Antimicrobial Effect of Phytochemicals from Edible Plants
Abstract
1. Introduction
2. Antimicrobial Properties of Edible Plants
3. Photosensitizers from Edible Plants as a Source of New Drugs
4. Use of Natural Plant Antimicrobials for Food Disinfection
5. Essential Oils
6. Antimicrobial Peptides
7. Plant Extracts
8. Limitations in Plant Antimicrobials as Food Preservatives
9. Ultrasonic Activation
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thomford, N.E.; Senthebane, D.A.; Rowe, A.; Munro, D.; Seele, P.; Maroyi, A.; Dzobo, K. Natural products for drug discovery in the 21st century: Innovations for novel drug discovery. Int. J. Mol. Sci. 2018, 19, 1578. [Google Scholar] [CrossRef] [PubMed]
- Verpoorte, R. Pharmacognosy in the new millennium: Leadfinding and biotechnology. J. Pharm. Pharmacol. 2000, 52, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Yarmolinsky, L.; Bronstein, M.; Gorelick, J. Review: Inhibition of bacterial quorum sensing by plant extracts. Isr. J. Plant Sci. 2015, 62, 294–297. [Google Scholar] [CrossRef]
- Yarmolinsky, L.L.; Budovsky, A.; Yarmolinsky, L.L.; Khalfin, B.; Glukhman, V.; Ben-Shabat, S. Effect of bioactive phytochemicals from phlomis viscosa poiret on wound healing. Plants 2019, 8, 609. [Google Scholar] [CrossRef] [PubMed]
- Zaccai, M.; Yarmolinsky, L.; Khalfin, B.; Budovsky, A.; Gorelick, J.; Dahan, A.; Ben-Shabat, S. Medicinal Properties of Lilium candidum L. and Its Phytochemicals. Plants 2020, 9, 959. [Google Scholar] [CrossRef]
- Sharma, H.B.; Panigrahi, S.; Sarmah, A.K.; Dubey, B.K. Antibacterial and antibiofilm activities of Mayan medicinal plants against Methicillinsusceptible and -resistant strains of Staphylococcus aureus. Sci. Total Environ. 2019, 135907. [Google Scholar] [CrossRef]
- Wang, Z.; Zhou, Y.; Shi, X.; Xia, X.; He, Y.; Zhu, Y.; Xie, T.; Liu, T.; Xu, X.; Luo, X. Comparison of chemical constituents in diverse zanthoxylum herbs, and evaluation of their relative antibacterial and nematicidal activity. Food Biosci. 2021, 42, 101206. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Escolà-Vergé, L.; Los-Arcos, I.; Almirante, B. New antibiotics for the treatment of infections by multidrug-resistant microorganisms. Med. Clín. Engl. Ed. 2020, 154, 351–357. [Google Scholar] [CrossRef]
- Neuman, H.; Forsythe, P.; Uzan, A.; Avni, O.; Koren, O. Antibiotics in early life: Dysbiosis and the damage done. FEMS Microbiol. Rev. 2018, 42, 489–499. [Google Scholar] [CrossRef]
- Abrahamse, H.; Hamblin, M.R. New photossensitizersfot photodynamic therapy. Biochem. J. 2017, 473, 347–364. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, A.; Grinholc, M. Combined Antimicrobial Activity of Photodynamic Inactivation and Antimicrobials-State of the Art. Front. Microbiol. 2018, 9, 930. [Google Scholar] [CrossRef] [PubMed]
- Polat, E.; Kang, K. Natural photosensitizers in antimicrobial photodynamic therapy. Biomedicines 2021, 9, 584. [Google Scholar] [CrossRef] [PubMed]
- Christina Pires Gonçalves, L. Photophysical properties and therapeutic use of natural photosensitizers. J. Photochem. Photobiol. 2021, 7, 100052. [Google Scholar] [CrossRef]
- Foresto, E.; Gilardi, P.; Ibarra, L.E.; Cogno, I.S. Light-activated green drugs: How we can use them in photodynamic therapy and mass-produce them with biotechnological tools. Phytomedicine Plus 2021, 1, 100044. [Google Scholar] [CrossRef]
- Nakonechny, F.; Nisnevitch, M. Different Aspects of Using Ultrasound to Combat Microorganisms. Adv. Funct. Mater. 2021, 2011042. [Google Scholar] [CrossRef]
- Yadav, A.; Kumar, A.; Das, M.; Tripathi, A. Sodium benzoate, a food preservative, affects the functional and activation status of splenocytes at non cytotoxic dose. Food Chem. Toxicol. 2016, 88, 40–47. [Google Scholar] [CrossRef]
- Ren, L.; Meng, M.; Wang, P.; Xu, Z.; Eremin, S.A.; Zhao, J.; Yin, Y.; Xi, R. Determination of sodium benzoate in food products by fluorescence polarization immunoassay. Talanta 2014, 121, 136–143. [Google Scholar] [CrossRef]
- Seidi Damyeh, M.; Mereddy, R.; Netzel, M.E.; Sultanbawa, Y. An insight into curcumin-based photosensitization as a promising and green food preservation technology. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1727–1759. [Google Scholar] [CrossRef]
- Ceccanti, C.; Landi, M.; Benvenuti, S.; Pardossi, A.; Guidi, L. Mediterranean Wild Edible Plants: Weeds or “New Functional Crops”? Molecules 2018, 23, 2299. [Google Scholar] [CrossRef]
- Abdel-Reheem, M.A.T.; Oraby, M.M. Anti-microbial, cytotoxicity, and necrotic ripostes of Pimpinella anisum essential oil. Ann. Agric. Sci. 2015, 60, 335–340. [Google Scholar] [CrossRef]
- Abdelrahman, M.; Jogaiah, S. Saponins Versus Plant Fungal Pathogens. Bioact. Mol. Plant Def. 2020, 37–45. [Google Scholar] [CrossRef]
- Budovsky, A.; Yarmolinsky, L.; Ben-Shabat, S. Effect of medicinal plants on wound healing. Wound Repair Regen. 2015, 23, 171–183. [Google Scholar] [CrossRef]
- Isah, T. Stress and defense responses in plant secondary metabolites production. Biol. Res. 2019, 52, 39. [Google Scholar] [CrossRef]
- Sánchez-Maldonado, A.F.; Schieber, A.; Gänzle, M.G. Structure-function relationships of the antibacterial activity of phenolic acids and their metabolism by lactic acid bacteria. J. Appl. Microbiol. 2011, 111, 1176–1184. [Google Scholar] [CrossRef]
- Liu, J.; Du, C.; Beaman, H.T.; Monroe, M.B.B. Characterization of Phenolic Acid Antimicrobial and Antioxidant Structure-Property Relationships. Pharmaceutics 2020, 12, 419. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, H.; Rao, S.; Sun, J.; Ma, C.; Li, J. p-Coumaric acid kills bacteria through dual damage mechanisms. Food Control 2012, 25, 550–554. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, M.; Zhao, Z.; Yu, S. The antibiotic activity and mechanisms of sugarcane (Saccharum officinarum L.) bagasse extract against food-borne pathogens. Food Chem. 2015, 185, 112–118. [Google Scholar] [CrossRef]
- Alves, M.J.; Ferreira, I.C.F.R.; Froufe, H.J.C.; Abreu, R.M.V.; Martins, A.; Pintado, M. Antimicrobial activity of phenolic compounds identified in wild mushrooms, SAR analysis and docking studies. J. Appl. Microbiol. 2013, 115, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Vamsi Krishna, N.A.; Saradhi, P.M. Cumulative activity of the p-coumaric acid and syringaldehyde for antimicrobial activity of different microbial strains. Pelagia Res. Libr. Eur. J. Exp. Biol. 2014, 4, 40–43. [Google Scholar]
- Kulapichitr, F.; Borompichaichartkul, C.; Fang, M.; Suppavorasatit, I.; Cadwallader, K.R. Effect of post-harvest drying process on chlorogenic acids, antioxidant activities and CIE-Lab color of Thai Arabica green coffee beans. Food Chem. 2022, 366, 130504. [Google Scholar] [CrossRef]
- Santana-Gálvez, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. Chlorogenic Acid: Recent Advances on Its Dual Role as a Food Additive and a Nutraceutical against Metabolic Syndrome. Molecules 2017, 22, 358. [Google Scholar] [CrossRef] [PubMed]
- Karunanidhi, A.; Thomas, R.; van Belkum, A.; Neela, V. In vitro antibacterial and antibiofilm activities of chlorogenic acid against clinical isolates of Stenotrophomonas maltophilia including the trimethoprim/sulfamethoxazole resistant strain. Biomed Res. Int. 2013, 2013, 392058. [Google Scholar] [CrossRef] [PubMed]
- Bajko, E.; Kalinowska, M.; Borowski, P.; Siergiejczyk, L.; Lewandowski, W. 5-O-Caffeoylquinic acid: A spectroscopic study and biological screening for antimicrobial activity. LWT-Food Sci. Technol. 2016, 65, 471–479. [Google Scholar] [CrossRef]
- Farzaei, M.H.; Abdollahi, M.; Rahimi, R. Role of dietary polyphenols in the management of peptic ulcer. World J. Gastroenterol. 2015, 21, 6499–6517. [Google Scholar] [CrossRef]
- Ayseli, M.T.; İpek Ayseli, Y. Flavors of the future: Health benefits of flavor precursors and volatile compounds in plant foods. Trends Food Sci. Technol. 2016, 48, 69–77. [Google Scholar] [CrossRef]
- Fu, L.; Lu, W.; Zhou, X. Phenolic Compounds and In Vitro Antibacterial and Antioxidant Activities of Three Tropic Fruits: Persimmon, Guava, and Sweetsop. Biomed Res. Int. 2016, 2016, 4287461. [Google Scholar] [CrossRef]
- Sousa, R.; Muñoz-Mahamud, E.; Quayle, J.; Dias da Costa, L.; Casals, C.; Scott, P.; Leite, P.; Vilanova, P.; Garcia, S.; Ramos, M.H.; et al. Is asymptomatic bacteriuria a risk factor for prosthetic joint infection? Clin. Infect. Dis. 2014, 59, 41–47. [Google Scholar] [CrossRef]
- Puupponen-Pimiä, R.; Nohynek, L.; Meier, C.; Kähkönen, M.; Heinonen, M.; Hopia, A.; Oksman-Caldentey, K.M. Antimicrobial properties of phenolic compounds from berries. J. Appl. Microbiol. 2001, 90, 494–507. [Google Scholar] [CrossRef]
- Aljadi, A.M.; Yusoff, K.M. Isolation and identification of phenolic acids in Malaysian honey with antibacterial properties. Turkish J. Med. Sci. 2003, 33, 229–236. [Google Scholar]
- Chun, S.-S.; Vattem, D.A.; Lin, Y.-T.; Shetty, K. Phenolic antioxidants from clonal oregano (Origanum vulgare) with antimicrobial activity against Helicobacter pylori. Process Biochem. 2005, 40, 809–816. [Google Scholar] [CrossRef]
- Amarowicz, R.; Dykes, G.A.; Pegg, R.B. Antibacterial activity of tannin constituents from Phaseolus vulgaris, Fagoypyrum esculentum, Corylus avellana and Juglans nigra. Fitoterapia 2008, 79, 217–219. [Google Scholar] [CrossRef] [PubMed]
- Pommier, Y.; Leo, E.; Zhang, H.; Marchand, C. DNA topoisomerases and their poisoning by anticancer and antibacterial drugs. Chem. Biol. 2010, 17, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Elmasri, W.A.; Zhu, R.; Peng, W.; Al-Hariri, M.; Kobeissy, F.; Tran, P.; Hamood, A.N.; Hegazy, M.F.; Paré, P.W.; Mechref, Y. Multitargeted Flavonoid Inhibition of the Pathogenic Bacterium Staphylococcus aureus: A Proteomic Characterization. J. Proteome Res. 2017, 16, 2579–2586. [Google Scholar] [CrossRef] [PubMed]
- Ramalhete, C.; Spengler, G.; Martins, A.; Martins, M.; Viveiros, M.; Mulhovo, S.; Ferreira, M.-J.U.; Amaral, L. Inhibition of efflux pumps in methicillin-resistant Staphylococcus aureus and Enterococcus faecalis resistant strains by triterpenoids from Momordica balsamina. Int. J. Antimicrob. Agents 2011, 37, 70–74. [Google Scholar] [CrossRef]
- Liu, B.; Chen, F.; Bi, C.; Wang, L.; Zhong, X.; Cai, H.; Deng, X.; Niu, X.; Wang, D. Quercitrin, an inhibitor of Sortase A, interferes with the adhesion of Staphylococcal aureus. Molecules 2015, 20, 6533–6543. [Google Scholar] [CrossRef]
- Cushnie, T.P.T.; Lamb, A.J. Recent advances in understanding the antibacterial properties of flavonoids. Int. J. Antimicrob. Agents 2011, 38, 99–107. [Google Scholar] [CrossRef]
- Tsuchiya, H. Membrane Interactions of Phytochemicals as Their Molecular Mechanism Applicable to the Discovery of Drug Leads from Plants. Molecules 2015, 20, 18923–18966. [Google Scholar] [CrossRef]
- Ouyang, J.; Sun, F.; Feng, W.; Sun, Y.; Qiu, X.; Xiong, L.; Liu, Y.; Chen, Y. Quercetin is an effective inhibitor of quorum sensing, biofilm formation and virulence factors in Pseudomonas aeruginosa. J. Appl. Microbiol. 2016, 120, 966–974. [Google Scholar] [CrossRef]
- Ilk, S.; Sağlam, N.; Özgen, M.; Korkusuz, F. Chitosan nanoparticles enhances the anti-quorum sensing activity of kaempferol. Int. J. Biol. Macromol. 2017, 94, 653–662. [Google Scholar] [CrossRef]
- Terán, W.; Krell, T.; Ramos, J.L.; Gallegos, M.-T. Effector-repressor interactions, binding of a single effector molecule to the operator-bound TtgR homodimer mediates derepression. J. Biol. Chem. 2006, 281, 7102–7109. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.; Tan, K.S. Effects of Epigallocatechin gallate against Enterococcus faecalis biofilm and virulence. Arch. Oral Biol. 2015, 60, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Rasul, A.; Millimouno, F.M.; Ali Eltayb, W.; Ali, M.; Li, J.; Li, X. Pinocembrin: A Novel Natural Compound with Versatile Pharmacological and Biological Activities. Biomed Res. Int. 2013, 2013, 379850. [Google Scholar] [CrossRef] [PubMed]
