Polyphenol Profile and Antimicrobial and Cytotoxic Activities of Natural Mentha × piperita and Mentha longifolia Populations in Northern Saudi Arabia
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Preparation
2.2. Analyses of Phenolic Compounds
2.3. Antiproliferative and Cytotoxic Activities
2.4. Antioxidant Activity
2.5. Antibacterial Effect
2.6. Antifungal Effect
2.7. Statistical Analyses
3. Results
3.1. Polyphenol Profiling of M. × piperita and M. longifolia Leaf Extracts
3.2. Antioxidant Effects
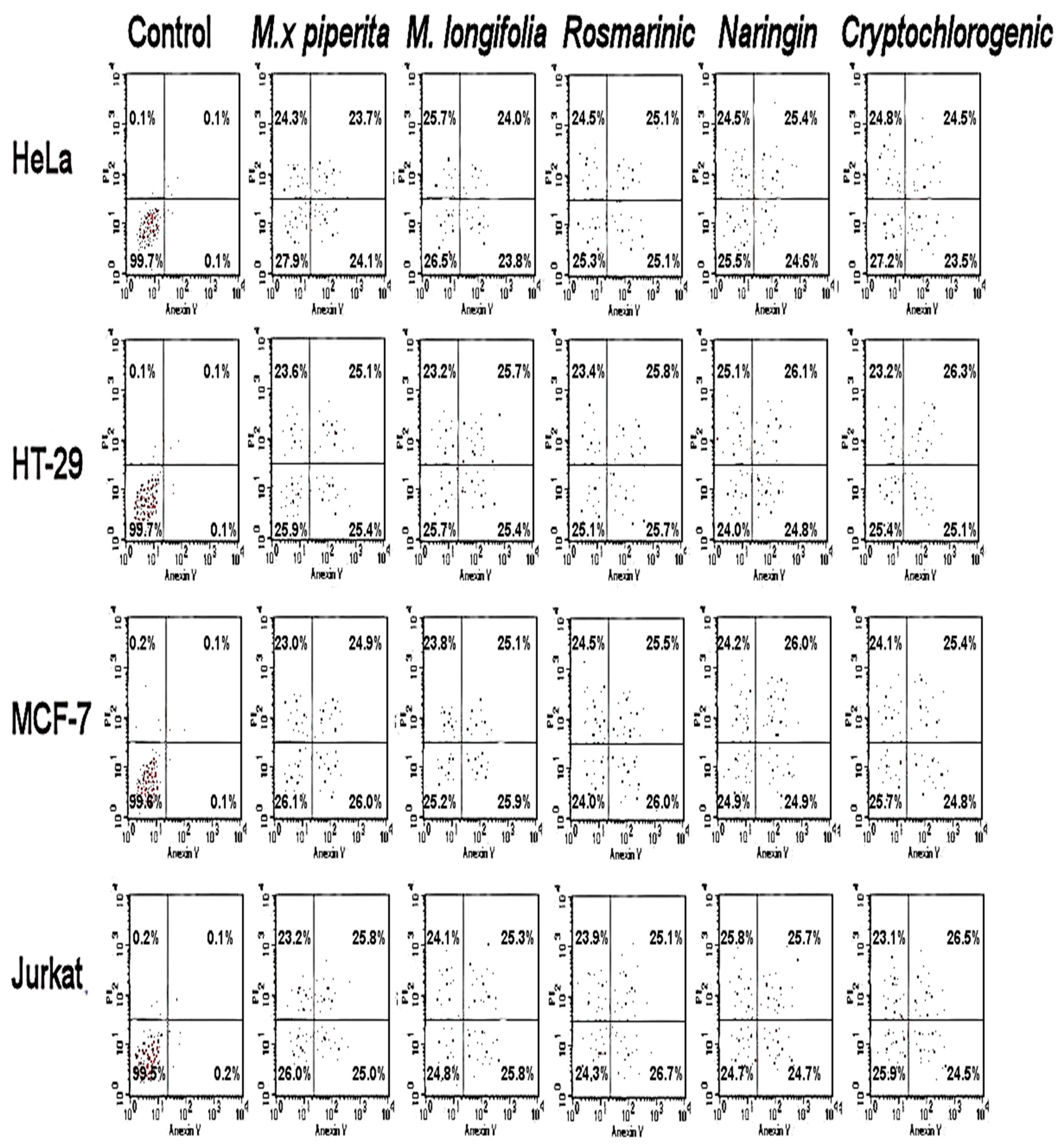
3.3. Antiproliferative and Cytotoxic Effects
3.4. Antibacterial Activities
3.5. Antifungal Effects
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Elansary, H.O.; Szopa, A.; Kubica, P.; Al-Mana, F.A.; Mahmoud, E.A.; El-Abedin, T.Z.; Mattar, M.A.; Ekiert, H.; El-Abedin, T.K.A.Z. Phenolic Compounds of Catalpa speciosa, Taxus cuspidata, and Magnolia acuminata have Antioxidant and Anticancer Activity. Molecules 2019, 24, 412. [Google Scholar] [CrossRef]
- Elansary, H.O.; Szopa, A.; Kubica, P.; Ekiert, H.; Mattar, M.A.; Al-Yafrasi, M.A.; El-Ansary, D.O.; Zin El-Abedin, T.K.; Yessoufou, K. Polyphenol Profile and Pharmaceutical Potential of Quercus spp. Bark Extracts. Plants 2019, 8, 486. [Google Scholar] [CrossRef] [PubMed]
- Elansary, H.O. Tree Bark Phenols Regulate the Physiological and Biochemical Performance of Gladiolus Flowers. Processes 2020, 8, 71. [Google Scholar] [CrossRef]
- Elansary, H.O.; Szopa, A.; Klimek-Szczykutowicz, M.; Jafernik, K.; Ekiert, H.; Mahmoud, E.; Barakat, A.A.; El-Ansary, D.O. Mammillaria Species—Polyphenols Studies and Anti-Cancer, Anti-Oxidant, and Anti-Bacterial Activities. Molecules 2019, 25, 131. [Google Scholar] [CrossRef]
- Okla, M.; Alamri, S.; Salem, M.Z.M.; Ali, H.M.; Behiry, S.I.; Nasser, R.A.; Alaraidh, I.A.; Al-Ghtani, S.M.; Soufan, W. Yield, Phytochemical Constituents, and Antibacterial Activity of Essential Oils from the Leaves/Twigs, Branches, Branch Wood, and Branch Bark of Sour Orange (Citrus aurantium L.). Processes 2019, 7, 363. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; Elansary, H.O.; Ali, H.M.; El-Settawy, A.A.; Elshikh, M.S.; Abdel-Salam, E.M.; Skalicka-Wozniak, K. Bioactivity of essential oils extracted from Cupressus macrocarpa branchlets and Corymbia citriodora leaves grown in Egypt. BMC Complement. Altern. Med. 2018, 18, 23. [Google Scholar] [CrossRef]
- Halagarda, M.; Groth, S.; Popek, S.; Rohn, S.; Pedan, V. Antioxidant Activity and Phenolic Profile of Selected Organic and Conventional Honeys from Poland. Antioxidants 2020, 9, 44. [Google Scholar] [CrossRef]
- Wang, S.; Moustaid-Moussa, N.; Chen, L.; Mo, H.; Shastri, A.; Su, R.; Bapat, P.; Kwun, I.; Shen, C.-L. Novel insights of dietary polyphenols and obesity. J. Nutr. Biochem. 2014, 25, 1–18. [Google Scholar] [CrossRef]
- Ebrahimi, A.; Schluesener, H. Natural polyphenols against neurodegenerative disorders: Potentials and pitfalls. Ageing Res. Rev. 2012, 11, 329–345. [Google Scholar] [CrossRef]
- Cao, H.; Ou, J.; Chen, L.; Zhang, Y.; Szkudelski, T.; Delmas, M.; Daglia, M.; Ulrih, N.P. Dietary polyphenols and type 2 diabetes: Human Study and Clinical Trial. Crit. Rev. Food Sci. Nutr. 2018, 59, 3371–3379. [Google Scholar] [CrossRef]
- Tangney, C.; Rasmussen, H. Polyphenols, inflammation, and cardiovascular disease. Curr. Atheroscler. Rep. 2013, 15, 324. [Google Scholar] [CrossRef] [PubMed]
