Cowpea Crop Response to Mineral and Organic Fertilization in SE Spain
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Experimental Design
2.2. Soil and Plant Sampling
2.3. Soil Analyses
2.4. Plant Analyses
2.5. Statistical Analyses
3. Results
3.1. Soil Physicochemical Properties
3.2. Soil Enzyme Activities
3.3. Cowpea Yield, Crop Quality and Nutritional Characteristics
3.4. Interrelationships between Soil and Crop Properties
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Boukar, O.; Coulibaly, O.; Fatokun, C.; Lopez, K.; Tamò, M. Innovative Research along the Cowpea Value Chain, Proceedings of the Fifth World Cowpea Conference on Improving Livelihoods in the Cowpea Value Chain through Advancement in Science, Saly, Senegal, 26 September–1 October 2010; Boukar, O., Coulibaly, O., Fatokun, C.A., Lopez, K., Tamò, M., Eds.; International Institute of Tropical Agriculture: Ibadan, Nigeria, 2012. [Google Scholar]
- Alayande, L.B.; Mustapha, K.B.; Dabak, J.D.; Ubom, G.A. Comparison of nutritional values of brown and white beans in Jos North Local Government markets. Afr. J. Biotechnol. 2012, 11, 10135–10140. [Google Scholar] [CrossRef]
- Avanza, M.; Acevedo, B.; Chaves, M.; Añón, M. Nutritional and anti-nutritional components of four cowpea varieties under thermal treatments: Principal component analysis. Food Sci. Technol. 2013, 51, 148–157. [Google Scholar] [CrossRef]
- Rivas, R.; Falcão, H.M.; Ribeiro, R.V.; Machado, E.C.; Pimentel, C.; Santos, M.G. Drought tolerance in cowpea species is driven by less sensitivity of leaf gas exchange to water deficit and rapid recovery of photosynthesis after rehydration. S. Afr. J. Bot. 2016, 103, 101–107. [Google Scholar] [CrossRef]
- Sawada, H.; Kuykendall, L.D.; Young, J.M. Changing concepts in the systematics of bacterial nitrogen-fixing legume symbionts. J. Gen. Appl. Microbiol. 2003, 49, 155–179. [Google Scholar] [CrossRef]
- Zhang, W.J.; Xang, X.J.; Xu, M.G.; Huang, S.M.; Liu, H.; Peng, C. Soil organic carbon dynamics under long-term fertilizations in arable land of northerm China. Biogeosci. Discuss. 2009, 6, 6539–6577. [Google Scholar]
- El-Metwally, I.M.; Abdelhamid, M.T. Weed control under integrated nutrient management systems in faba bean (Vicia faba) production in Egypt. Planta. Daninha. 2008, 26, 585–594. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; Han, W.; Tang, A.; Shen, J.; Cui, Z.; Vitousek, P.; Erisman, J.W.; Goulding, K.; Goulding, P.; et al. Enhanced nitrogen deposition over China. Nature 2013, 494, 459–462. [Google Scholar] [CrossRef]
- Leita, L.; De Nobili, M.; Mondini, C.; Muhlbachova, G.; Marchiol, L.; Bragato, G.; Contin, M. Influence of inorganic and organic fertilization on soil microbial biomass, metabolic quotient and heavy metal bioavailability. Biol. Fertil Soils 1999, 28, 371–376. [Google Scholar] [CrossRef]
- Parham, J.A.; Deng, S.P.; Raun, W.R.; Johnson, G.V. Long-term cattle manure application in soil. I. Effect on soil phosphorus levels, microbial biomass C, and dehydrogenase and phosphatase activity. Biol. Fertil Soils 2002, 35, 328–337. [Google Scholar]
- Tong, X.; Xu, M.; Wang, X.; Bhattacharyya, R.; Zhang, W.; Cong, R. Long-term fertilization effects on organic carbon fractions in a red soil of China. Catena 2014, 113, 251–259. [Google Scholar] [CrossRef]
- Bouwman, A.F.; Boumans, L.J.M.; Batjes, N.H. Emissions of N2O and NO from fertilized fields: Summary of available measurement data. Glob. Biogeochem. Cycle 2002, 16, 1058. [Google Scholar] [CrossRef]
- Meng, L.; Ding, W.; Cai, Z. Long-term application of organic manure and nitrogen fertilizer on N2O emissions, soil quality and crop production in a sandy loam soil. Soil Biol. Biochem. 2005, 37, 2037–2045. [Google Scholar] [CrossRef]