- Soromou, L.W.; Zhang, Y.; Cui, Y.; Wei, M.; Chen, N.; Yang, X.; Huo, M.; Baldé, A.; Guan, S.; Deng, X.; et al. Subinhibitory concentrations of pinocembrin exert anti-Staphylococcus aureus activity by reducing α-toxin expression. J. Appl. Microbiol. 2013, 115, 41–49. [Google Scholar] [CrossRef]
- Cherubin, P.; Garcia, M.C.; Curtis, D.; Britt, C.B.T.; Craft Jr, J.W.; Burress, H.; Berndt, C.; Reddy, S.; Guyette, J.; Zheng, T.; et al. Inhibition of Cholera Toxin and Other AB Toxins by Polyphenolic Compounds. PLoS ONE 2016, 11, e0166477. [Google Scholar] [CrossRef]
- Yang, J.Y.; Lee, H.S. Evaluation of antioxidant and antibacterial activities of morin isolated from mulberry fruits (Morus alba L.). J. Korean Soc. Appl. Biol. Chem. 2012, 55, 485–489. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, X.; Liu, S.; Li, G.; Shi, L.; Dong, J.; Li, W.; Deng, X.; Niu, X. Morin hydrate attenuates Staphylococcus aureus virulence by inhibiting the self-assembly of α-hemolysin. J. Appl. Microbiol. 2015, 118, 753–763. [Google Scholar] [CrossRef]
- Brynildsen, M.P.; Winkler, J.A.; Spina, C.S.; MacDonald, I.C.; Collins, J.J. Potentiating antibacterial activity by predictably enhancing endogenous microbial ROS production. Nat. Biotechnol. 2013, 31, 160–165. [Google Scholar] [CrossRef]
- Chalal, M.; Klinguer, A.; Echairi, A.; Meunier, P.; Vervandier-Fasseur, D.; Adrian, M. Antimicrobial Activity of Resveratrol Analogues. Molecules 2014, 19, 7679–7688. [Google Scholar] [CrossRef]
- Chong, J.; Poutaraud, A.; Hugueney, P. Metabolism and roles of stilbenes in plants. Plant Sci. 2009, 177, 143–155. [Google Scholar] [CrossRef]
- Petersen, A.; Wang, C.; Crocoll, C.; Halkier, B.A. Biotechnological approaches in glucosinolate production. J. Integr. Plant Biol. 2018, 60, 1231–1248. [Google Scholar] [CrossRef] [PubMed]
- Verkerk, R.; Schreiner, M.; Krumbein, A.; Ciska, E.; Holst, B.; Rowland, I.; De Schrijver, R.; Hansen, M.; Gerhäuser, C.; Mithen, R.; et al. Glucosinolates in Brassica vegetables: The influence of the food supply chain on intake, bioavailability and human health. Mol. Nutr. Food Res. 2009, 53 (Suppl. 2), S219. [Google Scholar] [CrossRef]
- Saladino, F.; Bordin, K.; Luciano, F.B.; Franzón, M.F.; Mañes, J.; Meca, G. Antimicrobial Activity of the Glucosinolates BT-Glucosinolates; Mérillon, J.-M., Ramawat, K.G., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 249–274. ISBN 978-3-319-25462-3. [Google Scholar]
- Eisenreich, W.; Rohdich, F.; Bacher, A. Deoxyxylulose phosphate pathway to terpenoids. Trends Plant Sci. 2001, 6, 78–84. [Google Scholar] [CrossRef]
- Hartmann, T. From waste products to ecochemicals: Fifty years research of plant secondary metabolism. Phytochemistry 2007, 68, 2831–2846. [Google Scholar] [CrossRef] [PubMed]
- Vilas, V.; Philip, D.; Mathew, J. Essential oil mediated synthesis of silver nanocrystals for environmental, anti-microbial and antioxidant applications. Mater. Sci. Eng. C. Mater. Biol. Appl. 2016, 61, 429–436. [Google Scholar] [CrossRef]
- Guimarães, A.C.; Meireles, L.M.; Lemos, M.F.; Guimarães, M.C.C.; Endringer, D.C.; Fronza, M.; Scherer, R. Antibacterial Activity of Terpenes and Terpenoids Present in Essential Oils. Molecules 2019, 24, 2471. [Google Scholar] [CrossRef]
- Ribeiro, M.; Malheiro, J.; Grenho, L.; Fernandes, M.H.; Simões, M. Cytotoxicity and antimicrobial action of selected phytochemicals against planktonic and sessile Streptococcus mutans. PeerJ 2018, 6, e4872. [Google Scholar] [CrossRef]
- Zore, G.B.; Thakre, A.D.; Jadhav, S.; Karuppayil, S.M. Terpenoids inhibit Candida albicans growth by affecting membrane integrity and arrest of cell cycle. Phytomedicine 2011, 18, 1181–1190. [Google Scholar] [CrossRef]
- Usmani, Y.; Ahmed, A.; Faizi, S.; Versiani, M.A.; Shamshad, S.; Khan, S.; Simjee, S.U. Antimicrobial and biofilm inhibiting potential of an amide derivative [N-(2′, 4′-dinitrophenyl)-3β-hydroxyurs-12-en-28-carbonamide] of ursolic acid by modulating membrane potential and quorum sensing against colistin resistant Acinetobacter baumannii. Microb. Pathog. 2021, 157, 104997. [Google Scholar] [CrossRef]
- Jäger, S.; Trojan, H.; Kopp, T.; Laszczyk, M.N.; Scheffler, A. Pentacyclic triterpene distribution in various plants-rich sources for a new group of multi-potent plant extracts. Molecules 2009, 14, 2016–2031. [Google Scholar] [CrossRef]
- Szakiel, A.; Pączkowski, C.; Pensec, F.; Bertsch, C. Fruit cuticular waxes as a source of biologically active triterpenoids. Phytochem. Rev. 2012, 11, 263–284. [Google Scholar] [CrossRef]
- Woźniak, Ł.; Skąpska, S.; Marszałek, K. Ursolic Acid--A Pentacyclic Triterpenoid with a Wide Spectrum of Pharmacological Activities. Molecules 2015, 20, 20614–20641. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Wang, W.; Zhang, J.; Wang, T.; Liu, M.; Yang, M.; Sun, Z.; Li, X.; Li, Y. Antimicrobial and antibiofilm activities of ursolic acid against carbapenem-resistant Klebsiella pneumoniae. J. Antibiot. 2020, 73, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Lyu, X.; Wang, L.; Shui, Y.; Jiang, Q.; Chen, L.; Yang, W.; He, X.; Zeng, J.; Li, Y. Ursolic acid inhibits multi-species biofilms developed by Streptococcus mutans, Streptococcus sanguinis, and Streptococcus gordonii. Arch. Oral Biol. 2021, 125, 105107. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-M.; Jhan, Y.-L.; Tsai, S.-J.; Chou, C.-H. The Pleiotropic Antibacterial Mechanisms of Ursolic Acid against Methicillin-Resistant Staphylococcus aureus (MRSA). Molecules 2016, 21, 884. [Google Scholar] [CrossRef]
- Kim, S.G.; Kim, M.J.; Jin, D.; Park, S.N.; Cho, E.; Freire, M.O.; Jang, S.J.; Park, Y.J.; Kook, J.K. Antimicrobial effect of ursolic acid and oleanolic acid against methicillin-resistant staphylococcus aureus. Korean J. Microbiol. 2012, 48, 212–215. [Google Scholar] [CrossRef][Green Version]
- Horiuchi, K.; Shiota, S.; Hatano, T.; Yoshida, T.; Kuroda, T.; Tsuchiya, T. Antimicrobial activity of oleanolic acid from Salvia officinalis and related compounds on vancomycin-resistant enterococci (VRE). Biol. Pharm. Bull. 2007, 30, 1147–1149. [Google Scholar] [CrossRef]
- Siddique, H.R.; Saleem, M. Beneficial health effects of lupeol triterpene: A review of preclinical studies. Life Sci. 2011, 88, 285–293. [Google Scholar] [CrossRef]
- Sharma, P.; Tyagi, A.; Bhansali, P.; Pareek, S.; Singh, V.; Ilyas, A.; Mishra, R.; Poddar, N.K. Saponins: Extraction, bio-medicinal properties and way forward to anti-viral representatives. Food Chem. Toxicol. 2021, 150, 112075. [Google Scholar] [CrossRef]
- Kim, J.; Joo, I.; Kim, H.; Han, Y. 18β-glycyrrhetinic acid induces immunological adjuvant activity of Th1 against Candida albicans surface mannan extract. Phytomedicine 2013, 20, 951–955. [Google Scholar] [CrossRef]
- Dong, S.; Yang, X.; Zhao, L.; Zhang, F.; Hou, Z.; Xue, P. Antibacterial activity and mechanism of action saponins from Chenopodium quinoa Willd. husks against foodborne pathogenic bacteria. Ind. Crops Prod. 2020, 149, 112350. [Google Scholar] [CrossRef]
- Zhou, T.; Deng, X.; Qiu, J. Antimicrobial activity of licochalcone E against Staphylococcus aureus and its impact on the production of staphylococcal alpha-toxin. J. Microbiol. Biotechnol. 2012, 22, 800–805. [Google Scholar] [CrossRef]
- Dai, X.-H.; Li, H.-E.; Lu, C.-J.; Wang, J.-F.; Dong, J.; Wei, J.-Y.; Zhang, Y.; Wang, X.; Tan, W.; Deng, X.-M.; et al. Liquiritigenin prevents Staphylococcus aureus-mediated lung cell injury via inhibiting the production of α-hemolysin. J. Asian Nat. Prod. Res. 2013, 15, 390–399. [Google Scholar] [CrossRef]
- Messier, C.; Grenier, D. Effect of licorice compounds licochalcone A, glabridin and glycyrrhizic acid on growth and virulence properties of Candida albicans. Mycoses 2011, 54, e801-6. [Google Scholar] [CrossRef] [PubMed]
- Wei, M.; Yu, H.; Guo, Y.; Cheng, Y.; Xie, Y.; Yao, W. Antibacterial activity of Sapindus saponins against microorganisms related to food hygiene and the synergistic action mode of Sapindoside A and B against Micrococcus luteus in vitro. Food Control 2021, 130, 108337. [Google Scholar] [CrossRef]
- Wei, M.-P.; Qiu, J.-D.; Li, L.; Xie, Y.-F.; Yu, H.; Guo, Y.-H.; Yao, W.-R. Saponin fraction from Sapindus mukorossi Gaertn as a novel cosmetic additive: Extraction, biological evaluation, analysis of anti-acne mechanism and toxicity prediction. J. Ethnopharmacol. 2021, 268, 113552. [Google Scholar] [CrossRef] [PubMed]
- Anju, V.T.; Busi, S.; Ranganathan, S.; Ampasala, D.R.; Kumar, S.; Suchiang, K.; Kumavath, R.; Dyavaiah, M. Sesamin and sesamolin rescues Caenorhabditis elegans from Pseudomonas aeruginosa infection through the attenuation of quorum sensing regulated virulence factors. Microb. Pathog. 2021, 155, 104912. [Google Scholar] [CrossRef]
- Farag, M.A.; Ali, S.E.; Hodaya, R.H.; El-Seedi, H.R.; Sultani, H.N.; Laub, A.; Eissa, T.F.; Abou-Zaid, F.O.F.; Wessjohann, L.A. Phytochemical Profiles and Antimicrobial Activities of Allium cepa Red cv. and A. sativum Subjected to Different Drying Methods: A Comparative MS-Based Metabolomics. Molecules 2017, 22, 761. [Google Scholar] [CrossRef]
- Persson, T.; Hansen, T.H.; Rasmussen, T.B.; Skindersø, M.E.; Givskov, M.; Nielsen, J. Rational design and synthesis of new quorum-sensing inhibitors derived from acylated homoserine lactones and natural products from garlic. Org. Biomol. Chem. 2005, 3, 253–262. [Google Scholar] [CrossRef]
- Birring, O.J.S.; Viloria, I.L.; Nunez, P. Anti-microbial efficacy of Allium sativum extract against Enterococcus faecalis biofilm and its penetration into the root dentin: An in vitro study. Indian J. Dent. Res. Off. Publ. Indian Soc. Dent. Res. 2015, 26, 477–482. [Google Scholar] [CrossRef]
- Bayan, L.; Koulivand, P.H.; Gorji, A. Garlic: A review of potential therapeutic effects. Avicenna J. phytomedicine 2014, 4, 1–14. [Google Scholar]
- Vadekeetil, A.; Kaur, G.; Chhibber, S.; Harjai, K. Applications of thin-layer chromatography in extraction and characterisation of ajoene from garlic bulbs. Nat. Prod. Res. 2015, 29, 768–771. [Google Scholar] [CrossRef]
- Dutta, S.; Bhattacharyya, D. Enzymatic, antimicrobial and toxicity studies of the aqueous extract of Ananas comosus (pineapple) crown leaf. J. Ethnopharmacol. 2013, 150, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Zorga, J.; Kunicka-Styczyńska, A.; Gruska, R.; Śmigielski, K. Ultrasound-Assisted Hydrodistillation of Essential Oil from Celery Seeds (Apium graveolens L.) and Its Biological and Aroma Profiles. Molecules 2020, 25, 5322. [Google Scholar] [CrossRef] [PubMed]
- Petrović, S.; Drobac, M.; Ušjak, L.; Filipović, V.; Milenković, M.; Niketić, M. Volatiles of roots of wild-growing and cultivated Armoracia macrocarpa and their antimicrobial activity, in comparison to horseradish, A. rusticana. Ind. Crops Prod. 2017, 109, 398–403. [Google Scholar] [CrossRef]
- Park, H.-W.; Choi, K.-D.; Shin, I.-S. Antimicrobial activity of isothiocyanates (ITCs) extracted from horseradish (Armoracia rusticana) root against oral microorganisms. Biocontrol Sci. 2013, 18, 163–168. [Google Scholar] [CrossRef]
- Jakobsen, T.H.; Bragason, S.K.; Phipps, R.K.; Christensen, L.D.; van Gennip, M.; Alhede, M.; Skindersoe, M.; Larsen, T.O.; Høiby, N.; Bjarnsholt, T.; et al. Food as a source for quorum sensing inhibitors: Iberin from horseradish revealed as a quorum sensing inhibitor of Pseudomonas aeruginosa. Appl. Environ. Microbiol. 2012, 78, 2410–2421. [Google Scholar] [CrossRef]
- Dekić, M.S.; Radulović, N.S.; Stojanović, N.M.; Randjelović, P.J.; Stojanović-Radić, Z.Z.; Najman, S.; Stojanović, S. Spasmolytic, antimicrobial and cytotoxic activities of 5-phenylpentyl isothiocyanate, a new glucosinolate autolysis product from horseradish (Armoracia rusticana P. Gaertn., B. Mey. & Scherb., Brassicaceae). Food Chem. 2017, 232, 329–339. [Google Scholar] [CrossRef]
