Despite its name, mustard gas is not produced from the mustard plant, but from its chemical substances. Mustard gas in its impure form is yellow-brown in color, with an odor similar to mustard plants. When exposed, mustard gas can cause serious injury to the eyes, skin, and respiratory tract [
4]. Sulfur mustards (bis [2-chloroethyl]-sulfide; 1,5-dichloro-3-thiapentane), which are related to the mustard gas group, may cause large skin blisters and, due to their high toxicity, are not suitable for medical use.
Mustard gas was initially developed as a devastating chemical warfare agent in World War I and was subsequently banned as part of the 1925 Geneva Protocol [
9]. However, during World War II, a ship explosion resulted in a vast number of military personnel being accidentally exposed to 2000 mustard gas bombs [
4,
8]. To prevent any further harm at the time, research to discover more efficient agents and protective measures was conducted, and it was found that nitrogen mustard agents were more stable (compared with sulfur mustards) [
9]. In 1942, Gilman, Goodman, and co-workers at Yale discovered that bis(2-chloromethyl) methylamine (or chlormethine) possessed anticancer effects in animal models [
10], and subsequent clinical trials showed encouraging results. Further clinical trials in patients with terminal malignant diseases resulted in successful outcomes. It is interesting to note that these findings were kept confidential due to the war, and were not disclosed for almost two decades when they were revealed by one of the discoverers [
11].
Nitrogen-based alkylators are the most frequently used alkylating agents in cancer therapy [
4,
12]. Following uptake, the nitrogen-based alkylator is metabolized to a highly reactive aziridinium derivative that alkylates DNA and inhibits DNA reduplication [
4]. There are several generations of nitrogen-based alkylating drugs (
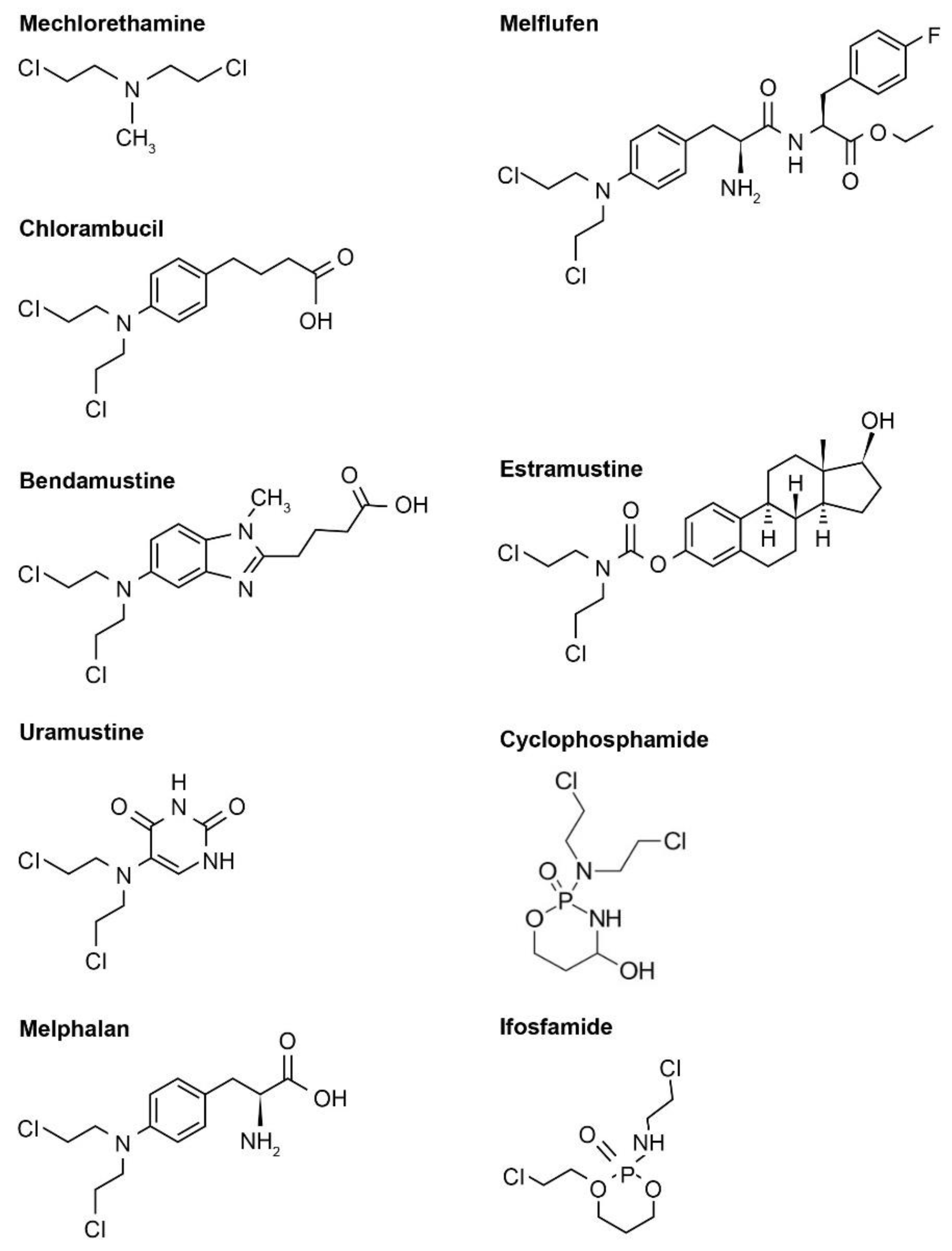
Table 1,
Figure 1).
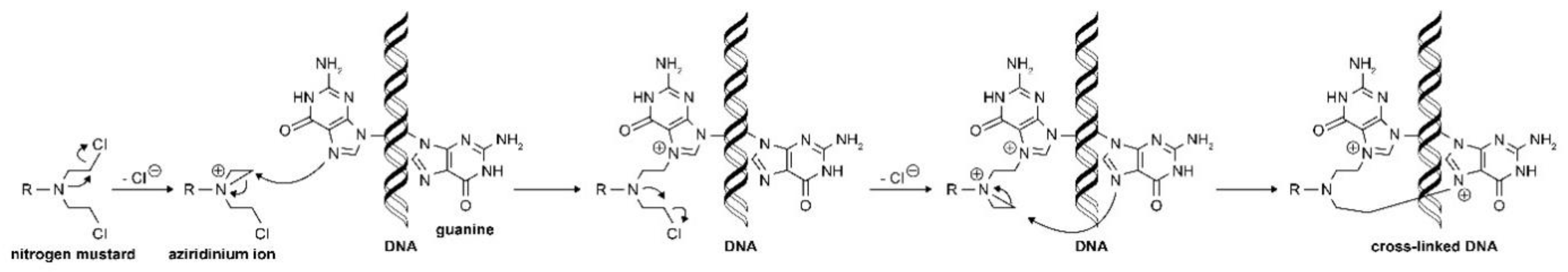
Among these highly reactive agents, the bis(2-chloroethyl) group is the main chemical constituent, and they all share a common mechanism of action, an in-situ generation of highly electrophilic cyclopropyl through an intramolecular nucleophilic substitution reaction [
