From Microbubbles to Nanobubbles: Effect on Flotation
Abstract
:1. Flotation—Introduction
2. Fundamentals
3. Wastewater Treatment
4. Nanobubbles
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Matis, K.A.; Zouboulis, A.I. Flotation Science and Engineering. In Flotation Science and Engineering; Matis, K.A., Ed.; Marcel Dekker: New York, NY, USA, 1995. [Google Scholar]
- Burns, S.E.; Yiacoumi, S.; Tsouris, C. Microbubble generation for environmental and industrial separations. Sep. Purif. Technol. 1997, 11, 221–232. [Google Scholar] [CrossRef]
- Rodrigues, R.T.; Rubio, J. DAF-dissolved air flotation: Potential applications in the mining and mineral processing industry. Int. J. Miner. Process. 2007, 82, 1–13. [Google Scholar] [CrossRef]
- Szpyrkowicz, L. Hydrodynamic effects on the performance of electro-coagulation/electro- flotation for the removal of dyes from textile wastewater. Ind. Eng. Chem. Res. 2005, 44, 7844–7853. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Matis, K.A. Methods of arsenic wastes recycling: Focus on flotation. J. Mol. Liq. 2016, 214, 37–45. [Google Scholar] [CrossRef]
- Peleka, E.N.; Matis, K.A. Water separation processes and sustainability. Ind. Eng. Chem. Res. 2011, 50, 421–430. [Google Scholar] [CrossRef]
- Yenial, Ü.; Bulut, G. Examination of flotation behavior of metal ions for process water remediation. J. Mol. Liq. 2017, 241, 130–135. [Google Scholar] [CrossRef]
- Edzwald, J.K. Dissolved air flotation and me. Water Res. 2010, 44, 2077–2106. [Google Scholar] [CrossRef] [PubMed]
- Matis, K.A.; Mavros, P. Recovery of metals by ion flotation from dilute aqueous solutions. Sep. Purif. Methods 1991, 20, 1–48. [Google Scholar] [CrossRef]
- Simões, C.R.; Hacha, R.R.; Merma, A.G.; Torem, M.L. On the recovery of hematite from an iron ore fine fraction by electroflotation using a biosurfactant. Minerals 2020, 10, 1057. [Google Scholar] [CrossRef]
- Peleka, E.N.; Matis, K.A. Hydrodynamic aspects of flotation separation. Open Chem. 2016, 14, 132–139. [Google Scholar] [CrossRef] [Green Version]
- Mesa, D.; Morrison, A.J.; Brito-Parada, P.R. The effect of impeller-stator design on bubble size: Implications for froth stability and flotation performance. Miner. Eng. 2020, 157, 106533. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, X.; Liu, H.; Zhang, H.; Miller, J.D. Some physicochemical aspects of water-soluble mineral flotation. Adv. Colloid Interface Sci. 2016, 235, 190–200. [Google Scholar] [CrossRef]
- Zhuo, Q.; Liu, W.; Zhang, H.; Zhang, W.; Cui, R. Effect of particle size on the relative motion between particles and bubbles. Colloids Surf. A Physicochem. Eng. Asp. 2020, 601, 124956. [Google Scholar] [CrossRef]
- Matis, K.A.; Gallios, G.P. Dissolved-air and electrolytic flotation. In Mineral Processing at a Crossroads; Wills, B.A., Barley, R.W., Eds.; Martinus Nijhoff: Dordrecht, Germany, 1986; pp. 37–70. [Google Scholar]
- Farrokhpay, S.; Filippov, L.; Fornasiero, D. Flotation of Fine Particles: A Review. Miner. Process. Extr. Metall. Rev. 2020, 1–11. [Google Scholar] [CrossRef]
- Rulyov, N.N.; Sadovskiy, D.Y.; Rulyova, N.A.; Filippov, L.O. Column flotation of fine glass beads enhanced by their prior heteroaggregation with microbubbles. Colloids Surf. A Physicochem. Eng. Asp. 2021, 617, 126398. [Google Scholar] [CrossRef]
- Da Mota, I.D.O.; de Castro, J.A.; de Góes Casqueira, R.; de Oliveira Junior, A.G. Study of electroflotation method for treatment of wastewater from washing soil contaminated by heavy metals. J. Mater. Res. Technol. 2015, 4, 109–113. [Google Scholar] [CrossRef] [Green Version]
- Kyzas, G.Z.; Matis, K.A. Electroflotation process: A review. J. Mol. Liq. 2016, 220, 657–664. [Google Scholar] [CrossRef]
- Shetty, S.; Chernyshova, I.V.; Ponnurangam, S. Foam flotation of rare earth elements by conventional and green surfactants. Miner. Eng. 2020, 158, 106585. [Google Scholar] [CrossRef]
- Milosavljević, M.M.; Marinković, A.D.; Rančić, M.; Milentijević, G.; Bogdanović, A.; Cvijetić, I.N.; Gurešić, D. New Eco-Friendly Xanthate-Based Flotation Agents. Minerals 2020, 10, 350. [Google Scholar] [CrossRef] [Green Version]
- Roberto, P.-G.; Arturo, B.-T. Coalescence of air bubbles: Effect of the electrical double layer. Miner. Eng. 2020, 150, 106301. [Google Scholar] [CrossRef]
- Brabcová, Z.; Karapantsios, T.; Kostoglou, M.; Basařová, P.; Matis, K. Bubble–particle collision interaction in flotation systems. Colloids Surf. A Physicochem. Eng. Asp. 2015, 473, 95–103. [Google Scholar] [CrossRef]
- Matis, K.A. Electrolytic flotation. In Innovations in Flotation Technology; Mavros, P., Matis, K.A., Eds.; Kluwer Academic: Dordrecht, The Netherlands, 1992; pp. 301–304. [Google Scholar]
- Swart, B.; Zhao, Y.; Khaku, M.; Che, E.; Maltby, R.; Chew, Y.J.; Wenk, J. In situ characterisation of size distribution and rise velocity of microbubbles by high-speed photography. Chem. Eng. Sci. 2020, 225, 115836. [Google Scholar] [CrossRef]
- Peleka, E.N.; Gallios, G.P.; Matis, K.A. A perspective on flotation: A review. J. Chem. Technol. Biotechnol. 2018, 93, 615–623. [Google Scholar] [CrossRef]
- Sebba, F. Ion Flotation. Available online: http://books.google.com/books?id=MYFKAAAAMAAJ (accessed on 10 May 2021).
