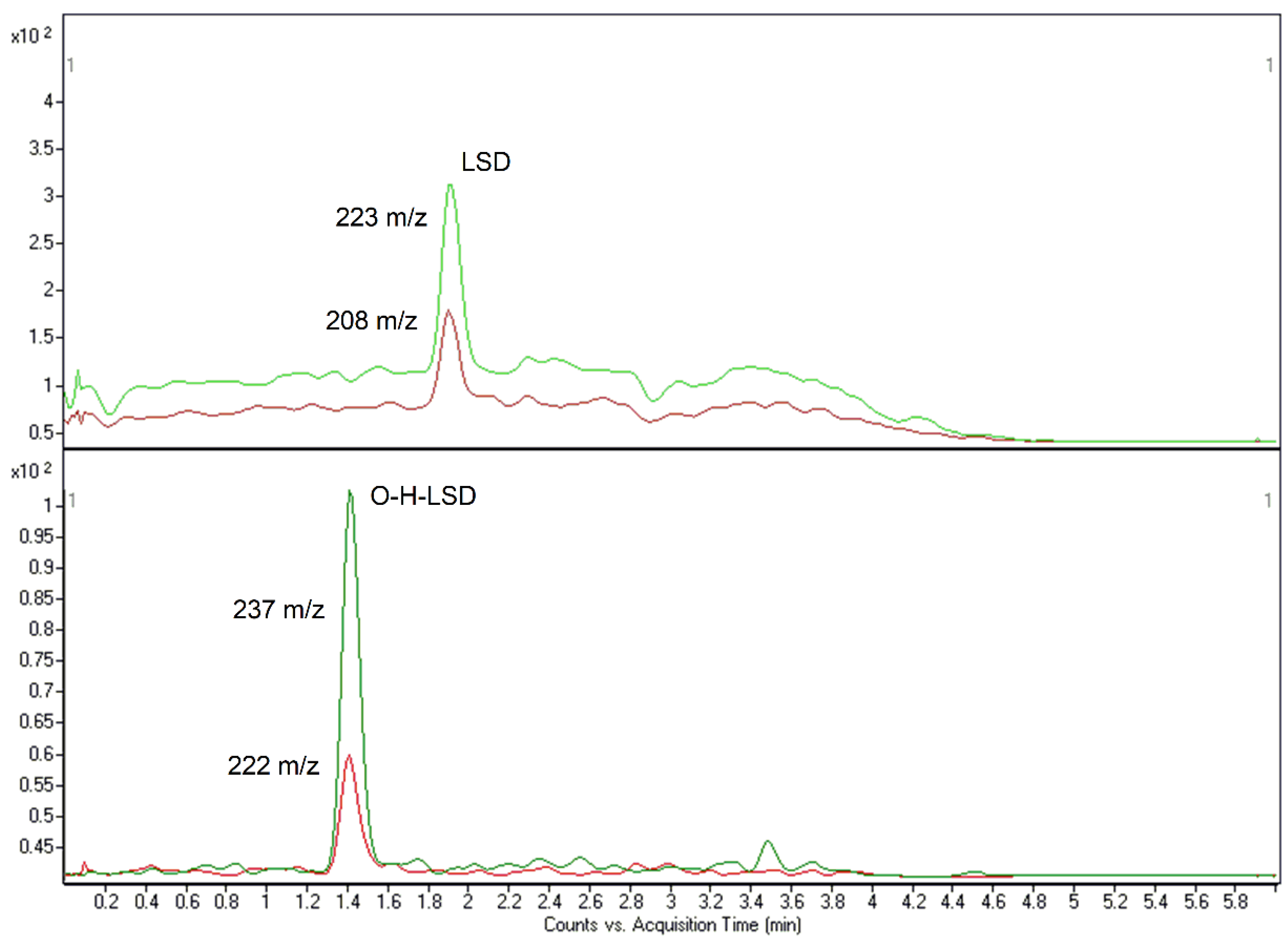
The methods were found to be highly specific and selective since neither endogenous nor exogenous interfering peaks were observed. Nevertheless, with the high number of substances, identification of LSD and O-H-LSD in method #1 was reliable and effective. Both methods were also sensitive enough to detect these compounds at common concentrations. LOD values were 0.01 (#1) and 0.005 (#2) ng/mL for LSD and 0.005 (#1) and 0.0025 (#2) ng/mL for its metabolite. LOQs were measured at 0.0375 (#1) and 0.025 (#2) ng/mL for LSD and 0.0187 (#1) and 0.0125 (#2) ng/mL for O-H-LSD (Figure 1 and Figure 2).
