Experimental Evaluation on the Catalytic Activity of a Novel CeZrK/rGO Nanocomposite for Soot Oxidation in Catalyzed Diesel Particulate Filter
Abstract
1. Introduction
2. Experimental Methods
2.1. Catalyst Preparation
2.2. Experimental Apparatus
3. Results and Discussion
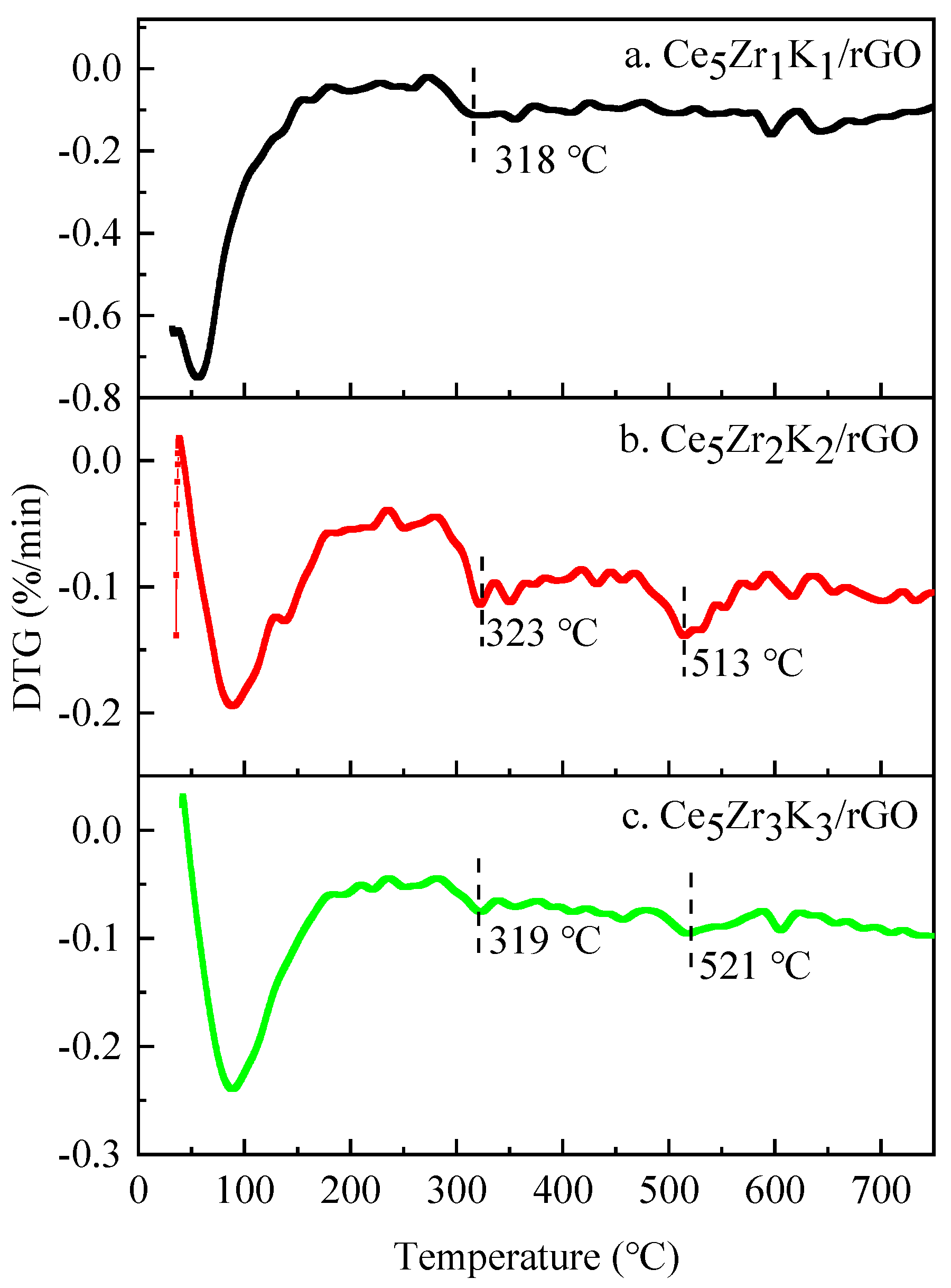
3.1. Characterization Studies
3.2. Evaluation on Catalytic Activity for Soot Oxidation
4. Conclusions
- The use of rGO as the carrier of CeZrK/rGO catalysts provides a large specific surface area and special pore structure, increasing the contact between soot and catalyst compared to the Ce-based catalysts without rGO.
- The doping of Zr and K ions in the CeZrK/rGO catalysts leads to the lattice distortion of CeO2, which can increase oxygen vacancies and active oxygen species, control the crystallite growth of CeO2, and ensure the dispersion quality of active sites.
- The characterization studies show that the CeZrK/rGO catalysts have nanoscale pore structure, superior dispersion quality, small particle size, large specific surface area, as well as sufficient oxygen vacancies.
