Valorization of Globe Artichoke (Cynara scolymus) Agro-Industrial Discards, Obtaining an Extract with a Selective Effect on Viability of Cancer Cell Lines
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material and Reagents
2.2. Hydroalcoholic Extraction
2.3. Bioactive Compound Determination
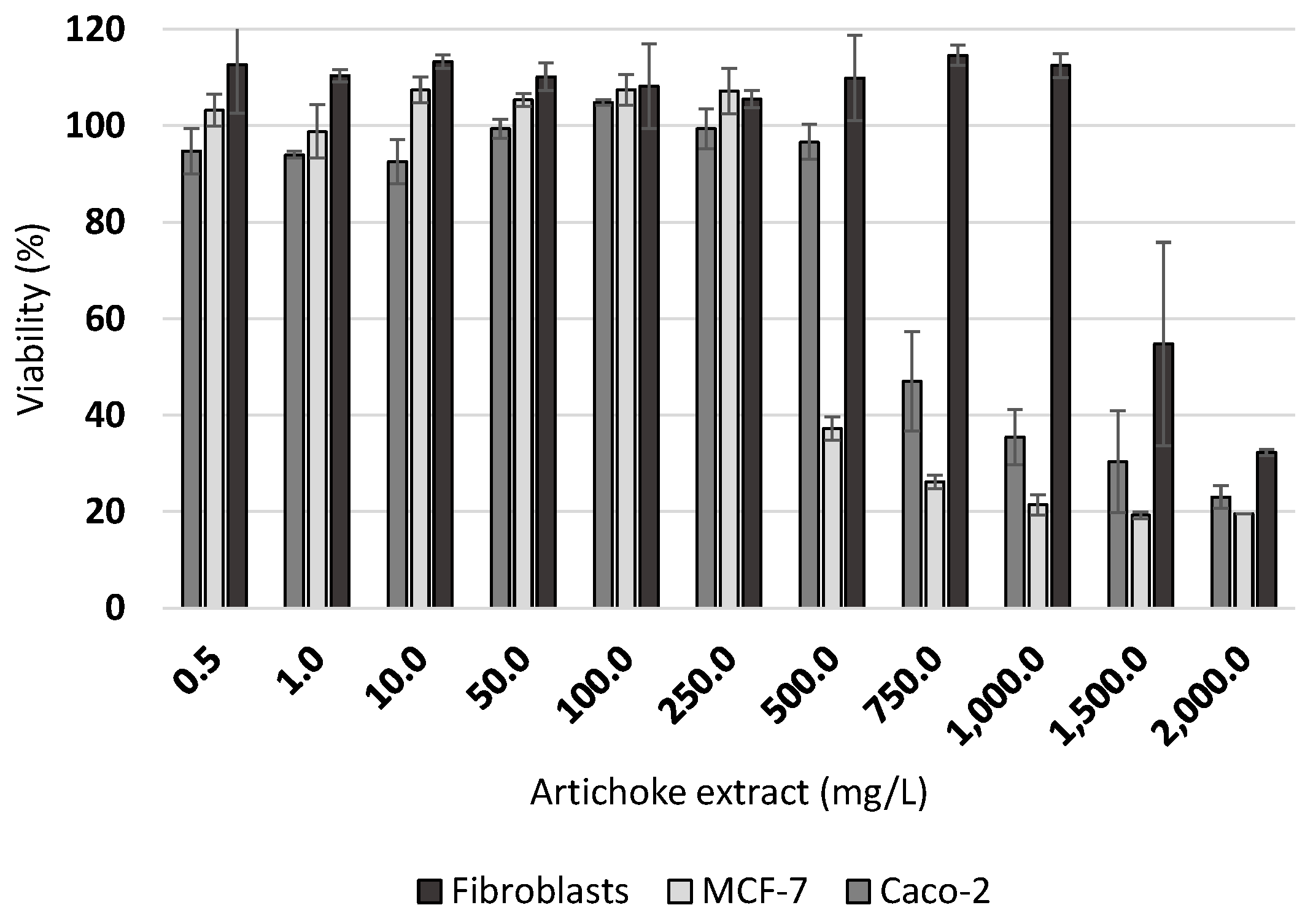
2.4. Carcinogenic Cell Lines Studies
2.5. Statistical Analysis
3. Results
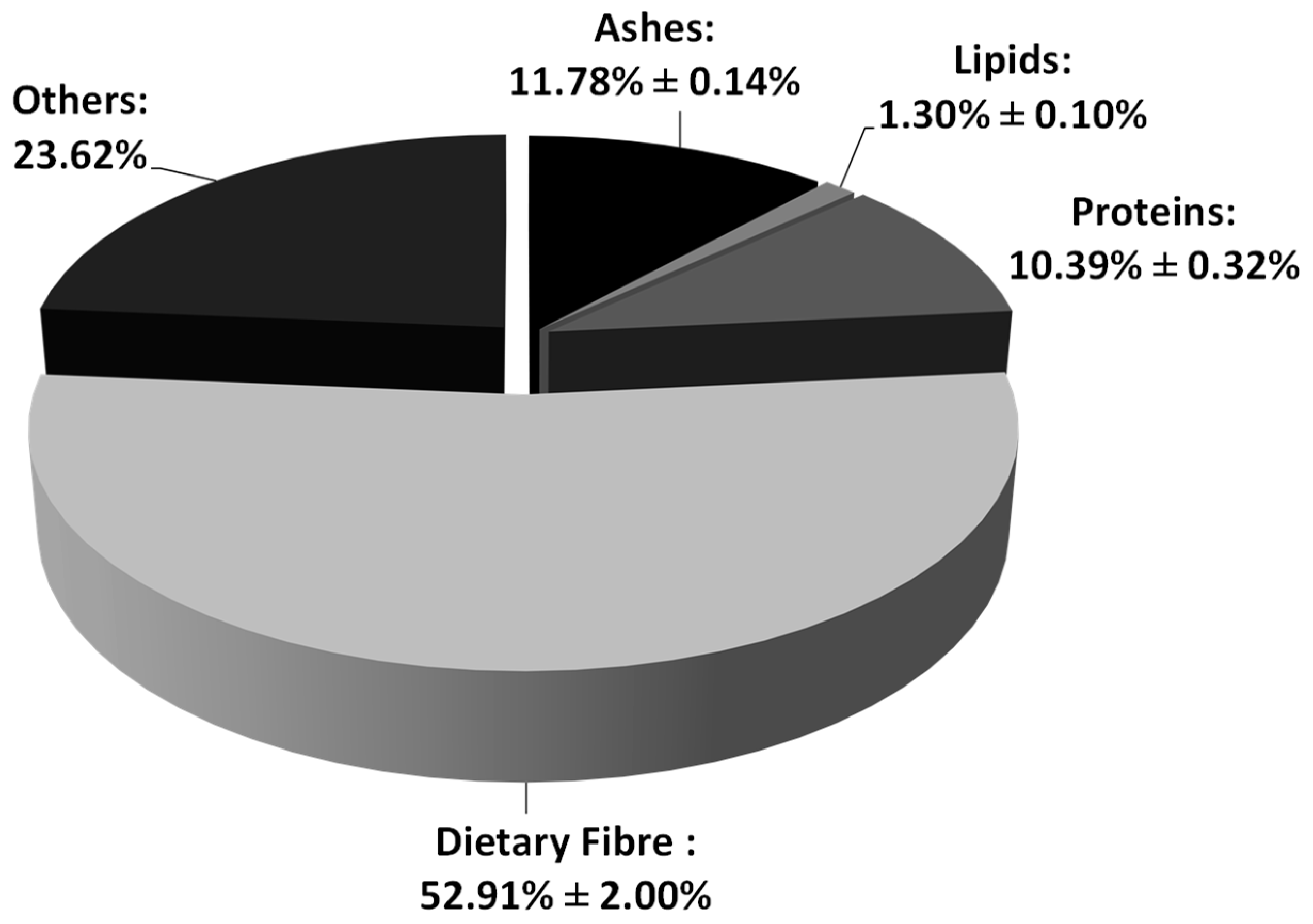
3.1. Proximal Characterization
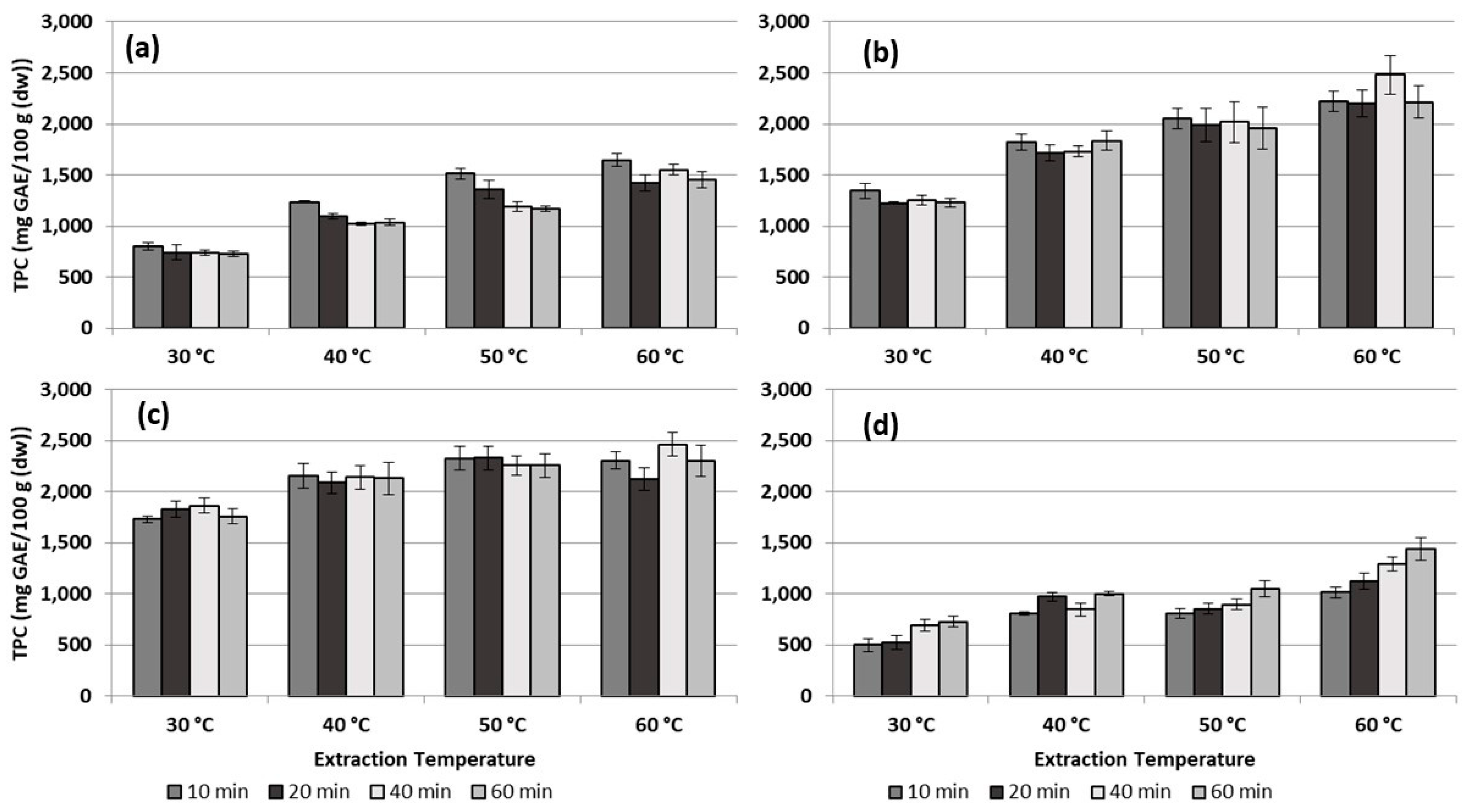
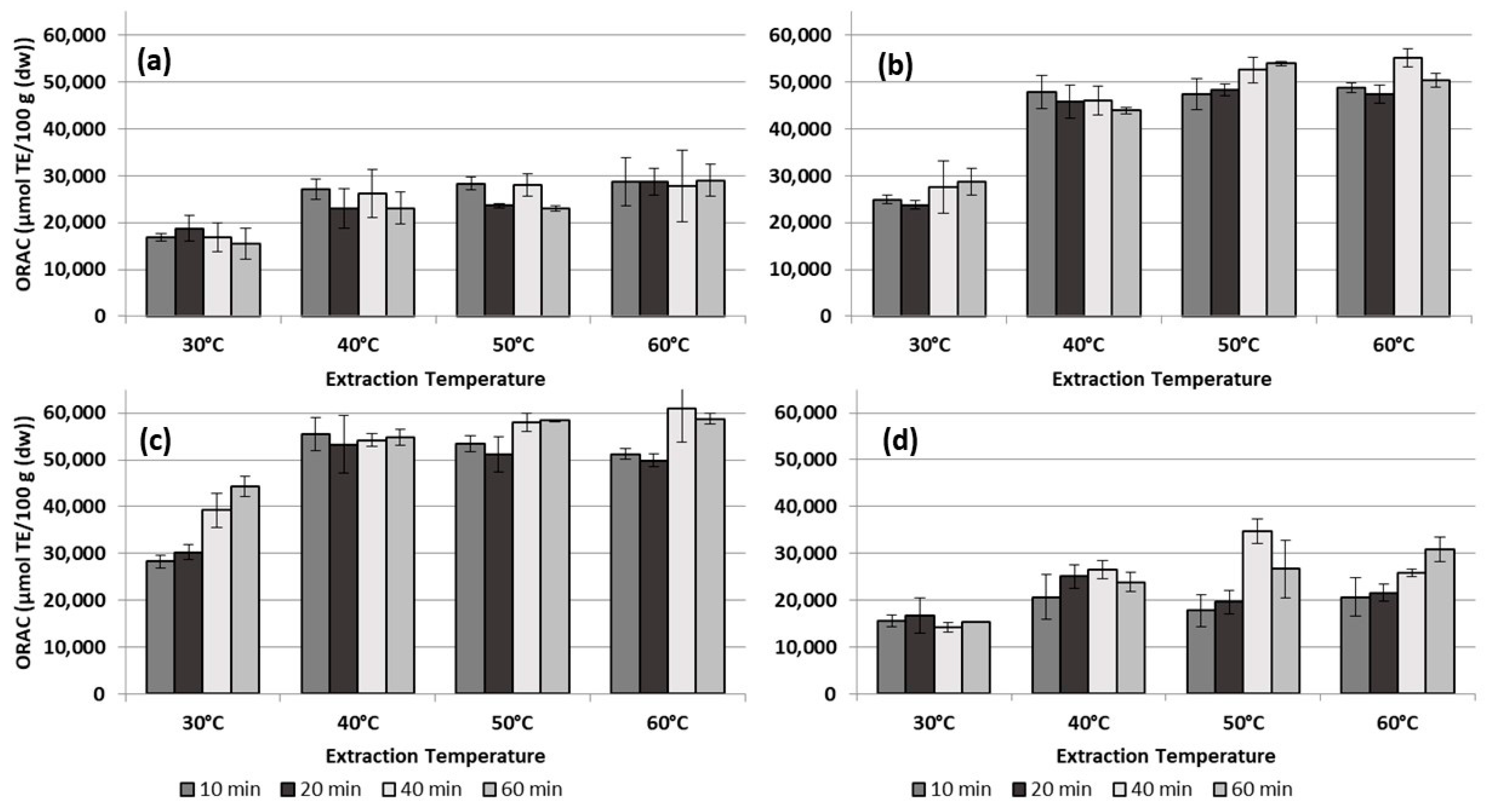
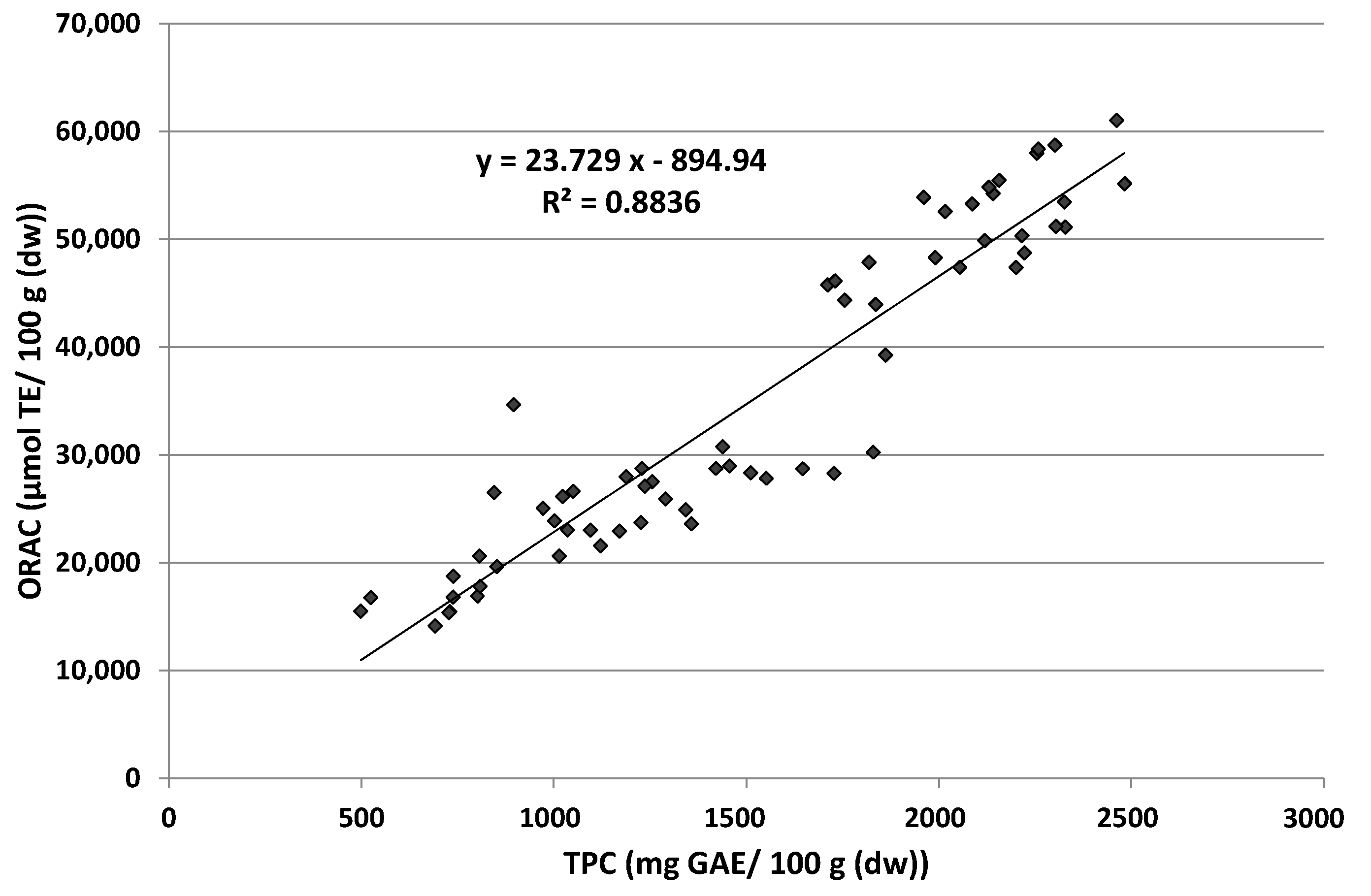
3.2. Total Phenolic Content (TPC) and Antioxidant Capacity (AOC)
3.3. Artichoke Extract Characterization
3.4. Effect of Artichoke Extracts on the Viability of Cancer Cell Lines
4. Discussion
4.1. Artichoke Discards Proximal Characterization
4.2. Total Phenolic Content (TPC) and Antioxidant Capacity (AOC)
4.3. Artichoke Extract Characterization
4.4. Effect of Artichoke Extracts on the Viability of Cancer Cell Lines
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ceccarelli, N.; Curadi, M.; Picciarelli, P.; Martelloni, L.; Sbrana, C.; Giovannetti, M. Globe artichoke as a functional food. Med. J. Nutrition Metab. 2010, 3, 197–201. [Google Scholar] [CrossRef]
- Ruiz-Cano, D.; Pérez-Llamas, F.; Frutos, M.J.; Arnao, M.B.; Espinosa, C.; López-Jiménez, J.Á.; Castillo, J.; Zamora, S. Chemical and functional properties of the different by-products of artichoke (Cynara scolymus L.) from industrial canning processing. Food Chem. 2014, 160, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Silva de Costa, R.; Ferreira Ozela, E.; Ramos Barbosa, W.L.; Pereira, N.L.; Carrera Silva Junior, J.O. Physical Characterization, Chemistry and Physic-chemistry of Cynara Scolymus L. (Asteraceae) dry extract by spray-drying. Rev. Bras. Farmácia 2009, 90, 169–174. [Google Scholar]