- Zouboulis, A.I.; Matis, K.A. Ion flotation in environmental technology. Chemosphere 1987, 16, 623–631. [Google Scholar] [CrossRef]
- Matis, K.A.; Zouboulis, A.I. Flotation techniques in water technology for metals recovery: The impact of speciation. Sep. Sci. Technol. 2001, 36, 3777–3800. [Google Scholar] [CrossRef]
- Matis, K.A.; Lazaridis, N.K. Flotation techniques in water technology for metals recovery: Dispersed-air vs. dissolved-air flotation. J. Min. Metall. A Min. 2002, 38, 1–27. [Google Scholar]
- Batjargal, K.; Guven, O.; Ozdemir, O.; Karakashev, S.I.; Grozev, N.A.; Boylu, F.; Çelik, M.S. Adsorption Kinetics of Various Frothers on Rising Bubbles of Different Sizes under Flotation Conditions. Minerals 2021, 11, 304. [Google Scholar] [CrossRef]
- Panjipour, R.; Karamoozian, M.; Albijanic, B. Bubble size distributions in gas–liquid–solid systems and their influence on flotation separation in a bubble column. Chem. Eng. Res. Des. 2021, 167, 96–106. [Google Scholar] [CrossRef]
- Zhu, H.; Valdivieso, A.L.; Zhu, J.; Song, S.; Min, F.; Corona Arroyo, M.A. A study of bubble size evolution in Jameson flotation cell. Chem. Eng. Res. Des. 2018, 137, 461–466. [Google Scholar] [CrossRef]
- Jain, S. Activation in the flotation of sphalerite. Flotat. Sulphide Miner. 1985, 6, 159–174. [Google Scholar]
- Gallios, G.P.; Kydros, K.A.; Matis, K.A. Electrokinetic behaviour of sulphide minerals: A contribution to the chemistry of flotation. In Mineral Processing and the Environment; Gallios, G.P., Matis, K.A., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1998; pp. 25–42. [Google Scholar]
