Anaerobic Degradation of Individual Components from 5-Hydroxymethylfurfural Process-Wastewater in Continuously Operated Fixed Bed Reactors
Abstract
1. Introduction
2. Materials and Methods
2.1. Reactor Setup
2.2. Analytical Methods
2.3. Startup of the Anaerobic Filters
2.4. Substrate and Stock Solution Preparation
2.5. Experimental Procedure
2.6. Analysis of Recorded Data
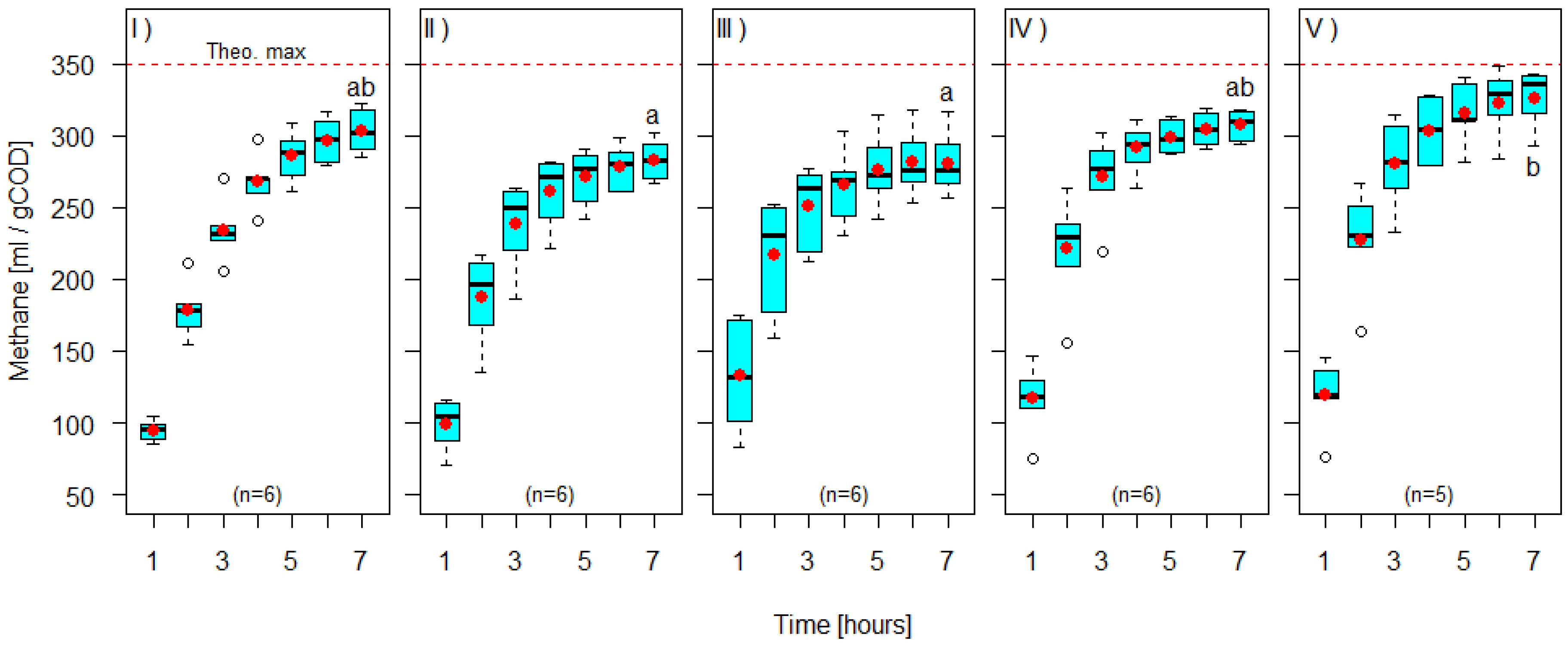
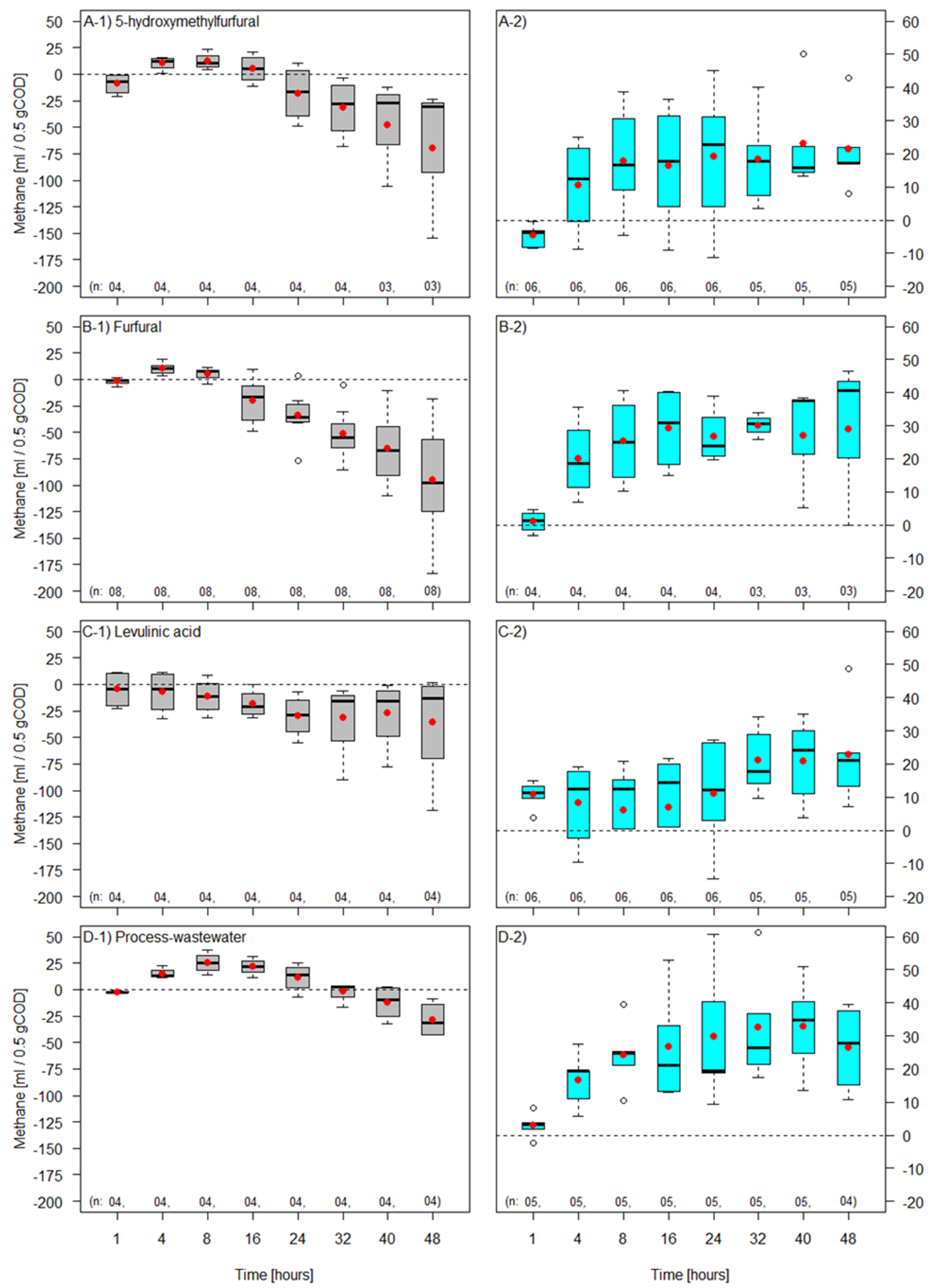
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xia, H.; Xu, S.; Hu, H.; An, J.; Li, C. Efficient conversion of 5-hydroxymethylfurfural to high-value chemicals by chemo- and bio-catalysis. RSC Adv. 2018, 8, 30875–30886. [Google Scholar] [CrossRef]
- Moreau, C.; Belgacem, M.N.; Gandini, A. Recent catalytic advances in the chemistry of substituted furans from carbohydrates and in the ensuing polymers. Top. Catal. 2004, 27, 11–30. [Google Scholar] [CrossRef]
- Yu, I.K.M.; Tsang, D.C.W. Conversion of biomass to hydroxymethylfurfural: A review of catalytic systems and underlying mechanisms. Bioresour. Technol. 2017, 238, 716–732. [Google Scholar] [CrossRef] [PubMed]
- Steinbach, D.; Kruse, A.; Sauer, J. Pretreatment technologies of lignocellulosic biomass in water in view of furfural and 5-hydroxymethylfurfural production- A review. Biomass Convers. Biorefinery 2017, 7, 247–274. [Google Scholar] [CrossRef]
- Stemann, J.; Putschew, A.; Ziegler, F. Hydrothermal carbonization: Process water characterization and effects of water recirculation. Bioresour. Technol. 2013, 143, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, H.; Sørensen, H.R.; Meyer, A.S. Formation of degradation compounds from lignocellulosic biomass in the biorefinery: Sugar reaction mechanisms. Carbohydr. Res. 2014, 385, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Wüst, D.; Correa, C.R.; Jung, D.; Zimmermann, M.; Kruse, A.; Fiori, L. Understanding the influence of biomass particle size and reaction medium on the formation pathways of hydrochar. Biomass Convers. Biorefinery 2020, 10, 1357–1380. [Google Scholar] [CrossRef]