12,
13]. The resulting aziridinium cation serves as the direct alkylating agent and, once formed, it undergoes nucleophilic attack by DNA bases, most often N-7 of guanine, to form a covalent bond (
Figure 2). The remaining molecule is important in determining the physical properties of the agent, including transport, distribution, lipophilicity, and reactivity [
12]. It is worth noting that a number of agents are prodrugs that have been designed to be selectively activated inside tumor cells. Overall, the underlying evolution of the nitrogen-based alkylator class is the design and development of molecules that are increasingly lipophilic and distributed effectively.
2.1. First-Generation Nitrogen-Based Alkylators
For first-generation agents, aliphatic groups were linked to the mustard pharmacophore, –N(CH
2CH
2Cl)
2. This group includes mechlorethamine hydrochloride (2-chloro-N-[2-chloroethyl]-N-methyl-ethanamine), a salt of a synthetic nitrogen-containing sulfur mustard derivative that has antineoplastic and lympholytic properties. Mechlorethamine hydrochloride, which was first approved in 1949 by the US Food and Drug Administration (FDA) [
13], induces a rapid response and is indicated for the treatment of lymphosarcoma, Hodgkin’s disease (stages III and IV), chronic myelocytic or chronic lymphocytic leukemia, polycythemia vera, and bronchogenic carcinoma. Due to its highly reactive profile, when mechlorethamine is combined with water in the body, it no longer exists in an active form. As a result, mechlorethamine cannot be taken orally, and is administered as an intravenous infusion over 20 min. However, dosage may vary depending on the clinical situation. Potential adverse events (AEs) include fatigue, nausea, and vomiting, which can be minimized by administering at bedtime, followed by a barbiturate and an anti-emetic. Serious AEs may include anaphylactic reactions or bleeding. Contraindications to mechlorethamine include hypersensitivity and known infectious diseases. Additionally, due to bone marrow suppression, administering vaccinations around mechlorethamine treatment should be avoided [
4].