Figure 1.
LOQ-related chromatograms of LSD (0.0375 ng/mL) and O-H-LSD (0.0187 ng/mL) for method #1.
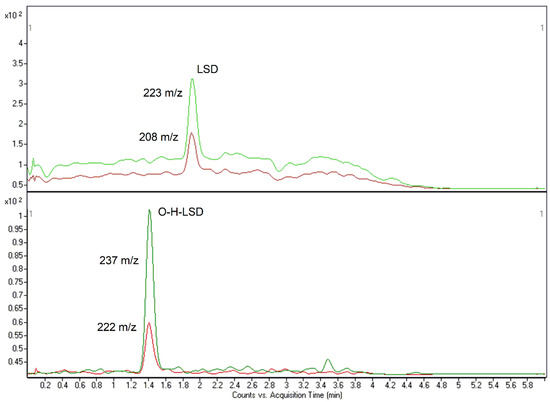
Figure 2.
LOQ-related chromatograms of LSD (0.025 ng/mL) and O-H-LSD (0.0125 ng/mL) for method #2.
Calibration curves for LSD and O-H-LSD were linear with average R2 > 0.9900 for all the analytical ranges (Table 2 and Table 3).

Table 2.
Main validation parameters for method #1 for each compound.

Table 3.
Main validation parameters for method #2 for each compound.
Bias and %CV were always within the acceptance criteria suggested by the AAFS guidelines [39]. RRs were >83.5%, except for O-H-LSD with method #1. In this case, RR ranged from 15.9% to 47.2% (mean: 31.5%) at QC1 and QC3, respectively. A slight IoE was observed for LSD regardless of the method (mean: 9.5% for #1; mean: 9.7% for #2), while it was higher for O-H-LSD (Table 2 and Table 3). Indeed, for method #2 its mean was 18.5% and it was even higher for method #1 (156.5%, exceeding the acceptance criteria, <+25%). This high ME could be due to the low clean-up efficiency of the PP which does not allow the removal of interfering matrix compounds. Carry over was not observed.
4. Discussion
The aim of the study was to test two methods, based on LC-MS/MS, for the combined determination of LSD and O-H-LSD, in blood. In particular, the first method (method 1) was an upgrade of a previous validated procedure for detection of 163 substances (120 of which new psychoactive substances); the second method (method 2) was developed for the specific and sensitive determination of LSD and O-H-LSD.
4.1. Method #1
LSD and its main metabolite were included in a previously validated LC-MS/MS-based multi-analyte method for identification and quantification of 163 substances of relevant forensic-toxicological interest. This procedure employs a simple and fast preparation (PP) of a small amount of matrix (200 µL). The short preparation time and the low resource consumption represent the main advantages. For these reasons, this method is suitable for all cases that require the detection of a wide range of compounds. Caspar et al. developed a procedure for identification in plasma of 15 phenethylamines (n. 10 SCs and n. 5 NBOMes), LSD and 3 derivatives (LSZ, AL-LAD and 1P-LSD), fentanyl and its analogue butyryl fentanyl (21 vs. 165 for the here described method) [36]. The treatment consisted of a double LLE (at neutral and pH 8–9) of 1 mL of blood plasma, which results in a more time- and sample-consuming procedure. Instrumental analysis was performed by a high-resolution mass spectrometer system. For all analytes, they stated an LOQ value of 0.25 ng/mL, higher than our method (0.0375 ng/mL for LSD and 0.01875 ng/mL for O-H-LSD). Drawbacks of the herein proposed procedure were the high IoE (165.5%) and the low RR (31.6%) for O-H-LSD. Thus, we adopted this method only for screening purposes; in the case of positivity, it is confirmed with method #2 (Table 4).