- Kyzas, G.Z.; Deliyanni, E.A.; Matis, K.A. Research activities related to flotation process. Trends Green Chem. 2015, 1, 1–5. [Google Scholar]
- Zamboulis, D.; Peleka, E.N.; Lazaridis, N.K.; Matis, K.A. Metal ion separation and recovery from environmental sources using various flotation and sorption techniques. J. Chem. Technol. Biotechnol. 2011, 86, 335–344. [Google Scholar] [CrossRef]
- Zouboulis, A.I.; Kydros, K.A.; Matis, K.A. Adsorbing Flotation of Copper Hydroxo Precipitates by Pyrite Fines. Sep. Sci. Technol. 1992, 27, 2143–2155. [Google Scholar] [CrossRef]
- Yu, L.; Liu, Q.; Li, S.; Deng, J.; Luo, B.; Lai, H. Adsorption performance of copper ions on arsenopyrite surfaces and implications for flotation. Appl. Surf. Sci. 2019, 488, 185–193. [Google Scholar] [CrossRef]
- Craioveanu, M.G.; Stoica, L.; Constantin, C.; Oprea, O. Cr(III)aq separation by flotation with multipolar collector. Sep. Sci. Technol. 2020, 55, 346–357. [Google Scholar] [CrossRef]
- Zouboulis, A.I.; Kydros, K.A.; Matis, K.A. Removal of hexavalent chromium anions from solutions by pyrite fines. Water Res. 1995, 29, 1755–1760. [Google Scholar] [CrossRef]
- Nazari, S.; Shafaei, S.Z.; Shahbazi, B.; Chelgani, S.C. Study relationships between flotation variables and recovery of coarse particles in the absence and presence of nanobubble. Colloids Surf. A Physicochem. Eng. Asp. 2018, 559, 284–288. [Google Scholar] [CrossRef]
- Matsuoka, K.; Hasegawa, S.; Yuma, T.; Goto, Y. Application of foam separation method for removal of alkaline earth metal ions from solution. J. Mol. Liq. 2019, 294, 111663. [Google Scholar] [CrossRef]