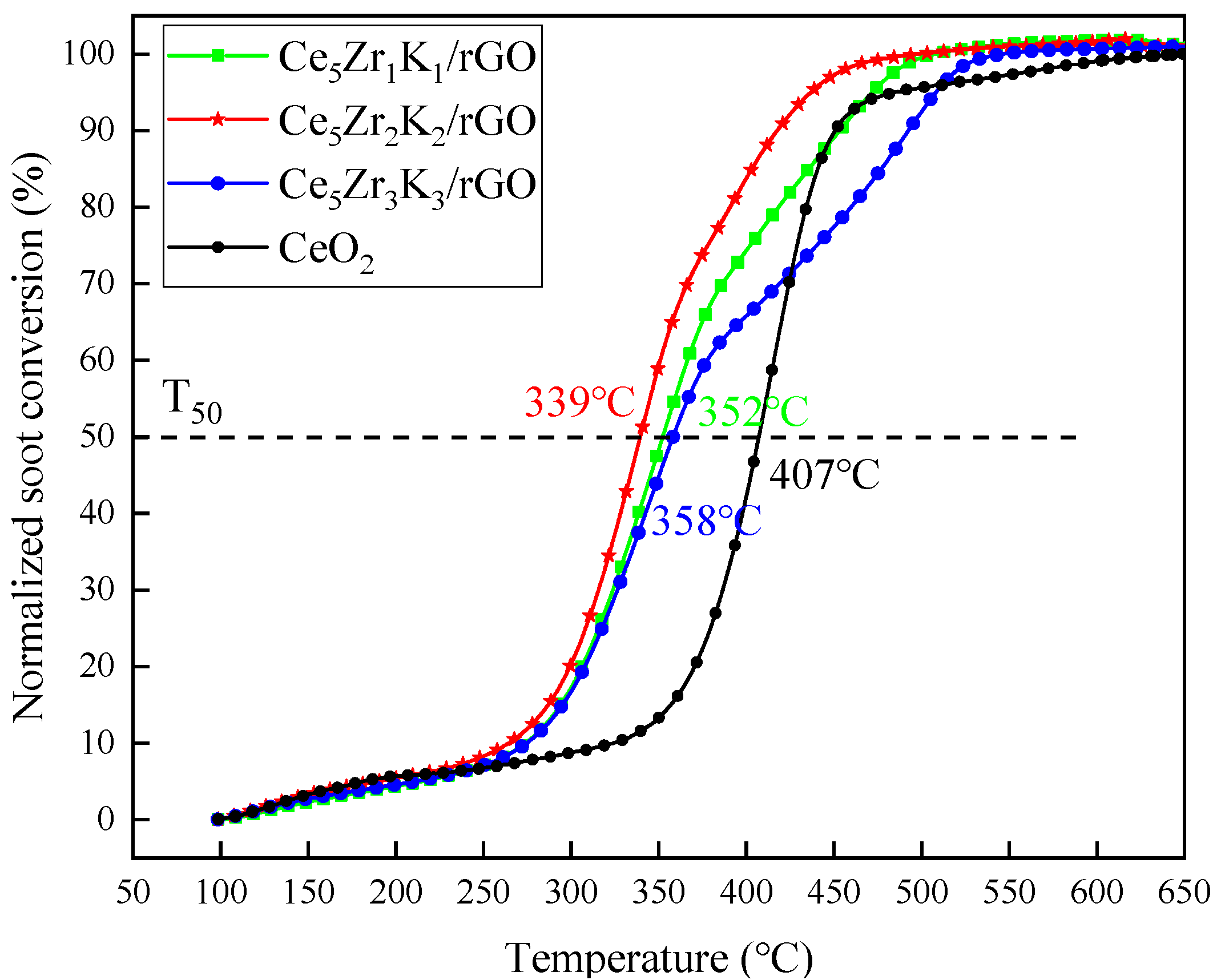
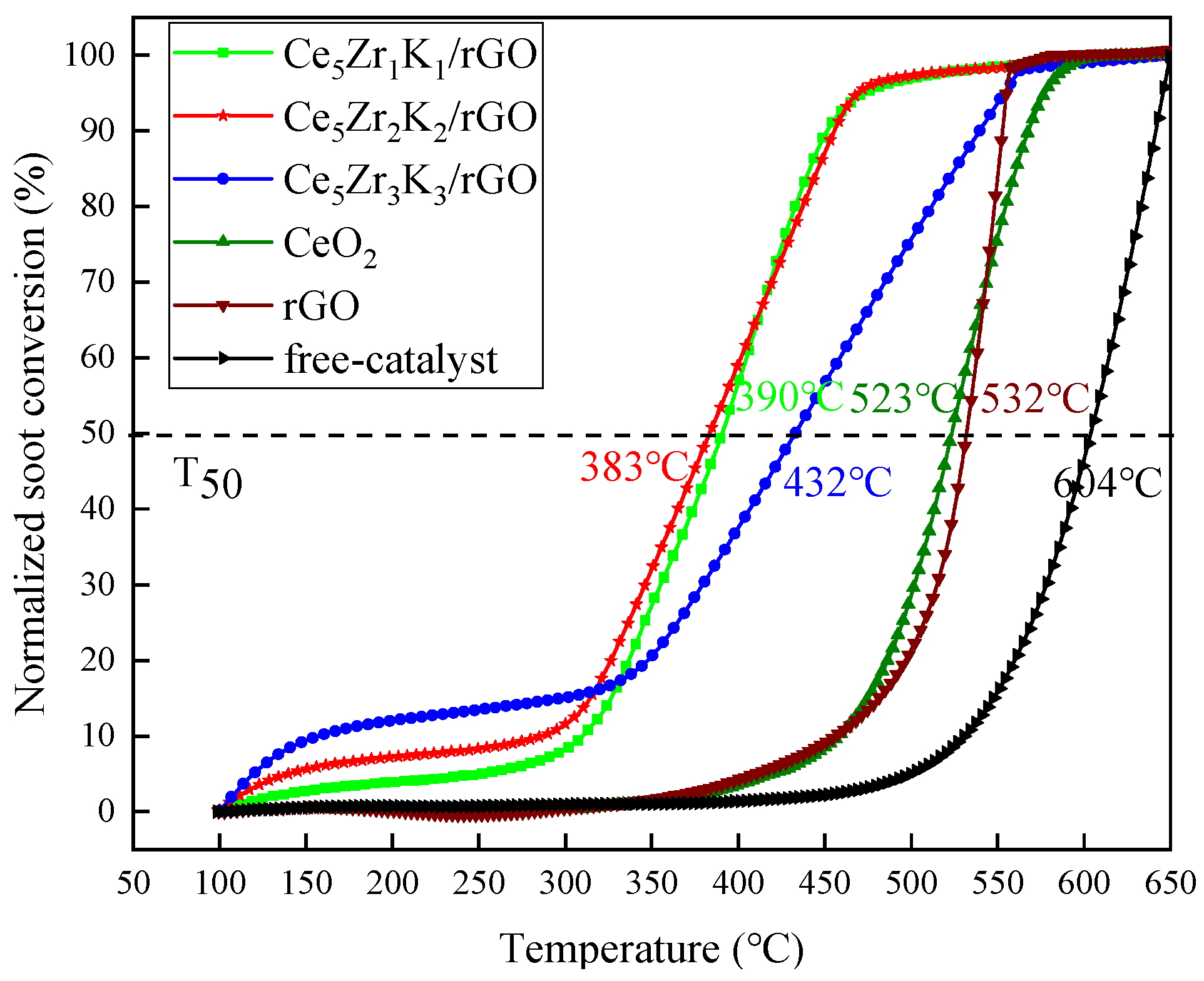
- The T50 related to soot conversion of Ce5Zr1K1/rGO, Ce5Zr2K2/rGO, and Ce5Zr3K3/rGO under tight contact and loose contact conditions were 352 °C, 339 °C, 358 °C and 390 °C, 383 °C, 432 °C, respectively, which are smaller than those of some typical rare earth, perovskite, and precious metal catalysts, such as CeO2, 3DOM La0.8Ce0.2FeO3, Pt/Al2O3, etc. It is indicated that the CeZrK/rGO catalysts have high catalytic activity for soot oxidation, especially for Ce5Zr2K2/rGO.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ratcliff, M.A.; Dane, A.J.; Williams, A.; Ireland, J.; Luecke, J.; McCormick, R.L.; Voorhees, K.J. Diesel Particle Filter and Fuel Effects on Heavy-Duty Diesel Engine Emissions. Environ. Sci. Technol. 2010, 44, 8343. [Google Scholar] [CrossRef]
- Wu, G.; Wang, X.; Li, Y.; Liu, Z. A Realistic Skeletal Mechanism for the Oxidation of Biodiesel Surrogate Composed of Long Carbon Chain and Polyunsaturated Compounds. Fuel 2021, 289, 119934. [Google Scholar] [CrossRef]
- Wu, G.; Wu, D.; Li, Y.; Meng, L. Effect of Acetone-n-butanol-ethanol (ABE) as an Oxygenate on Combustion, Performance, and Emission Characteristics of a Spark Ignition Engine. J. Chem. 2020, 2020, 7468651. [Google Scholar] [CrossRef]
- Johnson, T.V. Review of Diesel Emissions and Control. Int. J. Engine Res. 2009, 10, 275. [Google Scholar] [CrossRef]
- Peck, R.; Becker, C. Experimental Investigations and Dynamic Simulation of Diesel Particulate Filter Systems. Chem. Eng. Technol. 2009, 32, 1411. [Google Scholar] [CrossRef]
- Cauda, E.; Fino, D.; Saracco, G.; Specchia, V. Preparation and Regeneration of a Catalytic Diesel Particulate filter. Chem. Eng. Sci. 2007, 62, 5182. [Google Scholar] [CrossRef]
- Shukla, P.C.; Gupta, T.; Labhasetwar, N.K.; Khobaragade, R.; Gupta, N.K.; Agarwal, A.K. Effectiveness of Non-Noble Metal Based Diesel Oxidation Catalysts on Particle Number Emissions from Diesel and Biodiesel Exhaust. Sci. Total Environ. 2017, 574, 1512. [Google Scholar] [CrossRef] [PubMed]
- Mamontov, E.; Brezny, R.; Koranne, M.; Egami, T. Nanoscale Heterogeneities and Oxygen Storage Capacity of Ce0.5Zr0.5O2. J. Phys. Chem. B. 2003, 107, 13007. [Google Scholar] [CrossRef]
- Wei, Y.; Liu, J.; Zhao, Z.; Chen, Y.; Xu, C.; Duan, A.; Jiang, G.; He, H. Highly Active Catalysts of Gold Nanoparticles Supported on Three-Dimensionally Ordered Macroporous LaFeO3 for Soot Oxidation. Angew. Chem. Int. Ed. 2011, 50, 2326. [Google Scholar] [CrossRef]
- Da, Y.; Zeng, L.; Wang, C.; Mao, T.; Chen, R.; Gong, C.; Fan, G. Catalytic oxidation of diesel soot particulates over Pt substituted LaMn1-xPtxO3 perovskite oxides. Catal. Today 2019, 327, 73. [Google Scholar] [CrossRef]
