Promising Catalytic Systems for CO2 Hydrogenation into CH4: A Review of Recent Studies
Abstract
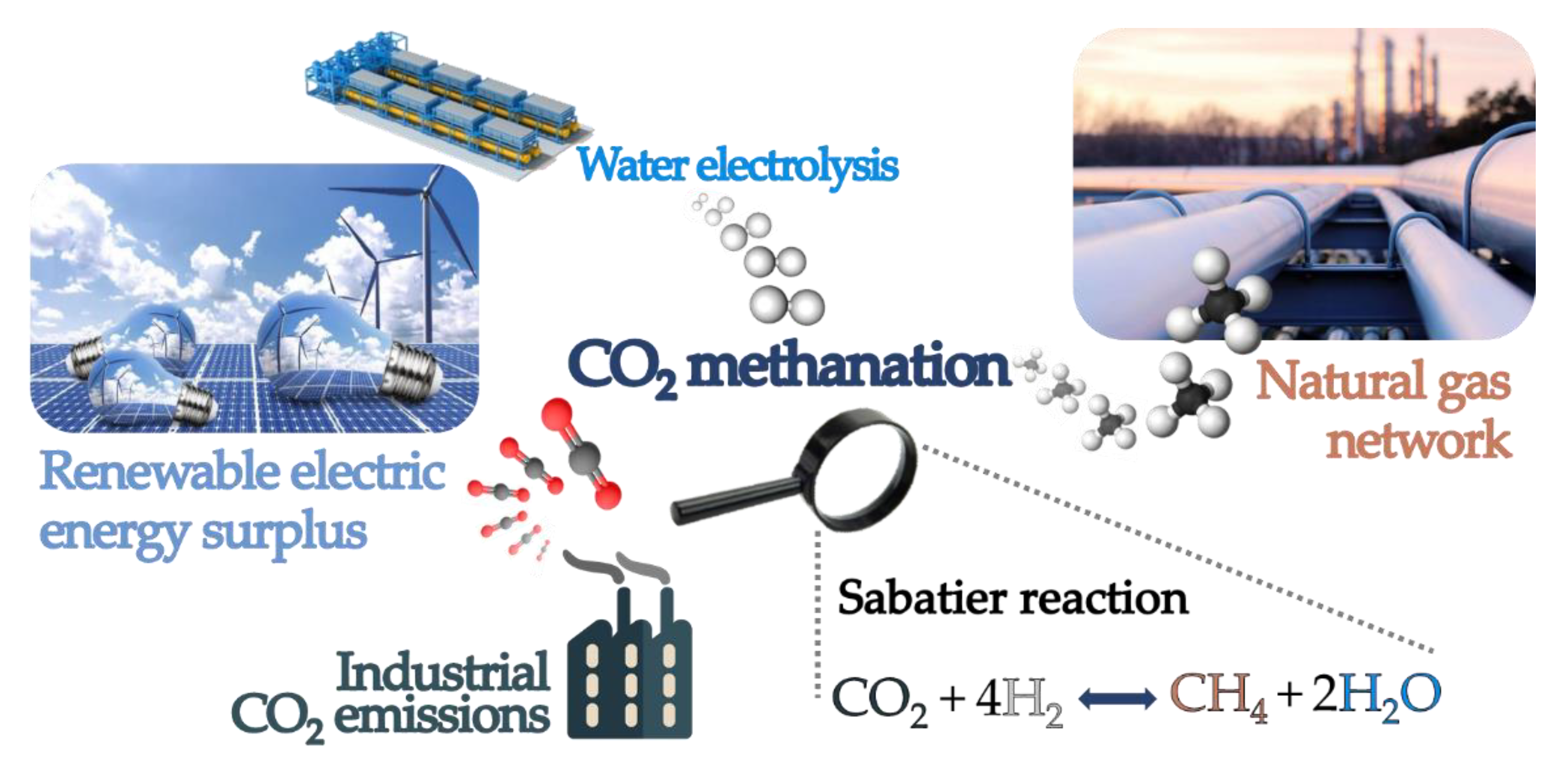
1. Introduction
2. SiO2-Based Catalysts
2.1. Monometallic SiO2-Supported Catalysts
2.2. Bi- and Trimetallic SiO2-Supported Catalysts
2.3. Mesoporous Silica-Based Catalysts
- Mesoporous silica nanoparticle (MSN)-based catalysts
- KIT–6-based catalysts
- SBA–15-based catalysts
- MCM–41-based catalysts
- KCC–1-based catalysts
- FDU–12-based catalysts
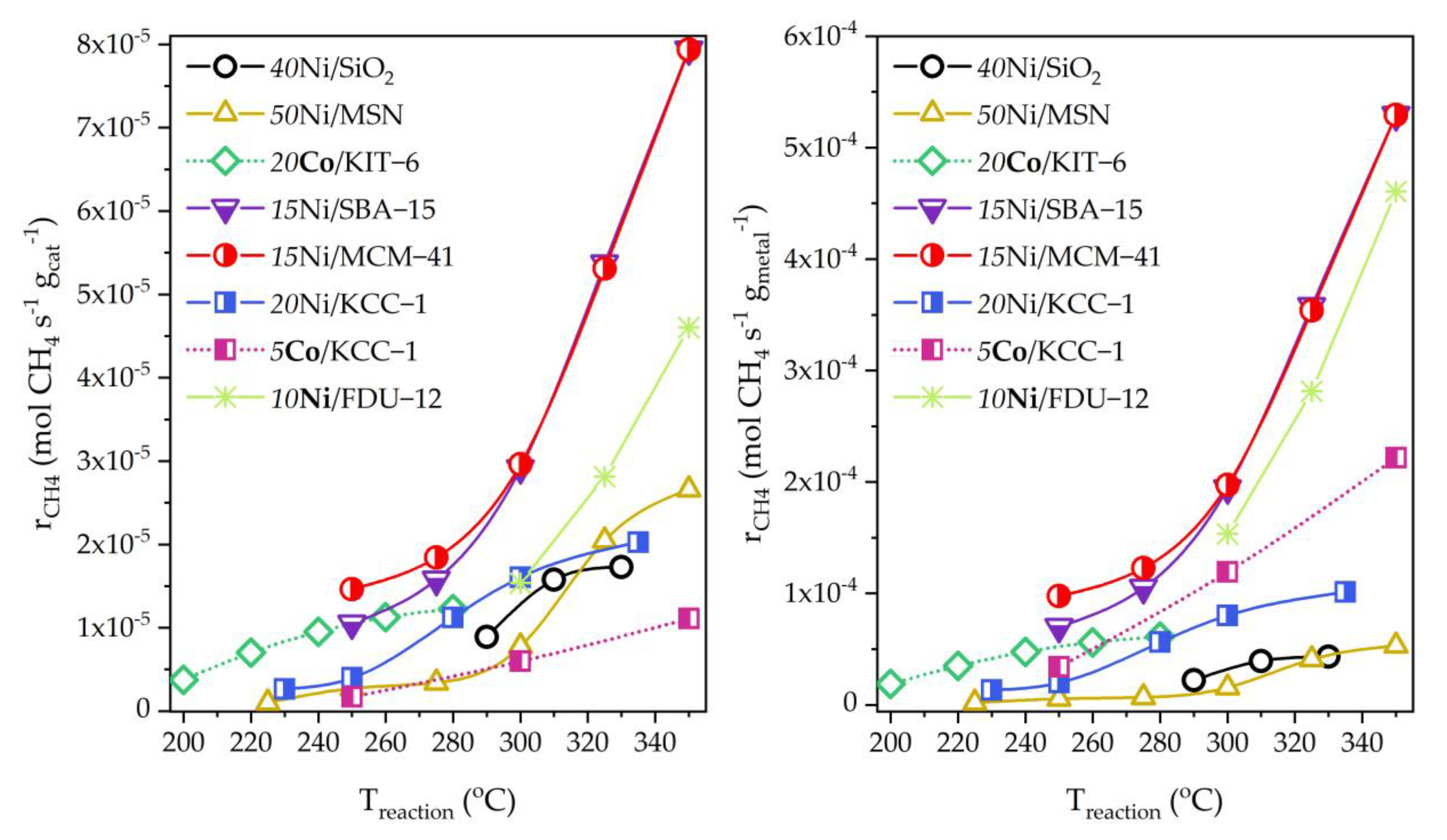
- Comparative analysis
2.4. Mechanistic Aspects
3. Al2O3-Based Catalysts
3.1. Monometallic Al2O3-Supported Catalysts
3.2. Bimetallic Al2O3-Supported Catalysts
3.3. Trimetallic Al2O3-Supported Catalysts and Composites
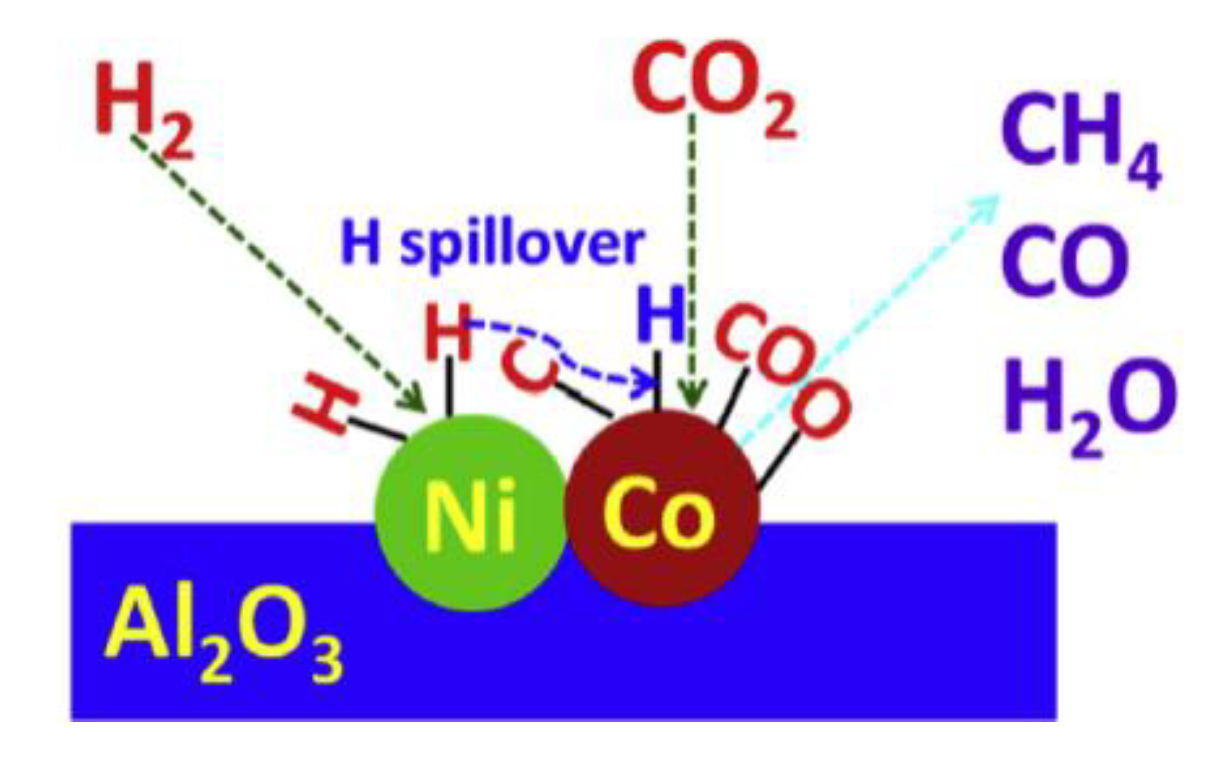
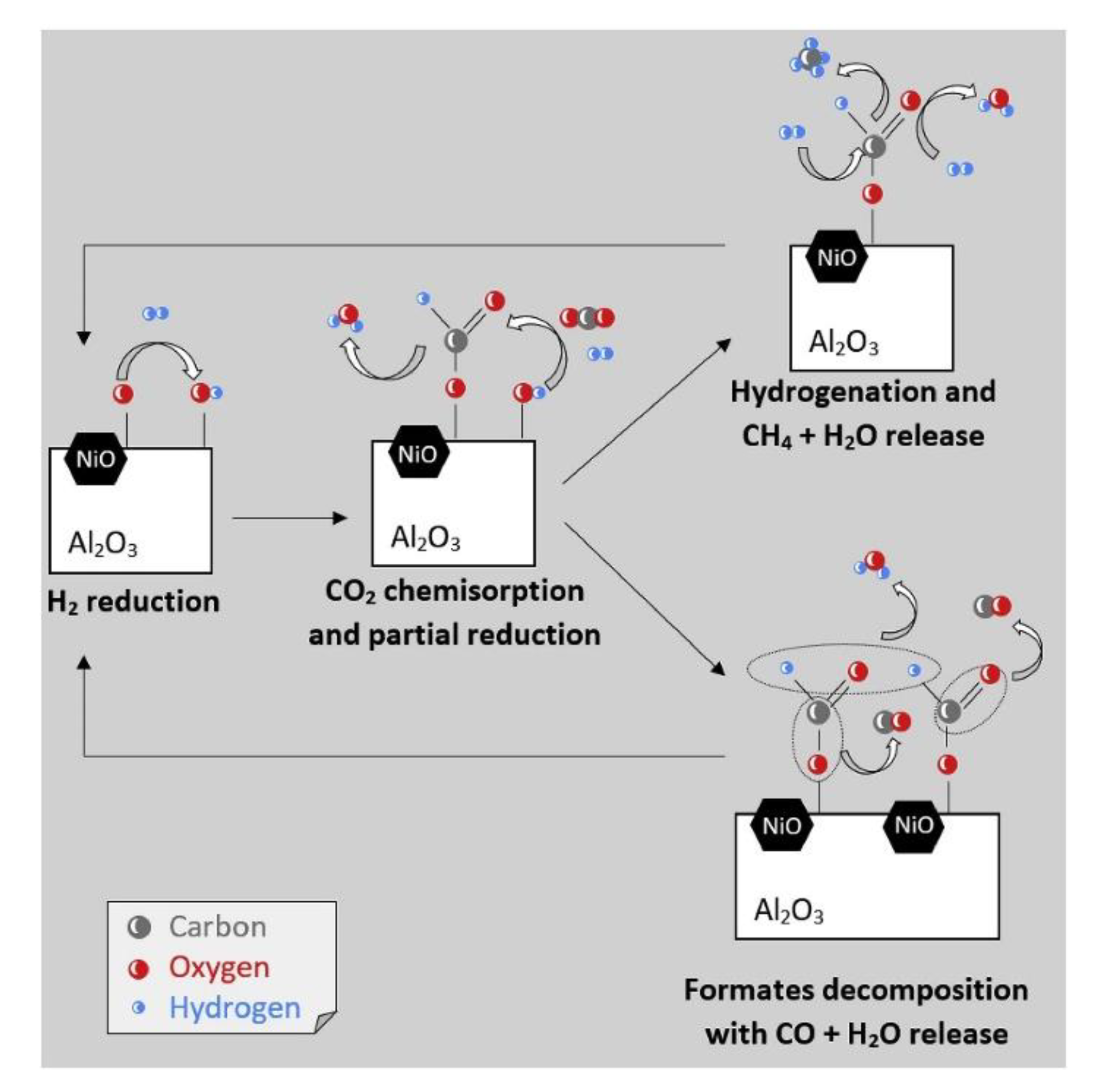
3.4. Mechanistic Aspects
4. CeO2-Based Catalysts
4.1. Monometallic CeO2-Supported Catalysts
4.2. Bi- and Trimetallic CeO2-Supported Catalysts
4.3. Mechanistic Aspects
5. ZrO2-Based Catalysts
5.1. Monometallic ZrO2-Supported Catalysts
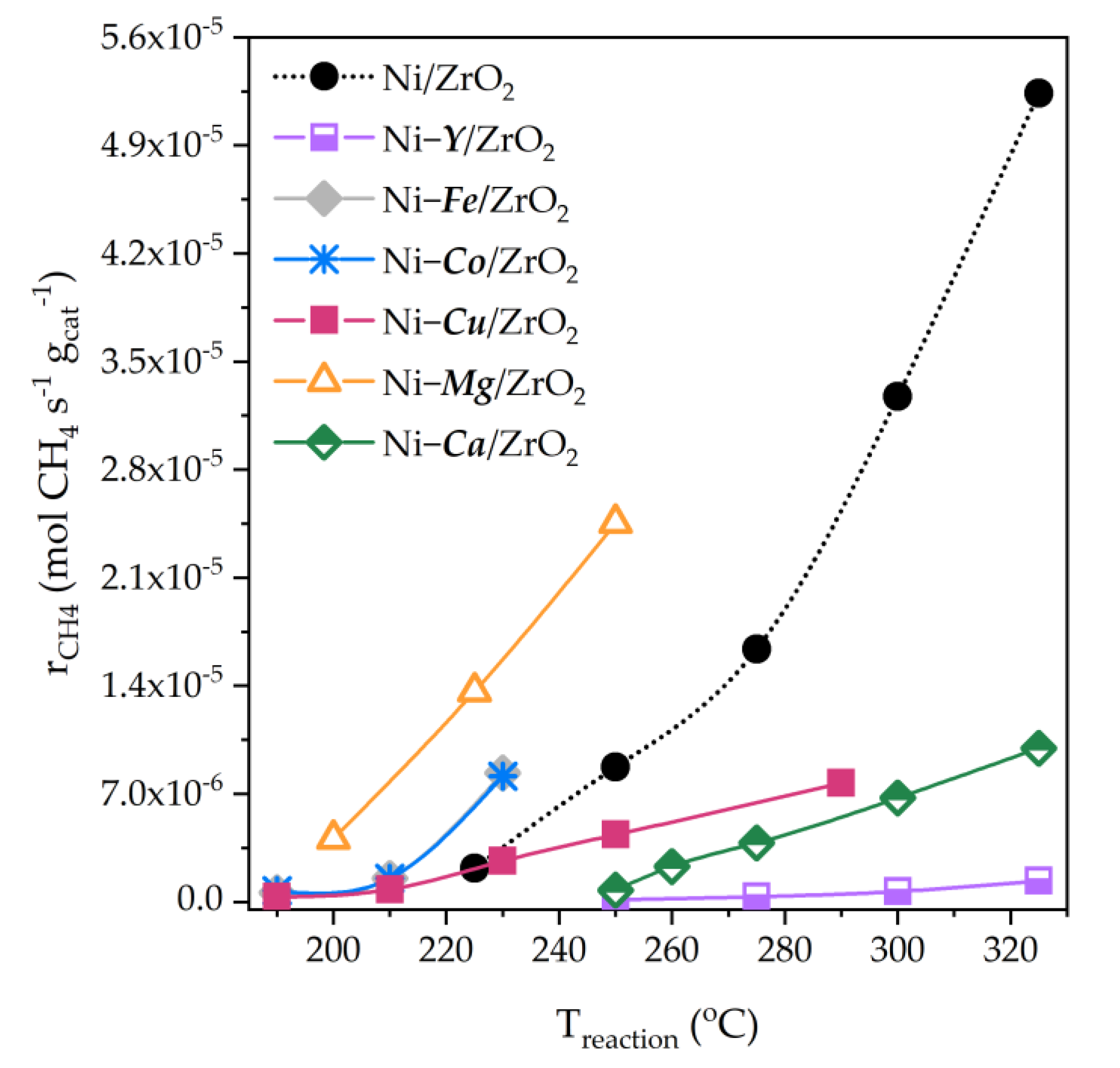
5.2. Bimetallic ZrO2-Supported Catalysts
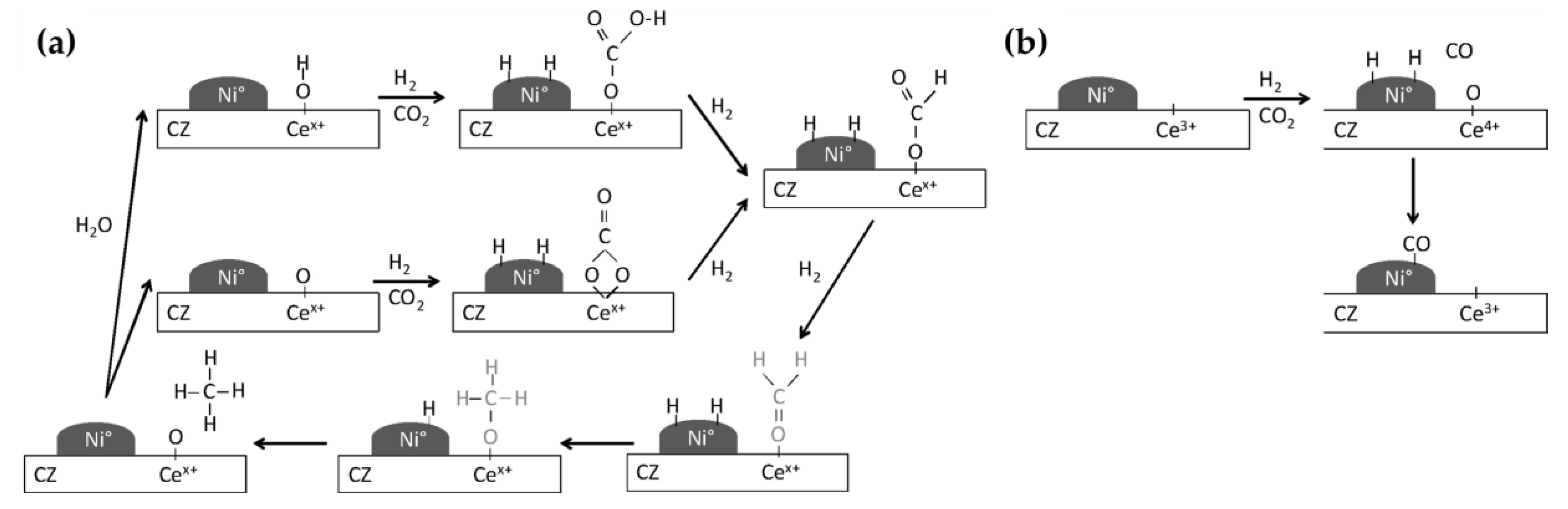
5.3. Mechanistic Aspects
6. MgO and Hydrotalcite-Based Catalysts
6.1. Monometallic MgO and Hydrotalcite-Derived Catalysts
6.2. Bi- and Trimetallic MgO and Hydrotalcite-Derived Catalysts
7. Carbon-Based Catalysts
7.1. Monometallic Carbon-Supported Catalysts
7.2. Bi- and Trimetallic Carbon-Supported Catalysts
7.3. Mechanistic Aspects
8. Zeolite-Based Catalysts
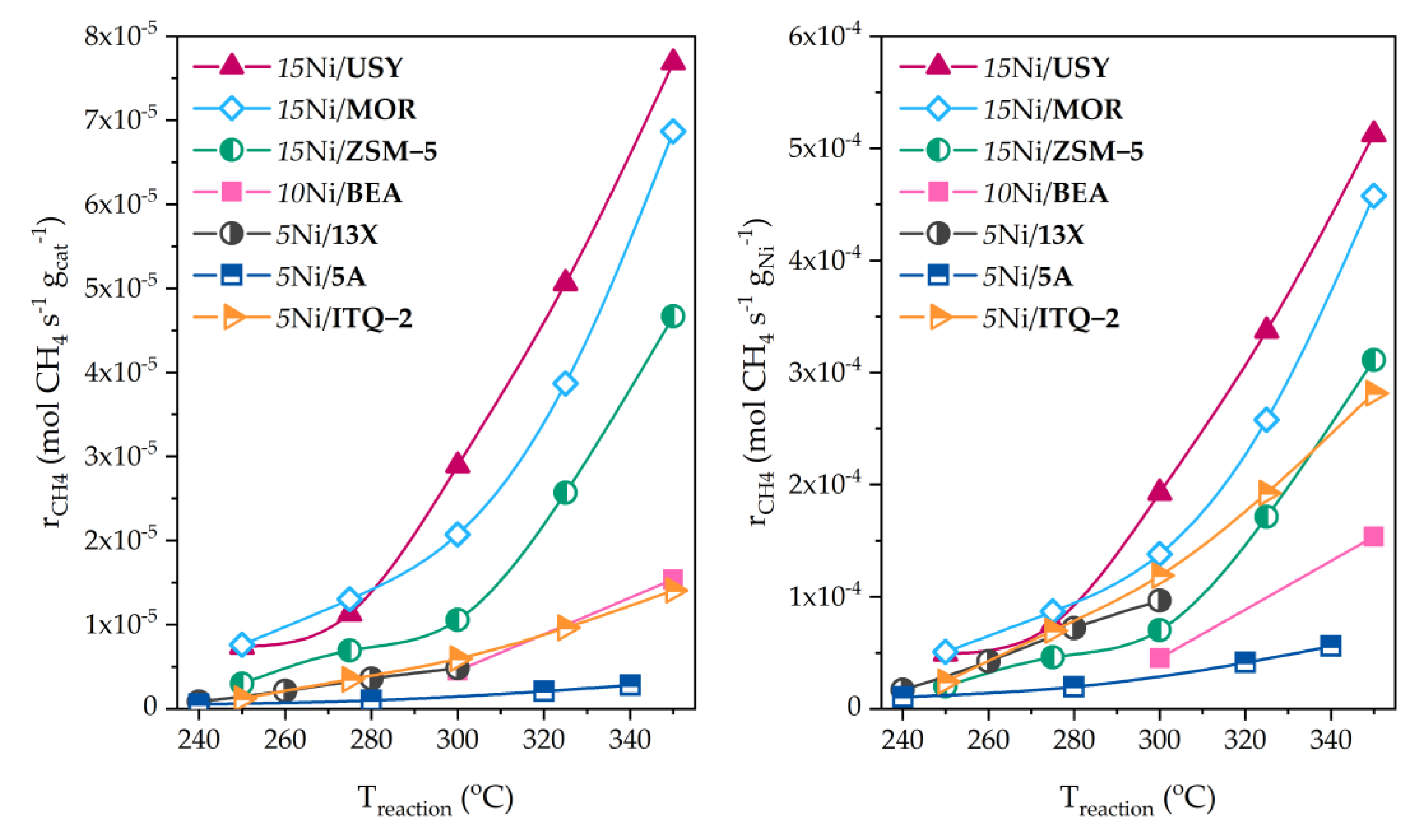
8.1. Monometallic Zeolite-Supported Catalysts
8.2. Bimetallic Zeolite-Supported Catalysts
9. Other Types of Supported Catalysts
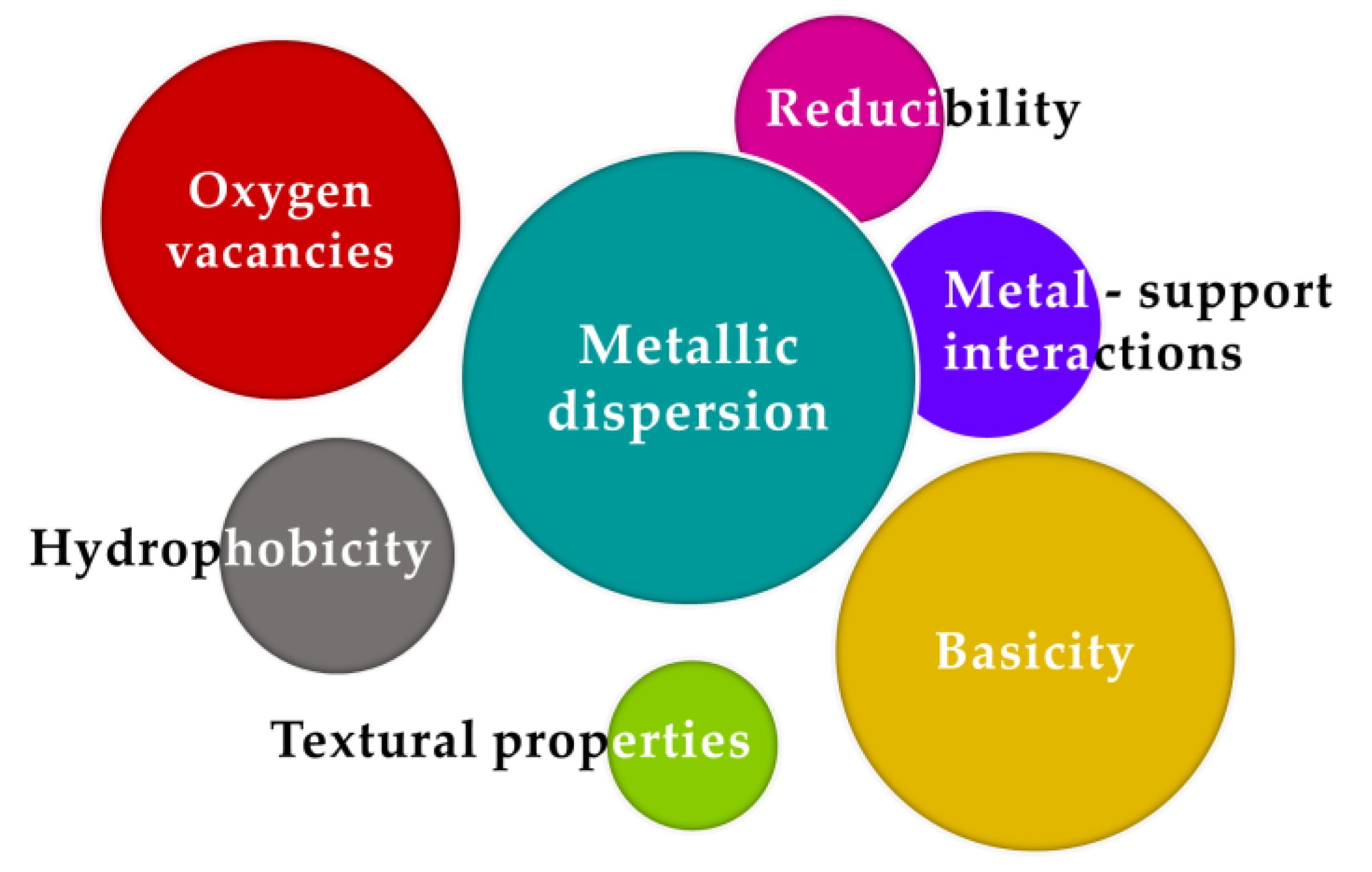
10. Final Remarks and Conclusions