Table 4.
Comparison between method #1 and other.
4.2. Method #2
This procedure proved to be more sensitive than #1. LLE at basic pH resulted in higher RR, especially for O-H-LSD; ME met all the acceptance criteria even for the metabolite which showed very high IoE levels with method #1. The chromatographic run was very short (6 min + 1.5-min post-time), reducing the overall time-consumption. Moreover, this method was less time-consuming than previously published GC-MS(/MS) procedures [26,27,28,40], which usually required longer sample treatments. The most recent validated method was published by Libong et al. [40]. The sample preparation process included a solid-phase extraction (SPE)—more expensive and time-consuming than LLE—of 2 mL of whole blood (vs. 200 µL for method #2) and quantification by a GC coupled with an ion-trap MS. The LOQ value for LSD was slightly lower than ours (0.02 vs. 0.025 ng/mL), with a lower RR (53–65% vs. 83.5–93.1%). LLE was also used by Canezin et al. [38]. In their method, they analyzed a 1-mL aliquot of blood for quantification of LSD iso-LSD and its main metabolites. LOQ values were stated at 0.02 ng/mL for LSD and O-H-LSD. Regarding LC-MS/MS-based methods, Favretto et al. reported the simultaneous determination of LSD, its epimer iso-LSD and its metabolites nor-LSD and O-H-LSD in blood, urine and vitreous humor samples [32]. A 2 mL aliquot of biological fluid (vs. 200 µL for method #2) was liquid-liquid extracted with 10 mL of chloroform. LSD’s and O-H-LSD’s LOQ values in blood were 0.02 (vs. 0.025) and 0.4 (vs. 0.0125) ng/mL. RR were in line with our data, while they observed IoS, instead of IoE, for O-H-LSD. For these reasons, method #2 presents a comparable sensitivity with a lower amount of analyzed matrix. A PP procedure was developed by Dolder et al. [35]. They added acetonitrile to 100 µL of plasma for the simultaneous detection of LSD and iso-LSD, nor-LSD and O-H-LSD by LC-MS/MS. LOQ was estimated at 0.05 ng/mL for LSD and 0.1 for O-H-LSD. They also estimated IoE at 125% for LSD and 103% for O-H-LSD. Dolder’s procedure is similar to method #2, even if it is focused only on LSD and its main metabolites. However, its sensitivity was slightly lower (0.05 vs. 0.025 ng/mL for LSD and 0.1 vs. 0.0125 ng/mL for O-H-LSD) (Table 5).

Table 5.
Comparison between method #2 and others.
