Technical Potential for Energy and GWP Reduction in Chemical–Pharmaceutical Industry in Germany and EU—Focused on Biologics and Botanicals Manufacturing
Abstract
1. Introduction
2. Biologics
2.1. Overview and Current State of Biologics
- Single-use plastic items are forbidden and reduced in daily life; in biotech manufacturing, however, they are booming due to single-use technology, advantages in cleaning efforts, personal resources and flexibility in various batches and products, as well as costs [46].
- Other studies point out that the pharmaceutical industry is inefficient compared to automotive manufacturing [47]: “The total global emissions of the pharma sector amounts to about 52 megatonnes of CO2e in 2015, more than the 46.4 megatonnes of CO2e generated by the automotive sector in the same year. The value of the pharma market, however, is smaller than the automotive market. By our calculations, the pharma market is 28 percent smaller yet 13 percent more polluting than the automotive sector”.
2.2. Technical Project Results for Biologics
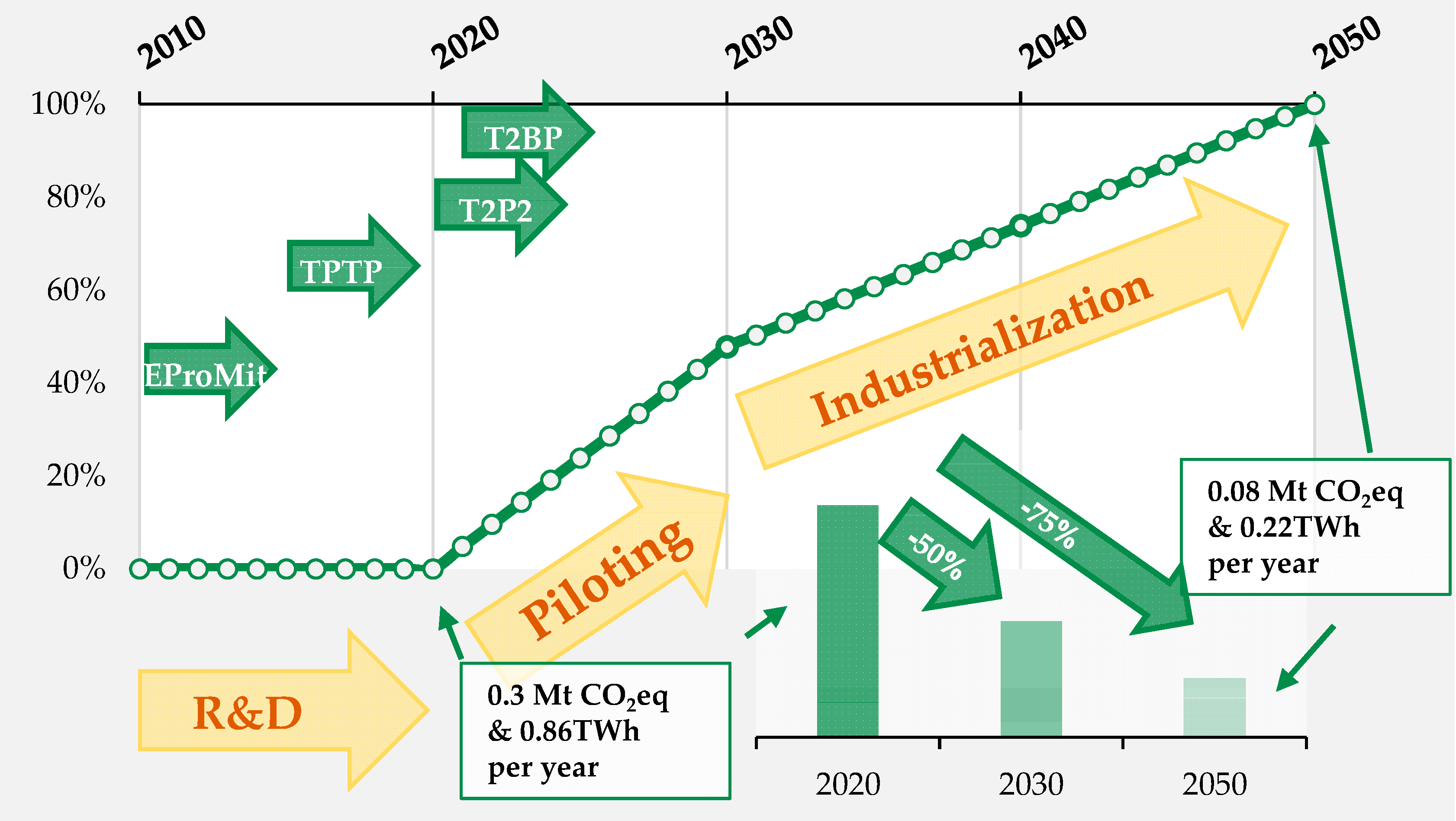
2.3. Industrialization of Biologics
3. Botanicals
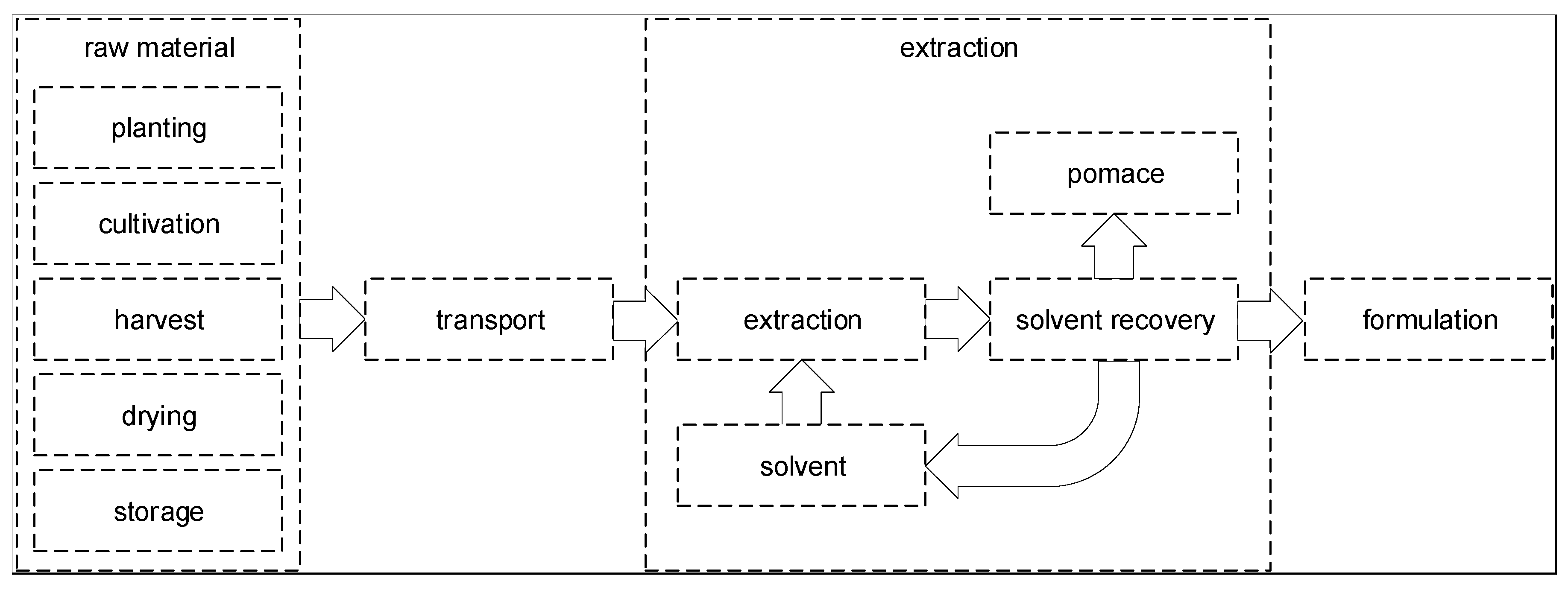
3.1. Overview and Current State of Botanicals
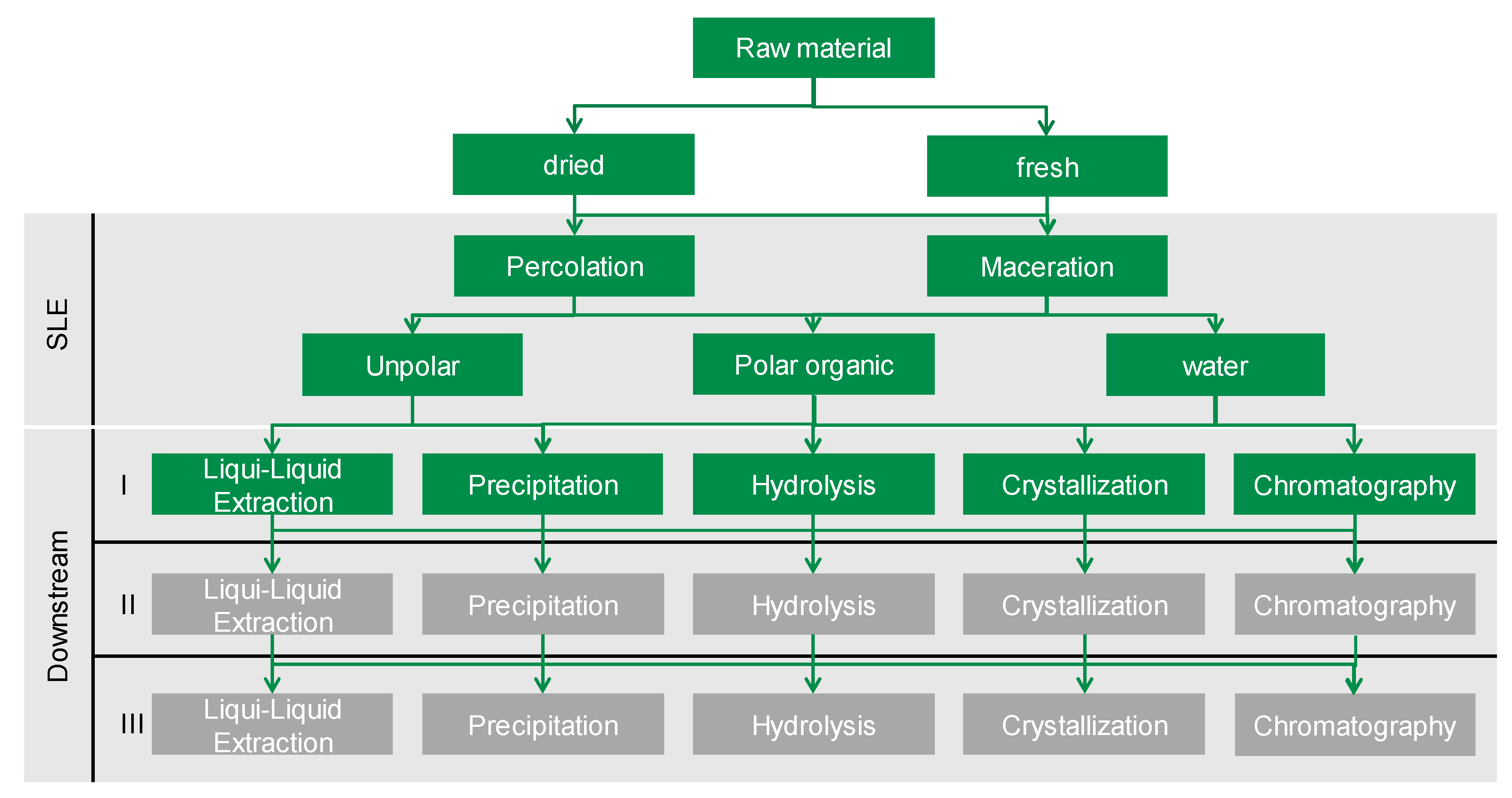
3.2. Technical Project Results for Botanicals
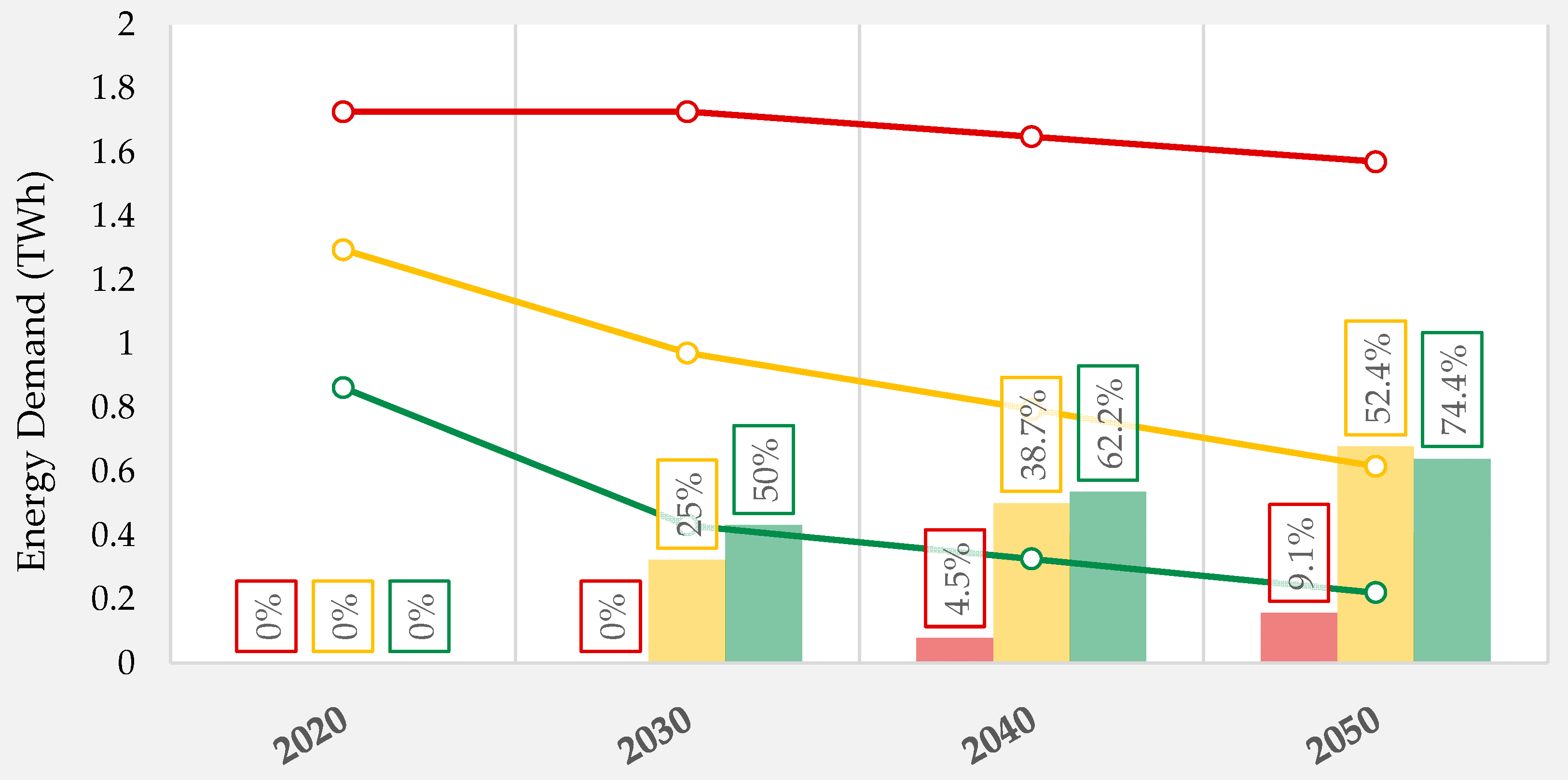
3.3. Industrialization Potential of Botanicals
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BMWi | Bundesministerium für Wirtschaft und Energie; English: Federal Ministry for Economic Affairs and Energy |
| CED | cumulative energy demand |
| CIP | cleaning-in-place |
| COG | cost of goods |
| DM | dried material |
| ED | energy demand |
| EH | electrolysis hydrogen |
| EHC | electrolysis hydrogen and carbon dioxide |
| EProMit | Energieeffiziente Prozesse durch Miktro-Trenntechnik; English: Energy-Efficient Processes by Micro-Separation Technology |
| GHG | greenhouse gas |
| GWP | global warming potential |
| HIC | hydrophobic interaction chromatography |
| IEX | ion-exchange |
| IF | improvement factor |
| LCA | life cycle assessment |
| mAb | monoclonal antibody |
| MP | methanol pyrolysis |
| PHWE | pressurized hot water extraction |
| SIP | sterilization-in-place |
| T2P2 | Total Traceless Phyto Plant |
| TPTP | Traceless Plant Traceless Production |
| TTBP | Total Traceless Biologics Plant |
| VCI | Verband der Chemischen Industrie e.V.; English: German Chemical Industry Association |
| WE | water electrolysis |
| WFI | water for injection |
References
- Umweltbundesamt. National trend tables for greenhouse gas emissions. In The German Climate Protection Act; Umweltbundesamt: Dessau-Rosslau, Germany, 2020. [Google Scholar]
- Bundesministerium für Umwelt, Naturschutz und Nukleare Sicherheit. Gesetz zur Einführung eines Bundes-Klimaschutzgesetzes und zur Änderung Weiterer Vorschriften. 2019. Available online: https://www.bmu.de/gesetz/bundes-klimaschutzgesetz/ (accessed on 1 June 2020).
