Application of Choline Chloride-Based Deep Eutectic Solvents in the Synthesis of Hydrazones
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of DESs and Detecting Water Content
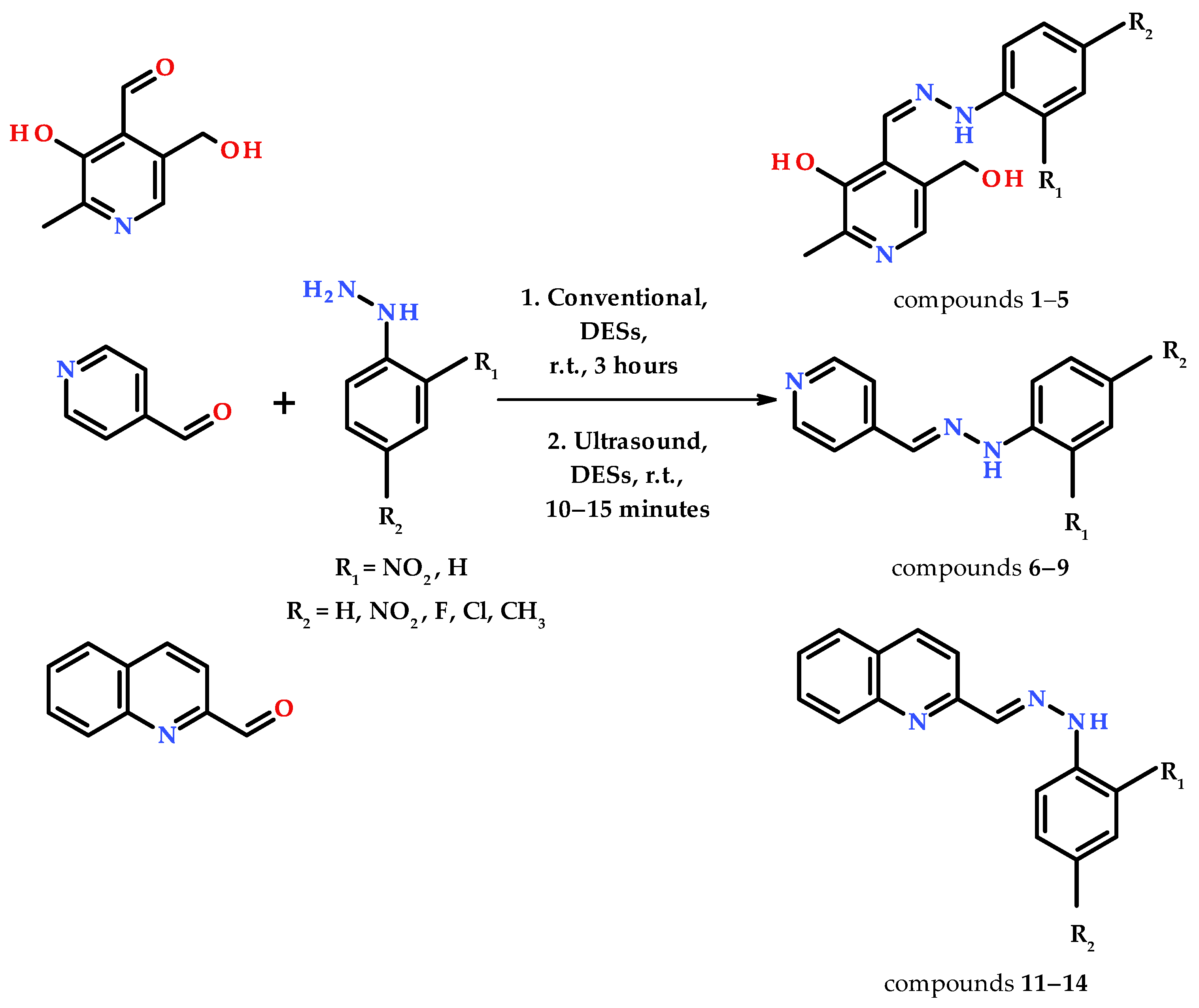
2.2. Conventional Method
2.3. Ultrasound Method
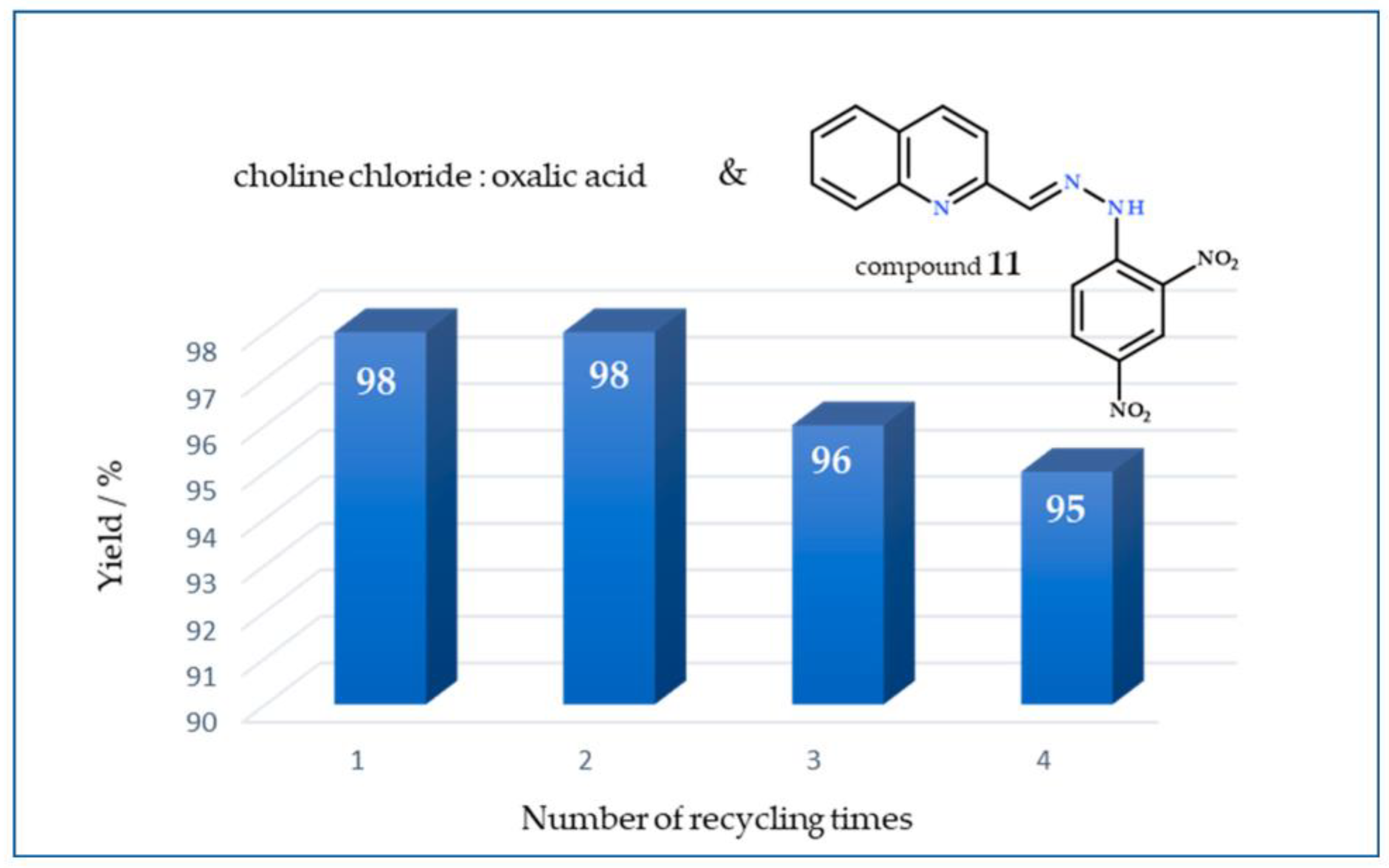
2.4. Recycling and Reuse of DESs
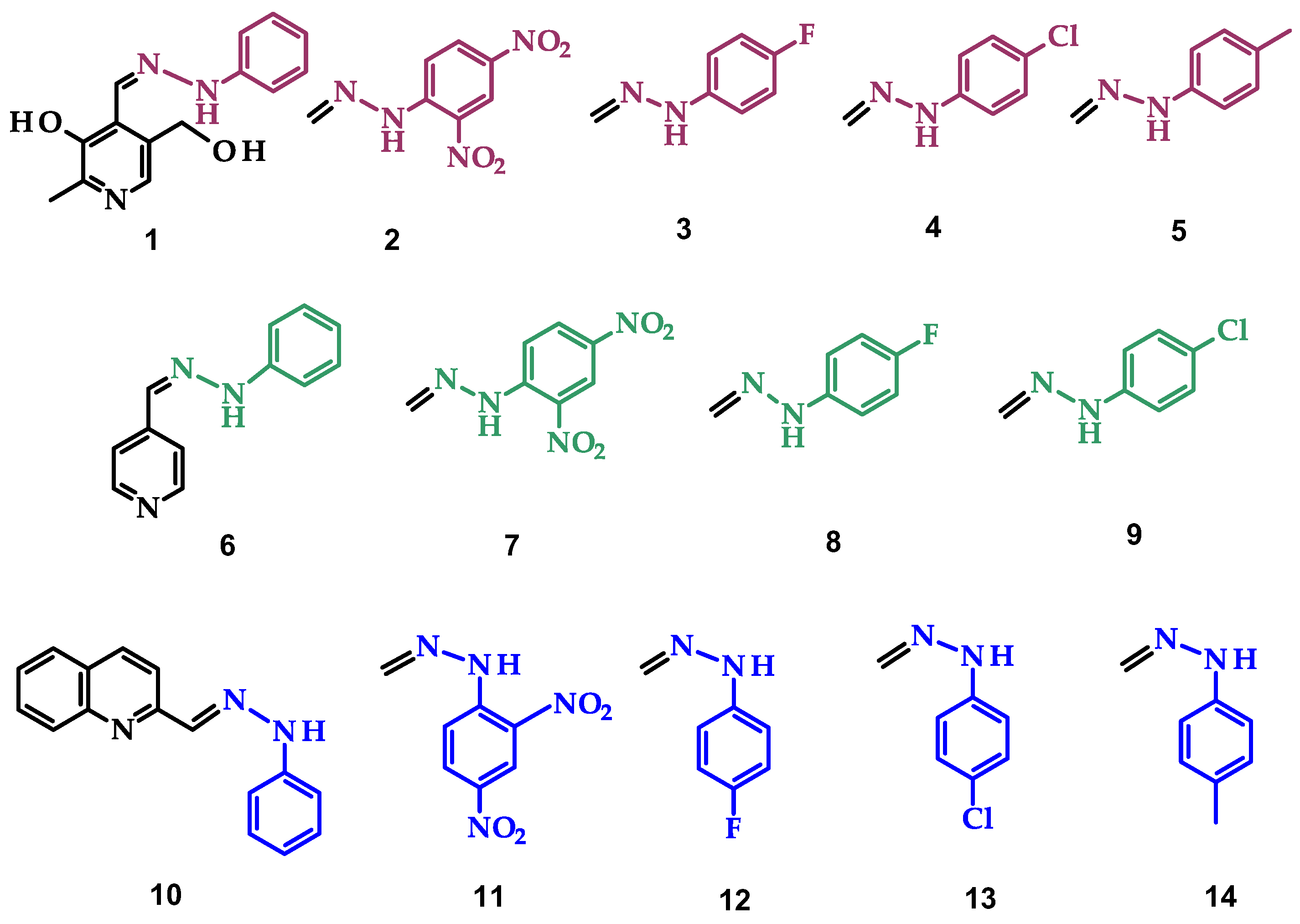
- 5-(hydroxymethyl)-2-methyl-4-((2-phenylhydrazineylidene)methyl)pyridin-3-ol (1)
- 4-((2-(2,4-dinitrophenyl)hydrazineylidene)methyl)-5-(hydroxymethyl)-2-methylpyridin-3-ol (2)
- 4-((2-(4-fluorophenyl)hydrazineylidene)methyl)-5-(hydroxymethyl)-2-methylpyridin-3-ol (3)
- 4-((2-(4-chlorophenyl)hydrazineylidene)methyl)-5-(hydroxymethyl)-2-methylpyridin-3-ol (4)
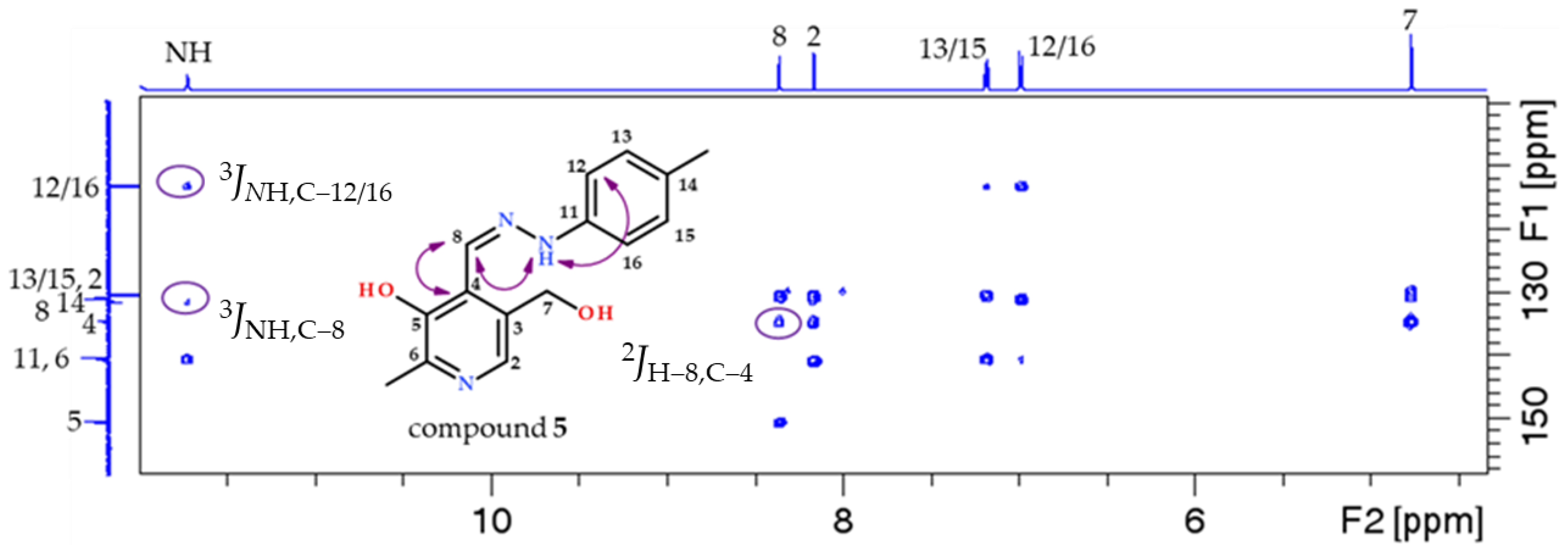
- 5-(hydroxymethyl)-2-methyl-4-((2-(p-tolyl)hydrazineylidene)methyl)pyridin-3-ol (5)
- Pyridine-4-carbaldehyde-phenylhydrazone (6)
- Pyridine-4-carbaldehyde-2,4-dinitrophenylhydrazone (7)
- Pyridine-4-carbaldehyde-4-fluorophenylhydrazone (8)
- Pyridine-4-carbaldehyde-4-chlorophenylhydrazone (9)
- Quinoline-2-carbaldehyde-phenylhydrazone (10)
- Quinoline-2-carbaldehyde-2,4-dinitrophenylhydrazone (11)
- Quinoline-2-carbaldehyde-4-fluorophenylhydrazone (12)
- Quinoline-2-carbaldehyde-4-chlorophenylhydrazone (13)
- Quinoline-2-carbaldehyde-4-methylphenylhydrazone (14)
3. Results and Discussion
3.1. Preparation of DESs and Water Content
3.2. Conventional Synthesis
3.3. Ultrasound Synthesis
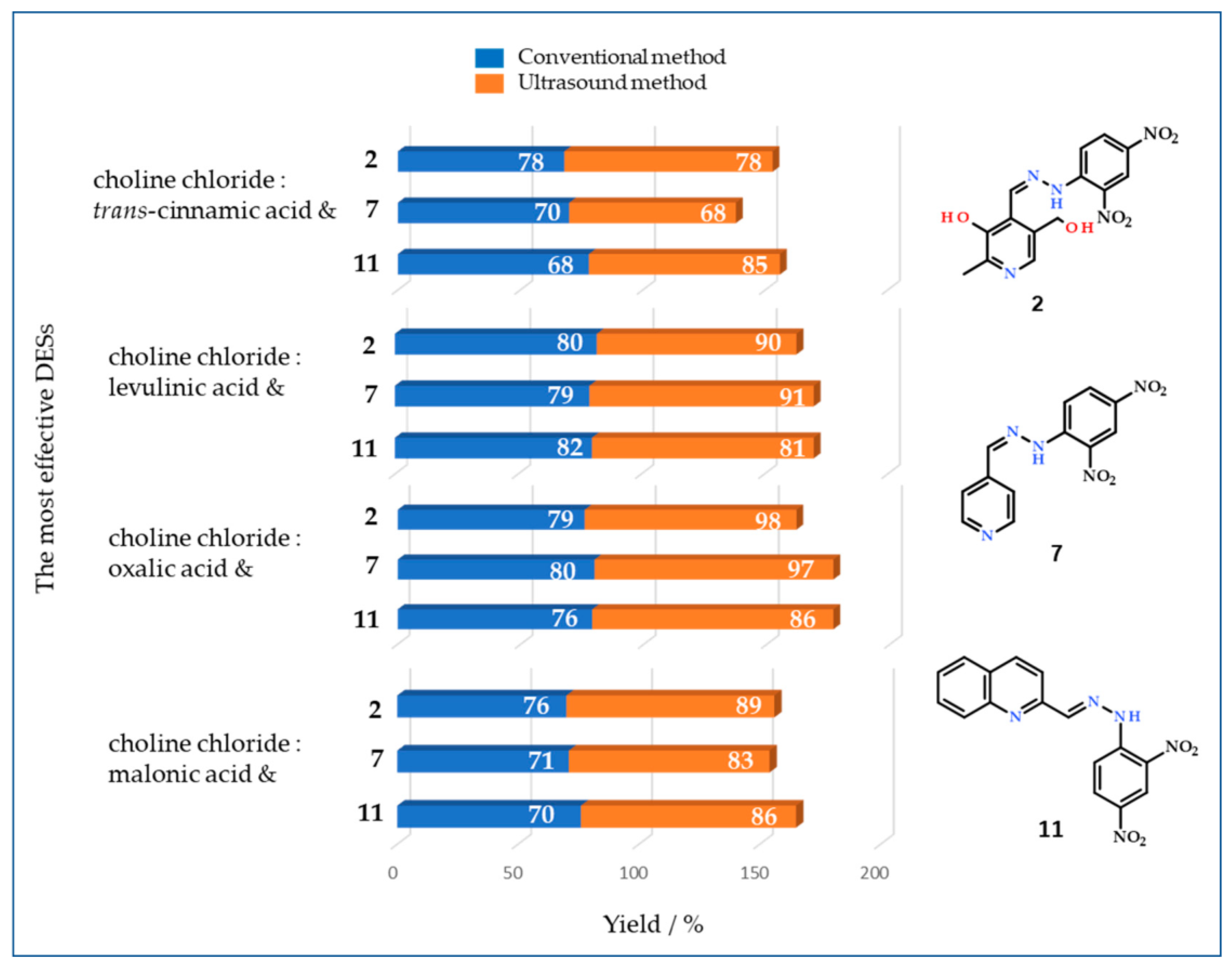
3.4. Recycling and Reuse of DESs Choline Chloride:Oxalic Acid
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sevim, R.; Küçükgüzel, G. Biological Activities of Hydrazone Derivatives. Molecules 2007, 12, 1910–1939. [Google Scholar]
- Dimmock, J.R.; Vashishtha, S.C.; Stables, J.P. Anticonvulsant properties of various acetylhydrazones, oxamoylhydrazones and semicarbazones derived from aromatic and unsaturated carbonyl compounds. Eur. J. Med. Chem. 2000, 35, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Jain, J.; Kumar, Y.; Sinha, R.; Kumar, R.; Stables, J. Menthone aryl acid hydrazones: A new class of anticonvulsants. Med. Chem. 2011, 7, 56–61. [Google Scholar] [CrossRef]
- Tributino, J.L.; Duarte, C.D.; Corrêa, R.S.; Doriguetto, A.C.; Ellena, J.; Romeiro, N.C.; Castro, N.G.; Miranda, A.L.P.; Barreiro, E.J.; Fraga, C.A. Novel 6-methanesulfonamide-3, 4-methylenedioxyphenyl-N-acylhydrazones: Orally effective anti-inflammatory drug candidates. Bioorg. Med. Chem. 2009, 17, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Gil-Longo, J.; Laguna, M.D.L.R.; Verde, I.; Castro, M.E.; Orallo, F.; Fontenla, J.A.; Calleja, J.M.; Ravina, E.; Teran, C. Pyridazine derivatives. XI: Antihypertensive activity of 3-hydrazinocycloheptyl [1, 2-c] pyridazine and its hydrazone derivatives. J. Pharm. Sci. 1993, 82, 286–290. [Google Scholar] [CrossRef][Green Version]