- Lattanzio, V.; Kroon, P.A.; Linsalata, V.; Cardinali, A. Globe artichoke: A functional food and source of nutraceutical ingredients. J. Funct. Foods 2009, 1, 131–144. [Google Scholar] [CrossRef]
- Pandino, G.; Lombardo, S.; Williamson, G.; Mauromicale, G. Polyphenol profile and content in wild and cultivated Cynara cardunculus L. Ital. J. Agron. 2012, 7, 254–261. [Google Scholar] [CrossRef]
- Negro, D.; Montesano, V.; Grieco, S.; Crupi, P.; Sarli, G.; De Lisi, A.; Sonnante, G. Polyphenol Compounds in Artichoke Plant Tissues and Varieties. J. Food Sci. 2012, 77, C244–C252. [Google Scholar] [CrossRef]
- Gaafar, A.A.; Salama, Z.A. Phenolic Compounds from Artichoke (Cynara scolymus L.) By-products and their Antimicrobial Activities. J. Biol. Agric. Healthc. 2013, 3, 1–6. [Google Scholar]
- Pereira, C.; Calhelha, R.C.; Barros, L.; Ferreira, I.C.F.R. Antioxidant properties, anti-hepatocellular carcinoma activity and hepatotoxicity of artichoke, milk thistle and borututu. Ind. Crops Prod. 2013, 49, 61–65. [Google Scholar] [CrossRef]
- Garbetta, A.; Capotorto, I.; Cardinali, A.; D’Antuono, I.; Linsalata, V.; Pizzi, F.; Minervini, F. Antioxidant activity induced by main polyphenols present in edible artichoke heads: Influence of in vitro gastro-intestinal digestion. J. Funct. Foods 2014, 10, 456–464. [Google Scholar] [CrossRef]
- Fratianni, F.; Pepe, R.; Nazzaro, F. Polyphenol Composition, Antioxidant, Antimicrobial and Quorum Quenching Activity of the “Carciofo di Montoro” (Cynara cardunculus var. scolymus) Global Artichoke of the Campania Region, Southern Italy. Food Nutr. Sci. 2014, 5, 2053–2062. [Google Scholar] [CrossRef][Green Version]