- Verband der chemischen Industrie e.V. Energiestatistik in Daten und Fakten; Verband der Chemischen Industrie e.V.: Frankfurt, Germany, 2020. [Google Scholar]
- Energieintensiven Industrien Deutschland (EID). Die Energieintensiven Industrien in Deutschland: Impulsgeber für den Industriestandort Deutschland; EID: Berlin, Germany, 2020. [Google Scholar]
- Deutschland in Zahlen (DIZ); Institut der Deutschen Wirtschaft Köln Medien GmbH (Eds.) Available online: https://www.deutschlandinzahlen.de/ (accessed on 1 June 2020).
- Destatis. Luftemissionen: Deutschland, Jahre, Luftemissionsart, Produktionsbereiche, Wiesbaden, Germany. Statistisches Bundesamt; Data Licence by-2-0. Available online: https://www-genesis.destatis.de/genesis/online?operation=sprachwechsel&language=en (accessed on 1 June 2020).
- Destatis. Energieverbrauch der Betriebe im Verarbeitenden Gewerbe: Deutschland, Jahre, Nutzung des Energieverbrauchs, Wirtschaftszweige, Energieträger, Wiesbaden, Germany. Statistisches Bundesamt; Data Licence by-2-0. Available online: https://www-genesis.destatis.de/genesis/online?operation=sprachwechsel&language=en (accessed on 1 June 2020).
- BASF. Our Climate Protection Goal. 2020. Available online: https://www.basf.com/global/en/who-we-are/sustainability/we-produce-safely-and-efficiently/energy-and-climate-protection/climate-protection-goals.html (accessed on 1 June 2020).
- LANXESS. LANXESS to Become Climate Neutral by 2040. 2019. Available online: https://lanxess.com/en/Media/Press-Releases/2019/11/LANXESS-to-become-climate-neutral-by-2040 (accessed on 1 June 2020).
- Gerbert, P.; Herhold, P.; Burchardt, J.; Schönberger, S.; Rechenmacher, F.; Kirchner, A.; Kemmler, A.; Wünsch, M. Klimapfade für Deutschland; Boston Consulting Group GmbH (BCG): München, Germany, 2018. [Google Scholar]
- Becks, H.; Gelbke, H. Die Ökoeffizienz-Analyse nach BASF. TATuP-Zeitschrift für Technikfolgenabschätzung in Theorie und Praxis. 2001, 10, 34–39. [Google Scholar] [CrossRef]
- Burger, J.; Asprion, N.; Blagov, S.; Böttcher, R.; Nowak, U.; Bortz, M.; Welke, R.; Küfer, K.-H.; Hasse, H. Multi-Objective Optimization and Decision Support in Process Engineering—Implementation and Application. Chem. Ing. Tech. 2014, 86, 1065–1072. [Google Scholar] [CrossRef]
- Linnhoff March. Introduction to Pinch Technology. Available online: https://www.ou.edu/class/che-design/a-design/Introduction%20to%20Pinch%20Technology-LinhoffMarch.pdf (accessed on 1 June 2020).
- Geres, R.; Kohn, A.; Lenz, S.; Ausfelder, F.; Bazzanella, A.M.; Möller, A. Roadmap Chemie 2050—Auf dem Weg zu einer Treibhausgasneutralen Chemischen Industrie in Deutschland; DECHEMA Gesellschaft für Chemische Technik und Biotetechnologie e.V.: Frankfurt, Germany, 2019. [Google Scholar]
- Verband der Chemischen Industrie e.V. (VCI) (Ed.) Available online: https://www.vci.de (accessed on 1 June 2020).
- Boehringer Ingelheim. Klimaschutz: Urkunde von Umweltministerin für Boehringer Ingelheim. Available online: https://www.boehringer-ingelheim.de/pressemitteilung/klimaschutz-urkunde-von-umweltministerin-fuer-boehringer-ingelheim (accessed on 1 June 2020).