- Sun, J.; Zhao, Z.; Li, Y.; Yu, X.; Zhao, L.; Li, J.; Wei, Y.; Liu, J. Synthesis and Catalytic Performance of Macroporous La1-xCexCoO3 Perovskite Oxide Catalysts with High Oxygen Mobility for Catalytic Combustion of Soot. J. Rare Earths 2020, 38, 584. [Google Scholar] [CrossRef]
- Mukherjee, D.; Reddy, B.M. Noble Metal-Free CeO2-Based Mixed Oxides for CO and Soot Oxidation. Catal. Today 2018, 309, 227. [Google Scholar] [CrossRef]
- Atribak, I.; Bueno-López, A.; García-García, A. Combined Removal of Diesel Soot Particulates and NOx Over CeO2–ZrO2 Mixed Oxides. J. Catal. 2008, 259, 123. [Google Scholar] [CrossRef]
- Gross, M.S.; Ulla, M.A.; Querini, C.A. Catalytic Oxidation of Diesel Soot: New Characterization and Kinetic Evidence Related to the Reaction Mechanism on K/CeO2 catalyst. Appl. Catal. A 2009, 360, 81. [Google Scholar] [CrossRef]
- Tan, L.; Wang, Y.; Liu, Q.; Wang, J.; Jing, X.; Liu, L.; Liu, J.; Song, D. Enhanced Adsorption of Uranium (VI) Using a Three-Dimensional Layered Double Hydroxide/Graphene Hybrid Material. Chem. Eng. J. 2015, 259, 752. [Google Scholar] [CrossRef]
- Mei, X.; Yan, Q.; Lu, P.; Wang, J.; Cui, Y.; Nie, Y.; Umar, A.; Wang, Q. Synthesis of Pt/K2CO3/MgAlOx–reduced Graphene Oxide Hybrids as Promising NOx Storage–Reduction Catalysts with Superior Catalytic Performance. Sci. Rep. 2017, 7, 42862. [Google Scholar] [CrossRef]
- Pan, M.; Qian, W.; Wei, H.; Feng, D.; Pan, J. Effects on Performance and Emissions of Gasoline Compression Ignition Engine over a Wide Range of Internal Exhaust Gas Recirculation Rates under Lean Conditions. Fuel 2020, 265, 116881. [Google Scholar] [CrossRef]
- Wang, P.; Yi, J.; Sun, C.; Luo, P.; Lei, L. Evaluation of H2 on the Evolution Mechanism of NOx Storage and Reduction over Pt-Ba-Ce/γ-Al2O3 Catalysts. Engineering 2019, 5, 568. [Google Scholar] [CrossRef]
- Pan, M.; Huang, R.; Liao, J.; Ouyang, T.; Lv, D.; Huang, H. Effect of EGR dilution on Combustion, Performance and Emission Characteristics of a Diesel Engine Fueled with n-Pentanol and 2-Ethylhexyl Nitrate Additive. Energy. Conv. Manag. 2018, 176, 246. [Google Scholar] [CrossRef]
- Zhang, P.; Yang, B.; Ma, H.; Wu, Z. Graphene Modified Porous Organic Polymer Supported Phosphotungstic Acid Catalyst for Alkylation Desulfurization. Fuel 2021, 293, 120438. [Google Scholar] [CrossRef]
- Li, X.; Wang, Z.; Wu, L.; Tsai, T. One-step in Situ Synthesis of a Novel α-zirconium Phosphate/Graphene Oxide Hybrid and its Application in Phenolic Foam with Enhanced Mechanical Strength, Flame Retardancy and Thermal Stability. RSC Adv. 2016, 6, 74903. [Google Scholar] [CrossRef]
- Wu, Z.S.; Ren, W.; Gao, L.; Zhao, J.; Chen, Z.; Liu, B.; Tang, D.; Yu, B.; Jiang, C.; Cheng, H.M. Synthesis of Graphene Sheets with High Electrical Conductivity and Good Thermal Stability by Hydrogen Arc Discharge Exfoliation. ACS Nano 2009, 3, 411. [Google Scholar] [CrossRef]
