Robust Model Selection: Flatness-Based Optimal Experimental Design for a Biocatalytic Reaction
Abstract
1. Introduction
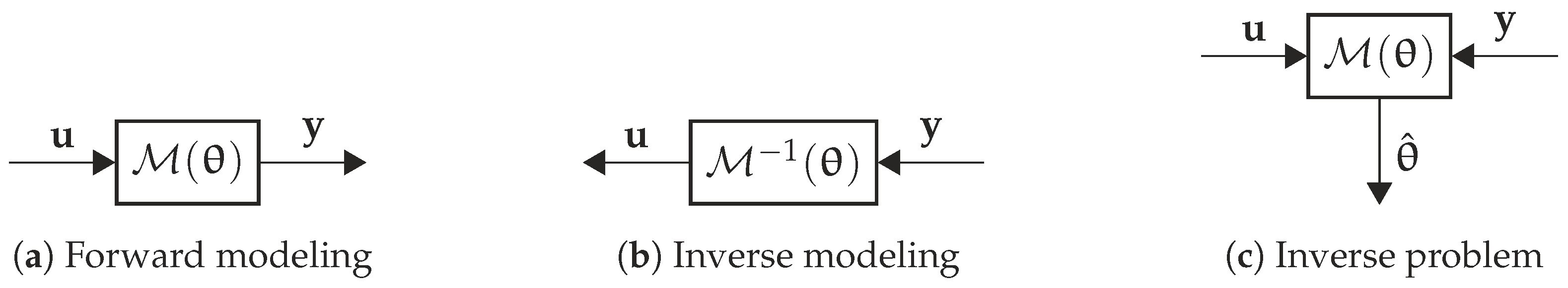
2. Inverse Modeling
2.1. Differential Flatness
- The states and the controls can be described as a function of the flat output and its derivatives:with the smooth mapping functions and .
- The dimensions of the control and the flat output vector are equal:
2.2. Flat Output Identification
2.3. Basis Splines
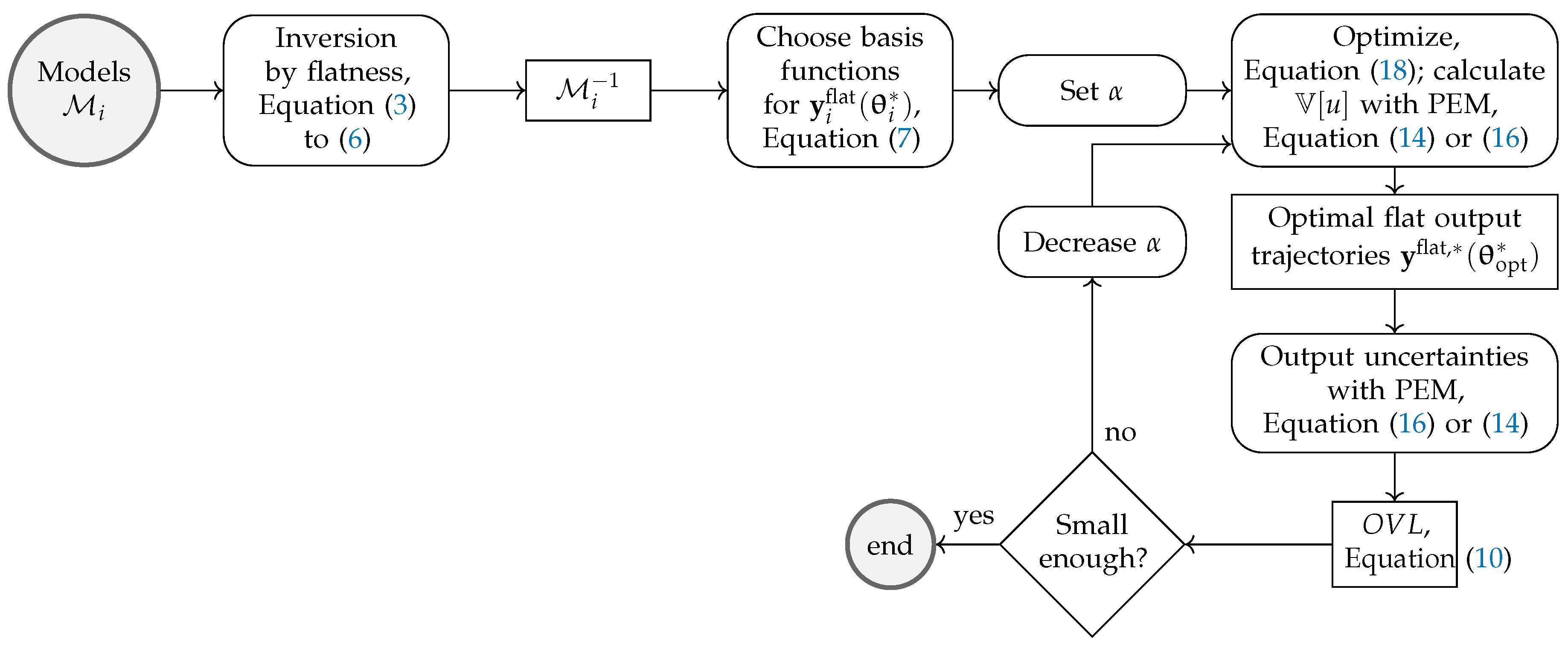
3. Robust Design of Experiments for Model Selection
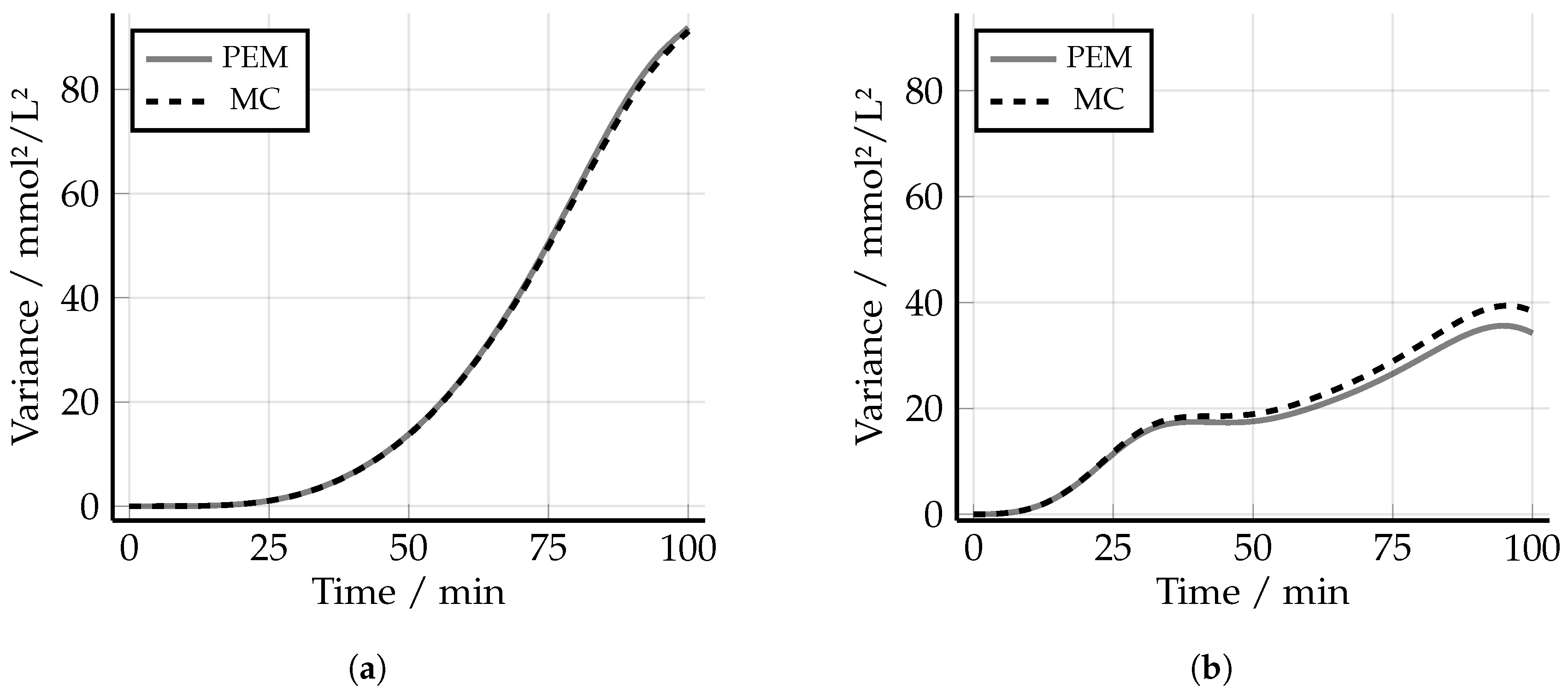
3.1. Point Estimate Method
3.2. Robust Flatness-Based Objective Function
3.3. Computational Methods
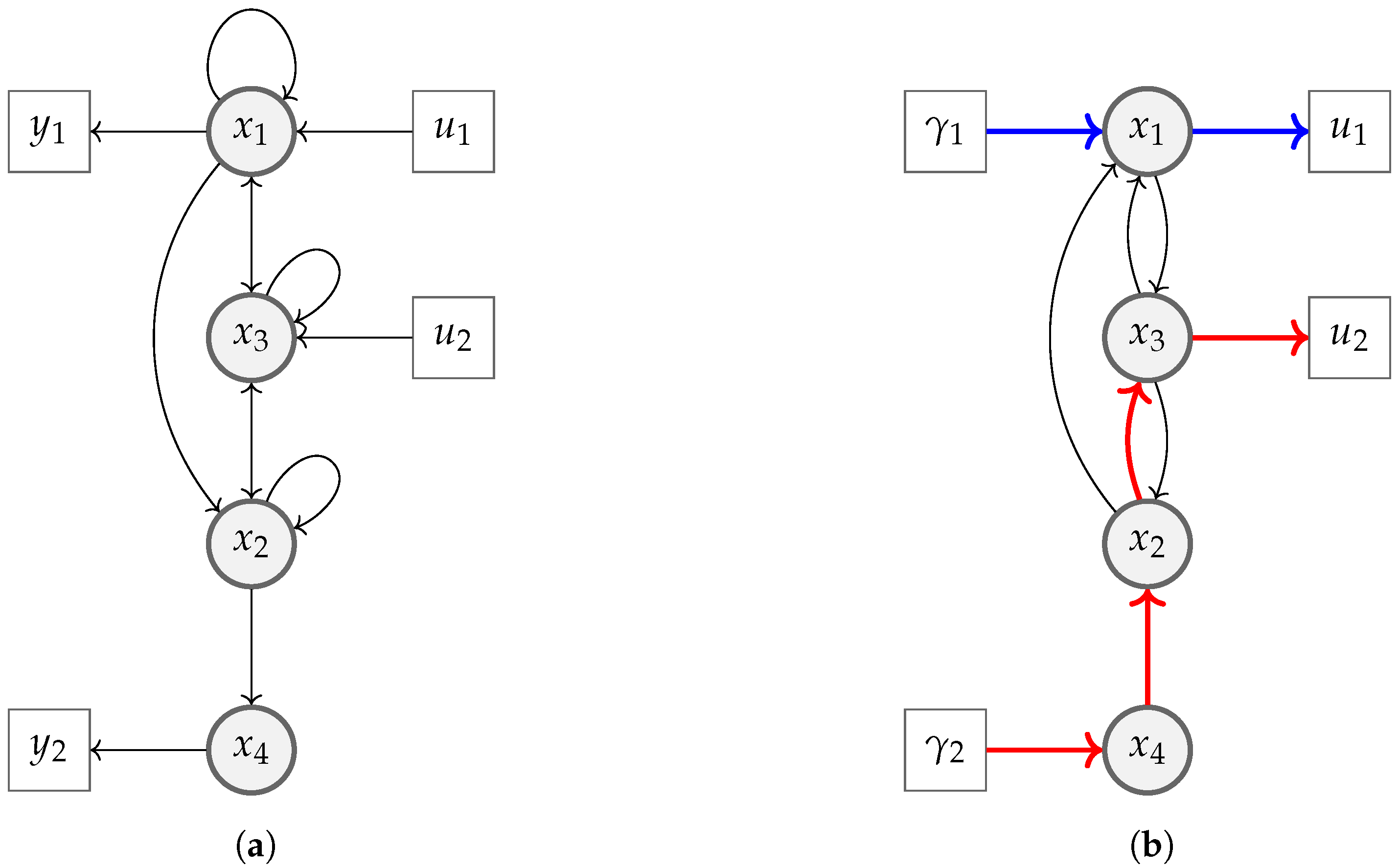
4. Case Study
5. Results
5.1. Differential Flatness Property
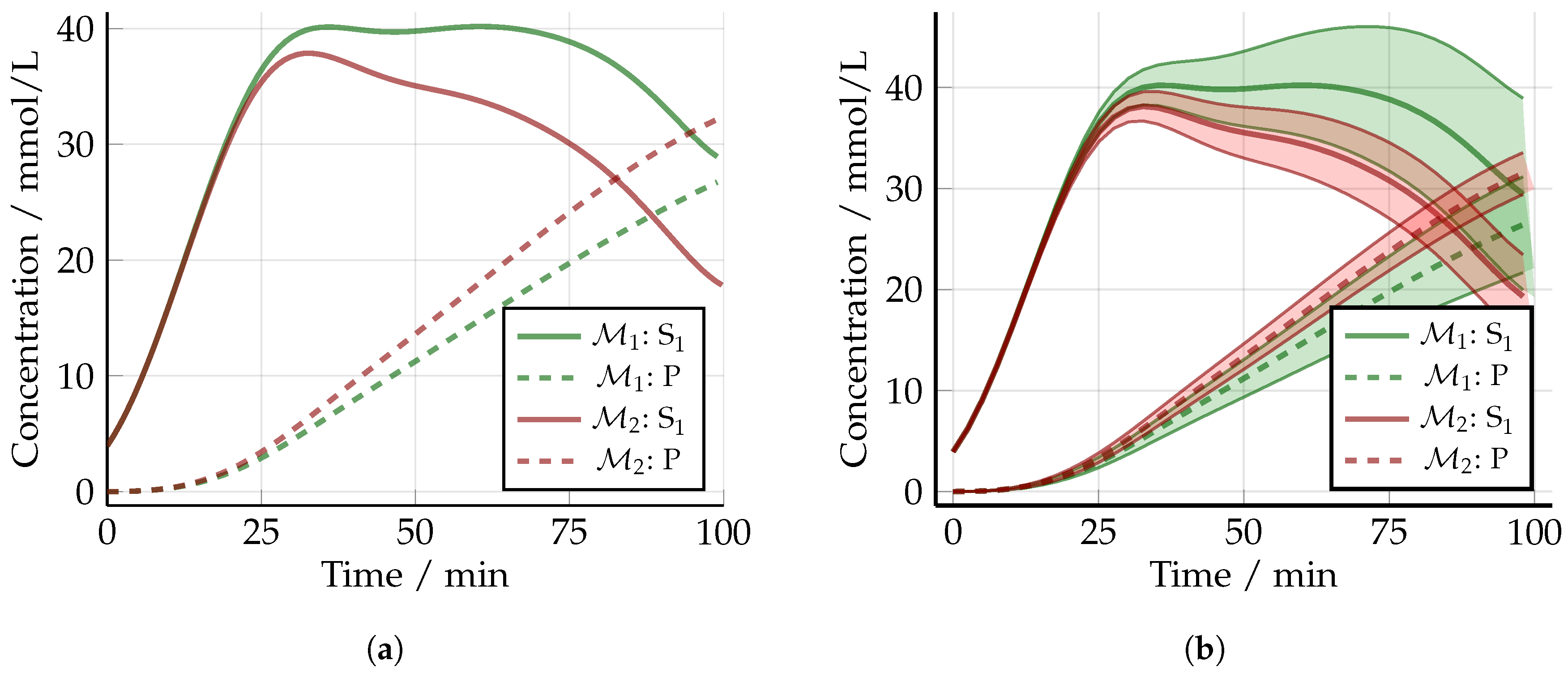
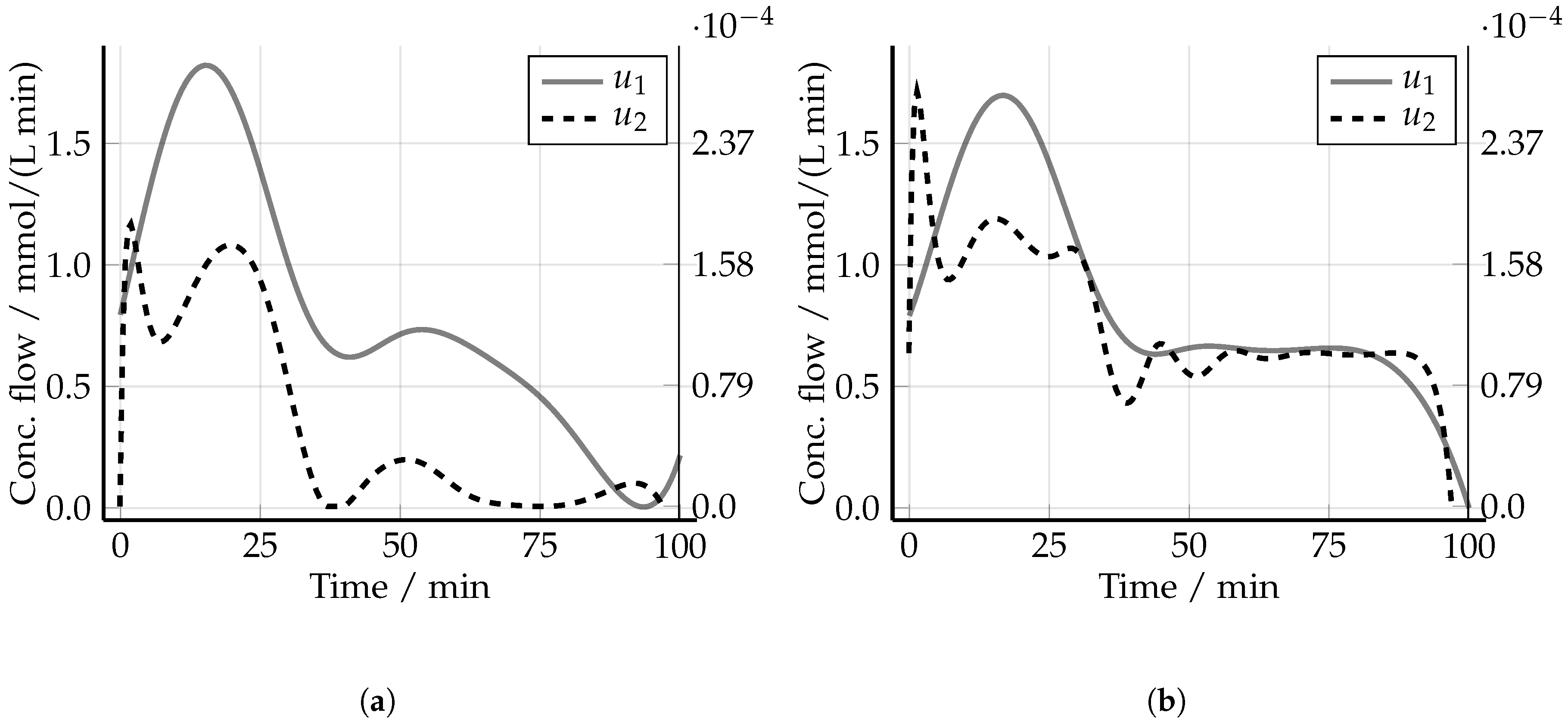
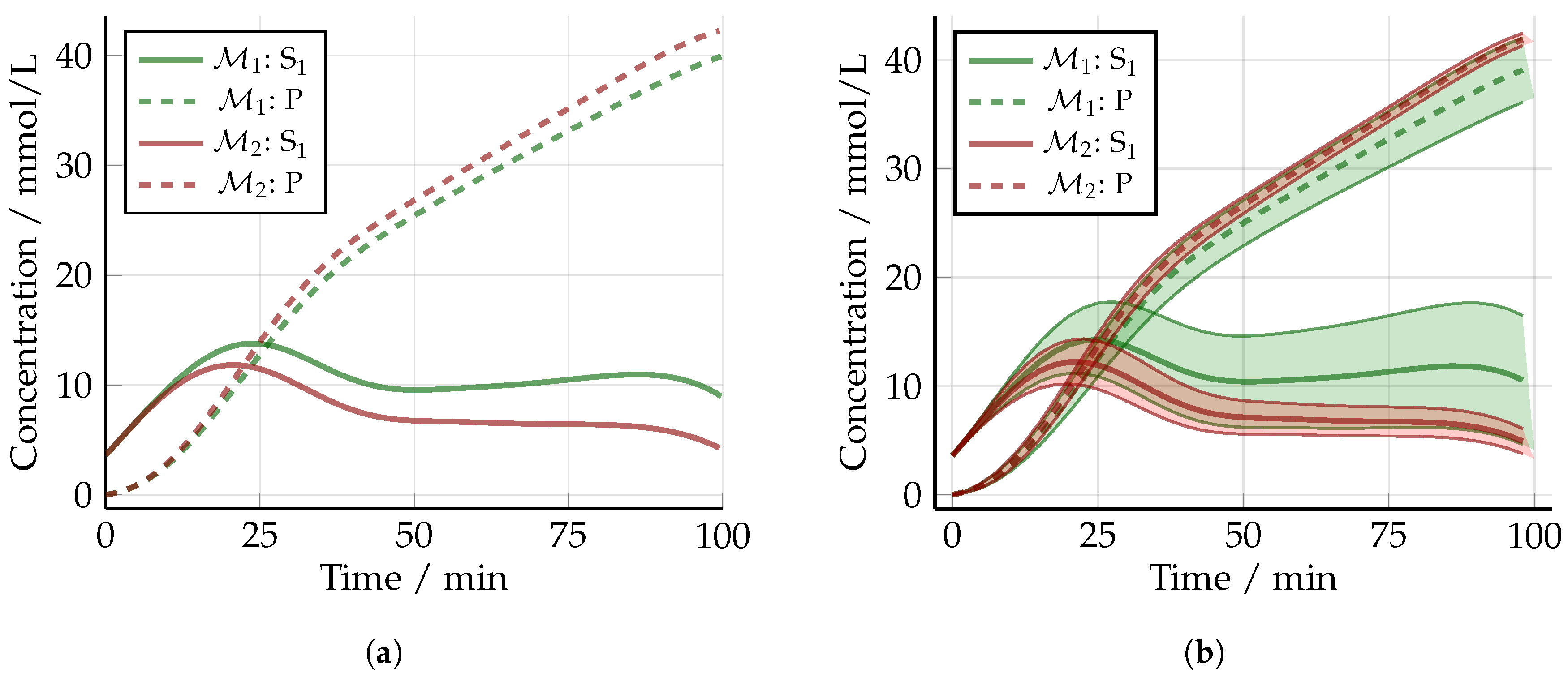
5.2. Non-Optimized Experimental Design
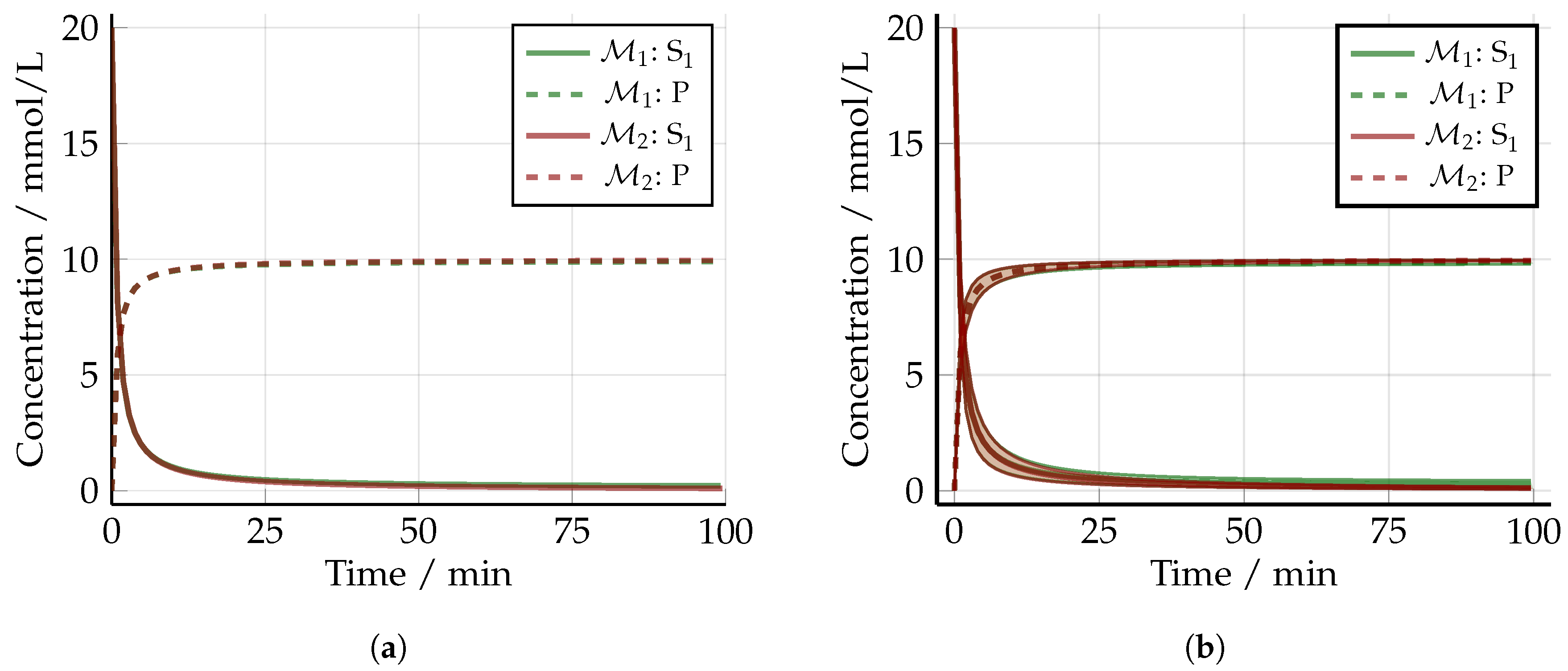
5.3. Optimized Experimental Design without Uncertainty: The Nominal Case
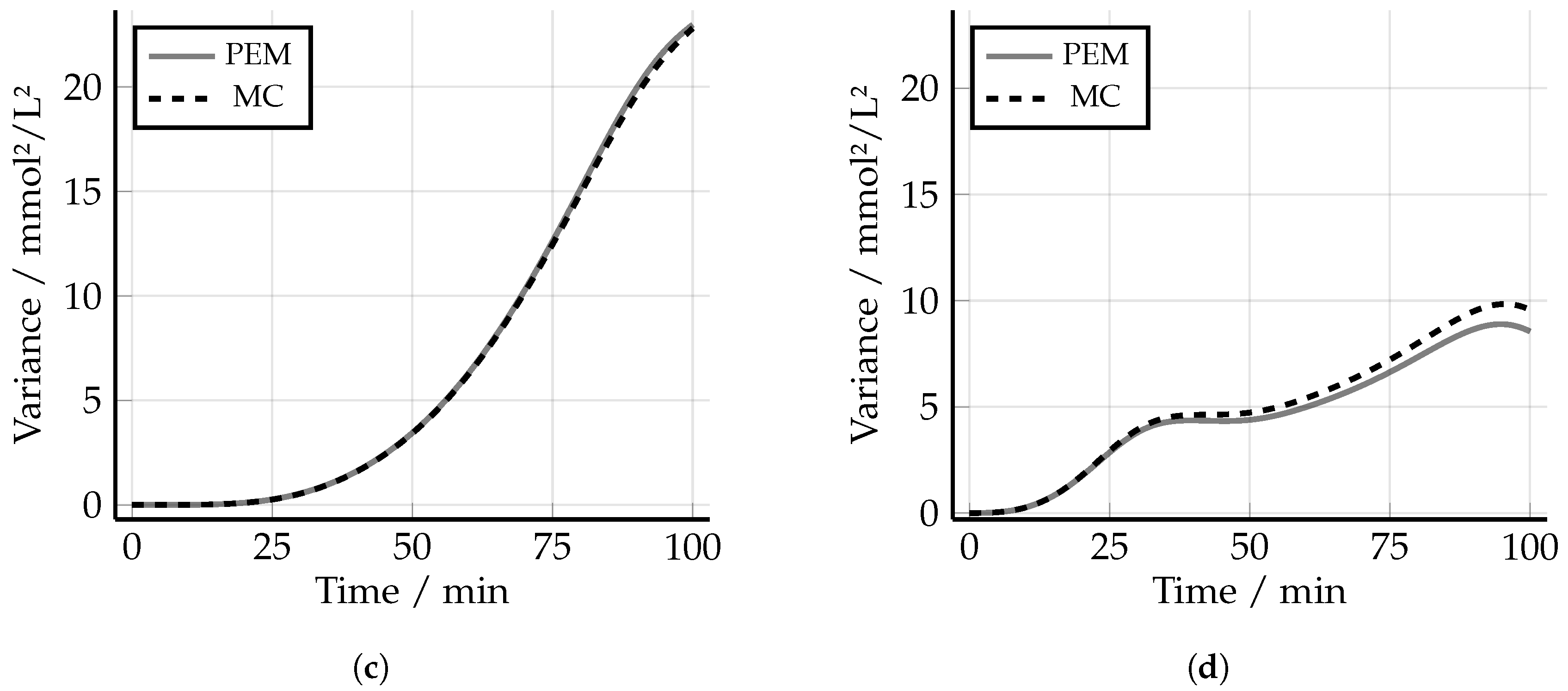
5.4. Robust Experimental Design
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Su, Q.; Ganesh, S.; Moreno, M.; Bommireddy, Y.; Gonzalez, M.; Reklaitis, G.V.; Nagy, Z.K. A Perspective on Quality-by-Control (QbC) in Pharmaceutical Continuous Manufacturing. Comput. Chem. Eng. 2019, 125, 216–231. [Google Scholar] [CrossRef]
- Taylor, D. The Pharmaceutical Industry and the Future of Drug Development. In Issues in Environmental Science and Technology; Hester, R.E., Harrison, R.M., Eds.; Royal Society of Chemistry: Cambridge, UK, 2015; pp. 1–33. [Google Scholar]
- Emenike, V.N.; Xie, X.; Schenkendorf, R.; Spiess, A.C.; Krewer, U. Robust Dynamic Optimization of Enzyme-Catalyzed Carboligation: A Point Estimate-Based Back-off Approach. Comput. Chem. Eng. 2019, 121, 232–247. [Google Scholar] [CrossRef]
- Kroll, P.; Hofer, A.; Ulonska, S.; Kager, J.; Herwig, C. Model-Based Methods in the Biopharmaceutical Process Lifecycle. Pharm. Res. 2017, 34, 2596–2613. [Google Scholar] [CrossRef] [PubMed]