- Liu, W.Y.; Li, H.Y.; Zhao, B.X.; Shin, D.S.; Lian, S.; Miao, J.Y. Synthesis of novel ribavirin hydrazone derivatives and anti-proliferative activity against A549 lung cancer cells. Carbohydr. Res. 2009, 344, 1270–1275. [Google Scholar] [CrossRef] [PubMed]
- Pandey, J.; Pal, R.; Dwivedi, A.; Hajela, K. Synthesis of some new diaryl and triaryl hydrazone derivatives as possible estrogen receptor modulators. Arzneimittelforsch 2002, 52, 39–44. [Google Scholar] [CrossRef]
- Terzioğlu, N.; Gürsoy, A. Synthesis and anticancer evaluation of some new hydrazone derivatives of 2,6-dimethylimidazo[2,1-b]-[1,3,4]thiadiazole-5-carbohydrazide. Eur. J. Med. Chem. 2003, 38, 781–786. [Google Scholar] [CrossRef]
- Cocco, M.T.; Congiu, C.; Lilliu, V.; Onnis, V. Synthesis and in vitro antitumoral activity of new hydrazinopyrimidine-5-carbonitrile derivatives. Bioorg. Med. Chem. 2005, 14, 366–372. [Google Scholar] [CrossRef]
- Gürsoy, E.; Güzeldemirci-Ulusoy, N. Synthesis and primary cytotoxicity evaluation of new imidazo[2,1-b]thiazole derivatives. Eur. J. Med. Chem. 2007, 42, 320–326. [Google Scholar] [CrossRef]
- El-Hawash, S.A.M.; Wahab, A.E.; El-Dewellawy, M.A. Cyanoacetic acid hydrazones of 3-(and 4-) acetylpyridine and some derived ring systems as potential antitumor and anti-HCV agents. Arch. Der Pharm. Int. J. Pharm. Med. Chem. 2006, 339, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Rane, R.A.; Telvekar, V.N. Synthesis and evaluation of novel chloropyrrole molecules designed by molecular hybridization of common pharmacophores as potential antimicrobial agents. Bioorg. Med. Chem. Lett. 2010, 20, 5681–5685. [Google Scholar] [CrossRef] [PubMed]
- Turan-Zitouni, G.; Blache, Y.; Güven, K. Synthesis and antimicrobial activity of some imidazo-[1,2-a]pyridine-2-carboxylic acid arylidenehydrazide derivatives. Boll. Chim. Farm. 2001, 140, 397–400. [Google Scholar]
- Fattorusso, C.; Campiani, G.; Kukreja, G.; Persico, M.; Butini, S.; Romano, M.P.; Altarelli, M.; Ros, S.; Brindisi, M.; Savini, L. Design, synthesis, and structure–activity relationship studies of 4-quinolinyl-and 9-acrydinylhydrazones as potent antimalarial agents. J. Med. Chem. 2008, 51, 1333–1343. [Google Scholar] [CrossRef]
- Walcourt, A.; Loyevsky, M.; Lovejoy, D.B.; Gordeuk, V.R.; Richardson, D.R. Novel aroylhydrazone and thiosemicarbazone iron chelators with anti-malarial activity against chloroquine-resistant and -sensitive parasites. Int. J. Biochem. Cell Biol. 2004, 36, 401–407. [Google Scholar] [CrossRef]