- Magrone, T.; Magrone, M.; Russo, M.A.; Jirillo, E. Magrone Recent Advances on the Anti-Inflammatory and Antioxidant Properties of Red Grape Polyphenols: In Vitro and In Vivo Studies. Antioxidants 2019, 9, 35. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Siddiqui, S. Concurrent chemoradiotherapy with or without induction chemotherapy for the management of cervical lymph node metastasis from unknown primary tumor. J. Cancer Res. Ther. 2018, 14, 1117. [Google Scholar] [CrossRef]
- Alvarado-Sansininea, J.J.; Sánchez, L.S.; López-Muñoz, H.; Escobar, M.L.; Flores-Guzmán, F.; Tavera-Hernández, R.; Jimenez-Estrada, M. Quercetagetin and Patuletin: Antiproliferative, Necrotic and Apoptotic Activity in Tumor Cell Lines. Molecules 2018, 23, 2579. [Google Scholar] [CrossRef]
- Sezer, E.D.; Oktay, L.M.; Karadadaş, E.; Memmedov, H.; Gunel, N.S.; Sözmen, E. Assessing Anticancer Potential of Blueberry Flavonoids, Quercetin, Kaempferol, and Gentisic Acid, Through Oxidative Stress and Apoptosis Parameters on HCT-116 Cells. J. Med. Food 2019, 22, 1118–1126. [Google Scholar] [CrossRef]
- Yessoufou, K.; Daru, B.H.; Tafirei, R.; Elansary, O.H.; Rampedi, I. Integrating biogeography, threat and evolutionary data to explore extinction crisis in the taxonomic group of cycad. Ecol. Evol. 2017, 7, 2735–2746. [Google Scholar] [CrossRef]
- Saleh, A.; Elfayoumi, H.M.; Youns, M.; Barakat, W. Rutin and orlistat produce antitumor effects via antioxidant and apoptotic actions. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2018, 392, 165–175. [Google Scholar] [CrossRef]
- Dayem, A.A.; Choi, H.Y.; Yang, G.-M.; Kim, K.; Saha, S.K.; Cho, S.G. The Anti-Cancer Effect of Polyphenols against Breast Cancer and Cancer Stem Cells: Molecular Mechanisms. Nutrients 2016, 8, 581. [Google Scholar] [CrossRef]
- Costea, T.; Nagy, P.; Ganea, C.; Szöllősi, J.; Mocanu, M.-M. Molecular Mechanisms and Bioavailability of Polyphenols in Prostate Cancer. Int. J. Mol. Sci. 2019, 20, 1062. [Google Scholar] [CrossRef]
- Kim, H.K.; Missiakas, M.; Schneewind, O. Mouse models for infectious diseases caused by Staphylococcus aureus. J. Immunol. Methods 2014, 410, 88–99. [Google Scholar] [CrossRef]
- Mahmoud, E.A.; Elansary, H.O.; Al-Mana, F.A.; Al-Mana, F.A. Elevated Bioactivity of Ruta graveolens against Cancer Cells and Microbes Using Seaweeds. Processes 2020, 8, 75. [Google Scholar] [CrossRef]
- Desai, A.N.; Anyoha, A.; Madoff, L.C.; Lassmann, B. Changing epidemiology of Listeria monocytogenes outbreaks, sporadic cases, and recalls globally: A review of ProMED reports from 1996 to 2018. Int. J. Infect. Dis. 2019, 84, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Elansary, H.O.; Szopa, A.; Kubica, P.; Ekiert, H.; Ali, H.M.; Elshikh, M.S.; Abdel-Salam, E.M.; El-Esawi, M.A.; El-Ansary, D.O. Bioactivities of Traditional Medicinal Plants in Alexandria. Evid.-Based Complement. Altern. Med. 2018, 2018, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Elansary, H.O.; Salem, M.Z.M.; Ashmawy, N.A.; Yessoufou, K.; El-Settawy, A.A. In vitro antibacterial, antifungal and antioxidant activities of Eucalyptus spp. leaf extracts related to phenolic composition. Nat. Prod. Res. 2017, 9, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277–283. [Google Scholar]
- Elansary, H.O.; El-Abedin, T.K.Z. Omeprazole alleviates water stress in peppermint and modulates the expression of menthol biosynthesis genes. Plant Physiol. Biochem. 2019, 139, 578–586. [Google Scholar] [CrossRef]
- Elansary, H.O.; Mahmoud, E.A.; El-Ansary, D.O.; Mattar, M.A. Effects of Water Stress and Modern Biostimulants on Growth and Quality Characteristics of Mint. Agronomy 2019, 10, 6. [Google Scholar] [CrossRef]