- Shukla, S.; Gupta, S. Apigenin: A promising molecule for cancer prevention. Pharm. Res. 2010, 27, 962–978. [Google Scholar] [CrossRef] [PubMed]
- Zuorro, A.; Maffei, G.; Lavecchia, R. Effect of solvent type and extraction conditions on the recovery of Phenolic compounds from artichoke waste. Chem. Eng. Trans. 2014, 39, 463–468. [Google Scholar] [CrossRef]
- Mabeau, S.; Baty-Julien, C.; Hélias, A.B.; Chodosas, O.; Surbled, M.; Metra, P.; Durand, D.; Morice, G.; Chesné, C.; Mekideche, K. Antioxidant activity of artichoke extracts and by-products. Acta Hortic. 2007, 730, 491–496. [Google Scholar] [CrossRef]
- Soto, C.; Caballero, E.; Pérez, E.; Zúñiga, M.E. Effect of extraction conditions on total phenolic content and antioxidant capacity of pretreated wild Peumus boldus leaves from Chile. Food Bioprod. Process. 2014, 92, 328–333. [Google Scholar] [CrossRef]
- Marakis, G.; Walker, A.F.; Middleton, R.W.; Booth, J.C.L.; Wright, J.; Pike, D.J. Artichoke leaf extract reduces mild dyspepsia in an open study. Phytomedicine 2002, 9, 694–699. [Google Scholar] [CrossRef]
- Schütz, K.; Muks, E.; Carle, R.; Schieber, A. Quantitative determination of phenolic compounds in artichoke-based dietary supplements and pharmaceuticals by high-performance liquid chromatography. J. Agric. Food Chem. 2006, 54, 8812–8817. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, H.; Lo, R. Phenolic compounds from the leaf extract of artichoke (Cynara scolymus L.) and their antimicrobial activities. J. Agric. Food Chem. 2004, 52, 7272–7278. [Google Scholar] [CrossRef]
