Abstract
Pharmaceutical manufacturing is evolving from traditional batch processes to continuous ones. The new global competition focused on throughput and quality of drug products is certainly the driving force behind this transition which, thus, represents the new challenge of pharmaceutical manufacturing and hence of lyophilization as a downstream operation. In this direction, the present review deals with the most recent technologies, based on spray freeze-drying, that can achieve this objective. It provides a comprehensive overview of the physics behind this process and of the most recent equipment design.
1. Introduction
The current worldwide problems with the coronavirus pandemic have demonstrated the fragility of the pharmaceutical supply chain. The implementation of continuous manufacturing of biopharmaceuticals would offer tremendous benefits, providing a more agile and reliable supply chain for both existing and new products, that can rapidly address emergencies. Despite having witnessed the development of continuous technologies for the manufacturing of specific products, many barriers remain to the formation of an overall strategy [1,2]. For example, the transition from the consolidated batch technology to a completely new strategy is hindered by the lack of personnel expertise and training. However, there is a great opportunity in the adoption of continuous platforms for the new generation of drug products, i.e., biopharmaceuticals [3].
Biopharmaceuticals are large and complex molecules that need to be treated with caution to maintain their effectiveness. The relatively short shelf-life stability of liquid biomolecular-based drugs, as well as cold chain storage and transport costs, makes it necessary to remove water by drying. Even though all drying techniques share a common goal, they are conceptually different and not always suitable to deal with temperature-sensitive products. Furthermore, although most of the drying techniques were designed for batch processes, some of them, such as spray freeze-drying (SFD), could theoretically be adapted to serve the continuous drug production.
SFD is a technique that takes advantage of characteristics of spray drying, which involves the atomization of a liquid to create smaller particles, and freeze-drying, which has particular value for drying thermally sensitive materials, to create dry powders in controlled size and enhanced stability. It has recently been proposed as a way for the production of powders with specific targets, preparing formulations of BCS Class II drugs to improve their dissolution behavior (e.g., phenytoin, ciclosporin, danazol, carbamazepine and THC), developing inhalable dry powders for drug substances that do not fall in the categories mentioned above (e.g., voriconazole), or for some necessary dosage forms (e.g., needle-free injection or pulmonary application) [4]. On the other hand, the disadvantages are that fast cooling promotes the formation of a large ice–glass interface at which protein denaturation can easily occur. At the same time, too low cooling rates might indeed result in phase separation. For this reason, an intermediate cooling rate of about 1 °C/min is recommended [5]. The use of excipients, for instance sugars that provide stabilization by means of vitrification and water replacement, is often necessary to prevent or minimize loss of biological activity during processing [6]. A limited number of organic solvents can be used in SFD, as a high vapor pressure and relatively high melting temperature (or eutectic temperature when mixtures with water are used) are needed to yield an acceptable processing time. On the other hand, from an industry point of view, this technology can effectively respond to the new challenge of pharmaceutical manufacturing; that is, moving from batch to continuous operations in order to meet the lack of effective manufacturing capacity and plants. From an industrial point of view, even if SFD is developing slowly, it might become part of the pharmaceutical industry standard. From this perspective, this work will give a comprehensive review of the current and potential technological applications of SFD, as well as a general discussion on how some recent advances in computational approaches and mathematical modelling may enhance the understanding of the processes taking place at scale of the porous matrix and of the influence of its complex structure [7,8]; the methodologies described in these recent works are now mature enough to construct reliable computational models for investigating those pore-scale dynamics which can be difficult to explore with experiments. These insights can be very useful for the equipment design, e.g., by giving insight into the freezing process and its impact on product morphology [9], or enhancing professionals in solving practical and technical issues or tackling complex problems, which are now slowing the development of continuous freeze-drying operations.
The freeze-drying process is constituted by different spatial scales and process steps. Thus, one obstacle to the reliable industrial exploitation of a fully computational approach is the difficulty in finding a link between models at different scales of resolution or simply referring to a different portion of the process, and in ensuring a coherent flow of information between them. This concept of connection between different temporal and spatial modelling scales is what is usually referred to as model interoperability, which is the necessary premise for the full exploitation of modelling workflows in the industry [10,11].
The purpose of the current review is, thus, to give a comprehensive discussion of both advantages and challenges of SFD as a continuous operation, presenting the new opportunities given by the most recent technologies.
2. Principles of Spray Freeze-Drying
The concept of SFD dates back to the late 1940s, when Benson and Ellis [12] successfully applied the process to produce protein particles having different values of specific surface area. Their process concept included the spray freezing of solution into a liquid nitrogen bath, followed by vacuum freeze-drying. SFD only started to be actively investigated in the 1990s [13], although its application was first demonstrated in the late 1950s by Meryman, who showed for the first time the possibility of freeze-drying at atmospheric pressure [14].
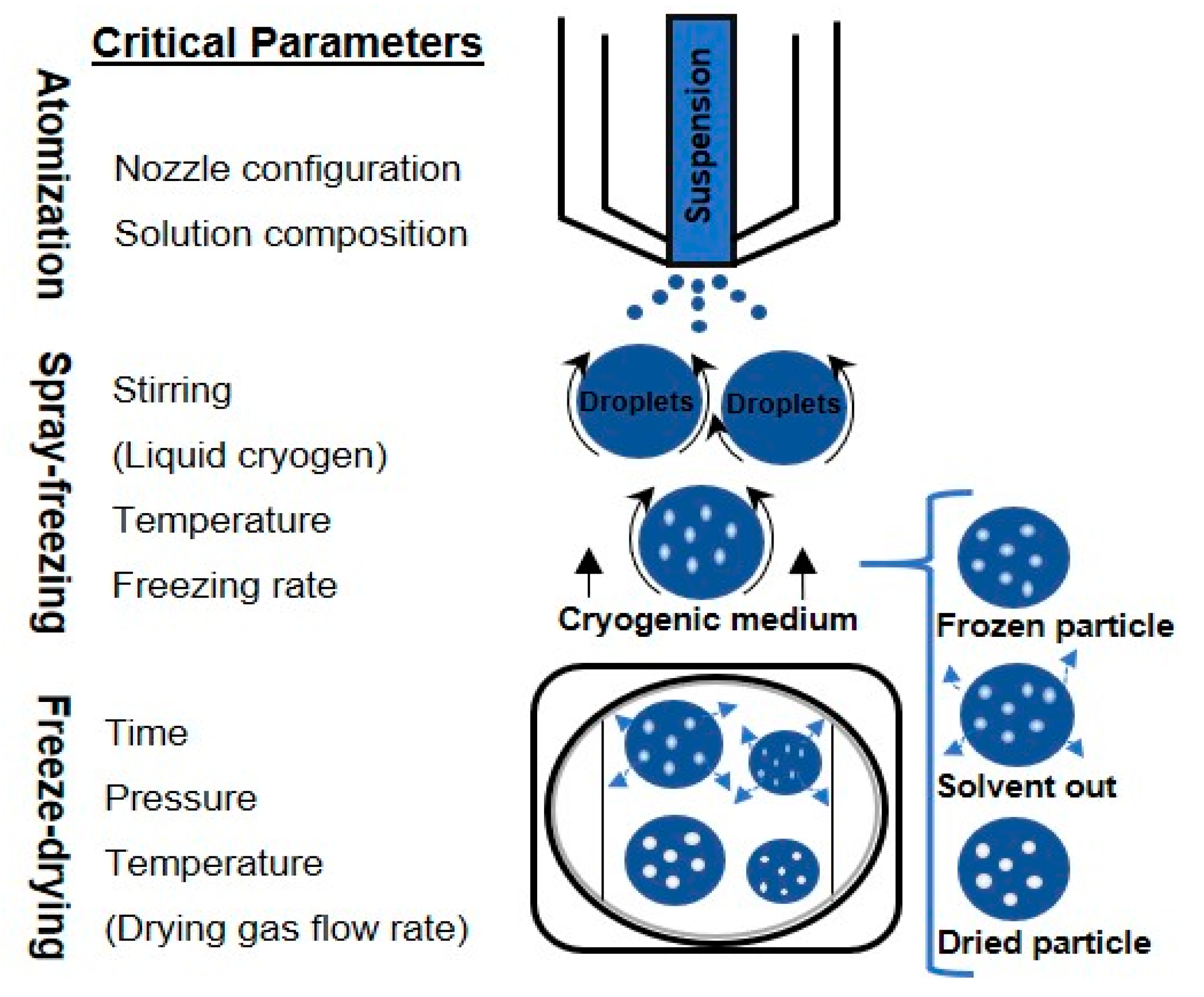
The SFD technique is a three-step operation [15]. The feed solution is initially sprayed through an atomizer, and the atomized droplets are frozen by contact with the low-temperature medium, locking in the spherical droplet shape. After cryogenic processing, the frozen droplets are transferred into a freeze-dryer to sublime water [16] and obtain a dried powder. While the physical and morphological properties of the obtained powder are mainly affected by the first two stages, the final stage relates to the drying time and the total energy required to dry the product [17]. This technique allows a high degree of control over the residual moisture content, mass density, and particle size; in fact, it allows easy manipulation of process parameters such as temperature of the cryogenic liquid, chemical composition and concentration of the solution, and the choice of the atomizer type [18].
The design and operation of each stage are discussed in the following sections, focusing on the atomization (type and size of the nozzle, and nominal flow rate), drying conditions, and formulations. SFD has been increasingly used as a process for pharmaceutical production: the current and proposed new applications of SFD are listed in Table 1.

Table 1.
Examples of spray freeze-drying techniques reported in the literature for different purposes: the active ingredient and carrier for each application are listed.
3. Atomization Techniques
Atomization is the first and critical step that breaks up the feed solution into individual droplets via an atomizer, strongly affecting the drying rate and final particle size distribution [41,42]. Since the freezing and freeze-drying steps do not significantly change the size of the final product, the droplet size distribution obtained during the spraying process determines the particle size of the final product. The density of the final particles is therefore largely determined by the concentration of feed solution. This is particularly important for the aerodynamic behavior of the particles when used for pulmonary delivery by inhalation where an aerodynamic particle size of 1–5 µm is required [25]. For spherical particles, while the aerodynamic diameter of a particle is directly proportional to the geometric size of the particle, it is inversely proportional to the density of the particle [43]. The flexibility in manipulating droplet size, and thus the particle size and surface morphology of the dried powder, is one of the benefits of SFD [44]. However, various atomization factors, including atomization conditions, feed concentration, flow rate and surface tension, should be considered in order to achieve the desired final powder formation [42].
The choice of the atomizer is critical in achieving the specific process and economic objectives, while achieving the desired particle characteristics. The effect of atomization variables on particle size and stability of dried bovine serum albumin (BSA) produced by spray freezing into vapor over liquid (SFV/L) was investigated by Costantino et al. [45]; during atomization, the increased mass flow rate, defined as the ratio of atomization gas to liquid feeding, was found to decrease the final size of the powder. In fact, it was proven that powders made of smaller particles were obtained by decreasing the liquid feed flow rate and increasing the atomization of nitrogen flow rate (or nitrogen atomization pressure).
Heldman and Hohner [46] showed that the reduction in the particle size reduces the drying time. Small droplets provide more homogenous freezing and increase both freezing and drying rates due to their high surface area. The freezing and freeze-drying times depend on the square of the sample thickness, thus reducing the size of the material results in shortening the time required by both steps. By spraying, microsphere powders can be obtained which can flow smoothly and freely after drying. Besides, the smooth spherical and porous structure of the particles increases the rehydration capacity of the powder product [47].