- Elansary, H.O. Green roof Petunia, Ageratum, and Mentha responses to water stress, seaweeds, and trinexapac-ethyl treatments. Acta Physiol. Plant. 2017, 39, 145. [Google Scholar] [CrossRef]
- Elansary, H.O.; Yessoufou, K.; Shokralla, S.; Mahmoud, E.; Skalicka-Wozniak, K. Enhancing mint and basil oil composition and antibacterial activity using seaweed extracts. Ind. Crop. Prod. 2016, 92, 50–56. [Google Scholar] [CrossRef]
- Elansary, H.O.; Ashmawy, N.A. Essential Oils of Mint between Benefits and Hazards. J. Essent. Oil Bear. Plants 2013, 16, 429–438. [Google Scholar] [CrossRef]
- McKay, D.L.; Blumberg, J.B. A review of the bioactivity and potential health benefits of peppermint tea (Mentha piperita L.). Phytother. Res. 2006, 20, 619–633. [Google Scholar] [CrossRef] [PubMed]
- Xhang, X. WHO Monographs on Selected Medicinal Plants WHO: Essential Medicines and Health Products Information Portal; World Health Organization: Geneva, Switzerland, 2004; Volume 2, p. 358. [Google Scholar]
- Medicines, E.D.f.t.Q.o. European Pharmacopoeia 9.0; Council of Europe: Strasburg, Germany, 2017. [Google Scholar]
- Mkaddem, M.; Bouajila, J.; Ennajar, M.; Lebrihi, A.; Mathieu, F.; Romdhane, M. Chemical Composition and Antimicrobial and Antioxidant Activities of Mentha (longifolia L. and viridis) Essential Oils. J. Food Sci. 2009, 74, M358–M363. [Google Scholar] [CrossRef] [PubMed]
- Moloudizargari, M.; Aghajanshakeri, S.; Mikaili, P.; Mojaverrostami, S. Pharmacological and therapeutic effects of Mentha longifolia L. and its main constituent, menthol. Anc. Sci. Life 2013, 33, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Zade, N.S.E.; Sadeghi, A.; Moradi, P. Streptomyces strains alleviate water stress and increase peppermint (Mentha piperita) yield and essential oils. Plant Soil 2018, 434, 441–452. [Google Scholar] [CrossRef]
- Rahimi, Y.; Taleei, A.; Ranjbar, M. Long-term water deficit modulates antioxidant capacity of peppermint (Mentha piperita L.). Sci. Hortic. 2018, 237, 36–43. [Google Scholar] [CrossRef]
- Elansary, H.O.; Mahmoud, E. Egyptian herbal tea infusions’ antioxidants and their antiproliferative and cytotoxic activities against cancer cells. Nat. Prod. Res. 2014, 29, 474–479. [Google Scholar] [CrossRef]
- Kamatou, G.P.; Vermaak, I.; Viljoen, A.; Lawrence, B.M. Menthol: A simple monoterpene with remarkable biological properties. Phytochemistry 2013, 96, 15–25. [Google Scholar] [CrossRef]
- Baliga, M.; Rao, S. Radioprotective potential of mint: A brief review. J. Cancer Res. Ther. 2010, 6, 255. [Google Scholar] [CrossRef]
- Fecka, I.; Kowalczyk, A.; Cisowski, W. Optimization of the separation of flavonoid glycosides and rosmarinic acid from Mentha piperita on HPTLC plates. J. Planar Chromatogr. Mod. TLC 2004, 17, 22–25. [Google Scholar] [CrossRef]
- Olennikov, D.N.; Tankhaeva, L.M. Quantitative determination of phenolic compounds in Mentha piperita leaves. Chem. Nat. Compd. 2010, 46, 22–27. [Google Scholar] [CrossRef]
- Elansary, H.O.M.; Adamec, L.; Štorchová, H. Uniformity of organellar DNA in Aldrovanda vesiculosa, an endangered aquatic carnivorous species, distributed across four continents. Aquat. Bot. 2010, 24, 214–220. [Google Scholar] [CrossRef]
- Sułkowska-Ziaja, K.; Maślanka, A.; Szewczyk, A.; Muszyńska, B. Physiologically Active Compounds in Four Species of Phellinus. Nat. Prod. Commun. 2017, 12, 363–366. [Google Scholar] [CrossRef] [PubMed]