- Gemma, S.; Kukreja, G.; Fattorusso, C.; Persico, M.; Romano, M.; Altarelli, M.; Savini, L.; Campiani, G.; Fattorusso, E.; Basilico, N. Synthesis of N1-arylidene-N2-quinolyl- and N2-acrydinylhydrazones as potent antimalarial agents active against CQ-resistant P. falciparum strains. Bioorg. Med. Chem. Let. 2006, 16, 5384–5388. [Google Scholar] [CrossRef]
- Ma, X.D.; Yang, S.Q.; Gu, S.X.; He, Q.Q.; Chen, F.E.; De Clercq, E.; Balzarini, J.; Pannecouque, C. Synthesis and anti-HIV activity of Aryl-2-[(4-cyanophenyl) amino]-4-pyrimidinone hydrazones as potent non-nucleoside reverse transcriptase inhibitors. Chem. Med. Chem. 2011, 6, 2225–2232. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, A.; Kremer, L.; Louw, S.; Guéradel, Y.; Chibale, K.; Biot, C. Synthesis and in vitro antitubercular activity of ferrocene-based hydrazones. Bioorg. Med. Chem. Lett. 2011, 21, 2866–2868. [Google Scholar] [CrossRef]
- Bukowski, L.; Janowiec, M. 1-Methyl-1H-2-imidazo[4,5-b]pyridinecarboxylic acid and some of derivatives with suspected antituberculotic activity. Pharmazie 1996, 51, 27–30. [Google Scholar]
- Bukowski, L.; Janowiec, M.; Zwolska-Kwiek, Z.; Andrzejczyk, Z. Synthesis and some reactions of 2- acetylimidazo[4,5-b]pyridine. Antituberculotic activity of the obtained compounds. Pharmazie 1999, 54, 651–654. [Google Scholar] [CrossRef]
- Mamolo, M.G.; Falagiani, V.; Zampieri, D.; Vio, L.; Banfi, E. Synthesis and antimycobacterial activity of [5-(pyridin-2-yl)-1,3,4-thiadiazole-2-ylthio]acetic acid arylidene-hydrazide derivatives. Farmaco 2001, 56, 587–592. [Google Scholar] [CrossRef]
- Mamolo, M.G.; Falagiani, V.; Zampieri, D.; Vio, L.; Banfi, E.; Scialino, G. Synthesis and antimycobacterial activity of (3,4-diaryl-3H-thiazole-2-ylidene)hydrazide derivatives. Farmaco 2003, 58, 631–637. [Google Scholar] [CrossRef]
- Musad, E.A.; Mohamed, R.; Saeed, B.A.; Vishwanath, B.S.; Rai, K.L. Synthesis and evaluation of antioxidant and antibacterial activities of new substituted bis (1, 3, 4-oxadiazoles), 3, 5-bis (substituted) pyrazoles and isoxazoles. Bioorg. Med. Chem. Lett. 2011, 21, 3536–3540. [Google Scholar] [CrossRef]
- El-Sabbagh, O.; Shabaan, M.A.; Kadry, H.H.; Al-Din, E.S. New octahydroquinazoline derivatives: Synthesis and hypotensive activity. Eur. J. Med. Chem. 2010, 45, 5390–5396. [Google Scholar] [CrossRef]
- De Oliveira, K.N.; Costa, P.; Santin, J.R.; Mazzambani, L.; Bürger, C.; Mora, C.; Nunes, R.J.; De Souza, M.M. Synthesis and antidepressant-like activity evaluation of sulphonamides and sulphonyl-hydrazones. Bioorg. Med. Chem. 2011, 19, 4295–4306. [Google Scholar] [CrossRef] [PubMed]
- Ergenç, N.; Günay, N.S. Synthesis and antidepressant evaluation of new 3-phenyl-5-sulfonamidoindole derivatives. Eur. J. Med. Chem. 1998, 33, 143–148. [Google Scholar] [CrossRef]