- Metallic dispersion. Based on the analyzed studies, catalysts presenting higher metallic dispersion and smaller particles present typically better performances due to the favored H2 dissociation capacity. These properties can be enhanced by tuning the preparation conditions (e.g., method, solvent, calcination and reduction temperatures), adding promoters (e.g., CeO2, MgO, ZrO2, La2O3) or, among all, through encapsulation strategies;
- Basicity. CO2 can be, based on the previously summarized works, adsorbed and activated on basic sites. Generally, authors have identified medium strength basic sites as the most promising for CO2 methanation, since stronger sites lead to the formation of inactive carbonate species. These types of sites can be obtained by adding a basic promoter, such as MgO, or using a support able to interact with CO2 (e.g., CeO2, zeolites, Al2O3);
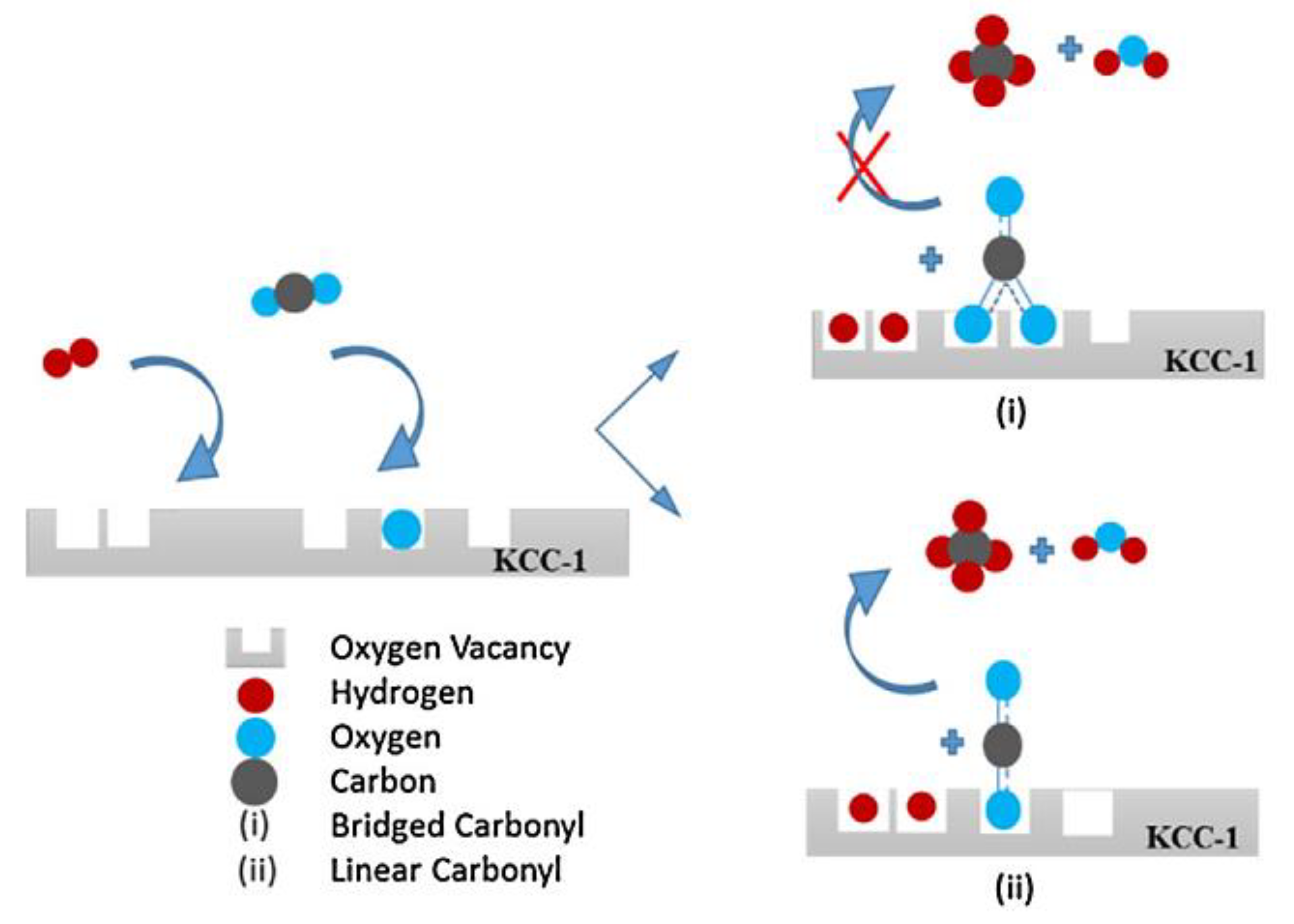
- Oxygen vacancies. Several authors referred to the presence of oxygen vacancies and its responsibility in the enhancement of CO2 activation. This can be achieved by using pure or modified CeO2, ZrO2 or TiO2 oxides or even KCC–1 mesoporous material;
- Metal-support interactions. Typically, this property is related to the metal dispersion and average particle sizes. Indeed, strong metal-support interactions (SMSI) can promote the formation of smaller and well-dispersed metallic particles, typically resisting sintering and carbon formation. However, the use of higher reduction temperatures could be required. SMSI can be obtained by tuning the preparation conditions. Indeed, while impregnation typically leads to weaker interactions, the insertion of the active metals into the support framework (e.g., ZrO2, CeO2, Al2O3, TiO2) represents a promising alternative;