2.2. Second-Generation Nitrogen-Based Alkylators
Chlormethine is too reactive for oral use and so must be administered intravenously. However, in second-generation agents, an electron-withdrawing aromatic ring is placed adjacent to the nitrogen atom, where the interaction between lone-pair electrons and the aromatic ring electrons makes the agent less prone to displacing the chloride ion that forms a transient aziridinium ring [
9]. Such modifications result in more stable molecules that are less reactive with serum and cellular constituents compared with the first-generation nitrogen mustards, allowing oral administration in tablet form. This group includes chlorambucil, melphalan, and bendamustine.
Chlorambucil (4-[bis(2-chloroethyl)amino] benzenebutanoic acid), approved by the FDA in 1957 [
13], is indicated for the palliative treatment of chronic lymphocytic leukemia, malignant lymphoma, lymphosarcoma, giant follicular lymphoma, and Hodgkin’s disease [
4]. Chlorambucil alkylates and cross-links DNA at all cell cycle phases, and the electron withdrawing properties of its aromatic aryl ring means that reactions with cellular components are slower compared with the first-generation nitrogen-based alkylator agents [
4]. Chlorambucil appears to be well tolerated by most patients [
4] and less toxic than mechlorethamine [
13]. Typically administered at 0.1–0.2 mg/kg/day for 3–6 weeks, the daily dose can be given in a single administration [
4]. Severe neutropenia seems to only occur in patients receiving a total dosage of 6.5 mg/kg or more as part of a continuous course of therapy. Once discontinued, this effect may be reversible following approximately 10 days from the last dose; bone marrow failure has been known to occur in rare cases. Other less frequent AEs include diarrhea, oral ulcerations, nausea, vomiting, central nervous system (CNS) damage (such as tremors, hallucinations, muscular twitching, increased risk of seizures in children with nephrotic syndrome), skin reactions, and infertility [
2,
4].
Melphalan (2-amino-3-[4-(bis[2-chloroethyl]amino) phenyl]propanoic acid) is a phenylalanine derivative of nitrogen mustard and was approved by the FDA in 1964 [
4,
13]. It is indicated for the palliative treatment of multiple myeloma and for the palliation of non-respectable epithelial ovarian carcinoma. Melphalan is also indicated to treat breast cancer, neuroblastoma, and rhabdomyosarcoma, and occasionally used for malignant melanoma due to lack of melanocytes [
2,
13]. Similar to other nitrogen-based alkylators, the cytotoxicity of melphalan is harnessed through DNA cross-linking, and melphalan is transported to tumor cells using L-leucine-type transporters [
13]. Melphalan can be given orally or intravenously, and dosing varies depending on route of administration, purpose, and the patient’s weight. Like with chlorambucil, daily dosage can be given at one time in an oral form. Melphalan is typically administered 6–10 mg daily for 2 to 3 weeks, followed by a rest interval of several weeks before starting the subsequent cycle. In combination with prednisone, melphalan has been used to enhance treatment effectiveness for multiple myeloma. Melphalan treatment has been linked with myelosuppression and lowered white blood cell count, increased risk of infection, and reduced platelet count; however, bone marrow suppression is generally reversible if melphalan is withdrawn early enough. Commonly reported AEs include nausea and vomiting, with possible allergic reactions following multiple rounds of treatment [
4].