- Kukić, J.; Popović, V.; Petrović, S.; Mucaji, P.; Ćirić, A.; Stojković, D.; Soković, M. Antioxidant and antimicrobial activity of Cynara cardunculus extracts. Food Chem. 2008, 107, 861–868. [Google Scholar] [CrossRef]
- López-Molina, D.; Navarro-Martínez, M.D.; Melgarejo, F.R.; Hiner, A.N.P.; Chazarra, S.; Rodríguez-López, J.N. Molecular properties and prebiotic effect of inulin obtained from artichoke (Cynara scolymus L.). Phytochemistry 2005, 66, 1476–1484. [Google Scholar] [CrossRef]
- Franck, A. Technological functionality of inulin and oligofructose. Br. J. Nutr. 2002, 87, S287–S291. [Google Scholar] [CrossRef] [PubMed]
- Pandino, G.; Lombardo, S.; Mauromicale, G.; Williamson, G. Profile of polyphenols and phenolic acids in bracts and receptacles of globe artichoke (Cynara cardunculus var. scolymus) germplasm. J. Food Compos. Anal. 2011, 24, 148–153. [Google Scholar] [CrossRef]
- Levine, V.E.; Becker, W.W. The determination of inulin in blood and in urine by means of vanillin in acid medium. Clin. Chem. 1959, 5, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Garret, A.; Murray, B.; Robinson, R.; O’Neill, K. Measuring Antioxidant Capacity Using the ORAC and TOSC Assays. Methods Mol. Biol. 2010, 594, 251–262. [Google Scholar] [CrossRef]
- Pandino, G.; Lombardo, S.; Mauromicale, G. Globe artichoke leaves and floral stems as a source of bioactive compounds. Ind. Crops Prod. 2013, 44, 44–49. [Google Scholar] [CrossRef]
- Cobs-Rosas, M.; Concha-Olmos, J.; Weinstein-Oppenheimer, C.; Zúñiga-Hansen, M.E. Assessment of antiproliferative activity of pectic substances obtained by different extraction methods from rapeseed cake on cancer cell lines. Carbohydr. Polym. 2015, 117, 923–932. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Ignat, I.; Volf, I.; Popa, V.I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem. 2011, 126, 1821–1835. [Google Scholar] [CrossRef]