- Boehringer Ingelheim. Bericht im Rahmen der Wirtschaftsinititative Nachhaltigkeit (WIN) Baden-Württemberg: Berichtsjahr 2016. Available online: https://www.win-bw.com/fileadmin/downloads/2_charta/berichte/Boehringer_Ingelheim_Nachhaltigkeitsbericht_2016.pdf (accessed on 1 June 2020).
- Positionspapier der Bundesregierung. Zwischenevaluierung von Horizont 2020; Federal Ministry of Education and Research (BMBF): Bonn, Germany, 2017. [Google Scholar]
- Renn, O. Ein Kompass für die Energiewende: Das Kopernikus-Projekt Energiewende-Navigationssystem (ENavi) ist gestartet. GAIA Ecol. Perspect. Sci. Soc. 2017, 26, 68–69. [Google Scholar] [CrossRef]
- Molins, F.; Schweers, E. Zu SunFuels aus CO2 in einer Ein-Schritt-Fotosynthese. Chem. Ing. Tech. 2018, 90, 1143. [Google Scholar] [CrossRef]
- Bundesministerium für Wirtschaft und Energie. Bekanntmachung Forschungsförderung im 6. Energieforschungsprogramm “Forschung Für Eine Umweltschonende, Zuverlässige Und Bezahlbare Energieversorgung”; Bundesministerium für Wirtschaft und Energie: Berlin, Germany, 2014. [Google Scholar]
- Sattler, K. Thermische Trennverfahren; John Wiley & Sons: Hoboken, NJ, USA, 2012; ISBN 978-3-527-30243-7. [Google Scholar]
- Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons: Hoboken, NJ, USA, 2000; ISBN 9780471238966.
- Sommerfeld, S.; Strube, J. Challenges in biotechnology production—Generic processes and process optimization for monoclonal antibodies. Chem. Eng. Process. Process Intensif. 2005, 44, 1123–1137. [Google Scholar] [CrossRef]
- Strube, J. Energieeffiziente Prozesse durch Mikro-Trenntechnik (EProMit): Schlussbericht zum Forschungsvorhaben; Technische Informationsbibliothek: Hannover, Germany, 2015. [Google Scholar]
- FDA. Guidance for Industry. PAT—A Framework for Innovative Pharmaceutical Development, Manufacturing, and Quality Assurance. 2004. Available online: https://www.fda.gov/downloads/drugs/guidances/ucm070305.pdf (accessed on 19 February 2018).
- Uhlenbrock, L.; Sixt, M.; Strube, J. Quality-by-Design (QbD) process evaluation for phytopharmaceuticals on the example of 10-deacetylbaccatin III from yew. Resour. Effic. Technol. 2017, 3, 137–143. [Google Scholar] [CrossRef]
- Sixt, M.; Strube, J. Systematic and Model-Assisted Evaluation of Solvent Based- or Pressurized Hot Water Extraction for the Extraction of Artemisinin from Artemisia annua L. Processes 2017, 5, 86. [Google Scholar] [CrossRef]
- Schmidt, A.; Sixt, M.; Huter, M.; Mestmäcker, F.; Strube, J. Systematic and Model-Assisted Process Design for the Extraction and Purification of Artemisinin from Artemisia annua L.—Part II: Model-Based Design of Agitated and Packed Columns for Multistage Extraction and Scrubbing. Processes 2018, 6, 179. [Google Scholar] [CrossRef]
- Mestmäcker, F.; Schmidt, A.; Huter, M.; Sixt, M.; Strube, J. Systematic and Model-Assisted Process Design for the Extraction and Purification of Artemisinin from Artemisia annua L.—Part III: Chromatographic Purification. Processes 2018, 6, 180. [Google Scholar] [CrossRef]
- Huter, M.; Schmidt, A.; Mestmäcker, F.; Sixt, M.; Strube, J. Systematic and Model-Assisted Process Design for the Extraction and Purification of Artemisinin from Artemisia annua L.—Part IV: Crystallization. Processes 2018, 6, 181. [Google Scholar] [CrossRef]
- Kornecki, M.; Schmidt, A.; Lohmann, L.; Huter, M.; Mestmäcker, F.; Klepzig, L.; Mouellef, L.; Zobel-Roos, S.; Strube, J. Accelerating Biomanufacturing by Modeling of Continuous Bioprocessing—Piloting Case Study of Monoclonal Antibody Manufacturing. Processes 2019, 7, 495. [Google Scholar] [CrossRef]
- Sixt, M.; Uhlenbrock, L.; Strube, J. Toward a Distinct and Quantitative Validation Method for Predictive Process Modelling—On the Example of Solid-Liquid Extraction Processes of Complex Plant Extracts. Processes 2018, 6, 66. [Google Scholar] [CrossRef]
- Kornecki, M.; Strube, J. Accelerating Biologics Manufacturing by Upstream Process Modelling. Processes 2019, 7, 166. [Google Scholar] [CrossRef]
- Cetinkaya, E.; Liu, N.; Simons, T.J.; Wallach, J. Petrochemicals 2030: Reinventing the Way to Win in a Changing Industry; McKinsey & Co: New York, NY, USA, 2018. [Google Scholar]
- Report of the Conference of the Parties on its twenty-first session, held in Paris from 30 November to 13 December 2015. Addendum. Part two: Action taken by the Conference of the Parties at its twenty-first session.
- Carbon Pollution Emission Guidelines for Existing Stationary Sources: Electric Utility Generating Units; 40 CFR Part 60. Available online: https://www.federalregister.gov/documents/2015/10/23/2015-22842/carbon-pollution-emission-guidelines-for-existing-stationary-sources-electric-utility-generating (accessed on 1 June 2020).
- Arbeitsgemeinschaft Energiebilanzen e.V. Auswertungstabellen zur Energiebilanz Deutschland. Available online: https://ag-energiebilanzen.de/10-0-Auswertungstabellen.html (accessed on 1 June 2020).
- Belkhir, L.; Elmeligi, A. Carbon footprint of the global pharmaceutical industry and relative impact of its major players. J. Clean. Prod. 2019, 214, 185–194. [Google Scholar] [CrossRef]
- Protocol, G.G. A Corporate Accounting and Reporting Standard; World Resources Institute and World Business Council for Sustainable Development: Geneva, Switzerland, 2004. [Google Scholar]
- Gao, Z.; Geng, Y.; Wu, R.; Chen, W.; Wu, F.; Tian, X. Analysis of energy-related CO2 emissions in China’s pharmaceutical industry and its driving forces. J. Clean. Prod. 2019, 223, 94–108. [Google Scholar] [CrossRef]
- Srai, J.S.; Badman, C.; Krumme, M.; Futran, M.; Johnston, C. Future Supply Chains Enabled by Continuous Processing—Opportunities and Challenges. May 20–21, 2014 Continuous Manufacturing Symposium. J. Pharm. Sci. 2015, 104, 840–849. [Google Scholar] [CrossRef] [PubMed]
- The Business Research Company. The Growing Pharmaceuticals Market: Expert Forecasts and Analysis. Available online: https://blog.marketresearch.com/the-growing-pharmaceuticals-market-expert-forecasts-and-analysis (accessed on 1 June 2020).
- Wenda, S.; Illner, S.; Mell, A.; Kragl, U. Industrial biotechnology—The future of green chemistry? Green Chem. 2011, 13, 3007–3047. [Google Scholar] [CrossRef]
- Pharmaceutical Online. Measuring COVID-19’s Impact on Availability of Drugs and API from India and China. Available online: https://www.pharmaceuticalonline.com/doc/measuring-covid-s-impact-on-availability-of-drugs-and-api-from-india-and-china-0001 (accessed on 1 June 2020).
- Pietrzykowski, M.; Flanagan, W.; Pizzi, V.; Brown, A.; Sinclair, A.; Monge, M. An environmental life cycle assessment comparison of single-use and conventional process technology for the production of monoclonal antibodies. J. Clean. Prod. 2013, 41, 150–162. [Google Scholar] [CrossRef]
- Bethir, L. Big Pharma Emits More Greenhouse Gases than the Automotive Industry. Available online: https://phys.org/news/2019-05-big-pharma-emits-greenhouse-gases.html (accessed on 1 June 2020).
- Grote, F.; Ditz, R.; Strube, J. Downstream of downstream processing: Development of recycling strategies for biopharmaceutical processes. J. Chem. Technol. Biotechnol. 2012, 87, 481–497. [Google Scholar] [CrossRef]
- Ramasamy, S.V.; Titchener-Hooker, N.J.; Lettieri, P. Life cycle assessment as a tool to support decision making in the biopharmaceutical industry: Considerations and challenges. Food Bioprod. Process. 2015, 94, 297–305. [Google Scholar] [CrossRef]
- Idris, A.; Chua, G.K.; Othman, M.R. Incorporating potential environmental impact from water for injection in environmental assessment of monoclonal antibody production. Chem. Eng. Res. Des. 2016, 109, 430–442. [Google Scholar] [CrossRef]
- Siegert, M.-W.; Lehmann, A.; Emara, Y.; Finkbeiner, M. Harmonized rules for future LCAs on pharmaceutical products and processes. Int. J. Life Cycle Assess. 2019, 24, 1040–1057. [Google Scholar] [CrossRef]
- Lalor, F.; Fitzpatrick, J.; Sage, C.; Byrne, E. Sustainability in the biopharmaceutical industry: Seeking a holistic perspective. Biotechnol. Adv. 2019, 37, 698–707. [Google Scholar] [CrossRef] [PubMed]
- Shirahata, H.; Hirao, M.; Sugiyama, H. Multiobjective decision-support tools for the choice between single-use and multi-use technologies in sterile filling of biopharmaceuticals. Comput. Chem. Eng. 2019, 122, 114–128. [Google Scholar] [CrossRef]
- Marques, C.M.; Moniz, S.; de Sousa, J.P.; Barbosa-Povoa, A.P.; Reklaitis, G. Decision-support challenges in the chemical-pharmaceutical industry: Findings and future research directions. Comput. Chem. Eng. 2020, 134, 106672. [Google Scholar] [CrossRef]
- Ho, S.V.; McLaughlin, J.M.; Cue, B.W.; Dunn, P.J. Environmental considerations in biologics manufacturing. Green Chem. 2010, 12, 755. [Google Scholar] [CrossRef]
- Pietrzykowski, V.P.; Flanagan, W.; Pietrzykowski, M.; Brown, A.; Sinclair, A.; Monge, M. An Environmental Lifecycle Assessment of Single-Use and Conventional Process Technology: Comprehensive Environmental Impacts. BioPharm Int. 2014, 27, 3. [Google Scholar]
- Bunnak, P.; Allmendinger, R.; Ramasamy, S.V.; Lettieri, P.; Titchener-Hooker, N.J. Life-cycle and cost of goods assessment of fed-batch and perfusion-based manufacturing processes for mAbs. Biotechnol. Prog. 2016, 32, 1324–1335. [Google Scholar] [CrossRef]
- Renteria Gamiz, A.G.; de Soete, W.; Heirman, B.; Dahlin, P.; de Meester, S.; Dewulf, J. Environmental sustainability assessment of the manufacturing process of a biological active pharmaceutical ingredient. J. Chem. Technol. Biotechnol. 2019, 94, 1937–1944. [Google Scholar] [CrossRef]
- Bundesverband der Pharmazeutischen Industrie e.V. Pharma-Daten 2019. Berlin. Available online: https://www.bpi.de/de/service/pharma-daten (accessed on 1 June 2020).