- Yan, G.; Yao, Z.; Zheng, X.; Liu, C. Characteristics of annual nitrous and nitric oxide emissions from major cereal crops in the North China Plain under alternative fertilizer management. Agric. Ecosyst. Environ. 2015, 207, 67–78. [Google Scholar] [CrossRef]
- Seufert, V.M.; Ramankutty, N.; Foley, J.A. Comparing the yields of organic and conventional agriculture. Nature 2012, 485, 229–232. [Google Scholar] [CrossRef]
- Alaru, M.; Talgre, A.; Eremeev, V.; Tein, B.; Luik, A.; Nemvalts, A.; Loit, E. Crop yield and supply of nitrogen compared in conventional and organic farming systems. Agric. Food Sci. 2014, 23, 317–326. [Google Scholar] [CrossRef]
- Jannoura, R.; Joergensen, R.G.; Bruns, C. Organic fertilizer effects on growth, crop yield, and soil microbial biomass indices in sole and intercropped peas and oats under organic farming conditions. Eur. J. Agron. 2014, 52, 259–270. [Google Scholar] [CrossRef]
- Cai, A.; Xu, M.; Wang, B.; Zhang, W.; Liang, G.; Hou, E.; Luo, Y. Manure acts as a better fertilizer for increasing crop yields than synthetic fertilizer does by improving soil fertility. Soil Tillage Res. 2019, 189, 168–175. [Google Scholar] [CrossRef]
- Brunori, E.; Farina, R.; Biasi, R. Sustainable viticulture: The carbon-sink function of the vineyard agro-ecosystem. Agric. Ecosyst. Environ. 2016, 223, 10–21. [Google Scholar] [CrossRef]
- Puig-Montserrat, X.; Stefanescu, C.; Torre, I.; Palet, J.; Fàbregas, E.; Dantart, J.; Arrizabalaga, A.; Flaquer, C. Effects of organic and conventional crop management on vineyard biodiversity. Agric. Ecosyst. Environ. 2017, 243, 19–26. [Google Scholar] [CrossRef]
- Mäder, P.; Fliebbach, A.; Dubois, D.; Gunst, L.; Fried, P.; Niggli, U. Soil fertility and biodiversity in organic farming. Science 2002, 296, 1694–1697. [Google Scholar] [CrossRef]
- Blair, N.; Faulkner, R.D.; Till, A.R.; Poulton, P.R. Long-term management impacts on soil C, N and physical fertility. Soil Tillage Res. 2006, 91, 30–38. [Google Scholar] [CrossRef]
- Wang, W.; Niu, J.; Zhou, X.; Wang, Y. Long-term change in land management from subtropical wetland to paddy field shifts soil microbial community structure as determined by PLFA and T-RFLP. Pol. J. Ecol. 2011, 59, 37–44. [Google Scholar]
- Bottinelli, N.; Angers, D.A.; Hallaire, V.; Michot, D.; Le Guillou, C.; Cluzeau, D.; Heddadj, D.; Menasseri-Aubry, S. Tillage and fertilization practices affect soil aggregate stability in a Humic Cambisol of Northwest France. Soil Tillage Res. 2017, 170, 14–17. [Google Scholar] [CrossRef]
- Abbona, E.A.; Sarandón, S.J.; Marasas, M.E.; Astier, M. Ecological sustainability evaluation of traditional management in different vineyard systems in Berisso, Argentina. Agric. Ecosyst. Environ. 2007, 119, 335–345. [Google Scholar] [CrossRef]
- Chiriacò, M.V.; Belli, C.; Chiti, T.; Trotta, C.; Sabbatini, S. The potential carbon neutrality of sustainable viticulture showed through a comprehensive assessment of the greenhouse gas (GHG) budget of wine production. J. Clean. Prod. 2019, 225, 435–450. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources, International soil classification system for naming soils and creating legends for soil maps. In World Soil Resources Reports; FAO: Rome, Italy, 2014; Volume 106. [Google Scholar]
- Zornoza, R.; Mataix-Solera, J.; Guerrero, C.; Arcenegui, V.; Mataix-Beneyto, J. Storage Effects on Biochemical Properties of Air-Dried Soil Samples from Southeastern Spain. Arid Land Res. Manag. 2009, 23, 213–222. [Google Scholar] [CrossRef]