- Bora, C.; Bharali, P.; Baglari, S.; Dolui, S.K.; Konwar, B.K. Strong and Conductive Reduced Graphene Oxide/Polyester Resin Composite Films with Improved Mechanical Strength, Thermal Stability and its Antibacterial Activity. Compos. Sci. Technol. 2013, 87, 1. [Google Scholar] [CrossRef]
- Yuan, B.; Song, L.; Liew, K.M.; Hu, Y. Solid Acid-Reduced Graphene Oxide Nanohybrid for Enhancing Thermal Stability, Mechanical Property and Flame Retardancy of Polypropylene. RSC Adv. 2015, 5, 41307. [Google Scholar] [CrossRef]
- Zong, M.; Huang, Y.; Zhao, Y.; Sun, X.; Qu, C.; Luo, D.; Zheng, J. Facile Preparation, High Microwave Absorption and Microwave Absorbing Mechanism of RGO–Fe3O4 Composites. RSC Adv. 2013, 3, 23638. [Google Scholar] [CrossRef]
- Khira, Z.; Nasr, S.; Dorra, J.; Karima, H. Investigations on electrical conductivity and dielectric properties ofgraphene oxide nanosheets synthetized from modified Hummer’s method. J. Mol. Struct. 2020, 1216, 128304. [Google Scholar]
- Xiao, Q.; Sun, Y.; Zhang, J.; Li, Q. Size-dependent of Chromium (VI) Adsorption on Nano α-Fe2O3 Surface. Appl. Surf. Sci. 2015, 356, 18. [Google Scholar] [CrossRef]
- Sajeevan, A.C.; Sajith, V. Synthesis of Stable Cerium Zirconium Oxide Nanoparticle—Diesel Suspension and Investigation of its Effects on Diesel Properties and Smoke. Fuel 2016, 183, 155. [Google Scholar] [CrossRef]
- Lian, P.; Zhu, X.; Liang, S.; Li, Z.; Yang, W.; Wang, H. High Reversible Capacity of SnO2/graphene Nanocomposite as an Anode Material for Lithium-Ion Batteries. Electrochim. Acta 2011, 56, 4532. [Google Scholar] [CrossRef]
- Rangaswamy, A.; Sudarsanam, P.; Reddy, B.M. Rare Earth Metal Doped CeO2-based Catalytic Materials for Diesel Soot Oxidation at Lower Temperatures. J. Rare Earths 2015, 33, 1162. [Google Scholar] [CrossRef]
- You, X.; Sheng, Z.; Yu, D.; Yang, L.; Xiao, X.; Wang, S. Influence of Mn/Ce ratio on the Physicochemical Properties and Catalytic Performance of Graphene Supported MnOx-CeO2 Oxides for NH3-SCR at Low Temperature. Appl. Surf. Sci. 2017, 423, 845. [Google Scholar] [CrossRef]
- Julkapli, N.M.; Bagheri, S. Graphene Supported Heterogeneous Catalysts: An Overview. Int. J. Hydrog. Energy 2015, 40, 948–979. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S. Physisorption of Gases, with Special Reference to the Evaluation of Surface Area and Pore Size Distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051. [Google Scholar] [CrossRef]
- Shi, Z.M.; Jin, L.N. Influence of La3+/Ce3+-doping on Phase Transformation and Crystal Growth in TiO2-15wt% ZnO Gels. J. Non-Cryst. Solids 2009, 355, 213. [Google Scholar] [CrossRef]
- Weng, D.; Li, J.; Wu, X.; Si, Z. Modification of CeO2-ZrO2 Catalyst by Potassium for NOx-Assisted Soot Oxidation. J. Environ. Sci. 2011, 23, 145. [Google Scholar] [CrossRef]