- Szopa, A.; Kokotkiewicz, A.; Bednarz, M.; Luczkiewicz, M.; Ekiert, H. Studies on the accumulation of phenolic acids and flavonoids in different in vitro culture systems of Schisandra chinensis (Turcz.) Baill. using a DAD-HPLC method. Phytochem. Lett. 2017, 20, 462–469. [Google Scholar] [CrossRef]
- Yessoufou, K.; Elansary, H.O.; Mahmoud, E.; Skalicka-Wozniak, K. Antifungal, antibacterial and anticancer activities of Ficus drupacea L. stem bark extract and biologically active isolated compounds. Ind. Crop. Prod. 2015, 74, 752–758. [Google Scholar] [CrossRef]
- Elansary, H.O.; Abdelgaleil, S.A.; Mahmoud, E.; Yessoufou, K.; Elhindi, K.; El-Hendawy, S. Effective antioxidant, antimicrobial and anticancer activities of essential oils of horticultural aromatic crops in northern Egypt. BMC Complement. Altern. Med. 2018, 18, 214. [Google Scholar] [CrossRef]
- Elansary, H.O.; Yessoufou, K.; Abdel-Hamid, A.; El-Esawi, M.A.; Ali, H.M.; Elshikh, M.S. Seaweed Extracts Enhance Salam Turfgrass Performance during Prolonged Irrigation Intervals and Saline Shock. Front. Plant Sci. 2017, 8, 830. [Google Scholar] [CrossRef]
- Ferreira, J.; Miranda, I.; Sousa, V.; Pereira, H. Chemical composition of barks from Quercus faginea trees and characterization of their lipophilic and polar extracts. PLoS ONE 2018, 13, e0197135. [Google Scholar] [CrossRef]
- El-Esawi, M.A.; Elkelish, A.; Soliman, M.H.; Elansary, H.O.; Zaid, A.; Wani, S.H. Serratia marcescens BM1 Enhances Cadmium Stress Tolerance and Phytoremediation Potential of Soybean Through Modulation of Osmolytes, Leaf Gas Exchange, Antioxidant Machinery, and Stress-Responsive Genes Expression. Antioxidants 2020, 9, 43. [Google Scholar] [CrossRef]
- Elansary, H.O.; Szopa, A.; Klimek-Szczykutowicz, M.; Ekiert, H.; Barakat, A.A.; Al-Mana, F.A. Antiproliferative, Antimicrobial, and Antifungal Activities of Polyphenol Extracts from Ferocactus Species. Processes 2020, 8, 138. [Google Scholar] [CrossRef]
- Elansary, H.O.; Mahmoud, E. Basil cultivar identification using chemotyping still favored over genotyping using core barcodes and possible resources of antioxidants. J. Essent. Oil Res. 2014, 27, 82–87. [Google Scholar] [CrossRef]
- El-Kareem, M.S.A.; Rabbih, M.A.; Elansary, H.O.; Al-Mana, F.A. Mass Spectral Fragmentation of Pelargonium graveolens Essential Oil Using GC–MS Semi-Empirical Calculations and Biological Potential. Processes 2020, 8, 128. [Google Scholar] [CrossRef]
- Chumpitazi, B.P.; Kearns, G.L.; Shulman, R.J. Review article: The physiological effects and safety of peppermint oil and its efficacy in irritable bowel syndrome and other functional disorders. Aliment. Pharmacol. Ther. 2018, 47, 738–752. [Google Scholar] [CrossRef] [PubMed]
- Aziz, M.; Sharma, S.; Ghazaleh, S.; Fatima, R.; Acharya, A.; Ghanim, M.; Sheikh, T.; Lee-Smith, W.; Hamdani, S.U.; Nawras, A. The anti-spasmodic effect of peppermint oil during colonoscopy: A systematic review and meta-analysis. Minerva Gastroenterol. Dietol. 2020. [Google Scholar] [CrossRef]
- Keifer, D.; Ulbricht, C.; Abrams, T.R.; Basch, E.; Giese, N.; Giles, M.; Kirkwood, C.D.; Miranda, M.; Woods, J. Peppermint (Mentha X piperita). J. Herb. Pharmacother. 2008, 7, 91–143. [Google Scholar] [CrossRef]
- Salem, M.Z.M.; El-Hefny, M.; Nasser, R.A.; Ali, H.M.; El-Shanhorey, N.A.; Elansary, H.O. Medicinal and biological values of Callistemon viminalis extracts: History, current situation and prospects. Asian Pac. J. Trop. Med. 2017, 10, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Badea, M.L.; Iconaru, S.; Groza, A.; Chifiriuc, M.C.; Beuran, M.; Predoi, M. Peppermint Essential Oil-Doped Hydroxyapatite Nanoparticles with Antimicrobial Properties. Molecules 2019, 24, 2169. [Google Scholar] [CrossRef] [PubMed]