- Matis, K.A.; Peleka, E.N. Alternative flotation techniques for wastewater treatment: Focus on electroflotation. Sep. Sci. Technol. 2010, 45, 2465–2474. [Google Scholar] [CrossRef]
- Levenspiel, O. Mixed models to represent flow of fluids through vessels. Can. J. Chem. Eng. 1962, 40, 135–138. [Google Scholar] [CrossRef]
- Gaydukova, A.; Kolesnikov, V.; Aung, H.T. Electroflotosorption method for removing organic and inorganic impurities from wastewater. Sep. Purif. Technol. 2021, 267, 118682. [Google Scholar] [CrossRef]
- Tadesse, B.; Albijanic, B.; Makuei, F.; Browner, R. Recovery of Fine and Ultrafine Mineral Particles by Electroflotation—A Review. Miner. Process. Extr. Metall. Rev. 2019, 40, 108–122. [Google Scholar] [CrossRef]
- Hoseinian, F.S.; Rezai, B.; Safari, M.; Deglon, D.; Kowsari, E. Effect of hydrodynamic parameters on nickel removal rate from wastewater by ion flotation. J. Environ. Manag. 2019, 244, 408–414. [Google Scholar] [CrossRef]
- Gulden, S.J.; Riedele, C.; Kopf, M.H.; Nirschl, H. Potential of flotation as alternative separation process in biotechnology with focus on cost and energy efficiency. Chem. Eng. Sci. 2020, 218, 115117. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Matis, K.A. Flotation of biological materials. Processes 2014, 2, 293–310. [Google Scholar] [CrossRef]
- Matis, K.A.; Mavros, P. Foam/Froth Flotation. Sep. Purif. Methods 1991, 20, 163–198. [Google Scholar] [CrossRef]