Bendamustine hydrochloride (4-[5-[bis(2-chloroethyl)amino]-1-methylbenzimidazol-2-yl]butanoic acid) was designed in the 1960s, first marketed in the early 1970s in East Germany, and approved by the FDA almost 40 years later in 2008 [
4]. Bendamustine is indicated for the treatment of chronic lymphocytic leukemia, as well as multiple myeloma and indolent B-cell non-Hodgkin lymphoma [
4]. While both bendamustine and chlorambucil are water-soluble, and share metabolic pathways and mechanism of action, bendamustine, with its benzimidazole moiety, possesses a more favorable safety profile [
4,
13]. Bendamustine is distributed freely in the blood and hydrolyzed to metabolites with low cytotoxic activity. The standard dose (100–120 mg/m
2 body surface area) for bendamustine is administered intravenously over 30 to 60 min, with varying cycles for each indicated disease. The most common hematologic complications include lymphopenia, leukopenia, anemia, neutropenia, and thrombocytopenia. The most common non-hematologic AEs included pyrexia, nausea, diarrhea or constipation, headache, fatigue, anorexia, cough, rash, dyspnea, and stomatitis. In order to address any Grade 3/4 AEs, dose reduction or delayed administration could be implemented [
4]. There is some caution around the use of bendamustine as an anticancer strategy, mainly due to its potentially genotoxic and neurotoxic characteristics, with a case report describing a patient developing ascending paralysis following treatment [
13].
2.3. Third-Generation Nitrogen Mustards
In third-generation nitrogen mustards, the pyrimidine nucleus was selected as the carrier for the pharmacophore, as an approach to support oral administration [
4]. This group includes uracil mustard (uramustine; 5-[bis(2-chloroethyl)amino]-1
H-pyrimidine-2,4-dione), a bifunctional alkylating agent that acts on the cell cycle non-specifically. The activity of uramustine is derived from the formation of an unstable ethylenimmonium ion. Uramustine is a current treatment option for chronic lymphocytic leukemia, chronic myelogenous leukemia, Hodgkin lymphoma, non-Hodgkin lymphomata of the histiocytic or lymphocytic type, lymphosarcoma, ovarian cancer, and breast cancer. Uramustine is administered orally as capsules and taken once weekly for at least 4 weeks; the typical adult dose is 150 μg/kg. Uramustine is considered to be well tolerated and does not cause alopecia. Reported AEs include nausea, vomiting, diarrhea, dermatitis, nervousness, irritability, and depression. Additionally, due to the bone suppression effects, there is the potential of increased risk of microbial infections, bleeding and delayed healing [
4].
2.4. Phosphoramide Mustards
Phosphoramide mustards (1,3,2-oxazaphosphinane) are formed by the conversion of the base nitrogen mustard into non-toxic prodrugs, and then actively transported into cancer cells where they are enzymatically converted into their active, cytotoxic forms [
4]. Phosphoramide mustards include cyclophosphamide (2-[bis(2-chloroethyl)amino]-1,3,2λ
5-oxazaphosphinan-2-one) and ifosfamide (3-(2-chloroethyl)-2-[(2-chloroethyl)amino]-1,3,2λ
5-oxazaphosphinan-2-one), and current phosphoramide prodrugs share a common 2H-oxazaphosphorine scaffold.