- Staszowska-Karkut, M.; Materska, M. Phenolic Composition, Mineral Content, and Beneficial Bioactivities of Leaf Extracts from Black Currant (Ribes nigrum L.), Raspberry (Rubus idaeus), and Aronia (Aronia melanocarpa). Nutrients 2020, 12, 463. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Lin, J.-H.; Chou, S.-C.; Chang, S.-J.; Chung, C.-C.; Chen, Y.-S.; Chang, C.-H. Berberine-loaded targeted nanoparticles as specific Helicobacter pylori eradication therapy: In vitro and in vivo study. Nanomedicine 2015, 10, 57–71. [Google Scholar] [CrossRef]
- Guan, Y.; Chye, M.-L. A Brassica juncea chitinase with two-chitin binding domains show anti-microbial properties against phytopathogens and Gram-negative bacteria. Plant Signal. Behav. 2008, 3, 1103–1105. [Google Scholar] [CrossRef][Green Version]
- Sharma, A.; Gupta, S.; Sarethy, I.P.; Dang, S.; Gabrani, R. Green tea extract: Possible mechanism and antibacterial activity on skin pathogens. Food Chem. 2012, 135, 672–675. [Google Scholar] [CrossRef]
- Cichewicz, R.H.; Thorpe, P.A. The antimicrobial properties of chile peppers (Capsicum species) and their uses in Mayan medicine. J. Ethnopharmacol. 1996, 52, 61–70. [Google Scholar] [CrossRef]
- Vasavi, H.S.; Arun, A.B.; Rekha, P.D. Anti-quorum sensing activity of Psidium guajava L. flavonoids against Chromobacterium violaceum and Pseudomonas aeruginosa PAO1. Microbiol. Immunol. 2014, 58, 286–293. [Google Scholar] [CrossRef]
- Sarumathi, A.; Anbu, S.; Saravanan, N. Antibacterial activity of Centella asiatica (Linn.,) leaves. Int. J. Chem. Pharm. Sci. 2013, 1, 393–395. [Google Scholar]
- Papetti, A.; Mascherpa, D.; Carazzone, C.; Stauder, M.; Spratt, D.A.; Wilson, M.; Pratten, J.; Ciric, L.; Lingström, P.; Zaura, E.; et al. Identification of organic acids in Cichorium intybus inhibiting virulence-related properties of oral pathogenic bacteria. Food Chem. 2013, 138, 1706–1712. [Google Scholar] [CrossRef]
- Alva, P.P.; Suresh, S.; MP, G.; Premanath, R. Evaluation of anti-quorum sensing activity of indigenous dietary plants against Pseudomonas aeruginosa. Eur. J. Integr. Med. 2019, 30, 100931. [Google Scholar] [CrossRef]
- Gao, Z.; Zhong, W.; Chen, K.; Tang, P.; Guo, J. Chemical composition and anti-biofilm activity of essential oil from Citrus medica L. var. sarcodactylis Swingle against Listeria monocytogenes. Ind. Crops Prod. 2020, 144, 112036. [Google Scholar] [CrossRef]
- Sultana, H.S.; Ali, M.; Panda, B.P. Influence of volatile constituents of fruit peels of Citrus reticulata Blanco on clinically isolated pathogenic microorganisms under In–vitro. Asian Pac. J. Trop. Biomed. 2012, 2, S1299–S1302. [Google Scholar] [CrossRef]
- Oikeh, E.I.; Oviasogie, F.E.; Omoregie, E.S. Quantitative phytochemical analysis and antimicrobial activities of fresh and dry ethanol extracts of Citrus sinensis (L.) Osbeck (sweet Orange) peels. Clin. Phytoscience 2020, 6, 46. [Google Scholar] [CrossRef]
- Duangjai, A.; Suphrom, N.; Wungrath, J.; Ontawong, A.; Nuengchamnong, N.; Yosboonruang, A. Comparison of antioxidant, antimicrobial activities and chemical profiles of three coffee (Coffea arabica L.) pulp aqueous extracts. Integr. Med. Res. 2016, 5, 324–331. [Google Scholar] [CrossRef]
- Delaquis, P.J.; Stanich, K.; Girard, B.; Mazza, G. Antimicrobial activity of individual and mixed fractions of dill, cilantro, coriander and eucalyptus essential oils. Int. J. Food Microbiol. 2002, 74, 101–109. [Google Scholar] [CrossRef]
- Rolnik, A.; Olas, B. Vegetables from the Cucurbitaceae family and their products: Positive effect on human health. Nutrition 2020, 78, 110788. [Google Scholar] [CrossRef]
- Amin, M.Z.; IslamRity, T.; RaselUddin, M.; Rahman, M.M.; Uddin, M.J. A comparative assessment of anti-inflammatory, anti-oxidant and anti-bacterial activities of hybrid and indigenous varieties of pumpkin (Cucurbita maxima Linn.) seed oil. Biocatal. Agric. Biotechnol. 2020, 28, 101767. [Google Scholar] [CrossRef]
- Morita, A.; Tai, A.; Ito, H.; Ganeko, N.; Aizawa, S.-I. Proanthocyanidins in an astringent persimmon inhibit Salmonella pathogenicity island 1 (SPI1) secretion. J. Sci. Food Agric. 2016, 96, 1798–1802. [Google Scholar] [CrossRef]
- Liu, F.; Liu, C.; Liu, W.; Ding, Z.; Ma, H.; Seeram, N.P.; Xu, L.; Mu, Y.; Huang, X.; Li, L. New Sesquiterpenoids from Eugenia jambolana Seeds and Their Anti-microbial Activities. J. Agric. Food Chem. 2017, 65, 10214–10222. [Google Scholar] [CrossRef]
- Di Vito, M.; Gentile, M.; Mattarelli, P.; Barbanti, L.; Micheli, L.; Mazzuca, C.; Garzoli, S.; Titubante, M.; Vitali, A.; Cacaci, M.; et al. Phytocomplex Influences Antimicrobial and Health Properties of Concentrated Glycerine Macerates. Antibiotics 2020, 9, 858. [Google Scholar] [CrossRef]
- Da-Costa-Rocha, I.; Bonnlaender, B.; Sievers, H.; Pischel, I.; Heinrich, M. Hibiscus sabdariffa L.–A phytochemical and pharmacological review. Food Chem. 2014, 165, 424–443. [Google Scholar] [CrossRef]
- Otsuka, N.; Liu, M.-H.; Shiota, S.; Ogawa, W.; Kuroda, T.; Hatano, T.; Tsuchiya, T. Anti-methicillin resistant Staphylococcus aureus (MRSA) compounds isolated from Laurus nobilis. Biol. Pharm. Bull. 2008, 31, 1794–1797. [Google Scholar] [CrossRef]
- Liu, M.-H.; Otsuka, N.; Noyori, K.; Shiota, S.; Ogawa, W.; Kuroda, T.; Hatano, T.; Tsuchiya, T. Synergistic effect of kaempferol glycosides purified from Laurus nobilis and fluoroquinolones on methicillin-resistant Staphylococcus aureus. Biol. Pharm. Bull. 2009, 32, 489–492. [Google Scholar] [CrossRef]
- Rafińska, K.; Pomastowski, P.; Rudnicka, J.; Krakowska, A.; Maruśka, A.; Narkute, M.; Buszewski, B. Effect of solvent and extraction technique on composition and biological activity of Lepidium sativum extracts. Food Chem. 2019, 289, 16–25. [Google Scholar] [CrossRef]
- Gao, M.; Teplitski, M.; Robinson, J.B.; Bauer, W.D. Production of substances by Medicago truncatula that affect bacterial quorum sensing. Mol. Plant. Microbe. Interact. 2003, 16, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Keshavan, N.D.; Chowdhary, P.K.; Haines, D.C.; González, J.E. L-Canavanine made by Medicago sativa interferes with quorum sensing in Sinorhizobium meliloti. J. Bacteriol. 2005, 187, 8427–8436. [Google Scholar] [CrossRef] [PubMed]
- Lucena Filho, J.H.; Medeiros, A.C.; Pereira, J.V.; Granville-Garcia, A.F.; Costa, E.M. Antimicrobial Potential of Momordica charantia L. against Multiresistant Standard Species and Clinical Isolates. J. Contemp. Dent. Pract. 2015, 16, 854–858. [Google Scholar] [CrossRef] [PubMed]
- Jabeen, U.; Khanum, A. Isolation and characterization of potential food preservative peptide from Momordica charantia L. Arab. J. Chem. 2017, 10, S3982–S3989. [Google Scholar] [CrossRef]
- Paikra, B.K.; Dhongade, H.K.J.; Gidwani, B. Phytochemistry and Pharmacology of Moringa oleifera Lam. J. pharmacopuncture 2017, 20, 194–200. [Google Scholar] [CrossRef]
- Ningappa, M.B.; Dhananjaya, B.L.; Dinesha, R.; Harsha, R.; Srinivas, L. Potent antibacterial property of APC protein from curry leaves (Murraya koenigii L.). Food Chem. 2010, 118, 747–750. [Google Scholar] [CrossRef]
- Avetisyan, A.; Markosian, A.; Petrosyan, M.; Sahakyan, N.; Babayan, A.; Aloyan, S.; Trchounian, A. Chemical composition and some biological activities of the essential oils from basil Ocimum different cultivars. BMC Complement. Altern. Med. 2017, 17, 60. [Google Scholar] [CrossRef]
- Peng, M.; Zhao, X.; Biswas, D. Polyphenols and tri-terpenoids from Olea europaea L. in alleviation of enteric pathogen infections through limiting bacterial virulence and attenuating inflammation. J. Funct. Foods 2017, 36, 132–143. [Google Scholar] [CrossRef]
- Alvarez, M.V.; Ortega-Ramirez, L.A.; Gutierrez-Pacheco, M.M.; Bernal-Mercado, A.T.; Rodriguez-Garcia, I.; Gonzalez-Aguilar, G.A.; Ponce, A.; Moreira, M.D.R.; Roura, S.I.; Ayala-Zavala, J.F. Oregano essential oil-pectin edible films as anti-quorum sensing and food antimicrobial agents. Front. Microbiol. 2014, 5, 699. [Google Scholar] [CrossRef]
- Bharti, V.; Vasudeva, N.; Kumar, S. Anti-oxidant studies and anti-microbial effect of Origanum vulgare Linn in combination with standard antibiotics. Ayu 2014, 35, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, L.N.; Alves, F.C.B.; Andrade, B.F.M.T.; Albano, M.; Rall, V.L.M.; Fernandes, A.A.H.; Buzalaf, M.A.R.; de Lima Leite, A.; de Pontes, L.G.; Dos Santos, L.D.; et al. Proteomic analysis and antibacterial resistance mechanisms of Salmonella Enteritidis submitted to the inhibitory effect of Origanum vulgare essential oil, thymol and carvacrol. J. Proteomics 2020, 214, 103625. [Google Scholar] [CrossRef]
- Kenari, H.M.; Kordafshari, G.; Moghimi, M.; Eghbalian, F.; TaherKhani, D. Review of Pharmacological Properties and Chemical Constituents of Pastinaca sativa. J. Pharmacopunct. 2021, 24, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Linde, G.A.; Gazim, Z.C.; Cardoso, B.K.; Jorge, L.F.; Tešević, V.; Glamoćlija, J.; Soković, M.; Colauto, N.B. Antifungal and antibacterial activities of Petroselinum crispum essential oil. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef]
- Zaki, A.A.; Shaaban, M.I.; Hashish, N.E.; Amer, M.A.; Lahloub, M.-F. Assessment of anti-quorum sensing activity for some ornamental and medicinal plants native to egypt. Sci. Pharm. 2013, 81, 251–258. [Google Scholar] [CrossRef]
- Rajaei, A.; Barzegar, M.; Mobarez, A.M.; Sahari, M.A.; Esfahani, Z.H. Antioxidant, anti-microbial and antimutagenicity activities of pistachio (Pistachia vera) green hull extract. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2010, 48, 107–112. [Google Scholar] [CrossRef]
- Hu, S.; Chai, W.C.; Xu, L.; Li, S.; Jin, C.; Zhu, R.; Yang, L.; Zhang, R.; Tang, K.; Li, P.; et al. Catecholic alkaloid sulfonates and aromatic nitro compounds from Portulaca oleracea and screening of their anti-inflammatory and anti-microbial activities. Phytochemistry 2021, 181, 112587. [Google Scholar] [CrossRef]
- Afonso, S.; Oliveira, I.V.; Meyer, A.S.; Aires, A.; Saavedra, M.J.; Gonçalves, B. Phenolic Profile and Bioactive Potential of Stems and Seed Kernels of Sweet Cherry Fruit. Antioxidants 2020, 9, 1295. [Google Scholar] [CrossRef]
- Mitani, T.; Ota, K.; Inaba, N.; Kishida, K.; Koyama, H.A. Antimicrobial Activity of the Phenolic Compounds of Prunus mume against Enterobacteria. Biol. Pharm. Bull. 2018, 41, 208–212. [Google Scholar] [CrossRef]
- Li, X.; Zhang, J.; Gao, W.; Wang, H. Study on chemical composition, anti-inflammatory and anti-microbial activities of extracts from Chinese pear fruit (Pyrus bretschneideri Rehd.). Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2012, 50, 3673–3679. [Google Scholar] [CrossRef]
- Iyda, J.H.; Fernandes, Â.; Ferreira, F.D.; Alves, M.J.; Pires, T.C.S.P.; Barros, L.; Amaral, J.S.; Ferreira, I.C.F.R. Chemical composition and bioactive properties of the wild edible plant Raphanus raphanistrum L. Food Res. Int. 2019, 121, 714–722. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.; Han, S.-W.; Kim, J. Sulforaphene identified from radish (Raphanus sativus L.) seeds possesses antimicrobial properties against multidrug-resistant bacteria and methicillin-resistant Staphylococcus aureus. J. Funct. Foods 2016, 24, 131–141. [Google Scholar] [CrossRef]
- Kuo, I.-P.; Lee, P.-T.; Nan, F.-H. Rheum officinale extract promotes the innate immunity of orange-spotted grouper (Epinephelus coioides) and exerts strong bactericidal activity against six aquatic pathogens. Fish Shellfish Immunol. 2020, 102, 117–124. [Google Scholar] [CrossRef]
- Cendrowski, A.; Kraśniewska, K.; Przybył, J.L.; Zielińska, A.; Kalisz, S. Antibacterial and Antioxidant Activity of Extracts from Rose Fruits (Rosa rugosa). Molecules 2020, 25, 1365. [Google Scholar] [CrossRef]
- Akroum, S. [Anti-microbial activity of Rosmarinus officinalis and Zingiber officinale extracts on the species of the genus Candida and on Streptococcus pneumonia]. Ann. Pharm. Fr. 2021, 79, 62–69. [Google Scholar] [CrossRef]