- Reducibility. Even if few works suggested a possible role of metal oxides in the reaction (e.g., NiO), metallic states of transition or noble metals are typically considered as the active phases for the CO2 methanation reaction. This property, hardly dependent on the metal-support interactions, could be responsible for a reduction in the number of available active sites. It is important to deem in mind that increasing the reduction temperature to maximize the amount of reduced species can lead to severe sintering processes, which can negatively influence the observed performances. Authors found improvements in reducibility by adding promoters (e.g., CeO2, MgO, ZrO2, La2O3);
- Hydrophobicity. Water inhibitory effect in the CO2 methanation reaction was proved by several research studies. From the literature about zeolite supports, it was pointed out that the lower the affinity of the support for water adsorption (higher hydrophobicity), the higher the methane yields produced. The adsorption of water on the same sites for CO2 adsorption may comprehensively induce a negative kinetic effect on the methanation process. Consequently, it is important to use supports and/or promoters presenting low and weak affinity with water;
- Textural properties. Mesoporous materials have gained attention in the last years for this reaction as they can be responsible for encapsulation effects able to reduce metallic particle sizes and strengthen metal-support interactions, resulting in higher catalytic performances. Apart from the use of conventional mesoporous materials, carbons or zeolites as supports, strategies for obtaining ordered mesoporous structures such as Al2O3 represent promising strategies.
- Supports. The optimization and further development of new catalysts based on promising but not deeply explored materials such as MOFs or mesoporous silicas would be a promising pathway [217,218,219]. In addition, the utilization of waste materials (e.g., fly ash, rice husk) as support precursors constitutes an interesting route towards higher cost-efficient catalysts [220];
