Adsorption of Tetracycline and Sulfadiazine onto Three Different Bioadsorbents in Binary Competitive Systems
Abstract
1. Introduction
2. Materials and Methods
2.1. Bioadsorbent Materials
2.2. Chemicals
2.3. Adsorption/Desorption Experiments
2.3.1. Adsorption
2.3.2. Desorption
2.4. Quantification of the Antibiotics
2.5. Data Analysis and Statistical Treatment
3. Results and Discussion
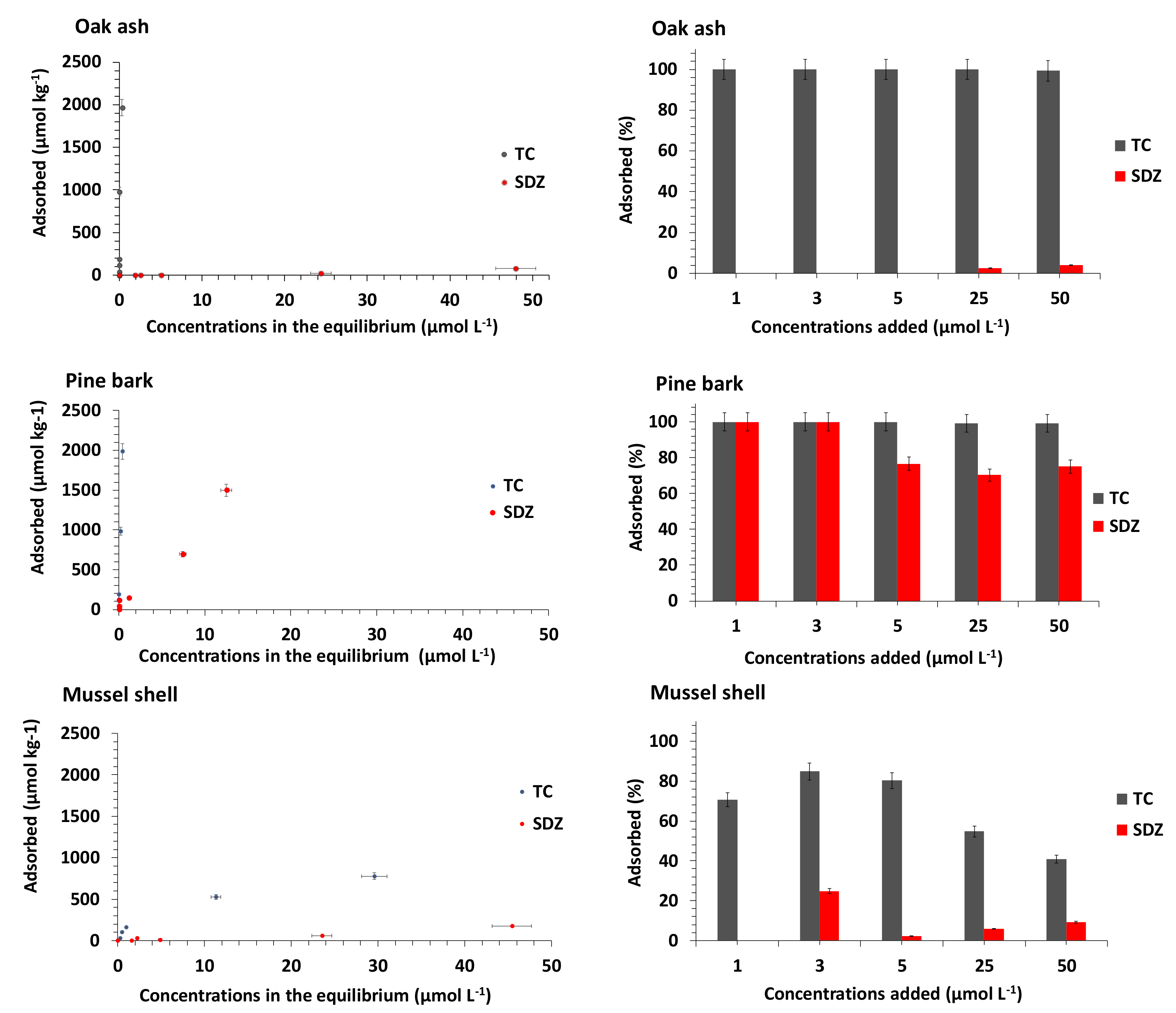
3.1. Adsorption of TC and SDZ onto the Bioadsorbents in Binary Systems
3.2. Fitting of TC and SDZ Adsorption Data to Different Models
3.3. Desorption of TC and SDZ from the Three Bioadsorbents in Binary Systems
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Albero, B.; Tadeo, J.L.; Escario, M.; Miguel, E.; Pérez, R.A. Persistence, and availability of veterinary anbitiotics in soil and soil-manure systems. Sci. Total Environ. 2018, 643, 1562–1570. [Google Scholar] [CrossRef] [PubMed]
- Cycon, M.; Mrozik, A.; Piotrowska-Seget, Z. Anbitiotics in the Soil Environment—Degradation and Their Impact on Microbial Activity and Diversity. Front. Microbiol. 2019, 10, 338. [Google Scholar] [CrossRef] [PubMed]
- Hanna, N.; Sun, P.; Sun, Q.; Li, X.W.; Yang, X.W.; Ji, X.; Zou, H.Y.; Ottoson, J.; Nilsson, L.E.; Berglund, B.; et al. Presence of antibiotic residues in various environmental compartments of Shandong province in eastern China: Its potential for resistance development and ecological and human risk. Environ. Int. 2018, 114, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Tasho, R.P.; Cho, J.Y. Veterinary antibiotics in animal waste, its distribution in soil and uptake by plants: A review. Sci. Total Environ. 2016, 563, 366–376. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, Z.; Zhu, Y. Changes in abiotic dissipation rates and bound fractions of antibiotics in biochar-amended soil. J. Clean. Prod. 2020, 256, 120312. [Google Scholar] [CrossRef]
- Conde-Cid, M.; Álvarez-Esmorís, C.; Paradelo-Núñez, R.; Novoa-Muñoz, J.C.; Arias-Estevez, M.; Álvarez-Rodríguez, E.; Fernández-Sanjurjo, M.J.; Núñez-Delgado, A. Occurrence of tetracyclines and sulfonamides in manures, agricultural soils and crops from different areas in Galicia (NW Spain). J. Clean. Prod. 2018, 197, 491–500. [Google Scholar] [CrossRef]
- Yang, Y.Y.; Song, W.J.; Lin, H.; Wang, W.B.; Du, L.N.; Xing, W. Antibiotics and antibiotic resistance genes in global lakes: A review and meta-analysis. Environ. Int. 2018, 116, 60–73. [Google Scholar] [CrossRef]