- Wei, Q.; Bhasme, P.; Wang, Z.; Wang, L.; Wang, S.; Zeng, Y.; Wang, Y.; Ma, L.Z.; Li, Y. Chinese medicinal herb extract inhibits PQS-mediated quorum sensing system in Pseudomonas aeruginosa. J. Ethnopharmacol. 2020, 248, 112272. [Google Scholar] [CrossRef]
- Zhang, X.; Guo, Y.; Guo, L.; Jiang, H.; Ji, Q. In Vitro Evaluation of Antioxidant and Antimicrobial Activities of Melaleuca alternifolia Essential Oil. Biomed Res. Int. 2018, 2018, 2396109. [Google Scholar] [CrossRef]
- de Carvalho Bernardo, W.L.; Boriollo, M.F.G.; Tonon, C.C.; da Silva, J.J.; Cruz, F.M.; Martins, A.L.; Höfling, J.F.; Spolidorio, D.M.P. Antimicrobial effects of silver nanoparticles and extracts of Syzygium cumini flowers and seeds: Periodontal, cariogenic and opportunistic pathogens. Arch. Oral Biol. 2021, 125, 105101. [Google Scholar] [CrossRef]
- Gopu, V.; Kothandapani, S.; Shetty, P.H. Quorum quenching activity of Syzygium cumini (L.) Skeels and its anthocyanin malvidin against Klebsiella pneumoniae. Microb. Pathog. 2015, 79, 61–69. [Google Scholar] [CrossRef]
- Sirdaarta, J.; Matthews, B.; Cock, I.E. Kakadu plum fruit extracts inhibit growth of the bacterial triggers of rheumatoid arthritis: Identification of stilbene and tannin components. J. Funct. Foods 2015, 17, 610–620. [Google Scholar] [CrossRef]
- Borugă, O.; Jianu, C.; Mişcă, C.; Goleţ, I.; Gruia, A.T.; Horhat, F.G. Thymus vulgaris essential oil: Chemical composition and antimicrobial activity. J. Med. Life 2014, 7, 56–60. [Google Scholar]
- Choo, J.H.; Rukayadi, Y.; Hwang, J.-K. Inhibition of bacterial quorum sensing by vanilla extract. Lett. Appl. Microbiol. 2006, 42, 637–641. [Google Scholar] [CrossRef]
- Uchida, K.; Miura, Y.; Nagai, M.; Tominaga, M. Isothiocyanates from Wasabia japonica activate transient receptor potential ankyrin 1 channel. Chem. Senses 2012, 37, 809–818. [Google Scholar] [CrossRef]
- Kumar, L.; Chhibber, S.; Harjai, K. Structural alterations in Pseudomonas aeruginosa by zingerone contribute to enhanced susceptibility to antibiotics, serum and phagocytes. Life Sci. 2014, 117, 24–32. [Google Scholar] [CrossRef]
- Macdonald, I.J.; Dougherty, T.J. Basic principles of photodynamic therapy. J. Porphyr. Phthalocyanines 2001, 5, 105–129. [Google Scholar] [CrossRef]
- Huang, L.; Xuan, Y.; Koide, Y.; Zhiyentayev, T.; Tanaka, M.; Hamblin, M.R. Type I and Type II mechanisms of antimicrobial photodynamic therapy: An in vitro study on gram-negative and gram-positive bacteria. Lasers Surg. Med. 2012, 44, 490–499. [Google Scholar] [CrossRef]
- Alves, E.; Faustino, M.A.; Neves, M.G.; Cunha, A.; Tome, J.; Almeida, A. An insight on bacterial cellular targets of photodynamic inactivation. Future Med. Chem. 2014, 6, 141–164. [Google Scholar] [CrossRef]
- Liu, Y.; Qin, R.; Zaat, S.A.J.; Breukink, E.; Heger, M. Antibacterial photodynamic therapy: Overview of a promising approach to fight antibiotic-resistant bacterial infections. J. Clin. Transl. Res. 2015, 1, 140–167. [Google Scholar] [PubMed]
- Hamblin, M.R.; Abrahamse, H. Oxygen-Independent Antimicrobial Photoinactivation: Type III Photochemical Mechanism? Antibiotics 2020, 9, 53. [Google Scholar] [CrossRef] [PubMed]
- Daniell, M.D.; Hill, J.S. A history of photodynamic therapy. Aust. N. Z. J. Surg. 1991, 61, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Moan, J.; Peng, Q. An outline of the hundred-year history of PDT. Anticancer Res. 2003, 23, 3591–3600. [Google Scholar] [PubMed]
- Ackroyd, R.; Kelty, C.; Brown, N.; Reed, M. The history of photodetection and photodynamic therapy. Photochem. Photobiol. 2001, 74, 656–669. [Google Scholar] [CrossRef]
- Hamblin, M.R. Potentiation of antimicrobial photodynamic inactivation by inorganic salts. Expert Rev. Anti. Infect. Ther. 2017, 15, 1059–1069. [Google Scholar] [CrossRef]
- Cieplik, F.; Deng, D.; Crielaard, W.; Buchalla, W.; Hellwig, E.; Al-Ahmad, A.; Maisch, T. Antimicrobial photodynamic therapy-what we know and what we don’t. Crit. Rev. Microbiol. 2018, 44, 571–589. [Google Scholar] [CrossRef] [PubMed]
- Hamblin, M.R. Antimicrobial photodynamic inactivation: A bright new technique to kill resistant microbes. Curr. Opin. Microbiol. 2016, 33, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Nisnevitch, M.; Nakonechny, F.; Nitzan, Y. Photodynamic antimicrobial chemotherapy by liposome-encapsulated water-soluble photosensitizers. Russ. J. Bioorganic Chem. 2010, 36, 363–369. [Google Scholar] [CrossRef]
- Ilizirov, Y.; Formanovsky, A.; Mikhura, I.; Paitan, Y.; Nakonechny, F.; Nisnevitch, M. Effect of photodynamic antibacterial chemotherapy combined with antibiotics on Gram-positive and gram-negative bacteria. Molecules 2018, 23, 3152. [Google Scholar] [CrossRef]
- Wainwright, M.; Maisch, T.; Nonell, S.; Plaetzer, K.; Almeida, A.; Tegos, G.P.; Hamblin, M.R. Photoantimicrobials-are we afraid of the light? Lancet. Infect. Dis. 2017, 17, e49–e55. [Google Scholar] [CrossRef]
- Ghorbani, J.; Rahban, D.; Aghamiri, S.; Teymouri, A.; Bahador, A. Photosensitizers in antibacterial photodynamic therapy: An overview. Laser Ther. 2018, 27, 293–302. [Google Scholar] [CrossRef]
- Siewert, B.; Stuppner, H. The photoactivity of natural products–An overlooked potential of phytomedicines? Phytomedicine 2019, 60, 152985. [Google Scholar] [CrossRef]
- Jha, A.; Mohapatra, P.P.; AlHarbi, S.A.; Jahan, N. Curcumin: Not So Spicy After All. Mini Rev. Med. Chem. 2017, 17, 1425–1434. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Wu, J.; Pang, X.; Jiang, Y.; Wang, P.; Leung, A.W.; Gao, L.; Jiang, S.; Xu, C. Discovery and Development of Natural Products and their Derivatives as Photosensitizers for Photodynamic Therapy. Curr. Med. Chem. 2018, 25, 839–860. [Google Scholar] [CrossRef]
- Kotha, R.R.; Luthria, D.L. Curcumin: Biological, Pharmaceutical, Nutraceutical, and Analytical Aspects. Molecules 2019, 24, 2930. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, P.; Singh, M.; Kumari, H.; Kumari, A.; Mukhopadhyay, K. Bactericidal activity of curcumin I is associated with damaging of bacterial membrane. PLoS ONE 2015, 10, e0121313. [Google Scholar] [CrossRef] [PubMed]
- Dias, L.D.; Blanco, K.C.; Mfouo-Tynga, I.S.; Inada, N.M.; Bagnato, V.S. Curcumin as a photosensitizer: From molecular structure to recent advances in antimicrobial photodynamic therapy. J. Photochem. Photobiol. C Photochem. Rev. 2020, 45, 100384. [Google Scholar] [CrossRef]
- Trigo Gutierrez, J.K.; Zanatta, G.C.; Ortega, A.L.M.; Balastegui, M.I.C.; Sanitá, P.V.; Pavarina, A.C.; Barbugli, P.A.; de Oliveira Mima, E.G. Encapsulation of curcumin in polymeric nanoparticles for antimicrobial Photodynamic Therapy. PLoS ONE 2017, 12, e0187418. [Google Scholar] [CrossRef]
- Rocha, M.P.; Santos, M.S.; Rodrigues, P.L.F.; Araújo, T.S.D.; de Oliveira, J.M.; Rosa, L.P.; Bagnato, V.S.; da Silva, F.C. Photodynamic therapry with curcumin in the reduction of enterococcus faecalis biofilm in bone cavity: rMicrobiological and spectral fluorescense analysis. Photodiagnosis Photodyn. Ther. 2021, 33, 102084. [Google Scholar] [CrossRef]
- Santezi, C.; Reina, B.D.; de Annunzio, S.R.; Calixto, G.; Chorilli, M.; Dovigo, L.N. Photodynamic potential of curcumin in bioadhesive formulations: Optical characteristics and antimicrobial effect against biofilms. Photodiagnosis Photodyn. Ther. 2021, 35, 102416. [Google Scholar] [CrossRef] [PubMed]
- Randazzo, W.; Aznar, R.; Sánchez, G. Curcumin-Mediated Photodynamic Inactivation of Norovirus Surrogates. Food Environ. Virol. 2016, 8, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Araújo, B.P.; Silva, E.A.; Rosa, L.P.; Inada, N.M.; Iermak, I.; Romano, R.A.; Mezzacappo, N.F.; Melo, F.F.; Silva, F.C.; Rocha, M.P.; et al. Morpho-molecular mechanisms study of photodynamic therapy and curcumin larvicide action on wild mosquitoes larvae of genus Aedes. bioRxiv 2020, 200295. [Google Scholar] [CrossRef]
- Buchovec, I.; Gricajeva, A.; Kalėdienė, L.; Vitta, P. Antimicrobial Photoinactivation Approach Based on Natural Agents for Control of Bacteria Biofilms in Spacecraft. Int. J. Mol. Sci. 2020, 21, 6932. [Google Scholar] [CrossRef] [PubMed]
- Bhavya, M.L.; Umesh Hebbar, H. Efficacy of blue LED in microbial inactivation: Effect of photosensitization and process parameters. Int. J. Food Microbiol. 2019, 290, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Duval, J.; Pecher, V.; Poujol, M.; Lesellier, E. Research advances for the extraction, analysis and uses of anthraquinones: A review. Ind. Crops Prod. 2016, 94, 812–833. [Google Scholar] [CrossRef]
- Otieno, W.; Liu, C.; Ji, Y. Aloe-Emodin-Mediated Photodynamic Therapy Attenuates Sepsis-Associated Toxins in Selected Gram-Positive Bacteria In Vitro. J. Microbiol. Biotechnol. 2021, 31, 1200–1209. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Qin, M.; Liu, C.; Ma, W.; Zeng, X.; Ji, Y. Antimicrobial photodynamic therapy against multidrug-resistant Acinetobacter baumannii clinical isolates mediated by aloe-emodin: An in vitro study. Photodiagnosis Photodyn. Ther. 2020, 29, 101632. [Google Scholar] [CrossRef]
- Wang, Y.; Li, J.; Geng, S.; Wang, X.; Cui, Z.; Ma, W.; Yuan, M.; Liu, C.; Ji, Y. Aloe-emodin-mediated antimicrobial photodynamic therapy against multidrug-resistant Acinetobacter baumannii: An in vivo study. Photodiagnosis Photodyn. Ther. 2021, 34, 102311. [Google Scholar] [CrossRef]
- Yao, J.-Y.; Lin, L.-Y.; Yuan, X.-M.; Ying, W.-L.; Xu, Y.; Pan, X.-Y.; Hao, G.-J.; Shen, J.-Y.; Wu, J.-C.; Ye, T.; et al. Antifungal Activity of Rhein and Aloe-Emodin from Rheum palmatum on Fish Pathogenic Saprolegnia sp. J. World Aquac. Soc. 2017, 48, 137–144. [Google Scholar] [CrossRef]
- Ma, W.; Liu, C.; Li, J.; Hao, M.; Ji, Y.; Zeng, X. The effects of aloe emodin-mediated antimicrobial photodynamic therapy on drug-sensitive and resistant Candida albicans. Photochem. Photobiol. Sci. 2020, 19, 485–494. [Google Scholar] [CrossRef]
- Ma, W.; Zhang, M.; Cui, Z.; Wang, X.; Niu, X.; Zhu, Y.; Yao, Z.; Ye, F.; Geng, S.; Liu, C. Aloe-emodin-mediated antimicrobial photodynamic therapy against dermatophytosis caused by Trichophyton rubrum. Microb. Biotechnol. 2021. [Google Scholar] [CrossRef]
- Mulrooey, C.A.; O’Brien, E.M.; Morgan, B.J.; Kozlowski, M.C. Perylenequinones: Isolation, Synthesis, and Biological Activity. European J. Org. Chem. 2012, 2012, 3887–3904. [Google Scholar] [CrossRef]
- Borghi-Pangoni, F.B.; Junqueira, M.V.; de Souza Ferreira, S.B.; Silva, L.L.; Rabello, B.R.; de Castro, L.V.; Baesso, M.L.; Diniz, A.; Caetano, W.; Bruschi, M.L. Preparation and characterization of bioadhesive system containing hypericin for local photodynamic therapy. Photodiagnosis Photodyn. Ther. 2017, 19, 284–297. [Google Scholar] [CrossRef]
- Bruni, R.; Barreca, D.; Protti, M.; Brighenti, V.; Righetti, L.; Anceschi, L.; Mercolini, L.; Benvenuti, S.; Gattuso, G.; Pellati, F. Botanical Sources, Chemistry, Analysis, and Biological Activity of Furanocoumarins of Pharmaceutical Interest. Molecules 2019, 24, 2163. [Google Scholar] [CrossRef]
- Kitamura, N.; Kohtani, S.; Nakagaki, R. Molecular aspects of furocoumarin reactions: Photophysics, photochemistry, photobiology, and structural analysis. J. Photochem. Photobiol. C Photochem. Rev. 2005, 6, 168–185. [Google Scholar] [CrossRef]
- AYa, P. Mechanisms of photodynamic effects of furocoumarins. J. Photochem. Photobiol. B. 1991, 9, 1–33. [Google Scholar] [CrossRef]
- Towers, G.H.; Graham, E.A.; Spenser, I.D.; Abramowski, Z. Phototoxic furanoquinolines of the Rutaceae. Planta Med. 1981, 41, 136–142. [Google Scholar] [CrossRef]
- An, Y.-W.; Jin, H.-T.; Yuan, B.; Wang, J.-C.; Wang, C.; Liu, H.-Q. Research progress of berberine mediated photodynamic therapy (Review). Oncol. Lett. 2021, 21, 359. [Google Scholar] [CrossRef]
- Lee, M.M.S.; Zheng, L.; Yu, B.; Xu, W.; Kwok, R.T.K.; Lam, J.W.Y.; Xu, F.; Wang, D.; Tang, B.Z. A highly efficient and AIE-active theranostic agent from natural herbs. Mater. Chem. Front. 2019, 3, 1454–1461. [Google Scholar] [CrossRef]