- Active metals. The systematic analysis of alternative transition (e.g., Co, Fe) or noble (e.g., Ru, Rh) active metals for CO2 methanation is a topic of high interest. Complementary, further efforts towards the identification and optimization of synergistic effects in bimetallic systems (e.g., Ni-Co, Ni–Fe) through advanced characterization techniques would be highly valuable;
- Mechanistic approaches. Modelling and in situ/Operando spectroscopy studies carried out using density-functional theory (DFT), DRIFTS or Operando FTIR under conventional and more realistic methanation conditions (e.g., incorporation of pollutants/minor compounds present in flue gases, biogas or hydrogen streams in the feed) will be advantageous. In addition, further studies dealing with the elucidation of the deactivation mechanisms over different types of catalysts, using as basis long-term and aging experiments, will be helpful;
- Catalytic testing. Although few works dealt with the effects of incorporating CO, CH4 or even steam in the reactor feed, evaluating CO2 methanation performances under realistic conditions will be key for identifying strategies to obtain catalysts with high resistance to, among all, oxygen, steam or H2S. In addition, further research on the preparation of scale-up catalysts (e.g., monoliths) will be important.
Author Contributions
Funding
Conflicts of Interest
References
- Denholm, P.; Ela, E.; Kirby, B.; Milligan, M. The Role of Energy Storage with Renewable Electricity Generation; Technical Report; National Renewable Energy Laboratory: Golden, CO, USA, 2010; pp. 1–61. [Google Scholar]
- Weitzel, T.; Glock, C.H. Energy management for stationary electric energy storage systems: A systematic literature review. Eur. J. Oper. Res. 2018, 264, 582–606. [Google Scholar] [CrossRef]