- Shtyrlin, V.; Khaziev, R.M.; Shtyrlin, V.G.; Gilyazetdinov, E.M.; Agafonova, M.N.; Usachev, K.S.; Islamov, D.R.; Klimovitskii, A.E.; Vinogradova, T.I.; Dogonadze, M.Z.; et al. Isonicotinoyl hydrazones of pyridoxine derivatives: Synthesis and antimycobacterial activity. Med. Chem. Res. 2021, 30, 952–963. [Google Scholar] [CrossRef]
- Aljuhani, A.; Rezki, N.; Al-Sodies, S.; Messali, M.; ElShafei, G.M.S.; Hagar, M.; Aouad, M.R. Dicationic Bis-Pyridinium Hydrazone-Based Amphiphiles Encompassing Fluorinated Counteranions: Synthesis, Characterization, TGA-DSC, and DFT Investigations. Molecules 2022, 27, 2492. [Google Scholar] [CrossRef]
- Ali, A.; Khalid, M.; Abid, S.; Tahir, N.M.; Ashfaq, M.; Kanwal, F.; Lu, C.; Rehman, M.F. Green Synthesis, SC-XRD, Non-Covalent Interactive Potential and Electronic Communication via DFT Exploration of Pyridine-Based Hydrazone. Crystals 2020, 10, 778. [Google Scholar] [CrossRef]
- Ali, A.; Khalid, M.; Abid, S.; Iqbal, J.; Tahir, N.M.; Raza, A.R.; Zukerman-Schpector, J.; Paixão, M.W. Facile synthesis, crystal growth, characterization and computational study of new pyridine-based halogenated hydrazones: Unveiling the stabilization behavior in terms of noncovalent interactions. Appl. Organometal Chem. 2020, 34, e5399. [Google Scholar] [CrossRef]
- Nogueira, T.C.M.; Cruz, L.S.; Lourenço, M.C.; de Souza, M.V.N. Design, Synthesis and Anti-tuberculosis Activity of Hydrazones and N-acylhydrazones Containing Vitamin B6 and Different Heteroaromatic Nucleus. Lett. Drug Des. Discov. 2019, 16, 792–798. [Google Scholar] [CrossRef]
- Alptüzün, V.; Parlar, S.; Taşli, H.; Erciyas, E. Synthesis and antimicrobial activity of some pyridinium salts. Molecules 2009, 14, 5203–5215. [Google Scholar] [CrossRef]
- Parlar, S.; Erzurumlu, Y.; Ilhan, R.; Ballar, P.; Kırmızıbayrak, P.B.; Alptüzün, V.; Erciyas, E. Synthesis and evaluation of pyridinium-hydrazone derivatives as potential antitumoral agents. Chem. Biol. Drug Des. 2018, 92, 1198–1205. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, T.; Milton, M.D. Logic gate based novel phenothiazine-pyridylhydrazones: Halochromism in solid and solution state. Dye. Pigment. 2019, 164, 305–318. [Google Scholar] [CrossRef]
- Mali, S.N.; Thorat, B.R.; Gupta, D.R.; Pandey, A. Mini-Review of the Importance of Hydrazides and Their Derivatives—Synthesis and Biological Activity. Eng. Proc. 2021, 11, 21. [Google Scholar]
- Mandewale, M.C.; Thorat, B.; Nivid, Y.; Jadhav, R.; Nagarsekar, A.; Yamgar, R. Synthesis, structural studies and antituberculosis evaluation of new hydrazone derivatives of quinolone and their Zn(II) complexes. J. Saudi Chem. Soc. 2018, 22, 218–228. [Google Scholar] [CrossRef]