- Świątek, K.; Gaag, S.; Klier, A.; Kruse, A.; Sauer, J.; Steinbach, D. Acid hydrolysis of lignocellulosic biomass: Sugars and furfurals formation. Catalysts 2020, 10, 437. [Google Scholar] [CrossRef]
- Menegazzo, F.; Ghedini, E.; Signoretto, M. 5-Hydroxymethylfurfural (HMF) production from real biomasses. Molecules 2018, 23, 2201. [Google Scholar] [CrossRef]
- McKeown, R.M.; Hughes, D.; Collins, G.; Mahony, T.; O’Flaherty, V. Low-temperature anaerobic digestion for wastewater treatment. Curr. Opin. Biotechnol. 2012, 23, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Passos, F.; Uggetti, E.; Carrère, H.; Ferrer, I. Algal Biomass: Physical Pretreatments. In Pretreatment of Biomass: Processes and Technologies, 1st ed.; Pandey, A., Negi, S., Binod, P., Larroche, C., Eds.; Elsevier Science Publishing Co Inc.: Amsterdam, The Netherlands, 2014; pp. 195–226. ISBN 978-0-12-800080-9. [Google Scholar]
- Stökle, K.; Hülsemann, B.; Steinbach, D.; Cao, Z.; Oechsner, H.; Kruse, A. A biorefinery concept using forced chicory roots for the production of biogas, hydrochar, and platform chemicals. Biomass Convers. Biorefinery 2019. [Google Scholar] [CrossRef]
- Erdogan, E.; Atila, B.; Mumme, J.; Reza, M.T.; Toptas, A.; Elibol, M.; Yanik, J. Characterization of products from hydrothermal carbonization of orange pomace including anaerobic digestibility of process liquor. Bioresour. Technol. 2015, 196, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Marin-Batista, J.D.; Villamil, J.A.; Rodriguez, J.J.; Mohedano, A.F.; de la Rubia, M.A. Valorization of microalgal biomass by hydrothermal carbonization and anaerobic digestion. Bioresour. Technol. 