- Mahendran, G.; Rahman, L. Ethnomedicinal, phytochemical and pharmacological updates on Peppermint (Mentha × piperita L.)-A review. Phytother. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Ellis, B.E.; Towers, G.H.N. Biogenesis of rosmarinic acid in Mentha. Biochem. J. 1970, 118, 291–297. [Google Scholar] [CrossRef]
- Guedon, D.J.; Pasquier, B.P. Analysis and Distribution of Flavonoid Glycosides and Rosmarinic Acid in 40 Mentha x piperita Clones. J. Agric. Food Chem. 1994, 42, 679–684. [Google Scholar] [CrossRef]
- Kapp, K.; Hakala, E.; Orav, A.; Pohjala, L.; Vuorela, P.; Püssa, T.; Vuorela, H.; Raal, A. Commercial peppermint (Mentha × piperita L.) teas: Antichlamydial effect and polyphenolic composition. Food Res. Int. 2013, 53, 758–766. [Google Scholar] [CrossRef]
- Zgórka, G.; Głowniak, K. Variation of free phenolic acids in medicinal plants belonging to the Lamiaceae family. J. Pharm. Biomed. Anal. 2001, 26, 79–87. [Google Scholar] [CrossRef]
- Mišan, A.; Mimica-Dukić, N.M.; Mandić, A.I.; Sakač, M.B.; Milovanović, I.L.; Sedej, I.J. Development of a rapid resolution HPLC method for the separation and determination of 17 phenolic compounds in crude plant extracts. Open Chem. 2011, 9, 133–142. [Google Scholar] [CrossRef]
- Inoue, T.; Sugimoto, Y.; Masuda, H.; Kamei, C. Antiallergic Effect of Flavonoid Glycosides Obtained from Mentha piperita L. Boil. Pharm. Bull. 2002, 25, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Kosar, M.; Dorman, H.J.D.; Baser, K.H.C.; Hiltunen, R. Screening of Free Radical Scavenging Compounds in Water Extracts of Mentha Samples Using a Postcolumn Derivatization Method. J. Agric. Food Chem. 2004, 52, 5004–5010. [Google Scholar] [CrossRef]
- Fecka, I.; Raj, D.; Krauze-Baranowska, M. Quantitative Determination of Four Water-Soluble Compounds in Herbal Drugs from Lamiaceae Using Different Chromatographic Techniques. Chromatographia 2007, 66, 87–93. [Google Scholar] [CrossRef]
- Pérez, M.G.F.; Rocha-Guzmán, N.E.; Mercado-Silva, E.; Loarca-Piña, G.; Reynoso–Camacho, R. Effect of chemical elicitors on peppermint (Mentha piperita) plants and their impact on the metabolite profile and antioxidant capacity of resulting infusions. Food Chem. 2014, 156, 273–278. [Google Scholar] [CrossRef]
- Gulluce, M.; Sahin, F.; Sokmen, M.; Ozer, H.; Daferera, D.; Sökmen, A.; Polissiou, M.; Adiguzel, A.; Ozkan, H. Antimicrobial and antioxidant properties of the essential oils and methanol extract from Mentha longifolia L. ssp. longifolia. Food Chem. 2007, 103, 1449–1456. [Google Scholar] [CrossRef]
- Akroum, S.; Bendjeddou, D.; Satta, D.; Lalaoui, K. Antibacterial activity and acute toxicity effect of flavonoids extracted from Mentha longifolia. Am.-Eurasian J. Sci. Res. 2009, 4, 93–96. [Google Scholar]
- Adham, A.N. Comparative extraction methods, phytochemical constituents, fluorescence analysis and HPLC validation of rosmarinic acid content in Mentha piperita, Mentha longifolia and Osimum basilicum. J. Pharmacogn. Phytochem. 2015, 3, 130–139. [Google Scholar]
- Zengin, G.; Bahadori, S.; Dinparast, L.; Movahhedin, N. Phenolic composition and functional properties of wild mint (Mentha longifolia var. calliantha (Stapf) Briq.). Int. J. Food Prop. 2018, 21, 183–193. [Google Scholar] [CrossRef]