- Wei, Y.; Zhao, Z.; Jiao, J.; Liu, J.; Duan, A.; Jiang, G. Preparation of Ultrafine Ce-based Oxide Nanoparticles and Their Catalytic Performances for Diesel Soot Combustion. J. Rare Earths 2014, 32, 124. [Google Scholar] [CrossRef]
- Zheng, C.; Zhou, X.; Cao, H.; Wang, G.; Liu, Z. Synthesis of Porous Graphene/Activated Carbon Composite with High Packing Density and Large Specific Surface Area for Supercapacitor Electrode Material. J. Power Sources 2014, 258, 290. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, F.; Yang, X.; Long, G.; Wu, Y.; Zhang, T.; Leng, K.; Huang, Y.; Ma, Y.; Yu, A.; et al. Porous 3D Graphene-Based Bulk Materials with Exceptional High Surface Area and Excellent Conductivity for Supercapacitors. Sci. Rep. 2013, 3, 1408. [Google Scholar] [CrossRef]
- Vallés, C.; Núñez, J.D.; Benito, A.M.; Maser, W.K. Flexible Conductive Graphene Paper Obtained by Direct and Gentle Annealing of Graphene Oxide Paper. Carbon 2012, 50, 835. [Google Scholar] [CrossRef]
- Xie, R.; Fan, G.; Ma, Q.; Yang, L.; Li, F. Facile Synthesis and Enhanced Catalytic Performance of Graphene-Supported Ni Nanocatalyst from a Layered Double Hydroxide-Based Composite Precursor. J. Mater. Chem. A. 2014, 2, 7880. [Google Scholar] [CrossRef]
- Strankowski, M.; Włodarczyk, D.; Piszczyk, Ł.; Strankowska, J. Polyurethane Nanocomposites Containing Reduced Graphene Oxide, FTIR, Raman, and XRD Studies. J. Spectrosc. 2016, 2016, 7520741. [Google Scholar] [CrossRef]
- Fan, X.; Peng, W.; Li, Y.; Li, X.; Wang, S.; Zhang, G.; Zhang, F. Deoxygenation of Exfoliated Graphite Oxide under Alkaline Conditions: A Green Route to Graphene Preparation. Adv. Mater. 2008, 20, 4490. [Google Scholar] [CrossRef]
- Larachi, F.; Pierre, J.; Adnot, A.; Bernis, A. Ce 3d XPS Study of Composite CexMn1−xO2−y Wet Oxidation Catalysts. Appl. Surf. Sci. 2002, 195, 236. [Google Scholar] [CrossRef]
- Atiqullah, M.; Akhtar, M.N.; Faiz, M.; Moman, A.; Abu-Raqabah, A.H.; Khan, J.H.; Wazeer, M.I. Surface Chemistry of Selected Supported Metallocene Catalysts Studied by DR-FTIR, CPMAS NMR, and XPS Techniques. Surf. Interface Anal. 2006, 38, 1319. [Google Scholar] [CrossRef]
- Hajjar, Z.; Kazemeini, M.; Rashidi, A.; Bazmi, M. In Situ and Simultaneous Synthesis of a Novel Graphene-Based Catalyst for Deep Hydrodesulfurization of Naphtha. Catal. Lett. 2015, 145, 1660. [Google Scholar] [CrossRef]
- Li, H.; Lu, G.; Dai, Q.; Wang, Y.; Guo, Y.; Guo, Y. Efficient Low-Temperature Catalytic Combustion of Trichloroethylene over Flower-Like Mesoporous Mn-doped CeO2 Microspheres. Appl. Catal. B 2011, 102, 475. [Google Scholar] [CrossRef]
- Jiao, J.Z.; Li, S.H.; Huang, B.C. Preparation of Manganese Oxides Supported on Graphene Catalysts and their Activity in Low-Temperature NH3-SCR. Acta Phys. Chim. Sin. 2015, 31, 1383. [Google Scholar]