2019, 274, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Hülsemann, B.; Wüst, D.; Illi, L.; Oechsner, H.; Kruse, A. Valorization of maize silage digestate from two-stage anaerobic digestion by hydrothermal carbonization. Energy Convers. Manag. 2020, 222, 113218. [Google Scholar] [CrossRef]
- Aragón-Briceño, C.; Ross, A.B.; Camargo-Valero, M.A. Evaluation and comparison of product yields and bio-methane potential in sewage digestate following hydrothermal treatment. Appl. Energy 2017, 208, 1357–1369. [Google Scholar] [CrossRef]
- Barakat, A.; Monlau, F.; Steyer, J.P.; Carrere, H. Effect of lignin-derived and furan compounds found in lignocellulosic hydrolysates on biomethane production. Bioresour. Technol. 2012, 104, 90–99. [Google Scholar] [CrossRef]
- Si, B.; Li, J.; Zhu, Z.; Shen, M.; Lu, J.; Duan, N.; Zhang, Y.; Liao, Q.; Huang, Y.; Liu, Z. Inhibitors degradation and microbial response during continuous anaerobic conversion of hydrothermal liquefaction wastewater. Sci. Total Environ. 2018, 630, 1124–1132. [Google Scholar] [CrossRef] [PubMed]
- Almeida, J.R.M.; Bertilsson, M.; Gorwa-Grauslund, M.F.; Gorsich, S.; Lidén, G. Metabolic effects of furaldehydes and impacts on biotechnological processes. Appl. Microbiol. Biotechnol. 2009, 82, 625–638. [Google Scholar] [CrossRef]
- Ibraheem, O.; Ndimba, B.K. Molecular adaptation mechanisms employed by ethanologenic bacteria in response to lignocellulose-derived inhibitory compounds. Int. J. Biol. Sci. 2013, 9, 598–612. [Google Scholar] [CrossRef] [PubMed]
- McCarty, P.L. Anaerobic Waste Treatment Fundamentals (parts 1–4). Public Works 1964, 95, 91–94, 95–99, 107–112, 123–126. [Google Scholar]