The atomization can be performed using different types of nozzles, the most employed are summarized below.
Hydraulic (Pressure) nozzles: Sprays are generated by forcing the fluid through an orifice. The required energy is provided by converting the pressure into kinetic energy, and the droplet size varies as a function of the feed rate and viscosity, and the spraying pressure.
Pneumatic nozzles: The atomization energy is provided by a compressed gas flow (usually air) that interacts with the liquid and produces a shear field that results in a wide range of droplet sizes. These devices are also known as multi-fluid nozzles. Two-fluid nozzles, where the liquid feed and the compressed gas are fed into the nozzle, are the most popular configuration for pharmaceutical applications. There are also configurations that allow the feeding of two independent feeds. If such a configuration has one gas channel, it is called three-fluid, if it has two gas passages, it is called four-fluid nozzle [48].
Ultrasonic nozzles: The liquid is broken into fine droplets when a high-frequency electrical signal is converted into mechanical energy and transferred into the liquid. Typically, ultrasonic nozzles consist of two piezoelectric transducers that receive electrical input placed between two electrodes. This causes simultaneous mechanical expansion and contraction of the transducers, resulting in ultrasonic vibrations sent to the nozzle tip in order to atomize the feed. The droplet size depends on the operating frequency and feed flow rate. The use of such a device allows advanced control over the particle size and provides a narrow droplet size distribution; however, it is only employable for low-viscosity fluids [48,49].
Monodisperse droplet-stream generations: The generation of monodisperse droplets is regulated using thermal ink-jet print-heads or piezoelectric droplet-stream generators. In thermal ink-jet print-heads, tiny resistors create heat that evaporates a small quantity of the ink to create a droplet and forces it out of the nozzle [50]. Although the use of these devices resulted in porous and uniformly sized powders for pulmonary delivery with a relatively heat-resistant active pharmaceutical ingredients [51,52], they may damage thermo-sensitive components. The main principle of piezoelectric droplet-stream generators is based on Rayleigh disintegration of a liquid jet exiting from the orifice of a capillary with the use of a mechanical vibration obtained by a piezoceramic oscillator [53,54].
These methods are broadly categorized atomization techniques being currently investigated; an in-depth description of the spray production according to the different nozzle types is given in [55].
Comparison of Atomizers
The advantages and disadvantages of the various atomizers used in SFD are listed in Table 2. Hydraulic nozzles may be problematic for the operation of highly viscous feeds, which can cause very rapid wear of the nozzle. Then, it must be further considered that the particle size depends on the viscosity. In a study conducted using an hydraulic nozzle, particles with diameters smaller than 100 µm could easily be obtained by using low viscosity feed solutions (1.65–3.85 cP), but similar particle sizes could not be achieved in the case of viscosity higher than 10 cP [38]. The only parameter that can be changed or adjusted in hydraulic nozzles is the feed rate, and this limits the control of the properties of the obtained particles [48].

Table 2.
Lists of different types of atomizers used in SFD.
In two-fluid nozzles, the atomizing gas flow rate and the liquid feed rate are the most important factors affecting the final particle size [56,57]. As already said, an increase in the atomization gas flow rate reduces the particle size, and the effect is expected to be more relevant when the liquid is moving at lower velocity [45]. Al-Hakim et al. [58] concluded that the spray freezing process with hydraulic and two-fluid nozzle results in different droplet sizes (a typical (d10) value of 12 µm for two-fluid and 50 µm for hydraulic nozzle were measured); besides, the necessity of a significant amount of gas into the pneumatic nozzle may affect spray freezing. It is important to note that droplet size measurements were performed at different feed flow rates of 0.53 cm3 s−1 and 12.5 cm3 s−1 for the tip of the two-fluid and hydraulic nozzles, respectively. The two-fluid nozzles have only one liquid supply line, so it is necessary to find a common solvent for the active pharmaceutical component and the carrier to ensure that both are dissolved; this could sometimes be challenging.
It may be preferable using four-fluid nozzles that have two liquid passages; various authors showed that polymeric particles with prolonged or controlled release could successfully be produced by using them [48,59,60,61]. It allows the production of particles within a significantly narrower size distribution than the two-flow nozzle [62]. Three-fluid nozzles, with two independent liquid and one gas passages, are also available, and used in spray drying, but are not documented for SFD application in the knowledge of the authors.
Although most of the published work has been conducted using hydraulic or pneumatic nozzles, ultrasonic nozzles have recently gained a great deal of popularity. Studies in which the SFD process is carried out utilizing ultrasonic nozzles have reported that this atomization technique has the advantage of producing droplets with a relatively uniform and narrow size distribution, resulting in a homogeneous size distribution of the powder produced. The control of the ultrasonic nozzle frequency provides an effective mean of control over particle size [44,47,63,64,65,66]. Moreover, atomization without additional airflow allows capturing the drops efficiently in a cryogenic liquid. However, using an ultrasonic nozzle also presents some disadvantages: the higher intensity of vibrational energy required for atomization or the recirculation of feed solution may heat the solution, which can cause deterioration of heat-labile drugs and only low viscosity liquids may be employed. For example, Leung et al. [67] noted that the loss of phage titer after the ultrasonic nozzle spraying was approximately 2 log, while it was approximately 0.75 log with the two-fluid nozzle, for the production of a Pseudomonas podoviridae phage.
A common focus of spray processes is to obtain narrow particle size distributions that are important in pharmaceutical processes for achieving homogeneous drying. Therefore, it is considered very promising that the piezoelectric droplet generator produces almost monodisperse droplets; in fact, it has been used to generate nearly monodisperse droplet with diameters in the range of 231 ± 3 µm and 310 ± 10 µm [31]. On the other hand, it has been evidenced that the final particle size may be strongly affected by droplet collision and merging occurring in the spray plume, and thus measures should be taken to reduce collision-related product changes which lead to loss of particle size uniformity and uncontrolled enlargement of the droplet diameter (increases up to three times the original diameter reported in [53]). Wanning et al. [68] proposed jet-vortex freezing of the droplet-stream, in other words, aerodynamic droplet-stream expansion, for the stabilization of droplet sizes and prevention of droplet collisions. This system injects a droplet stream into a vortex of cold gas created by two tangentially positioned gas-jet nozzles inside a swirl tube, speeds up droplets horizontally and reduces their temperature below the freezing point. The authors highlighted the importance of the position of droplet-stream generator, temperature and velocity of the cold gas vortex on the droplet sizes and collisions of the resulting powders. It is also noted that tightly spaced droplets in fast-moving streams may collide and coalesce before they are frozen, which results in the loss of mono-dispersity. The resulting heterogeneity can be reduced by quick cooling since the temperature of gaseous environment affects both the nucleation rate and the freezing rate of micro-droplets [69]. Another study showed that reducing the horizontal distance between the droplet stream generator orifice and the gas-jet nozzle and increasing the volumetric gas flow rates successfully reduce droplet collisions and the mean particle size from 160 to 50 μm [4]. The proof-of-concept study at laboratory scale also described the use of jet-vortex freezing of droplet-stream for a continuous atmospheric freeze-drying system by simulating the process parameters [70].
4. Spray Freezing Techniques
In SFD, the quench into a cryogen provides rapid freezing and thus mitigates the undesirable effects of large ice crystals, pH shift, and phase separation of drug and excipients. Among the different types of cryogens, e.g., liquid argon, propane, pentane, carbon dioxide, the ones most used are liquid and gas nitrogen. The reasons for this can be found in its relative inertness, density and viscosity, which vary considerably with pressure and temperature, and its desirable low boiling point (−196 °C) [35,38,71,72].
Freezing takes place very quickly as a consequence of the very low temperature of the cryogenic medium and the increased surface area of the droplets formed during the spraying phase. Thanks to their microscopic dimensions and high specific surface areas, thermal equilibrium between the droplet and the environment is quickly reached. This situation allows for accomplishing nucleation and freezing of the droplets in a span of milliseconds or seconds [58]. Rapid freezing prevents the damaging effects of phase separation on biomolecular structure by preventing, or at least minimizing, crystallization of excipients and also minimizing solutes partitioning and pH change [73,74,75]. Figure 1 depicts possible methods for performing the spray freezing step.
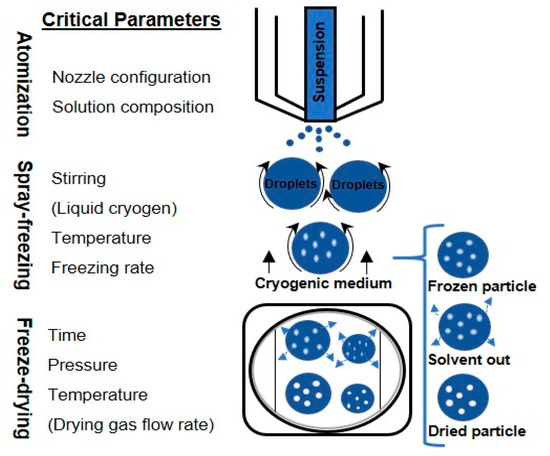
Figure 1.
Schematic diagram of the spray freeze-drying process.