5. Application to Real Samples
5.1. Case 1
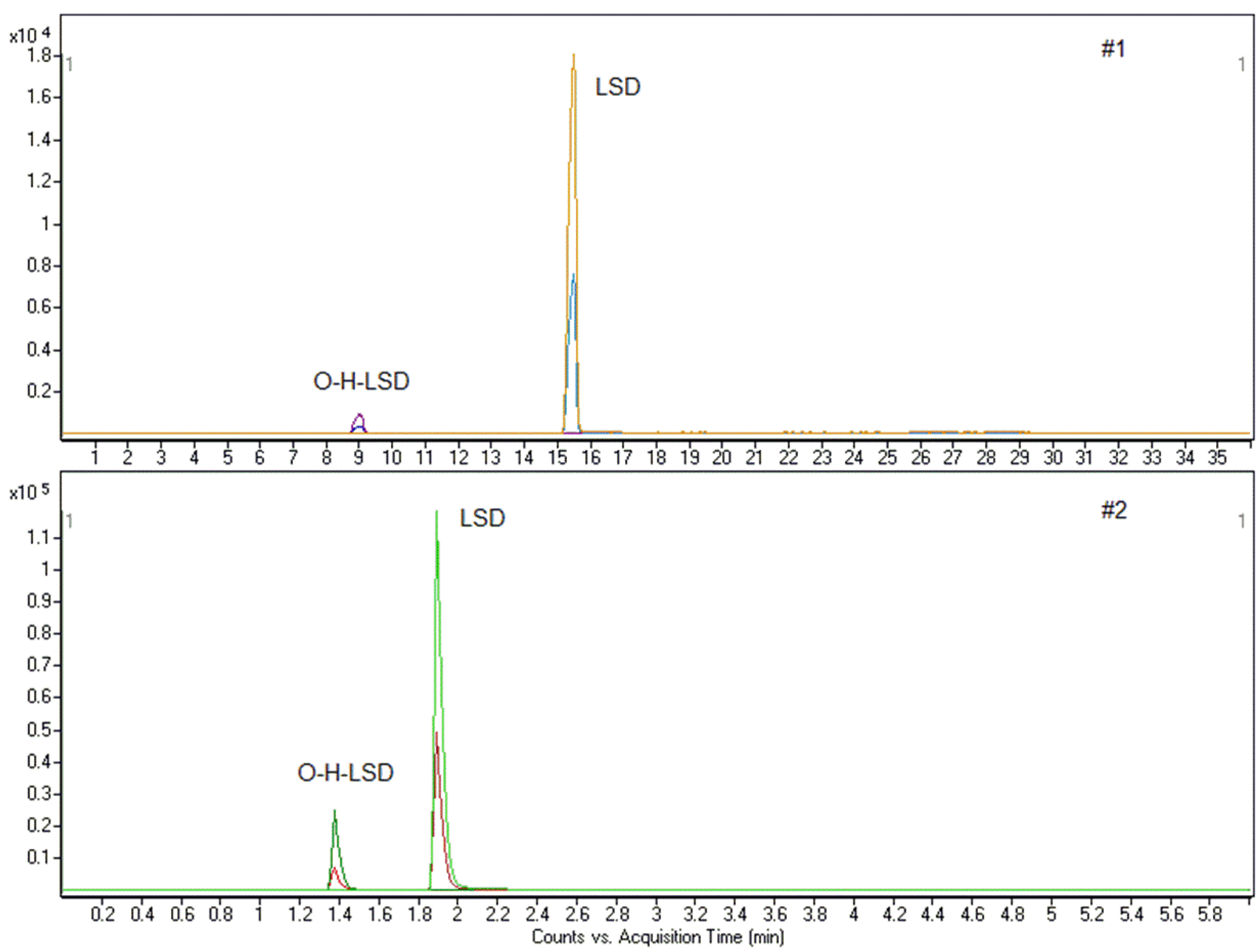
A 45-year-old male, reportedly a habitual cocaine and alcohol consumer, was hospitalized for time and space disorientation. At hospitalization, he reported the intake of one LSD-stamp 2 h. Analysis of whole blood with method #1 highlighted the presence of LSD and O-H-LSD, which were then quantified with method #2 at 8.60 and 1.14 ng/mL, respectively. The biological fluid resulted negative for cocaine, alcohol and other drugs of abuse by specific analytical methods [41,42] (Figure 3).
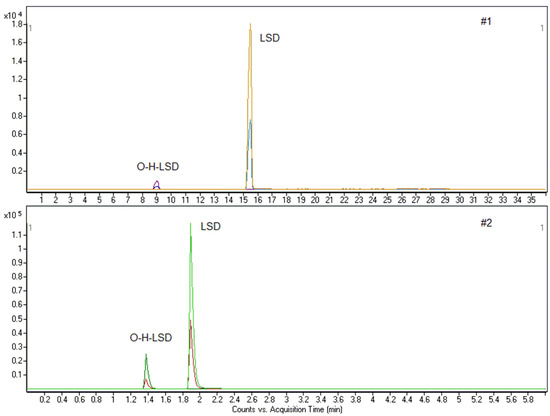
Figure 3.
