A Rapidly Growing Nodule on the Eyebrow of a Pediatric Patient
Abstract
1. Introduction
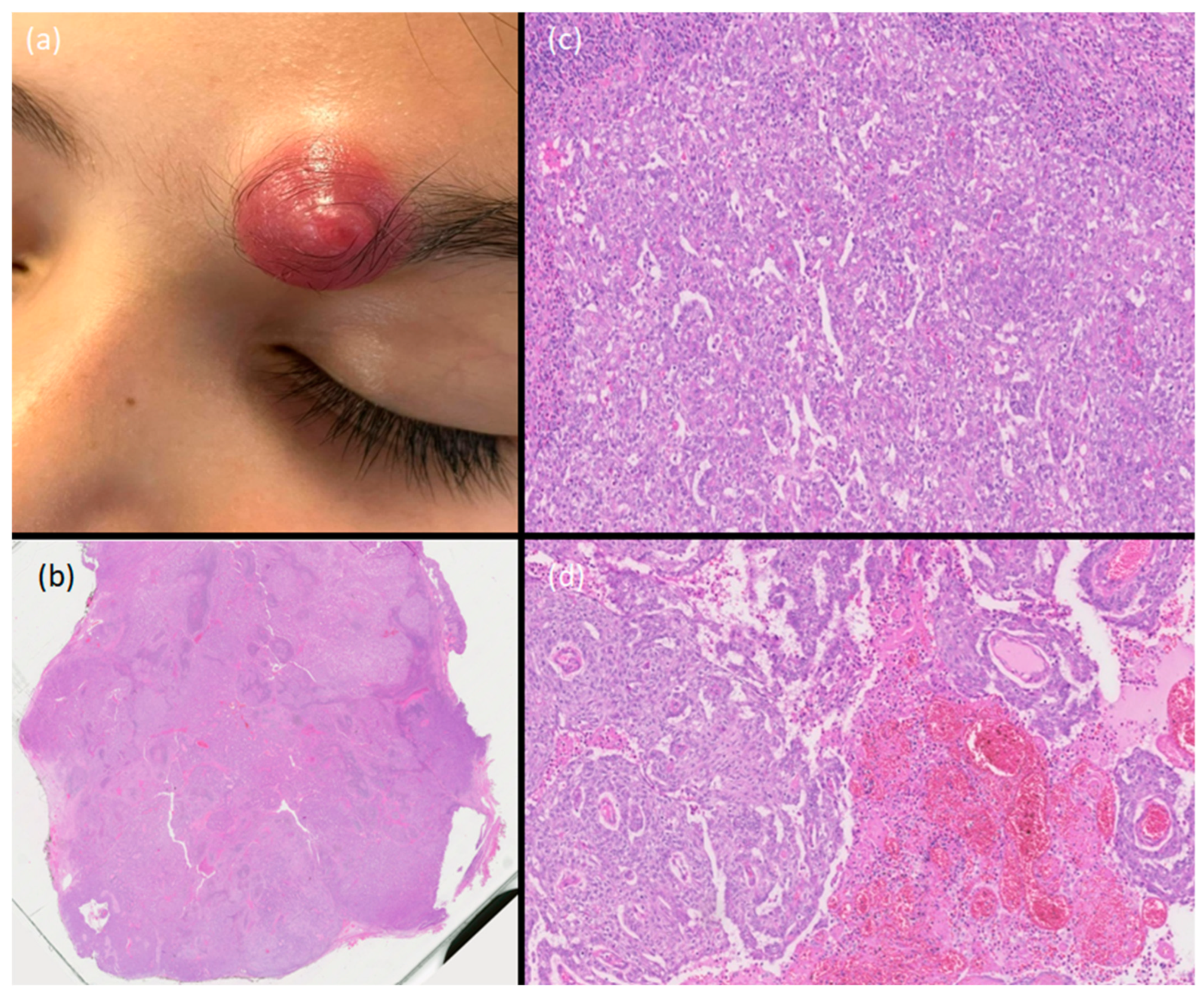
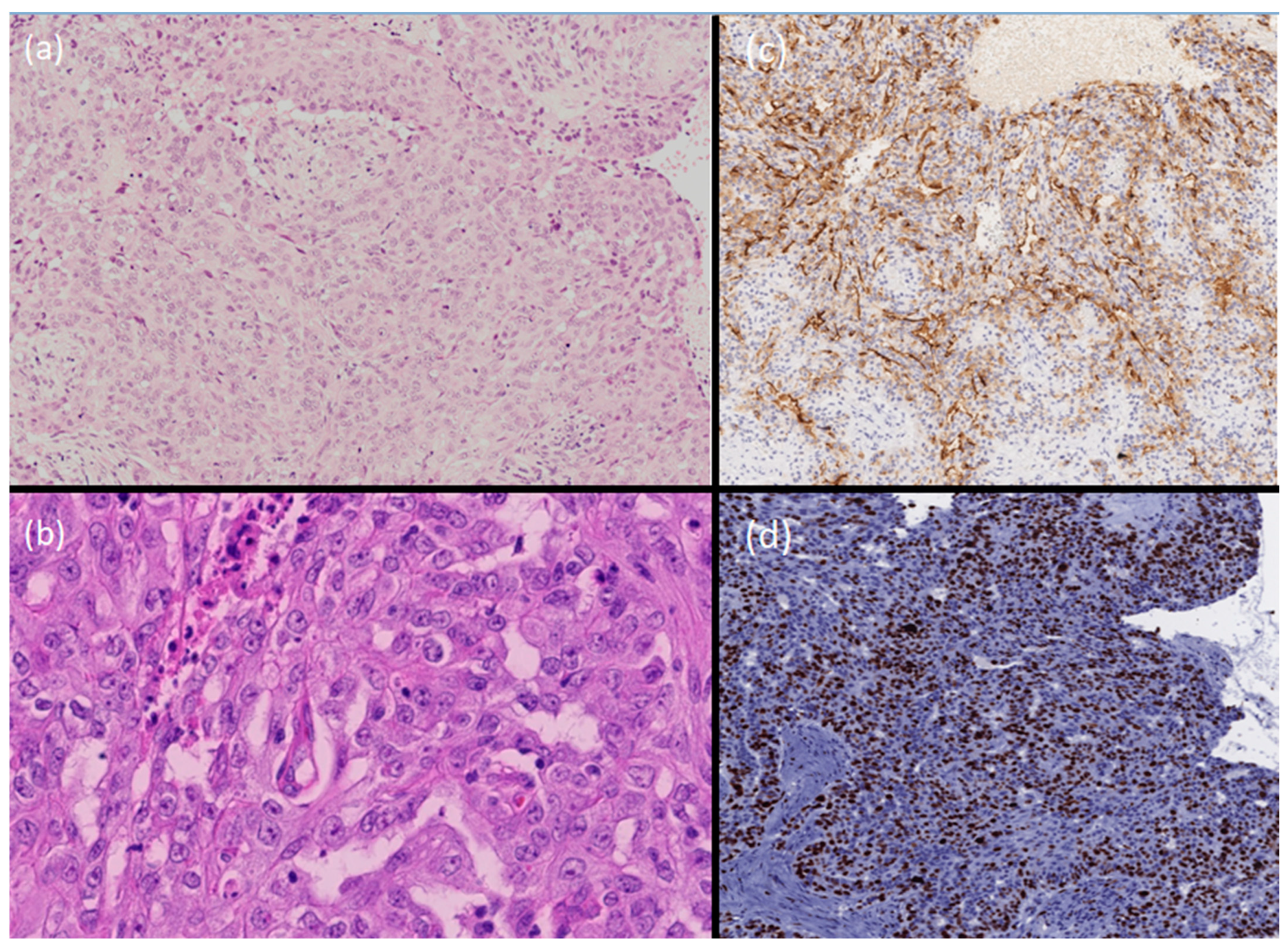
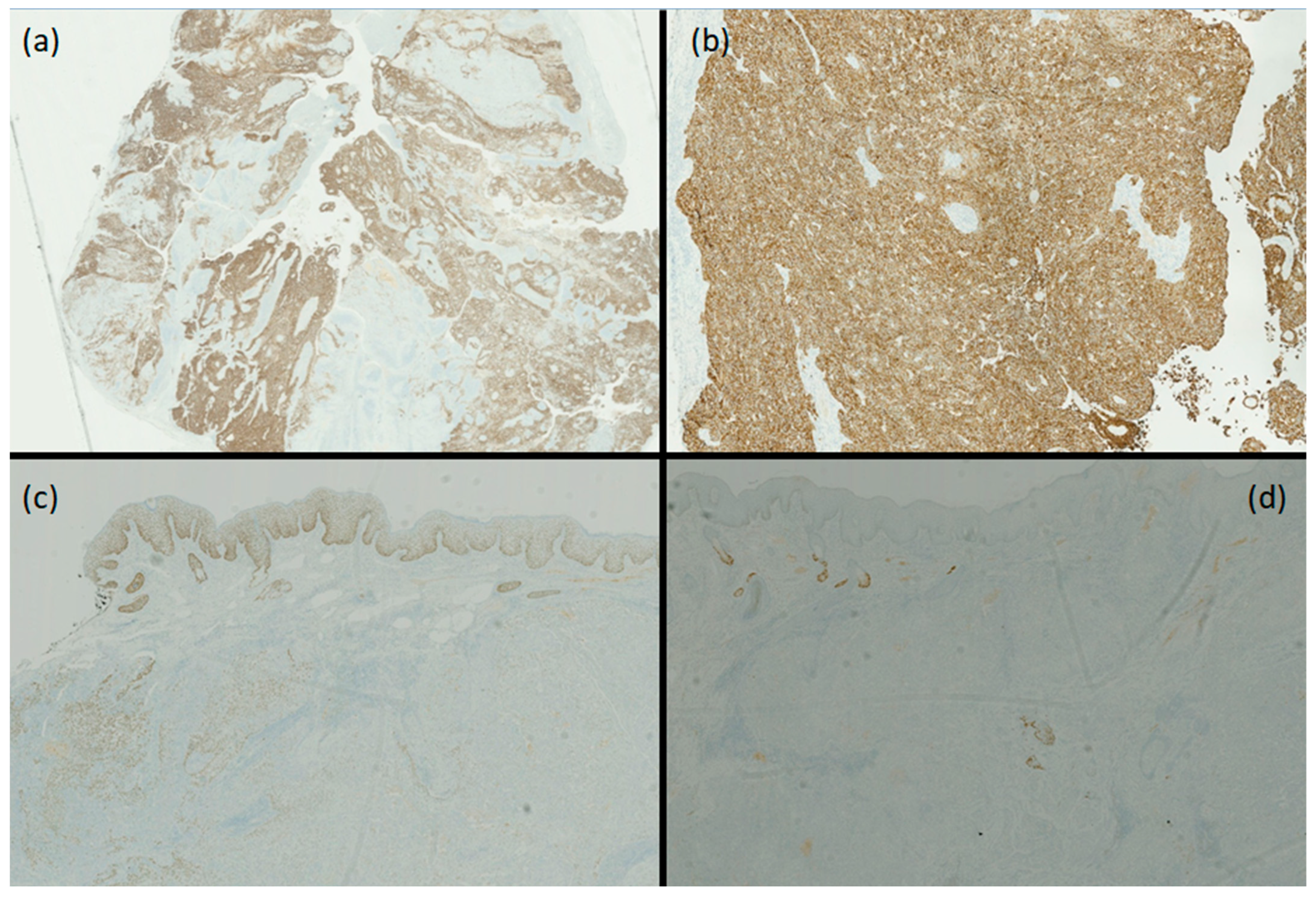
2. What Is the Diagnosis?
- (a)
- Hidradenoma;
- (b)
- Hidradenocarcinoma;
- (c)
- Clear-cell squamous cell carcinoma;
- (d)
- Porocarcinoma;
- (e)
- Trichilemmal carcinoma.
3. Diagnosis
- (b)
- Hidradenocarcinoma.
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moore, J.A.; Cui, S.; Berger, N.; Kim, S.; O’Guinn, D.; Labow, D.; Kamath, A. Hidradenocarcinoma: A rare but challenging diagnosis. Clin. Imaging 2021, 75, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Knops, A.M.; McClain, K.E.; Aaronson, N.L. Atypical Nodular Hidradenoma Versus Low-Grade Hidradenocarcinoma in a Young Female Patient: A Case Report and Review of Literature. Cureus 2022, 14, e32772. [Google Scholar] [CrossRef] [PubMed]