- Campbell, D.J.; Hensall, J.K. Bulk density. In Soil Analysis; Smith, K.A., Mullis, C.E., Eds.; Marcel Dekker: New York, NY, USA, 1991; pp. 329–366. [Google Scholar]
- Dewis, J.; Freitas, F. Physical and chemical methods of soil and water analyses. FAO Soils Bulletin 1970, 10, 275. [Google Scholar]
- Cobertera, E. Edafología Aplicada; Ediciones Cátedra: Madrid, Spain, 1993. [Google Scholar]
- Walkley, A.; Black, I.A. An examination of the Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Rovira, P.; Vallejo, V.R. Labile, recalcitrant, and inert organic matter in Mediterranean forest soils. Soil Biol. Biochem. 2007, 39, 202–215. [Google Scholar] [CrossRef]
- Roldán, A.; García-Orenes, F.; Lax, A. An incubation experiment to determinate factor involving aggregation changes in an arid soil receiving urban refuse. Soil Biol. Biochem. 1994, 26, 1699–1707. [Google Scholar] [CrossRef]
- Hoeger, R. Büchi Training Papers: Nitrogen Determination According to Kjeldahl; BÜCHI Labortechnik AG: Flawil, Switzerland, 1998. [Google Scholar]
- Roig, A.; Romero, M.; Lax, A.; Fernández, F.G. Estudio comparativo de métodos de determinación de capacidad de cambio catiónica en suelos calizos. Anal. Edaf. Agrobiol. 1980, 39, 2021–2032. [Google Scholar]
- Keeney, D.R.; Nelson, D.W. Nitrogen inorganic forms, Methods of soil analysis part 2 Chemical and microbiological properties, second edition. Agronomy 1982, 9, 643–698. [Google Scholar]
- Kandeler, E.; Gerber, E. Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol. Fertil. Soils 1988, 6, 68–72. [Google Scholar] [CrossRef]
- Díez, J.A. Consideraciones sobre la utilización de la técnica extractiva de Burriel-Hernando para la evaluación de fósforo asimilable en suelos. Anal. Edaf. Agrobiol. 1982, 41, 1345–1353. [Google Scholar]
- Porta, J.; López-Acevedo, M.; Rodríguez, R. Técnicas y Experimentos en Edafología; Collegi Official d’Enginyers Agrònoms de Catalunya: Barcelona, Spain, 1986; p. 282. [Google Scholar]
- Tabatabai, M.A. Soil Enzymes in Methods of Soil Analyses part 2: Chemical and Microbiological Properties. Soil Sci. Soc. Am. J. 1982, 903–947. [Google Scholar] [CrossRef]
- Parham, J.A.; Deng, S.P. Detection, quantification and characterization of β-glucosaminidase activity in soil. Soil Biol. Biochem. 2000, 32, 1183–1190. [Google Scholar] [CrossRef]
- Von Merci, W.; Schinner, F. An improved and accurate method for determining the dehydrogenase activity of soils with iodonitrotetrazolium chloride. Biol. Fert. Soils 1991, 11, 216–220. [Google Scholar] [CrossRef]
- Zornoza, R.; Landi, L.; Nannipieri, P.; Renella, G. A protocol for the assay of arylesterase activity in soil. Soil Biol. Biochem. 2009, 41, 659–662. [Google Scholar] [CrossRef]
- García-Álvarez, A.; Ibáñez, J.J. Seasonal fluctuations and crop influence on microbiota and enzyme activity in fully developed soils of central Spain. Arid. Soil Res. Rehab. 1994, 8, 161–178. [Google Scholar] [CrossRef]
- Nelson, N. A photometric adaptation of the Somogyi method for determination of glucose. J. Biol. Chem. 1994, 153, 375–380. [Google Scholar] [CrossRef]
- Nannipieri, P.; Johnson, R.L.; Paul, E.A. Criteria for measurement of microbial growth and activity in soil. Soil Biol. Biochem. 1978, 10, 223–229. [Google Scholar] [CrossRef]
- Haichar, F.Z.; Marol, C.; Berge, O.; Rangel-Castro, J.; Prosser, J.I.; Balesdent, J.; Heulin, T.; Achouak, W. Plant host habitat and root exudates shape soil bacterial community structure. ISME J. 2008, 2, 1221–1230. [Google Scholar] [CrossRef] [PubMed]
- Mwafulirwa, L.; Baggs, E.M.; Russell, J.; George, T.; Morley, N.; Sim, A.; Cant, C.D.L.F.; Paterson, E. Barley genotype influences stabilization of rhizodeposition-derived C and soil organic matter mineralization. Soil Biol. Biochem. 2016, 95, 60–69. [Google Scholar] [CrossRef]