- Xie, Y.-Y.; Zhang, Y.-W.; Liu, X.-Z.; Ma, X.-F.; Qin, X.-T.; Jia, S.-R.; Zhong, C. Aggregation-induced emission-active amino acid/berberine hydrogels with enhanced photodynamic antibacterial and anti-biofilm activity. Chem. Eng. J. 2021, 413, 127542. [Google Scholar] [CrossRef]
- Ma, K.; Zhe, T.; Li, F.; Zhang, Y.; Yu, M.; Li, R.; Wang, L. Sustainable films containing AIE-active berberine-based nanoparticles: A promising antibacterial food packaging. Food Hydrocoll. 2022, 123, 107147. [Google Scholar] [CrossRef]
- Sobotta, L.; Skupin-Mrugalska, P.; Piskorz, J.; Mielcarek, J. Porphyrinoid photosensitizers mediated photodynamic inactivation against bacteria. Eur. J. Med. Chem. 2019, 175, 72–106. [Google Scholar] [CrossRef]
- Leite, A.C.; Ferreira, A.M.; Morais, E.S.; Khan, I.; Freire, M.G.; Coutinho, J.A.P. Cloud Point Extraction of Chlorophylls from Spinach Leaves Using Aqueous Solutions of Nonionic Surfactants. ACS Sustain. Chem. Eng. 2018, 6, 590–599. [Google Scholar] [CrossRef]
- Garcia de Carvalho, G.; Sanchez-Puetate, J.C.; Donatoni, M.C.; Maquera Huacho, P.M.; de Souza Rastelli, A.N.; de Oliveira, K.T.; Palomari Spolidorio, D.M.; Leal Zandim-Barcelos, D. Photodynamic inactivation using a chlorin-based photosensitizer with blue or red-light irradiation against single-species biofilms related to periodontitis. Photodiagnosis Photodyn. Ther. 2020, 31, 101916. [Google Scholar] [CrossRef]
- Edwards, A.M. Structure and general properties of flavins. Methods Mol. Biol. 2014, 1146, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Hiltunen, H.-M.; Illarionov, B.; Hedtke, B.; Fischer, M.; Grimm, B. Arabidopsis RIBA proteins: Two out of three isoforms have lost their bifunctional activity in riboflavin biosynthesis. Int. J. Mol. Sci. 2012, 13, 14086–14105. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Choe, E.; Min, D.B. Kinetics for Singlet Oxygen Formation by Riboflavin Photosensitization and the Reaction between Riboflavin and Singlet Oxygen. J. Food Sci. 2004, 69, C726–C732. [Google Scholar] [CrossRef]
- Cardoso, D.R.; Libardi, S.H.; Skibsted, L.H. Riboflavin as a photosensitizer. Effects on human health and food quality. Food Funct. 2012, 3, 487–502. [Google Scholar] [CrossRef] [PubMed]
- Powers, H.J. Riboflavin (vitamin B-2) and health. Am. J. Clin. Nutr. 2003, 77, 1352–1360. [Google Scholar] [CrossRef]
- Saedisomeolia, A.; Ashoori, M. Riboflavin in Human Health: A Review of Current Evidences. Adv. Food Nutr. Res. 2018, 83, 57–81. [Google Scholar] [CrossRef]
- Suwannasom, N.; Kao, I.; Pruß, A.; Georgieva, R.; Bäumler, H. Riboflavin: The Health Benefits of a Forgotten Natural Vitamin. Int. J. Mol. Sci. 2020, 21, 950. [Google Scholar] [CrossRef]
- Chang, K.-C.; Cheng, Y.-Y.; Lai, M.-J.; Hu, A. Identification of carbonylated proteins in a bactericidal process induced by curcumin with blue light irradiation on imipenem-resistant Acinetobacter baumannii. Rapid Commun. Mass Spectrom. 2020, 34 (Suppl. 1), e8548. [Google Scholar] [CrossRef]
- Pan, H.; Wang, D.; Zhang, F. In vitro antimicrobial effect of curcumin-based photodynamic therapy on Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans. Photodiagnosis Photodyn. Ther. 2020, 32, 102055. [Google Scholar] [CrossRef]
- Najafi, S.; Khayamzadeh, M.; Paknejad, M.; Poursepanj, G.; Kharazi Fard, M.J.; Bahador, A. An In Vitro Comparison of Antimicrobial Effects of Curcumin-Based Photodynamic Therapy and Chlorhexidine, on Aggregatibacter actinomycetemcomitans. J. lasers Med. Sci. 2016, 7, 21–25. [Google Scholar] [CrossRef]
- Condat, M.; Mazeran, P.-E.; Malval, J.-P.; Lalevée, J.; Morlet-Savary, F.; Renard, E.; Langlois, V.; Abbad Andalloussi, S.; Versace, D.-L. Photoinduced curcumin derivative-coatings with antibacterial properties. RSC Adv. 2015, 5, 85214–85224. [Google Scholar] [CrossRef]
- Penha, C.B.; Bonin, E.; da Silva, A.F.; Hioka, N.; Zanqueta, É.B.; Nakamura, T.U.; de Abreu Filho, B.A.; Campanerut-Sá, P.A.Z.; Mikcha, J.M.G. Photodynamic inactivation of foodborne and food spoilage bacteria by curcumin. LWT-Food Sci. Technol. 2017, 76, 198–202. [Google Scholar] [CrossRef]
- de Oliveira, E.F.; Tosati, J.V.; Tikekar, R.V.; Monteiro, A.R.; Nitin, N. Antimicrobial activity of curcumin in combination with light against Escherichia coli O157:H7 and Listeria innocua: Applications for fresh produce sanitation. Postharvest Biol. Technol. 2018, 137, 86–94. [Google Scholar] [CrossRef]
- Tao, R.; Zhang, F.; Tang, Q.-J.; Xu, C.-S.; Ni, Z.-J.; Meng, X.-H. Effects of curcumin-based photodynamic treatment on the storage quality of fresh-cut apples. Food Chem. 2019, 274, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Le, T.D.; Phasupan, P.; Nguyen, L.T. Antimicrobial photodynamic efficacy of selected natural photosensitizers against food pathogens: Impacts and interrelationship of process parameters. Photodiagnosis Photodyn. Ther. 2020, 32, 102024. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Wu, J.; Li, Z.; Zhang, X.; Lu, N.; Xue, C.; Leung, A.W.; Xu, C.; Tang, Q. Curcumin-mediated photodynamic inactivation (PDI) against DH5α contaminated in oysters and cellular toxicological evaluation of PDI-treated oysters. Photodiagnosis Photodyn. Ther. 2019, 26, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.P.; Ruela, A.L.M.; Rosa, L.P.; Santos, G.P.O.; Rosa, F.C.S. Antimicrobial photodynamic therapy in dentistry using an oil-in-water microemulsion with curcumin as a mouthwash. Photodiagnosis Photodyn. Ther. 2020, 32, 101962. [Google Scholar] [CrossRef]
- Pileggi, G.; Wataha, J.C.; Girard, M.; Grad, I.; Schrenzel, J.; Lange, N.; Bouillaguet, S. Blue light-mediated inactivation of Enterococcus faecalis in vitro. Photodiagnosis Photodyn. Ther. 2013, 10, 134–140. [Google Scholar] [CrossRef]
- da Frota, M.F.; Guerreiro-Tanomaru, J.M.; Tanomaru-Filho, M.; Bagnato, V.S.; Espir, C.G.; Berbert, F.L.C.V. Photodynamic therapy in root canals contaminated with Enterococcus faecalis using curcumin as photosensitizer. Lasers Med. Sci. 2015, 30, 1867–1872. [Google Scholar] [CrossRef]
- Neelakantan, P.; Cheng, C.Q.; Ravichandran, V.; Mao, T.; Sriraman, P.; Sridharan, S.; Subbarao, C.; Sharma, S.; Kishen, A. Photoactivation of curcumin and sodium hypochlorite to enhance antibiofilm efficacy in root canal dentin. Photodiagnosis Photodyn. Ther. 2015, 12, 108–114. [Google Scholar] [CrossRef] [PubMed]
- de Annunzio, S.R.; de Freitas, L.M.; Blanco, A.L.; da Costa, M.M.; Carmona-Vargas, C.C.; de Oliveira, K.T.; Fontana, C.R. Susceptibility of Enterococcus faecalis and Propionibacterium acnes to antimicrobial photodynamic therapy. J. Photochem. Photobiol. B Biol. 2018, 178, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Pourhajibagher, M.; Kazemian, H.; Chiniforush, N.; Hosseini, N.; Pourakbari, B.; Azizollahi, A.; Rezaei, F.; Bahador, A. Exploring different photosensitizers to optimize elimination of planktonic and biofilm forms of Enterococcus faecalis from infected root canal during antimicrobial photodynamic therapy. Photodiagnosis Photodyn. Ther. 2018, 24, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Cusicanqui Méndez, D.A.; Cardenas Cuéllar, M.R.; Feliz Pedrinha, V.; Velásquez Espedilla, E.G.; Bombarda de Andrade, F.; de Almeida Rodrigues, P.; Cruvinel, T. Effects of curcumin-mediated antimicrobial photodynamic therapy associated to different chelators against Enterococcus faecalis biofilms. Photodiagnosis Photodyn. Ther. 2021, 35, 102464. [Google Scholar] [CrossRef]
- Ghorbanzadeh, R.; Assadian, H.; Chiniforush, N.; Parker, S.; Pourakbari, B.; Ehsani, B.; Alikhani, M.Y.; Bahador, A. Modulation of virulence in Enterococcus faecalis cells surviving antimicrobial photodynamic inactivation with reduced graphene oxide-curcumin: An ex vivo biofilm model. Photodiagnosis Photodyn. Ther. 2020, 29, 101643. [Google Scholar] [CrossRef]
- Vetvicka, V.; Vetvickova, J.; Fernandez-Botran, R. Effects of curcumin on Helicobacter pylori infection. Ann. Transl. Med. 2016, 4, 479. [Google Scholar] [CrossRef]
- Bonifácio, D.; Martins, C.; David, B.; Lemos, C.; Neves, M.G.P.M.S.; Almeida, A.; Pinto, D.C.G.A.; Faustino, M.A.F.; Cunha, Â. Photodynamic inactivation of Listeria innocua biofilms with food-grade photosensitizers: A curcumin-rich extract of Curcuma longa vs commercial curcumin. J. Appl. Microbiol. 2018, 125, 282–294. [Google Scholar] [CrossRef]
- Gao, J.; Matthews, K.R. Effects of the photosensitizer curcumin in inactivating foodborne pathogens on chicken skin. Food Control 2020, 109, 106959. [Google Scholar] [CrossRef]
- Huang, J.; Chen, B.; Li, H.; Zeng, Q.-H.; Wang, J.J.; Liu, H.; Pan, Y.; Zhao, Y. Enhanced antibacterial and antibiofilm functions of the curcumin-mediated photodynamic inactivation against Listeria monocytogenes. Food Control 2020, 108, 106886. [Google Scholar] [CrossRef]
- Yang, M.-Y.; Chang, K.-C.; Chen, L.-Y.; Hu, A. Low-dose blue light irradiation enhances the antimicrobial activities of curcumin against Propionibacterium acnes. J. Photochem. Photobiol. B Biol. 2018, 189, 21–28. [Google Scholar] [CrossRef]
- Gong, C.; Li, Y.; Gao, R.; Xiao, F.; Zhou, X.; Wang, H.; Xu, H.; Wang, R.; Huang, P.; Zhao, Y. Inactivation of specific spoilage organism (Pseudomonas) of sturgeon by curcumin-mediated photodynamic inactivation. Photodiagnosis Photodyn. Ther. 2020, 31, 101827. [Google Scholar] [CrossRef]
- Mirzahosseinipour, M.; Khorsandi, K.; Hosseinzadeh, R.; Ghazaeian, M.; Shahidi, F.K. Antimicrobial photodynamic and wound healing activity of curcumin encapsulated in silica nanoparticles. Photodiagnosis Photodyn. Ther. 2020, 29, 101639. [Google Scholar] [CrossRef]
- Dos Santos, R.F.; Campos, B.S.; Francisco de Assis Filho, M.G.; de Oliveira Moraes, J.; Albuquerque, A.L.I.; da Silva, M.C.D.; Dos Santos, P.V.; de Araujo, M.T. Photodynamic inactivation of S. aureus with a water-soluble curcumin salt and an application to cheese decontamination. Photochem. Photobiol. Sci. Off. J. Eur. Photochem. Assoc. Eur. Soc. Photobiol. 2019, 18, 2707–2716. [Google Scholar] [CrossRef]
- Freitas, M.A.; Pereira, A.H.; Pinto, J.G.; Casas, A.; Ferreira-Strixino, J. Bacterial viability after antimicrobial photodynamic therapy with curcumin on multiresistant Staphylococcus aureus. Future Microbiol. 2019, 14, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Paolillo, F.R.; Rodrigues, P.G.S.; Bagnato, V.S.; Alves, F.; Pires, L.; Corazza, A.V. The effect of combined curcumin-mediated photodynamic therapy and artificial skin on Staphylococcus aureus-infected wounds in rats. Lasers Med. Sci. 2021, 36, 1219–1226. [Google Scholar] [CrossRef] [PubMed]
- Soares, J.M.; Inada, N.M.; Bagnato, V.S.; Blanco, K.C. Evolution of surviving Streptoccocus pyogenes from pharyngotonsillitis patients submit to multiple cycles of antimicrobial photodynamic therapy. J. Photochem. Photobiol. B Biol. 2020, 210, 111985. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Song, Z.; Zhi, X.; Du, B. Photoinduced Antimicrobial Activity of Curcumin-Containing Coatings: Molecular Interaction, Stability and Potential Application in Food Decontamination. ACS Omega 2020, 5, 31044–31054. [Google Scholar] [CrossRef]
- Ribeiro, A.P.D.; Pavarina, A.C.; Dovigo, L.N.; Brunetti, I.L.; Bagnato, V.S.; Vergani, C.E.; Costa, C.A. de S. Phototoxic effect of curcumin on methicillin-resistant Staphylococcus aureus and L929 fibroblasts. Lasers Med. Sci. 2013, 28, 391–398. [Google Scholar] [CrossRef]
- Jiang, Y.; Leung, A.W.; Hua, H.; Rao, X.; Xu, C. Photodynamic action of LED-activated curcumin against staphylococcus aureus involving intracellular ROS increase and membrane damage. Int. J. Photoenergy 2014, 2014, 9–11. [Google Scholar] [CrossRef]
- Almeida, P.P.; Pereira, Í.S.; Rodrigues, K.B.; Leal, L.S.; Marques, A.S.; Rosa, L.P.; da Silva, F.C.; da Silva, R.A.A. Photodynamic therapy controls of Staphylococcus aureus intradermal infection in mice. Lasers Med. Sci. 2017, 32, 1337–1342. [Google Scholar] [CrossRef] [PubMed]