- Bart, H.J.; Bäcker, W.; Bischoff, F.; Godecke, R.; Johannisbau, W.; Jordan, V.; Stockfleth, R.; Strube, J.; Wiesmet, V. Phytoextrakte—Produkte und Prozesse: Vorschlag für einen neuen, fachübergreifenden Forschungsschwerpunkt; Dechema e.V., Ed.; Dechema e.V.: Frankfurt am Main, Germany, 2012. [Google Scholar]
- Dar, R.A.; Shahnawaz, M.; Rasool, S.; Qazi, P.H. Natural product medicines: A literature update. J. Phytopharm. 2017, 6, 349–351. [Google Scholar]
- FNR. Fachagentur Nachwachsende Rohstoffe: Ein Projektträger des Bundesministeriums für Ernährung und Landwirtschaft. Available online: https://www.fnr.de/ (accessed on 1 June 2020).
- proplanta. Das Informationszentrum für Landwirtschaft. Available online: https://www.proplanta.de/ (accessed on 1 June 2020).
- Zafeiridou, M.; Hopkinson, N.S.; Voulvoulis, N. Cigarette Smoking: An Assessment of Tobacco’s Global Environmental Footprint Across Its Entire Supply Chain. Environ. Sci. Technol. 2018, 52, 8087–8094. [Google Scholar] [CrossRef] [PubMed]