- Zorro, S.; Matias, R.; Sousa, C.; Aguiar, A. Malignant Transformation of Retroauricular Hidradenoma in Hidradenocarcinoma in a Nine-Year-Old Patient: A Case Report. Cureus 2023, 15, e37160. [Google Scholar] [CrossRef] [PubMed]
- Nazarian, R.M.; Kapur, P.; Rakheja, D.; Piris, A.; Duncan, L.M.; Mihm, M.C., Jr.; Hoang, M.P. Atypical and malignant hidradenomas: A histological and immunohistochemical study. Mod. Pathol. 2009, 22, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Gauerke, S.; Driscoll, J.J. Hidradenocarcinomas: A Brief Review and Future Directions. Arch. Pathol. Lab. Med. 2010, 134, 781–785. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Pan, S.; Li, M.; Su, R. Prognostic analysis of hidradenocarcinoma: A SEER-based observational study. Ann. Med. 2022, 54, 454–463. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Published by MDPI on behalf of the European Society of Dermatopathology. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aromolo, I.F.; Brena, M.; Monzani, N.A.; Caviggioli, F.; Berti, E.; Micello, D.; Cavalli, R. A Rapidly Growing Nodule on the Eyebrow of a Pediatric Patient. Dermatopathology 2024, 11, 266-271. https://doi.org/10.3390/dermatopathology11040028
Aromolo IF, Brena M, Monzani NA, Caviggioli F, Berti E, Micello D, Cavalli R. A Rapidly Growing Nodule on the Eyebrow of a Pediatric Patient. Dermatopathology. 2024; 11(4):266-271. https://doi.org/10.3390/dermatopathology11040028
Chicago/Turabian StyleAromolo, Italo Francesco, Michela Brena, Nicola Adriano Monzani, Fabio Caviggioli, Emilio Berti, Donata Micello, and Riccardo Cavalli. 2024. "A Rapidly Growing Nodule on the Eyebrow of a Pediatric Patient" Dermatopathology 11, no. 4: 266-271. https://doi.org/10.3390/dermatopathology11040028
APA StyleAromolo, I. F., Brena, M., Monzani, N. A., Caviggioli, F., Berti, E., Micello, D., & Cavalli, R. (2024). A Rapidly Growing Nodule on the Eyebrow of a Pediatric Patient. Dermatopathology, 11(4), 266-271. https://doi.org/10.3390/dermatopathology11040028