- Ninfali, P.; Mea, G.; Giorgini, S.; Rocchi, M.; Bacchiocca, M. Antioxidant capacity of vegetables, spices and dressings relevant to nutrition. Br. J. Nutr. 2005, 93, 257–266. [Google Scholar] [CrossRef]
- El Sohaimy, S.A. Chemical composition, antioxidant and antimicrobial potential of artichoke. Open Nutraceuticals J. 2014, 7, 15–20. [Google Scholar] [CrossRef]
- Curadi, M.; Picciarelli, P.; Lorenzi, R.; Graifenberg, A.; Ceccarelli, N. Antioxidant activity and phenolic compounds in the edible parts of early and late italian artichoke (Cynara scolymus L.) varieties. Ital. J. Food Sci. 2005, 17, 33–44. [Google Scholar]
- Lutz, M.; Henríquez, C.; Escobar, M. Chemical composition and antioxidant properties of mature and baby artichokes (Cynara scolymus L.), raw and cooked. J. Food Compos. Anal. 2011, 24, 49–54. [Google Scholar] [CrossRef]
- Pandino, G.; Courts, F.L.; Lombardo, S.; Mauromicale, G.; Williamson, G. Caffeoylquinic acids and flavonoids in the immature Inflorescence of globe artichoke, wild cardoon, and cultivated cardoon. J. Agric. Food Chem. 2010, 58, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Schütz, K.; Kammerer, D.; Carle, R.; Schieber, A. Identification and quantification of caffeoylquinic acids and flavonoids from artichoke (Cynara scolymus L.) heads, juice, and pomace by HPLC-DAD-ESI/MSn. J. Agric. Food Chem. 2004, 52, 4090–4096. [Google Scholar] [CrossRef]
- Lombardo, S.; Pandino, G.; Mauro, R.; Mauromicale, G. Variation of phenolic content in globe artichoke in relation to biological, technical and environmental factors. Ital. J. Agron. 2009, 4, 181–189. [Google Scholar] [CrossRef]