- Araújo, T.S.D.; Rodrigues, P.L.F.; Santos, M.S.; de Oliveira, J.M.; Rosa, L.P.; Bagnato, V.S.; Blanco, K.C.; da Silva, F.C. Reduced methicillin-resistant Staphylococcus aureus biofilm formation in bone cavities by photodynamic therapy. Photodiagnosis Photodyn. Ther. 2018, 21, 219–223. [Google Scholar] [CrossRef]
- Tonon, C.C.; Paschoal, M.A.; Correia, M.; Spolidório, D.M.P.; Bagnato, V.S.; Giusti, J.S.M.; Santos-Pinto, L. Comparative effects of photodynamic therapy mediated by curcumin on standard and clinical isolate of Streptococcus mutans. J. Contemp. Dent. Pract. 2015, 16, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Paschoal, M.A.; Tonon, C.C.; Spolidório, D.M.P.; Bagnato, V.S.; Giusti, J.S.M.; Santos-Pinto, L. Photodynamic potential of curcumin and blue LED against Streptococcus mutans in a planktonic culture. Photodiagnosis Photodyn. Ther. 2013, 10, 313–319. [Google Scholar] [CrossRef]
- Paschoal, M.A.; Lin, M.; Santos-Pinto, L.; Duarte, S. Photodynamic antimicrobial chemotherapy on Streptococcus mutans using curcumin and toluidine blue activated by a novel LED device. Lasers Med. Sci. 2015, 30, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Paschoal, M.A.; Santos-Pinto, L.; Lin, M.; Duarte, S. Streptococcus mutans photoinactivation by combination of short exposure of a broad-spectrum visible light and low concentrations of photosensitizers. Photomed. Laser Surg. 2014, 32, 175–180. [Google Scholar] [CrossRef]
- Soria-Lozano, P.; Gilaberte, Y.; Paz-Cristobal, M.P.; Pérez-Artiaga, L.; Lampaya-Pérez, V.; Aporta, J.; Pérez-Laguna, V.; García-Luque, I.; Revillo, M.J.; Rezusta, A. In vitro effect photodynamic therapy with differents photosensitizers on cariogenic microorganisms. BMC Microbiol. 2015, 15, 187. [Google Scholar] [CrossRef]
- Manoli, E.; Samara, C. The removal of Polycyclic Aromatic Hydrocarbons in the wastewater treatment process: Experimental calculations and model predictions. Environ. Pollut. 2008, 151, 477–485. [Google Scholar] [CrossRef]
- Araújo, N.C.; Fontana, C.R.; Bagnato, V.S.; Gerbi, M.E.M. Photodynamic antimicrobial therapy of curcumin in biofilms and carious dentine. Lasers Med. Sci. 2014, 29, 629–635. [Google Scholar] [CrossRef]
- Araújo, N.C.; de Menezes, R.F.; Carneiro, V.S.M.; Dos Santos-Neto, A.P.; Fontana, C.R.; Bagnato, V.S.; Harvey, C.M.; Gerbi, M.E.M. Photodynamic Inactivation of Cariogenic Pathogens Using Curcumin as Photosensitizer. Photomed. Laser Surg. 2017, 35, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-J.; Kang, S.-M.; Jeong, S.-H.; Chung, K.-H.; Kim, B.-I. Antibacterial photodynamic therapy with curcumin and Curcuma xanthorrhiza extract against Streptococcus mutans. Photodiagnosis Photodyn. Ther. 2017, 20, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Jia, Y.; Li, W.; Zhang, M. Antimicrobial photodynamic inactivation with curcumin against Staphylococcus saprophyticus, in vitro and on fresh dough sheet. LWT 2021, 147, 111567. [Google Scholar] [CrossRef]
- Wu, J.; Mou, H.; Xue, C.; Leung, A.W.; Xu, C.; Tang, Q.-J. Photodynamic effect of curcumin on Vibrio parahaemolyticus. Photodiagnosis Photodyn. Ther. 2016, 15, 34–39. [Google Scholar] [CrossRef]
- Chen, B.; Huang, J.; Li, H.; Zeng, Q.-H.; Wang, J.J.; Liu, H.; Pan, Y.; Zhao, Y. Eradication of planktonic Vibrio parahaemolyticus and its sessile biofilm by curcumin-mediated photodynamic inactivation. Food Control 2020, 113, 107181. [Google Scholar] [CrossRef]
- Bulit, F.; Grad, I.; Manoil, D.; Simon, S.; Wataha, J.C.; Filieri, A.; Feki, A.; Schrenzel, J.; Lange, N.; Bouillaguet, S. Antimicrobial activity and cytotoxicity of 3 photosensitizers activated with blue light. J. Endod. 2014, 40, 427–431. [Google Scholar] [CrossRef]
- Ricci Donato, H.A.; Pratavieira, S.; Grecco, C.; Brugnera-Júnior, A.; Bagnato, V.S.; Kurachi, C. Clinical Comparison of Two Photosensitizers for Oral Cavity Decontamination. Photomed. Laser Surg. 2017, 35, 105–110. [Google Scholar] [CrossRef]
- Cusicanqui Méndez, D.A.; Gutierres, E.; José Dionisio, E.; Afonso Rabelo Buzalaf, M.; Cardoso Oliveira, R.; Andrade Moreira Machado, M.A.; Cruvinel, T. Curcumin-mediated antimicrobial photodynamic therapy reduces the viability and vitality of infected dentin caries microcosms. Photodiagnosis Photodyn. Ther. 2018, 24, 102–108. [Google Scholar] [CrossRef]
- Méndez, D.A.C.; Rizzato, V.L.; Lamarque, G.C.C.; Dionisio, E.J.; Buzalaf, M.A.R.; Rios, D.; Machado, M.A.A.M.; Cruvinel, T. Could a chelant improve the effect of curcumin-mediated photodynamic antimicrobial chemotherapy against dental intact biofilms? Lasers Med. Sci. 2019, 34, 1185–1192. [Google Scholar] [CrossRef]
- Cusicanqui Méndez, D.A.; Gutierrez, E.; Campos Chaves Lamarque, G.; Lopes Rizzato, V.; Afonso Rabelo Buzalaf, M.; Andrade Moreira Machado, M.A.; Cruvinel, T. The effectiveness of curcumin-mediated antimicrobial photodynamic therapy depends on pre-irradiation and biofilm growth times. Photodiagnosis Photodyn. Ther. 2019, 27, 474–480. [Google Scholar] [CrossRef]
- Oda, D.F.; Duarte, M.A.H.; Andrade, F.B.; Moriyama, L.T.; Bagnato, V.S.; de Moraes, I.G. Antimicrobial action of photodynamic therapy in root canals using LED curing light, curcumin and carbopol gel. Int. Endod. J. 2019, 52, 1010–1019. [Google Scholar] [CrossRef]
- Dou, F.; Huang, K.; Nitin, N. Targeted Photodynamic Treatment of Bacterial Biofilms Using Curcumin Encapsulated in Cells and Cell Wall Particles. ACS Appl. Bio Mater. 2021, 4, 514–522. [Google Scholar] [CrossRef]
- Dovigo, L.N.; Pavarina, A.C.; Ribeiro, A.P.D.; Brunetti, I.L.; de S. Costa, C.A.; Jacomassi, D.P.; Bagnato, V.S.; Kurachi, C. Investigation of the photodynamic effects of curcumin against Candida albicans. Photochem. Photobiol. 2011, 87, 895–903. [Google Scholar] [CrossRef]
- Jordão, C.C.; Viana de Sousa, T.; Inêz Klein, M.; Mendonça Dias, L.; Pavarina, A.C.; Carmello, J.C. Antimicrobial photodynamic therapy reduces gene expression of Candida albicans in biofilms. Photodiagnosis Photodyn. Ther. 2020, 31, 101825. [Google Scholar] [CrossRef]
- Chukwujekwu, J.C.; Coombes, P.H.; Mulholland, D.A.; van Staden, J. Emodin, an antibacterial anthraquinone from the roots of Cassia occidentalis. South African J. Bot. 2006, 72, 295–297. [Google Scholar] [CrossRef]
- Azelmat, J.; Larente, J.F.; Grenier, D. The anthraquinone rhein exhibits synergistic antibacterial activity in association with metronidazole or natural compounds and attenuates virulence gene expression in Porphyromonas gingivalis. Arch. Oral Biol. 2015, 60, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Aponiene, K.; Paskeviciute, E.; Reklaitis, I.; Luksiene, Z. Reduction of microbial contamination of fruits and vegetables by hypericin-based photosensitization: Comparison with other emerging antimicrobial treatments. J. Food Eng. 2015, 144, 29–35. [Google Scholar] [CrossRef]
- Yow, C.M.N.; Tang, H.M.; Chu, E.S.M.; Huang, Z. Hypericin-mediated photodynamic antimicrobial effect on clinically isolated pathogens. Photochem. Photobiol. 2012, 88, 626–632. [Google Scholar] [CrossRef] [PubMed]
- de Melo, W. de C.M.A.; Lee, A.N.; Perussi, J.R.; Hamblin, M.R. Electroporation enhances antimicrobial photodynamic therapy mediated by the hydrophobic photosensitizer, hypericin. Photodiagnosis Photodyn. Ther. 2013, 10, 647–650. [Google Scholar] [CrossRef]
- Kashef, N.; Borghei, Y.S.; Djavid, G.E. Photodynamic effect of hypericin on the microorganisms and primary human fibroblasts. Photodiagnosis Photodyn. Ther. 2013, 10, 150–155. [Google Scholar] [CrossRef]
- Barroso, R.A.; Navarro, R.; Tim, C.R.; de Paula Ramos, L.; de Oliveira, L.D.; Araki, Â.T.; Fernandes, K.G.C.; Macedo, D.; Assis, L. Antimicrobial photodynamic therapy against Propionibacterium acnes biofilms using hypericin (Hypericum perforatum) photosensitizer: In vitro study. Lasers Med. Sci. 2021, 36, 1235–1240. [Google Scholar] [CrossRef]
- García, I.; Ballesta, S.; Gilaberte, Y.; Rezusta, A.; Pascual, Á. Antimicrobial photodynamic activity of hypericin against methicillin-susceptible and resistant Staphylococcus aureus biofilms. Future Microbiol. 2015, 10, 347–356. [Google Scholar] [CrossRef]
- Kashef, N.; Karami, S.; Djavid, G.E. Phototoxic effect of hypericin alone and in combination with acetylcysteine on Staphylococcus aureus biofilms. Photodiagnosis Photodyn. Ther. 2015, 12, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Plenagl, N.; Seitz, B.S.; Duse, L.; Pinnapireddy, S.R.; Jedelska, J.; Brüßler, J.; Bakowsky, U. Hypericin inclusion complexes encapsulated in liposomes for antimicrobial photodynamic therapy. Int. J. Pharm. 2019, 570, 118666. [Google Scholar] [CrossRef] [PubMed]
- Rezusta, A.; López-Chicón, P.; Paz-Cristobal, M.P.; Alemany-Ribes, M.; Royo-Díez, D.; Agut, M.; Semino, C.; Nonell, S.; Revillo, M.J.; Aspiroz, C.; et al. In vitro fungicidal photodynamic effect of hypericin on Candida species. Photochem. Photobiol. 2012, 88, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Sytar, O.; Kotta, K.; Valasiadis, D.; Kosyan, A.; Brestic, M.; Koidou, V.; Papadopoulou, E.; Kroustalaki, M.; Emmanouilidou, C.; Pashalidis, A.; et al. The Effects of Photosensitizing Dyes Fagopyrin and Hypericin on Planktonic Growth and Multicellular Life in Budding Yeast. Molecules 2021, 26, 4708. [Google Scholar] [CrossRef] [PubMed]
- Sytar, O.; Švedienė, J.; Ložienė, K.; Paškevičius, A.; Kosyan, A.; Taran, N. Antifungal properties of hypericin, hypericin tetrasulphonic acid and fagopyrin on pathogenic fungi and spoilage yeasts. Pharm. Biol. 2016, 54, 3121–3125. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, S.; Lee, K.; Kim, R.H.; Hwang, K.T. Antibacterial Photodynamic Inactivation of Fagopyrin F from Tartary Buckwheat (Fagopyrum tataricum) Flower against Streptococcus mutans and Its Biofilm. Int. J. Mol. Sci. 2021, 22, 6205. [Google Scholar] [CrossRef]
- Oginsky, E.L.; Green, G.S.; Griffith, D.G.; Fowlks, W.L. Lethal photosensitization of bacteria with 8-methoxypsoralen to long wave length ultraviolet radiation. J. Bacteriol. 1959, 78, 821–833. [Google Scholar] [CrossRef]
- Bianchi, L.; Melli, R.; Pizzala, R.; Stivala, L.A.; Rehak, L.; Quarta, S.; Vannini, V. Effects of beta-carotene and alpha-tocopherol on photogenotoxicity induced by 8-methoxypsoralen: The role of oxygen. Mutat. Res. 1996, 369, 183–194. [Google Scholar] [CrossRef]
- Park, J.-H.; Moon, Y.-H.; Bang, I.-S.; Kim, Y.-C.; Kim, S.-A.; Ahn, S.-G.; Yoon, J.-H. Antimicrobial effect of photodynamic therapy using a highly pure chlorin e6. Lasers Med. Sci. 2010, 25, 705–710. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.-M.; Lee, H.-S.; Jeong, D.; Oh, H.-K.; Ra, K.-H.; Lee, M.-Y. Antimicrobial photodynamic therapy using chlorin e6 with halogen light for acne bacteria-induced inflammation. Life Sci. 2015, 124, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-H.; Ahn, M.-Y.; Kim, Y.-C.; Kim, S.-A.; Moon, Y.-H.; Ahn, S.-G.; Yoon, J.-H. In vitro and in vivo antimicrobial effect of photodynamic therapy using a highly pure chlorin e6 against Staphylococcus aureus Xen29. Biol. Pharm. Bull. 2012, 35, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Nie, M.; Deng, D.M.; Wu, Y.; de Oliveira, K.T.; Bagnato, V.S.; Crielaard, W.; Rastelli, A.N. de S. Photodynamic inactivation mediated by methylene blue or chlorin e6 against Streptococcus mutans biofilm. Photodiagnosis Photodyn. Ther. 2020, 31, 101817. [Google Scholar] [CrossRef] [PubMed]
- Luke-Marshall, N.R.; Hansen, L.A.; Shafirstein, G.; Campagnari, A.A. Antimicrobial Photodynamic Therapy with Chlorin e6 Is Bactericidal against Biofilms of the Primary Human Otopathogens. mSphere 2020, 5, e00492-20. [Google Scholar] [CrossRef]
- Eichner, A.; Gollmer, A.; Späth, A.; Bäumler, W.; Regensburger, J.; König, B.; Maisch, T. Fast and effective inactivation of Bacillus atrophaeus endospores using light-activated derivatives of vitamin B2. Photochem. Photobiol. Sci. Off. J. Eur. Photochem. Assoc. Eur. Soc. Photobiol. 2015, 14, 387–396. [Google Scholar] [CrossRef]