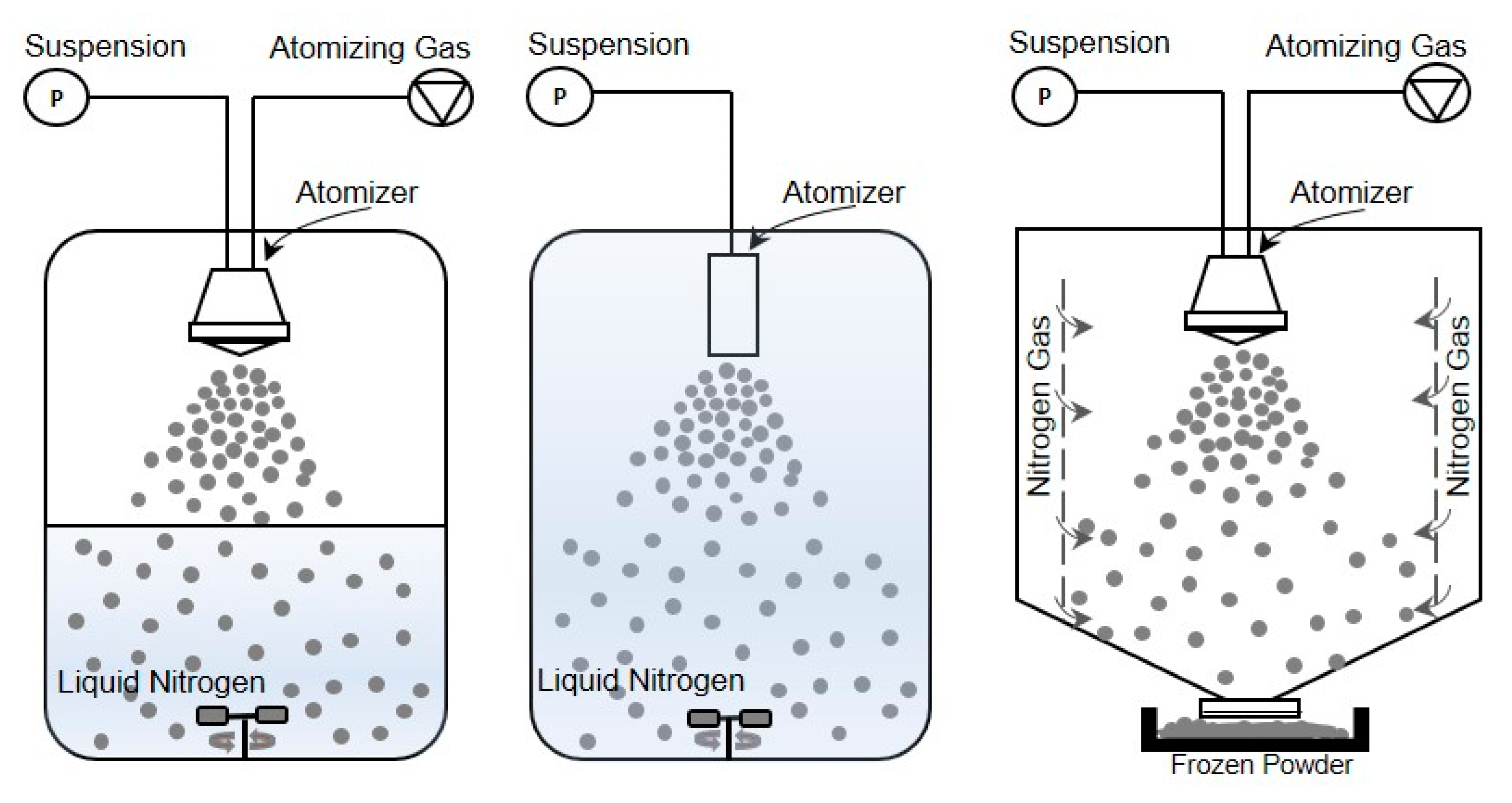
4.1. Spray Freezing into Vapor (SFV)
In this configuration, the atomization of a feed solution occurs in a chamber containing a cold vapor (Figure 2-right) [31]. Mumenthaler and Leuenberger [13] proposed spray freezing into the cold gas stream (air in their case) for the purpose of freezing and, then, drying the frozen particles in a fluidized bed at atmospheric pressure. Cold gas passes through the drying chamber for sublimation of frozen solvents and transports the vapor into a recycling chamber. After condensing on a cooled surface, the recycled gas re-enters the chamber for further sublimation. In the case of food products, improved aroma retention, fine powders, free-flowing material, and shorter drying times were achieved compared to the traditional freeze-drying methods. In this configuration, since the counter-current flow is used, collection efficiency and particle elutriation are major concerns. These difficulties were also evidenced by Leuenberger’s [16] group when they successfully obtained an instant water-soluble drug (10–30 µm size) using the SFV technique at atmospheric pressure, capturing the particles with a gas filter. To overcome the above mentioned difficulties, Wang et al. [36] developed a co-current flow process to convey the frozen powder to the exit filter. In this case, cooled nitrogen gas was fed from lateral porous walls into the chamber, where frozen particles are formed and then dried on an exit filter disc at atmospheric pressure. They obtained free-flowing porous powders, preserving the relevant biological properties of BSA and skim milk formulations.
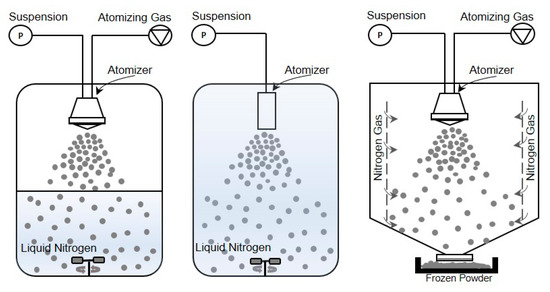
Figure 2.
Illustration of different spray freezing techniques. (left) Spray freezing into vapor over liquid, (middle) Spray freezing into liquid, (right) Spray freezing into vapor.
In another approach [31], a liquid solution is sprayed through a piezoelectric droplet generator nozzle into a very cold environment, a jacketed system cooled by liquid nitrogen, and subsequently dried by sublimation. The principle of this process allows to avoid direct contact between the product and liquid or vapor nitrogen, and to easily remove the product after spray freezing. The researchers tested lysozyme and stabilizers (BSA, polyvinylpyrrolidone or dextran) in various formulations and observed free-flowing and porous particles with a diameter ranging between 231 ± 3 µm and 310 ± 10 µm depending on the composition. In addition, enzyme activity was maintained throughout the process for all formulations.
4.2. Spray Freezing into Liquid (SFL)
Another possible method, developed and patented by the University of Texas at Austin in 2001 and commercialized by Dow Chemical Company [76,77], is SFL (Figure 2-middle) in which the insulated nozzle is positioned directly into a cryogenic liquid to produce frozen microparticles [38]. Then the frozen particles are collected and lyophilized [73,78]. During the SFL process, the cryogenic liquid may be stirred by an impeller to prevent clumping of the frozen particles. This method results in ultra-rapid freezing rates and intense atomization where tiny droplets can be obtained due to the higher viscosity and density of a liquid compared to gas and high velocity through an orifice nozzle [47]. This also means that phase separation and pH changes of dissolved substances and crystalline growth of water can be minimized [37,40]. Some studies have evidenced that rapid cooling rates can favor the formation of an amorphous glass [40,79,80], while others concluded that increased freezing rate and ice water interface area increase protein and peptide denaturation [73].
During the SFL process, generally, a capillary nozzle made from polyether–ether ketone (PEEK) characterized by very low thermal conduction is used to spray the liquid formulation because of the risk of clogging of the nozzle by ice formation. Alternatively, the liquid solution may be sprayed into a cryogen through a heated nozzle. However, this may further contribute to the evaporation of cryogenic fluid gases, which will act as an insulating layer around the droplets, strengthening the Leidenfrost effect, which gives more time to form ice crystals due to low thermal conductivity, resulting in low freezing rate [50,72]. It should be noted that when this technique is applied to water-soluble peptides and proteins, no organic solvent or high temperature is used [73].
Rogers et al. [40] compared SFL and slow freezing of danazol formulations in the presence of hydroxypropyl-β-cyclodextrin (HPbCD) for enhanced dissolution. The morphology of the micronized powder obtained from SFL has been found to be better in terms of reconstruction time compared to that obtained by slow freezing. The diameter of the SFL particles was estimated to be approximately 7 µm. The specific surface area of the SFL particles (113.50 m2/g) was significantly larger than that obtained by slow freezing (0.17 m2/g). Due to its high surface area, SFL danazol particles exhibited the best dissolution profiles. In another SFL investigation of danazol formulations containing danazol/polyvinylpyrrolidone (PVP) K-15, danazol/PVP K-15/poloxamer 407, and danazol/PVP K-15/sodium lauryl sulphate, the surface area of the micronized powders was found to be 79.9 m2/g, 30.0 m2/g, and 48.0 m2/g, respectively. Although these powders have different specific surface areas, each of them exhibited enhanced dissolution rates compared to the micronized crystalline danazol [81].
4.3. Spray Freezing into Vapor over Liquid (SFV/L)
The method most employed for spray freezing (Figure 2-left) in pharmaceutical applications is defined as the atomization of a feed solution into a cryogenic vapor over liquid. In the process, the droplet sprayed from a nozzle positioned at a distance above the boiling cryogenic liquid that is stirred or not stirred begins to freeze slowly while falling down through the cold vapor phase before it comes into contact with the cryogen [82]. When they pass through the cold vapor phase, the droplets begin to freeze and become completely frozen as contact is made with the liquid cryogen. Finally, the suspended frozen particles are captured by sieving, or by letting the cryogen evaporate and then transferring them to a freeze-dryer to obtain a dry powder [6].
Yu et al. [83] used SFV/L and SFL to produce lysozyme powders in order to investigate the morphology and stability of powders. The specific surface area of the SFV/L and SFL samples ranged between 25 and 90 m2/g, and was slightly smaller in SFL particles compared to the particles produced by SFV/L. The particle morphology and particle size observed (via SEM imaging) for SFV/L and SFL indicated similar results and particles varied between 100–300 nm size depending on the formulation. In another research, the particle size distribution for seven types of hydrophobic amino acid (L-alanine, glycine, L-isoleucine, L-leucine, L-phenylalanine, L-tryptophan, or L-valine) obtained by SFV/L, varied again with formulation, but the particle diameters ranged between 5 to 10 µm [84].
5. Freeze-Drying Techniques
Freeze-drying (FD) is performed as the final step of SFD after the completion of spray freezing. The resulting frozen suspension is collected, and the residual cryogen properly removed. Subsequently, the frozen powder is transferred to pre-chilled shelves or directly filled into pre-cooled standard freeze-drying vials using a pre-cooled plastic spoon and tongs or a separating funnel type bowl, introduced by Gieseler in 2004 [85,86].
5.1. Vacuum Freeze-Drying
Vacuum freeze-drying, also known as conventional freeze-drying, which is the most widely used technique, applies vacuum during the drying process to remove the solvent [87]. Primary drying removes the water from formulations by sublimation of ice, and then secondary drying removes the unfrozen bound water [50]. Any type of freeze-dryer from conventional chamber, to rotary, tunnel, or fluid bed can be selected with a suitable condition for performing vacuum freeze-drying.
When frozen particles are packed in a tray, freeze-drying can be carried out in a vacuum chamber; in this manner, the sublimation energy is provided by conduction from the lower heating plate and/or by radiation from a radiating shelf. Yeom and Song [17] studied the SFD process both experimentally and numerically by spraying liquid droplets and packing them into the tray as frozen particles. They investigated the effects of product height, heating plate features and wall temperature on drying time. It was found that the drying time decreased as the product height decreased. Furthermore, the drying time exponentially decreased as the temperature of the heating plate increased and slightly decreased as the upper wall temperature increased.
Capozzi et al. [88] proposed a new concept, the continuous freeze-drying/lyophilization of suspended vials, as a response to the current trend in the pharmaceutical industry. A functional version of the continuous plant was created to examine the heat transfer process during freezing and primary drying as well as its impact on product quality. With this configuration, the primary drying time was found approximately 3−4 times shorter than that observed for the batch lyophilizer, resulting in a final product with properties similar to those traditionally obtained by batch processing. SEM micrographs of lyophilized samples with suspended vial freezing showed that this technology yielded a final product with a more uniform pore structure and characterized by larger pores with respect to the one obtained with conventional freezing. Furthermore, this process concept is potentially suitable for particle-based materials and, hence, compatible with spray freezing technology.
Bruttini and Liapis [89] suggested that the use of a layered packed bed structure where the smallest particles are at the bottom in contact with heating plate while the largest particles are placed on the upper part of the bed in the SFD process may result in higher average total heat and mass transfer rates than those obtained by using a single packed bed with the same particle size and, thus, may speed up both primary and secondary drying. In another study, the impact of using inert particles on drying time was investigated by Zhang et al. [18] to reduce the poor heat transfer through the large particle-to-particle voids within a packed bed. The liquid feed was directly atomized on the surface of cold particles to ensure better thermal conductivity during FD and to prevent agglomeration during SFD. The aim was to investigate the possibility of using inert particles as both thermal conductors and material dispersion carriers. It was found that the drying time decreased with the increase in stainless-steel inert particles diameter from 1 to 5 mm. With 5-mm stainless steel inert particles, the average drying time was found about to be 37% shorter than that observed for the SFD process without inert particles. These results confirm that the packed bed thermal conductivity play a key role in the definition of the SFD rate, even if the process configuration proposed in [18] is not compatible with the pharmaceutical manufacturing requirements.
Langford et al. [90] showed that a rotary freeze-dryer can effectively be used to carry out the SFD of a complex biological material. This study was carried out at both laboratory and commercial scales, using a rotary dryer commercialized by Meridion Technologies. They have reported that this technology can produce lyophilized products having very low residual moisture (below 1%). It should be noted that biopharmaceuticals may become “overdried”. At residual moisture levels below 1 wt-%, stability of biopharmaceuticals can be compromised [91]. The disadvantage here is that the yield is reduced due to the very light particles leaving the drum trapped in the high steam flow.