- Pörtner, R.; Barradas, O.P.; Frahm, B.; Hass, V. 16–Advanced Process and Control Strategies for Bioreactors. In Current Developments in Biotechnology and Bioengineering; Larroche, C., Sanromán, M.Á., Du, G., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 463–493. [Google Scholar]
- Abt, V.; Barz, T.; Cruz, N.; Herwig, C.; Kroll, P.; Möller, J.; Pörtner, R.; Schenkendorf, R. Model-Based Tools for Optimal Experiments in Bioprocess Engineering. Curr. Opin. Chem. Eng. 2018, 22, 244–252. [Google Scholar] [CrossRef]
- Stamati, I.; Logist, F.; Akkermans, S.; Fernández, E.N.; Impe, J.V. On the Effect of Sampling Rate and Experimental Noise in the Discrimination between Microbial Growth Models in the Suboptimal Temperature Range. Comput. Chem. Eng. 2016, 85, 84–93. [Google Scholar] [CrossRef][Green Version]
- Telen, D.; Logist, F.; Van Derlinden, E.; Van Impe, J. Approximate Robust Optimal Experiment Design in Dynamic Bioprocess Models. In Proceedings of the 20th Mediterranean Conference on Control & Automation (MED), Barcelona, Spain, 3–6 July 2012; pp. 157–162. [Google Scholar]
- Skanda, D.; Lebiedz, D. An Optimal Experimental Design Approach to Model Discrimination in Dynamic Biochemical Systems. Bioinformatics 2010, 26, 939–945. [Google Scholar] [CrossRef]
- Franceschini, G.; Macchietto, S. Model-Based Design of Experiments for Parameter Precision: State of the Art. Chem. Eng. Sci. 2008, 63, 4846–4872. [Google Scholar] [CrossRef]
- Biral, F.; Bertolazzi, E.; Bosetti, P. Notes on Numerical Methods for Solving Optimal Control Problems. IEEJ J. Ind. Appl. 2016, 5, 154–166. [Google Scholar] [CrossRef]
- Rodrigues, D.; Billeter, J.; Bonvin, D. Maximum-Likelihood Estimation of Kinetic Parameters via the Extent-Based Incremental Approach. Comput. Chem. Eng. 2018, 122, 152–171. [Google Scholar] [CrossRef]
- Wang, Y.; Biegler, L.T.; Patel, M.; Wassick, J. Parameters Estimation and Model Discrimination for Solid-Liquid Reactions in Batch Processes. Chem. Eng. Sci. 2018, 187, 455–469. [Google Scholar] [CrossRef]
- Barz, T.; Bournazou, M.N.C.; Körkel, S.; Walter, S.F. Real-Time Adaptive Input Design for the Determination of Competitive Adsorption Isotherms in Liquid Chromatography. Comput. Chem. Eng. 2016, 94, 104–116. [Google Scholar] [CrossRef]