- Pisk, J.; Đilović, I.; Hrenar, T.; Cvijanović, D.; Pavlović, G.; Vrdoljak, V. Effective methods for the synthesis of hydrazones, quinazolines, and Schiff bases: Reaction monitoring using a chemometric approach. RCS Adv. 2020, 10, 38566–38577. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003, 9, 70–71. [Google Scholar] [CrossRef]
- Hansen, B.B.; Spittle, S.; Chen, B.; Poe, D.; Zhang, Y.; Klein, J.M.; Horton, A.; Adhikari, L.; Zelovich, T.; Doherty, B.W.; et al. Deep Eutectic Solvents: A Review of Fundamentals and Applications. Chem. Rev. 2021, 121, 1232–1285. [Google Scholar] [CrossRef]
- Smith, E.L.; Abbott, A.P.; Ryder, K.S. Deep Eutectic Solvents (DESs) and Their Applications. Chem. Rev. 2014, 114, 11060–11082. [Google Scholar] [CrossRef]
- El Achkar, T.; Greige-Gerges, H.; Fourmentin, S. Basics and properties of deep eutectic solvents: A review. Environ. Chem. Lett. 2021, 19, 3397–3408. [Google Scholar] [CrossRef]
- Lomba, L.; Ribate, M.P.; Sangüesa, E.; Concha, J.; Garralaga, M.P.; Errazquin, D.; García, C.B.; Giner, B. Deep Eutectic Solvents: Are They Safe? Appl. Sci. 2021, 11, 10061. [Google Scholar] [CrossRef]
- Vieira Sanches, M.; Freitas, R.; Oliva, M.; Mero, A.; De Marchi, L.; Cuccaro, A.; Fumagalli, G.; Mezzetta, A.; Dugoni, G.C.; Ferro, M.; et al. Are natural deep eutectic solvents always a sustainable option? A bioassay-based study. Environ. Sci. Pollut. Res. 2023, 30, 17268–17279. [Google Scholar] [CrossRef]
- Lapeña, D.; Errazquin, D.; Lomba, L.; Lafuente, C.; Giner, B. Ecotoxicity and biodegradability of pure and aqueous mixtures of deep eutectic solvents: Glyceline, ethaline and reline. Environ. Sci. Pollut. Res. 2021, 28, 8812–8821. [Google Scholar] [CrossRef] [PubMed]
- Wen, Q.; Chen, J.X.; Tang, Y.-L.; Wang, J.; Yang, Z. Assessing the toxicity and biodegradability of deep eutectic solvents. Chemosphere 2015, 132, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Kudlak, B.; Owczarek, K.; Namiesnik, J. Selected issues related to the toxicity of ionic liquids and deep eutectic solvents-a review. Environ. Sciand Pollut. Res. 2015, 22, 11975–11992. [Google Scholar] [CrossRef] [PubMed]
- Juneidi, I.; Hayyan, M.; Hashim, M.A. Intensification of biotransformations using deep eutectic solvents: Overview and outlook. Process Biochem. 2018, 66, 33–60. [Google Scholar] [CrossRef]
- Marchel, M.; Cieśliński, H.; Boczkaj, G. Thermal Instability of Choline Chloride-Based Deep Eutectic Solvents and Its Influence on Their Toxicity─Important Limitations of DESs as Sustainable Materials. Ind. Eng. Chem. Res. 2022, 61, 11288–11300. [Google Scholar] [CrossRef]
- Chen, W.J.; Xue, Z.M.; Xue, J.F.; Wang, J.Y.; Jiang, X.H.; Zhao, T.C.; Mu, T. Investigation on the Thermal Stability of Deep Eutectic Solvents. Acta Phys.-Chim. Sin. 2018, 34, 904–911. [Google Scholar] [CrossRef]