- Nenov, V.; Lazaridis, N.K.; Blöcher, C.; Bonev, B.; Matis, K.A. Metal recovery from a copper mine effluent by a hybrid process. Chem. Eng. Process. Process Intensif. 2008, 47, 596–602. [Google Scholar] [CrossRef]
- Ahmed, S.F.; Mofijur, M.; Nuzhat, S.; Chowdhury, A.T.; Rafa, N.; Uddin, M.A.; Inayat, A.; Mahlia, T.M.I.; Ong, H.C.; Chia, W.Y.; et al. Recent developments in physical, biological, chemical, and hybrid treatment techniques for removing emerging contaminants from wastewater. J. Hazard. Mater. 2021, 416, 125912. [Google Scholar] [CrossRef]
- Zhang, M.; Yang, J.; Kang, Z.; Wu, X.; Tang, L.; Qiang, Z.; Zhang, D.; Pan, X. Removal of micron-scale microplastic particles from different waters with efficient tool of surface-functionalized microbubbles. J. Hazard. Mater. 2021, 404, 124095. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jin, X.; Yang, S.; Wang, G.; Xu, L.; Jin, P.; Shi, X.; Shi, Y. Interactions between flocs and bubbles in the separation zone of dissolved air flotation system. Sci. Total Environ. 2021, 761, 143222. [Google Scholar] [CrossRef]
- Kostoglou, M.; Karapantsios, T.D.; Matis, K.A. CFD model for the design of large scale flotation tanks for water and wastewater treatment. Ind. Eng. Chem. Res. 2007, 46, 6590–6599. [Google Scholar] [CrossRef]
- Nazari, A.M.; Cox, P.W.; Waters, K.E. Biosorptive flotation of copper ions from dilute solution using BSA-coated bubbles. Miner. Eng. 2015, 75, 140–145. [Google Scholar] [CrossRef]
- Hu, N.; Liu, W.; Jin, L.; Li, Y.; Li, Z.; Liu, G.; Huang, D.; Wu, Z.; Yin, H. Recovery of trace Cu2+ using a process of nano-adsorption coupled with flotation: SNP as an adsorbing carrier. Sep. Purif. Technol. 2017, 184, 257–263. [Google Scholar] [CrossRef]