- Liu, J.; Mendoza, S.; Li, G.; Fathy, H. Efficient total least squares state and parameter estimation for differentially flat systems. In Proceedings of the American Control Conference, Boston, MA, USA, 6–8 July 2016; pp. 5419–5424. [Google Scholar] [CrossRef]
- Schenkendorf, R.; Mangold, M. Parameter Identification for Ordinary and Delay Differential Equations by Using Flat Inputs. Theor. Found. Chem. Eng. 2014, 48, 594–607. [Google Scholar] [CrossRef]
- Fliess, M.; Lévine, J.; Martin, P.; Rouchon, P. Sur Les Systèmes Non Linéaires Différentiellement Plats. C. R. Acad. Sci. Paris 1992, 619–624. [Google Scholar]
- Biegler, L.T. Nonlinear Programming: Concepts, Algorithms, and Applications to Chemical Processes; MOS-SIAM Series on Optimization; Society for Industrial and Applied Mathematics: Philadelphia, PA, USA, 2010. [Google Scholar]
- Franke, M.; Robenack, K. On the Computation of Flat Outputs for Nonlinear Control Systems. In Proceedings of the 2013 European Control Conference (ECC), Zurich, Switzerland, 17–19 July 2013; pp. 167–172. [Google Scholar]
- Schulze, M.; Schenkendorf, R. Flatness-Based Design of Experiments for Model Selection. IFAC-PapersOnLine 2018, 51, 233–238. [Google Scholar] [CrossRef]
- Schenkendorf, R.; Reichl, U.; Mangold, M. Parameter Identification of Time-Delay Systems: A Flatness Based Approach. IFAC Proc. Vol. 2012, 45, 165–170. [Google Scholar] [CrossRef]
- Hildebrand, F.; Kühl, S.; Pohl, M.; Vasic-Racki, D.; Müller, M.; Wandrey, C.; Lütz, S. The Production of (R)-2-Hydroxy-1-Phenyl-Propan-1-One Derivatives by Benzaldehyde Lyase from Pseudomonasfluorescens in a Continuously Operated Membrane Reactor. Biotechnol. Bioeng. 2007, 96, 835–843. [Google Scholar] [CrossRef]
- Švarc, A.; Findrik Blažević, Z.; Vasić-Rački, Đ.; Szekrenyi, A.; Fessner, W.D.; Charnock, S.J.; Vrsalović Presečki, A. 2-Deoxyribose-5-phosphate Aldolase from Thermotogamaritima in the Synthesis of a Statin Side-chain Precursor: Characterization, Modeling and Optimization. J. Chem. Technol. Biotechnol. 2019, 94, 1832–1842. [Google Scholar] [CrossRef]
- Hampel, S.; Steitz, J.P.; Baierl, A.; Lehwald, P.; Wiesli, L.; Richter, M.; Fries, A.; Pohl, M.; Schneider, G.; Dobritzsch, D.; et al. Structural and Mutagenesis Studies of the Thiamine-Dependent, Ketone-Accepting YerE from Pseudomonas protegens. ChemBioChem 2018, 19, 2283–2292. [Google Scholar] [CrossRef]