Tunnel vacuum freeze-drying is a convenient alternative to reduce drying time and product loss. In this concept heat is supplied to the product by infrared and microwave radiation. The frozen product, loaded in trays placed in an automatic airlock, goes through a long cylinder chamber designed to operate under vacuum. Although the continuous freeze-drying tunnels are manufactured by GEA, ALD, Bucher, Devex, SSP, SPX, and Cuddon for food products, it is still challenging its application to pharmaceutical products, because of the stringent aseptic requirements of pharmaceutical manufacturing.
5.2. Atmospheric and Sub-Atmospheric Freeze-Drying
Conventional freeze-drying takes place under vacuum, and this is one of the most critical factors increasing the cost of the drying process. It is thermodynamically possible to carry out the freeze-drying at atmospheric pressure, reducing the process energy consumptions, if the partial pressure of water is kept below the ice vapor pressure [13]. The applicability of atmospheric freeze-drying was first proposed by Meryman [14] using a circulating cold and dry air stream; it was demonstrated that the drying rate is a function of the ice temperature as well as of the vapor pressure gradient between the material and the bulk of the drying medium (cold gas).
In a different study [92], it was emphasized that the atmospheric freeze-drying process had a very long holding time due to internal resistance to mass transfer, and the controlling parameter was molecular diffusion of steam through the porous product structure. Similarly, in another work, it has been reported that the effectiveness of the drying process may vary depending on the pore structure of the material, the specific outer surface area, and the flow rate of the cold gas [47].
Sebastiao et al. [93] presented a numerical model calibrated with experimental data to investigate drying dynamics for single droplets sprayed into the chamber, then freeze-dried at atmospheric pressure. The model considers droplet freezing, heat transfer within the chamber, and powder drying. The results reveal that less than 100 ms are required to freeze spray droplets, and the gas temperature in the chamber and the total time required to achieve a predefined powder moisture content can be estimated.
Although it is thought that the atmospheric freeze-drying technique can easily be applied to continuous processes as it eliminates interlock systems required for vacuum [77], large amounts of cold and dry gas (usually <–30 °C, as the product must remain below the collapse temperature), should be circulated at high velocity for a relatively long time. This situation adversely affects the economic aspects of this manufacturing approach.
The uniform heat transfer to the drying product eliminates the risk of particle melting and collapse, allowing for faster drying. In this respect, fluidized bed systems in which the contact of particles with dry and cold airflow and the heat-mass transfer rate is increased can also be used for freeze-drying, either under atmospheric or sub-atmospheric conditions [94,95].
The use of a fluidized bed with the integration of spray freezing was initially proposed by Leuenberger [16] to freeze-dry liquid pharmaceutical products using an upward flowing cold desiccated gas. In this system, the velocity of the circulated gas exceeds the terminal velocity of the frozen particles. It quickly transports them to the filter system so that at the end of drying, most of the dried particles are recovered from the surface of a subsequent filter. Another problem encountered is that strong adhesion forces among the particles cause them to adhere to the system walls, which reduces product yield and may increase contamination of the particles. As a solution to these problems, Wang et al. [36] proposed a system by adding air outlets to the wall, thereby reducing the build-up of particles on the sidewall and increasing the efficiency of the system.
Sub-atmospheric conditions can also be implemented by applying low vacuum conditions to reduce the large amount of dry gas required by atmospheric freeze-drying. Anandharamakrishnan et al. [94] analyzed the consumption of dry gas for a pilot-scale spray freeze-dryer based on a fluidized bed dryer and working at sub-atmospheric pressure (0.1 bar absolute) in the case of SFD of whey protein. The authors found that a tenfold reduction in pressure reduces the consumption of dry gas by the same factor. It is stated that since the density of the gas is also lower at reduced pressure, particles elutriation from the bed (a phenomenon observed at atmospheric pressure) can be prevented with a sufficiently low pressure, as a consequence of the decrease in inert (non-viscous) drag forces on the particles.
6. Developments of a Continuous Approach in Freeze-Drying
SFD can improve process efficiency and product quality, as a result of increased heat and mass transfer during the drying step thanks to the larger surface area of the frozen particles. Upscaling the SFD process to a commercial scale is likely to be complicated due to the limitations of different factors at each step. The proper design of new technology for continuous manufacturing seems to be a possible solution to meet the changing dynamics of the industry coupled with some practical scale-up considerations.
In the context of upscaling, the numerous spray freezing and freeze-drying methods described above are batch processes, which may be challenging to adapt and implement for continuous manufacturing. In the next section, we will focus on some recent concepts for continuous SFD which can potentially be implemented in a pharmaceutical manufacturing infrastructure, and present practical considerations for a successful SFD scale-up.
6.1. Rey’s Concept
In the process suggested by Rey (Figure 3), the frozen spherical material is placed on the heated conveyors and transported along a vacuum chamber. In this configuration, in order to obtain the frozen material, since there is no atomization, the feed liquid can be formed by dropping and subsequently frozen in counter-current cold air stream. The energy for sublimation is supplied by electrical heaters mounted onto the top of the drying chamber. After the drying process is completed, the dried powder goes to the section where vials are continuously filled [96,97]. It is believed that this concept, including the drying of frozen particles as well as the filling of final powder, is suitable for the design of continuous spray freeze-drying process.
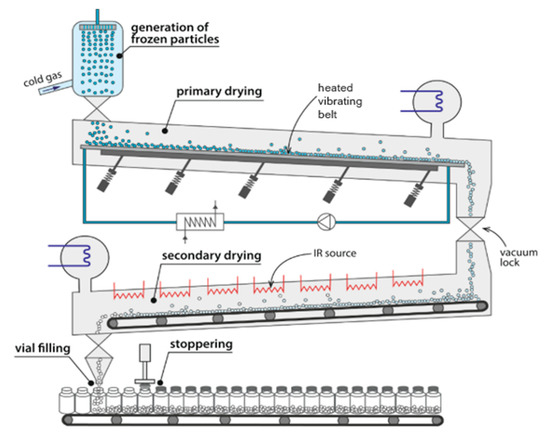
Figure 3.
Illustration of the invention of Rey. Reprinted with permission from Pisano et al., Eur. J. Pharm. Biopharm. 2019, 142, 265–279. Copyright (2019) Elsevier [97].
6.2. Spray Freezing and Dynamic Freeze-Drying
Meridion Technologies designed and manufactured SFD technology to generate frozen microspheres in the range of 300–600 µm diameter. The technique integrates a spray freezing chamber (SprayCon) and a cylindrical rotary drum (LyoMotion) in a fully contained line. In the spray freezing section, laminar jet break-up of the liquid occurs using a droplet formation nozzle placed on the top of the chamber. The droplets are frozen while falling through the double walled cylindrical chamber cooled with gaseous and/or liquid nitrogen (−110 °C). In the next step, dynamic bulk lyophilization is conducted: the frozen material is discharged continuously from the freezing chamber into the precooled drum of the rotary dryer, where it is freeze-dried by constant gentle mixing under vacuum. Radiating sources and temperature-controlled surfaces supply the required sublimation energy. After the completion of drying, the resulting homogeneous bulk product is loaded into a container that can be coupled to a sterile isolator for powder filling [91].
The LyoMotion FD system is known as the first industrial scale unit, which has been tested in industry as a batch technology. In fact, if the spray freezing is continuous in its nature, the separation of particles, as soon as they are fully dried, is currently unfeasible. Therefore, to the best of our knowledge, the current plant configuration could not realize a continuous flow of material going through the drum dryer.
Nevertheless, this technology may potentially be converted into a continuous process if an appropriate system is designed for the continuous discharge of dried particles or, as an alternative, if the spray freezing unit serves multiple batch drum dryers, which work in parallel and are scheduled to give a continuous flow of material exiting the system. Figure 4 introduces the conceptual design of dynamic freeze-drying process with continuous removal of dried particle fraction.
Figure 4.
Scheme of the conceptual design of dynamic freeze-drying process with continuous removal of dried particle fraction.
6.3. Fine-Spray Freeze-Drying
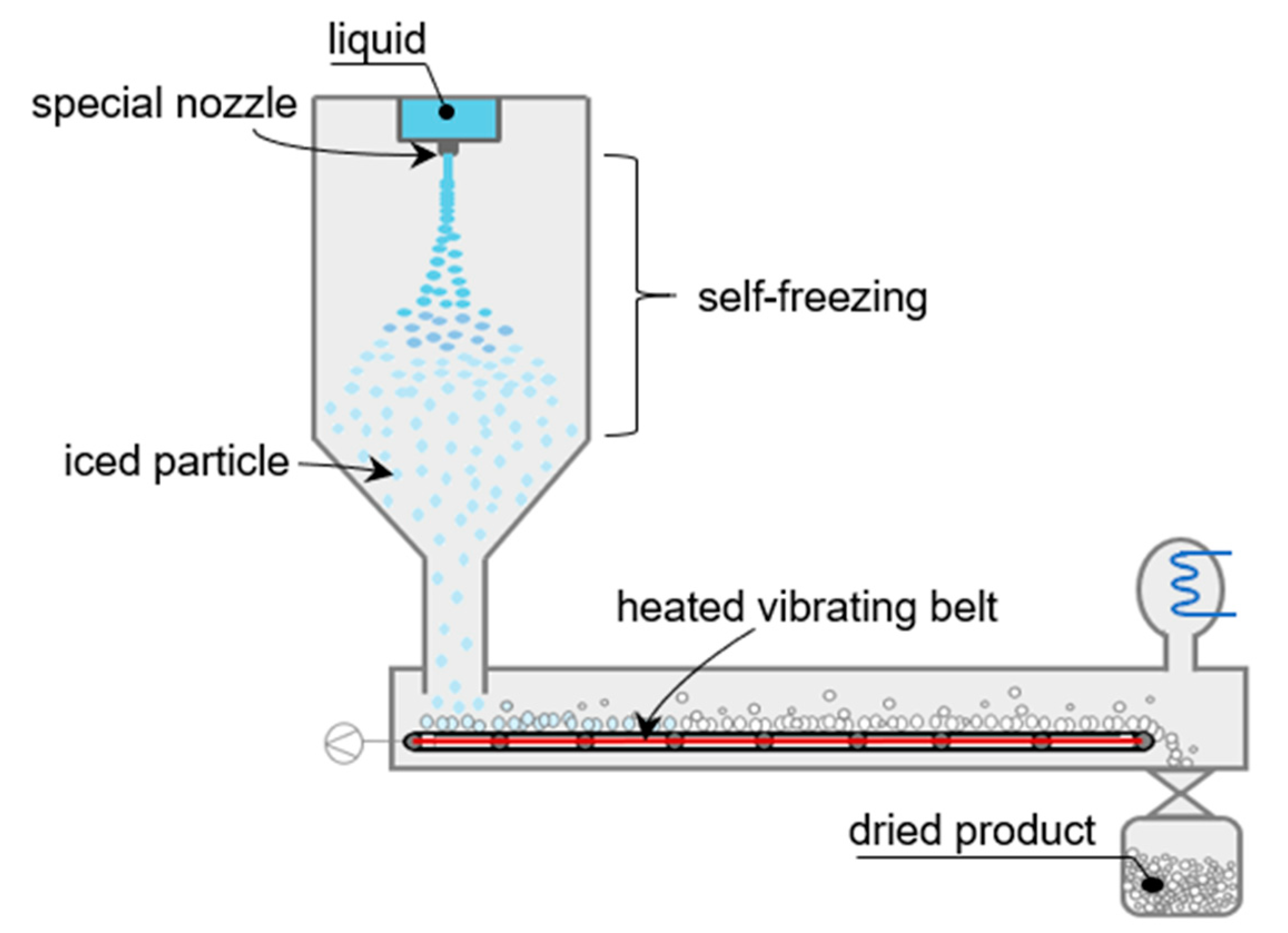
ULVAC Technologies recently introduced a fully closed system for sterile microparticles production by spraying a liquid directly into a vacuum chamber (Figure 5). In the suggested system, a self-freezing process takes place to form frozen particles. The liquid sample sprayed as a liquid column from a special nozzle into the vacuum chamber is dispersed gradually by forming uniform water particles. These particles are self-freezing because some vapor flashes off which cools the droplet while falling in the vacuum chamber, and then they are formed into frozen particles. When the spraying process is finished, and the self-freezing frozen particles are gathered on the heating/cooling belt in the chamber, the belt is heated to obtain the dry sample. The fully closed design for the sterile formulation was established by the agreement between ULVAC Technologies and Azbil Telstar Technologies as a combination of a mass-production system and the fine-spray freeze-drying technology [98]. On the other hand, the system is still under development regarding its transition to a continuous production concept.