- Monlau, F.; Steyer, J.-P.; Carrère, H.; Quéméneur, M.; Sambusiti, C.; Trably, E.; Barakat, A. Do furanic and phenolic compounds of lignocellulosic and algae biomass hydrolyzate inhibit anaerobic mixed cultures? A comprehensive review. Biotechnol. Adv. 2014, 32, 934–951. [Google Scholar] [CrossRef] [PubMed]
- Mah, T.-F.C.; O’Toole, G.A. Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol. 2001, 9, 34–39. [Google Scholar] [CrossRef]
- Cresson, R.; Carrère, H.; Delgenès, J.P.; Bernet, N. Biofilm formation during the start-up period of an anaerobic biofilm reactor-Impact of nutrient complementation. Biochem. Eng. J. 2006, 30, 55–62. [Google Scholar] [CrossRef]
- Ercan, D.; Demirci, A. Current and future trends for biofilm reactors for fermentation processes. Crit. Rev. Biotechnol. 2015, 35, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Krümpel, J.; Schäufele, F.; Schneider, J.; Jungbluth, T.; Zielonka, S.; Lemmer, A. Kinetics of biogas production in Anaerobic Filters. Bioresour. Technol. 2016, 200, 230–234. [Google Scholar] [CrossRef]
- Lemmer, A.; Krümpel, J. Demand-driven biogas production in anaerobic filters. Appl. Energy 2017, 185, 885–894. [Google Scholar] [CrossRef]
- Chen, Y.; Rößler, B.; Zielonka, S.; Wonneberger, A.M.; Lemmer, A. Effects of organic loading rate on the performance of a pressurized anaerobic filter in two-phase anaerobic digestion. Energies 2014, 7, 736–750. [Google Scholar] [CrossRef]
- Rittmann, B.E.; McCarty, P.L. Environmental Biotechnology: Principles and Applications; International Edition, McGraw-Hill Education: New York, NY, USA, 2001; pp. 126–164. ISBN 0-07-118184-9. [Google Scholar]
- VDI VDI 4630—Fermentation of organic materials: Characterisation of the substrate, sampling, collection of material data, fermentation tests. In VDI-Handbuch Energietechnik; Beuth Verlag GmbH: Berlin, Germany, 2006; pp. 44–59.