- Galvanin, F.; Barolo, M.; Bezzo, F.; Macchietto, S. A backoff strategy for model-based experiment design under parametric uncertainty. AIChE J. 2009, 56, 2088–2102. [Google Scholar] [CrossRef]
- Jost, F.; Sager, S.; Le, T.T.T. A Feedback Optimal Control Algorithm with Optimal Measurement Time Points. Processes 2017, 5, 10. [Google Scholar] [CrossRef]
- Buzzi-Ferraris, G.; Forzatti, P. A new sequential experimental design procedure for discriminating among rival models. Chem. Eng. Sci. 1983, 38, 225–232. [Google Scholar] [CrossRef]
- Donckels, B.M.; Pauw, D.J.D.; Baets, B.D.; Maertens, J.; Vanrolleghem, P.A. An anticipatory approach to optimal experimental design for model discrimination. Chemom. Intell. Lab. Syst. 2009, 95, 53–63. [Google Scholar] [CrossRef]
- Violet, L.; Loubière, K.; Rabion, A.; Samuel, R.; Hattou, S.; Cabassud, M.; Prat, L. Stoichio-kinetic model discrimination and parameter identification in continuous microreactors. Chem. Eng. Res. Des. 2016, 114, 39–51. [Google Scholar] [CrossRef]
- Schenkendorf, R.; Xie, X.; Rehbein, M.; Scholl, S.; Krewer, U. The Impact of Global Sensitivities and Design Measures in Model-Based Optimal Experimental Design. Processes 2018, 6, 27. [Google Scholar] [CrossRef]
- Schöniger, A.; Wöhling, T.; Samaniego, L.; Nowak, W. Model Selection on Solid Ground: Rigorous Comparison of Nine Ways to Evaluate Bayesian Model Evidence. Water Resour. Res. 2014, 50, 9484–9513. [Google Scholar] [CrossRef]
- Brunetti, C.; Linde, N. Impact of petrophysical uncertainty on Bayesian hydrogeophysical inversion and model selection. Adv. Water Resour. 2018, 111, 346–359. [Google Scholar] [CrossRef]
- Lerner, U.N. Hybrid Bayesian Networks for Reasoning about Complex Systems. Ph.D. Thesis, Stanford University Stanford, Stanford, CA, USA, 2002. [Google Scholar]
- Xie, X.; Schenkendorf, R.; Krewer, U. Toward a Comprehensive and Efficient Robust Optimization Framework for (Bio)chemical Processes. Processes 2018, 6, 183. [Google Scholar] [CrossRef]
- Walter, E.E.; Pronzato, L. Identification of Parametric Models from Experimental Data; Springer: Berlin, Germany, 1997; p. 413. [Google Scholar]