Figure 5.
Schematic design of the fine-spray freeze-drying system.
6.4. Stirred Freeze-Drying
Another technique, which in principle allows continuous freeze-drying, is named Active Freeze Drying (Figure 6). In the concept by Hosokawa Micron, the material is first frozen through a freezing medium and/or controlled cooling, and then freeze-dried in a jacketed and conical vacuum dryer, Vrieco-Nauta, under stirred conditions at low temperature and pressure. The heat required for sublimation is supplied from the jacketed walls and distributed by a stirrer which also helps to carry the dried material to the integrated filter system. When the vacuum is broken, the resulting loose and free-flowing product is poured directly into a sterilized container, which allows an aseptic and sterile operation. The main advantage of operating the process under stirred conditions is that the heat is better distributed than in conventional tray-type lyophilizers, resulting in shorter and homogeneous drying. In addition, continuous mixing ensures that the particle size of the final product is extremely fine without an additional crushing stage. In the case of spray frozen particles, the mixing system should be redesigned to avoid crushing. This technique proposes continuous production of dry particles by collecting them in a filter alongside the actual dryer [98].
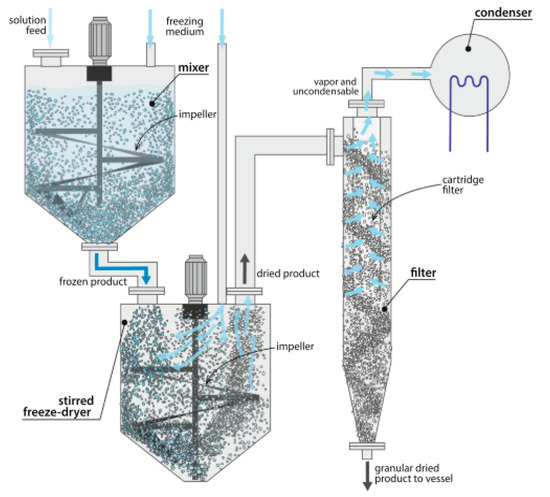
Figure 6.
Schematic design of the stirred freeze-drying process. Reprinted with permission from Pisano et al., Eur. J. Pharm. Biopharm. 2019, 142, 265–279. Copyright (2019) Elsevier [97].
6.5. LYnfinity
A new approach that can be considered as the improvement of Rey’s concept with some modifications has been introduced by the IMA Group (Figure 7), which succeeds in the continuous processing of spray freeze-dried products. The design starts with a spray freezing step into a cryogenically cooled freezing tower in which the bulk liquid product is fed to a temperature-controlled droplet zone utilizing a specially designed nozzle mounted on the top of the freezing column. The nozzle operated at a specific frequency divides the liquid into uniform droplets for consistent drying. The IMA Group claims that care had been taken to identify a nozzle design that does not require high process feed pressure to minimize atomization shear stress. Moreover, they stated that it is possible to control droplet formation for a wide variety of product formulations with selected nozzle and process parameters. After spraying, the frozen particles are continuously loaded into a heated and inclined conveyor in the drying chamber. During drying, continuous motion of particles on the inclined shelf together with the direct thermal contact between frozen particles and shelf supports rapid sublimation and prevent agglomeration. Finally, the dried spherical particles are filled into various containers for direct powder dosage or bulk storage, assuring a higher level of sterility. It seems that the spraying technology, which ensures the uniform production of dried particles, is crucial not only for consistent drying but also for downstream powder filling. In order to accelerate the drying process and make it more efficient, it was concluded that the important contribution of radiation at low pressure should be taken into account when designing the process equipment. It is believed that this proposed concept provides a necessary and essential capability to take a step forward for the continuous aseptic production of SFD, which shows that technology is at the edge of commercialization [99].
Figure 7.
Schematic design of the LYnfinity.
7. Application of Spray Freeze-Drying to Pharmaceutical and Biological Products
The SFD method is ideal for processing proteins, antibodies, vaccines, dry powder aerosols, and nanoparticles since it enables the production of small spherical powders with a relatively size-controlled porous shape. SFD has been effectively employed to produce dry biomacromolecule powders for specific applications such as pulmonary delivery, microencapsulation, and enhancing the solubility of poorly water-soluble drugs. It has also been used to produce microparticles with characteristic morphology and adequate stability by developing particle formulations [100].
Various technologies can be selected and used for each step, depending on the desired formulation and particulate properties; this can result in numerous alternatives that can be obtained by combining the different concepts. The different combinations of SFD steps that have been reported in the literature and the bioproducts processed are summarized in Table 3. Emami et al. [26] produced immunoglobulin G (IgG) powder using various combinations of trehalose and different amino acids (leucine, phenylalanine, arginine, cysteine, and glycine) for improving the stability of IgG using the SFD method. The formulated powders were stored at 40 °C and 75% RH for two months. The powder showed the best stability in the presence of phenylalanine during the SFD process and after storage. Trehalose, leucine, phenylalanine, and glycine during the SFD procedure resulted to be good stabilizers for IgG, while arginine caused the aggregation of IgG.

Table 3.
Combination of applied steps of SFD processes.
SFD can also be used for dissolution-enhanced formulation strategies. Wei et al. [82] suggested a combination of homogenization and the spray freeze-drying technique to produce poorly soluble and temperature-sensitive diterpenoid lactone drug nanocrystals with improved dissolution using low viscosity hydroxypropyl cellulose (HPC) as a matrix carrier. They reported that higher ratios of HPC (1:2 and 1:1) possessed excellent dispersibility, and it can be an effective stabilizer for agglomeration during storage due to the immobilization of drug nanocrystals in its cage-like structure.
The SFD method is widely employed for the preparation of dry powder in biopharmaceutics for the delivery of aerosolized particles. Parsian et al. [32] examined the effect of hydroxypropyl-β-cyclodextrin (HPbCD) and/or L-leucine excipients on the aerodynamic behavior of budesonide powders in a mannitol-based matrix. Micronized porous particles containing both L-leucine and HPbCD showed improved aerosolization behavior. Ogienko et al. [101] also concluded that the use of zwitterionic glycine (carrier) prior to SFD in the formulation of salbutamol and budesonide (active drug) powders, in cases where the mixed solvent can form clathrate hydrates on freezing, enhances aerosol performance and improves air-flow properties. Okuda et al. [25] prepared RNA (siRNA) inhalation powder containing polyethyleneimine (PEI) using SFD. The powder was characterized for its aerosol performance and showed a porous structure with high aerosol performance with a spherical morphology of approximately 10 μm in diameter. In addition, in vivo studies against tumors metastasized in the lungs of mice showed specific and robust gene silencing activity at a low dosage of 3 μg siRNA per mouse, which did not cause serious injury to the lungs.
SFD can also be considered as an alternative method for microencapsulation with improved bioavailability. Parthasarathi and Anandharamakrishnan [102] suggested an SFD technique to produce Vitamin E microcapsules with highly porous internal structures and excellent dissolution behavior, much better than those produced by spray drying and freeze-drying. In vivo oral bioavailability studies supported this conclusion. In the same study, the moisture content of the powders obtained by SD, FD, and SFD was 7.16, 6.99, and 5.41%, respectively. Semyonov et al. [103] investigated the viability of Lactobacillus paracasei microcapsules produced by SFD and FD. When SFD and FD were compared, the FD process showed a slightly higher cell survival; however, they noted that this technique requires a secondary particle size reduction process, which would significantly reduce viability. It was also shown that the spraying stage did not affect the survival of the cells, and SFD was a successful method to generate dry Lactobacillus paracasei microcapsules with high viability (>60%). On the other hand, another study reported that the survival rate of Lactobacillus casei in the final SFD probiotic powder, obtained by spraying the probiotic solution containing glucose, sucrose, and mannose, and measured using plate agar counting was found to be 97.7% [104]. Similarly, another comparison of three different techniques; spray drying, FD, and SFD in terms of thermal stability was conducted by Isleroglu and Turker [105], regarding microencapsulation of microbial transglutaminase (mTG) using a mixture of inulin and arabic gum as coating materials. When the specific surface area and thermal stability of the samples were established, SFD microencapsulated mTG showed the highest thermal stability. Furthermore, those microcapsules with porous internal structure showed better dissolution characteristics and in vivo bioavailability.
8. Conclusions
The SFD process has attracted a keen interest as a potential continuous technology for pharmaceutical manufacturing. However, most of the SFD technologies have been designed to operate in batch mode and their application in a real industrial environment is critical due to the strict requirements of sterility and accurate dosage of pharmaceutical manufacturing. As widely discussed in this review, some of these technologies can potentially be converted, with minor modifications, to the continuous production of pharmaceuticals, reducing the capital and operational costs associated with long batch cycles, as well as the risk of product contamination, and increasing the uniformity of the final product quality. Overall, we acknowledge the fact that the development of a continuous spray freeze-dryer for pharmaceuticals is still a challenge, but the most recent advancements are paving the way towards the design of fully closed integrated spray freezing and freeze-drying units. Of course, a multitude of factors has to be addressed in this transition, e.g., the process validation protocols, good manufacturing practices, operational cost and energy consumption.
Author Contributions
M.B.A. wrote the manuscript with support from all the other co-authors. All authors have contributed equally to its review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Badman, C.; Cooney, C.L.; Florence, A.; Konstantinov, K.; Krumme, M.; Mascia, S.; Nasr, M.; Trout, B.L. Why we need continuous pharmaceutical manufacturing and how to make it happen. J. Pharm. Sci. 2019, 108, 3521–3523. [Google Scholar] [CrossRef]
- David, T. The pharmaceutical industry and the future of drug development. In Pharmaceuticals in Environment; Hester, R.E., Harrison, R.M., Eds.; The Royal Society of Chemistry: Cambridge, UK, 2015; pp. 1–33. [Google Scholar]
- Pisano, R. Continuous manufacturing of lyophilized products: Why and how to make it happen. Am. Phar. Rev. 2020, 23, 20–22. Available online: https://www.americanpharmaceuticalreview.com/1505-Archives/563953-April-2020/ (accessed on 9 May 2020).