- Khan, S.; Rayis, M.; Rizvi, A.; Alam, M.M.; Rizvi, M.; Naseem, I. ROS mediated antibacterial activity of photoilluminated riboflavin: A photodynamic mechanism against nosocomial infections. Toxicol. Rep. 2019, 6, 136–142. [Google Scholar] [CrossRef]
- Banerjee, S.; Ghosh, D.; Vishakha, K.; Das, S.; Mondal, S.; Ganguli, A. Photodynamic antimicrobial chemotherapy (PACT) using riboflavin inhibits the mono and dual species biofilm produced by antibiotic resistant Staphylococcus aureus and Escherichia coli. Photodiagnosis Photodyn. Ther. 2020, 32, 102002. [Google Scholar] [CrossRef]
- Kim, M.-J.; Da Jeong, M.; Zheng, Q.; Yuk, H.-G. Antimicrobial activity of 405 nm light-emitting diode (LED) in the presence of riboflavin against Listeria monocytogenes on the surface of smoked salmon. Food Sci. Biotechnol. 2021, 30, 609–618. [Google Scholar] [CrossRef]
- Su, L.; Huang, J.; Li, H.; Pan, Y.; Zhu, B.; Zhao, Y.; Liu, H. Chitosan-riboflavin composite film based on photodynamic inactivation technology for antibacterial food packaging. Int. J. Biol. Macromol. 2021, 172, 231–240. [Google Scholar] [CrossRef]
- Kamran, M.A.; Qasim, M.; Udeabor, S.E.; Hameed, M.S.; Mannakandath, M.L.; Alshahrani, I. Impact of riboflavin mediated photodynamic disinfection around fixed orthodontic system infected with oral bacteria. Photodiagnosis Photodyn. Ther. 2021, 34, 102232. [Google Scholar] [CrossRef]
- Li, H.; Tan, L.; Chen, B.; Huang, J.; Zeng, Q.; Liu, H.; Zhao, Y.; Wang, J.J. Antibacterial potency of riboflavin-mediated photodynamic inactivation against Salmonella and its influences on tuna quality. LWT 2021, 146, 111462. [Google Scholar] [CrossRef]
- Alshehri, A.H. Mechanical and antimicrobial effects of riboflavin-mediated photosensitization of in vitro C. albicans formed on polymethyl methacrylate resin. Photodiagnosis Photodyn. Ther. 2021, 36, 102488. [Google Scholar] [CrossRef] [PubMed]
- Dhiman, R.; Kumar, N. Efficacy of Plant Antimicrobials as Preservative in Food. Intech 2019, 13. [Google Scholar]
- do Prado-Silva, L.; Brancini, G.T.P.; Braga, G.Ú.L.; Liao, X.; Ding, T.; Sant’Ana, A.S. Antimicrobial photodynamic treatment (aPDT) as an innovative technology to control spoilage and pathogenic microorganisms in agri-food products: An updated review. Food Control 2022, 132, 108527. [Google Scholar] [CrossRef]
- da Silveira, S.M.; Luciano, F.B.; Fronza, N.; Cunha, A.; Scheuermann, G.N.; Vieira, C.R.W. Chemical composition and antibacterial activity of Laurus nobilis essential oil towards foodborne pathogens and its application in fresh Tuscan sausage stored at 7 C. LWT-Food Sci. Technol. 2014, 59, 86–93. [Google Scholar] [CrossRef]
- Shi, M.; Chen, L.; Wang, Y.; Wang, W.; Yan, S. Effect of low-frequency pulsed ultrasound on drug delivery, antibacterial efficacy, and bone cement degradation in vancomycin-loaded calcium phosphate cement. Med. Sci. Monit. 2018, 24, 797–802. [Google Scholar] [CrossRef]
- Hernández-Ochoa, L.; Aguirre-Prieto, Y.B.; Nevárez-Moorillón, G.V.; Gutierrez-Mendez, N.; Salas-Muñoz, E. Use of essential oils and extracts from spices in meat protection. J. Food Sci. Technol. 2014, 51, 957–963. [Google Scholar] [CrossRef][Green Version]
- Kedia, A.; Prakash, B.; Mishra, P.K.; Dubey, N.K. Antifungal and antiaflatoxigenic properties of Cuminum cyminum (L.) seed essential oil and its efficacy as a preservative in stored commodities. Int. J. Food Microbiol. 2014, 168–169, 1–7. [Google Scholar] [CrossRef]
- Govaris, A.; Solomakos, N.; Pexara, A.; Chatzopoulou, P.S. The antimicrobial effect of oregano essential oil, nisin and their combination against Salmonella Enteritidis in minced sheep meat during refrigerated storage. Int. J. Food Microbiol. 2010, 137, 175–180. [Google Scholar] [CrossRef]
- El Moussaoui, N.; El Ouardy Khay, N.A.; Amajoud, N.; Boujida, N.; Charfi, S.; Senhaji, N.; Abrini, J. Effect of Origanum elongatum essential oil and heating on Pomegranate Juice Quality. Int. J. Curr. Microbiol. Appl. Sci. 2016, 5, 1–8. [Google Scholar] [CrossRef]
- Tserennadmid, R.; Takó, M.; Galgóczy, L.; Papp, T.; Vágvölgyi, C.; Gerő, L.; Krisch, J. Antibacterial effect of essential oils and interaction with food components. Open Life Sci. 2010, 5, 641–648. [Google Scholar] [CrossRef]
- Solomakos, N.; Govaris, A.; Koidis, P.; Botsoglou, N. The antimicrobial effect of thyme essential oil, nisin, and their combination against Listeria monocytogenes in minced beef during refrigerated storage. Food Microbiol. 2008, 25, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Selim, S. Antimicrobial activity of essential oils against vancomycin-resistant enterococci (vre) and Escherichia coli o157:h7 in feta soft cheese and minced beef meat. Brazilian J. Microbiol. Publ. Brazilian Soc. Microbiol. 2011, 42, 187–196. [Google Scholar] [CrossRef]
- Tajik, H.; Aminzare, M.; Mounesi Raad, T.; Hashemi, M.; Hassanzad Azar, H.; Raeisi, M.; Naghili, H. Effect of Zataria multiflora Boiss Essential Oil and Grape Seed Extract on the Shelf Life of Raw Buffalo Patty and Fate of Inoculated Listeria monocytogenes. J. Food Process. Preserv. 2015, 39, 3005–3013. [Google Scholar] [CrossRef]
- Thery, T.; Arendt, E.K. Antifungal activity of synthetic cowpea defensin Cp-thionin II and its application in dough. Food Microbiol. 2018, 73, 111–121. [Google Scholar] [CrossRef] [PubMed]
- López-Solanilla, E.; González-Zorn, B.; Novella, S.; Vázquez-Boland, J.A.; Rodríguez-Palenzuela, P. Susceptibility of Listeria monocytogenes to antimicrobial peptides. FEMS Microbiol. Lett. 2003, 226, 101–105. [Google Scholar] [CrossRef]
- Shwaiki, L.N.; Arendt, E.K.; Lynch, K.M. Study on the characterisation and application of synthetic peptide Snakin-1 derived from potato tubers–Action against food spoilage yeast. Food Control 2020, 118, 107362. [Google Scholar] [CrossRef]
- Hintz, T.; Matthews, K.K.; Di, R. The Use of Plant Antimicrobial Compounds for Food Preservation. Biomed Res. Int. 2015, 2015, 246264. [Google Scholar] [CrossRef]
- Tshabalala, R.; Kabelinde, A.; Kaptchouang Tchatchouang, C.-D.; Ateba, C.N.; Manganyi, M.C. Effect of Clove (Syzygium aromaticum) spice as microbial inhibitor of resistant bacteria and Organoleptic Quality of meat. Saudi J. Biol. Sci. 2021, 28, 3855–3863. [Google Scholar] [CrossRef]
- Temba, B.A.; Fletcher, M.T.; Fox, G.P.; Harvey, J.; Okoth, S.A.; Sultanbawa, Y. Curcumin-based photosensitization inactivates Aspergillus flavus and reduces aflatoxin B1 in maize kernels. Food Microbiol. 2019, 82, 82–88. [Google Scholar] [CrossRef]
- Lin, Y.; Hu, J.; Li, S.; Hamzah, S.S.; Jiang, H.; Zhou, A.; Zeng, S.; Lin, S. Curcumin-based photodynamic sterilization for preservation of fresh-cut hami melon. Molecules 2019, 24, 2374. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.; Wolf-Hall, C.; Hall, C.A. 3rd Application of raisin extracts as preservatives in liquid bread and bread systems. J. Food Sci. 2009, 74, M177–M184. [Google Scholar] [CrossRef] [PubMed]
- Bhavya, M.L.; Hebbar, H.U. Sono-photodynamic inactivation of Escherichia coli and Staphylococcus aureus in orange juice. Ultrason. Sonochem. 2019, 57, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ip, M.; Leung, A.W.; Yang, Z.; Wang, P.; Zhang, B.; Ip, S.; Xu, C. Sonodynamic action of curcumin on foodborne bacteria Bacillus cereus and Escherichia coli. Ultrasonics 2015, 62, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ip, M.; Leung, A.W.; Xu, C. Sonodynamic inactivation of methicillin-resistant Staphylococcus aureus in planktonic condition by curcumin under ultrasound sonication. Ultrasonics 2014, 54, 2109–2114. [Google Scholar] [CrossRef]
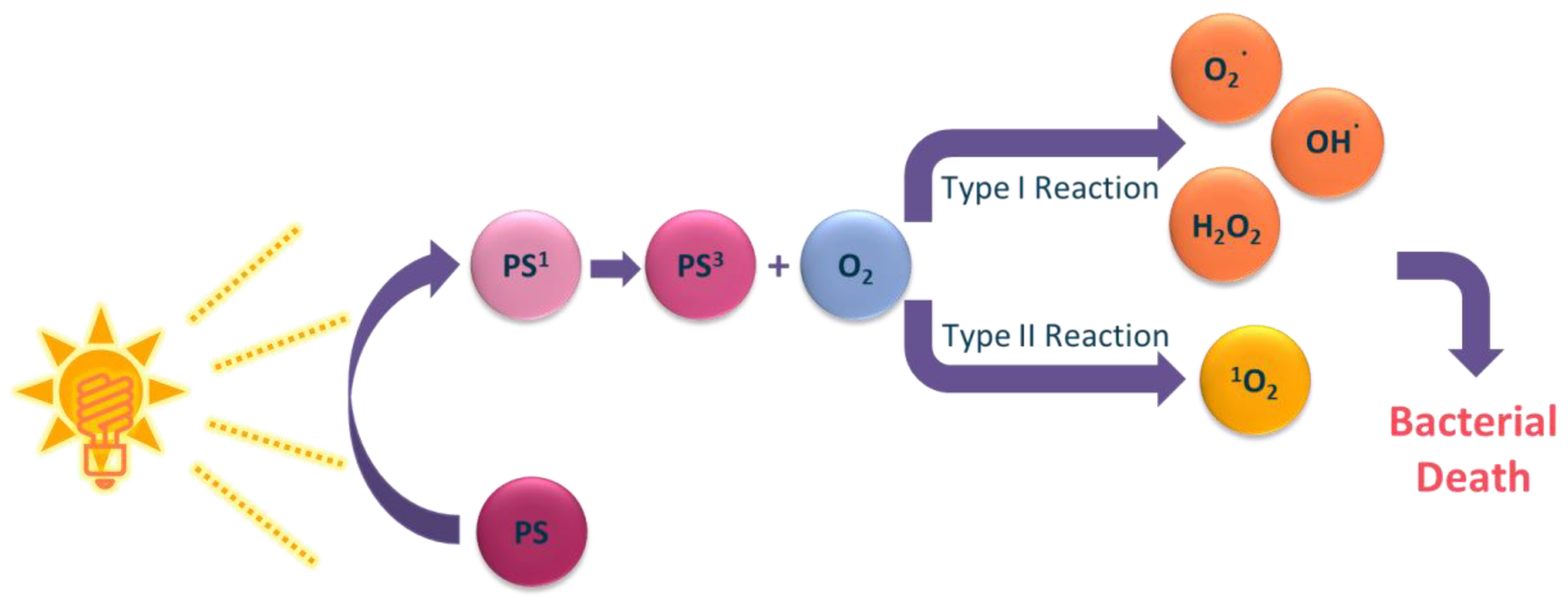
| Phytochemicals | Plant | Microorganism | References |
|---|---|---|---|
| 2-vinyl-2,4-dihydro-1,3-dithiin, 3-vinyl-3,4- dihydro-1,2-dithiin, and ajoene | Allium cepa | Bacillus subtilis | [89] |
| Ajoene, kaempferol, and allicin | Allium sativum | Pseudomonas aeruginosa Enterococcus faecalis Salmonella, Escherichia coli, Pseudomonas, Proteus, Staphylococcus aureus, Helicobacter | [90,91,92,93] |
| Proteins | Ananas comosus | Saccharomyces cerevisiae, Escherichia coli | [94] |
| Phenolic compounds | Annona squamosa | Staphylococcus aureus, Bacillus cereus, Staphylococcus epidermidis, Monilia albican, Escherichia coli, Salmonella typhimurium, Shigella flexneri, Pseudomonas aeruginosa | [37] |
| Limonene, pinene (-α, -β), and selinene (-α, -β) | Apium graveolens | Staphylococcus aureus, Bacillus subtilis, Escherichia coli, Pseudomonas aeruginosa | [95] |
| Berteroin and lesquerellin | Armoracia macrocarpa | Bacillus subtilis, Escherichia coli | [96] |
| Iberin and some undetermined compounds 5-phenylpentyl isothiocyanate | Armoracia rusticana | Pseudomonas aeruginosa 6 strains of facultative anaerobic bacteria, Streptococcus mutans, Streptococcus sobrinus, Lactobacillus casei, Staphylococcus aureus, Enterococcus faecalis, and Aggregatibacter actinomycetemcomitans; one strain of yeast, Candida albicans, and 3 strains of anaerobic bacteria, Fusobacterium nucleatum, Prevotella nigrescens, and Clostridium perfringens Staphylococcus aureus, Bacillus subtilis, Bacillus cereus, Proteus vulgaris, Escherichia coli, Salmonella enterica | [97,98,99] |
| Phenolic compounds | Aronia melanocarpa | Proteus mirabilis | [100] |
| Berberine | Berberis vulgaris | Helicobacter pylori | [101] |
| Protein: BjCHI1 | Brassica juncea | Several Gram-negative bacteria | [102] |
| Epicatechin, epicatechin gallate, epigallocatechin, and epigallocatechin gallate | Camellia sinensis | Staphylococcus epidermidis, Micrococcus luteus, Brevibacterium linens, Pseudomonas fluorescens, Bacillus subtilis | [103] |
| Capsaicin and dihydrocapsaicin | Capsicum species | Bacillus cereus, Bacillus subtilis, Clostridium sporogenes, Clostridium tetani, Streptococcus pyogenes | [104] |
| Saponins | Chenopodium quinoa | Staphylococcus aureus, Staphylococcus epidermidis, Bacillus cereus, Salmonella enteritidis, Pseudomonas aeruginosa, Listeria ivanovii | [82] |
| Kaempferol and some undetermined compounds presumably, tannins, saponins, flavonoids, alkaloids | Centella asiatica L. | Chromobacterium violaceum Escherichia coli | [105,106] |
| Oxalic, succinic, shikimic, and quinic acids | Cichorium intybus | Streptococcus mutans, Actinomyces naeslundii | [107] |
| Unknown | Cinnamomum verum | Pseudomonas aeruginosa | [108] |
| Unknown | Citrus medica | Listeria monocytogenes | [109] |
| l-limonene | Citrus reticulata | Escherichia coli, Staphylococcus aureus | [110] |
| Polyphenols | Citrus sinensis | Staphylococcus aureus, Enterococcus faecalis, Pseudomonas aeruginosa, Escherichia coli, and Salmonella typhimurium | [111] |
| Unknown | Coffea arabica | Staphylococcus aureus, Staphylococcus epidermidis, Pseudomonas aeruginosa, Escherichia coli | [112] |
| α-pinene, camphene, and linalool (2E)-dodecenal | Coriandrum sativum L. | Listeria monocytogenes, Salmonella choleraesuis | [113] |
| Protein PR-5 | Cucurbita moschata | Fusarium oxysporum | [114] |
| Unknown | Cucurbita pepo | Escherichia coli BL21, Escherichia coli B-23, Escherichia coli BL24 JPN, Shigella sonnei | [115] |
| The octamers of epigallocatechin and its gallate Phenolic compounds | Diospyros kaki | Salmonella enterica Staphylococcus aureus, Bacillus cereus, Staphylococcus epidermidis, Monilia albican, Escherichia coli, Salmonella typhimurium, Shigella flexneri, Pseudomonas aeruginosa | [37,116] |
| Sesquiterpenoids: jambolanins | Eugenia jambolana | Staphylococcus aureus | [117] |
| Unknown | Ficus carica | Streptococcus pyogenes, 10 Lactobacillus strains | [118] |
| Unknown | Hibiscus sabdariffa | Staphylococcus aureus, Klebsiella pneumoniae, Pseudomonas aeruginosa, Acinetobacter baumannii, Streptococcus mutans, Campylobacter jejuni, Campylobacter coli, Campylobacter fetus, Pseudomonas fluorescence | [119] |