- Núñez-Delgado, A.; López-Periago, E.; Díaz-Fierros Viqueira, F. Chloride, sodium, potassium and faecal bacteria levels in surface runoff and subsurface percolates from grassland plots amended with cattle slurry. Bioresour. Technol. 2002, 82, 261–271. [Google Scholar] [CrossRef]
- Pousada-Ferradás, Y.; Seoane-Labandeira, S.; Mora-Gutierrez, A.; Núñez-Delgado, A. Risk of water pollution due to ash–sludge mixtures: Column trials. Int. J. Environ. Sci. Technol. 2012, 9, 21–29. [Google Scholar] [CrossRef][Green Version]
- Ben, Y.J.; Fu, C.X.; Hu, M.; Liu, L.; Wong, M.H.; Zheng, C.M. Human health risk assessment of antibiotic resistance associated with antibiotic residues in the environment: A review. Environ. Res. 2019, 169, 483–493. [Google Scholar] [CrossRef]
- Ding, H.J.; Wu, Y.X.; Zou, B.C.; Lou, Q.; Zhang, W.H.; Zhong, J.Y.; Lu, L.; Dai, G.F. Simultaneous removal and degradation characteristics of sulfonamide, tetracycline, and quinolone antibiotics by laccase-mediated oxidation coupled with soil adsorption. J. Hazard. Mater. 2016, 307, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Conde-Cid, M.; Ferreira-Coelho, G.; Arias-Estévez, M.; Fernández-Calvinho, D.; Núñez-Delgado, A.; Álvarez-Rodríguez, E.; Fernández- Sanjurjo, M.J. Adsorption/desorption of three tetracycline antibiotics on different soils in binary competitive systems. J. Environ. Manag. 2020, 262, 110337. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Zhang, Q.; Deng, X.; Nan, Z.; Liang, X.; Wen, H.; Huang, K.; Wu, Y. Single and competitive sorption of sulfadiazine and chlortetracycline on loess soil from Northwest China. Environ. Pollut. 2020, 263, 114650. [Google Scholar] [CrossRef]
- Quintáns-Fondo, A.; Ferreira-Coelho, G.; Paradelo-Núñez, R.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. As(V)/Cr(VI) pollution control in soils, hemp waste, and other by-products: Competitive sorption trials. Environ. Sci. Pollut. Res. 2016, 23, 18182–19192. [Google Scholar] [CrossRef]
- Conde-Cid, M.; Fernández-Calviño, D.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Díaz-Raviña, M.; Fernández-Sanjurjo, M.J.; Núñez-Delgado, A.; Álvarez-Rodríguez, E. Biotic and abiotic dissipation of tetracyclines using simulated sunlight and in the dark. Sci. Total Environ. 2018, 635, 1520–1529. [Google Scholar] [CrossRef] [PubMed]
- Conde-Cid, M.; Fernández-Calviño, D.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Díaz-Raviña, M.; Núñez-Delgado, A.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E. Degradation of sulfadiazine, sulfachloropyridazine and sulfamethazine in aqueous media. J. Environ. Manag. 2018, 228, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Calviño, D.; Bermúdez-Couso, A.; Arias-Estévez, M.; Nóvoa-Muñoz, J.C.; Fernández- Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Kinetics of tetracycline, oxytetracycline and chlortetracycline adsorption and desorption on two acid soils. Environ. Sci. Pollut. Res. 2015, 22, 425–433. [Google Scholar] [CrossRef]