- Atelge, M.R.; Krisa, D.; Kumar, G.; Eskicioglu, C.; Nguyen, D.D.; Chang, S.W.; Atabani, A.E.; Al-Muhtaseb, A.H.; Unalan, S. Biogas Production from Organic Waste: Recent Progress and Perspectives. Waste Biomass Valorization 2020, 11, 1019–1040. [Google Scholar] [CrossRef]
- Habe, H.; Sato, S.; Morita, T.; Fukuoka, T.; Kirimura, K.; Kitamoto, D. Bacterial production of short-chain organic acids and trehalose from levulinic acid: A potential cellulose-derived building block as a feedstock for microbial production. Bioresour. Technol. 2015, 177, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Kim, S.H.; Park, H.D.; Lim, D.J.; Yoon, J.J. Feasibility of anaerobic digestion from bioethanol fermentation residue. Bioresour. Technol. 2013, 141, 177–183. [Google Scholar] [CrossRef]
- Ghasimi, D.S.M.; Aboudi, K.; de Kreuk, M.; Zandvoort, M.H.; van Lier, J.B. Impact of lignocellulosic-waste intermediates on hydrolysis and methanogenesis under thermophilic and mesophilic conditions. Chem. Eng. J. 2016, 295, 181–191. [Google Scholar] [CrossRef]
- Boopathy, R.; Bokang, H.; Daniels, L. Biotransformation of furfural and 5-hydroxymethyl furfural by enteric bacteria. J. Ind. Microbiol. 1993, 11, 147–150. [Google Scholar] [CrossRef]
- Boopathy, R. Anaerobic biotransformation of furfural to furfuryl alcohol by a methanogenic archaebacterium. Int. Biodeterior. Biodegrad. 2009, 63, 1070–1072. [Google Scholar] [CrossRef]
- Gutiérrez, T.; Buszko, M.L.; Ingram, L.O.; Preston, J.F. Reduction of furfural to furfuryl alcohol by ethanologenic strains of bacteria and its effect on ethanol production from xylose. Appl. Biochem. Biotechnol. Part A Enzym. Eng. Biotechnol. 2002, 98–100, 327–340. [Google Scholar] [CrossRef]
- Zhang, Y.; Han, B.; Ezeji, T.C. Biotransformation of furfural and 5-hydroxymethyl furfural (HMF) by Clostridium acetobutylicum ATCC 824 during butanol fermentation. N. Biotechnol. 2012, 29, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Cheng, J.J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef] [PubMed]
- Kayembe, K.; Basosila, L.; Mpiana, P.T.; Sikulisimwa, P.C.; Mbuyu, K. Inhibitory Effects of Phenolic Monomers on Methanogenesis in Anaerobic Digestion. Br. Microbiol. Res. J. 2013, 3, 32–41. [Google Scholar] [CrossRef]
- Taherzadeh, M.J.; Gustafsson, L.; Niklasson, C.; Lidén, G. Conversion of furfural in aerobic and anaerobic batch fermentation of glucose by Saccharomyces cerevisiae. J. Biosci. Bioeng. 1999, 87, 169–174. [Google Scholar] [CrossRef]
- Taherzadeh, M.J.; Eklund, R.; Gustafsson, L.; Niklasson, C.; Lidén, G. Characterization and Fermentation of Dilute-Acid Hydrolyzates from Wood. Ind. Eng. Chem. Res. 1997, 36, 4659–4665. [Google Scholar] [CrossRef]
- Modig, T.; Lidén, G.; Taherzadeh, M.J. Inhibition effects of furfural on alcohol dehydrogenase, aldehyde dehydrogenase and pyruvate dehydrogenase. Biochem. J. 2002, 363, 769–776. [Google Scholar] [CrossRef]