- Shekarchi, M.; Hajimehdipoor, H.; Saeidnia, S.; Gohari, A.; Hamedani, M. Comparative study of rosmarinic acid content in some plants of Labiatae family. Pharmacogn. Mag. 2012, 8, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Dudai, N.; Segey, D.; Haykin-Frenkel, D.; Eshel, A. Genetic Variation of Phenolic Compounds Content, Essential Oil Composition and Anti Oxidative Activity in Israel-Grown Mentha longifolia L. Acta Hortic. 2006, 69–78. [Google Scholar] [CrossRef]
- Canelas, V.; Teixeira da Costa, C. Quantitative HPLC Analysis of Rosmarinic Acid in Extracts of Melissa officinalis and Spectrophotometric Measurement of Their Antioxidant Activities. J. Chem. Educ. 2007, 84, 1502. [Google Scholar] [CrossRef]
- Shim, J.-H.; Chae, J.-I.; Cho, S.-S. Identification and Extraction Optimization of Active Constituents in Citrus junos Seib ex TANAKA Peel and Its Biological Evaluation. Molecules 2019, 24, 680. [Google Scholar] [CrossRef] [PubMed]
- De Martino, L.; Mencherini, T.; Mancini, E.; Aquino, R.P.; De Almeida, L.F.R.; De Feo, V. In Vitro Phytotoxicity and Antioxidant Activity of Selected Flavonoids. Int. J. Mol. Sci. 2012, 13, 5406–5419. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Zhang, M.; Zhang, T.; Wu, J.; Wang, J.; Liu, K.; Zhan, N. Antioxidant and Anti-inflammatory Activities of Cynaroside from Elsholtiza bodinieri. Nat. Prod. Commun. 2018, 13. [Google Scholar] [CrossRef]
- Wang, J.; Cao, X.; Jiang, H.; Qi, Y.; Chin, K.L.; Yue, Y.-D. Antioxidant Activity of Leaf Extracts from Different Hibiscus sabdariffa Accessions and Simultaneous Determination Five Major Antioxidant Compounds by LC-Q-TOF-MS. Molecules 2014, 19, 21226–21238. [Google Scholar] [CrossRef]
- Valdés, A.; Sullini, G.; Ibáñez, E.; Cifuentes, A.; Garcia-Cañas, V. Rosemary polyphenols induce unfolded protein response and changes in cholesterol metabolism in colon cancer cells. J. Funct. Foods 2015, 15, 429–439. [Google Scholar] [CrossRef]
- Afonso, M.; Silva, A.; Carvalho, E.B.; Rivelli, D.P.; Barros, S.B.D.M.; Rogero, M.M.; Lottenberg, A.; Torres, R.P.; Mancini-Filho, J. Phenolic compounds from Rosemary (Rosmarinus officinalis L.) attenuate oxidative stress and reduce blood cholesterol concentrations in diet-induced hypercholesterolemic rats. Nutr. Metab. 2013, 10, 19. [Google Scholar] [CrossRef]
- Berdowska, I.; Zielinski, B.; Fecka, I.; Kulbacka, J.; Saczko, J.; Gamian, A. Cytotoxic impact of phenolics from Lamiaceae species on human breast cancer cells. Food Chem. 2013, 141, 1313–1321. [Google Scholar] [CrossRef]
- Scheckel, K.A.; Degner, S.C.; Romagnolo, D.F. Rosmarinic acid antagonizes activator protein-1-dependent activation of cyclooxygenase-2 expression in human cancer and nonmalignant cell lines. J. Nutr. 2008, 138, 2098–2105. [Google Scholar] [CrossRef] [PubMed]
- Rajamani, S.; Radhakrishnan, A.; Sengodan, T.; Thangavelu, S. Augmented anticancer activity of naringenin-loaded TPGS polymeric nanosuspension for drug resistive MCF-7 human breast cancer cells. Drug Dev. Ind. Pharm. 2018, 44, 1752–1761. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Zhen, Y.; Chen, Y.; Zou, L.; Zhang, Y.; Hu, F.; Feng, J.; Shen, J.; Wei, B. Naringin inhibits growth and induces apoptosis by a mechanism dependent on reduced activation of NF-κB/COX-2-caspase-1 pathway in HeLa cervical cancer cells. Int. J. Oncol. 2014, 45, 1929–1936. [Google Scholar] [CrossRef] [PubMed]