- Fernández-Calviño, D.; Bermúdez-Couso, A.; Arias-Estévez, M.; Nóvoa-Muñoz, J.C.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Competitive adsorption/desorption of tetracycline, oxytetracycline and chlortetracycline on two acid soils: Stirred flow chamber experiments. Chemosphere 2015, 134, 361–366. [Google Scholar] [CrossRef]
- Arias, M.; Pérez-Novo, C.; López, E.; Soto, B. Competitive adsorption and desorption of copper and zinc in acid soils. Geoderma 2006, 133, 151–159. [Google Scholar] [CrossRef]
- Murali, V.; Aylmore, A.G. Competitive adsorption during solute transport in soils: 1. Mathematical models. Soil Sci. 1983, 135, 143–150. [Google Scholar] [CrossRef]
- Conde-Cid, M.; Fernández-Sanjurjo, M.J.; Ferreira-Coelho, G.; Fernández-Calviño, D.; Arias-Estevez, M.; Núñez-Delgado, A.; Álvarez-Rodríguez, E. Competitive adsorption and desorption of three tetracycline antibiotics on bio-sorbent materials in binary systems. Environ. Res. 2020, 190, 110003. [Google Scholar] [CrossRef] [PubMed]
- Conde-Cid, M.; Fernández-Calviño, D.; Núñez-Delgado, A.; Fernández-Sanjurjo, M.J.; Arias-Estévez, M.; Álvarez-Rodríguez, E. Influence of mussel shell, oak ash and pine bark on the adsorption and desorption of sulfonamides in agricultural soils. J. Environ. Manag. 2020, 261, 110221. [Google Scholar] [CrossRef] [PubMed]
- Sarmah, A.K.; Meyer, M.T.; Boxall, A.B. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006, 65, 725–759. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, I.T.; Santos, L. Antibiotics in the aquatic environments: A review of the European scenario. Environ. Int. 2016, 94, 736–757. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, H. Adsorption behavior of antibiotic in soil environment: A critical review. Front. Environ. Sci. Eng. 2015, 9, 565–574. [Google Scholar] [CrossRef]
- Wegst-Uhrich, S.R.; Navarro, D.A.G.; Zimmerman, L.; Aga, D.S. Assessing antibiotic sorption in soil: A literature review and new case studies on sulfonamides and macrolides. Chem. Cent. J. 2014, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Rath, S.; Fostier, A.H.; Pereira, L.A.; Dioniso, A.C.; Ferreira, F.D.O.; Doretto, K.M. Sorption behaviors of antimicrobial and antiparasitic veterinary drugs on subtropical soils. Chemosphere 2019, 214, 111–122. [Google Scholar] [CrossRef]