| Kaempferol 3-O-alphaL-(2″,4″-di-E-p-coumaroyl)-rhamnoside (C2), and Kaempferol 3-O-alphaL-(2″-Z -p-coumaroyl)-rhamnoside (C3) | Laurus nobilis | Streptococcus pneumonia, Pseudomonas aeruginosa, Serratia marcessens | [120,121] |
| Flavonoids | Lepidium sativum | Proteus mirabilis, Staphylococcus epidermidis, Staphylococcus aureus | [122] |
| Unknown | Medicago truncatula | Chromobacterium violaceum, Pseudomonas putida, Escherichia coli | [123] |
| L-canvanine | Medigo sativa | Sinochrizobium melioti | [124] |
| Unknown Peptide | Momordica charantia | Microorganisms of clinical interest (standard strains and multiresistant isolates) Escherichia coli, Staphylococcus aureus | [125,126] |
| Unknown | Moringa oleifera | Pseudomonas aeruginosa, Staphylococcus aureus | [127] |
| Morin | Morus alba | Staphylococcus aureus | [56] |
| APC protein | Murraya koenigii L | Escherichia coli, Staphylococcus aureus, Vibrio cholerae, Klebsiella pneumoniae, Salmonella typhi, Bacillus subtilis | [128] |
| Estragol | Ocimum basilicum | Bacillus subtilis, Staphylococcus aureus | [129] |
| Hydroxytyrosol, oleuropein | Olea europaea | Salmonella enterica, Escherichia coli | [130] |
| Unknown Thymol and carvacrol | Origanum vulgare | Chromobacterium violaceum, Bacillus species, Salmonella enteritidis | [131,132,133] |
| Anglicin and psoralen | Pastinaca sativa | Staphylococcus aureus | [134] |
| Apiol, myristicin, and b-phellandrene | Petroselinum crispum | Staphylococcus aureus, Listeria monocytogenes, Salmonella enterica | [135] |
| Unknown | Piper longum L. | Chromobacterium violaceum | [136] |
| Unknown | Pistachia vera | Bacillus cereus, Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, Salmonella typhi, Candida albicans, Neurospora intermedia | [137] |
| 3-methoxy-4,5-dinitrophenol | Portulaca oleracea | Staphylococcus aureus, Staphylococcus sonnei, Acinetobacter baumannii, Bacillus subtilis | [138] |
| Phenolics, flavonoids, ortho-diphenols, and saponins. | Prunus avium | Staphylococcus aureus, Escherichia coli | [139] |
| Citric acid | Prunus mume | Enterobacteria | [140] |
| Quercetin and quercetin-3-O-arabinoside | Psidium guajava L. | Chromobacterium violaceum, Pseudomonas aeruginosa | [105] |
| α-amyrin and flavonoid compounds, steroidal compounds | Pyrus bretschneideri Rehd. | Staphylococcus aureus, Escherichia coli | [141] |
| Unknown | Raphanus raphanistrum L. | Klebsiella pneumoniae, Pseudomonas aeruginosa | [142] |
| Sulforaphene | Raphanus sativus | Staphylococcus aureus | [143] |
| Unknown | Rheum officinale | Vibrio parahaemolyticus, Vibrio vulnificus, Vibrio alginolyticus, Vibrio carchariae, Aeromonas hydrophila, Edwardsiella tarda | [144] |
| Unknown | Ribes nigrum | Streptococcus pyogenes, 10 Lactobacillus strains | [118] |
| Unknown | Rosa rugosa | Bacillus cereus | [145] |
| Unknown | Rosmarinus officinalis | Streptococcus pneumoniae | [146] |
| Unknown | Solanum melongena | Pseudomonas aeruginosa | [147] |
| Eugenol | Syzygium aromaticum | Streptococcus pneumoniae | [148] |
| Unknown Squalene, campesterol, tocopherol, isooctyl phthalate, ethyl glicopyranoside, stigmasterol, hexadecanoic acid, malvidin | Syzygium cumini | Chromobacterium violaceum Actinomyces naeslundii, Fusobacterium nucleatum, Staphylococcus aureus, Staphylococcus epidermidis, Veillonella dispar, Klebsiella pneumoniae | [105,149,150] |
| Chebulic acid, combretastatin A1, corilagin, diethylstilbestrol, ellagic acid, ethyl gallate, gallic acid, piceid, resveratrol | Terminalia ferdinandiana | Proteus mirabilis, Proteus vulgaris | [151] |
| Phenolic compounds, γ-terpinene | Thymus vulgaris | Staphylococcus aureus, Pseudomonas aeruginosa, Salmonella typhimurium, Escherichia coli, Klebsiella pneumoniae, Enterococcus faecalis | [152] |
| Unknown | Vanilla planifolia Andrews | Chromobacterium violaceum | [153] |
| Isothiocyanates | Wasabia japonica | Staphylococcus aureus, Pseudomonas aeruginosa, Salmonella typhimurium, Escherichia coli | [154] |
| Phenolic components | Zingiber officinale Roscoe | Chromobacterium violaceum, Pseudomonas aeruginosa | [155] |
| Photosensitizer | Structure |
|---|---|
| Aloe emodin | 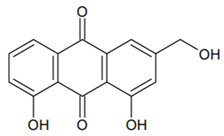 |
| Anthocyanidin | 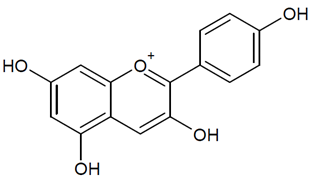 |
| Berberine |  |
| Chlorin e6 | 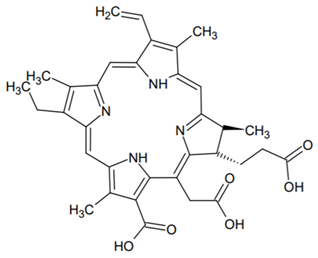 |
| Curcumin | 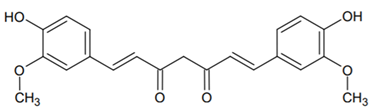 |
| Emodin | 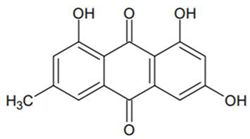 |
| Fagopyrin |  |
| Flavan-3-ol |  |
| Flavone | 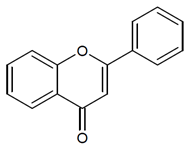 |
| Hypericin | 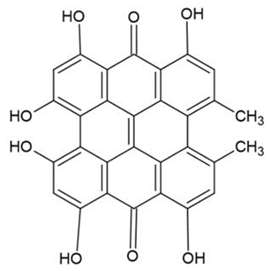 |
| Rhein | 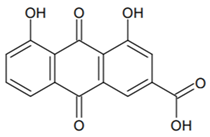 |
| Riboflavin | 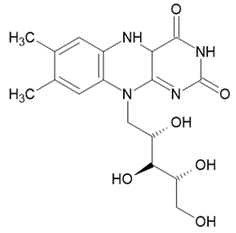 |
| Chemical Class | PS-Phytochemicals | Edible Plants Containing the PS-Phytochemicals 1 | Tested Microorganisms | References |
|---|---|---|---|---|
| Curcuminoids | Curcumin | Turmeric (Curcuma longa) | Acinetobacter baumannii | [211] |
| Aggregatibacter actinomycetemcomitans | [212,213] | |||
| Escherichia coli | [214,215,216,217,218,219,220] | |||
| Enterococcus faecalis | [221,222,223,224,225,226,227] | |||
| Helicobacter pylori | [228] | |||
| Lactobacillus casei | [179] | |||
| Listeria innocua | [216,229] | |||
| Listeria monocytogenes | [230,231] | |||
| Porphyromonas gingivalis | [212] | |||
| Propionibacterium acnes | [224,232] | |||
| Pseudomonas | [233] | |||
| Pseudomonas aeruginosa | [234] | |||
| Staphylococcus aureus | [177,214,215,218,220,234,235,236,237,238,239,240,241,242,243] | |||
| Salmonella strains | [230] | |||
| Salmonella typhimurium | [239] | |||
| Streptococcus mutans | [177,179,244,245,246,247,248,249,250,251,252] | |||
| Staphylococcus saprophyticus | [253] | |||
| Vibrio parahaemolyticus | [254,255] | |||
| Oral bacteria | [202,256,257,258,259,260,261] | |||
| Bacterial Biofilms | [262] | |||
| Candida albicans | [179,263,264] | |||
| Anthraquinones | Aloe emodin | Rheum palmatum | Acinetobacter baumannii | [186,187] |
| Escherichia coli | [218] | |||
| Enterococcus faecalis | [185] | |||
| Staphylococcus aureus | [185,218] | |||
| Streptococcus pneumonia | [185] | |||
| Trichophyton rubrum | [190] | |||
| Candida albicans | [189] | |||
| Emodin | Cassia occidentalis | Bacillus subtilis | [265] | |
| Staphylococcus aureus | [265] | |||
| Rhein | Rheum palmatum | Porphyromonas gingivalis | [266] | |
| Saprolegnia sp. | [188] | |||
| Perylenequinones | Hypericin | Hypericum perforatum, Hypericum erectum, Hypericum perforatum L. | Bacillus cereus | [267] |
| Escherichia coli | [268,269,270] | |||
| Enterococcus faecalis | [270] | |||
| Propionibacterium acnes | [271] | |||
| Pseudomonas aeruginosa | [270] | |||
| Staphylococcus aureus | [268,269,270,272,273] | |||
| Staphylococcus saprophyticus subsp. bovis | [274] | |||
| Candida species (Candida albicans, Candida parapsilosis, and Candida krusei) | [275] | |||
| Saccharomyces cerevisiae | [276] | |||
| Pathogenic fungi (Microsporum canis, Trichophyton rubrum, Fusarium oxysporum) and spoilage yeasts (Exophiala dermatitidis, Candida albicans, Kluyveromyces marxianus, Pichia fermentans, Saccharomyces cerevisiae) | [277] | |||
| Fagopyrin | Fagopyrum esculentum Moench | Pathogenic fungi (Microsporum canis, Trichophyton rubrum, Fusarium oxysporum) and spoilage yeasts (Exophiala dermatitidis, Candida albicans, Kluyveromyces marxianus, Pichia fermentans, Saccharomyces cerevisiae) | [277] | |
| Saccharomyces cerevisiae | [276] | |||
| Fagopyrin F | Tartary buckwheat (Fagopyrum tataricum) | Streptococcus mutans | [278] | |
| Furanocoumarins | 8-methoxy-psoralen | Species of the genus Heracleum in the family Apiaceae | Escherichia coli | [279] |
| Staphylococcus aureus | [279] | |||
| Salmonella typhimurium | [280] | |||
| Saccharomyces cerevisiae | [280] | |||
| Alkaloids | Berberine | Mahonia aquifolium, Berberis vulgaris | Escherichia coli | [198,199,200] |
| Staphylococcus aureus | [198,199,200] | |||
| Chlorins | Chlorin e6 | Spinach, green cabbage, dandelion | Escherichia coli | [281] |
| Propionibacterium acnes | [282] | |||
| Pseudomonas aeruginosa | [281] | |||
| Staphylococcus aureus | [281,283] | |||
| Staphylococcus aureus | [282] | |||
| Streptococcus mutans | [284] | |||
| Salmonella typhimurium | [281] | |||
| Upper respiratory opportunistic pathogens (Moraxella catarrhalis, Streptococcus pneumoniae, Haemophilus influenzae) | [285] | |||
| Flavins | Riboflavin (vitamin B2) | Spinach, asparagus | Bacillus atrophaeus | [286] |
| Escherichia coli | [287,288] | |||
| Listeria monocytogenes | [289,290] | |||
| Staphylococcus aureus | [288] | |||
| Streptococcus mutans | [291] | |||
| Streptococcus sanguinis | [291] | |||
| Salmonella strains (Salmonella typhimurium and Salmonella enteritidis) | [292] | |||
| Shewanella baltica | [290] | |||
| Vibrio parahaemolyticus | [290] | |||
| Candida albicans | [293] |
| Antimicrobial Type | Plant Antimicrobials | Plant Source 1 | Target Microorganism | Food | Reference |
|---|---|---|---|---|---|
| Essential oil | Bay leaf essential oil | Laurus nobilis, Lauraceae | Coliforms | Tuscan sausage | [296] |
| Clove oil | Syzygium aromaticum | Listeria monocytogenes | Minced fish | [297] | |
| Native microflora | Meat | [298] | |||
| Cuminum (cumin) seed essential oil | Cuminum cyminum | Spoilage moulds | Wheat and chickpea samples | [299] | |
| Oregano essential oil | Origanum elongatum | Salmonela enteritidis | Minced sheep meat | [300] | |
| Origanum elongatum essential oil | Origanum elongatum | Lactic acid bacteria, yeasts and molds | Pomegranate juice | [301] | |
| Thyme or marjoram essential oils | Thymus vulgaris and Origanum majorana | Escherichia coli | Minced pork | [302] | |
| Thyme essential oil | Thymus vulgaris | Escherichia coli | Minced beef | [303] | |
| Vancomycin-resistant Enterococci and E. coli | Feta soft cheese | [304] | |||
| Vancomycin-resistant Enterococci and Escherichia coli | Minced beef meat | [304] | |||
| Zataria multiflora Boiss essential oil | Zataria multiflora | Listeria monocytogenes | Buffalo patties | [305] | |
| Antimicrobial peptides (AMPs) | Defensin KT43C | Cowpea seeds | Fusarium culmorum, Penicillium expansum, and Aspergillus niger | Dough | [306] |
| Snakin | Potato tubers | Listeria monocytogenes and Listeria ivanovii | Potato | [307] | |
| spoilage yeast; Zygosaccharomyces bailii | Beverages | [308] | |||
| Thionins | Triticum aestivum (wheat) | Listeria monocytogenes and Listeria ivanovii | Wheat | [309] | |
| Plant extract | Black seed cumin | Cuminum cyminum | Escherichia coli and Enterococci spp. | Meat | [310] |
| Clove | Syzygium aromaticum | Escherichia coli and Enterococci spp. | Meat | [310] | |
| Curcumin | Turmeric (Curcuma longa L.) | Aspergillus flavus | Maize kernels | [311] | |
| Escherichia coli, Salmonella, and Listeria monocytogenes | Hami-melons | [312] | |||
| Grape seeds | Grape seeds | Listeria monocytogenes | Buffalo patties | [305] | |
| Raisin | Raisin (species Hovenia dulcis) | Mold | Wheat | [313] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hochma, E.; Yarmolinsky, L.; Khalfin, B.; Nisnevitch, M.; Ben-Shabat, S.; Nakonechny, F. Antimicrobial Effect of Phytochemicals from Edible Plants. Processes 2021, 9, 2089. https://doi.org/10.3390/pr9112089
Hochma E, Yarmolinsky L, Khalfin B, Nisnevitch M, Ben-Shabat S, Nakonechny F. Antimicrobial Effect of Phytochemicals from Edible Plants. Processes. 2021; 9(11):2089. https://doi.org/10.3390/pr9112089
Chicago/Turabian StyleHochma, Efrat, Ludmila Yarmolinsky, Boris Khalfin, Marina Nisnevitch, Shimon Ben-Shabat, and Faina Nakonechny. 2021. "Antimicrobial Effect of Phytochemicals from Edible Plants" Processes 9, no. 11: 2089. https://doi.org/10.3390/pr9112089
APA StyleHochma, E., Yarmolinsky, L., Khalfin, B., Nisnevitch, M., Ben-Shabat, S., & Nakonechny, F. (2021). Antimicrobial Effect of Phytochemicals from Edible Plants. Processes, 9(11), 2089. https://doi.org/10.3390/pr9112089