Chromatograms for case 1 (LSD: 8.60 ng/mL; O-H-LSD: 1.14 ng/mL).
5.2. Case 2
A blood sample was obtained from a 23-year-old girl hospitalized for delirium. She only reported to have consumed a gummy candy whose content was unknown. Screening of blood using method #1 resulted in positivity for LSD and O-H-LSD, which was then confirmed at 1.75 and 0.55 ng/mL.
6. Conclusions
Reliable monitoring of drugs of abuse requires a specific and sensitive analytical procedure. This is particularly true for LSD determination, since this compound is active at very low levels. In this paper, validation of two methods for simultaneous determination of LSD and O-H-LSD in blood were fully validated. The first procedure proved to be very useful for screening purposes. It allows the identification of 165 substances of forensic-toxicological interest. The simple sample preparation (PP) and the small sample volume (200 µL) are the main method’s advantages. Though LC-MS/MS is often used for quantitation, it is increasingly used to screen a large number of compounds, even at low concentrations. The second proposed method was highly sensitive and specific for the quantification of LSD and O-H-LSD in blood. It is characterized by a rapid and low-cost sample preparation (LLE) and an efficient chromatographic separation in a relatively short time (6 min + 1.5-min post-time). Both methods were successfully applied to two real cases and currently are part of our routinary protocols. It must be underlined that these methods could be complementary as demonstrated by case #2. Indeed, we had no information about the possible intake of LSD. Only the application of the screening method #1 allowed us to detect the presence of this substance, which was then confirmed by method #2.
Author Contributions
Conceptualization, F.V. and A.D.; methodology, F.V. and M.G.D.M.; validation, F.V. and M.G.D.M.; formal analysis, A.D.; resources, F.V., B.G. and R.R.; data curation, F.V. and A.D.; writing—original draft preparation, F.V. and A.D.; writing—review and editing, F.V., A.D. and S.G.; supervision, F.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy issues.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Jastrzębski, M.K.; Kaczor, A.A.; Wróbel, T.M. Methods of Lysergic Acid Synthesis-The Key Ergot Alkaloid. Molecules 2022, 27, 7322. [Google Scholar] [CrossRef]
- Schmid, Y.; Enzler, F.; Gasser, P.; Grouzmann, E.; Preller, K.H.; Vollenweider, F.X.; Brenneisen, R.; Müller, F.; Borgwardt, S.; Liechti, M.E. Acute Effects of Lysergic Acid Diethylamide in Healthy Subjects. Biol. Psychiatry 2015, 78, 544–553. [Google Scholar] [CrossRef]
- Dolder, P.C.; Liechti, M.E.; Rentsch, K.M. Development and Validation of a Rapid Turboflow LC-MS/MS Method for the Quantification of LSD and 2-Oxo-3-Hydroxy LSD in Serum and Urine Samples of Emergency Toxicological Cases. Anal. Bioanal. Chem. 2014, 407, 1577–1584. [Google Scholar] [CrossRef][Green Version]
- Dolder, P.C.; Schmid, Y.; Haschke, M.; Rentsch, K.M.; Liechti, M.E. Pharmacokinetics and Concentration-Effect Relationship of Oral LSD in Humans. Int. J. Neuropsychopharmacol. 2015, 19, pyv072. [Google Scholar] [CrossRef]