- Teixidó, M.; Granados, M.; Prat, M.D.; Beltrán, J.L. Sorption of tetracyclines onto natural soil: Date analysis and prediction. Environ. Sci. Pollut. Res. 2012, 19, 3087–3095. [Google Scholar] [CrossRef]
- Chen, M.; Yi, Q.; Hong, J.; Zhang, L.; Lin, K.; Yuan, D. Simultaneous determination of 32 antibiotics and 12 pesticides in sediment using ultrasonic-assisted extraction and high performance liquid chromatography-tandem mass spectrometry. Anal. Methods 2015, 7, 1896–1905. [Google Scholar] [CrossRef]
- Babić, S.; Horvat, A.J.M.; Mutavdžić-Pavlović, D.; Kaštelan-Macan, M. Determination of pKa values of active pharmaceutical ingredients. Trends Anal. Chem. 2007, 26, 1043–1061. [Google Scholar] [CrossRef]
- Figueroa-Diva, R.A.; Vasudevan, D.; MacKay, A.A. Trends in soil sorption coefficients within common antimicrobial families. Chemosphere 2010, 79, 786–793. [Google Scholar] [CrossRef] [PubMed]
- Białk-Bielińska, A.; Stolte, S.; Matzke, M.; Fabiańska, A.; Maszkowska, J.; Kołodziejska, M.; Liberek, B.; Stepnowski, P.; Kumirska, J. Hydrolysis of sulphonamides in aqueous solutions. J. Hazard. Mater. 2012, 221, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W.S.; Johir, M.A.H.; Sornalingam, K. Single and competitive sorption properties and mechanism of functionalized biochar for removing sulfonamide antibiotics from water. Chem. Eng. J. 2017, 311, 348–358. [Google Scholar] [CrossRef]
- Edwards, M.; Benjamin, M.M.; Ryan, J.N. Role of organic acidity in sorption of natural organic matter (NOM) to oxide surfaces. Colloids Surf. Physicochem. Eng. Asp. 1996, 107, 297–307. [Google Scholar] [CrossRef]
- Conde-Cid, M.; Ferreira-Coelho, G.; Arias-Estévez, M.; Álvarez-Esmorís, C.; Nóvoa-Muñoz, J.C.; Núñez-Delgado, A.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E. Competitive adsorption/desorption of tetracycline, oxytetracycline and chlortetracycline on pine bark, oak ash and mussel shell. J. Environ. Manag. 2019, 250, 109509. [Google Scholar] [CrossRef] [PubMed]
- Conde-Cid, M.; Ferreira-Coelho, G.; Fernández-Calviño, D.; Núñez-Delgado, A.; Fernández-Sanjurjo, M.J.; Arias-Estévez, M.; Álvarez-Rodríguez, E. Single and simultaneous adsorption of three sulfonamides in agricultural soils: Effects of pH and organic matter content. Sci. Total Environ. 2020, 744, 140872. [Google Scholar] [CrossRef]
- Sukul, P.; Lamshöft, M.; Zühlke, S.; Spiteller, M. Sorption and desorption of sulfadiazine in soil and soil-manure systems. Chemosphere 2008, 73, 1344. [Google Scholar] [CrossRef]
- Leal, R.M.P.; Alleoni, L.R.F.; Tornisielo, V.L.; Regitano, J.B. Sorption of fluoroquinolones and sulfonamides in 13 Brazilian soils. Chemosphere 2013, 92, 979. [Google Scholar] [CrossRef]