- Kraatz, S.; Sinistore, J.C.; Reinemann, D.J. Energy intensity and global warming potential of corn grain ethanol production in Wisconsin (USA). Food Energy Secur. 2013, 2, 207–219. [Google Scholar] [CrossRef]
- Tongwane, M.; Mdlambuzi, T.; Moeletsi, M.; Tsubo, M.; Mliswa, V.; Grootboom, L. Greenhouse gas emissions from different crop production and management practices in South Africa. Environ. Dev. 2016, 19, 23–35. [Google Scholar] [CrossRef]
- Leibniz-Institut für Agrartechnik und Bioökonomie e.V. Leitfaden Trocknung von Arznei- und Gewürzpflanzen; Leibniz-Institut für Agrartechnik und Bioökonomie e.V.: Potsdam, Germany, 2017. [Google Scholar]
- Uhlenbrock, L.; Ditz, R.; Strube, J. Process Engineering Accelerating an Economic Industrialization Towards a Bio-Based World. Molecules 2019, 24, 1853. [Google Scholar] [CrossRef] [PubMed]
- Sixt, M.; Strube, J. Systematic Design and Evaluation of an Extraction Process for Traditionally Used Herbal Medicine on the Example of Hawthorn (Crataegus monogyna JACQ.). Processes 2018, 6, 73. [Google Scholar] [CrossRef]
- Sixt, M.; Strube, J. Pressurized hot water extraction of 10-deacetylbaccatin III from yew for industrial application. Resour. Effic. Technol. 2017, 3, 177–186. [Google Scholar] [CrossRef]