- Hintzen, A.; Passie, T. Pharmacology of LSD: A Critical Review; Oxford University Press: Oxford, UK, 2010. [Google Scholar]
- Upshall, D.G.; Wailling, D.G. The Determination of LSD in Human Plasma Following Oral Administration. Clin. Chim. Acta 1972, 36, 67–73. [Google Scholar] [CrossRef]
- Libânio Osório Marta, R.F. Metabolism of Lysergic Acid Diethylamide (LSD): An Update. Drug Metab. Rev. 2019, 51, 378–387. [Google Scholar] [CrossRef]
- Boyd, E.S.; Rothlin, E.; Bonner, J.F.; Slater, I.H.; Hodge, H.C. Preliminary Studies of the Metabolism of Lysergic Acid Diethylamide Using Radioactive Carbon-Marked Molecules. J. Nerv. Ment. Dis. 1955, 122, 470–471. [Google Scholar] [CrossRef]
- Axelrod, J.; Brady, R.O.; Witkop, B.; Evarts, E.V. The Distribution and Metabolism of Lysergic Acid Diethylamide. Ann. N. Y. Acad. Sci. 1957, 66, 435–444. [Google Scholar] [CrossRef]
- Siddik, Z.H.; Barnes, R.D.; Dring, L.G.; Smith, R.L.; Williams, R.T. The Metabolism of Lysergic Acid DI[14C]Ethylamide ([14C]LSD) in the Isolated Perfused Rat Liver. Biochem. Pharmacol. 1979, 28, 3081–3091. [Google Scholar] [CrossRef]
- Aghajanian, G.K.; L Bing, O.H. Persistence of Lysergic Acid Diethylamide in the Plasma of Human Subjects. Clin. Pharmacol. Ther. 1964, 5, 611–614. [Google Scholar] [CrossRef]
- Schiff, P.L. Ergot and Its Alkaloids. Am. J. Pharm. Educ. 2006, 70, 98. [Google Scholar] [CrossRef] [PubMed]
- Reiff, C.M.; Richman, E.E.; Nemeroff, C.B.; Carpenter, L.L.; Widge, A.S.; Rodriguez, C.I.; Kalin, N.H.; McDonald, W.M. Psychedelics and Psychedelic-Assisted Psychotherapy. Am. J. Psychiatry 2020, 177, 391–410. [Google Scholar] [CrossRef] [PubMed]
- Mertens, L.J.; Preller, K.H. Classical Psychedelics as Therapeutics in Psychiatry—Current Clinical Evidence and Potential Therapeutic Mechanisms in Substance Use and Mood Disorders. Pharmacopsychiatry 2021, 54, 176–190. [Google Scholar] [CrossRef] [PubMed]
- De Gregorio, D.; Aguilar-Valles, A.; Preller, K.H.; Heifets, B.D.; Hibicke, M.; Mitchell, J.; Gobbi, G. Hallucinogens in Mental Health: Preclinical and Clinical Studies on LSD, Psilocybin, MDMA, and Ketamine. J. Neurosci. 2021, 41, 891–900. [Google Scholar] [CrossRef] [PubMed]
- De Gregorio, D.; Inserra, A.; Enns, J.P.; Markopoulos, A.; Pileggi, M.; El Rahimy, Y.; Lopez-Canul, M.; Comai, S.; Gobbi, G. Repeated Lysergic Acid Diethylamide (LSD) Reverses Stress-Induced Anxiety-like Behavior, Cortical Synaptogenesis Deficits and Serotonergic Neurotransmission Decline. Neuropsychopharmacology 2022, 47, 1188–1198. [Google Scholar] [CrossRef]
- Markopoulos, A.; Inserra, A.; De Gregorio, D.; Gobbi, G. Evaluating the Potential Use of Serotonergic Psychedelics in Autism Spectrum Disorder. Front. Pharmacol. 2022, 12, 749068. [Google Scholar] [CrossRef]
- Doyon, W.M.; Dong, Y.; Ostroumov, A.; Thomas, A.M.; Zhang, T.A.; Dani, J.A. Nicotine Decreases Ethanol-Induced Dopamine Signaling and Increases Self-Administration via Stress Hormones. Neuron 2013, 79, 530–540. [Google Scholar] [CrossRef]