- Buchholz, J.; Grünhagen, W.V. Inversion Impossible; GRIN Publishing GmbH: Munich, Germany, 2007. [Google Scholar]
- Lu, L.; Murray-Smith, D.J.; Thomson, D. Issues of numerical accuracy and stability in inverse simulation. Simul. Model. Pract. Theory 2008, 16, 1350–1364. [Google Scholar] [CrossRef]
- Lu, L. Inverse Modelling and Inverse Simulation for System Engineering and Control Applications. Ph.D. Thesis, University of Glasgow, Glasgow, UK, 2007. [Google Scholar]
- Wey, T. Nichtlineare Regelungssysteme: Ein Differentialalgebraischer Ansatz; mit 13 Tabellen, 1st ed.; OCLC: 76389242; Teubner: Stuttgart, Germany, 2002. [Google Scholar]
- Adamy, J. Nichtlineare Systeme und Regelungen; Springer Vieweg: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Waldherr, S.; Zeitz, M. Conditions for the Existence of a Flat Input. Int. J. Control. 2008, 81, 439–443. [Google Scholar] [CrossRef]
- Kaminski, Y.J.; Lévine, J.; Ollivier, F. Intrinsic and Apparent Singularities in Differentially Flat Systems, and Application to Global Motion Planning. Syst. Control. Lett. 2018, 113, 117–124. [Google Scholar] [CrossRef]
- Rigatos, G.G. Nonlinear Control and Filtering Using Differential Flatness Approaches. In Studies in Systems, Decision and Control; Springer International Publishing: Cham, Switzerland, 2015; Volume 25. [Google Scholar]
- Fliess, M.; Mounier, H.; Rouchon, P.; Rudolph, J. A Distributed Parameter Approach to the Control of a Tubular Reactor: A Multivariable Case. In Proceedings of the 37th IEEE Conference on Decision and Control (Cat. No. 98CH36171), Tampa, FL, USA, 18 December 1998; pp. 439–442. [Google Scholar]
- Andrej, J.; Meurer, T. Flatness-Based Constrained Optimal Control of Reaction-Diffusion Systems. In Proceedings of the 2018 Annual American Control Conference (ACC), Milwaukee, WI, USA, 27–29 June 2018; p. 6. [Google Scholar]
- Kolar, B.; Diwold, J.; Schöberl, M. Necessary and Sufficient Conditions for Difference Flatness. arXiv 2019, arXiv:1909.02868. [Google Scholar]
- Wagner, M.O.; Meurer, T.; Kugi, A. Trajectory Planning for Semilinear PDEs Modeling a Countercurrent Heat Exchanger. IFAC Proc. Vol. 2010, 43, 593–598. [Google Scholar] [CrossRef]