- Claus, T.; Maruyama, S.A.; Palombini, S.V.; Montanher, P.F.; Bonafé, E.G.; de Oliveira Santos Junior, O.; Matsushita, M.; Visentainer, J.V. Chemical characterization and use of artichoke parts for protection from oxidative stress in canola oil. LWT—Food Sci. Technol. 2015, 61, 346–351. [Google Scholar] [CrossRef]
- López, G.; Ros, G.; Rincón, F.; Periago, M.J.; Martínez, M.C.; Ortuño, J. Relationship between Physical and Hydration Properties of Soluble and Insoluble Fiber of Artichoke. J. Agric. Food Chem. 1996, 44, 2773–2778. [Google Scholar] [CrossRef]
- Jiménez-Escrig, A.; Dragsted, L.O.; Daneshvar, B.; Pulido, R.; Saura-Calixto, F. In vitro antioxidant activities of edible artichoke (Cynara scolymus L.) and effect on biomarkers of antioxidants in rats. J. Agric. Food Chem. 2003, 51, 5540–5545. [Google Scholar] [CrossRef]
- Zuorro, A.; Maffei, G.; Lavecchia, R. Reuse potential of artichoke (Cynara scolymus L.) waste for the recovery of phenolic compounds and bioenergy. J. Clean. Prod. 2016, 111, 279–284. [Google Scholar] [CrossRef]
- Romani, A.; Pinelli, P.; Cantini, C.; Cimato, A.; Heimler, D. Characterization of Violetto di Toscana, a typical Italian variety of artichoke (Cynara scolymus L.). Food Chem. 2006, 95, 221–225. [Google Scholar] [CrossRef]
- Chang, S.K.; Alasalvar, C.; Shahidi, F. Superfruits: Phytochemicals, antioxidant efficacies, and health effects–A comprehensive review. Crit. Rev. Food Sci. Nutr. 2019, 59, 1580–1604. [Google Scholar] [CrossRef]