- García-Álvarez, J. Deep Eutectic Mixtures: Promising Sustainable Solvents for Metal-Catalysed and Metal-Mediated Organic Reactions. Eur. J. Inorg. Chem. 2015, 2015, 5147–5157. [Google Scholar] [CrossRef]
- Nolan, M.D.; Mezzetta, A.; Lorenzo Guazzelli, L.; Scanlan, E.M. Radical-mediated thiol–ene ‘click’ reactions in deep eutectic solvents for bioconjugation. Green. Chem. 2022, 24, 1456–1462. [Google Scholar] [CrossRef]
- Pedro, S.N.; Freire, C.R.S.; Silvestre, A.J.D.; Freire, M.G. Deep Eutectic Solvents and Pharmaceuticals. Encyclopedia 2021, 1, 942–963. [Google Scholar] [CrossRef]
- Sheldon, R.A. Biocatalysis and Biomass Conversion in Alternative Reaction Media. Chem.—A Eur. J. 2016, 22, 12984–12999. [Google Scholar] [CrossRef] [PubMed]
- Cicco, L.; Ríos-Lombardía, N.; Rodríguez-Álvarez, M.J.; Morís, F.; Perna, F.M.; Capriati, V.; García-Álvarez, J.; González-Sabín, J. Programming cascade reactions interfacing biocatalysis with transition-metal catalysis in Deep Eutectic Solvents as biorenewable reaction media. Green. Chem. 2018, 20, 3468–3475. [Google Scholar] [CrossRef]
- Martínez, R.; Berbegal, L.; Guillena, G.; Ramón, D.J. Bio-renewable enantioselective aldol reaction in natural deep eutectic solvents. Green. Chem. 2016, 18, 1724–1730. [Google Scholar] [CrossRef]
- Massolo, E.; Palmieri, S.; Benaglia, M.; Capriati, V.; Perna, F.M. Stereoselective organocatalysed reactions in deep eutectic solvents: Highly tunable and biorenewable reaction media for sustainable organic synthesis. Green. Chem. 2016, 18, 792–797. [Google Scholar] [CrossRef]
- Millia, L.; Dall’Asta, V.; Ferrara, C.; Berbenni, V.; Quartarone, E.; Perna, F.M.; Capriati, V.; Mustarelli, P. Bio-inspired choline chloride-based deep eutectic solvents as electrolytes for lithium-ion batteries. Solid State Ion. 2018, 323, 44–48. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Nguyen, C.T.; Tran, P.H. Synthesis of a new series of 2-hydroxy-5-iodo-N’-(1-arylethylidene)benzohydrazides using a deep eutectic solvent as solvent/catalyst under sonication. Heliyon 2019, 5, e02353. [Google Scholar] [CrossRef] [PubMed]
- Hammond, O.S.; Bowron, D.T.; Edler, K.J. The Effect of Water upon Deep Eutectic Solvent Nanostructure: An Unusual Transition from Ionic Mixture to Aqueous Solution. Angew. Chem. Int. Ed. Engl. 2017, 56, 9782–9785. [Google Scholar] [CrossRef]
- Protsenko, V.S.; Bobrova, L.S.; Baskevich, A.S.; Korniy, S.A.; Danilov, F.I. Electrodeposition of chromium coatings from a choline chloride based ionic liquid with the addition of water. J. Chem. Technol. Metall. 2018, 53, 906–915. [Google Scholar]