- Wanning, S.; Süverkrüp, R.; Lamprecht, A. Aerodynamic droplet stream expansion for the production of spray freeze-dried powders. AAPS PharmSciTech 2017, 18, 1760–1769. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Pikal, M.J. Design of freeze-drying processes for pharmaceuticals: Practical advice. Pharm. Res. 2004, 21, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Ferrati, S.; Wu, T.; Fuentes, O.; Brunaugh, A.D.; Kanapuram, S.R.; Smyth, H.D.C. Influence of formulation factors on the aerosol performance and stability of lysozyme powders: A systematic approach. AAPS PharmSciTech 2018, 19, 2755–2766. [Google Scholar] [CrossRef]
- Capozzi, L.C.; Barresi, A.A.; Pisano, R. A multi-scale computational framework for modeling the freeze-drying of microparticles in packed-beds. Powder Technol. 2019, 343, 834–846. [Google Scholar] [CrossRef]
- Boccardo, G.; Sethi, R.; Marchisio, D.L. Fine and ultrafine particle deposition in packed-bed catalytic reactors. Chem. Eng. Sci. 2019, 198, 290–304. [Google Scholar] [CrossRef]
- Capozzi, L.C.; Pisano, R. Looking inside the ’black box’: Freezing engineering to ensure the quality of freeze-dried biopharmaceuticals. Eur. J. Pharm. Biopharm. 2018, 129, 58–65. [Google Scholar] [CrossRef]
- Horsch, M.T.; Niethammer, C.; Boccardo, G.; Carbone, P.; Chiacchiera, S.; Chiricotto, M.; Elliott, J.D.; Lobaskin, V.; Neumann, P.; Schiffels, P. Semantic interoperability and characterization of data provenance in computational molecular engineering. J. Chem. Eng. Data 2000, 65, 1313–1329. [Google Scholar] [CrossRef]
- De Baas, A.F. What Makes a Material Function? Let Me Compute the Ways: Modelling in H2020 LEIT-NMBP Programme Materials and Nanotechnology Projects, 6th ed.; Office of the European Union: Luxembourg, 2017. [Google Scholar]
- Benson, S.W.; Ellis, D.A. Surface areas of proteins. I. Surface areas and heats of absorption. J. Am. Chem. Soc. 1948, 70, 3563–3569. [Google Scholar] [CrossRef]
- Mumenthaler, M.; Leuenberger, H. Atmospheric spray-freeze drying: A suitable alternative in freeze-drying technology. Int. J. Pharm. 1991, 72, 97–110. [Google Scholar] [CrossRef]
- Meryman, H.T. Sublimation freeze-drying without vacuum. Science 1956, 130, 628–629. [Google Scholar] [CrossRef]
- Emami, F.; Vatanara, A.; Park, E.J.; Na, D.H. Drying technologies for the stability and bioavailability of biopharmaceuticals. Pharmaceutics 2018, 10, 131. [Google Scholar] [CrossRef]
- Leuenberger, H. Spray freeze-drying—The process of choice for low water soluble drugs? J. Nanoparticle Res. 2002, 4, 111–119. [Google Scholar] [CrossRef]
- Yeom, G.S.; Song, C.S. Experimental and numerical investigation of the characteristics of spray-freeze drying for various parameters: Effects of product height, heating plate temperature, and wall temperature. Drying Technol. 2010, 28, 165–179. [Google Scholar] [CrossRef]
- Zhang, F.; Ma, X.; Wu, X.; Xu, Q.; Tian, W.; Li, Z. Inert particles as process aid in spray-freeze drying. Drying Technol. 2020, 38, 71–79. [Google Scholar] [CrossRef]
- Liao, Q.; Yip, L.; Chow, M.Y.T.; Chow, S.F.; Chan, H.K.; Kwok, P.C.L.; Lam, J.K.W. Porous and highly dispersible voriconazole dry powders produced by spray freeze drying for pulmonary delivery with efficient lung deposition. Int. J. Pharm. 2019, 560, 144–154. [Google Scholar] [CrossRef]
- Ito, T.; Okuda, T.; Takashima, Y.; Okamoto, H. Naked pDNA inhalation powder composed of hyaluronic acid exhibits high gene expression in the lungs. Mol. Pharm. 2019, 16, 489–497. [Google Scholar] [CrossRef]
- Emami, F.; Vatanara, A.; Vakhshiteh, F.; Kim, Y.; Kim, T.W.; Na, D.H. Amino acid-based stable adalimumab formulation in spray freeze-dried microparticles for pulmonary delivery. J. Drug Deliv. Sci. Technol. 2019, 54, 101249. [Google Scholar] [CrossRef]
- Ibrahim, M.; Hatipoglu, M.K.; Garcia-Contreras, L. Cryogenic fabrication of dry powders to enhance the solubility of a promising anticancer drug, SHetA2, for oral administration. AAPS PharmSciTech 2019, 20, 20. [Google Scholar] [CrossRef]
- Braig, V.; Konnerth, C.; Peukert, W.; Lee, G. Can spray freeze-drying improve the re-dispersion of crystalline nanoparticles of pure naproxen? Int. J. Pharm. 2019, 564, 293–298. [Google Scholar] [CrossRef]
- Brunaugh, A.D.; Wu, T.; Kanapuram, S.R.; Smyth, H.D.C. Effect of particle formation process on characteristics and aerosol performance of respirable protein powders. Mol. Pharm. 2019, 16, 4165–4180. [Google Scholar] [CrossRef] [PubMed]
- Okuda, T.; Morishita, M.; Mizutani, K.; Shibayama, A.; Okazaki, M.; Okamoto, H. Development of spray-freeze-dried siRNA/PEI powder for inhalation with high aerosol performance and strong pulmonary gene silencing activity. J. Control. Release 2018, 279, 99–113. [Google Scholar] [CrossRef] [PubMed]
- Emami, F.; Vatanara, A.; Najafabadi, A.R.; Kim, Y.; Park, E.J.; Sardari, S.; Na, D.H. Effect of amino acids on the stability of spray freeze-dried immunoglobulin G in sugar-based matrices. Eur. J. Pharm. Sci. 2018, 119, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Moghaddam, S.P.H.; Farhat, S.; Vatanara, A. Porous microparticles containing raloxifene hydrochloride tailored by spray freeze drying for solubility enhancement. Adv. Pharm. Bull. 2018, 8, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Chan, A.Y.L.; Chow, M.Y.T.; Lo, F.F.K.; Qiu, Y.; Kwok, P.C.L.; Lam, J.K.W. Spray freeze drying of small nucleic acids as inhaled powder for pulmonary delivery. Asian J. Pharm. Sci. 2018, 13, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Adeli, E. The use of spray freeze drying for dissolution and oral bioavailability improvement of Azithromycin. Powder Technol. 2017, 319, 323–331. [Google Scholar] [CrossRef]
- Ye, T.; Yu, J.; Luo, Q.; Wang, S.; Chan, H.K. Inhalable clarithromycin liposomal dry powders using ultrasonic spray freeze drying. Powder Technol. 2017, 305, 63–70. [Google Scholar] [CrossRef]
- Eggerstedt, S.N.; Dietzel, M.; Sommerfeld, M.; Süverkrüp, R.; Lamprecht, A. Protein spheres prepared by drop jet freeze drying. Int. J. Pharm. 2012, 438, 160–166. [Google Scholar] [CrossRef]
- Parsian, A.R.; Vatanara, A.; Rahmati, M.R.; Gilani, K.; Khosravi, K.M.; Najafabadi, A.R. Inhalable budesonide porous microparticles tailored by spray freeze drying technique. Powder Technol. 2014, 260, 36–41. [Google Scholar] [CrossRef]
- Tonnis, W.F.; Amorij, J.P.; Vreeman, M.A.; Frijlink, H.W.; Kersten, G.F.; Hinrichs, W.L.J. Improved storage stability and immunogenicity of hepatitis B vaccine after spray-freeze drying in presence of sugars. Eur. J. Pharm. Sci. 2014, 55, 36–45. [Google Scholar] [CrossRef]
- Her, J.Y.; Song, C.S.; Lee, S.J.; Lee, K.G. Preparation of kanamycin powder by an optimized spray freeze-drying method. Powder Technol. 2010, 199, 159–164. [Google Scholar] [CrossRef]
- Engstrom, J.D.; Simpson, D.T.; Cloonan, C.; Lai, E.S.; Williams, R.O.; Barrie Kitto, G.; Johnston, K.P. Stable high surface area lactate dehydrogenase particles produced by spray freezing into liquid nitrogen. Eur. J. Pharm. Biopharm. 2007, 65, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.L.; Finlay, W.H.; Peppler, M.S.; Sweeney, L.G. Powder formation by atmospheric spray-freeze-drying. Powder Technol. 2006, 170, 45–52. [Google Scholar] [CrossRef]
- Yu, Z.; Garcia, A.S.; Johnston, K.P.; Williams, R.O. Spray freezing into liquid nitrogen for highly stable protein nanostructured microparticles. Eur. J. Pharm. Biopharm. 2004, 58, 529–537. [Google Scholar] [CrossRef]
- Barron, M.K.; Young, T.J.; Johnston, K.P.; Williams, R.O. Investigation of processing parameters of spray freezing into liquid to prepare polyethylene glycol polymeric particles for drug delivery. AAPS PharmSciTech 2003, 4, 1–13. [Google Scholar] [CrossRef]
- Hu, J.; Johnston, K.P.; Williams, R.O. Spray freezing into liquid (SFL) particle engineering technology to enhance dissolution of poorly water soluble drugs: Organic solvent versus organic/aqueous co-solvent systems. Eur. J. Pharm. Sci. 2003, 20, 295–303. [Google Scholar] [CrossRef]
- Rogers, T.L.; Nelsen, A.C.; Sarkari, M.; Young, T.J.; Johnston, K.P.; Williams, R.O. Enhanced aqueous dissolution of a poorly water soluble drug by novel particle engineering technology: Spray-freezing into liquid with atmospheric freeze-drying. Pharm. Res. 2003, 20, 485–493. [Google Scholar] [CrossRef]
- Wan, F.; Yang, M. Design of PLGA-based depot delivery systems for biopharmaceuticals prepared by spray drying. Int. J. Pharm. 2016, 498, 82–95. [Google Scholar] [CrossRef]
- Vishali, D.A.; Monisha, J.; Sivakamasundari, S.K.; Moses, J.A.; Anandharamakrishnan, C. Spray freeze drying: Emerging applications in drug delivery. J. Control. Release 2019, 300, 93–101. [Google Scholar] [CrossRef]
- Wan, F.; Maltesen, M.J.; Bjerregaard, S.; Foged, C.; Rantanen, J.; Yang, M. Particle engineering technologies for improving the delivery of peptide and protein drugs. J. Drug Deliv. Sci. Technol. 2013, 23, 355–363. [Google Scholar] [CrossRef]
- Schiffter, H.; Condliffe, J.; Vonhoff, S. Spray-freeze-drying of nanosuspensions: The manufacture of insulin particles for needle-free ballistic powder delivery. J. R. Soc. Interface 2010, 7, S483–S500. [Google Scholar] [CrossRef]
- Costantino, H.R.; Firouzabadian, L.; Hogeland, K.; Wu, C.; Beganski, C.; Carrasquillo, K.G.; Córdova, M.; Griebenow, K.; Zale, S.E.; Tracy, M.A. Protein spray-freeze drying. Effect of atomization conditions on particle size and stability. Pharm. Res. 2000, 17, 1374–1383. [Google Scholar] [CrossRef] [PubMed]
- Heldman, D.R.; Hohner, G.A. An analysis of atmospheric freeze drying. J. Food Sci. 1974, 39, 147–155. [Google Scholar] [CrossRef]
- Ishwarya, S.P.; Anandharamakrishnan, C.; Stapley, A.G.F. Spray-freeze-drying: A novel process for the drying of foods and bioproducts. Trends Food Sci. Technol. 2015, 41, 161–181. [Google Scholar] [CrossRef]
- Cal, K.; Sollohub, K. Spray drying technique. I: Hardware and process parameters. J. Pharm. Sci. 2010, 99, 575–586. [Google Scholar] [CrossRef] [PubMed]
- Huimin, L. Science and Engineering of Droplets: Fundamentals and Applications, 1st ed.; William Andrew: New York, NY, USA, 1999; p. 536. [Google Scholar]
- Wanning, S.; Süverkrüp, R.; Lamprecht, A. Pharmaceutical spray freeze drying. Int. J. Pharm. 2015, 488, 136–153. [Google Scholar] [CrossRef]
- Mueannoom, W.; Srisongphan, A.; Taylor, K.M.G.; Hauschild, S.; Gaisford, S. Thermal ink-jet spray freeze-drying for preparation of excipient-free salbutamol sulphate for inhalation. Eur. J. Pharm. Biopharm. 2012, 80, 149–155. [Google Scholar] [CrossRef]
- Sharma, G.; Mueannoom, W.; Buanz, A.B.M.; Taylor, K.M.G.; Gaisford, S. In vitro characterisation of terbutaline sulphate particles prepared by thermal ink-jet spray freeze drying. Int. J. Pharm. 2013, 447, 165–170. [Google Scholar] [CrossRef]