- Merbach, W.; Schulze, J.; Richert, M.; Rrocco, E.; Mengel, K. A comparison of different 15N application techniques to study the N net rhizodeposition in the plant–soil system. J. Plant Nutr. Soil Sci. 2000, 163, 375–379. [Google Scholar] [CrossRef]
- Nguyen, C. Rhizodeposition of organic C by plants: Mechanisms and controls. Agronomie 2003, 23, 375–393. [Google Scholar] [CrossRef]
- Bukert, A.; Bationo, A.; Possa, K. Mechanism of residue Mulch-induced cereal growth increases in West-African. Soil Sci. Soc. Am. J. 2000, 64, 1–42. [Google Scholar]
- Shah, Z.; Shah, S.H.; Peoples, M.B.; Schwenke, G.D.; Herriedge, D.F. Crop residue and fertilizer N effects on nitrogen fixation and yields of legume-cereal rotations and soil organic fertility. Field Crops Res. 2003, 83, 1–11. [Google Scholar] [CrossRef]
- Shafi, M.; Bakht, J.; Jan, M.T.; Shah, Z. Soil C and N dynamics and maize (Zea may L.) yields as affected by cropping systems and residue management in North-western Pakistan. Soil Tillage Res. 2007, 94, 520–529. [Google Scholar] [CrossRef]
- Galloway, N.; Townsend, A.R.; Erisman, J.W.; Bekunda, M.; Cai, Z.; Freney, J.R.; Martinelli, L.A.; Seitzinger, S.P.; Sutton, M.A. Transformation of the nitrogen cycle: Recent trends, questions, and potential solutions. Science 2008, 80, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Wang, X.; Han, B.; Ouyang, Z.; Duan, X.; Zheng, H.; Miao, H. Soil carbon sequestrations by nitrogen fertilizer application, straw return and no-tillage in China’s cropland. Glob. Chang. Biol. 2009, 15, 281–305. [Google Scholar] [CrossRef]
- Ladha, J.K.; Reddy, C.K.; Padre, A.T.; van Kessel, C. Role of nitrogen fertilization in sustaining organic matter in cultivated soils. J. Environ. Qual. 2011, 40, 1756–1766. [Google Scholar] [CrossRef] [PubMed]
- Mazzoncini, M.; Sapkota, T.B.; Bàrberi, P.; Antichi, D.; Risaliti, R. Long-term effect of tillage, nitrogen fertilization and cover crops on soil organic carbon and total nitrogen content. Soil Tillage Res. 2011, 114, 165–174. [Google Scholar] [CrossRef]
- Quemada, M.; Menacho, E. Soil respiration 1 year after sewage sludge application. Biol. Fertil Soils 2001, 33, 344–346. [Google Scholar] [CrossRef]
- Cela, S.; Santiveri, F.; Lloveras, J. Residual effects of pig slurry and mineral nitrogen fertilizer on irrigated wheat. Eur. J. Agron. 2011, 34, 257–262. [Google Scholar] [CrossRef]
- Zhao, W.; Liang, B.; Yang, X.; Zhou, J. Fate of residual 15N-labeled fertilizer in dryland farming systems on soils of contrasting fertility. Soil Sci. Plant Nutr. 2015, 61, 846–855. [Google Scholar] [CrossRef]
- Riley, H. Residual value of inorganic fertilizer and farmyard manure for crop yields and soil fertility after long-term use on a loam soil in Norway. Nutr. Cycl. Agroecosystems 2016, 104, 25–37. [Google Scholar] [CrossRef]
- Khaliq, A.; Abbasi, M.K.; Hussain, T. Effects of integrated use of organic and inorganic nutrient sources with effective microorganisms (EM) on seed cotton yield in Pakistan. Bioresour. Technol. 2006, 97, 967–972. [Google Scholar] [CrossRef] [PubMed]
- Maltas, A.; Charles, R.; Jeangros, B.; Sinaj, S. Effect of organic fertilizers and reduced-tillage on soil properties, crop nitrogen response and crop yield: Results of a 12-year experiment in Changins, Switzerland. Soil Tillage Res. 2013, 126, 11–18. [Google Scholar] [CrossRef]
- Jiang, G.; Xu, M.; He, X.; Zhang, W.; Huang, S.; Yang, X.; Liu, H.; Peng, C.; Shirato, Y.; Iizumi, T.; et al. Soil organic carbon sequestration in upland soils of northern China under variable fertilizer management and climate change scenarios. Glob. Biogeochem. Cycles 2014, 28, 319–333. [Google Scholar] [CrossRef]