- Chen, H.; Cong, T.N.; Yang, W.; Tan, C.; Li, Y.; Ding, Y. Progress in electrical energy storage system: A critical review. Prog. Nat. Sci. 2009, 19, 291–312. [Google Scholar] [CrossRef]
- Blanco, H.; Faaij, A. A review at the role of storage in energy systems with a focus on Power to Gas and long-term storage. Renew. Sustain. Energy Rev. 2018, 81, 1049–1086. [Google Scholar] [CrossRef]
- Schaaf, T.; Grünig, J.; Schuster, M.R.; Rothenfluh, T.; Orth, A. Methanation of CO2 - storage of renewable energy in a gas distribution system. Energy Sustain. Soc. 2014, 4, 2. [Google Scholar] [CrossRef]
- Ghaib, K.; Ben-Fares, F.-Z. Power-to-Methane: A state-of-the-art review. Renew. Sustain. Energy Rev. 2018, 81, 433–446. [Google Scholar] [CrossRef]
- Sterner, M. Bioenergy and Renewable Power Methane in Integrated 100% Renewable Energy Systems: Limiting Global Warming By Transforming Energy Systems; Kassel University Press GmbH: Kassel, Germany, 2009; ISBN 978-3-89958-799-9. [Google Scholar]
- International Renewable Energy Agency (IRENA). Global Energy Transformation: A Roadmap to 2050 (2019 edition); International Renewable Energy Agency: Abu Dhabi, UAE, 2019. [Google Scholar]
- U.S. Energy Information Administration (EIA). International Energy Outlook 2019; U.S. Energy Information Administration (EIA): Washington, DC, USA, 2019.
- Pachauri, R.K.; Meyer, L.A. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Intergovernmental Panel on Climate Change (IPCC): Geneva, Switzerland, 2014. [Google Scholar]
- Andrew, R.M. Global CO2 emissions from cement production, 1928–2018. Earth Syst. Sci. Data 2019, 11, 1675–1710. [Google Scholar] [CrossRef]
- IEA. Technology Roadmap-Low-Carbon Transition in the Cement Industry–Analysis, IEA, Paris. 2018. Available online: https://www.iea.org/reports/technology-roadmap-low-carbon-transition-in-the-cement-industry (accessed on 30 June 2020).