- Noldin, V.F.; Cechinel Filho, V.; Delle Monache, F.; Benassi, J.C.; Christmann, I.L.; Pedrosa, R.C.; Yunes, R.A. Chemical Composition and Biological Activities of the Leaves of Cynara scolymus L. (Artichoke). Cultivated in Brazil. Quim. Nova 2003, 26, 331–334. [Google Scholar] [CrossRef]
- Lombardo, S.; Pandino, G.; Mauromicale, G.; Knödler, M.; Carle, R.; Schieber, A. Influence of genotype, harvest time and plant part on polyphenolic composition of globe artichoke [Cynara cardunculus L. var. scolymus (L.) Fiori]. Food Chem. 2010, 119, 1175–1181. [Google Scholar] [CrossRef]
- Sánchez-Rabaneda, F.; Jauregui, O.; Lamuela-Raventos, R.M.; Bastida, J.; Viladomat, F.; Codina, C. Identification of phenolic compounds in artichoke waste by high- performance liquid chromatography—Tandem mass spectrometry. J. Chromatogr. A 2003, 1008, 57–72. [Google Scholar]
- Michalska, A.; Wojdyło, A.; Brzezowska, J.; Majerska, J.; Ciska, E. The influence of inulin on the retention of polyphenolic compounds during the drying of blackcurrant juice. Molecules 2019, 24, 4167. [Google Scholar] [CrossRef] [PubMed]
- Giammona, G.; Mauro, N.; Scialabba, C. Inulin for Cancer Therapy: Present and Perspectives. Int. J. Pharma Res. Rev. 2016, 5, 63–69. [Google Scholar]
- Mileo, A.M.; Di Venere, D.; Linsalata, V.; Fraioli, R.; Miccadei, S. Artichoke polyphenols induce apoptosis and decrease the invasive potential of the human breast cancer cell line MDA-MB231. J. Cell. Physiol. 2012, 227, 3301–3309. [Google Scholar] [CrossRef]
| p-Value | KW Chi | ||
|---|---|---|---|
| TPC | % Ethanol | 2.23 × 10−16 | 107.62 |
| Temperature | 1.37 × 10−2 | 10.66 | |
| Time | 0.96 | 0.32 | |
| AOC | % Ethanol | 2.21 × 10−16 | 90.17 |
| Temperature | 1.14 × 10−4 | 20.86 | |
| Time | 0.78 | 1.01 |
| Ax/AT (%) | |
|---|---|
| Neo-Chlorogenic acid (5-O-Caffeoylquinic Acid) | 2.25 ± 0.25 |