- Khan, P.; Zhu, W.; Huang, F.; Gao, W.; Khan, N.A. Micro–nanobubble technology and water-related application. Water Supply 2020, 20, 2021–2035. [Google Scholar] [CrossRef]
- Liu, L.; Hu, S.; Wu, C.; Liu, K.; Weng, L.; Zhou, W. Aggregates characterizations of the ultra-fine coal particles induced by nanobubbles. Fuel 2021, 297, 120765. [Google Scholar] [CrossRef]
- Zhang, F.; Sun, L.; Yang, H.; Gui, X.; Schönherr, H.; Kappl, M.; Cao, Y.; Xing, Y. Recent advances for understanding the role of nanobubbles in particles flotation. Adv. Colloid Interface Sci. 2021, 291, 102403. [Google Scholar] [CrossRef] [PubMed]
- Sobhy, A.; Wu, Z.; Tao, D. Statistical analysis and optimization of reverse anionic hematite flotation integrated with nanobubbles. Miner. Eng. 2021, 163, 06799. [Google Scholar] [CrossRef]
- Pourkarimi, Z.; Rezai, B.; Noaparast, M.; Nguyen, A.V.; Chelgani, S.C. Proving the existence of nanobubbles produced by hydrodynamic cavitation and their significant effects in powder flotation. Adv. Powder Technol. 2021, 32, 1810–1818. [Google Scholar] [CrossRef]
- Li, C.; Li, X.; Xu, M.; Zhang, H. Effect of ultrasonication on the flotation of fine graphite particles: Nanobubbles or not? Ultrason. Sonochemistry 2020, 69, 105243. [Google Scholar] [CrossRef]
- Wu, L.; Han, Y.; Zhang, Q.; Zhao, S. Effect of external electric field on nanobubbles at the surface of hydrophobic particles during air flotation. RSC Adv. 2019, 9, 1792–1798. [Google Scholar] [CrossRef] [Green Version]