- Sixt, M.; Schmidt, A.; Mestmäcker, F.; Huter, M.; Uhlenbrock, L.; Strube, J. Systematic and Model-Assisted Process Design for the Extraction and Purification of Artemisinin from Artemisia annua L.—Part I: Conceptual Process Design and Cost Estimation. Processes 2018, 6, 161. [Google Scholar] [CrossRef]
- BASF. BASF Online Report 2019. Available online: https://report.basf.com/2019/en/ (accessed on 1 June 2020).
- Bayer. Sustainability Report 2019. Bayer AG, Ed.; Available online: https://www.investor.bayer.de/en/reports/sustainability-reports/ (accessed on 1 June 2020).
- Boehringer Ingelheim (Ed.) Corporate Social Responsibility Report. Available online: http://csrreport.us.boehringer-ingelheim.com/ (accessed on 1 June 2020).
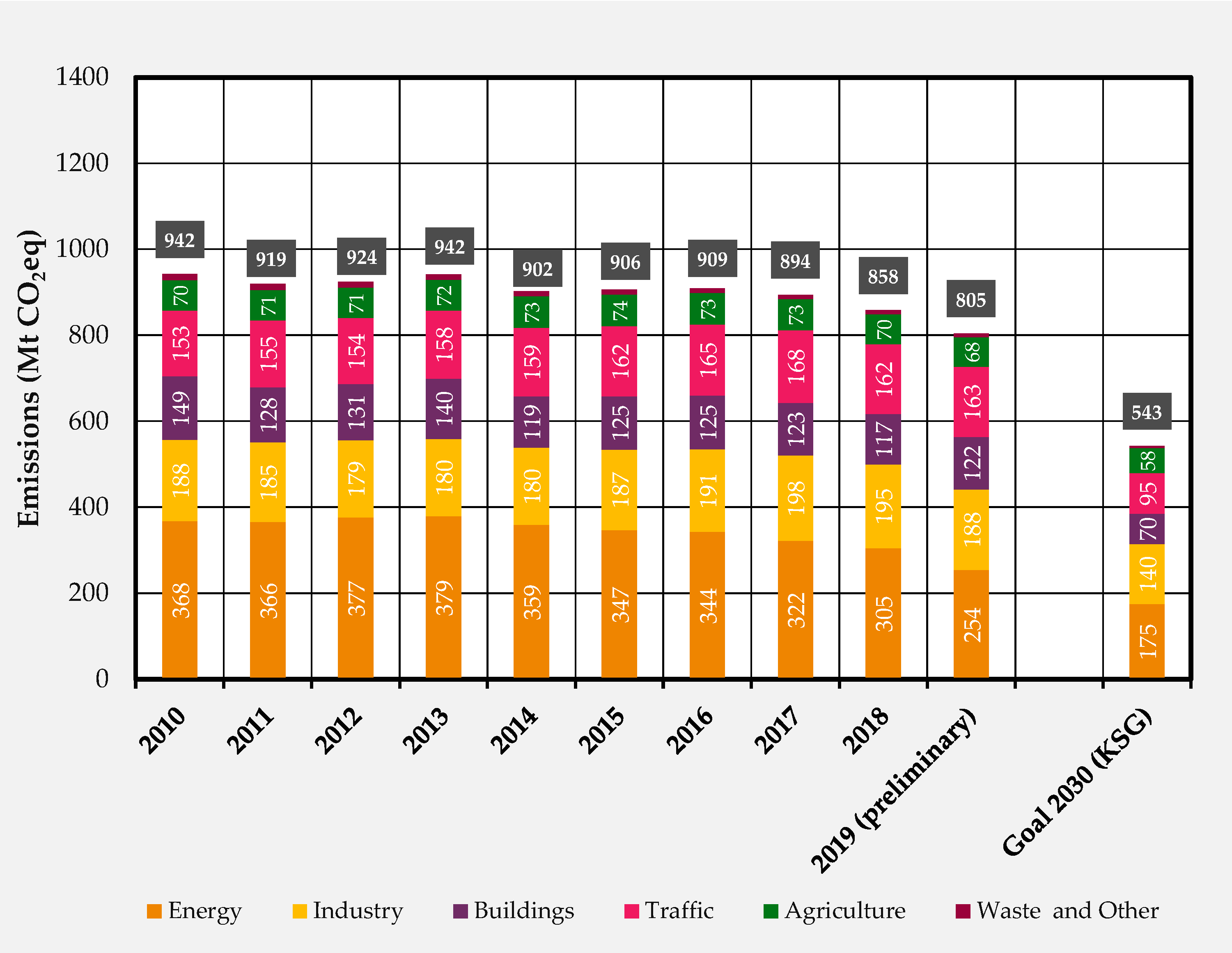
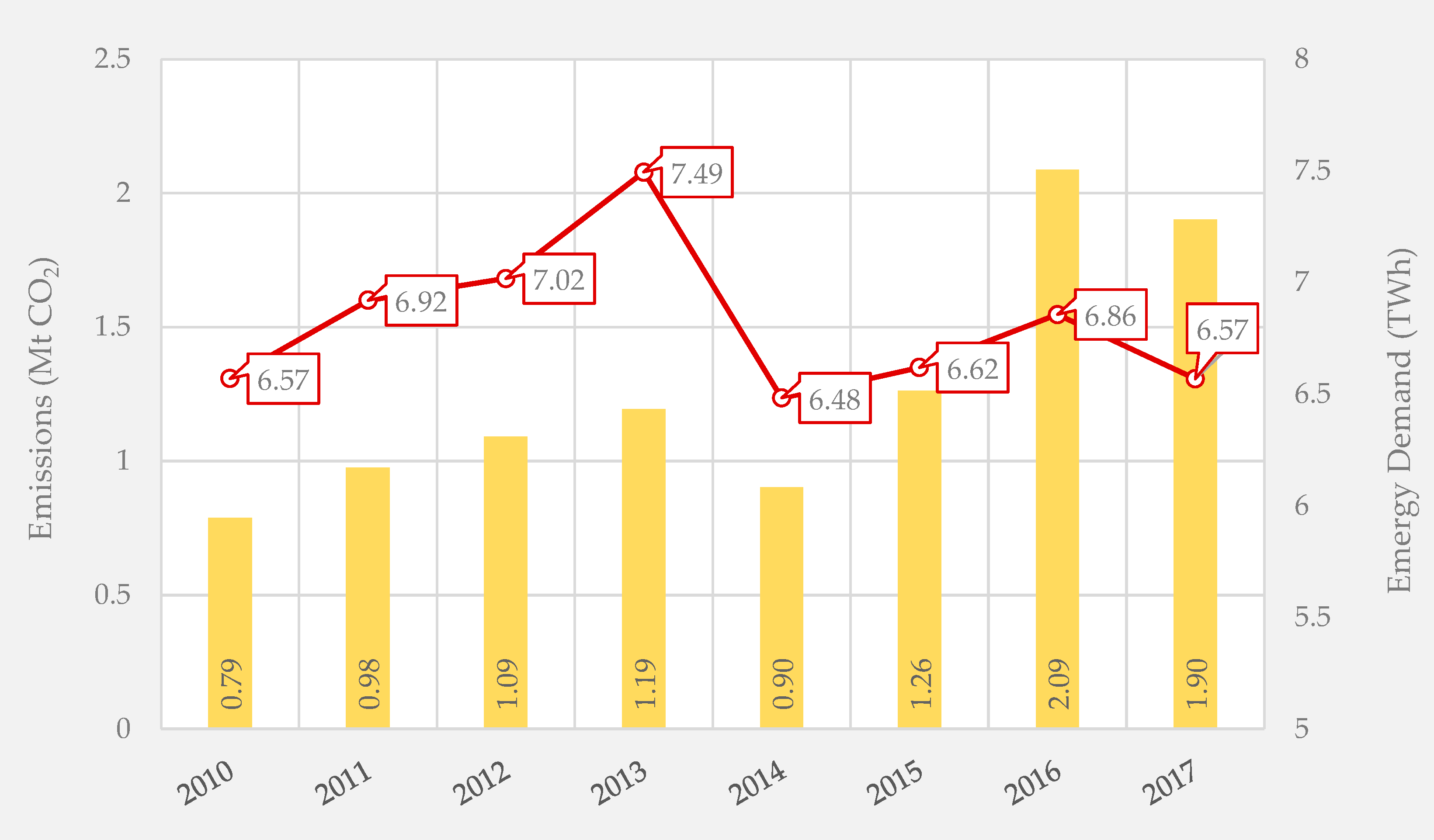
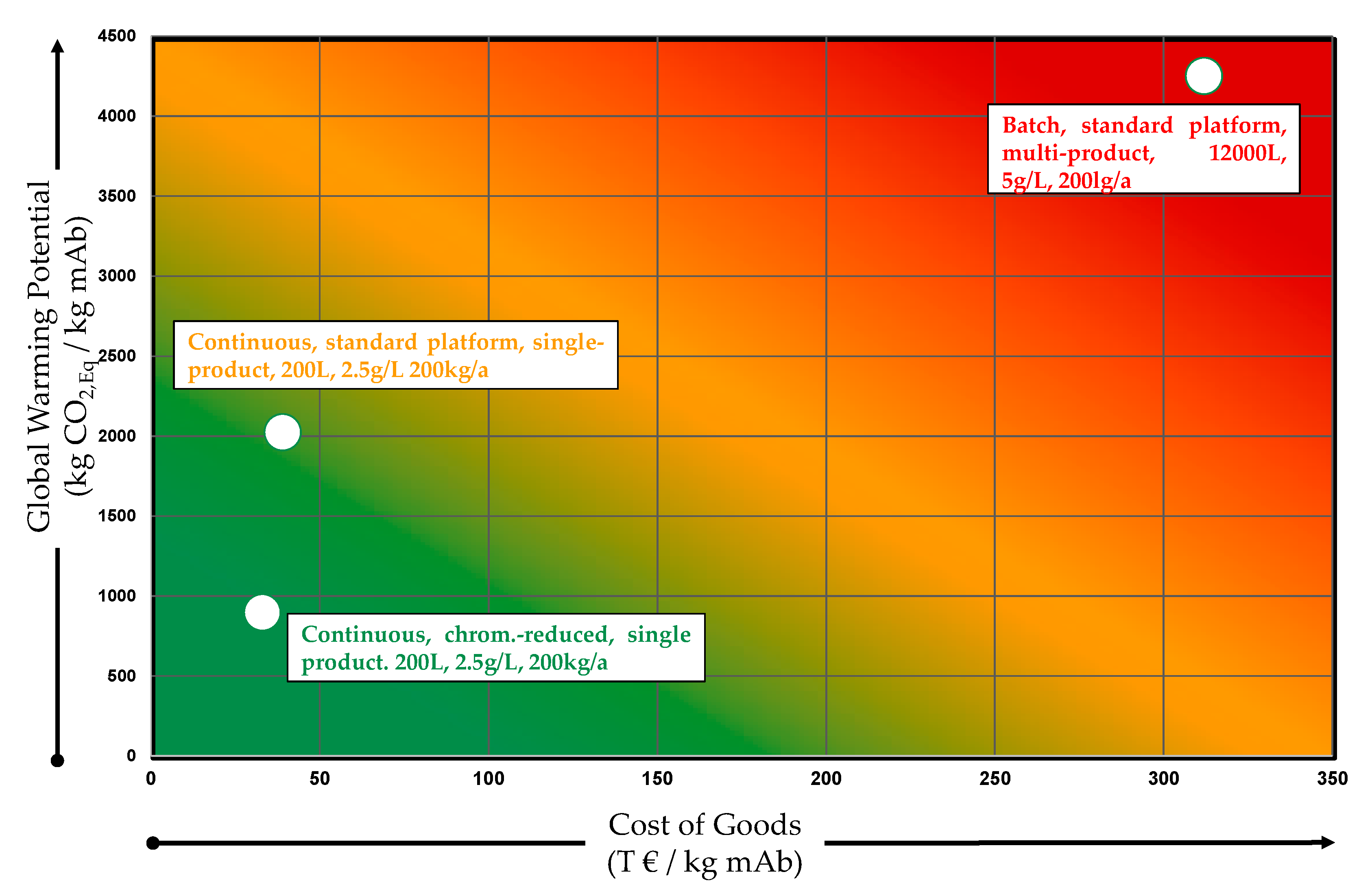
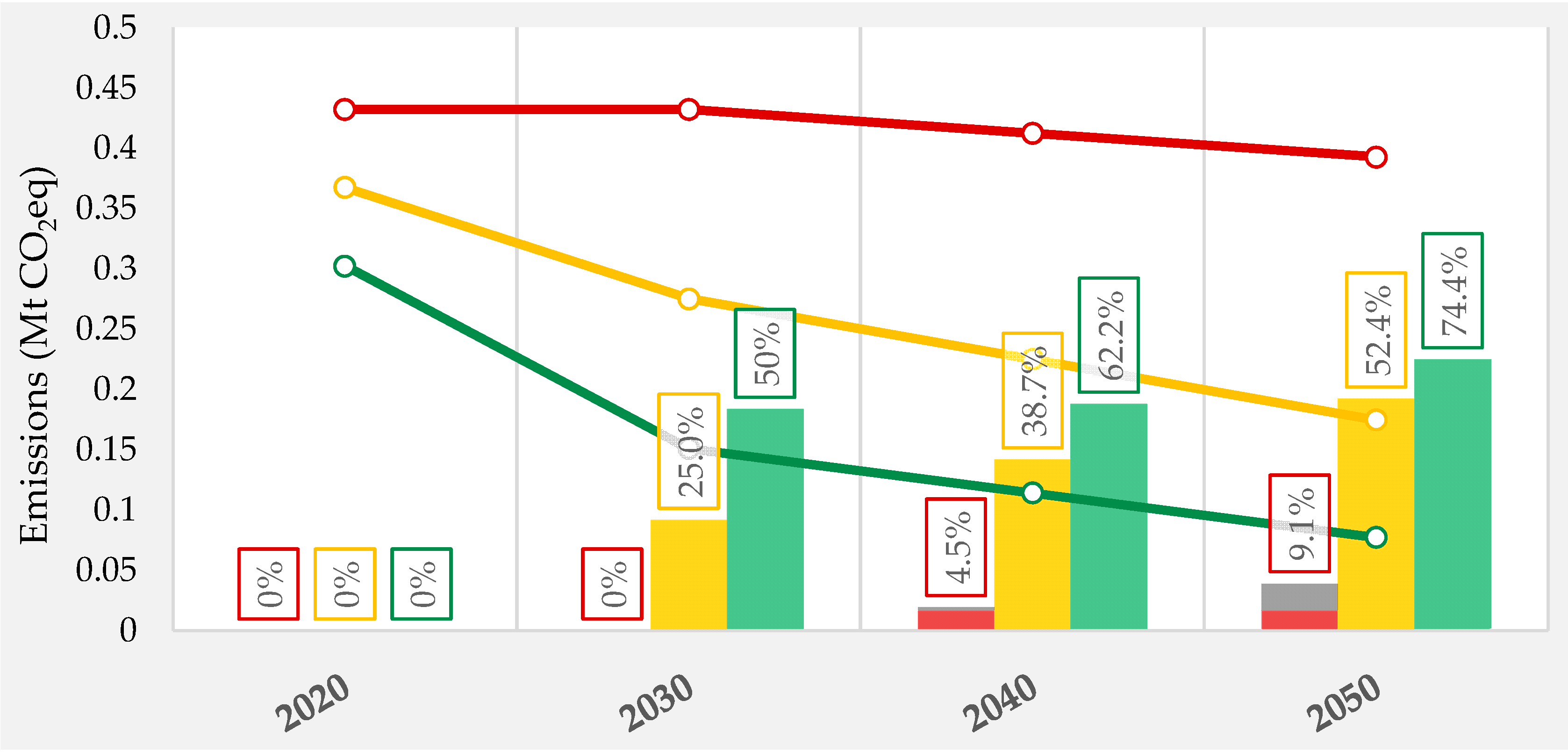
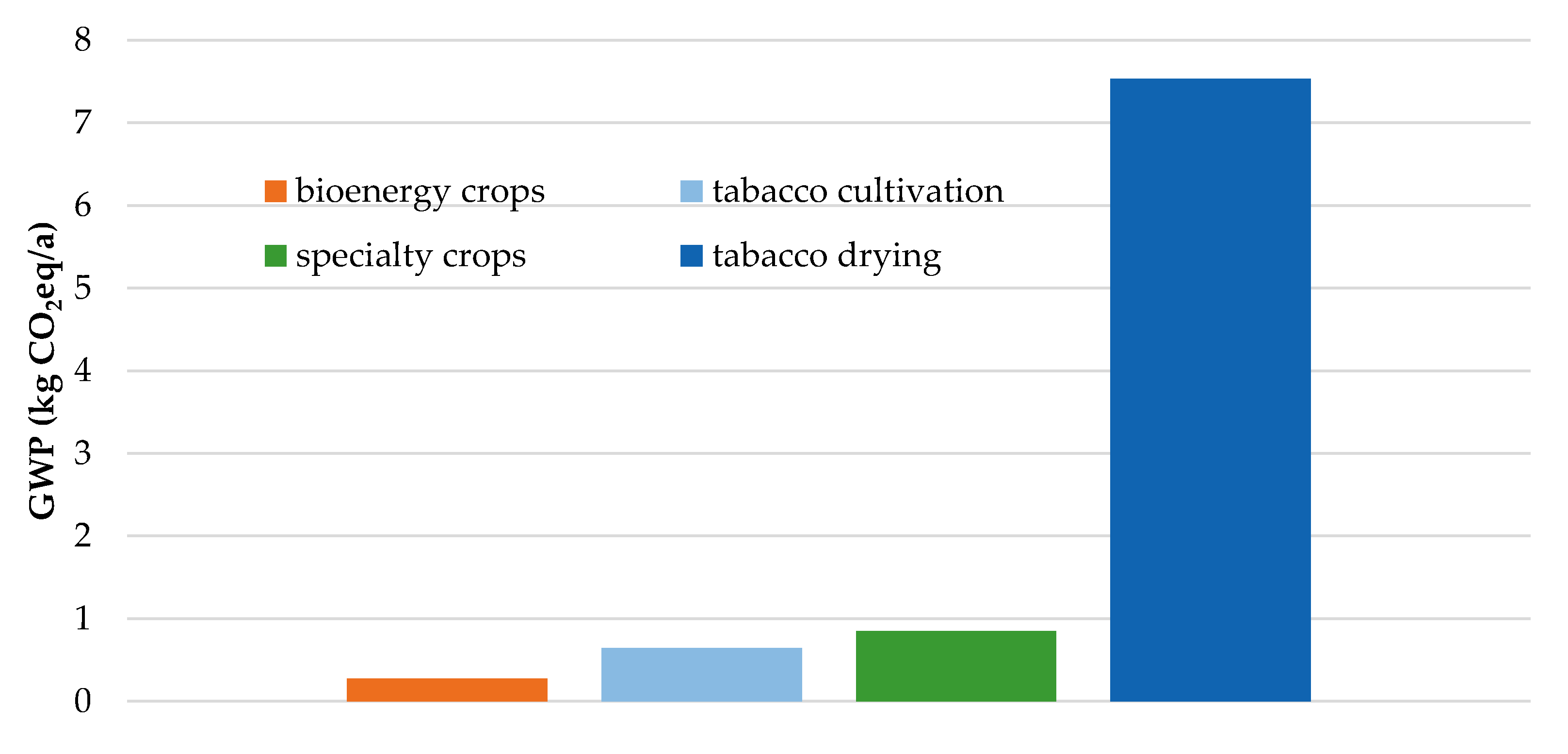
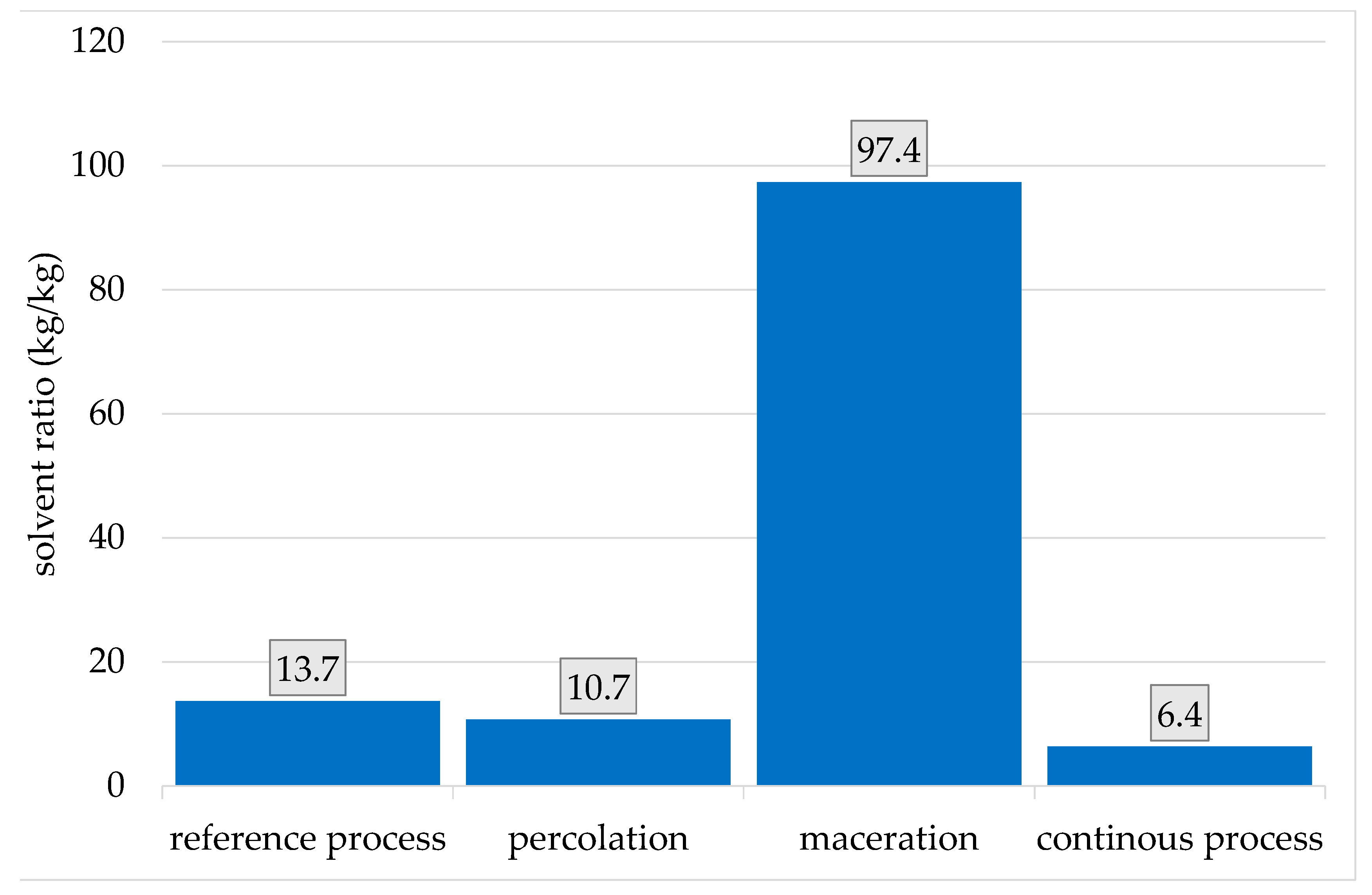
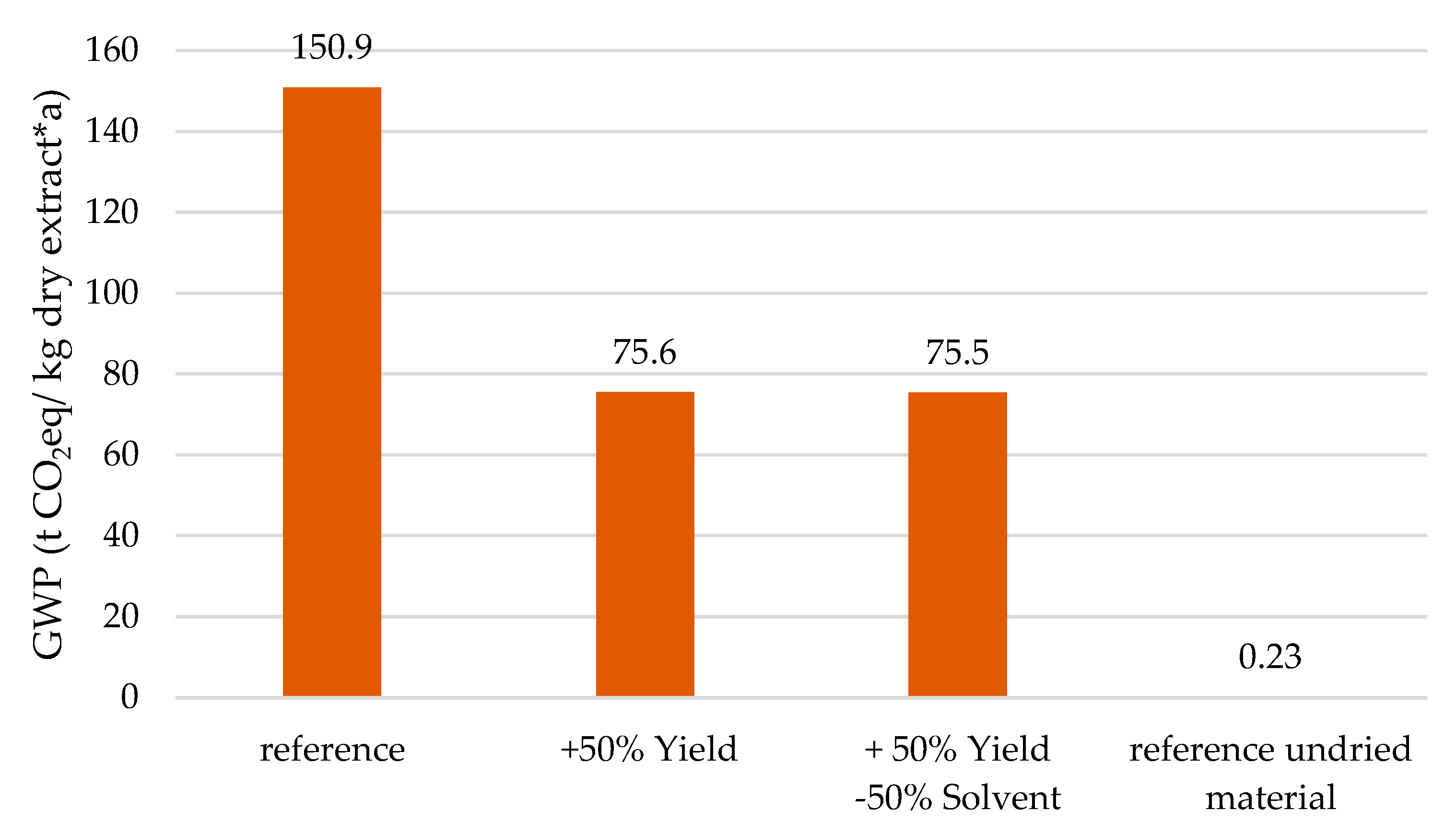
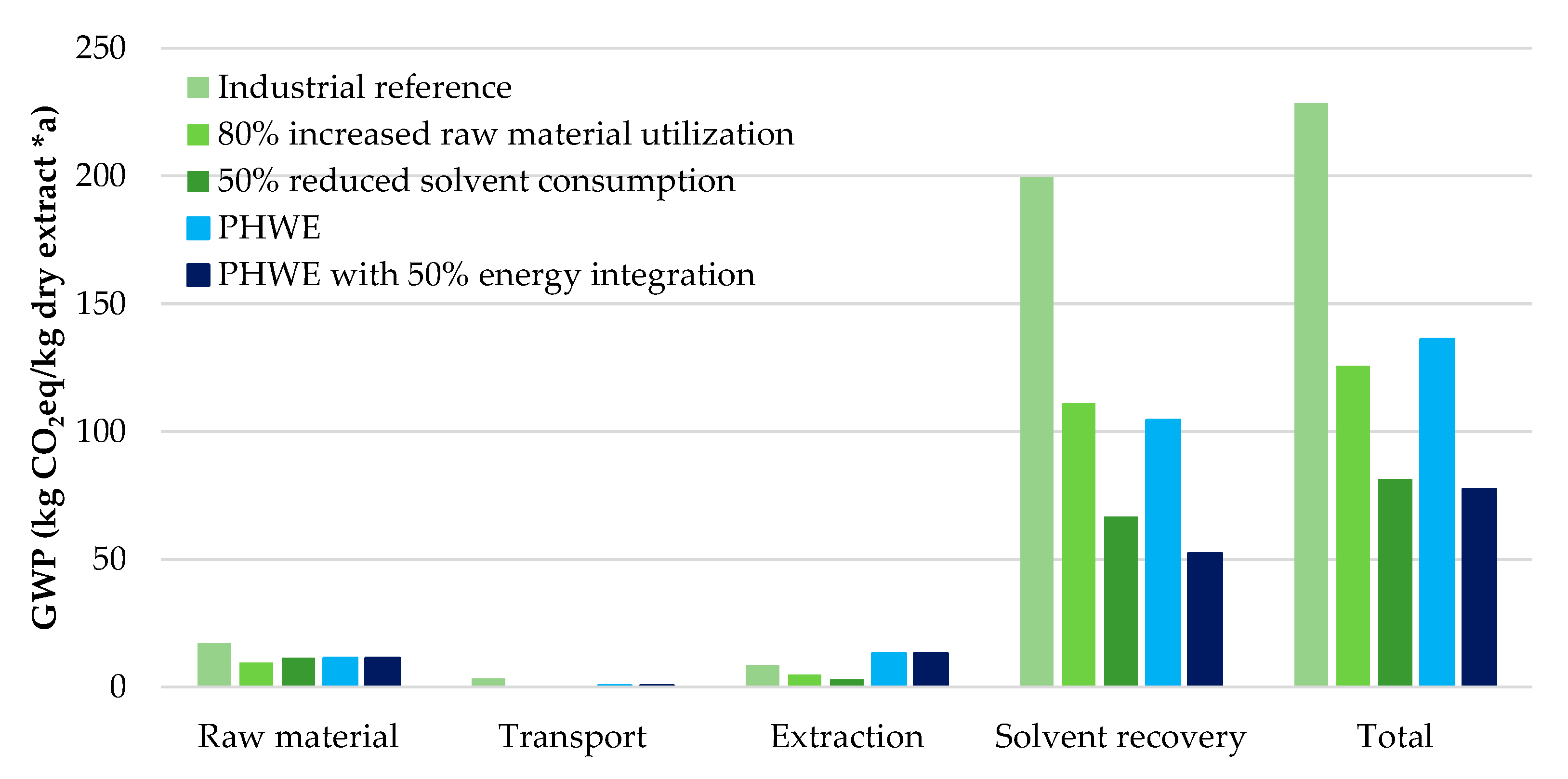