- Dyck, E. ‘Hitting Highs at Rock Bottom’: LSD Treatment for Alcoholism, 1950–1970. Soc. Hist. Med. 2006, 19, 313–329. [Google Scholar] [CrossRef]
- Dolder, P.C.; Schmid, Y.; Steuer, A.E.; Kraemer, T.; Rentsch, K.M.; Hammann, F.; Liechti, M.E. Pharmacokinetics and Pharmacodynamics of Lysergic Acid Diethylamide in Healthy Subjects. Clin. Pharmacokinet. 2017, 56, 1219–1230. [Google Scholar] [CrossRef]
- Verstraete, A.G.; Van De Velde, E.J. 2-Oxo-3-Hydroxy-LSD: An Important LSD Metabolite? Acta Clin. Belg. 1999, 53 (Suppl. 1), 94–96. [Google Scholar]
- Loeffler, L.J.; Pierce, J. V Radioimmunoassay for Lysergide (LSD) in Illicit Drugs and Biological Fluids. J. Pharm. Sci. 1973, 62, 1817–1820. [Google Scholar] [CrossRef] [PubMed]
- Grobosch, T.; Lemm-Ahlers, U. Immunoassay Screening of Lysergic Acid Diethylamide (LSD) and Its Confirmation by HPLC and Fluorescence Detection Following LSD ImmunElute Extraction. J. Anal. Toxicol. 2002, 26, 181–186. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Frost, M.; Köhler, H.; Blaschke, G. Determination of LSD in Blood by Capillary Electrophoresis with Laser-Induced Fluorescence Detection. J. Chromatogr. B Biomed. Sci. Appl. 1997, 693, 313–319. [Google Scholar] [CrossRef]
- Steuer, A.E.; Poetzsch, M.; Stock, L.; Eisenbeiss, L.; Schmid, Y.; Liechti, M.E.; Kraemer, T. Development and Validation of an Ultra-Fast and Sensitive Microflow Liquid Chromatography-Tandem Mass Spectrometry (MFLC-MS/MS) Method for Quantification of LSD and Its Metabolites in Plasma and Application to a Controlled LSD Administration Study in Humans. Drug Test Anal. 2017, 9, 788–797. [Google Scholar] [CrossRef]
- Nelson, C.C.; Foltz, R.L. Determination of Lysergic Acid Diethylamide (LSD), Iso-LSD, and N-Demethyl-LSD in Body Fluids by Gas Chromatography/Tandem Mass Spectrometry. Anal. Chem. 1992, 64, 1578–1585. [Google Scholar] [CrossRef] [PubMed]
- Papac, D.I.; Foltz, R.L. Measurement of Lysergic Acid Diethylamide (LSD) in Human Plasma by Gas Chromatography/Negative Ion Chemical Ionization Mass Spectrometry. J. Anal. Toxicol. 1990, 14, 189–190. [Google Scholar] [CrossRef]
- Reuschel, S.A.; Percey, S.E.; Liu, S.; Eades, D.M.; Foltz, R.L. Quantitative Determination of LSD and a Major Metabolite, 2-Oxo-3-Hydroxy-LSD, in Human Urine by Solid-Phase Extraction and Gas Chromatography-Tandem Mass Spectrometry. J. Anal. Toxicol. 1999, 23, 306–312. [Google Scholar] [CrossRef]
- Favretto, D.; Frison, G.; Maietti, S.; Ferrara, S.D. LC-ESI-MS/MS on an Ion Trap for the Determination of LSD, Iso-LSD, nor-LSD and 2-Oxo-3-Hydroxy-LSD in Blood, Urine and Vitreous Humor. Int. J. Legal Med. 2007, 121, 259–265. [Google Scholar] [CrossRef]
- Grumann, C.; Henkel, K.; Stratford, A.; Hermanns-Clausen, M.; Passie, T.; Brandt, S.D.; Auwärter, V. Validation of an LC-MS/MS Method for the Quantitative Analysis of 1P-LSD and Its Tentative Metabolite LSD in Fortified Urine and Serum Samples Including Stability Tests for 1P-LSD under Different Storage Conditions. J. Pharm. Biomed. Anal. 2019, 174, 270–276. [Google Scholar] [CrossRef]