- Azevedo, A.; Etchepare, R.; Rubio, J. Raw water clarification by flotation with microbubbles and nanobubbles generated with a multiphase pump. Water Sci. Technol. 2017, 75, 2342–2349. [Google Scholar] [CrossRef] [PubMed]
- Etchepare, R.; Azevedo, A.; Calgaroto, S.; Rubio, J. Removal of ferric hydroxide by flotation with micro and nanobubbles. Sep. Purif. Technol. 2017, 184, 347–353. [Google Scholar] [CrossRef]
- Favvas, E.P.; Kyzas, G.Z.; Efthimiadou, E.K.; Mitropoulos, A.C. Bulk nanobubbles, generation methods and potential applications. Curr. Opin. Colloid Interface Sci. 2021, 54, 101455. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Favvas, E.P.; Kostoglou, M.; Mitropoulos, A.C. Effect of agitation on batch adsorption process facilitated by using nanobubbles. Colloids Surfaces A Physicochem. Eng. Asp. 2020, 607, 125440. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Bomis, G.; Kosheleva, R.I.; Efthimiadou, E.K.; Favvas, E.P.; Kostoglou, M.; Mitropoulos, A.C. Nanobubbles effect on heavy metal ions adsorption by activated carbon. Chem. Eng. J. 2019, 356, 91–97. [Google Scholar] [CrossRef]
- Michailidi, E.D.; Bomis, G.; Varoutoglou, A.; Kyzas, G.Z.; Mitrikas, G.; Mitropoulos, A.C.; Efthimiadou, E.K.; Favvas, E.P. Bulk nanobubbles: Production and investigation of their formation/stability mechanism. J. Colloid Interface Sci. 2020, 564, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Chang, G.; Xing, Y.; Zhang, F.; Yang, Z.; Liu, X.; Gui, X. Effect of nanobubbles on the flotation performance of oxidized coal. ACS Omega 2020, 5, 20283–20290. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Zhang, M.; Lei, W.; Yao, W.; Fan, R. Effect of Nanobubbles on the Slime Coating of Kaolinite in Coal Flotation. ACS Omega 2020, 5, 24773–24779. [Google Scholar] [CrossRef]