- McCalman, D.C.; Sun, L.; Zhang, Y.; Brennecke, J.F.; Maginn, E.J.; Schneider, W.F. Speciation, conductivities, diffusivities, and electrochemical reduction as a function of water content in mixtures of hydrated chromium chloride/choline chloride. J. Phys. Chem. B 2015, 119, 6018–6023. [Google Scholar] [CrossRef] [PubMed]
- Bušić, V.; Gašo-Sokač, D. Menshutkin Reaction in Choline Chloride-based Deep Eutectic Solvents. Org. Prep. Proced. Int. 2022, 55, 160–166. [Google Scholar] [CrossRef]
- Florindo, C.; Oliveira, F.S.; Rebelo, L.P.N.; Fernandes, A.M.; Marrucho, I.M. Insights into the Synthesis and Properties of Deep Eutectic Solvents Based on Cholinium Chloride and Carboxylic Acids. ACS Sustain. Chem. Eng. 2014, 2, 2416–2425. [Google Scholar] [CrossRef]
- Nolasco, M.N.; Pedro, S.N.; Vilela, C.; Vaz, P.D.; Ribeiro-Claro, P.; Rudić, S.; Parker, S.F.; Freire, C.R.S.; Freire, M.G.; Silvestre, A.D.J. Water in Deep Eutectic Solvents: New Insights from Inelastic Neutron Scattering Spectroscopy. Front. Phys. 2022, 10, 834571. [Google Scholar] [CrossRef]
- Gabriele, F.; Chiarini, M.; Germani, R.; Tiecco, M.; Spreti, N. Effect of Water Addition on Choline Chloride/glycol Deep Eutectic Solvents: Characterization of Their Structural and Physicochemical Properties. J. Mol. Liq. 2019, 291, 111301. [Google Scholar] [CrossRef]
- Nayyar, A.; Malde, A.; Coutinho, E.; Jain, R. Synthesis, antituberculosis activity, and 3D-QSAR study of ring-substituted-2/4-quinolinecarbaldehyde derivatives. Bioorg. Med. Chem. 2006, 14, 7302–7310. [Google Scholar] [CrossRef] [PubMed]
- Puskullu, M.O.; Shirinzadeh, H.; Nenni, M.; Gurer-Orhan, H.; Suzen, S. Synthesis and evaluation of antioxidant activity of new quinoline-2-carbaldehyde hydrazone derivatives: Bioisosteric melatonin analogues. J. Enzym. Inhib. Med. Chem. 2016, 31, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez Rodriguez, N.; van den Bruinhorst, A.; Kollau, J.B.M.L.; Kroon, C.M.; Binnemans, K. Degradation of deep-eutectic solvents based on choline chloride and carboxylic acids. ACS Sustain. Chem. Eng. 2019, 7, 11521–11528. [Google Scholar] [CrossRef]

| HBD in Prepared DES with Applied Molar Ratio | wt */% | Yields in DESs (ChCl:HBDs) per Compound/% | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | ||
| urea (1:2) | 0.99 | 21 | 30 | 40 | 57 | 20 | 31 | 41 | 44 | 17 | 43 | 50 | 37 | 48 | 11 |
| N-methylurea (1:3) | 1.86 | 35 | 68 | 34 | 53 | 19 | 15 | 58 | 50 | 24 | 56 | 54 | 46 | 56 | 18 |
| thiourea (1:2) | 4.84 | 48 | 54 | 46 | 57 | 36 | 34 | 52 | 39 | 27 | 63 | 51 | 54 | 62 | 23 |
| glycerol (1:2) | 0.76 | 51 | 60 | 47 | 50 | 43 | 36 | 61 | 60 | 34 | 48 | 65 | 78 | 68 | 24 |
| acetamide (1:2) | 1.86 | 57 | 22 | 37 | 59 | 36 | 51 | 32 | 67 | 15 | 37 | 30 | 67 | 58 | 16 |
| malic acid (1:1) | 0.79 | 61 | 65 | 62 | 52 | 40 | 38 | 45 | 54 | 22 | 36 | 54 | 57 | 63 | 37 |