- Martin, R.; Schürenkamp, J.; Gasse, A.; Pfeiffer, H.; Köhler, H. Determination of Psilocin, Bufotenine, LSD and Its Metabolites in Serum, Plasma and Urine by SPE-LC-MS/MS. Int. J. Legal Med. 2013, 127, 593–601. [Google Scholar] [CrossRef]
- Johansen, S.S.; Jensen, J.L. Liquid Chromatography–Tandem Mass Spectrometry Determination of LSD, ISO-LSD, and the Main Metabolite 2-Oxo-3-Hydroxy-LSD in Forensic Samples and Application in a Forensic Case. J. Chromatogr. B 2005, 825, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Canezin, J.; Cailleux, A.; Turcant, A.; Le Bouil, A.; Harry, P.; Allain, P. Determination of LSD and Its Metabolites in Human Biological Fluids by High-Performance Liquid Chromatography with Electrospray Tandem Mass Spectrometry. J. Chromatogr. B Biomed. Sci. Appl. 2001, 765, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Jang, M.; Kim, J.; Han, I.; Yang, W. Simultaneous Determination of LSD and 2-Oxo-3-Hydroxy LSD in Hair and Urine by LC-MS/MS and Its Application to Forensic Cases. J. Pharm. Biomed. Anal. 2015, 115, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Dolder, P.C.; Liechti, M.E.; Rentsch, K.M. Development and Validation of an LC-MS/Ms Method to Quantify Lysergic Acid Diethylamide (LSD), Iso-LSD, 2-Oxo-3-Hydroxy-LSD, and nor-LSD and Identify Novel Metabolites in Plasma Samples in a Controlled Clinical Trial. J. Clin. Lab. Anal. 2018, 32, e22265. [Google Scholar] [CrossRef]
- Caspar, A.T.; Kollas, A.B.; Maurer, H.H.; Meyer, M.R. Development of a Quantitative Approach in Blood Plasma for Low-Dosed Hallucinogens and Opioids Using LC-High Resolution Mass Spectrometry. Talanta 2018, 176, 635–645. [Google Scholar] [CrossRef]
- Vaiano, F.; Bertol, E.; Mineo, M.; Pietrosemoli, L.; Rubicondo, J.; Supuran, C.T.; Carta, F. Development of a New Lc-Ms/Ms Screening Method for Detection of 120 Nps and 43 Drugs in Blood. Separations 2021, 8, 221. [Google Scholar] [CrossRef]
- Rubicondo, J.; Scuffi, L.; Pietrosemoli, L.; Mineo, M.; Terranova, F.; Bartucca, M.; Trignano, C.; Bertol, E.; Vaiano, F. A New Multi-Analyte LC-MS-MS Screening Method for the Detection of 120 NPSs and 49 Drugs in Hair. J. Anal. Toxicol. 2023, 46, e262–e273. [Google Scholar] [CrossRef]
- ANSI/ASB STANDARD 036; Standard Board Standard Practices for Method Validation in Forensic Toxicology. Academy Standards Board: Colorado Springs, CO, USA, 2019.
- Libong, D.; Bouchonnet, S.; Ricordel, I. A Selective and Sensitive Method for Quantitation of Lysergic Acid Diethylamide (LSD) in Whole Blood by Gas Chromatography-Ion Trap Tandem Mass Spectrometry. J. Anal. Toxicol. 2003, 27, 24–29. [Google Scholar] [CrossRef][Green Version]
- Bertol, E.; Di Milia, M.G.; Fioravanti, A.; Mari, F.; Palumbo, D.; Pascali, J.P.; Vaiano, F. Proactive Drugs in DFSA Cases: Toxicological Findings in an Eight-Years Study. Forensic Sci. Int. 2018, 291, 207–215. [Google Scholar] [CrossRef]
- Bertol, E.; Pascali, J.P.; Palumbo, D.; Catalani, V.; Di Milia, M.G.; Fioravanti, A.; Mari, F.; Vaiano, F. 3-MeO-PCP Intoxication in Two Young Men: First in Vivo Detection in Italy. Forensic Sci. Int. 2017, 274, 7–12. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).