- Cucci, G.; Lacolla, G.; Summo, C.; Pasqualone, A. Effect of organic and mineral fertilization on faba bean (Vicia faba L.). Sci. Hortic. 2019, 243, 338–343. [Google Scholar] [CrossRef]
- Pimentel, D. Environmental and economic costs of the application of pesticides primarily in the United States. Environ. Develop. Sustain. 2005, 7, 229–252. [Google Scholar] [CrossRef]
- Berry, P.M.; Sylvester-Bradley, R.; Philipps, L.; Hatch, D.J.; Cuttle, S.P.; Rayans, F.W.; Gosling, P. Is the productivity of organic farms restricted by the supply of available nitrogen. Soil Use Manag. 2002, 18, 248–255. [Google Scholar] [CrossRef]
- Yang, B.; Xiong, Z.; Wang, J.; Xu, X.; Huang, Q.; Shen, Q. Mitigating net global warming potential and greenhouse gas intensities by substituting chemical nitrogen fertilizers with organic fertilization strategies in rice-wheat annual rotation systems in China: A 3-year field experiment. Ecol. Eng. 2015, 81, 289–297. [Google Scholar] [CrossRef]
- Tirol-Padre, A.; Ladha, J.K.; Regmi, A.P.; Bhandari, A.L.; Inubushi, K. Organic amendments affect soil parameters in two long-term rice-wheat experiments. Soil Sci. Soc. Am. J. 2007, 71, 442–452. [Google Scholar] [CrossRef]
- Amoach, A.A.; Senge, M.; Miyagawa, S.; Itou, K. Effects of soil fertility management on growth, yield, and water-use efficiency of maize (Zea mays L.) and selected soil properties. Commun. Soil Sci. Plant Anal. 2012, 43, 924–935. [Google Scholar] [CrossRef]
- Bravo, K.; Toselli, M.; Baldi, E.; Marcolini, G.; Sorrenti, G.; Quartieri, M.; Marangoni, B. Effect of organic fertilization on carbon assimilation and partitioning in bearing nectarine trees. Sci. Hortic. 2012, 137, 100–106. [Google Scholar] [CrossRef]
- Lu, H.J.; Ye, Z.Q.; Zhang, X.L.; Lin, X.Y.; Ni, W.Z. Growth and yield responses of crops and macronutrient balance influenced by commercial organic manure used as a partial substitute for chemical fertilizers in an intensive vegetable cropping system. Phys. Chem. Earth Parts A B C 2011, 36, 387–394. [Google Scholar] [CrossRef]
- Liang, H.; Hu, K.; Batchelor, W.D.; Hu, K.; Qi, Z.; Li, B. An integrated soil-crop system model for water and nitrogen management in North China. Sci. Rep. 2016, 6, 25755. [Google Scholar] [CrossRef]
| Parameters a | |
|---|---|
| pH | 8.40 ± 0.09 |
| EC (dS m−1) | 0.34 ± 0.07 |
| SOC (%) | 1.19 ± 0.18 |
| Bulk density (Mg m−3) | 1.01 ± 0.03 |
| CEC (cmol kg−1) | 7.80 ± 1.20 |
| CaCO3 (%) | 30.2 ± 1.2 |
| Clay (%) | 34.5 ± 0.16 |
| Silt (%) | 21.3 ± 1.06 |
| Sand (%) | 44.2 ± 0.92 |
| Nt (%) | 0.094 ± 0.007 |
| NO3− (mg kg−1) | 156 ± 45 |
| NH4+ (mg kg−1) | 5.69 ± 1.12 |
| Available P (mg kg−1) | 30.0 ± 5.4 |
| Exchangeable K (mg kg−1) | 369 ± 63 |
| Exchangeable Ca (mg kg−1) | 2726 ± 126 |
| Exchangeable Mg (mg kg−1) | 606 ± 24 |
| Exchangeable Na (mg kg−1) | 301 ± 31 |
| Cowpea Cultivar a | Fertilizer Type | SOC (g kg−1) | Nt (g kg−1) | AS (%) | RC (%) | LC (%) | NH₄+ (mg kg−1) | NO₃− (mg kg−1) |
|---|---|---|---|---|---|---|---|---|
| 2014 | ||||||||
| FP | Mineral | 13.2 ± 1.2 | 1.2 ± 0.1 | 24.7 ± 13.4 | 0.58 ± 0.06 | 0.74 ± 0.09 | 842 ± 134 | 6.7 ± 2.6 |
| FC | Mineral | 13.0 ± 0.4 | 1.1 ± 0.0 | 17.3 ± 4.7 | 0.56 ± 0.02 | 0.74 ± 0.05 | 576 ± 213 | 7.3 ± 1.0 |
| FP | Organic | 12.7 ± 1.0 | 1.1 ± 0.1 | 18.3 ± 6.5 | 0.59 ± 0.09 | 0.53 ± 0.22 | 473 ± 185 | 11.2 ± 4.1 |
| FC | Organic | 11.6 ± 0.7 | 1.0 ± 0.1 | 7.8 ± 4.4 | 0.53 ± 0.09 | 0.63 ± 0.07 | 499 ± 249 | 13.2 ± 3.6 |
| 2015 | ||||||||
| FP | Mineral | 11.7 ± 0.4 | 1.1 ± 0.1 | 26.7 ± 1.7 | 0.69 ± 0.14 | 0.70 ± 0.49 | 58 ± 13 | 4.9 ± 0.6 |