- Süverkrüp, R.; Eggerstedt, S.N.; Gruner, K.; Kuschel, M.; Sommerfeld, M.; Lamprecht, A. Collisions in fast droplet streams for the production of spherolyophilisates. Eur. J. Pharm. Sci. 2013, 49, 535–541. [Google Scholar] [CrossRef]
- Strutt, J.W. On the instability of jets. Proc. Lond. Math. Soc. 1879, 10, 4–13. [Google Scholar]
- Overhoff, K.A.; Johnston, K.P.; Tam, J.; Engstrom, J.; Williams, R.O. Use of thin film freezing to enable drug delivery: A review. J. Drug Deliv. Sci. Technol. 2009, 19, 89–98. [Google Scholar] [CrossRef]
- Kemp, I.C.; Wadley, R.; Hartwig, T.; Cocchini, U.; See-Toh, Y.; Gorringe, L.; Fordham, K.; Ricard, F. Experimental study of spray drying and atomization with a two-fluid nozzle to produce inhalable particles. Drying Technol. 2013, 31, 930–941. [Google Scholar] [CrossRef]
- Liang, W.; Chow, M.Y.T.; Chow, S.F.; Chan, H.K.; Kwok, P.C.L.; Lam, J.K.W. Using two-fluid nozzle for spray freeze drying to produce porous powder formulation of naked siRNA for inhalation. Int. J. Pharm. 2018, 552, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Al-Hakim, K.; Wigley, G.; Stapley, A.G.F. Phase doppler anemometry studies of spray freezing. Chem. Eng. Res. Des. 2006, 84, 1142–1151. [Google Scholar] [CrossRef]
- Kondo, M.; Niwa, T.; Okamoto, H.; Danjo, K. Particle characterization of poorly water-soluble drugs using a spray freeze drying technique. Chem. Pharm. Bull. 2009, 57, 657–662. [Google Scholar] [CrossRef][Green Version]
- Niwa, T.; Shimabara, H.; Kondo, M.; Danjo, K. Design of porous microparticles with single-micron size by novel spray freeze-drying technique using four-fluid nozzle. Int. J. Pharm. 2009, 382, 88–97. [Google Scholar] [CrossRef]
- Niwa, T.; Shimabara, H.; Danjo, K. Novel spray freeze-drying technique using four-fluid nozzle—Development of organic solvent system to expand its application to poorly water soluble drugs. Chem. Pharm. Bull. 2010, 58, 195–200. [Google Scholar] [CrossRef][Green Version]
- Ozeki, T.; Beppu, S.; Mizoe, T.; Takashima, Y.; Yuasa, H.; Okada, H. Preparation of two-drug composite microparticles to improve the dissolution of insoluble drug in water for use with a 4-fluid nozzle spray drier. J. Control. Release 2005, 107, 387–394. [Google Scholar] [CrossRef]
- Shoyele, S.A.; Cawthorne, S. Particle engineering techniques for inhaled biopharmaceuticals. Adv. Drug Deliv. Rev. 2006, 58, 1009–1029. [Google Scholar] [CrossRef]
- Maa, Y.F.; Ameri, M.; Shu, C.; Payne, L.G.; Chen, D. Influenza vaccine powder formulation development: Spray-freeze-drying and stability evaluation. J. Pharm. Sci. 2004, 93, 1912–1923. [Google Scholar] [CrossRef]
- D’Addio, S.M.; Chan, J.G.Y.; Kwok, P.C.L.; Prud’Homme, R.K.; Chan, H.K. Constant size, variable density aerosol particles by ultrasonic spray freeze drying. Int. J. Pharm. 2012, 427, 185–191. [Google Scholar] [CrossRef]
- Isleroglu, H.; Turker, I.; Tokatli, M.; Koc, B. Ultrasonic spray-freeze drying of partially purified microbial transglutaminase. Food Bioprod. Process. 2018, 111, 153–164. [Google Scholar] [CrossRef]
- Leung, S.S.Y.; Parumasivam, T.; Gao, F.G.; Carrigy, N.B.; Vehring, R.; Finlay, W.H.; Morales, S.; Britton, W.J.; Kutter, E.; Chan, H.K. Production of inhalation phage powders using spray freeze drying and spray drying techniques for treatment of respiratory infections. Pharm. Res. 2016, 33, 1486–1496. [Google Scholar] [CrossRef]
- Wanning, S.; Süverkrüp, R.; Lamprecht, A. Jet-vortex spray freeze drying for the production of inhalable lyophilisate powders. Eur. J. Pharm. Sci. 2017, 96, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Süverkrüp, R.; Eggerstedt, S.; Wanning, S.; Kuschel, M.; Sommerfeld, M.; Lamprecht, A. Collisions and coalescence in droplet streams for the production of freeze-dried powders. Colloids Surf. B 2016, 141, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Süverkrüp, R.; Wanning, S.; Lamprecht, A. Continuous atmospheric droplet–stream freeze-drying processes development and laboratory system. Pharm. Ind. 2016, 78, 741–749. [Google Scholar]
- Rogers, S.; Wu, W.D.; Saunders, J.; Chen, X.D. Characteristics of milk powders produced by spray freeze drying. Drying Technol. 2008, 26, 404–412. [Google Scholar] [CrossRef]
- Engstrom, J.D.; Simpson, D.T.; Lai, E.S.; Williams, R.O.; Johnston, K.P. Morphology of protein particles produced by spray freezing of concentrated solutions. Eur. J. Pharm. Biopharm. 2007, 65, 149–162. [Google Scholar] [CrossRef]
- Yu, Z.; Rogers, T.L.; Hu, J.; Johnston, K.P.; Williams, R.O. Preparation and characterization of microparticles containing peptide produced by a novel process: Spray freezing into liquid. Eur. J. Pharm. Biopharm. 2002, 54, 221–228. [Google Scholar] [CrossRef]
- Heller, M.C.; Carpenter, J.F.; Randolph, T.W. Protein formulation and lyophilization cycle design: Prevention of damage due to freeze-concentration induced phase separation. Biotechnol. Bioeng. 1999, 63, 166–174. [Google Scholar] [CrossRef]
- Sebastião, I.B.; Bhatnagar, B.; Tchessalov, S.; Ohtake, S.; Plitzko, M.; Luy, B.; Alexeenko, A. Bulk dynamic spray freeze-drying. Part 2: Model-based parametric study for spray-freezing process characterization. J. Pharm. Sci. 2019, 108, 2075–2085. [Google Scholar] [CrossRef]
- Williams, R.; Johnston, K.; Young, T.; Rogers, T.; Barron, M.; Yu, Z.; Hu, J. Process for Production of Nanoparticles and Microparticles by Spray Freezing into Liquid. US Patent 6862890 B2, 8 March 2005. [Google Scholar]
- Hu, J.; Johnston, K.P.; To, R.O.W., III. Nanoparticle engineering processes for enhancing the dissolution rates of poorly water soluble drugs. Drug Dev. Ind. Pharm. 2004, 30, 233–245. [Google Scholar] [CrossRef]
- Hu, J.; Johnston, K.P.; Williams, R.O. Stable amorphous danazol nanostructured powders with rapid dissolution rates produced by spray freezing into liquid. Drug Dev. Ind. Pharm. 2004, 30, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, J.M.; Gao, X.; Yacaman, M.J.; Johnston, K.P.; Williams, R.O. Comparison of powder produced by evaporative precipitation into aqueous solution (EPAS) and spray freezing into liquid (SFL) technologies using novel Z-contrast STEM and complimentary techniques. Eur. J. Pharm. Biopharm. 2005, 60, 81–89. [Google Scholar] [CrossRef]
- Hu, J.; Johnston, K.P.; Williams, R.O. Rapid dissolving high potency danazol powders produced by spray freezing into liquid process. Int. J. Pharm. 2004, 271, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Johnston, K.P.; Williams, R.O. Rapid release tablet formulation of micronized danazol powder produced by spray-freezing into liquid (SFL). J. Drug Deliv. Sci. Technol. 2004, 14, 305–311. [Google Scholar] [CrossRef]
- Wei, S.; Ma, Y.; Luo, J.; He, X.; Yue, P.; Guan, Z.; Yang, M. Hydroxypropylcellulose as matrix carrier for novel cage-like microparticles prepared by spray-freeze-drying technology. Carbohydr. Polym. 2017, 157, 953–961. [Google Scholar] [CrossRef]
- Yu, Z.; Johnston, K.P.; Williams, R.O. Spray freezing into liquid versus spray-freeze drying: Influence of atomization on protein aggregation and biological activity. Eur. J. Pharm. Sci. 2006, 27, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Otake, H.; Okuda, T.; Okamoto, H. Development of spray-freeze-dried powders for inhalation with high inhalation performance and antihygroscopic property. Chem. Pharm. Bull. 2016, 64, 239–245. [Google Scholar] [CrossRef]
- Gieseler, H. Product Morphology and Drying Behavior Delineated by a New Freeze-Drying Microbalance. Ph.D. Thesis, Friedrich-Alexander University, Nuremberg-Erlangen, Germany, June 2004. [Google Scholar]
- Schiffter, H.A. Spray-freeze-drying in the manufacture of pharmaceuticals. Eur. Pharm. Rev. 2007, 12, 67–71. [Google Scholar]
- Walters, R.H.; Bhatnagar, B.; Tchessalov, S.; Izutsu, K.I.; Tsumoto, K.; Ohtake, S. Next generation drying technologies for pharmaceutical applications. J. Pharm. Sci. 2014, 103, 2673–2695. [Google Scholar] [CrossRef] [PubMed]
- Capozzi, L.C.; Trout, B.L.; Pisano, R. From batch to continuous: Freeze-drying of suspended vials for pharmaceuticals in unit-doses. Ind. Eng. Chem. Res. 2019, 58, 1635–1649. [Google Scholar] [CrossRef]
- Bruttini, R.; Liapis, A.I. The drying rates of spray freeze drying systems increase through the use of stratified packed bed structures. Int. J. Heat Mass Transf. 2015, 90, 515–522. [Google Scholar] [CrossRef]
- Langford, A.; Balthazor, B.; Bhatnagar, B.; Tchessalov, S.; Hageman, M.J.; Lukas, A.; Plitzko, M.; Luy, B.; Ohtake, S. Beyond freeze-drying of biologics: Vacuum-foam drying and spray freeze-drying. In Proceedings of the IDS’2018, 21st International Drying Symposium, Valencia, Spain, 11–14 September 2018; Carcel, J.A., Clemente, G., Garcia-Perez, J.V., Mulet, A., Rossello, C., Eds.; Universitat Politècnica de València: Valencia, Spain, 2019; pp. 11–14. [Google Scholar] [CrossRef]
- Duralliu, A.; Matejtschuk, P.; Stickings, P.; Hassall, L.; Tierney, R.; Williams, D.R. The influence of moisture content and temperature on the long-term storage stability of freeze-dried high concentration immunoglobulin G (IgG). Pharmaceutics 2020, 12, 303. [Google Scholar] [CrossRef] [PubMed]
- Claussen, I.C.; Ustad, T.S.; Strømmen, I.; Walde, P.M. Atmospheric freeze drying—A review. Drying Technol. 2007, 25, 947–957. [Google Scholar] [CrossRef]
- Borges Sebastião, I.; Robinson, T.D.; Alexeenko, A. Atmospheric spray freeze-drying: Numerical modeling and comparison with experimental measurements. J. Pharm. Sci. 2017, 106, 183–192. [Google Scholar] [CrossRef]
- Anandharamakrishnan, C.; Rielly, C.D.; Stapley, A.G.F. Spray-freeze-drying of whey proteins at sub-atmospheric pressures. Dairy Sci. Technol. 2010, 90, 321–334. [Google Scholar] [CrossRef]
- Leuenberger, H.; Plitzko, M.; Puchkov, M. Spray freeze drying in a fluidized bed at normal and low pressure. Drying Technol. 2006, 24, 711–719. [Google Scholar] [CrossRef]
- Rey, L. Glimpses into the realm of freeze-drying: Classical issues and new ventures. In Freeze-Drying/Lyophilization of Pharmaceutical and Biological Products, 3rd ed.; Rey, L., May, J.C., Eds.; CRC Press: New York, NY, USA, 2010; pp. 1–32. [Google Scholar]
- Pisano, R.; Arsiccio, A.; Capozzi, L.C.; Trout, B.L. Achieving continuous manufacturing in lyophilization: Technologies and approaches. Eur. J. Pharm. Biopharm. 2019, 142, 265–279. [Google Scholar] [CrossRef]
- Bullich, R. Telstar industry session: Continuous freeze drying. In Proceedings of the Innovation Forum in Pharmaceutical Process Professional, Pharmaprocess Forum, Barcelona, Spain, 27–28 October 2015. [Google Scholar]
- IMA Group Web Page. Available online: https://ima.it/pharma/machine/lynfinity/ (accessed on 9 May 2020).