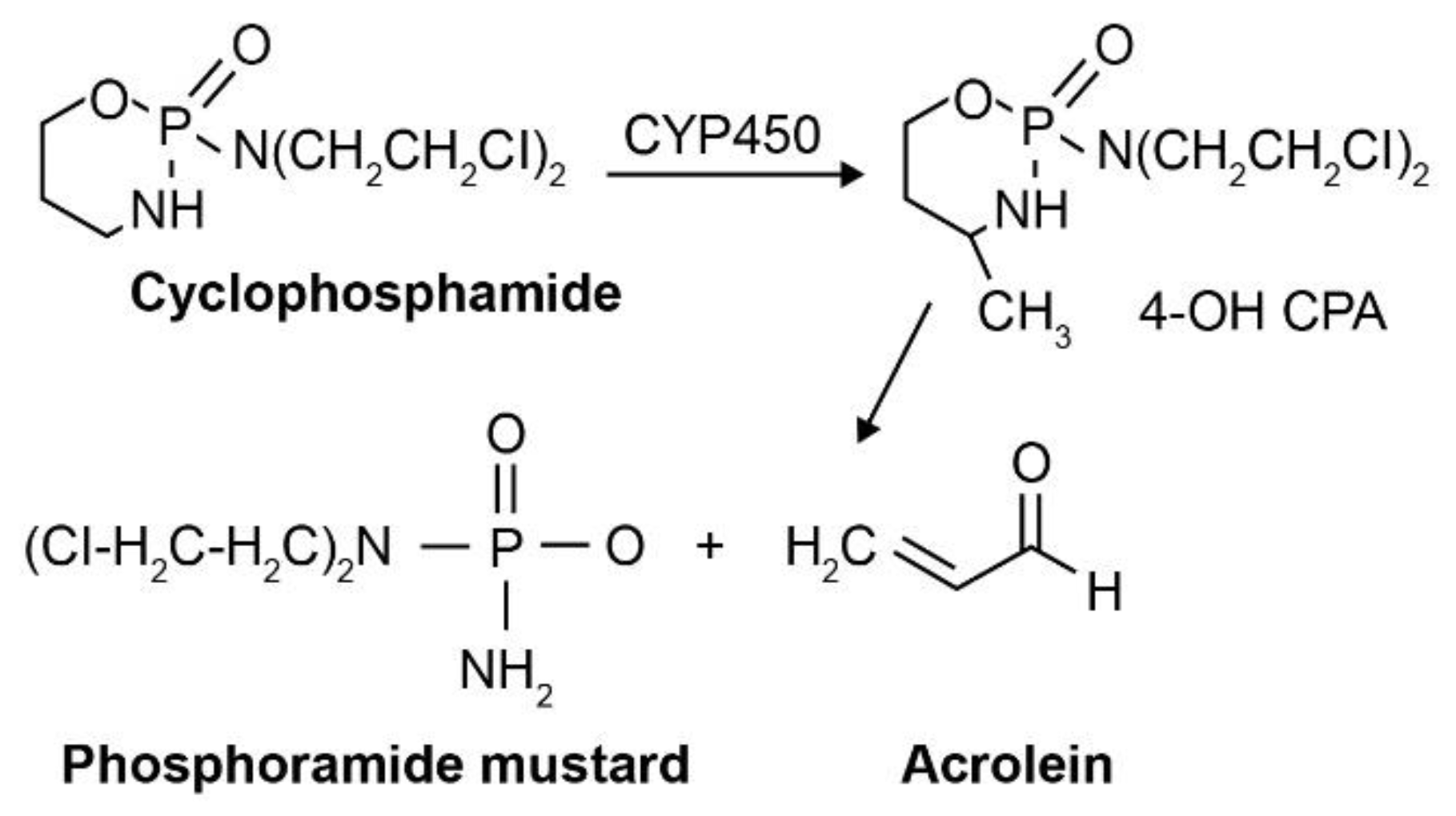
As a synthetic alkylating agent approved by the FDA in 1959 [
13], cyclophosphamide (N, N-bise[2-chloroethyl]-2-oxo-1-oxa-3-aza-2u{5}-phosphacyclohexan-2-amine) can be administered orally or intravenously. It was initially assumed that cyclophosphamide would be activated by phosphoramidase enzymes present in target tumor cells; however, this turned out not to be the case [
13]. Instead, activation occurs via cytochrome P450 (CYP450)-mediated hydroxylation in the liver [
14]. Upon cellular uptake, cyclophosphamide is metabolized by the CYP450 oxidase enzyme to form an active phosphoramide mustard product and acrolein, an inactive yet toxic metabolite that affects the bladder epithelium (
Figure 3) [
3,
13]. The active phosphoramide mustard is subsequently metabolized into an active nornitrogen mustard (normeclorethamine) [
13]. Cyclophosphamide targets various cancers, including lymphomata (e.g., multiple myeloma, Stage 3–4 malignant lymphomata, Hodgkin’s disease, Burkitt lymphoma, nodular or diffuse lymphocytic lymphoma) and some leukemias (chronic lymphocytic leukemia, acute myelogenous and monocytic leukemia) [
2,
4]. Depending on the clinical situation, doses vary and can be adjusted to account for antitumor activity and tolerable myelosuppression. Nausea, vomiting, and leukopenia are commonly reported AEs associated with cyclophosphamide treatment, and there is an increased risk of infections. Hematologic effects, such as thrombocytopenia or anemia, are known to occur occasionally, which can usually be reversed using dose reductions or interrupting treatment [
4]. Cyclophosphamide is often administered in combination with chemotherapy in order to address drug resistance in tumor cells and minimize AEs. For example, the standard combination treatment for advanced non-Hodgkin lymphoma is cyclophosphamide, doxorubicin, vincristine and prednisone (also known as CHOP) [
13].