- Aresta, M.; Dibenedetto, A.; Quaranta, E. State of the art and perspectives in catalytic processes for CO2 conversion into chemicals and fuels: The distinctive contribution of chemical catalysis and biotechnology. J. Catal. 2016, 343, 2–45. [Google Scholar] [CrossRef]
- Aresta, M.; Dibenedetto, A. Utilisation of CO2 as a chemical feedstock: opportunities and challenges. Dalton Trans. 2007, 2975. [Google Scholar] [CrossRef]
- Aresta, M.; Dibenedetto, A.; Angelini, A. The changing paradigm in CO2 utilization. J. CO2 Util. 2013, 3–4, 65–73. [Google Scholar] [CrossRef]
- Wang, W.; Wang, S.; Ma, X.; Gong, J. Recent advances in catalytic hydrogenation of carbon dioxide. Chem. Soc. Rev. 2011, 40, 3703–3727. [Google Scholar] [CrossRef]
- Frontera, P.; Macario, A.; Ferraro, M.; Antonucci, P. Supported Catalysts for CO2 Methanation: A Review. Catalysts 2017, 7, 59. [Google Scholar] [CrossRef]
- Ghaib, K.; Nitz, K.; Ben-Fares, F.-Z. Chemical Methanation of CO2: A Review. ChemBioEng Rev. 2016, 3, 266–275. [Google Scholar] [CrossRef]
- Jin Lee, W.; Li, C.; Prajitno, H.; Yoo, J.; Patel, J.; Yang, Y.; Lim, S. Recent trend in thermal catalytic low temperature CO2 methanation: A Critical Review. Catal. Today 2020. [Google Scholar] [CrossRef]
- Rönsch, S.; Schneider, J.; Matthischke, S.; Schlüter, M.; Götz, M.; Lefebvre, J.; Prabhakaran, P.; Bajohr, S. Review on methanation – From fundamentals to current projects. Fuel 2016, 166, 276–296. [Google Scholar] [CrossRef]
- Malara, A.; Frontera, P.; Antonucci, P.; Macario, A. Smart recycling of carbon oxides: Current status of methanation reaction. Curr. Opin. Green Sustain. Chem. 2020, 26, 100376. [Google Scholar] [CrossRef]
- Ashok, J.; Pati, S.; Hongmanorom, P.; Tianxi, Z.; Junmei, C.; Kawi, S. A review of recent catalyst advances in CO2 methanation processes. Catal. Today 2020. [Google Scholar] [CrossRef]
- Huynh, H.L.; Yu, Z. CO2 Methanation on Hydrotalcite-Derived Catalysts and Structured Reactors: A Review. Energy Technol. 2020, 8, 1901475. [Google Scholar] [CrossRef]
- Bacariza, M.C.; Graça, I.; Lopes, J.M.; Henriques, C. Tuning Zeolite Properties towards CO2 Methanation: An Overview. Chem. Cat. Chem. 2019, 11, 2388–2400. [Google Scholar] [CrossRef]
- Tsiotsias, A.I.; Charisiou, N.D.; Yentekakis, I.V.; Goula, M.A. The Role of Alkali and Alkaline Earth Metals in the CO2 Methanation Reaction and the Combined Capture and Methanation of CO2. Catalysts 2020, 10, 812. [Google Scholar] [CrossRef]
- Lv, C.; Xu, L.; Chen, M.; Cui, Y.; Wen, X.; Li, Y.; Wu, C.; Yang, B.; Miao, Z.; Hu, X.; et al. Recent Progresses in Constructing the Highly Efficient Ni Based Catalysts With Advanced Low-Temperature Activity Toward CO2 Methanation. Front. Chem. 2020, 8. [Google Scholar] [CrossRef]
- Solis-Garcia, A.; Fierro-Gonzalez, J.C. Mechanistic Insights into the CO2 Methanation Catalyzed by Supported Metals: A Review. Available online: https://www.ingentaconnect.com/content/asp/jnn/2019/00000019/00000006/art00005 (accessed on 28 April 2020).
- Kuznecova, I.; Gusca, J. Property based ranking of CO and CO2 methanation catalysts. Energy Procedia 2017, 128, 255–260. [Google Scholar] [CrossRef]
- Erdőhelyi, A. Hydrogenation of Carbon Dioxide on Supported Rh Catalysts. Catalysts 2020, 10, 155. [Google Scholar] [CrossRef]
- Choe, S.-J.; Kang, H.-J.; Kim, S.-J.; Park, S.-B.; Park, D.-H.; Huh, D.-S. Adsorbed Carbon Formation and Carbon Hydrogenation for CO2 Methanation on the Ni(111) Surface: ASED-MO Study. Bull. Korean Chem. Soc. 2005, 26, 1682–1688. [Google Scholar] [CrossRef]
- Ren, J.; Guo, H.; Yang, J.; Qin, Z.; Lin, J.; Li, Z. Insights into the mechanisms of CO2 methanation on Ni(111) surfaces by density functional theory. Appl. Surf. Sci. 2015, 351, 504–516. [Google Scholar] [CrossRef]
- Zhao, K.; Calizzi, M.; Moioli, E.; Li, M.; Borsay, A.; Lombardo, L.; Mutschler, R.; Luo, W.; Züttel, A. Unraveling and optimizing the metal-metal oxide synergistic effect in a highly active Cox(CoO)1–x catalyst for CO2 hydrogenation. J. Energy Chem. 2021, 53, 241–250. [Google Scholar] [CrossRef]
- Wu, H.C.; Chang, Y.C.; Wu, J.H.; Lin, J.H.; Lin, I.K.; Chen, C.S. Methanation of CO2 and reverse water gas shift reactions on Ni/SiO2 catalysts: The influence of particle size on selectivity and reaction pathway. Catal. Sci. Technol. 2015, 5, 4154–4163. [Google Scholar] [CrossRef]
- Ye, R.-P.; Gong, W.; Sun, Z.; Sheng, Q.; Shi, X.; Wang, T.; Yao, Y.; Razink, J.J.; Lin, L.; Zhou, Z.; et al. Enhanced stability of Ni/SiO2 catalyst for CO2 methanation: Derived from nickel phyllosilicate with strong metal-support interactions. Energy 2019, 188, 116059. [Google Scholar] [CrossRef]
- Guo, M.; Lu, G. The effect of impregnation strategy on structural characters and CO2 methanation properties over MgO modified Ni/SiO2 catalysts. Catal. Commun. 2014, 54, 55–60. [Google Scholar] [CrossRef]
- Kim, H.Y.; Lee, H.M.; Park, J.-N. Bifunctional Mechanism of CO2 Methanation on Pd-MgO/SiO2 Catalyst: Independent Roles of MgO and Pd on CO2 Methanation. J. Phys. Chem. C 2010, 114, 7128–7131. [Google Scholar] [CrossRef]
- Park, J.-N.; McFarland, E.W. A highly dispersed Pd–Mg/SiO2 catalyst active for methanation of CO2. J. Catal. 2009, 266, 92–97. [Google Scholar] [CrossRef]
- Ali, S.A.M.; Hamid, K.H.K.; Ismail, K.N. Effect of Calcination Temperature on The Structure and Catalytic Performance Of 80Ni20CO/SiO2 Catalyst for CO2 Methanation. In 3rd Electronic and Green Materials International Conference 2017 (egm 2017); Abdullah, M.M.A., Ramli, M.M., AbdRahim, S.Z., Isa, S.S.M., Saad, M.N.M., Ismail, R.C., Ghazli, M.F., Eds.; Amer Inst Physics: Melville, NY, USA, 2017; Volume 1885, p. UNSP 020272-1. ISBN 978-0-7354-1565-2. [Google Scholar]
- Trovarelli, A.; Deleitenburg, C.; Dolcetti, G.; Lorca, J.L. CO2 Methanation Under Transient and Steady-State Conditions over Rh/CeO2 and CeO2-Promoted Rh/SiO2: The Role of Surface and Bulk Ceria. J. Catal. 1995, 151, 111–124. [Google Scholar] [CrossRef]
- Li, S.; Guo, S.; Gong, D.; Kang, N.; Fang, K.-G.; Liu, Y. Nano composite composed of MoOx-La2O3Ni on SiO2 for storing hydrogen into CH4 via CO2 methanation. Int. J. Hydrogen Energy 2019, 44, 1597–1609. [Google Scholar] [CrossRef]
- Branco, J.B.; Brito, P.E.; Ferreira, A.C. Methanation of CO2 over nickel-lanthanide bimetallic oxides supported on silica. Chem. Eng. J. 2020, 380, 122465. [Google Scholar] [CrossRef]
- Vogt, C.; Wijten, J.; Madeira, C.L.; Kerkenaar, O.; Xu, K.; Holzinger, R.; Monai, M.; Weckhuysen, B.M. Alkali Promotion in the Formation of CH4 from CO2 and Renewably Produced H2 over Supported Ni Catalysts. Chem. Cat. Chem. 2020, 12, 2792–2800. [Google Scholar] [CrossRef]