- Zhang, F.; Xing, Y.; Chang, G.; Yang, Z.; Cao, Y.; Gui, X. Enhanced lignite flotation using interfacial nanobubbles based on temperature difference method. Fuel 2021, 293, 120313. [Google Scholar] [CrossRef]
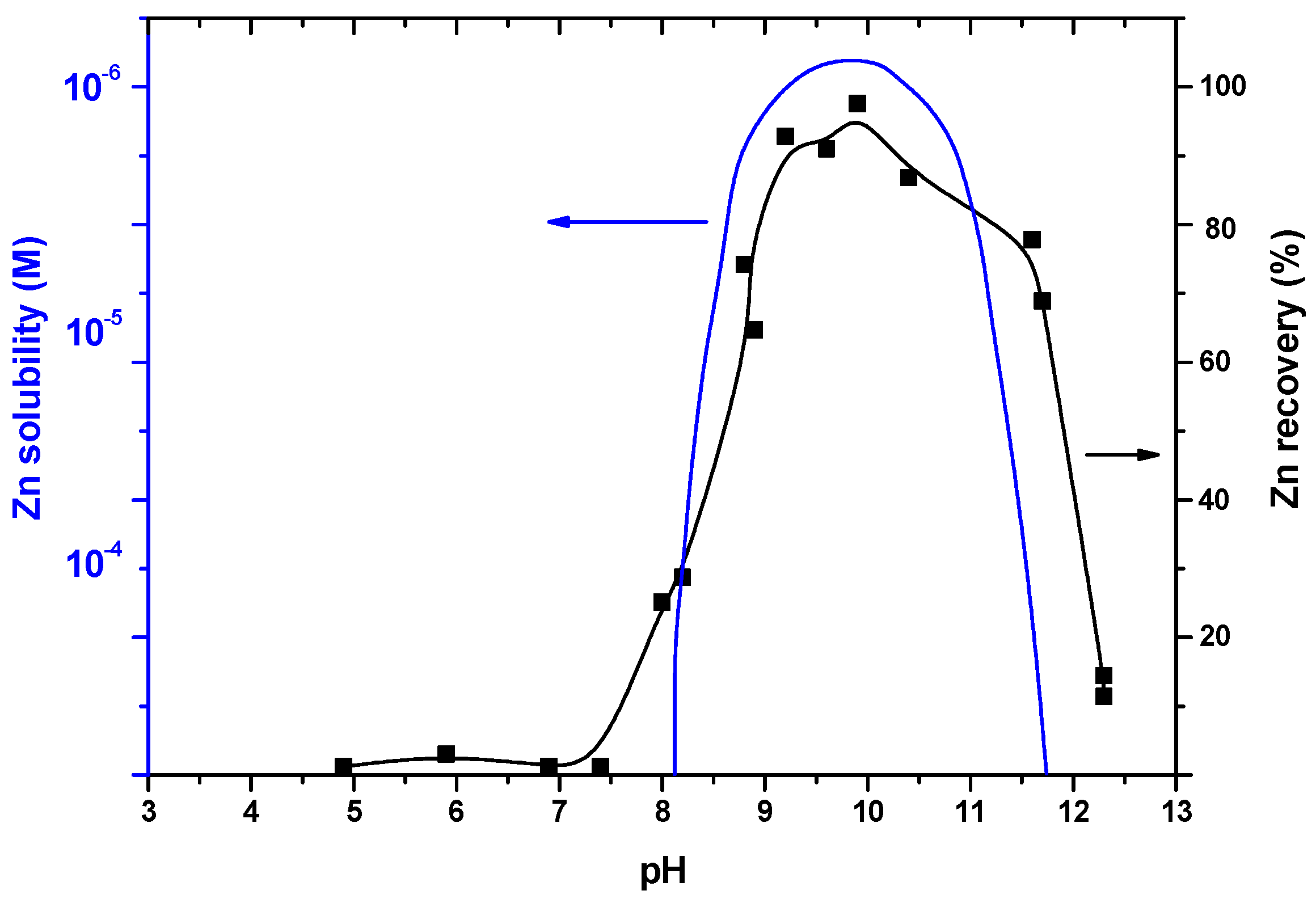
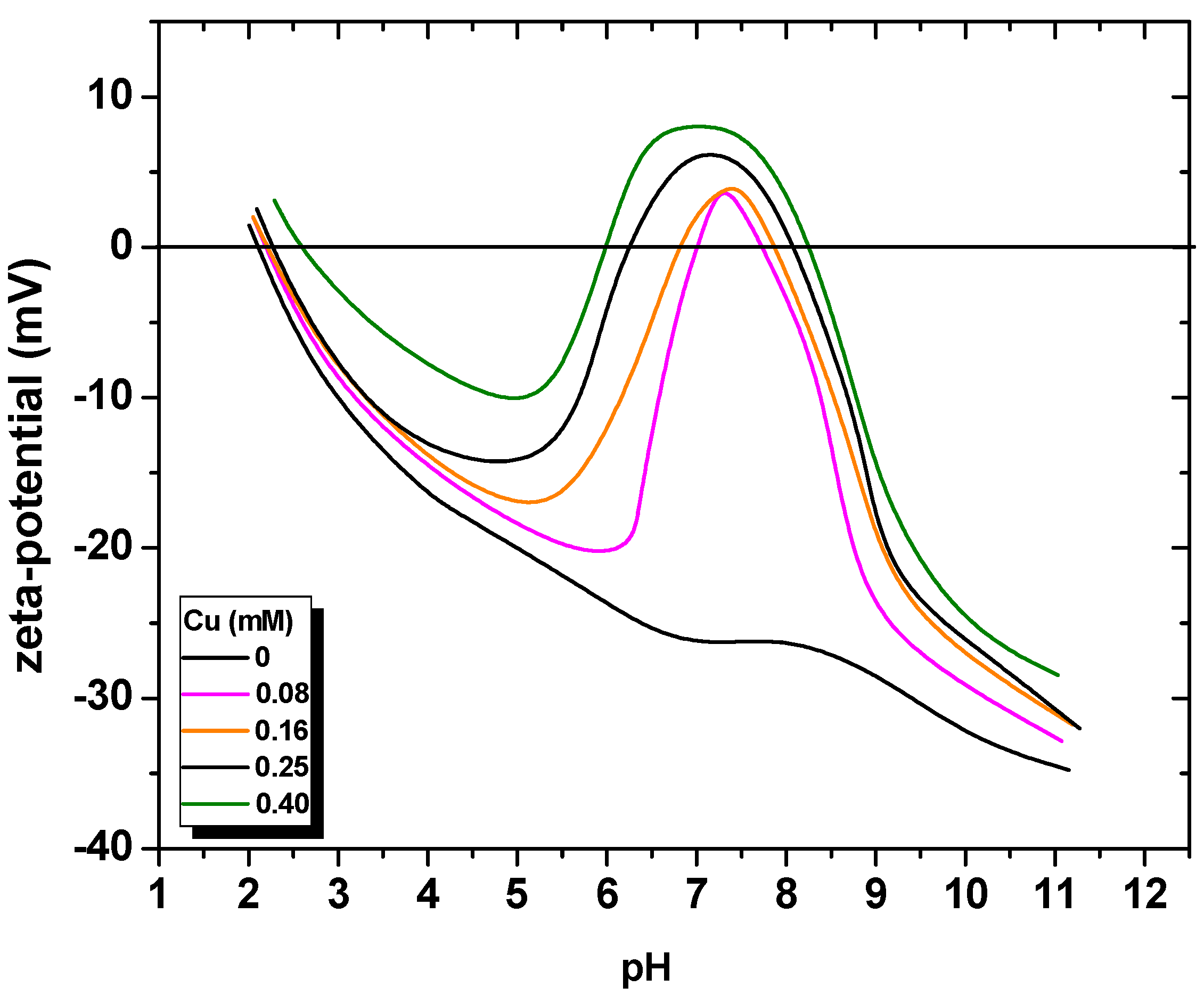
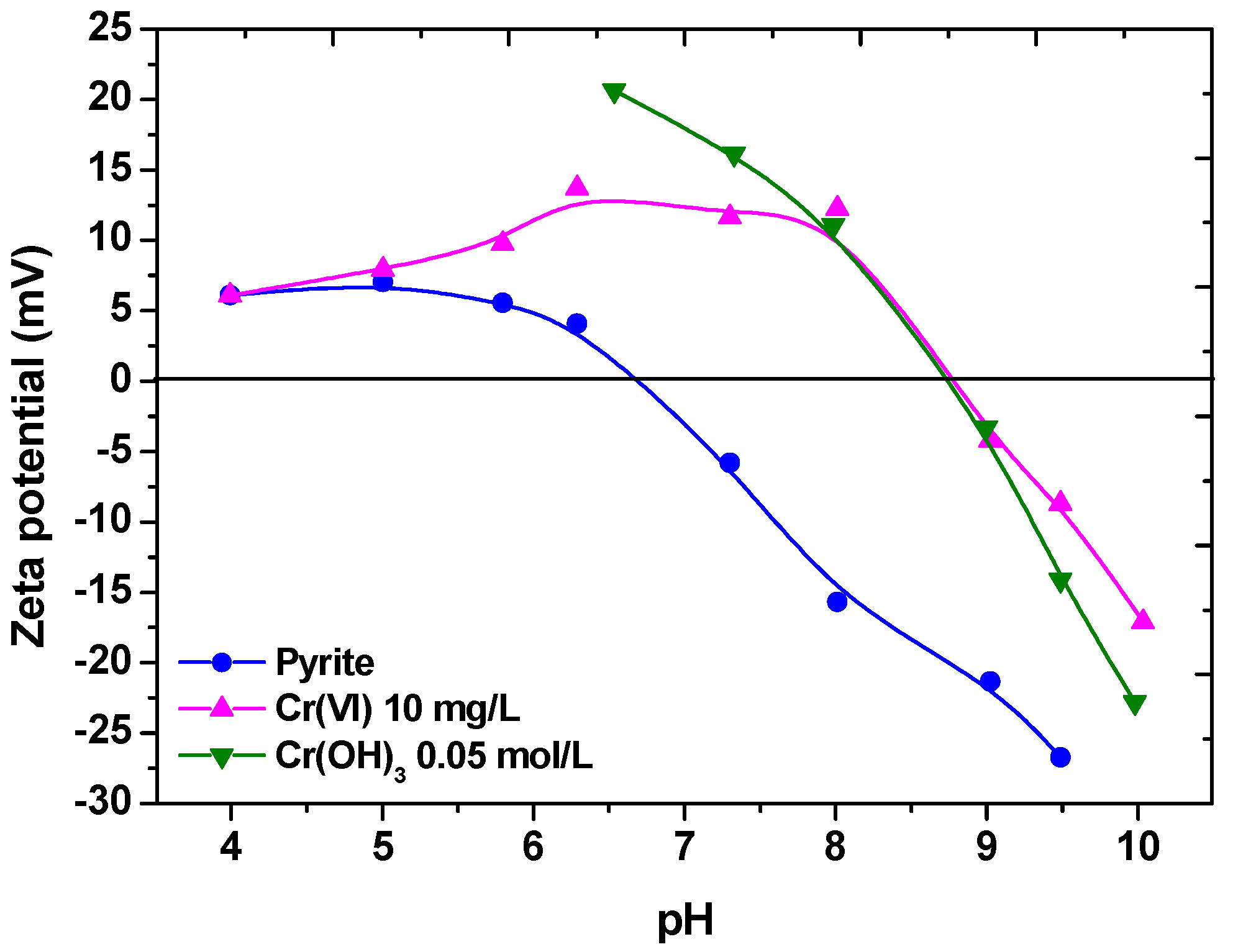
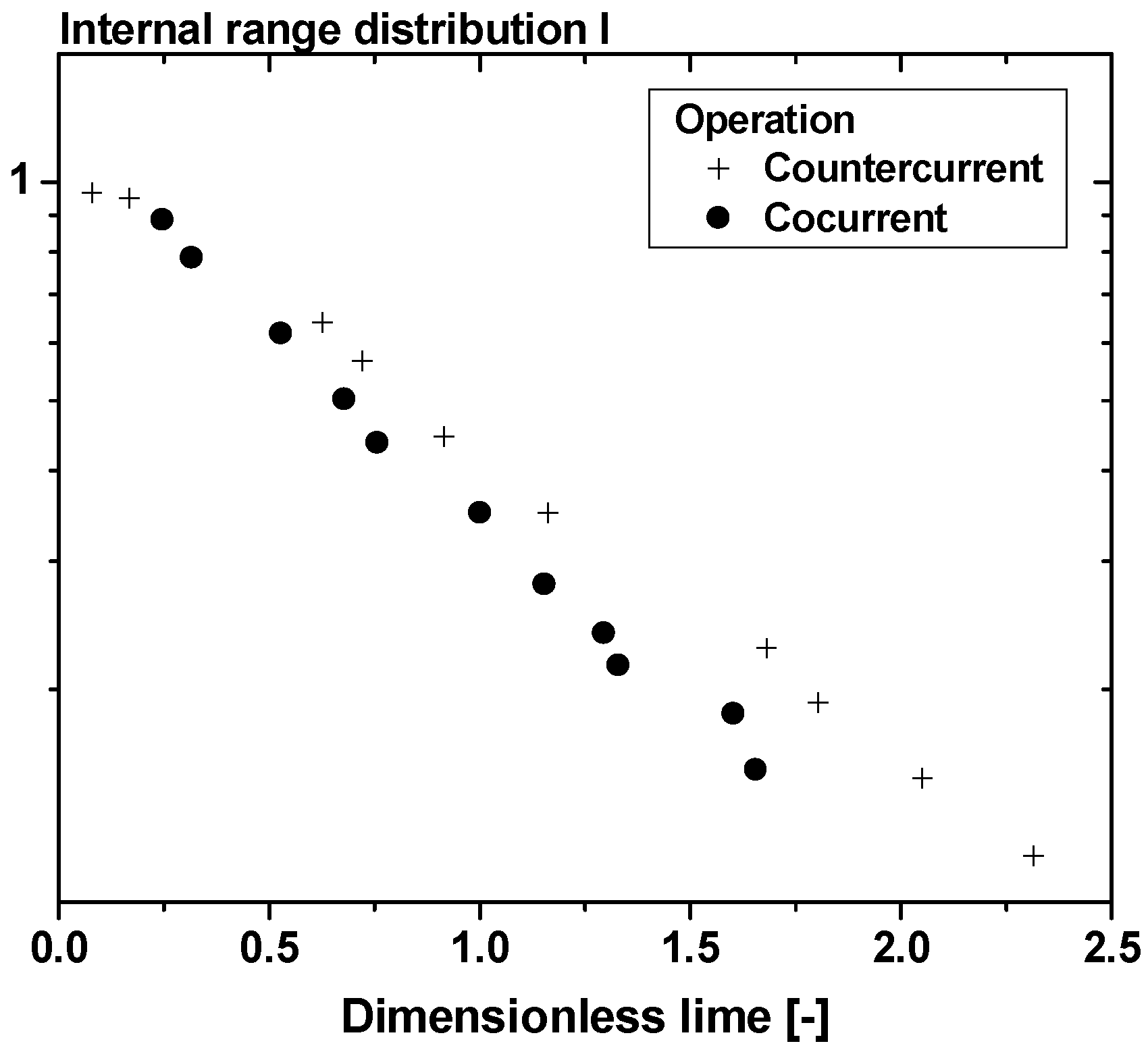
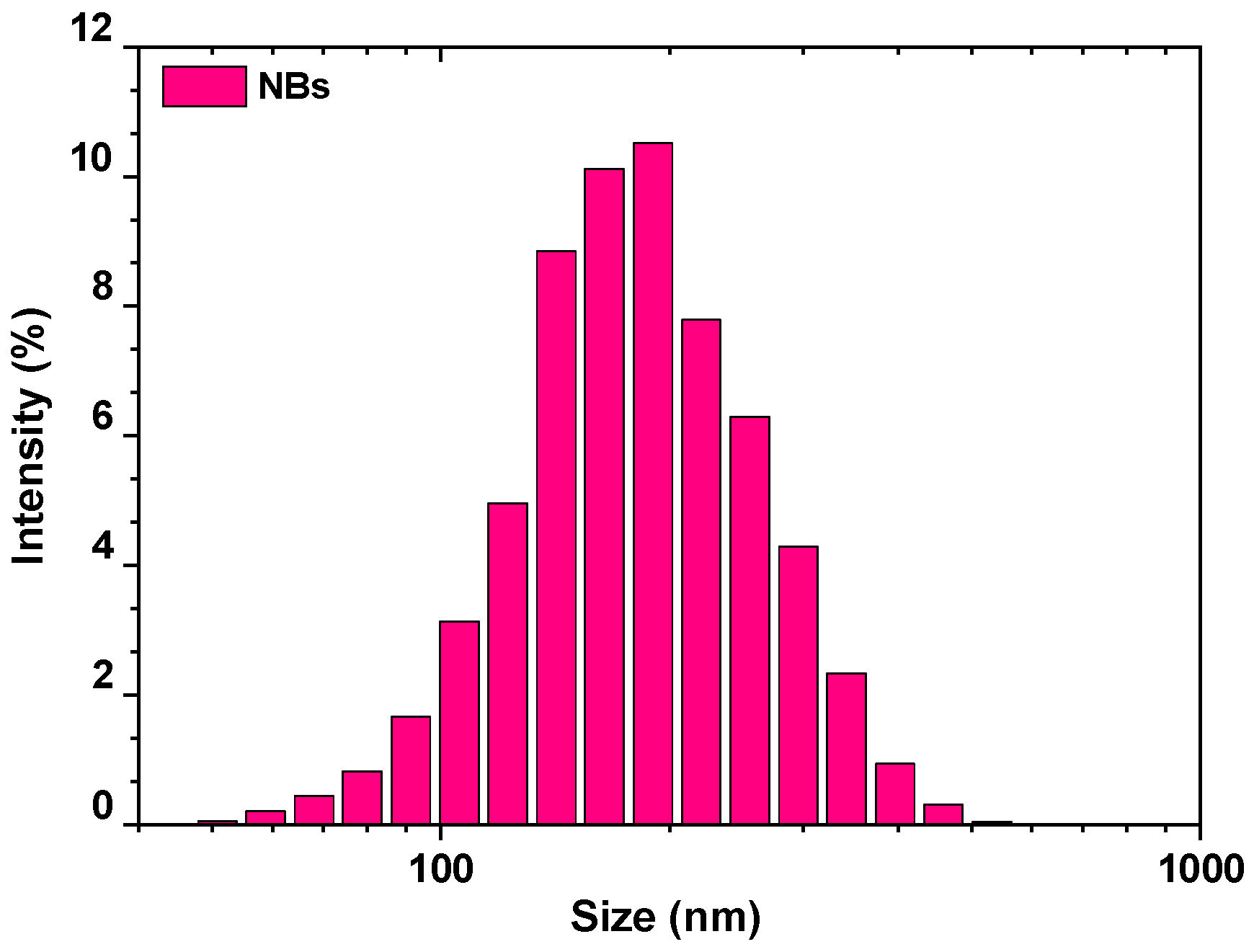
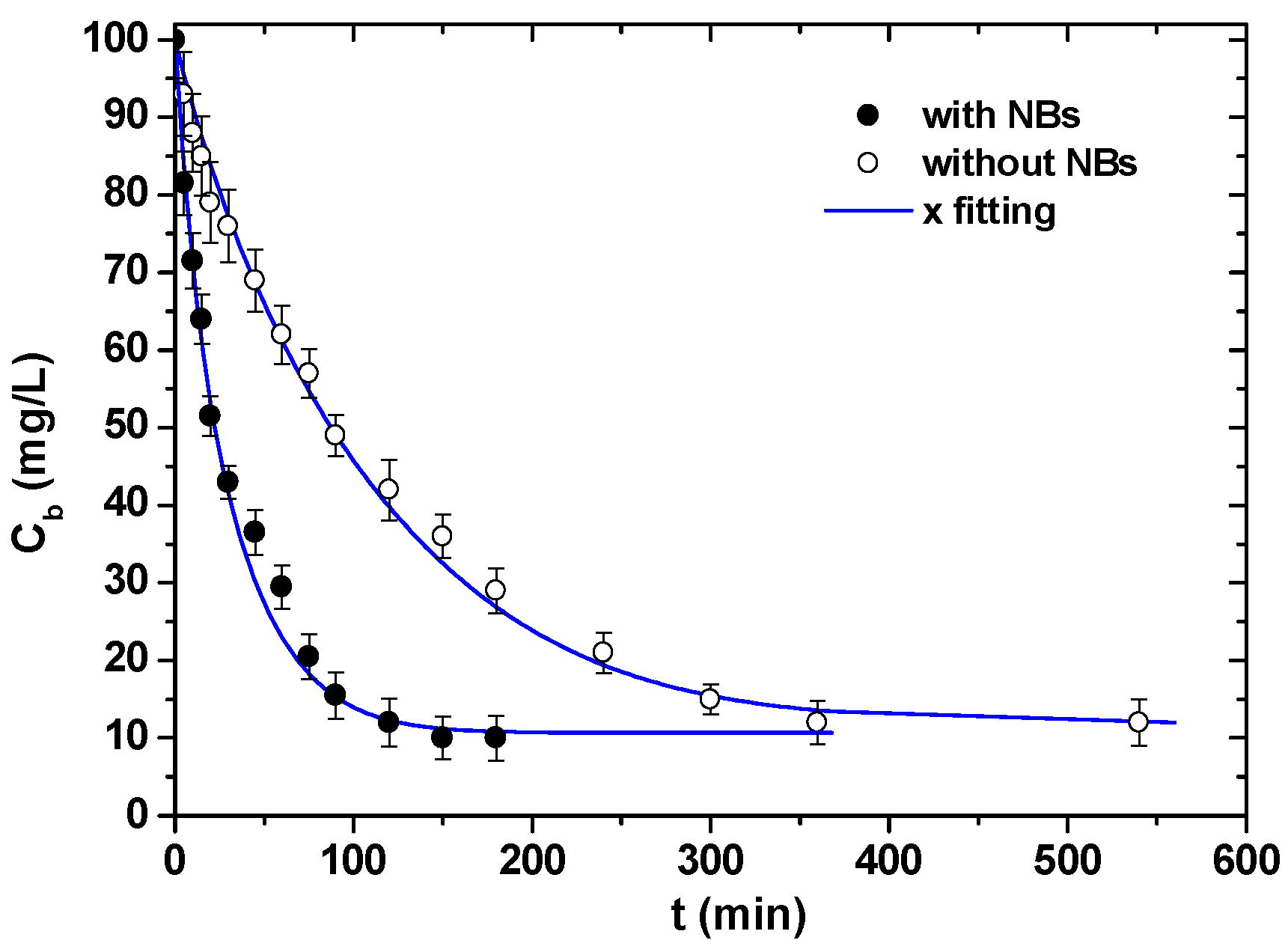
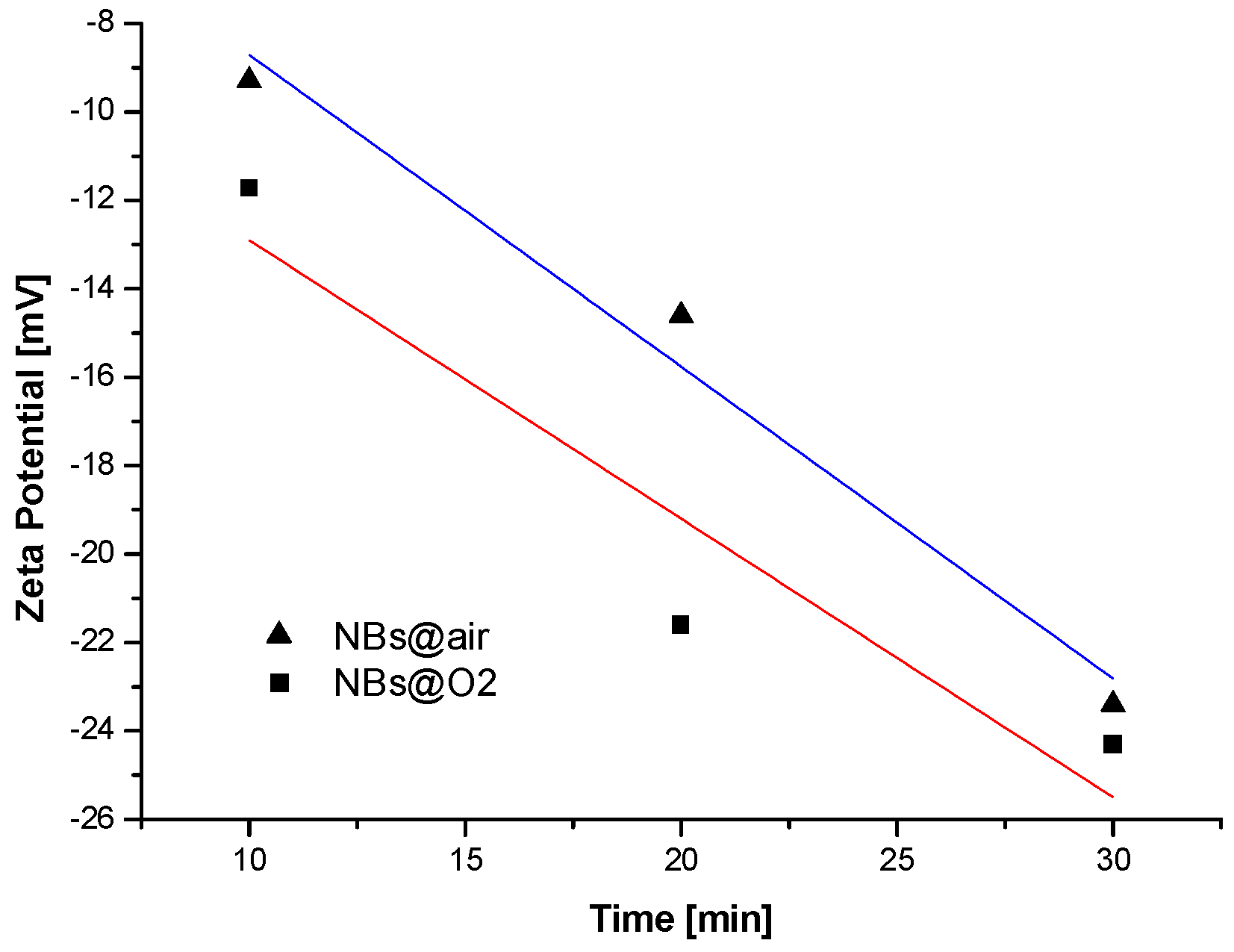
| Size, in µm | 20 | 50 | 100 |
| Number of bubbles (×103) | 1250 | 100 | 14 |
| Surface area (cm2 cm−3) | 23 | 12 | 6.6 |
| Rising rate (m h−1) | 1 | 5 | 20 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kyzas, G.Z.; Mitropoulos, A.C.; Matis, K.A. From Microbubbles to Nanobubbles: Effect on Flotation. Processes 2021, 9, 1287. https://doi.org/10.3390/pr9081287
Kyzas GZ, Mitropoulos AC, Matis KA. From Microbubbles to Nanobubbles: Effect on Flotation. Processes. 2021; 9(8):1287. https://doi.org/10.3390/pr9081287
Chicago/Turabian StyleKyzas, George Z., Athanasios C. Mitropoulos, and Kostas A. Matis. 2021. "From Microbubbles to Nanobubbles: Effect on Flotation" Processes 9, no. 8: 1287. https://doi.org/10.3390/pr9081287
APA StyleKyzas, G. Z., Mitropoulos, A. C., & Matis, K. A. (2021). From Microbubbles to Nanobubbles: Effect on Flotation. Processes, 9(8), 1287. https://doi.org/10.3390/pr9081287