| FC | Mineral | 11.2 ± 0.1 | 1.1 ± 0.1 | 22.6 ± 1.1 | 0.89 ± 0.02 | 0.70 ± 0.83 | 64 ± 16 | 6.9 ± 4.2 |
| FP | Organic | 11.6 ± 0.9 | 0.8 ± 0.0 | 14.7 ± 4.8 | 0.21 ± 0.08 | 0.95 ± 0.14 | 71 ± 48 | 8.7 ± 2.2 |
| FC | Organic | 12.6 ± 1.0 | 0.9 ± 0.1 | 14.7 ± 6.9 | 0.44 ± 0.25 | 0.82 ± 0.34 | 116 ± 68 | 5.8 ± 0.4 |
| 2016 | ||||||||
| FP | Mineral | 14.6 ± 2.2 | 1.5 ± 0.1 | 50.7 ± 9.3 | 0.99 ± 0.13 | 0.47 ± 0.35 | 640 ± 110 | 10.4 ± 3.2 |
| FC | Mineral | 14.1 ± 1.6 | 1.4 ± 0.1 | 44.8 ± 6.8 | 1.12 ± 0.08 | 0.29 ± 0.08 | 644 ± 149 | 10.0 ± 1.1 |
| FP | Organic | 12.8 ± 2.2 | 1.2 ± 0.1 | 43.7 ± 9.9 | 0.92 ± 0.13 | 0.36 ± 0.09 | 315 ± 96 | 8.5 ± 1.5 |
| FC | Organic | 14.5 ± 1.1 | 1.3 ± 0.2 | 56.0 ± 3.0 | 1.04 ± 0.05 | 0.41 ± 0.10 | 424 ± 109 | 18.0 ± 2.8 |
| F-value b | ||||||||
| Between Subjects | ||||||||
| Cowpea Cultivar (CC) | 0.03 ns | 0.04 ns | 0.71 ns | 13.67 ** | 0.05 ns | 6.74 * | 0.05 ns | |
| Fertilizer Type (FT) | 0.28 ns | 24.28 ** | 2.87 ns | 46.67 *** | 0.11 ns | 22.22 ** | 15.60 ** | |
| CC × FT | 0.62 ns | 0.26 ns | 1.07 ns | 0.02 ns | 0.09 ns | 2.45 ns | 3.48 ns | |
| Within Subjects | ||||||||
| Year (Y) | 21.13 *** | 44.31 *** | 128.76 *** | 53.12 *** | 4.85 * | 9.85 ** | 44.03 *** | |
| Y × CC | 1.80 ns | 1.05 ns | 3.85 * | 3.19 ns | 0.11 ns | 2.37 ns | 1.26 ns | |
| Y × FT | 3.07 ns | 1.78 ns | 4.31 * | 11.67 ** | 0.79 ns | 1.42 ns | 3.82 * | |
| Y × CC × FT | 2.58 ns | 0.96 ns | 2.99 ns | 0.05 ns | 0.22 ns | 4.99 * | 0.61 ns | |
| Cowpea Cultivar a | Fertilizer Type | Exc Ca (mg kg−1) | Exc Mg (mg kg−1) | Exc K (mg kg−1) | Exc Na (mg kg−1) | P (mg kg−1) |
|---|---|---|---|---|---|---|
| 2014 | ||||||
| FP | Mineral | 3614 ± 291 | 91 ± 10 | 524 ± 138 | 680 ± 144 | 24.8 ± 7.5 |
| FC | Mineral | 3372 ± 30 | 84 ± 6 | 430 ± 43 | 554 ± 34 | 16.0 ± 3.5 |
| FP | Organic | 3414 ± 220 | 83 ± 2 | 422 ± 32 | 563 ± 143 | 30.6 ± 2.8 |
| FC | Organic | 3411 ± 112 | 85 ± 6 | 434 ± 117 | 565 ± 159 | 32.4 ± 8.1 |
| 2015 | ||||||
| FP | Mineral | 6471 ± 1158 | 1513 ± 290 | 927 ± 79 | 662 ± 64 | 25.0 ± 4.4 |
| FC | Mineral | 5555 ± 365 | 1222 ± 103 | 860 ± 31 | 506 ± 101 | 22.4 ± 5.9 |
| FP | Organic | 5308 ± 112 | 1185 ± 70 | 698 ± 145 | 635 ± 24 | 19.7 ± 1.5 |
| FC | Organic | 5396 ± 922 | 1140 ± 132 | 792 ± 168 | 569 ± 67 | 15.8 ± 3.2 |
| 2016 | ||||||
| FP | Mineral | 877 ± 186 | 190 ± 33 | 196 ± 30 | 177 ± 56 | 72.6 ± 9.4 |
| FC | Mineral | 925 ± 282 | 202 ± 76 | 205 ± 65 | 200 ± 111 | 66.0 ± 7.1 |
| FP | Organic | 979 ± 352 | 200 ± 84 | 167 ± 80 | 154 ± 80 | 89.9 ± 30.7 |
| FC | Organic | 673 ± 54 | 115 ± 12 | 127 ± 14 | 88 ± 12 | 94.5 ± 28.0 |
| F-value b | ||||||
| Between Subjects | ||||||
| Cowpea Cultivar (CC) | 2.16 ns | 4.19 ns | 0.68 ns | 4.97 ns | 0.32 ns | |
| Fertilizer Type (FT) | 3.26 ns | 5.95 * | 22.89 ** | 1.37 ns | 4.21 ns | |
| CC × FT | 0.96 ns | 0.61 ns | 4.29 ns | 0.55 ns | 0.56 ns | |
| Within Subjects | ||||||
| Year (Y) | 292.13 *** | 453.64 *** | 107.37 *** | 77.38 *** | 80.21 *** | |
| Y × CC | 0.35 ns | 2.01 ns | 0.19 ns | 0.60 ns | 0.34 ns | |
| Y × FT | 1.42 ns | 3.05 ns | 0.81 ns | 0.63 ns | 3.82 ns | |
| Y × CC × FT | 1.45 ns | 2.03 ns | 0.76 ns | 1.01 ns | 0.21 ns | |
| Previous Cowpea Cultivar a | Fertilizer Type | Dhs (µmol INTF g−1 h−1) | Glm (µmol PNP g−1 h−1) | Glu (µmol PNP g−1 h−1) | Aryl (µmol PNP g−1 h−1) | Cel (nmol Gearbox Sugars g−1 h−1) |
|---|---|---|---|---|---|---|
| 2014 | ||||||
| FP | Mineral | 0.16 ± 0.05 | 0.12 ± 0.04 | 0.59 ± 0.07 | 128 ± 32 | 171 ± 50 |
| FC | Mineral | 0.18 ± 0.04 | 0.11 ± 0.05 | 0.54 ± 0.13 | 97 ± 30 | 190 ± 47 |
| FP | Organic | 0.16 ± 0.09 | 0.15 ± 0.04 | 0.48 ± 0.05 | 176 ± 45 | 165 ± 96 |