- Matejczyk, M.; Świsłocka, R.; Golonko, A.; Lewandowski, W.; Hawrylik, E. Cytotoxic, genotoxic and antimicrobial activity of caffeic and rosmarinic acids and their lithium, sodium and potassium salts as potential anticancer compounds. Adv. Med Sci. 2018, 63, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Fialová, S.; Kello, M.; Čoma, M.; Slobodníková, L.; Drobná, E.; Holková, I.; Garajová, M.; Garajová, M.; Zachar, V.; Lukáč, M. Derivatization of Rosmarinic Acid Enhances its in vitro Antitumor, Antimicrobial and Antiprotozoal Properties. Molecules 2019, 24, 1078. [Google Scholar] [CrossRef]
- Celiz, G.; Daz, M.; Audisio, M. Antibacterial activity of naringin derivatives against pathogenic strains. J. Appl. Microbiol. 2011, 111, 731–738. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, H.; Zhu, S.; Ma, C.; Wang, Z. Antibacterial Activity and Mechanism of Action of Chlorogenic Acid. J. Food Sci. 2011, 76, M398–M403. [Google Scholar] [CrossRef]
- Martínez, G.; Regente, M.; Jacobi, S.; Del Rio, M.; Pinedo, M.; De La Canal, L.; De Rio, M. Chlorogenic acid is a fungicide active against phytopathogenic fungi. Pestic. Biochem. Physiol. 2017, 140, 30–35. [Google Scholar] [CrossRef]
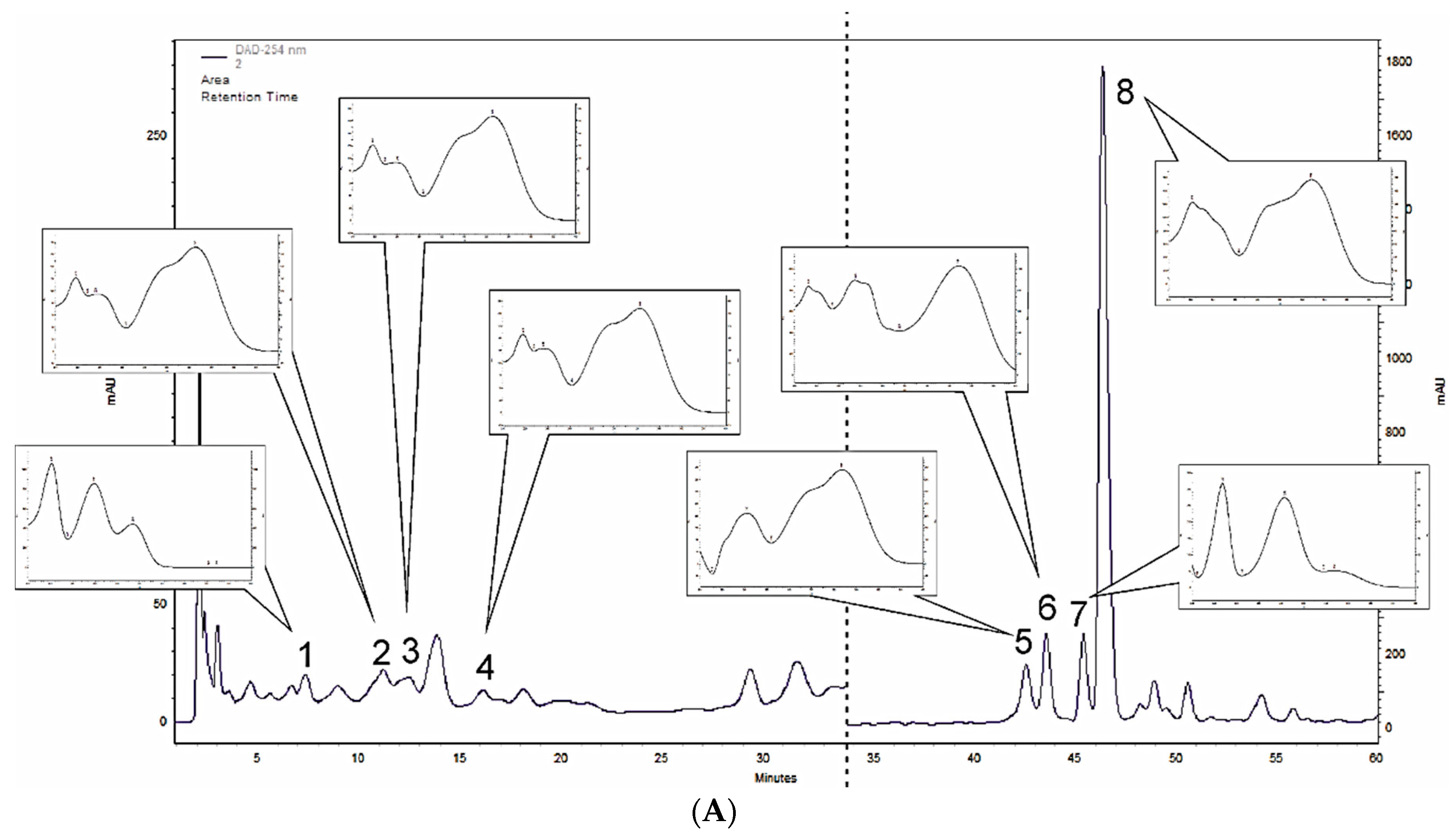
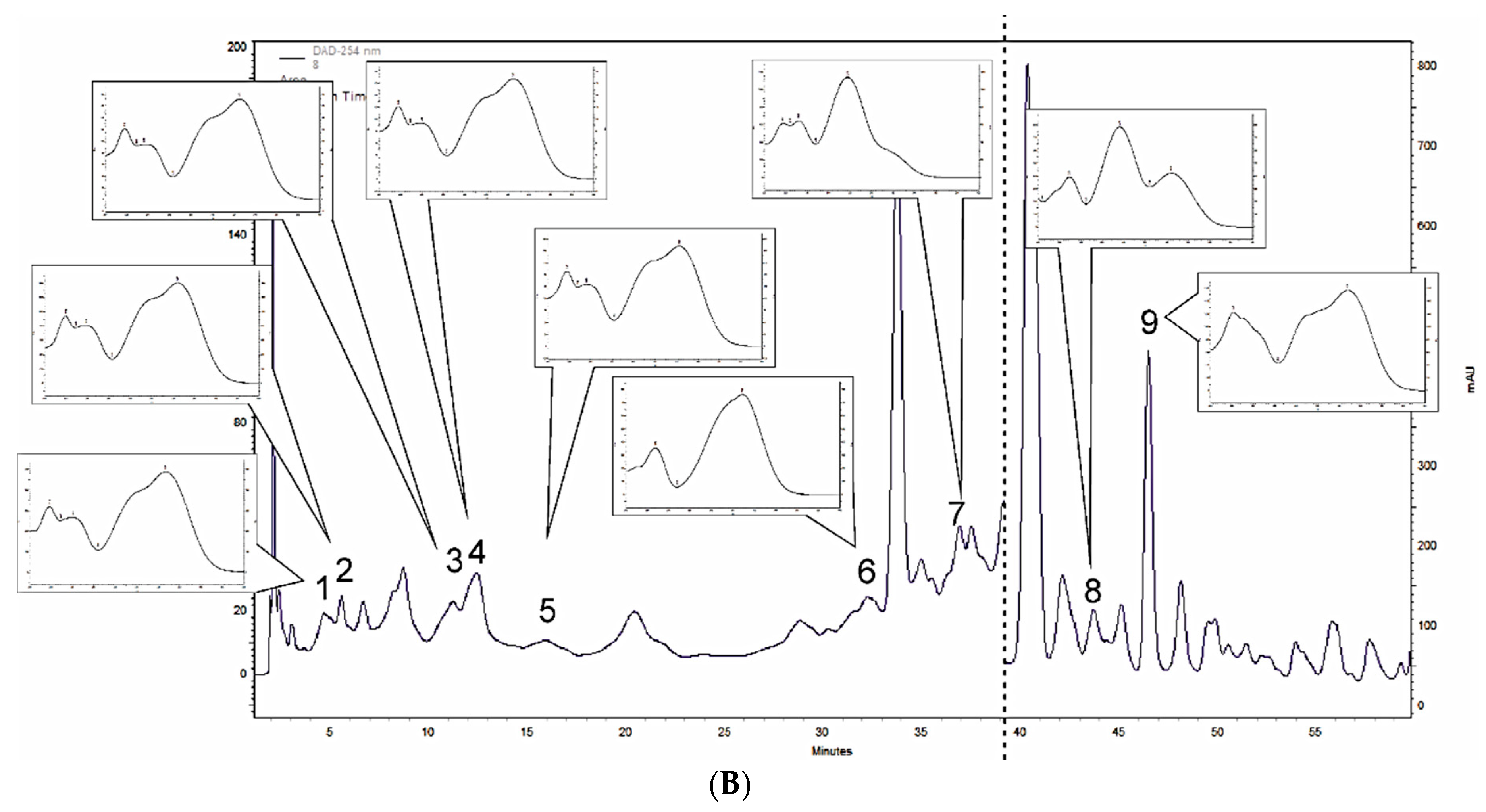
| Compound | M. × piperita | M. longifolia |
|---|---|---|
| Caffeic acid | 17.4 ± 1.5 | 19.6 ± 0.8 |
| Caftaric acid | nd * | 47.6 ± 5.7 |
| Chlorogenic acid | 69.4 ± 10.3 | 63.8 ± 9.3 |
| m-Coumaric acid | nd | 112.2 ± 4.7 |
| o-Coumaric acid | nd | 39.8 ± 0.7 |
| p-Coumaric acid | nd | 113.0 ± 15.2 |
| Cryptochlorogenic acid | 91.7 ± 6.4 | 191.1 ± 12.8 |
| Isochlorogenic acid | 10.3 ± 0.3 | nd |
| Neochlorogenic acid | nd | 56.8 ± 4.6 |
| Protocatechuic acid | 24.9 ± 2.1 | nd |
| Rosmarinic acid | 1 547.6 ± 59.5 | 781.6 ± 26.8 |
| Cynaroside(Luteolin-7-glucoside) | 162.8 ± 18.1 | nd |
| Naringin (Naringenin 7-rhamnoglucoside) | 328.8 ± 32.8 | nd |
| DPPH (IC50, µg/mL) | β-Carotene-Bleaching Assay (IC50, µg/mL) | FRAP (IC50, mM TEAC/g Extract) | |
|---|---|---|---|
| M. × piperita | 9.6 ± 0.1c | 11.7 ± 0.3c | 12.8 ± 0.3e |
| M. longifolia | 9.1 ± 0.1cd | 11.3 ± 0.2d | 12.0 ± 0.3e |
| Naringin | 117.3 ± 0.1d | 137.5 ± 0.1d | 158.1 ± 0.1d |
| Cynaroside | 6.3 ± 0.1d | 6.9 ± 0.1d | 8.2 ± 0.1d |
| Rosmarinic acid | 2.7 ± 0.1d | 3.1 ± 0.1d | 3.5 ± 0.7f |
| Cryptochlorogenic acid | 5.29 ± 0.1d | 6.3 ± 0.1d | 7.4 ± 0.2d |
| BHT | 2.6 ± 0.1e | 3.2 ± 0.1e | - |
| Trolox | - | - | 3.2 ± 0.1g |
| HeLa | HT-29 | MCF-7 | Jurkat | HEK-293 | |
|---|---|---|---|---|---|
| M. × piperita | 67.8 ± 3.5 | 44.62 ± 1.9 | 74.35 ± 2.1 | 89.66 ± 2.3 | >400 |
| M. longifolia | 79.31 ± 2.1 | 58.47 ± 1.3 | 83.61 ± 1.2 | 97.53 ± 2.7 | >400 |
| Naringin | 2.91 ± 2.1 | 20.54 ± 2.1 | 14.97 ± 0.5 | 43.79 ± 2.3 | >400 |
| Rosmarinic acid | 37.56 ± 1.0 | 25.98 ± 0.7 | 27.61 ± 1.1 | 49.58 ± 0.9 | >400 |
| Cryptochlorogenic acid | 5.7 ± 2.1 | 19.64 ± 0.3 | 47.31 ± 1.5 | 53.64 ± 1.1 | >400 |
| Vinblastine sulfate | 2.2 ± 0.05 | 17.13 ± 0.9 | ‒ | 0.1 ± 0.02 | 44.7 ± 0.7 |
| Taxol | ‒ | ‒ | 0.06 ± 0.005 | ‒ | ‒ |
| B. cereus MIC MBC | P. aeruginosa MIC MBC | L. monocytogenes MIC MBC | E. coli MIC MBC | M. flavus MIC MBC | S. aureus MIC MBC | |
|---|---|---|---|---|---|---|
| M. × piperita | 0.28 ± 0.01 | 0.16 ± 0.01 | 0.24 ± 0.01 | 0.21 ± 0.01 | 0.19 ± 0.01 | 0.17 ± 0.02 |
| 0.63 ± 0.03 | 0.47 ± 0.03 | 0.59 ± 0.03 | 0.49 ± 0.03 | 0.45 ± 0.03 | 0.37 ± 0.03 | |
| M. longifolia | 0.33 ± 0.02 | 0.26 ± 0.03 | 0.30 ± 0.02 | 0.31± 0.03 | 0.24 ± 0.03 | 0.25 ± 0.02 |
| 0.75 ± 0.04 | 0.63 ± 0.03 | 0.65 ± 0.03 | 0.67 ± 0.03 | 0.56 ± 0.02 | 0.55 ± 0.03 | |
| Naringin | 34.3 ± 2.1 | 43.1 ± 1.13 | 46.8 ± 6.11 | 41.7 ± 1.32 | 40.2 ± 1.42 | 43.9 ± 2.11 |
| >500 | >500 | >500 | >500 | >500 | >500 | |
| Rosmarinic acid | 40.5 ± 0.95 | 38.5 ± 2.12 | 48.5 ± 2.86 | 43.5 ± 2.33 | 30.5 ± 0.64 | 24.5 ± 1.53 |