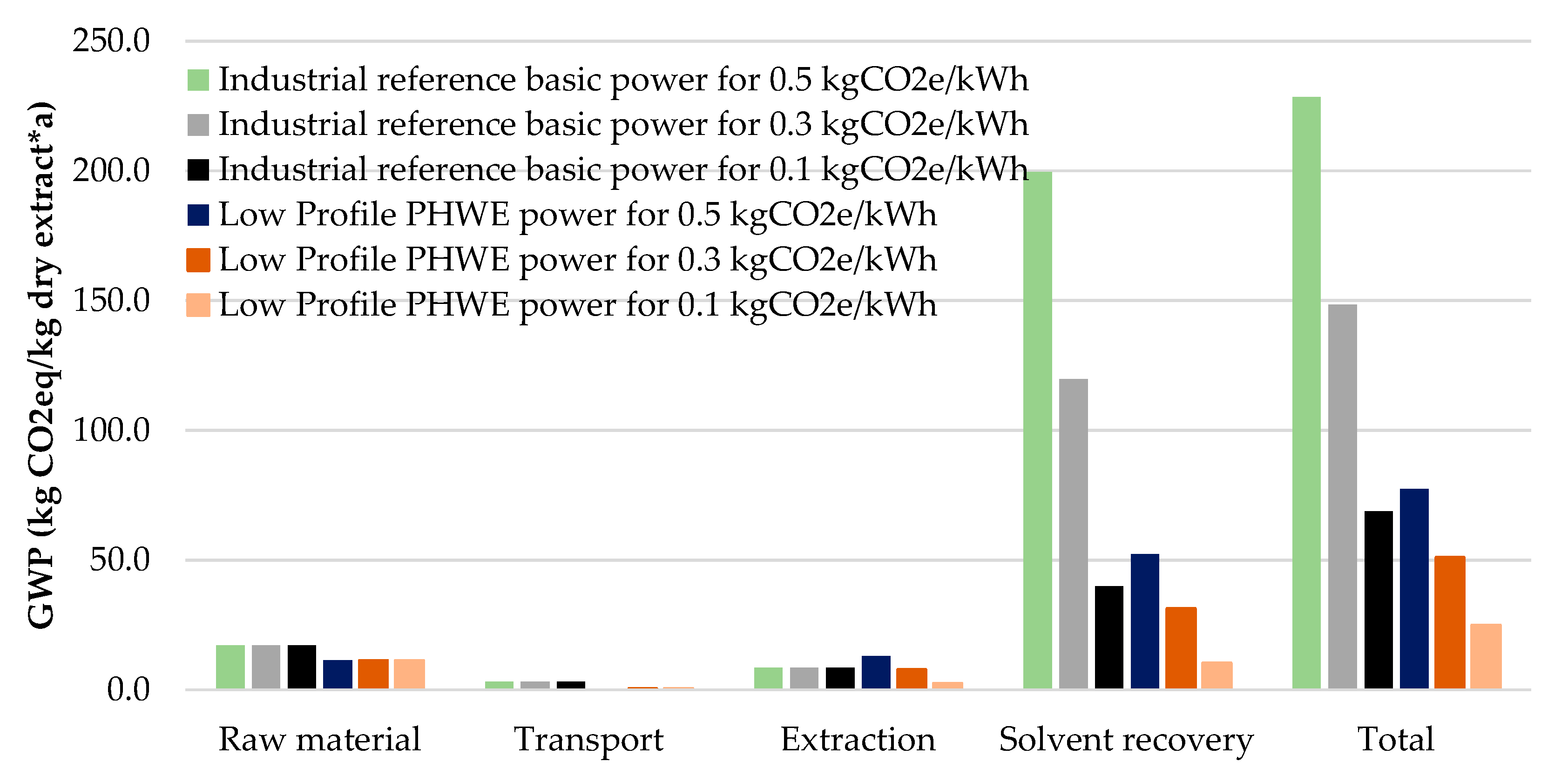
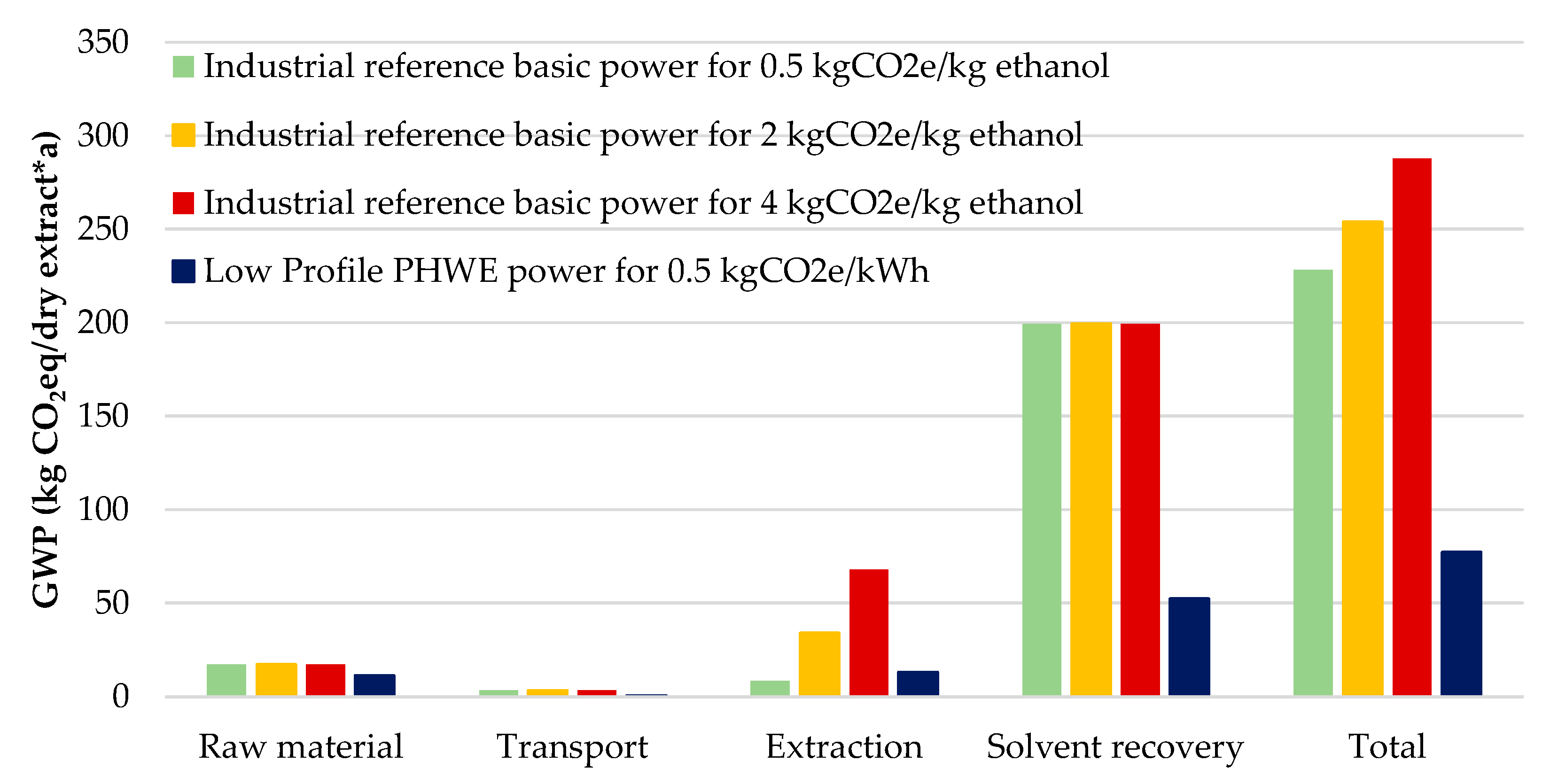
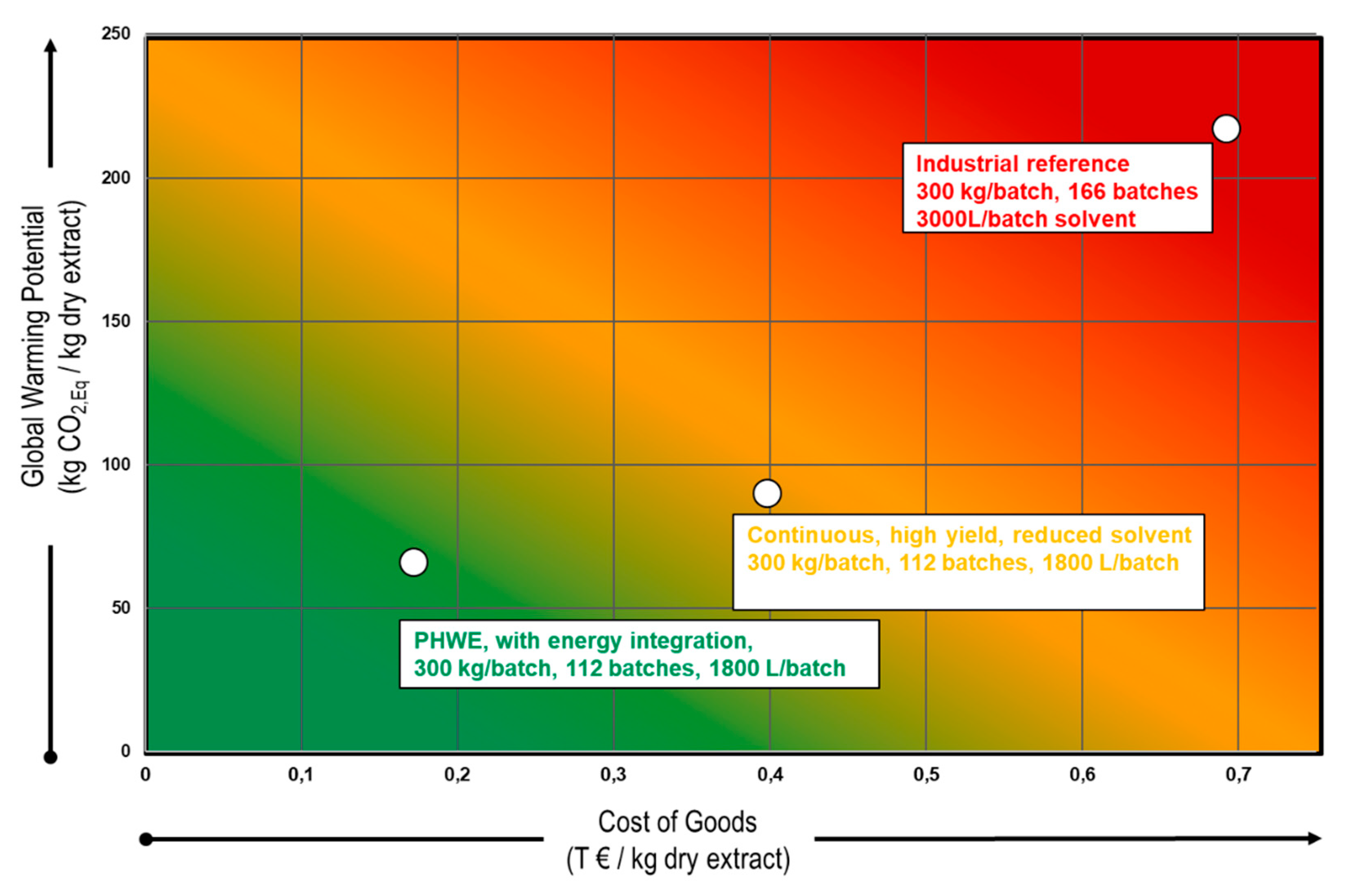
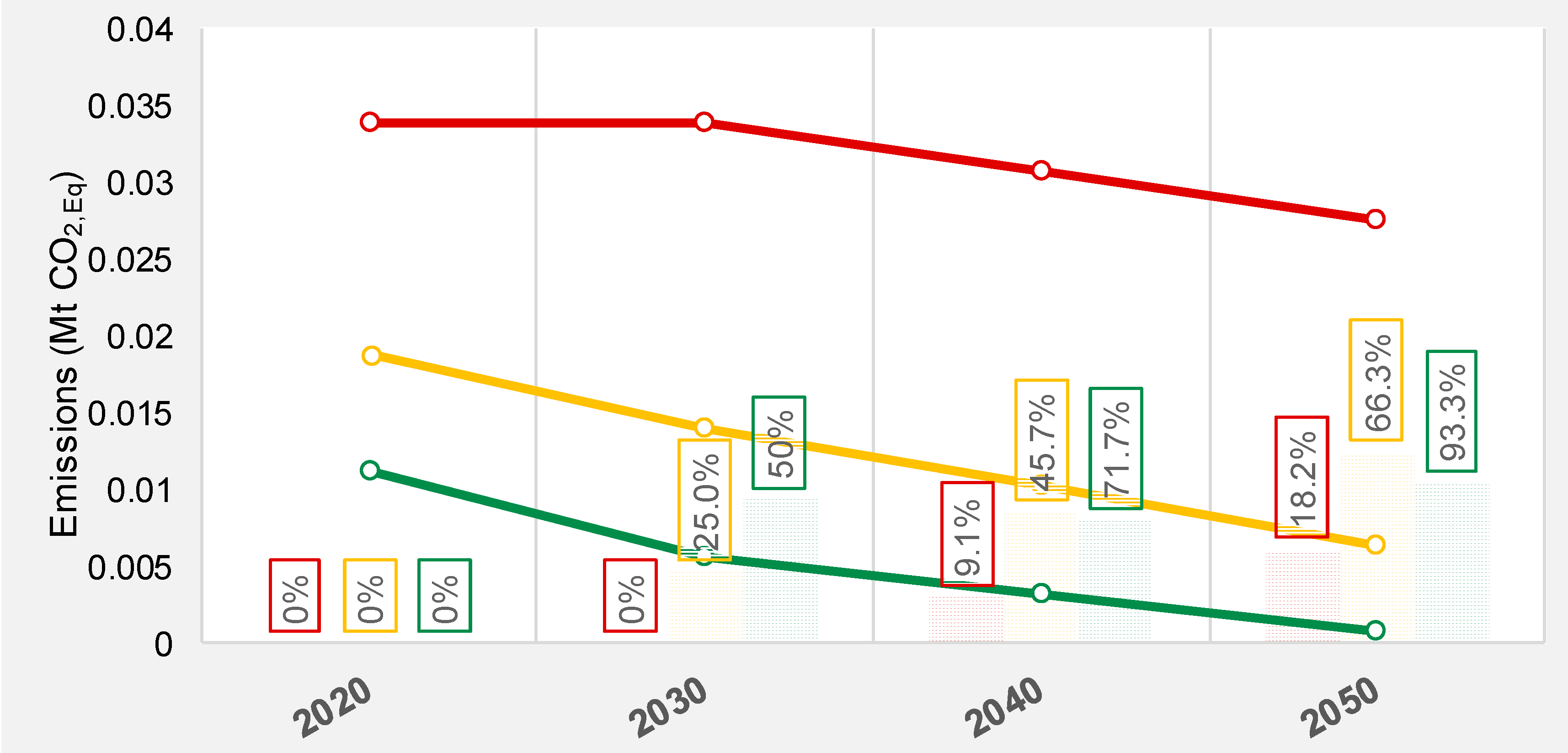
| Product | GWP (kg/kg) | ED (MWh/t) |
|---|---|---|
| Hydrogen WE | 24.4 | 51.60 |
| Hydrogen MP | 4.82 | 9.50 |
| Ammonia EH | 5.16 | 9.17 |
| Ammonia MP | 1.67 | 3.40 |
| Urea | 0.46 | 0.97 |
| MeOH EHC | 5.23 | 9.52 |
| MeOH MP | 1.62 | 3.30 |
| Range | GWP (t/kg) | CED (MWh/kg) | IFyield (%) | IFWFI (%) | GWPWFI (%) | CEDWFI (%) |
|---|---|---|---|---|---|---|
| Min | 3.50 | 10 | 10 | 50 | 50 | 50 |
| Avg | 4.25 | 15 | 20 | 60 | 70 | 70 |
| Max | 5.00 | 20 | 30 | 70 | 90 | 90 |
| Case | GWP (Mt CO2eq) | CED (TWh) |
|---|---|---|
| Best | 0.30 | 0.09 |
| Avg | 0.37 | 1.3 |
| Worst | 0.43 | 1.7 |
| Optimization Pathway | Yield | Solvent | Temperature | Energy Recovery |
|---|---|---|---|---|
| Reference | 0% | 0% | 20 °C | 0% |
| Increased Yield | +80% | 0% | 20 °C | 0% |
| Decreased Solvent | +50% | −50% | 20 °C | 0% |
| PHWE | +50% | −50% | 120 °C | 0% |