| Chlorogenic Acid (3-O-Caffeoylquinic Acid) | 32.93 ± 0.72 |
| Dicaffeoylquinic Acid | 45.97 ± 0.22 |
| Flavone | 5.36 ± 0.15 |
| Total 4 Main Compounds | 86.51 |
| Total Area | 100 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noriega-Rodríguez, D.; Soto-Maldonado, C.; Torres-Alarcón, C.; Pastrana-Castro, L.; Weinstein-Oppenheimer, C.; Zúñiga-Hansen, M.E. Valorization of Globe Artichoke (Cynara scolymus) Agro-Industrial Discards, Obtaining an Extract with a Selective Effect on Viability of Cancer Cell Lines. Processes 2020, 8, 715. https://doi.org/10.3390/pr8060715
Noriega-Rodríguez D, Soto-Maldonado C, Torres-Alarcón C, Pastrana-Castro L, Weinstein-Oppenheimer C, Zúñiga-Hansen ME. Valorization of Globe Artichoke (Cynara scolymus) Agro-Industrial Discards, Obtaining an Extract with a Selective Effect on Viability of Cancer Cell Lines. Processes. 2020; 8(6):715. https://doi.org/10.3390/pr8060715
Chicago/Turabian StyleNoriega-Rodríguez, Diana, Carmen Soto-Maldonado, Cristian Torres-Alarcón, Lorenzo Pastrana-Castro, Caroline Weinstein-Oppenheimer, and María Elvira Zúñiga-Hansen. 2020. "Valorization of Globe Artichoke (Cynara scolymus) Agro-Industrial Discards, Obtaining an Extract with a Selective Effect on Viability of Cancer Cell Lines" Processes 8, no. 6: 715. https://doi.org/10.3390/pr8060715
APA StyleNoriega-Rodríguez, D., Soto-Maldonado, C., Torres-Alarcón, C., Pastrana-Castro, L., Weinstein-Oppenheimer, C., & Zúñiga-Hansen, M. E. (2020). Valorization of Globe Artichoke (Cynara scolymus) Agro-Industrial Discards, Obtaining an Extract with a Selective Effect on Viability of Cancer Cell Lines. Processes, 8(6), 715. https://doi.org/10.3390/pr8060715