- Vollmer, U.; Raisch, J. Control of Batch Crystallization—A System Inversion Approach. Chem. Eng. Process. Process. Intensif. 2006, 45, 874–885. [Google Scholar] [CrossRef]
- Mahadevan, R.; Agrawal, S.K.; Doyle III, F.J. Differential Flatness Based Nonlinear Predictive Control of Fed-Batch Bioreactors. Control. Eng. Pract. 2001, 9, 889–899. [Google Scholar] [CrossRef]
- Liu, J.; Li, G.; Fathy, H.K. An Extended Differential Flatness Approach for the Health-Conscious Nonlinear Model Predictive Control of Lithium-Ion Batteries. IEEE Trans. Control. Syst. Technol. 2017, 25, 1882–1889. [Google Scholar] [CrossRef]
- Kolar, B.; Kaldmäe, A.; Schöberl, M.; Kotta, Ü.; Schlacher, K. Construction of Flat Outputs of Nonlinear Discrete-Time Systems in a Geometric and an Algebraic Framework. IFAC-PapersOnLine 2016, 49, 796–801. [Google Scholar] [CrossRef]
- Victor, S.; Melchior, P.; Lévine, J.; Oustaloup, A. Flat Output Computation for Fractional Linear Systems: Application to a Thermal System. IFAC Proc. Vol. 2014, 47, 2891–2896. [Google Scholar] [CrossRef]
- Waldherr, S.; Zeitz, M. Flat Inputs in the MIMO Case. IFAC Proc. Vol. 2010, 43, 695–700. [Google Scholar] [CrossRef]
- Richard, P.; Buisson, J.; Cormerais, H. Analysis of Flatness Using Bond Graphs and Bicausality. IFAC Proc. Vol. 2002, 35, 25–30. [Google Scholar] [CrossRef]
- Csercsik, D.; Szederkényi, G.; Hangos, K.M. Determining Flat Outputs of MIMO Nonlinear Systems Using Directed Graphs. In Proceedings of the 8th Portuguese Conference on Automatic Control (CONTROLO), Vila Real, Portugal, 21–23 July 2008; p. 6. [Google Scholar]
- Schizas, C.; Evans, F. A graph theoretic approach to multivariable control system design. Automatica 1981, 17, 371–377. [Google Scholar] [CrossRef]
- Reinschke, K.J. Multivariable Control: A Graph-Theoretic Approach; Springer: Berlin, Germany, 1988. [Google Scholar]
- Dion, J.M.; Commault, C.; van der Woude, J. Generic properties and control of linear structured systems: A survey. Automatica 2003, 39, 1125–1144. [Google Scholar] [CrossRef]
- de Boor, C. A Practical Guide to Splines; Number 27 in Applied Mathematical Sciences; Springer: New York, NY, USA, 2001. [Google Scholar]