Ifosfamide (3-[2-chloroethyl]-2-[(2-chloroethyl)amino] tetrahydro-2H-1,3,2-oxazaphosphorine 2-oxide) is an isomeric derivative of cyclophosphamide and was approved by the FDA in 1987 [
4,
13]. Ifosfamide produces 7-atom DNA cross-links (compared to 5-atom cross-links with cyclophosphamide). As with cyclophosphamide, ifosfamide requires hepatic activation by CYP450; however, due to its distinct metabolic pathway, generation of the cytotoxic metabolite occurs at a much slower rate than for cyclophosphamide [
13,
15]. Ifosfamide also displays dose-dependent pharmacokinetics, which may be seen at high concentrations in relevant metabolic pathways [
4]. Ifosfamide is used across a range of cancers, including germ-cell testicular cancer, breast cancer, non-Hodgkin lymphoma, soft tissue sarcoma, ovarian cancer, cervical cancer and lung cancer. Administered intravenously, the daily dose for ifosfamide is 1.2 g/m
2 body surface area for five consecutive days. Ifosfamide induces less myelotoxicity (mainly leukopenia) compared with cyclophosphamide, as well as exhibiting little cross-resistance. Commonly reported AEs include alopecia, nausea, vomiting, and transient nail ridging. There have been reports of neurotoxicity and urotoxicity following ifosfamide treatment, potentially connected to the formation of chloroacetaldehyde via N-dechloroethylation [
4].
2.5. Steroid-Coupled Nitrogen Mustards
Recent modifications highlight steroid-coupled nitrogen mustards as a promising approach that, for bifunctionality, bring together the alkylating agent effects and a steroid for selective uptake, or combined anticancer action. By acting as a biological carrier, the steroid allows the nitrogen mustard moiety to be easily transported across the cellular barrier [
8]. This group includes estramustine phosphate sodium (estradiol 3-[bis(2-chloroethyl)carbamate] 17-(dihydrogen phosphate), an oral, synthetic drug combining estradiol and mechlorethamine with a carbamate link. It is phosphorylated for improved water solubility and displays anti-androgenic effects. Estramustine phosphate sodium is primarily used for the palliative treatment of metastatic or progressive prostate cancer. It has been shown to be effective as an anticancer treatment option in combination with a 24-hydroxylase inhibitor (such as N-[4-(4-chlorophenyl)benzoyl]-2-(1H-imidazol-1-yl)-2-phenyl-1-aminoethane) [
3]. It is taken orally, either before or after food, and the recommended daily dose is 14 mg/kg body weight, administered across three or four doses. Once absorbed, estramustine phosphate is dephosphorylated into estramustine, its estrone analog and estradiol [
4]. Reported AEs include difficulty breathing, allergic reactions, and blood clots, where hypertension should be regularly assessed. Contraindications for estramustine include hypersensitivity to estradiol or nitrogen mustard [
4].
2.6. Peptide Conjugation
Several aminopeptidases have been known to be overexpressed in malignancies, implicating their role as potential antitumor targets. The multifunctional protein aminopeptidase N (APN) has received the most attention due to its association with the varying phenotypes of human malignancies, particularly the fast-growing, aggressive types [
16]. As research into this area develops, peptides are emerging as a promising therapeutic approach, exhibiting traits that make them ideal carriers of anticancer drugs to primary tumor sites (such as ease of synthesis, size, biocompatibility, and ability to target tumor cells).