| FC | Organic | 0.13 ± 0.01 | 0.11 ± 0.02 | 0.45 ± 0.10 | 173 ± 60 | 172 ± 51 |
| 2015 | ||||||
| FP | Mineral | 0.79 ± 0.13 | 0.29 ± 0.02 | 0.52 ± 0.10 | 70 ± 16 | 23 ± 17 |
| FC | Mineral | 0.71 ± 0.04 | 0.17 ± 0.06 | 0.36 ± 0.08 | 84 ± 20 | 30 ± 24 |
| FP | Organic | 0.73 ± 0.20 | 0.24 ± 0.11 | 0.28 ± 0.04 | 84 ± 30 | 22 ± 11 |
| FC | Organic | 0.89 ± 0.25 | 0.21 ± 0.01 | 0.41 ± 0.03 | 56 ± 20 | 22 ± 12 |
| 2016 | ||||||
| FP | Mineral | 0.04 ± 0.01 | 0.24 ± 0.08 | 0.36 ± 0.06 | 131 ± 44 | 1 ± 2 |
| FC | Mineral | 0.02 ± 0.01 | 0.21 ± 0.12 | 0.38 ± 0.12 | 154 ± 14 | 5 ± 1 |
| FP | Organic | 0.02 ± 0.00 | 0.27 ± 0.06 | 0.24 ± 0.09 | 105 ± 51 | 6 ± 2 |
| FC | Organic | 0.02 ± 0.00 | 0.12 ± 0.05 | 0.36 ± 0.11 | 125 ± 33 | 7 ± 3 |
| F-value b | ||||||
| Between Subjects | ||||||
| Cowpea Cultivar (CC) | 0.137 ns | 9.41 * | 0.10 ns | 0.00 ns | 0.22 ns | |
| Fertilizer Type (FT) | 0.04 ns | 0.03 ns | 6.81 * | 0.47 ns | 0.10 ns | |
| CC × FT | 1.34 ns | 0.24 ns | 4.22 ns | 0.04 ns | 0.07 ns | |
| Within Subjects | ||||||
| Year (Y) | 146.77 *** | 10.28 ** | 17.09 *** | 14.55 *** | 72.74 *** | |
| Y × CC | 0.20 ns | 0.91 ns | 1.58 ns | 1.09 ns | 0.07 ns | |
| Y × FT | 0.47 ns | 0.35 ns | 0.10 ns | 5.74 * | 0.13 ns | |
| Y × CC × FT | 1.22 ns | 2.24 ns | 2.41 ns | 0.82 ns | 0.01 ns | |
| Cowpea Cultivar a | Fertilizer Type | Crop Yield (kg ha−1) | Protein Content in Seeds (%) | Weight of 100 Seeds (g) |
|---|---|---|---|---|
| 2014 | ||||
| FP | Mineral | 2333 ± 133 | 20.06 ± 1.25 | 21.27 ± 1.66 |
| FC | Mineral | 2490 ± 167 | 19.66 ± 3.79 | 18.73 ± 0.59 |
| FP | Organic | 2135 ± 306 | 19.64 ± 1.78 | 18.07 ± 0.70 |
| FC | Organic | 2371 ± 161 | 18.54 ± 2.17 | 18.27 ± 0.81 |
| 2015 | ||||
| FP | Mineral | 100 ± 52 | 19.48 ± 1.93 | 19.63 ± 1.40 |
| FC | Mineral | 216 ± 144 | 20.15 ± 0.57 | 20.53 ± 2.29 |
| FP | Organic | 121 ± 70 | 23.86 ± 3.57 | 24.11 ± 5.95 |
| FC | Organic | 366 ± 152 | 21.55 ± 0.98 | 22.80 ± 3.03 |
| 2016 | ||||
| FP | Mineral | 3033 ± 757 | 21.05 ± 0.74 | 27.79 ± 0.89 |
| FC | Mineral | 4166 ± 1171 | 19.72 ± 1.80 | 21.08 ± 1.06 |
| FP | Organic | 2533 ± 321 | 20.75 ± 1.71 | 25.11 ± 0.50 |
| FC | Organic | 3366 ± 1167 | 18.94 ± 0.64 | 21.87 ± 1.38 |
| F-value b | ||||
| Between Subjects | ||||
| Cowpea Cultivar (CC) | 6.48 * | 2.01 ns | 9.72 * | |
| Fertilizer Type (FT) | 1.82 ns | 0.50 ns | 0.08 ns | |
| CC × FT | 0.01 ns | 0.87 ns | 0.96 ns | |
| Within Subjects | ||||
| Year (Y) | 95.76 * | 2.71 ns | 13.14 *** | |
| Y × CC | 2.03 ns | 0.17 ns | 3.51 ns | |
| Y × FT | 1.35 ns | 3.47 ns | 4.27 ns | |
| Y × CC × FT | 0.13 ns | 0.39 ns | 1.31 ns | |
| Cowpea Cultivar a | Fertilizer Type | N (g kg−1) | Ca (mg kg−1) | Mg (mg kg−1) | K (mg kg−1) | P (mg kg−1) |
|---|---|---|---|---|---|---|
| 2014 | ||||||
| FP | Mineral | 32.10 ± 2.01 | 754 ± 76 | 1985 ± 56 | 2977 ± 368 | 4651 ± 267 |
| FC | Mineral | 31.45 ± 6.06 | 882 ± 67 | 1953 ± 48 | 13,468 ± 579 | 4751 ± 173 |
| FP | Organic | 31.43 ± 2.85 | 716 ± 116 | 2029 ± 205 | 13,300 ± 486 | 4735 ± 658 |
| FC | Organic | 29.66 ± 3.47 | 853 ± 38 | 2015 ± 56 | 13,801 ± 175 | 4886 ± 199 |
| 2015 | ||||||
| FP | Mineral | 31.17 ± 3.09 | 2205 ± 397 | 3889 ± 457 | 28,302 ± 2575 | 11,208 ± 1264 |
| FC | Mineral | 32.24 ± 0.91 | 2111 ± 86 | 3905 ± 346 | 28,321 ± 2832 | 10,225 ± 1174 |
| FP | Organic | 38.17 ± 5.71 | 3068 ± 642 | 4575 ± 744 | 32,436 ± 5906 | 10,911 ± 1783 |
| FC | Organic | 34.48 ± 1.57 | 2834 ± 109 | 4822 ± 311 | 32,963 ± 3009 | 11,735 ± 2724 |
| 2016 | ||||||
| FP | Mineral | 33.69 ± 1.19 | 412 ± 65 | 1276 ± 134 | 3711 ± 230 | 1855 ± 115 |
| FC | Mineral | 31.55 ± 2.88 | 604 ± 356 | 1826 ± 1110 | 4962 ± 2441 | 2481 ± 1220 |