- Hu, S.; Zhang, Y.; Shen, G.; Zhang, H.; Yuan, Z.; Zhang, W. Adsorption/desorption behavior cand mechanisms of sulfadiazine and sulfamethoxazole in agricultural soil systems. Soil Tillage Res. 2019, 186, 233. [Google Scholar] [CrossRef]
- Conde-Cid, M.; Fernández-Calviño, D.; Nóvoa-Muñoza, J.C.; Núñez-Delgado, A.; Fernández-Sanjurjo, M.J. Arias-Estévez, M.; Álvarez-Rodríguez, E. Experimental data and model prediction of tetracycline adsorption and desorption in agricultural soils. Environ. Res. 2019, 177, 108607. [Google Scholar] [CrossRef]
- Bao, Y.J.; Ding, H.S.; Bao, Y.Y. Effects of Temperature on the Adsorption and Desorption of Tetracycline in Soils. Adv. Mater. Res. 2013, 726–731, 344–347. [Google Scholar] [CrossRef]
- Bhaumik, R.; Mondal, N.K.; Das, B.; Roy, P.K.C. Eggshell powder as an adsorbent for removal of fluoride from aqueous solution: Equilibrium, kinetic and thermodynamic studies. J. Chem. 2012, 9, 1457–1480. [Google Scholar] [CrossRef]
- Na, C.J.; Yoo, M.J.; Tsang, D.C.W.; Kim, H.W.; Kim, K.H. High-performance materials for effective sorptive removal of formaldehyde in air. J. Hazard. Mater. 2019, 366, 452–465. [Google Scholar] [CrossRef] [PubMed]
- Szulejko, J.E.; Kim, K.H.; Parise, J. Seeking the most powerful and practical realworld sorbents for gaseous benzene as a representative volatile organic compound based on performance metrics. Sep. Purif. Technol. 2019, 212, 980–985. [Google Scholar] [CrossRef]
- Vikrant, K.; Kim, K.H. Nanomaterials for the adsorptive treatment of Hg (II) ions from water. Chem. Eng. J. 2019, 358, 264–282. [Google Scholar] [CrossRef]
- Khezami, L.; Capart, R. Removal of chromium (VI) from aqueous solution by activated carbons: Kinetic and equilibrium studies. J. Hazard. Mater. 2005, 123, 223–231. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Behnajady, M.A.; Bimeghdar, S. Synthesis of mesoporous NiO nanoparticles and their application in the adsorption of Cr(VI). Chem. Eng. J. 2014, 239, 105–113. [Google Scholar] [CrossRef]
- Conde-Cid, M.; Fernandez-Calviño, D.; Fernandez-Sanjúrjo, M.J.; Núñez-Delgado, A.; Álvarez-Rodríguez, E.; Arias-Estevez, M. Adsorption/desorption and transport of sulfadiazine, sulfachloropyridazine, and sulfamethazine, in acid agricultural soils. Chemosphere 2019, 234, 978. [Google Scholar] [CrossRef]

| Bioadsorbent | Antibiotic | Kd (L kg−1) | Error | R2 |
|---|---|---|---|---|
| TC | - | - | - | |
| Oak ash | SDZ TC + SDZ | 1.51 42.06 | 0.16 0.46 | 0.939 0.999 |
| TC | 4805.01 | 275.58 | 0.979 | |