- Vassiliadis, V.; Sargent, R.; Pantelides, C. Solution of a class of multistage dynamic optimization problems. 1. Problems without path constraints. Ind. Eng. Chem. Res. 1994, 33, 2111–2122. [Google Scholar] [CrossRef]
- Vassiliadis, V.; Sargent, R.; Pantelides, C. Solution of a class of multistage dynamic optimization problems. 2. Problems with path constraints. Ind. Eng. Chem. Res. 1994, 33, 2123. [Google Scholar] [CrossRef]
- Kremling, A.; Fischer, S.; Gadkar, K.; Doyle, F.J.; Sauter, T.; Bullinger, E.; Allgöwer, F.; Gilles, E.D. A benchmark for methods in reverse engineering and model discrimination: Problem formulation and solutions. Genome Res. 2004, 14, 1773–1785. [Google Scholar] [CrossRef] [PubMed]
- Alberton, A.L.; Schwaab, M.; Lobão, M.W.N.; Pinto, J.C. Experimental design for the joint model discrimination and precise parameter estimation through information measures. Chem. Eng. Sci. 2011, 66, 1940–1952. [Google Scholar] [CrossRef]
- Schenkendorf, R.; Mangold, M. Online model selection approach based on Unscented Kalman Filtering. J. Process. Control. 2013, 23, 44–57. [Google Scholar] [CrossRef]
- Vanlier, J.; Tiemann, C.A.; Hilbers, P.A.; van Riel, N.A. Optimal experiment design for model selection in biochemical networks. BMC Syst. Biol. 2014, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Martin-Casas, M.; Mesbah, A. Discrimination Between Competing Model Structures of Biological Systems in the Presence of Population Heterogeneity. IEEE Life Sci. Lett. 2016, 2, 23–26. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; OCLC: ocm48557578; Springer: New York, NY, USA, 2002. [Google Scholar]
- Lorenz, S.; Diederichs, E.; Telgmann, R.; Schütte, C. Discrimination of Dynamical System Models for Biological and Chemical Processes: Discrimination of Dynamical System Models. J. Comput. Chem. 2007, 28, 1384–1399. [Google Scholar] [CrossRef]
- Rao, M.M.; Swift, R.J. Probability Theory with Applications; Springer: Berlin, Germany, 2006. [Google Scholar]
- Xie, X.; Krewer, U.; Schenkendorf, R. Robust Optimization of Dynamical Systems with Correlated Random Variables Using the Point Estimate Method. IFAC-PapersOnLine 2018, 51, 427–432. [Google Scholar] [CrossRef]
- Xie, X.; Schenkendorf, R. Robust Process Design in Pharmaceutical Manufacturing under Batch-to-Batch Variation. Processes 2019, 7, 509. [Google Scholar] [CrossRef]