| FP | Organic | 33.21 ± 2.73 | 233 ± 88 | 599 ± 217 | 2135 ± 596 | 1067 ± 298 |
| FC | Organic | 30.31 ± 1.03 | 349 ± 103 | 877 ± 317 | 3133 ± 772 | 1566 ± 386 |
| F-value b | ||||||
| Between Subjects | ||||||
| Cowpea Cultivar (CC) | 2.01 ns | 0.20 ns | 0.81 ns | 0.46 ns | 0.23 ns | |
| Fertilizer Type (FT) | 0.50 ns | 3.91 ns | 0.00 ns | 1.18 ns | 0.01 ns | |
| CC × FT | 0.87 ns | 0.14 ns | 0.00 ns | 0.00 ns | 0.48 ns | |
| Within Subjects | ||||||
| Year (Y) | 2.71 ns | 271.24 *** | 235.52 *** | 503.09 *** | 221.67 *** | |
| Y × CC | 0.17 ns | 0.14 ns | 1.08 ns | 0.13 ns | 0.26 ns | |
| Y × FT | 3.47 ns | 14.96 *** | 14.44 *** | 6.47 ** | 1.35 ns | |
| Y × CC × FT | 0.39 ns | 0.07 ns | 0.35 ns | 0.02 ns | 0.71 ns | |
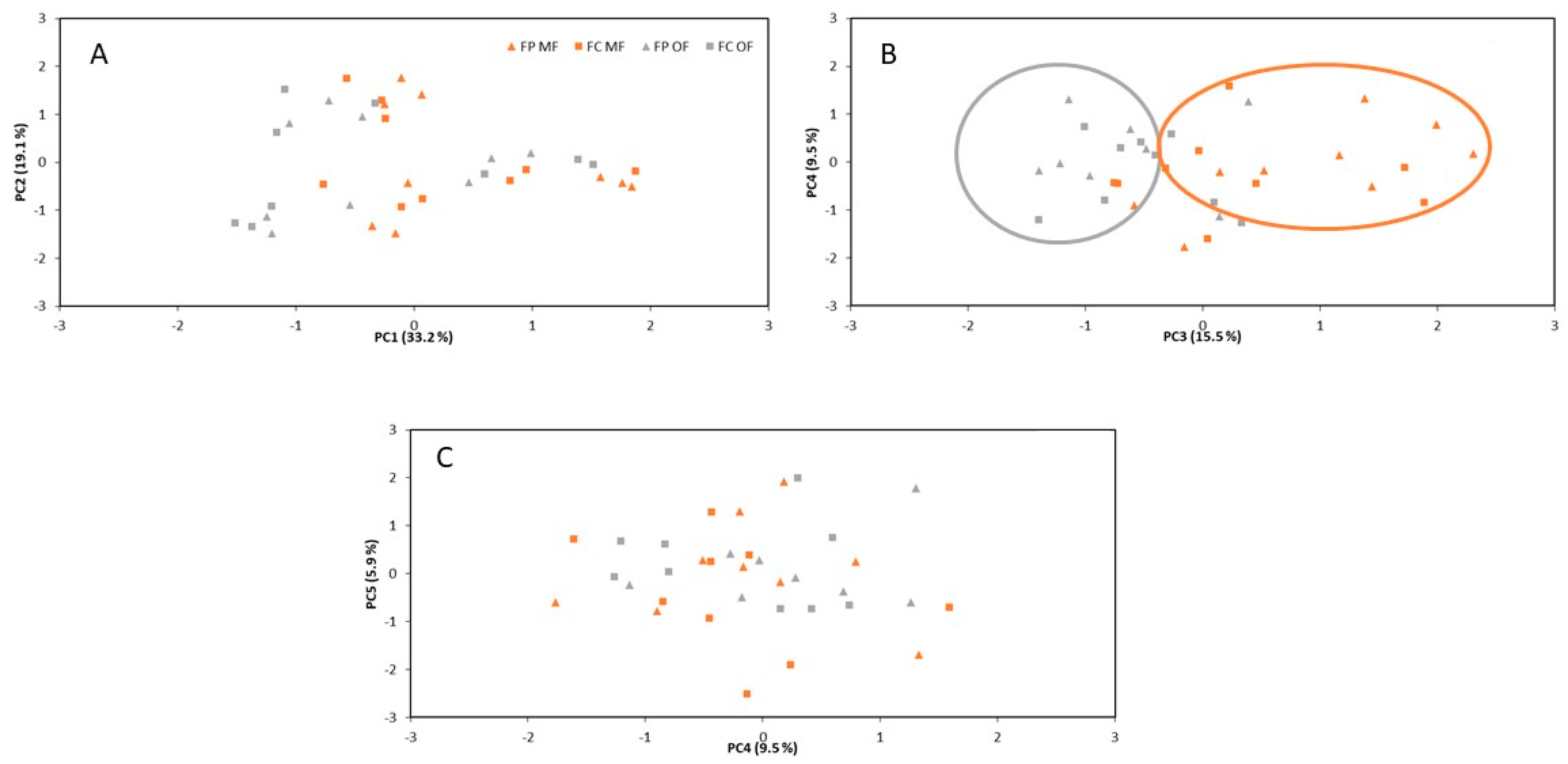
| Variance Explained | PC1 (33.2%) | PC2 (19.1%) | PC3 (15.5%) | PC4 (9.5%) | PC5 (5.9%) |
|---|---|---|---|---|---|
| Exc K (mg kg−1) | 0.884 | 0.286 | −0.269 | −0.068 | 0.051 |
| Exc Mg (mg kg−1) | 0.876 | −0.208 | −0.365 | 0.065 | 0.094 |
| Dhs Activity (µmol INTF g−1 h−1) | 0.852 | 0.033 | −0.376 | 0.167 | 0.175 |
| P in Seeds (mg kg−1) | 0.849 | 0.173 | −0.385 | 0.037 | 0.205 |
| Mg in Seeds (mg kg−1) | 0.827 | 0.143 | −0.325 | 0.107 | 0.229 |
| K in Seeds (mg kg−1) | 0.822 | 0.229 | −0.420 | 0.070 | 0.183 |
| Ca in Seeds (mg kg−1) | 0.807 | 0.064 | −0.391 | 0.161 | 0.262 |
| Exc Ca (mg kg−1) | 0.797 | 0.372 | −0.378 | −0.029 | 0.162 |
| Crop Yield (kg ha−1) | −0.773 | −0.081 | 0.427 | −0.044 | −0.269 |
| Ure Activity (µmol NH4+ g−1 h−1) | 0.749 | −0.386 | 0.049 | 0.106 | −0.165 |
| NH4+ (mg kg−1) | −0.672 | −0.187 | −0.052 | −0.119 | 0.054 |
| Aryl Activity (µmol PNP g−1 h−1) | −0.603 | 0.141 | 0.135 | −0.327 | 0.015 |
| B (mg kg−1) | 0.211 | 0.928 | −0.121 | −0.05 | 0.131 |
| Cel Activity (nmol gearbox sugars g−1 h−1) | −0.280 | 0.855 | −0.013 | −0.120 | 0.058 |
| AS (%) | −0.361 | −0.723 | 0.354 | −0.016 | −0.220 |
| Exc Na (mg kg−1) | 0.557 | 0.723 | −0.115 | −0.016 | 0.209 |
| P (mg kg−1) | −0.605 | −0.658 | 0.167 | 0.047 | −0.211 |
| β-Glm Activity (µmol PNP g−1 h−1) | 0.332 | −0.587 | −0.042 | 0.071 | 0.276 |
| RC (%) | −0.261 | −0.584 | 0.400 | −0.232 | −0.473 |
| β-Glu Activity (µmol PNP g−1 h−1) | 0.087 | 0.532 | 0.238 | −0.516 | 0.217 |
| EC (µS m−1) | −0.449 | 0.077 | 0.822 | −0.063 | −0.069 |
| pH | 0.236 | 0.347 | −0.798 | −0.057 | 0.127 |
| NO3− | −0.496 | 0.354 | 0.731 | 0.00 | −0.182 |
| Nt (g kg−1) | −0.338 | −0.433 | 0.688 | −0.218 | −0.193 |
| SOC (g kg−1) | −0.338 | −0.344 | 0.653 | −0.046 | 0.305 |
| Protein in Seeds (%) | −0.368 | −0.029 | −0.056 | 0.942 | 0.091 |
| N in Seeds (g kg−1) | 0.187 | −0.029 | −0.056 | 0.942 | 0.091 |
| Weight of 100 Seeds (g) | −0.026 | −0.556 | 0.235 | 0.581 | 0.177 |
| LC (%) | 0.261 | 0.242 | −0.132 | 0.154 | 0.784 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Navarro, V.; Zornoza, R.; Faz, Á.; Fernández, J.A. Cowpea Crop Response to Mineral and Organic Fertilization in SE Spain. Processes 2021, 9, 822. https://doi.org/10.3390/pr9050822
Sánchez-Navarro V, Zornoza R, Faz Á, Fernández JA. Cowpea Crop Response to Mineral and Organic Fertilization in SE Spain. Processes. 2021; 9(5):822. https://doi.org/10.3390/pr9050822
Chicago/Turabian StyleSánchez-Navarro, Virginia, Raúl Zornoza, Ángel Faz, and Juan Antonio Fernández. 2021. "Cowpea Crop Response to Mineral and Organic Fertilization in SE Spain" Processes 9, no. 5: 822. https://doi.org/10.3390/pr9050822
APA StyleSánchez-Navarro, V., Zornoza, R., Faz, Á., & Fernández, J. A. (2021). Cowpea Crop Response to Mineral and Organic Fertilization in SE Spain. Processes, 9(5), 822. https://doi.org/10.3390/pr9050822