- Aziz, M.A.A.; Jalil, A.A.; Triwahyono, S.; Sidik, S.M. Methanation of carbon dioxide on metal-promoted mesostructured silica nanoparticles. Appl. Catal. A 2014, 486, 115–122. [Google Scholar] [CrossRef]
- Aziz, M.A.A.; Jalil, A.A.; Triwahyono, S.; Saad, M.W.A. CO2 methanation over Ni-promoted mesostructured silica nanoparticles: Influence of Ni loading and water vapor on activity and response surface methodology studies. Chem. Eng. J. 2015, 260, 757–764. [Google Scholar] [CrossRef]
- Nguyen, P.-A.; Luu, C.-L.; Nguyen, T.-T.-V.; Nguyen, T.; Hoang, T.-C. Improving the performance of nickel catalyst supported on mesostructured silica nanoparticles in methanation of CO2-rich gas by urea–nitrate combustion. Chem. Pap. 2020. [Google Scholar] [CrossRef]
- Zhou, G.; Wu, T.; Xie, H.; Zheng, X. Effects of structure on the carbon dioxide methanation performance of Co-based catalysts. Int. J. Hydrogen Energy 2013, 38, 10012–10018. [Google Scholar] [CrossRef]
- Zhou, G.; Wu, T.; Zhang, H.; Xie, H.; Feng, Y. Carbon Dioxide Methanation on Ordered Mesoporous Co/KIT-6 Catalyst. Chem. Eng. Commun. 2014, 201, 233–240. [Google Scholar] [CrossRef]
- Liu, H.; Xu, S.; Zhou, G.; Xiong, K.; Jiao, Z.; Wang, S. CO2 hydrogenation to methane over Co/KIT-6 catalysts: Effect of Co content. Fuel 2018, 217, 570–576. [Google Scholar] [CrossRef]
- Liu, H.; Xu, S.; Zhou, G.; Huang, G.; Huang, S.; Xiong, K. CO2 hydrogenation to methane over Co/KIT-6 catalyst: Effect of reduction temperature. Chem. Eng. J. 2018, 351, 65–73. [Google Scholar] [CrossRef]
- Merkache, R.; Fechete, I.; Maamache, M.; Bernard, M.; Turek, P.; Al-Dalama, K.; Garin, F. 3D ordered mesoporous Fe-KIT-6 catalysts for methylcyclopentane (MCP) conversion and carbon dioxide (CO2) hydrogenation for energy and environmental applications. Appl. Catal. A-Gen. 2015, 504, 672–681. [Google Scholar] [CrossRef]
- Cao, H.; Wang, W.; Cui, T.; Wang, H.; Zhu, G.; Ren, X. Enhancing CO2 Hydrogenation to Methane by Ni-Based Catalyst with V Species Using 3D-mesoporous KIT-6 as Support. Energies 2020, 13, 2235. [Google Scholar] [CrossRef]
- Liu, Q.; Tian, Y. One-pot synthesis of NiO/SBA-15 monolith catalyst with a three-dimensional framework for CO2 methanation. Int. J. Hydrogen Energy 2017, 42, 12295–12300. [Google Scholar] [CrossRef]
- Lu, B.; Ju, Y.; Abe, T.; Kawamoto, K. Grafting Ni particles onto SBA-15, and their enhanced performance for CO2 methanation. RSC Adv. 2015, 5, 56444–56454. [Google Scholar] [CrossRef]
- Bacariza, M.C.; Graça, I.; Bebiano, S.S.; Lopes, J.M.; Henriques, C. Micro- and mesoporous supports for CO2 methanation catalysts: A comparison between SBA-15, MCM-41 and USY zeolite. Chem. Eng. Sci. 2018, 175, 72–83. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, H.; Zhang, L.; Zhang, H. Bimetallic NiPd/SBA-15 alloy as an effective catalyst for selective hydrogenation of CO2 to methane. Int. J. Hydrogen Energy 2019, 44, 13354–13363. [Google Scholar] [CrossRef]
- Du, G.; Lim, S.; Yang, Y.; Wang, C.; Pfefferle, L.; Haller, G.L. Methanation of carbon dioxide on Ni-incorporated MCM-41 catalysts: The influence of catalyst pretreatment and study of steady-state reaction. J. Catal. 2007, 249, 370–379. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, L.; Liu, Y.; Wang, S. CO2 methanation on the catalyst of Ni/MCM-41 promoted with CeO2. Sci. Total Environ. 2018, 625, 686–695. [Google Scholar] [CrossRef]
- Taherian, Z.; Khataee, A.; Orooji, Y. Promoted nickel-based catalysts on modified mesoporous silica support: The role of yttria and magnesia on CO2 methanation. Microporous Mesoporous Mater. 2020, 306, 110455. [Google Scholar] [CrossRef]
- Hamid, M.Y.S.; Firmansyah, M.L.; Triwahyono, S.; Jalil, A.A.; Mukti, R.R.; Febriyanti, E.; Suendo, V.; Setiabudi, H.D.; Mohamed, M.; Nabgan, W. Oxygen vacancy-rich mesoporous silica KCC-1 for CO2 methanation. Appl. Catal. A 2017, 532, 86–94. [Google Scholar] [CrossRef]
- Shahul Hamid, M.Y.; Triwahyono, S.; Jalil, A.A.; Che Jusoh, N.W.; Izan, S.M.; Tuan Abdullah, T.A. Tailoring the Properties of Metal Oxide Loaded/KCC-1 toward a Different Mechanism of CO2 Methanation by in Situ IR and ESR. Inorg. Chem. 2018, 57, 5859–5869. [Google Scholar] [CrossRef] [PubMed]
- Lv, C.; Xu, L.; Chen, M.; Cui, Y.; Wen, X.; Wu, C.; Yang, B.; Wang, F.; Miao, Z.; Hu, X.; et al. Constructing highly dispersed Ni based catalysts supported on fibrous silica nanosphere for low-temperature CO2 methanation. Fuel 2020, 278, 118333. [Google Scholar] [CrossRef]
- Liu, Q.; Dong, H. In Situ Immobilizing Ni Nanoparticles to FDU-12 via Trehalose with Fine Size and Location Control for CO2 Methanation. ACS Sustainable Chem. Eng. 2020, 8, 2093–2105. [Google Scholar] [CrossRef]
- Aziz, M.A.A.; Jalil, A.A.; Triwahyono, S.; Mukti, R.R.; Taufiq-Yap, Y.H.; Sazegar, M.R. Highly active Ni-promoted mesostructured silica nanoparticles for CO2 methanation. Appl. Catal. B 2014, 147, 359–368. [Google Scholar] [CrossRef]
- Wang, W.; Qi, R.; Shan, W.; Wang, X.; Jia, Q.; Zhao, J.; Zhang, C.; Ru, H. Synthesis of KIT-6 type mesoporous silicas with tunable pore sizes, wall thickness and particle sizes via the partitioned cooperative self-assembly process. Microporous Mesoporous Mater. 2014, 194, 167–173. [Google Scholar] [CrossRef]
- Soni, K.; Rana, B.S.; Sinha, A.K.; Bhaumik, A.; Nandi, M.; Kumar, M.; Dhar, G.M. 3-D ordered mesoporous KIT-6 support for effective hydrodesulfurization catalysts. Appl. Catal. B 2009, 90, 55–63. [Google Scholar] [CrossRef]
- Singh, S.; Kumar, R.; Setiabudi, H.D.; Nanda, S.; Vo, D.-V.N. Advanced synthesis strategies of mesoporous SBA-15 supported catalysts for catalytic reforming applications: A state-of-the-art review. Appl. Catal. A 2018, 559, 57–74. [Google Scholar] [CrossRef]
- Vallet-Regi, M.; Rámila, A.; Del Real, R.P.; Pérez-Pariente, J. A New Property of MCM-41: Drug Delivery System. Chem. Mater. 2001, 13, 308–311. [Google Scholar] [CrossRef]
- Corma, A.; Fornes, V.; Navarro, M.T.; Perezpariente, J. Acidity and Stability of MCM-41 Crystalline Aluminosilicates. J. Catal. 1994, 148, 569–574. [Google Scholar] [CrossRef]
- Gao, J.; Wang, Y.; Ping, Y.; Hu, D.; Xu, G.; Gu, F.; Su, F. A thermodynamic analysis of methanation reactions of carbon oxides for the production of synthetic natural gas. RSC Adv. 2012, 2, 2358. [Google Scholar] [CrossRef]
- Ocampo, F. Développement de catalyseurs pour la réaction de méthanation du dioxyde de carbone; University of Strasbourg: Strasbourg, France, 2011. [Google Scholar]
- Polshettiwar, V.; Cha, D.; Zhang, X.; Basset, J.M. High-Surface-Area Silica Nanospheres (KCC-1) with a Fibrous Morphology. Angew. Chem. Int. Ed. 2010, 49, 9652–9656. [Google Scholar] [CrossRef] [PubMed]