- Bezanson, J.; Edelman, A.; Karpinski, S.; Shah, V.B. Julia: A Fresh Approach to Numerical Computing. SIAM Rev. 2017, 59, 65–98. [Google Scholar] [CrossRef]
- Rackauckas, C.; Nie, Q. Differential Equations. Jl—A Performant and Feature-Rich Ecosystem for Solving Differential Equations in Julia. J. Open Res. Softw. 2017, 5, 15. [Google Scholar] [CrossRef]
- Dunning, I.; Huchette, J.; Lubin, M. JuMP: A Modeling Language for Mathematical Optimization. SIAM Rev. 2017, 59, 295–320. [Google Scholar] [CrossRef]
- Wächter, A.; Biegler, L.T. On the Implementation of an Interior-Point Filter Line-Search Algorithm for Large-Scale Nonlinear Programming. Math. Program. 2006, 106, 25–57. [Google Scholar] [CrossRef]
- Ohs, R.; Leipnitz, M.; Schöpping, M.; Spiess, A.C. Simultaneous Identification of Reaction and Inactivation Kinetics of an Enzyme-catalyzed Carboligation. Biotechnol. Prog. 2018, 34, 1081–1092. [Google Scholar] [CrossRef]
- Fang, J.J.; Hung, C.L. An Improved Parameterization Method for B-Spline Curve and Surface Interpolation. Comput.-Aided Des. 2013, 45, 1005–1028. [Google Scholar] [CrossRef]

| Approximation Scheme | ||||
|---|---|---|---|---|
| PEM5 (Equations (14a) and (14b)) | ||||
| PEM3 (Equations (16a) and (16b)) | - |
| Model |
|---|
| Model |
|---|
| Design | None | Nominal | PEM3 | PEM5 |
|---|---|---|---|---|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schulze, M.; Schenkendorf, R. Robust Model Selection: Flatness-Based Optimal Experimental Design for a Biocatalytic Reaction. Processes 2020, 8, 190. https://doi.org/10.3390/pr8020190
Schulze M, Schenkendorf R. Robust Model Selection: Flatness-Based Optimal Experimental Design for a Biocatalytic Reaction. Processes. 2020; 8(2):190. https://doi.org/10.3390/pr8020190
Chicago/Turabian StyleSchulze, Moritz, and René Schenkendorf. 2020. "Robust Model Selection: Flatness-Based Optimal Experimental Design for a Biocatalytic Reaction" Processes 8, no. 2: 190. https://doi.org/10.3390/pr8020190
APA StyleSchulze, M., & Schenkendorf, R. (2020). Robust Model Selection: Flatness-Based Optimal Experimental Design for a Biocatalytic Reaction. Processes, 8(2), 190. https://doi.org/10.3390/pr8020190