| Test Component | IUPAC Name | Component Molecular Formula | Component Molecular Mass |
|---|---|---|---|
| [g mol−1] | |||
| 5-Hydroxymethylfurfural | 5-(Hydroxymethyl)furan-2-carbaldehyde | C6H6O3 | 126.11 |
| Furfural | Furan-2-carbaldehyde | C5H4O2 | 96.08 |
| Levunilinc acid | 4-Oxopentanoic acid | C5H8O3 | 116.11 |
| Butyric acid (control) | Butanoic acid | C4H8O2 | 88.11 |
| Component/Parameter | Base Feed | Process-Wastewater | ||
|---|---|---|---|---|
| Concentration | Carbon | Concentration | Carbon | |
| [g L−1] | [%] | [g L−1] | [%] | |
| Formic acid | - | - | 2.66 | 2.46 |
| Acetic acid | 5.83 | 34.60 | 1.67 | 2.38 |
| Glycolic acid | - | - | 0.49 | 0.55 |
| Propionic acid | 1.56 | 11.26 | - | - |
| Lactic acid | - | - | 1.53 | 2.18 |
| Iso-butyric acid | 0.02 | 0.18 | - | - |
| Butyric acid | 5.20 | 42.11 | - | - |
| Furfural | - | - | 0.65 | 1.51 |
| Levulinic acid | - | - | 8.90 | 16.35 |
| n-Valeric acid | 0.05 | 0.46 | - | - |
| 5-Hydroxymethylfurfural | - | - | 14.79 | 30.01 |
| Caproic acid | 0.16 | 1.48 | - | - |
| Fructose | - | - | 3.87 | 5.50 |
| Unknown fractions | 9.91 | 39.1 | ||
| DOC (dissolved organic carbon) | 6.74 | 100 | 28.17 | 100 |
| TN (total nitrogen) | 0.37 | 0.08 | ||
| COD | 21.8 | 70.15 | ||
| Test Component | Specific Gas Yield | Gas Quality | Methane Conversion | ||
|---|---|---|---|---|---|
| CH4 | Biogas | CH4 | CO2 | ||
| [mL gCOD−1] | [%] | [%] | |||
| 5-Hydroxymethylfurfural | 208.74 ± 65.04 | 402.30 ± 98.40 | 51.31 ± 6.52 | 48.75 ± 6.39 | 59.64 ± 18.58 |
| Furfural | 258.31 ± 48.67 | 479.89 ± 84.97 | 54.34 ± 8.33 | 45.76 ± 8.73 | 73.80 ± 13.90 |
| Levulinic acid | 274.67 ± 66.66 | 517.73 ± 115.83 | 52.90 ± 3.68 | 46.84 ± 3.65 | 78.47 ± 19.04 |
| Process-wastewater | 191.25 ± 52.40 | 344.76 ± 83.21 | 55.36 ± 6.95 | 45.18 ± 6.85 | 54.64 ± 14.97 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.T.; Krümpel, J.; Wüst, D.; Lemmer, A. Anaerobic Degradation of Individual Components from 5-Hydroxymethylfurfural Process-Wastewater in Continuously Operated Fixed Bed Reactors. Processes 2021, 9, 677. https://doi.org/10.3390/pr9040677
Khan MT, Krümpel J, Wüst D, Lemmer A. Anaerobic Degradation of Individual Components from 5-Hydroxymethylfurfural Process-Wastewater in Continuously Operated Fixed Bed Reactors. Processes. 2021; 9(4):677. https://doi.org/10.3390/pr9040677
Chicago/Turabian StyleKhan, Muhammad Tahir, Johannes Krümpel, Dominik Wüst, and Andreas Lemmer. 2021. "Anaerobic Degradation of Individual Components from 5-Hydroxymethylfurfural Process-Wastewater in Continuously Operated Fixed Bed Reactors" Processes 9, no. 4: 677. https://doi.org/10.3390/pr9040677
APA StyleKhan, M. T., Krümpel, J., Wüst, D., & Lemmer, A. (2021). Anaerobic Degradation of Individual Components from 5-Hydroxymethylfurfural Process-Wastewater in Continuously Operated Fixed Bed Reactors. Processes, 9(4), 677. https://doi.org/10.3390/pr9040677