- Krishna, K.; Bueno-López, A.; Makkee, M.; Moulijn, J.A. Potential Rare Earth Modified CeO2 Catalysts for Soot Oxidation: I. Characterisation and Catalytic Activity with O2. Appl. Catal. B 2007, 75, 189. [Google Scholar] [CrossRef]
- Sui, L.; Yu, L. Diesel Soot Oxidation Catalyzed by Co-Ba-K Catalysts: Evaluation of the Performance of the Catalysts. Chem. Eng. J. 2008, 142, 327. [Google Scholar] [CrossRef]
- Liu, S.; Wu, X.; Weng, D.; Li, M.; Fan, J. Sulfation of Pt/Al2O3 Catalyst for Soot Oxidation: High Utilization of NO2 and Oxidation of Surface Oxygenated Complexes. Appl. Catal. B 2013, 138, 199. [Google Scholar] [CrossRef]
- Guilhaume, N.; Bassou, B.; Bergeret, G.; Bianchi, D.; Bosselet, F.; Desmartin-Chomel, A.; Jouguet, B.; Mirodatos, C. In situ Investigation of Diesel Soot Combustion Over an AgMnOx Catalyst. Appl. Catal. B 2012, 119, 287. [Google Scholar] [CrossRef]
- Zeng, L.; Cui, L.; Wang, C.; Guo, W.; Gong, C. In-situ Modified the Surface of Pt-Doped Perovskite Catalyst for Soot Oxidation. J. Hazard. Mater. 2020, 383, 121210. [Google Scholar] [CrossRef] [PubMed]
- Feng, N.; Chen, C.; Meng, J.; Liu, G.; Fang, F.; Wang, L.; Wan, H.; Guan, G. K–Mn Supported on Three-Dimensionally Ordered Macroporous La0.8Ce0.2FeO3 Catalysts for the Catalytic Combustion of Soot. Appl. Surf. Sci. 2017, 399, 114. [Google Scholar] [CrossRef]
- Xu, J.; Liu, J.; Zhao, Z.; Zheng, J.; Zhang, G.; Duan, A.; Jiang, G. Three-Dimensionally Ordered Macroporous LaCoxFe1−xO3 Perovskite-Type Complex Oxide Catalysts for Diesel Soot Combustion. Catal. Today 2010, 153, 136. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, Z.; Liu, J.; Jiang, G.; Duan, A.; Zheng, J.; Chen, S.; Zhou, R. Three Dimensionally Ordered Macroporous Ce1−xZrxO2 Solid Solutions for Diesel Soot Combustion. Chem. Commun. 2010, 46, 457. [Google Scholar] [CrossRef]
- Zhao, P.; Feng, N.; Fang, F.; Liu, G.; Chen, L.; Meng, J.; Chen, C.; Wang, L.; Wan, H.; Guan, G. Facile Synthesis of Three-Dimensional Ordered Macroporous Sr1−xKxTiO3 Perovskites with Enhanced Catalytic Activity for Soot Combustion. Catal. Sci. Technol. 2018, 8, 5462. [Google Scholar] [CrossRef]
- Mei, X.; Xiong, J.; Wei, Y.; Wang, C.; Wu, Q.; Zhao, Z.; Liu, J. Three-Dimensional Ordered Macroporous Perovskite-Type La1–xKxNiO3 Catalysts with Enhanced Catalytic Activity for Soot Combustion: The Effect of K-Substitution. Chin. J. Catal. 2019, 40, 722. [Google Scholar] [CrossRef]
- Alinezhadchamazketi, A.; Khodadadi, A.A.; Mortazavi, Y.; Nemati, A. Catalytic Evaluation of Promoted CeO2-ZrO2 by Transition, Alkali, and Alkaline-Earth Metal Oxides for Diesel Soot Oxidation. J. Environ. Sci. 2013, 25, 2498. [Google Scholar] [CrossRef]
- Matarrese, R.; Morandi, S.; Castoldi, L.; Villa, P.; Lietti, L. Removal of NOx and Soot Over Ce/Zr/K/Me (Me = Fe, Pt, Ru, Au) Oxide Catalysts. Appl. Catal. B 2017, 201, 318. [Google Scholar] [CrossRef]