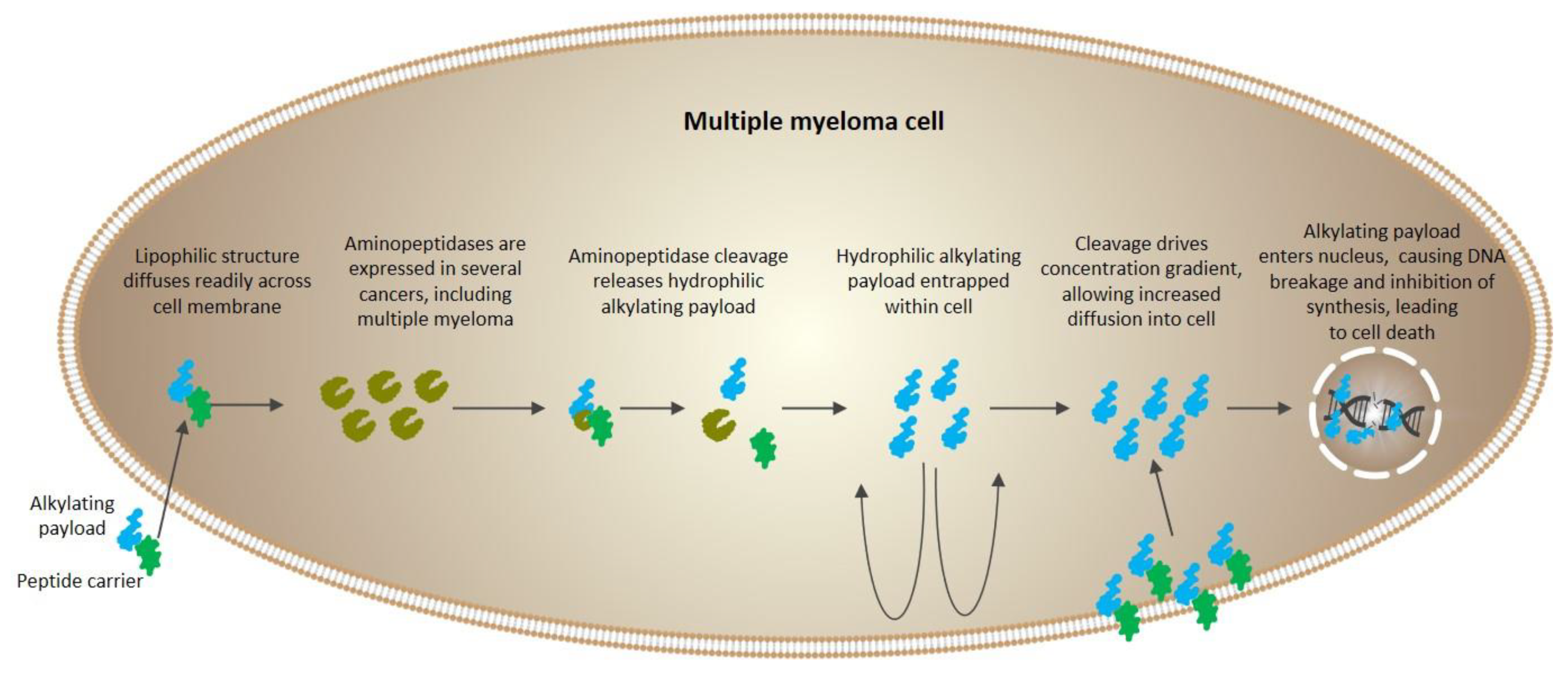
Melphalan flufenamide (L-melphalanyl-p-L-fluorophenylalanine ethyl ester hydrochloride), also known as melflufen and previously denoted J1, is the ethyl ester of a dipeptide consisting of melphalan and
para-fluoro-L-phenylalanine. Melflufen is highly lipophilic, and therefore is rapidly taken up by myeloma cells [
16]. Inside the cell, cleavage of melflufen by aminopeptidases leads to enrichment of hydrophilic alkylating metabolites (such as melphalan) (
Figure 4). In essence, by employing this simple peptide bond directly to APN-expressing cells, the agent provides a peptidase-potentiated effect, enriching its anti-myeloma activity [
16,
17,
18].
Melflufen has been shown from in vitro studies to be more cytotoxic to myeloma cells than to peripheral blood mononuclear cells, as well as exhibiting activity in melphalan-resistant myeloma cells [
19,
20]. Studies using myeloma tumor samples have shown melphalan exposure to be 50 times higher with melflufen compared with direct administration of melphalan in culture. Melflufen has also shown to be substantially more cytotoxic than melphalan (approximately 50-fold) [
19,
21].
Melflufen is currently in late-stage clinical development, and US marketing approval was submitted in June 2020 [
22]. Priority review was granted in August 2020 [
23].