| citric acid (1:2) | 1.05 | 65 | 58 | 59 | 48 | 39 | 26 | 56 | 56 | 34 | 66 | 65 | 55 | 63 | 26 |
| malonic acid (1:1) | 1.07 | 58 | 70 | 60 | 37 | 36 | 54 | 71 | 45 | 33 | 72 | 76 | 70 | 69 | 31 |
| oxalic acid (1:1) | 1.92 | 65 | 76 | 72 | 64 | 42 | 44 | 80 | 60 | 32 | 75 | 79 | 72 | 75 | 42 |
| lactic acid (1:2) | 6.58 | 47 | 34 | 45 | 46 | 32 | 42 | 64 | 56 | 24 | 50 | 59 | 53 | 70 | 28 |
| levulinic acid (1:2) | 0.83 | 63 | 82 | 78 | 56 | 34 | 45 | 79 | 69 | 36 | 61 | 80 | 58 | 70 | 38 |
| trans-cinnamic acid (1: 1) | 0.92 | 62 | 68 | 72 | 62 | 58 | 50 | 70 | 45 | 45 | 47 | 78 | 60 | 57 | 56 |
| HBD in Prepared DES with Applied Molar Ratio | Yields in DESs (ChCl:HBDs) per Compound/% | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | |
| urea (1:2) | 70 | 74 | 60 | 69 | 60 | 70 | 89 | 67 | 73 | 64 | 69 | 82 | 70 | 45 |
| N-methylurea (1:3) | 57 | 71 | 70 | 65 | 59 | 35 | 64 | 66 | 79 | 80 | 71 | 81 | 73 | 67 |
| thiourea (1:2) | 60 | 78 | 66 | 62 | 54 | 64 | 75 | 73 | 64 | 78 | 67 | 67 | 70 | 63 |
| glycerol (1:2) | 68 | 75 | 57 | 78 | 49 | 46 | 76 | 82 | 59 | 58 | 73 | 87 | 65 | 55 |
| acetamide (1:2) | 70 | 34 | 45 | 49 | 76 | 62 | 78 | 70 | 32 | 56 | 80 | 71 | 68 | 34 |
| malic acid (1:1) | 71 | 83 | 80 | 66 | 58 | 59 | 60 | 76 | 40 | 38 | 72 | 67 | 74 | 42 |
| citric acid (1:2) | 68 | 80 | 75 | 72 | 66 | 47 | 48 | 57 | 46 | 71 | 43 | 56 | 73 | 45 |
| malonic acid (1:1) | 78 | 86 | 80 | 70 | 66 | 72 | 83 | 64 | 59 | 65 | 89 | 67 | 70 | 65 |
| oxalic acid (1:1) | 72 | 86 | 80 | 56 | 55 | 75 | 97 | 78 | 80 | 92 | 98 | 86 | 78 | 61 |
| lactic acid (1:2) | 54 | 80 | 64 | 65 | 46 | 72 | 78 | 89 | 60 | 80 | 77 | 67 | 59 | 51 |
| levulinic acid (1:2) | 70 | 81 | 88 | 67 | 70 | 82 | 91 | 95 | 56 | 79 | 90 | 47 | 85 | 40 |
| trans-cinnamic acid (1: 1) | 68 | 85 | 89 | 69 | 85 | 40 | 68 | 55 | 34 | 67 | 78 | 62 | 81 | 58 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bušić, V.; Roca, S.; Gašo-Sokač, D. Application of Choline Chloride-Based Deep Eutectic Solvents in the Synthesis of Hydrazones. Separations 2023, 10, 551. https://doi.org/10.3390/separations10110551
Bušić V, Roca S, Gašo-Sokač D. Application of Choline Chloride-Based Deep Eutectic Solvents in the Synthesis of Hydrazones. Separations. 2023; 10(11):551. https://doi.org/10.3390/separations10110551
Chicago/Turabian StyleBušić, Valentina, Sunčica Roca, and Dajana Gašo-Sokač. 2023. "Application of Choline Chloride-Based Deep Eutectic Solvents in the Synthesis of Hydrazones" Separations 10, no. 11: 551. https://doi.org/10.3390/separations10110551
APA StyleBušić, V., Roca, S., & Gašo-Sokač, D. (2023). Application of Choline Chloride-Based Deep Eutectic Solvents in the Synthesis of Hydrazones. Separations, 10(11), 551. https://doi.org/10.3390/separations10110551