| >500 | >500 | >500 | >500 | >500 | >500 | |
| Cryptochlorogenic acid | 0.15 ± 0.01 | 0.14 ± 0.02 | 0.14 ± 0.01 | 0.12 ± 0.01 | 0.12 ± 0.01 | 0.12 ± 0.01 |
| 0.37 ± 0.03 | 0.37 ± 0.03 | 0.37 ± 0.03 | 0.33 ± 0.03 | 0.32 ± 0.02 | 0.33 ± 0.03 | |
| Caffeic acid | 0.15 ± 0.01 | 0.07 ± 0.01 | 0.30 ± 0.03 | 0.12 ± 0.01 | 0.15 ± 0.02 | 0.21 ± 0.02 |
| 0.31 ± 0.03 | 0.14 ± 0.02 | 0.62 ± 0.05 | 0.26 ± 0.03 | 0.33 ± 0.02 | 0.42 ± 0.03 | |
| Streptomycin | 0.08 ± 0.01 | 0.09 ± 0.01 | 0.11 ± 0.02 | 0.11 ± 0.01 | 0.10 ± 0.01 | 0.15 ± 0.01 |
| 0.18 ± 0.02 | 0.18 ± 0.01 | 0.25 ± 0.01 | 0.22 ± 0.01 | 0.20 ± 0.01 | 0.32 ± 0.02 |
| A. flavus MIC MFC | A. ochraceus MIC MFC | A. niger MIC MFC | C. albicans MIC MFC | P. funiculosum MIC MFC | P. ochrochloron MIC MFC | |
|---|---|---|---|---|---|---|
| M. × piperita | 0.33 ± 0.02 | 0.56 ± 0.05 | 0.76 ± 0.04 | 0.98 ± 0.05 | 0.31 ± 0.01 | 0.39 ± 0.03 |
| 0.86 ± 0.03 | 1.36 ± 0.11 | 1.51 ± 0.13 | 1.34 ± 0.11 | 0.67 ± 0.03 | 0.95 ± 0.02 | |
| M. longifolia | 0.42 ± 0.03 | 0.74 ± 0.09 | 0.89 ± 0.05 | 1.06 ± 0.05 | 0.41 ± 0.01 | 0.47 ± 0.01 |
| 0.97 ± 0.05 | 1.88 ± 0.07 | 1.97 ± 0.07 | 1.72 ± 0.13 | 0.87 ± 0.03 | 1.07 ± 0.05 | |
| Naringin | 0.27 ± 0.01 | 0.25 ± 0.02 | 0.31 ± 0.01 | 0.35 ± 0.03 | 0.25 ± 0.02 | 0.47 ± 0.05 |
| 0.71 ± 0.03 | 0.64 ± 0.03 | 0.70 ± 0.03 | 0.86 ± 0.03 | 0.63 ± 0.04 | 1.11 ± 0.12 | |
| Rosmarinic acid | 189.31 ± 7.13 | 227.54 ± 7.85 | 236.13 ± 7.53 | 397.5 ± 9.32 | 223.54 ± 14.85 | 358.7 ± 8.21 |
| >1000 | >1000 | >1000 | >1000 | >1000 | >1000 | |
| Cryptochlorogenic acid | 5.11 ± 0.3 | 6.03 ± 0.21 | 6.47 ± 0.15 | 11.42 ± 0.18 | 25.74 ± 1.02 | 32.21 ± 2.53 |
| 25.45 ± 0.53 | 32.42 ± 1.64 | 39.62 ± 2.28 | 53.55 ± 1.16 | 64.32 ± 3.03 | 84.32 ± 2.65 | |
| Caffeic acid | 0.21 ± 0.01 | 0.24 ± 0.02 | 0.23 ± 0.02 | 0.33 ± 0.01 | 0.30 ± 0.03 | 0.21 ± 0.03 |
| 0.41 ± 0.05 | 0.50 ± 0.05 | 0.42 ± 0.02 | 0.70 ± 0.03 | 0.68 ± 0.03 | 0.43 ± 0.03 | |
| KTZ (Ketoconazolum) | 0.20 ± 0.01 | 0.19 ± 0.01 | 0.10± 0.01 | 0.20± 0.02 | 2.01 ± 0.16 | 0.21 ± 0.01 |
| 0.43 ± 0.03 | 0.42 ± 0.03 | 0.22 ± 0.02 | 0.45 ± 0.03 | 3.64 ± 0.18 | 0.43 ± 0.03 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elansary, H.O.; Szopa, A.; Kubica, P.; Ekiert, H.; Klimek-Szczykutowicz, M.; El-Ansary, D.O.; Mahmoud, E.A. Polyphenol Profile and Antimicrobial and Cytotoxic Activities of Natural Mentha × piperita and Mentha longifolia Populations in Northern Saudi Arabia. Processes 2020, 8, 479. https://doi.org/10.3390/pr8040479
Elansary HO, Szopa A, Kubica P, Ekiert H, Klimek-Szczykutowicz M, El-Ansary DO, Mahmoud EA. Polyphenol Profile and Antimicrobial and Cytotoxic Activities of Natural Mentha × piperita and Mentha longifolia Populations in Northern Saudi Arabia. Processes. 2020; 8(4):479. https://doi.org/10.3390/pr8040479
Chicago/Turabian StyleElansary, Hosam O., Agnieszka Szopa, Paweł Kubica, Halina Ekiert, Marta Klimek-Szczykutowicz, Diaa O. El-Ansary, and Eman A. Mahmoud. 2020. "Polyphenol Profile and Antimicrobial and Cytotoxic Activities of Natural Mentha × piperita and Mentha longifolia Populations in Northern Saudi Arabia" Processes 8, no. 4: 479. https://doi.org/10.3390/pr8040479
APA StyleElansary, H. O., Szopa, A., Kubica, P., Ekiert, H., Klimek-Szczykutowicz, M., El-Ansary, D. O., & Mahmoud, E. A. (2020). Polyphenol Profile and Antimicrobial and Cytotoxic Activities of Natural Mentha × piperita and Mentha longifolia Populations in Northern Saudi Arabia. Processes, 8(4), 479. https://doi.org/10.3390/pr8040479