- Liu, S.; Wu, X.; Lin, Y.; Li, M.; Weng, D. Active Oxygen-Assisted NO-NO2 Recycling and Decomposition of Surface Oxygenated Species on Diesel Soot with Pt/Ce0.6Zr0.4O2 Catalyst. Chin. J. Catal. 2014, 35, 407. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, Z.; Xu, C.; Duan, A.; Liu, J.; Chi, Y. Simultaneous Removal of Soot Particles and NO From Diesel Engines Over LaBO3 Perovskite-Type Oxides. J. Phys. Chem. 2008, 29, 649. [Google Scholar]
- Han, D.; E, J.; Deng, Y.; Chen, J.; Leng, E.; Liao, G.; Zhao, X.; Feng, C.; Zhang, F. A review of studies using hydrocarbon adsorption material for reducing hydrocarbon emissions from cold start of gasoline engine. Renew. Sustain. Energy Rev. 2021, 135, 110079. [Google Scholar] [CrossRef]
- Zhao, X.; E, J.; Liao, G.; Zhang, F.; Chen, J.; Deng, Y. Numerical simulation study on soot continuous regeneration combustion model of diesel particulate filter under exhaust gas heavy load. Fuel 2021, 287, 119795. [Google Scholar] [CrossRef]
- E, J.; Zhao, M.; Zuo, Q.; Zhang, B.; Zhang, Z.; Peng, Q.; Han, D.; Zhao, X.; Deng, Y. Effects analysis on diesel soot continuous regeneration performance of a rotary microwave-assisted regeneration diesel particulate filter. Fuel 2020, 260, 116353. [Google Scholar] [CrossRef]
- Zhang, Z.; Ye, J.; Tan, D.; Feng, Z.; Luo, J.; Tan, Y.; Huang, Y. The effects of Fe2O3 based DOC and SCR catalyst on the combustion and emission characteristics of a diesel engine fueled with biodiesel. Fuel 2021, 290, 120039. [Google Scholar] [CrossRef]
- Wei, K.; Yang, Y.; Hong, Y.; Zhong, D. A review on ice detection technology and ice elimination technology for wind turbine. Wind Energy 2019, 23, 433–457. [Google Scholar] [CrossRef]
- Zuo, H.; Liu, G.; E, J.; Zuo, W.; Wei, K.; Hu, W.; Tan, J.; Zhong, D. Catastrophic analysis on the stability of a large dish solar thermal power generation system with wind-induced vibration. Sol. Energy 2019, 183, 40–49. [Google Scholar] [CrossRef]
- Zuo, H.; Tan, J.; Wei, K.; Huang, Z.; Zhong, D.; Xie, F. Effects of different poses and wind speeds on wind-induced vibration characteristics of a dish solar concentrator system. Renew. Energy 2021, 168, 1308–1326. [Google Scholar] [CrossRef]

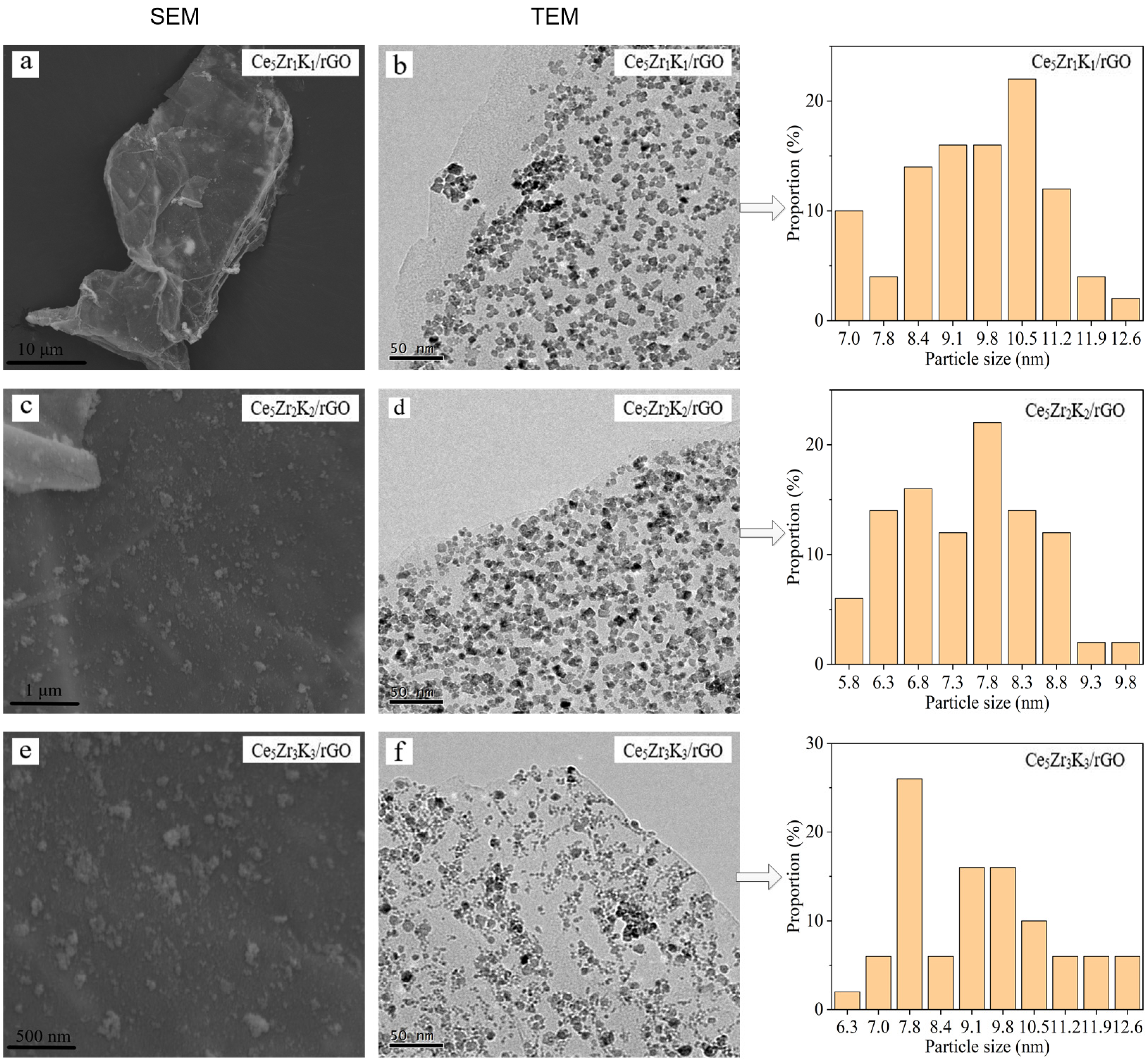


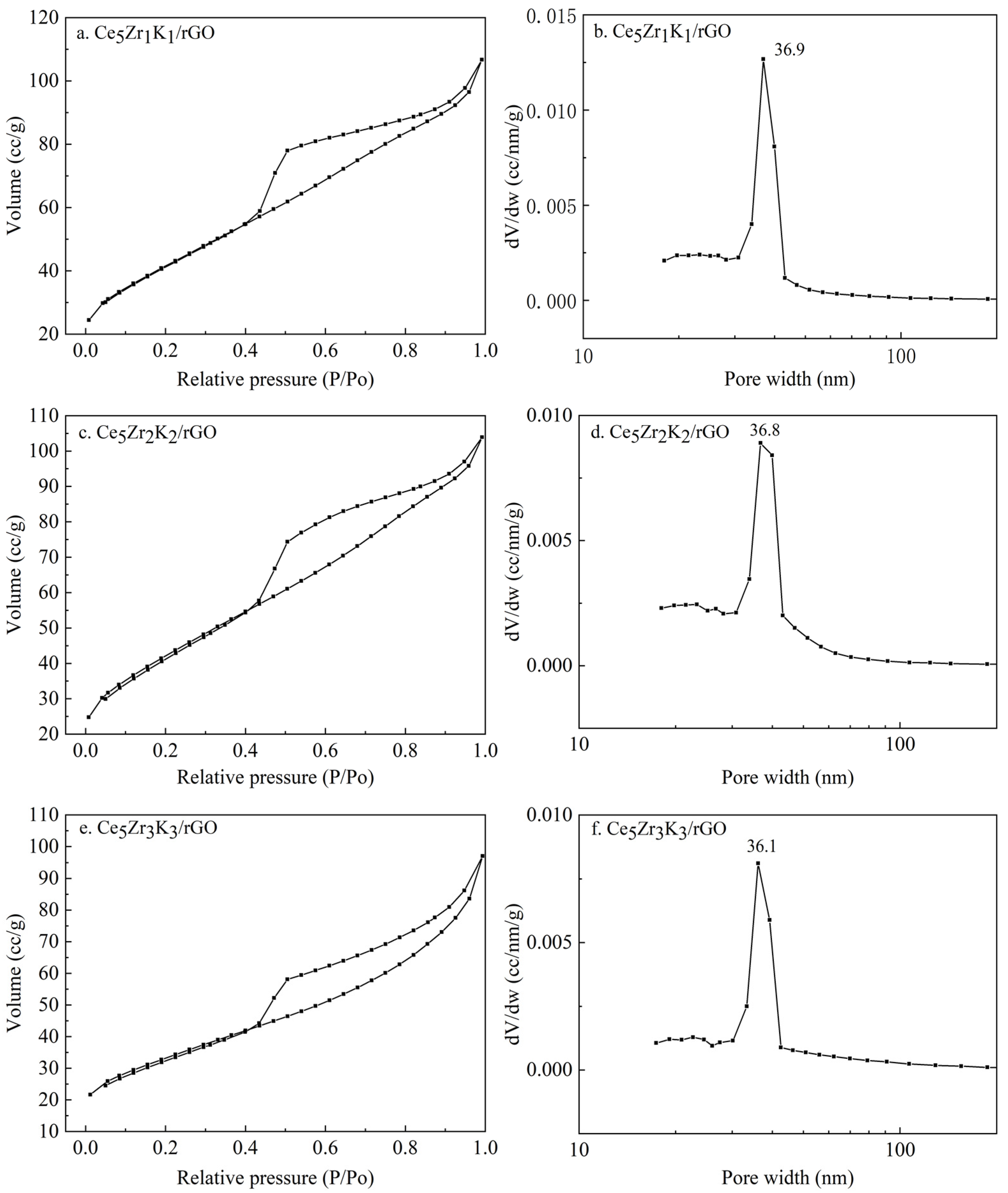

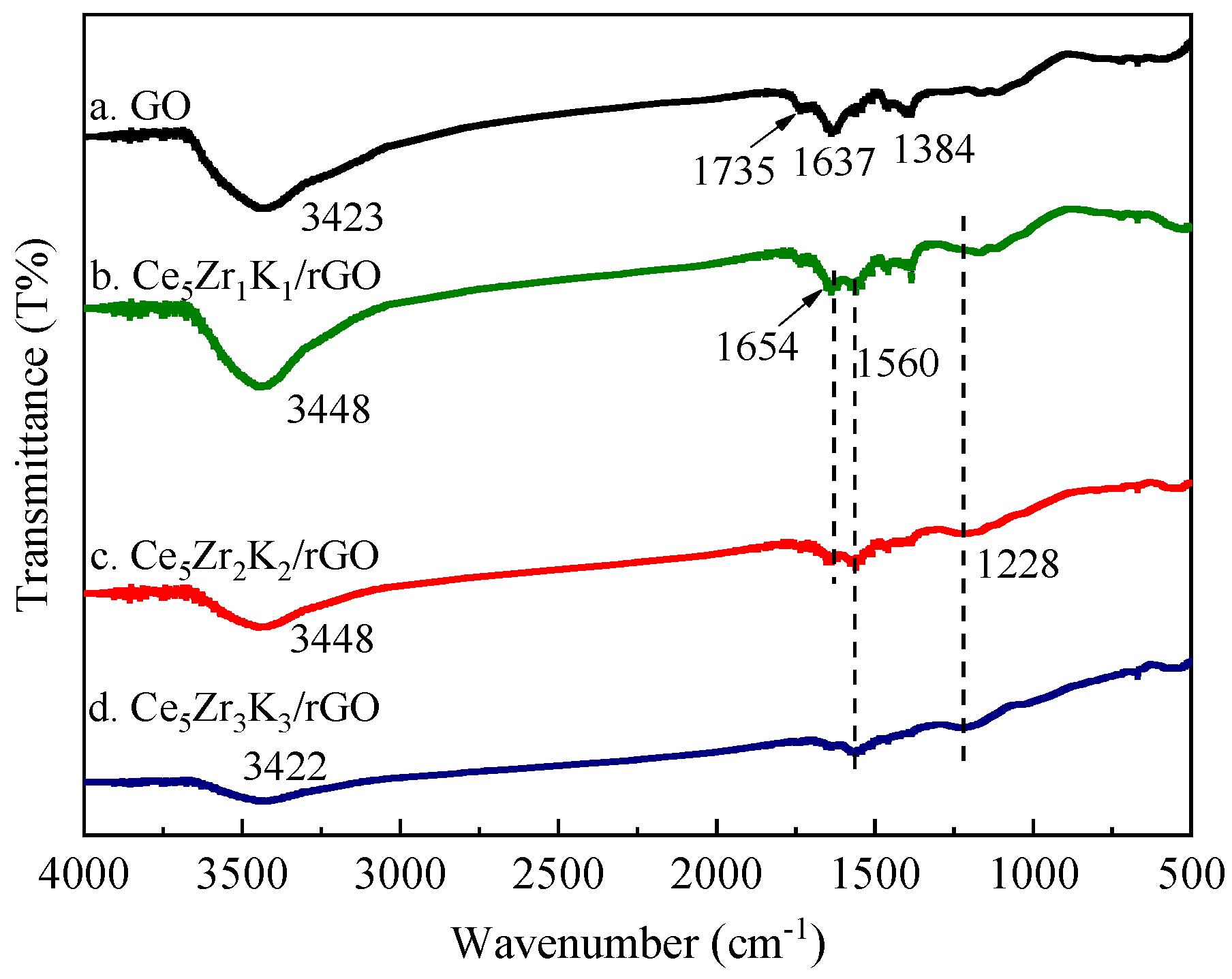
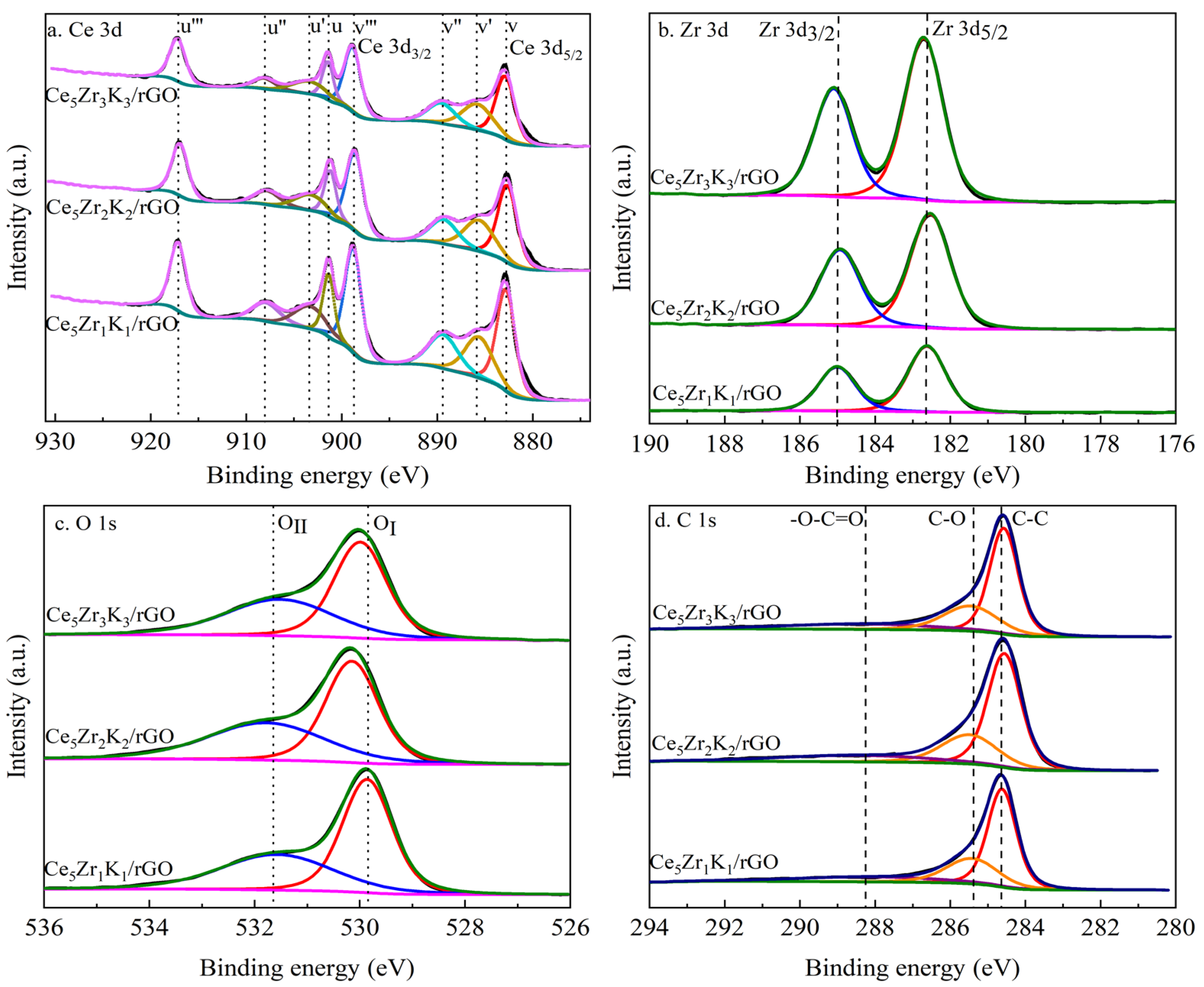

| Catalysts | Average Crystallite Size/nm | SBET/m2·g−1 |
|---|---|---|
| Ce5Zr1K1 | 8.3 | 117.2 |
| Ce5Zr2K2 | 6.9 | 151.5 |
| Ce5Zr3K3 | 6.7 | 152.4 |
| CeO2 | 8.9 | - |
| Catalysts | Atomic Surface Composition (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ce | Zr | O | C | Ce | O | C | |||||
| Ce3+ | Ce4+ | OⅠ | OⅡ | C-C | C-O | -O-C=O | |||||
| Ce5Zr1K1/rGO | 7.3 | 1.35 | 25.96 | 65.39 | 61.26 | 38.74 | 41.45 | 58.55 | 57.35 | 26.89 | 15.76 |