| Pine bark | SDZ TC + SDZ | 113.09 256.45 | 7.17 12.42 | 0.975 0.982 |
| TC | 29.04 | 4.18 | 0.858 | |
| Mussel shell | SDZ TC + SDZ | 3.50 13.49 | 0.37 0.96 | 0.934 0.955 |
| Bioadsorbent | Antibiotic | Langmuir Parameter | ||||
|---|---|---|---|---|---|---|
| Qm (µmol kg−1) | Error | KL (L µmol−1) | Error | R2 | ||
| Mussel shell | TC | 934.35 | 97.63 | 0.14 | 0.05 | 0.986 |
| Sorbent | Antibiotic | Freundlich Parameter | ||||
|---|---|---|---|---|---|---|
| KF (Ln µmol1−n kg−1) | Error | n | Error | R2 | ||
| Oak ash | TC | - | - | - | - | - |
| SDZ | 0.07 | 0.01 | 1.82 | 0.05 | 1 | |
| TC + SDZ * | 35.16 | 3.12 | 1.05 | 0.02 | 1 | |
| Pine bark | TC | 6036.53 | 1411.70 | 1.22 | 0.22 | 0.985 |
| SDZ | 46.07 | 23.98 | 1.37 | 0.21 | 0.988 | |
| TC + SDZ * | 123.29 | 54.40 | 1.30 | 0.6 | 0.991 | |
| Mussel shell | TC | 148.88 | 18.95 | 0.50 | 0.04 | 0.992 |
| SDZ | - | - | 1.67 | 0.37 | 0.997 | |
| TC + SDZ * | 50.18 | 9.16 | 0.70 | 0.04 | 0.997 | |
| Bioadsorbent | a12 | R2 | a21 | R2 |
|---|---|---|---|---|
| Oak ash | −0.956 | 0.907 | 0 | - |
| Pine bark | −0.555 | 0.991 | −0.013 | 0.975 |
| Mussel shell | −0.501 | 0.725 | −0.045 | 0.98 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cela-Dablanca, R.; Conde-Cid, M.; Ferreira-Coelho, G.; Arias-Estévez, M.; Fernández-Calviño, D.; Núñez-Delgado, A.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E. Adsorption of Tetracycline and Sulfadiazine onto Three Different Bioadsorbents in Binary Competitive Systems. Processes 2021, 9, 28. https://doi.org/10.3390/pr9010028
Cela-Dablanca R, Conde-Cid M, Ferreira-Coelho G, Arias-Estévez M, Fernández-Calviño D, Núñez-Delgado A, Fernández-Sanjurjo MJ, Álvarez-Rodríguez E. Adsorption of Tetracycline and Sulfadiazine onto Three Different Bioadsorbents in Binary Competitive Systems. Processes. 2021; 9(1):28. https://doi.org/10.3390/pr9010028
Chicago/Turabian StyleCela-Dablanca, Raquel, Manuel Conde-Cid, Gustavo Ferreira-Coelho, Manuel Arias-Estévez, David Fernández-Calviño, Avelino Núñez-Delgado, María J. Fernández-Sanjurjo, and Esperanza Álvarez-Rodríguez. 2021. "Adsorption of Tetracycline and Sulfadiazine onto Three Different Bioadsorbents in Binary Competitive Systems" Processes 9, no. 1: 28. https://doi.org/10.3390/pr9010028
APA StyleCela-Dablanca, R., Conde-Cid, M., Ferreira-Coelho, G., Arias-Estévez, M., Fernández-Calviño, D., Núñez-Delgado, A., Fernández-Sanjurjo, M. J., & Álvarez-Rodríguez, E. (2021). Adsorption of Tetracycline and Sulfadiazine onto Three Different Bioadsorbents in Binary Competitive Systems. Processes, 9(1), 28. https://doi.org/10.3390/pr9010028