- Gieseler, H.; Lee, G. Gravimetric measurement of momentary drying rate of spray freeze-dried powders in vials. J. Pharm. Sci. 2009, 98, 3447–3455. [Google Scholar] [CrossRef] [PubMed]
- Ogienko, A.G.; Bogdanova, E.G.; Trofimov, N.A.; Myz, S.A.; Ogienko, A.A.; Kolesov, B.A.; Yunoshev, A.S.; Zubikov, N.V.; Manakov, A.Y.; Boldyrev, V.V. Large porous particles for respiratory drug delivery. Glycine-based formulations. Eur. J. Pharm. Sci. 2017, 110, 148–156. [Google Scholar] [CrossRef]
- Parthasarathi, S.; Anandharamakrishnan, C. Enhancement of oral bioavailability of vitamin E by spray-freeze drying of whey protein microcapsules. Food Bioprod. Process. 2016, 100, 469–476. [Google Scholar] [CrossRef]
- Semyonov, D.; Ramon, O.; Kaplun, Z.; Levin-Brener, L.; Gurevich, N.; Shimoni, E. Microencapsulation of Lactobacillus paracasei by spray freeze drying. Food Res. Int. 2010, 43, 193–202. [Google Scholar] [CrossRef]
- Her, J.Y.; Kim, M.S.; Lee, K.G. Preparation of probiotic powder by the spray freeze-drying method. J. Food Eng. 2015, 150, 70–74. [Google Scholar] [CrossRef]
- Isleroglu, H.; Turker, I. Thermal inactivation kinetics of microencapsulated microbial transglutaminase by ultrasonic spray-freeze drying. LWT 2019, 101, 653–662. [Google Scholar] [CrossRef]
- Rahmati, M.R.; Vatanara, A.; Parsian, A.R.; Gilani, K.; Khosravi, K.M.; Darabi, M.; Najafabadi, A.R. Effect of formulation ingredients on the physical characteristics of salmeterol xinafoate microparticles tailored by spray freeze drying. Adv. Powder Technol. 2013, 24, 36–42. [Google Scholar] [CrossRef]
- Her, J.Y.; Lee, K.G. Effect of the solvent composition and annealing process on the preparation of spray freeze-dried acetaminophen powder. Drying Technol. 2017, 35, 625–630. [Google Scholar] [CrossRef]
- Maa, Y.F.; Nguyen, P.A.; Sweeney, T.; Shire, S.J.; Hsu, C. Protein inhalation powders: Spray drying vs spray freeze drying. Pharm. Res. 1999, 16, 249–254. [Google Scholar] [CrossRef]
- Saluja, V.; Amorij, J.P.; Kapteyn, J.C.; De Boer, A.H.; Frijlink, H.W.; Hinrichs, W.L.J. A comparison between spray drying and spray freeze drying to produce an influenza subunit vaccine powder for inhalation. J. Control. Release 2010, 144, 127–133. [Google Scholar] [CrossRef]
- Sweeney, L.G.; Wang, Z.; Loebenberg, R.; Wong, J.P.; Lange, C.F.; Finlay, W.H. Spray-freeze-dried liposomal ciprofloxacin powder for inhaled aerosol drug delivery. Int. J. Pharm. 2005, 305, 180–185. [Google Scholar] [CrossRef]
- Van Drooge, D.J.; Hinrichs, W.L.J.; Dickhoff, B.H.J.; Elli, M.N.A.; Visser, M.R.; Zijlstra, G.S.; Frijlink, H.W. Spray freeze drying to produce a stable Δ9- tetrahydrocannabinol containing inulin-based solid dispersion powder suitable for inhalation. Eur. J. Pharm. Sci. 2005, 26, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Karthik, P.; Anandharamakrishnan, C. Microencapsulation of docosahexaenoic acid by spray-freeze-drying method and comparison of its stability with spray-drying and freeze-drying methods. Food Bioprocess. Technol. 2013, 6, 2780–2790. [Google Scholar] [CrossRef]
- Pouya, M.A.; Daneshmand, B.; Aghababaie, S.; Faghihi, H.; Vatanara, A. Spray-freeze drying: A suitable method for aerosol delivery of antibodies in the presence of trehalose and cyclodextrins. AAPS PharmSciTech 2018, 19, 2247–2254. [Google Scholar] [CrossRef]
- Amorij, J.P.; Saluja, V.; Petersen, A.H.; Hinrichs, W.L.J.; Huckriede, A.; Frijlink, H.W. Pulmonary delivery of an inulin-stabilized influenza subunit vaccine prepared by spray-freeze drying induces systemic, mucosal humoral as well as cell-mediated immune responses in BALB/c mice. Vaccine 2007, 25, 8707–8717. [Google Scholar] [CrossRef]
- Wang, Y.; Kho, K.; Cheow, W.S.; Hadinoto, K. A comparison between spray drying and spray freeze drying for dry powder inhaler formulation of drug-loaded lipid-polymer hybrid nanoparticles. Int. J. Pharm. 2012, 424, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Okuda, T.; Okamoto, H. Development of new formulation dry powder for pulmonary delivery using amino acids to improve stability. Biol. Pharm. Bull. 2016, 39, 394–400. [Google Scholar] [CrossRef]
- Okuda, T.; Suzuki, Y.; Kobayashi, Y.; Ishii, T.; Uchida, S.; Itaka, K.; Kataoka, K.; Okamto, H. Development of biodegradable polycation-based inhalable dry gene powders by spray freeze drying. Pharmaceutics 2015, 7, 233–254. [Google Scholar] [CrossRef]
- Kho, K.; Hadinoto, K. Optimizing aerosolization efficiency of dry-powder aggregates of thermally-sensitive polymeric nanoparticles produced by spray-freeze-drying. Powder Technol. 2011, 214, 169–176. [Google Scholar] [CrossRef]
- Poursina, N.; Vatanara, A.; Rouini, M.R.; Gilani, K.; Najafabadi, A.R. The effect of excipients on the stability and aerosol performance of salmon calcitonin dry powder inhalers prepared via the spray freeze drying process. Acta Pharm. 2016, 66, 207–218. [Google Scholar] [CrossRef]
- Cheow, W.S.; Ng, M.L.L.; Kho, K.; Hadinoto, K. Spray-freeze-drying production of thermally sensitive polymeric nanoparticle aggregates for inhaled drug delivery: Effect of freeze-drying adjuvants. Int. J. Pharm. 2011, 404, 289–300. [Google Scholar] [CrossRef]
- Patil, H.P.; Murugappan, S.; De Vries-Idema, J.; Meijerhof, T.; De Haan, A.; Frijlink, H.W.; Wilschut, J.; Hinrichs, W.L.J.; Huckriede, A. Comparison of adjuvants for a spray freeze-dried whole inactivated virus influenza vaccine for pulmonary administration. Eur. J. Pharm. Biopharm. 2015, 93, 231–241. [Google Scholar] [CrossRef]
- Niwa, T.; Mizutani, D.; Danjo, K. Spray freeze-dried porous microparticles of a poorly water-soluble drug for respiratory delivery. Chem. Pharm. Bull. 2012, 60, 870–876. [Google Scholar] [CrossRef][Green Version]
- Burke, P.A.; Klumb, L.A.; Herberger, J.D.; Nguyen, X.C.; Harrell, R.A.; Zordich, M. Poly(lactide-co-glycolide) microsphere formulations of darbepoetin alfa: Spray drying is an alternative to encapsulation by spray-freeze drying. Pharm. Res. 2004, 21, 500–506. [Google Scholar] [CrossRef]
- Nguyen, X.C.; Herberger, J.D.; Burke, P.A. Protein powders for encapsulation: A comparison of spray-freeze drying and spray drying of darbepoetin alfa. Pharm. Res. 2004, 21, 507–514. [Google Scholar] [CrossRef]
- Cleland, J.L.; Johnson, O.L.; Putney, S.; Jones, A.J.S. Recombinant human growth hormone poly(lactic-co-glycolic acid) microsphere formulation development. Adv. Drug Deliv. Rev. 1997, 28, 71–84. [Google Scholar] [CrossRef]
- Sonner, C.; Maa, Y.F.; Lee, G. Spray-freeze-drying for protein powder preparation: Particle characterization and a case study with trypsinogen stability. J. Pharm. Sci. 2002, 91, 2122–2139. [Google Scholar] [CrossRef]
- Rogers, T.L.; Hu, J.; Yu, Z.; Johnston, K.P.; Williams, R.O. A novel particle engineering technology: Spray-freezing into liquid. Int. J. Pharm. 2002, 242, 93–100. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).