| Ce5Zr2K2/rGO | 4.68 | 2.11 | 27.00 | 66.21 | 61.83 | 38.17 | 40.84 | 59.16 | 59.07 | 25.30 | 15.62 |
| Ce5Zr3K3/rGO | 4.47 | 3.34 | 24.50 | 67.68 | 60.14 | 39.86 | 43.51 | 56.49 | 59.99 | 24.78 | 15.23 |
| Catalysts | Ce 3d3/2 (u‴)/eV | Zr 3d5/2/eV | OⅠ 1s/eV | OⅡ 1s/eV |
|---|---|---|---|---|
| Ce5Zr1K1/rGO | 917.3 | 182.6 | 529.8 | 531.6 |
| Ce5Zr2K2/rGO | 916.9 | 182.5 | 530.0 | 531.8 |
| Ce5Zr3K3/rGO | 917.0 | 182.7 | 529.9 | 531.5 |
| Catalysts | Ce3+/Ce4+ | OⅡ/OⅠ/ | Ce/Zr |
|---|---|---|---|
| Ce5Zr1K1/rGO | 1.58 | 1.41 | 5.4 |
| Ce5Zr2K2/rGO | 1.62 | 1.45 | 2.2 |
| Ce5Zr3K3/rGO | 1.51 | 1.30 | 1.3 |
| Catalyst Name | T50 (°C) | Refs. | |
|---|---|---|---|
| Tight | Loose | ||
| Catalyst-free | - | 604 | This work |
| CeO2-Sm2O3 | 417 | 517 | [30] |
| Pt/Al2O3 | - | 464 | [50] |
| Ag/MnOx | - | 498 | [51] |
| LaCo0.94Pt0.06O3 | - | 437 | [52] |
| 3DOM La0.8Ce0.2FeO3 | - | 434 | [53] |
| 3DOM LaCo0.5 Fe0.5O3 | - | 397 | [54] |
| 3DOM Ce0.8Zr0.2O2 | - | 396 | [55] |
| 3DOM Sr0.8K0.2TiO3 | - | 382 | [56] |
| CeO2 a | 419 | 523 | This work |
| Ce5Zr1K1/rGO a | 352 | 390 | This work |
| Ce5Zr2K2/rGO a | 339 | 383 | This work |
| Ce5Zr3K3/rGO a | 358 | 432 | This work |
| Catalyst Name | Tm (°C) | Refs. | |
|---|---|---|---|
| Tight | Loose | ||
| Au0.08/LaFeO3 | - | 359 | [9] |
| Pt0.1Ce0.68Zr0.31 | - | 430 | [60] |
| Pt0.1Al0.99 | - | 464 | [60] |
| 3DOM La0.95K0.05NiO3 | - | 341 | [57] |
| LaNiO3 | - | 431 | [61] |
| CoMgAl-LDH/rGO | 433 | 477 | [19] |
| Ce0.68Zr0.32O2 | 440 | 620 | [58] |
| K0.2Ce0.544Zr0.256O2 | 440 | 490 | [58] |
| CeZrK mixed oxides | - | 445 | [35] |
| CeZrKRu mixed oxides | - | 391 | [59] |
| CeO2 | 416 | 588 | This work |
| Ce5Zr1K1/rGO | 342 | 347 | This work |
| Ce5Zr2K2/rGO | 329 | 344 | This work |
| Ce5Zr3K3/rGO | 339 | 330 | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, G.; Tang, K.; Wu, D.; Li, Y.; Li, Y. Experimental Evaluation on the Catalytic Activity of a Novel CeZrK/rGO Nanocomposite for Soot Oxidation in Catalyzed Diesel Particulate Filter. Processes 2021, 9, 674. https://doi.org/10.3390/pr9040674
Wu G, Tang K, Wu D, Li Y, Li Y. Experimental Evaluation on the Catalytic Activity of a Novel CeZrK/rGO Nanocomposite for Soot Oxidation in Catalyzed Diesel Particulate Filter. Processes. 2021; 9(4):674. https://doi.org/10.3390/pr9040674
Chicago/Turabian StyleWu, Gang, Ke Tang, Deng Wu, Yuelin Li, and Yuqiang Li. 2021. "Experimental Evaluation on the Catalytic Activity of a Novel CeZrK/rGO Nanocomposite for Soot Oxidation in Catalyzed Diesel Particulate Filter" Processes 9, no. 4: 674. https://doi.org/10.3390/pr9040674
APA StyleWu, G., Tang, K., Wu, D., Li, Y., & Li, Y. (2021). Experimental Evaluation on the Catalytic Activity of a Novel CeZrK/rGO Nanocomposite for Soot Oxidation in Catalyzed Diesel Particulate Filter. Processes, 9(4), 674. https://doi.org/10.3390/pr9040674