| PHWE with energy integration | +50% | −50% | 120 °C | 50% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmidt, A.; Uhlenbrock, L.; Strube, J. Technical Potential for Energy and GWP Reduction in Chemical–Pharmaceutical Industry in Germany and EU—Focused on Biologics and Botanicals Manufacturing. Processes 2020, 8, 818. https://doi.org/10.3390/pr8070818
Schmidt A, Uhlenbrock L, Strube J. Technical Potential for Energy and GWP Reduction in Chemical–Pharmaceutical Industry in Germany and EU—Focused on Biologics and Botanicals Manufacturing. Processes. 2020; 8(7):818. https://doi.org/10.3390/pr8070818
Chicago/Turabian StyleSchmidt, Axel, Lukas Uhlenbrock, and Jochen Strube. 2020. "Technical Potential for Energy and GWP Reduction in Chemical–Pharmaceutical Industry in Germany and EU—Focused on Biologics and Botanicals Manufacturing" Processes 8, no. 7: 818. https://doi.org/10.3390/pr8070818
APA StyleSchmidt, A., Uhlenbrock, L., & Strube, J. (2020). Technical Potential for Energy and GWP Reduction in Chemical–Pharmaceutical Industry in Germany and EU—Focused on Biologics and Botanicals Manufacturing. Processes, 8(7), 818. https://doi.org/10.3390/pr8070818