- Riani, P.; Garbarino, G.; Lucchini, M.A.; Canepa, F.; Busca, G. Unsupported versus alumina-supported Ni nanoparticles as catalysts for steam/ethanol conversion and CO2 methanation. J. Mol. Catal. A 2014, 383–384, 10–16. [Google Scholar] [CrossRef]
- Garbarino, G.; Riani, P.; Magistri, L.; Busca, G. A study of the methanation of carbon dioxide on Ni/Al2O3 catalysts at atmospheric pressure. Int. J. Hydrogen Energy 2014, 39, 11557–11565. [Google Scholar] [CrossRef]
- Quindimil, A.; De-La-Torre, U.; Pereda-Ayo, B.; Davó-Quiñonero, A.; Bailón-García, E.; Lozano-Castelló, D.; González-Marcos, J.A.; Bueno-López, A.; González-Velasco, J.R. Effect of metal loading on the CO2 methanation: A comparison between alumina supported Ni and Ru catalysts. Catal. Today 2019. [Google Scholar] [CrossRef]
- Garbarino, G.; Bellotti, D.; Riani, P.; Magistri, L.; Busca, G. Methanation of carbon dioxide on Ru/Al2O3 and Ni/Al2O3 catalysts at atmospheric pressure: Catalysts activation, behaviour and stability. Int. J. Hydrogen Energy 2015, 40, 9171–9182. [Google Scholar] [CrossRef]
- Hwang, S.; Hong, U.G.; Lee, J.; Seo, J.G.; Baik, J.H.; Koh, D.J.; Lim, H.; Song, I.K. Methanation of carbon dioxide over mesoporous Ni–Fe–Al2O3 catalysts prepared by a coprecipitation method: Effect of precipitation agent. J. Ind. Eng. Chem. 2013, 19, 2016–2021. [Google Scholar] [CrossRef]
- Serrer, M.-A.; Gaur, A.; Jelic, J.; Weber, S.; Fritsch, C.; Clark, A.H.; Saraçi, E.; Studt, F.; Grunwaldt, J.-D. Structural dynamics in Ni–Fe catalysts during CO2 methanation – role of iron oxide clusters. Catal. Sci. Technol. 2020, 10, 7542–7554. [Google Scholar] [CrossRef]
- Burger, T.; Augenstein, H.M.S.; Hnyk, F.; Döblinger, M.; Köhler, K.; Hinrichsen, O. Targeted Fe-Doping of Ni−Al Catalysts via the Surface Redox Reaction Technique for Unravelling its Promoter Effect in the CO2 Methanation Reaction. ChemCatChem 2020, 12, 649–662. [Google Scholar] [CrossRef]
- Burger, T.; Koschany, F.; Thomys, O.; Köhler, K.; Hinrichsen, O. CO2 methanation over Fe- and Mn-promoted co-precipitated Ni-Al catalysts: Synthesis, characterization and catalysis study. Appl. Catal. A: General 2018, 558, 44–54. [Google Scholar] [CrossRef]
- Xu, L.; Lian, X.; Chen, M.; Cui, Y.; Wang, F.; Li, W.; Huang, B. CO2 methanation over CoNi bimetal-doped ordered mesoporous Al2O3 catalysts with enhanced low-temperature activities. Int. J. Hydrogen Energy 2018, 43, 17172–17184. [Google Scholar] [CrossRef]
- Alrafei, B.; Polaert, I.; Ledoux, A.; Azzolina-Jury, F. Remarkably stable and efficient Ni and Ni-Co catalysts for CO2 methanation. Catal. Today 2020, 346, 23–33. [Google Scholar] [CrossRef]
- Rahmani, S.; Rezaei, M.; Meshkani, F. Preparation of promoted nickel catalysts supported on mesoporous nanocrystalline gamma alumina for carbon dioxide methanation reaction. J. Ind. Eng. Chem. 2014, 20, 4176–4182. [Google Scholar] [CrossRef]
- Liu, H.; Zou, X.; Wang, X.; Lu, X.; Ding, W. Effect of CeO2 addition on Ni/Al2O3 catalysts for methanation of carbon dioxide with hydrogen. J. Nat. Gas Chem. 2012, 21, 703–707. [Google Scholar] [CrossRef]
- Guo, X.; He, H.; Traitangwong, A.; Gong, M.; Meeyoo, V.; Li, P.; Li, C.; Peng, Z.; Zhang, S. Ceria imparts superior low temperature activity to nickel catalysts for CO2 methanation. Catal. Sci. Technol. 2019, 9, 5636–5650. [Google Scholar] [CrossRef]
- Tada, S.; Ochieng, O.J.; Kikuchi, R.; Haneda, T.; Kameyama, H. Promotion of CO2 methanation activity and CH4 selectivity at low temperatures over Ru/CeO2/Al2O3 catalysts. Int. J. Hydrogen Energy 2014, 39, 10090–10100. [Google Scholar] [CrossRef]
- Ahmad, W.; Younis, M.N.; Shawabkeh, R.; Ahmed, S. Synthesis of lanthanide series (La, Ce, Pr, Eu & Gd) promoted Ni/gamma-Al2O3 catalysts for methanation of CO2 at low temperature under atmospheric pressure. Catal. Commun. 2017, 100, 121–126. [Google Scholar] [CrossRef]
- Karam, L.; Bacariza, M.C.; Lopes, J.M.; Henriques, C.; Massiani, P.; El Hassan, N. Assessing the potential of xNi-yMg-Al2O3 catalysts prepared by EISA-one-pot synthesis towards CO2 methanation: An overall study. Int. J. Hydrogen Energy 2020. [Google Scholar] [CrossRef]
- Nie, W.-X.; Zou, X.-J.; Wang, X.-G.; Ding, W.-Z.; Lu, X.-G. Preparation of Highly Dispersed Ni-Ce-Zr Oxides over Mesoporous Y-Alumina and Their Catalytic Properties for CO2 Methanation. Acta Phys.-Chim. Sin. 2016, 32, 2803–2810. [Google Scholar] [CrossRef]
- Toemen, S.; Bakar, W.A.W.A.; Ali, R. Investigation of Ru/Mn/Ce/Al2O3 catalyst for carbon dioxide methanation: Catalytic optimization, physicochemical studies and RSM. J. Taiwan Inst. Chem. Eng. 2014, 45, 2370–2378. [Google Scholar] [CrossRef]
- Wan Abu Bakar, W.A.; Ali, R.; Mohammad, N.S. The effect of noble metals on catalytic methanation reaction over supported Mn/Ni oxide based catalysts. Arab. J. Chem. 2015, 8, 632–643. [Google Scholar] [CrossRef]
- Do, J.Y.; Park, N.-K.; Seo, M.W.; Lee, D.; Ryu, H.-J.; Kang, M. Effective thermocatalytic carbon dioxide methanation on Ca-inserted NiTiO3 perovskite. Fuel 2020, 271, 117624. [Google Scholar] [CrossRef]
- Cai, M.; Wen, J.; Chu, W.; Cheng, X.; Li, Z. Methanation of carbon dioxide on Ni/ZrO2-Al2O3 catalysts: Effects of ZrO2 promoter and preparation method of novel ZrO2-Al2O3 carrier. J. Nat. Gas Chem. 2011, 20, 318–324. [Google Scholar] [CrossRef]
- Lin, J.; Ma, C.; Wang, Q.; Xu, Y.; Ma, G.; Wang, J.; Wang, H.; Dong, C.; Zhang, C.; Ding, M. Enhanced low-temperature performance of CO2 methanation over mesoporous Ni/Al2O3-ZrO2 catalysts. Appl. Catal. B 2019, 243, 262–272. [Google Scholar] [CrossRef]
- Xu, J.; Lin, Q.; Su, X.; Duan, H.; Geng, H.; Huang, Y. CO2 methanation over TiO2-Al2O3 binary oxides supported Ru catalysts. Chin. J. Chem. Eng. 2016, 24, 140–145. [Google Scholar] [CrossRef]
- Yang, W.; Feng, Y.; Chu, W. Promotion Effect of CaO Modification on Mesoporous Al2O3-Supported Ni Catalysts for CO2 Methanation. Int. J. Chem. Eng. 2016, 2041821. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, S.; Zhao, G.; Yang, H.; Yuan, M.; An, X.; Zhou, H.; Qiao, Y.; Tian, Y. CO2 methanation over ordered mesoporous NiRu-doped CaO-Al2O3 nanocomposites with enhanced catalytic performance. Int. J. Hydrogen Energy 2018, 43, 239–250. [Google Scholar] [CrossRef]
- Abate, S.; Mebrahtu, C.; Giglio, E.; Deorsola, F.; Bensaid, S.; Perathoner, S.; Pirone, R.; Centi, G. Catalytic Performance of gamma-Al2O3-ZrO2-TiO2-CeO2 Composite Oxide Supported Ni-Based Catalysts for CO2 Methanation. Ind. Eng. Chem. Res. 2016, 55, 4451–4460. [Google Scholar] [CrossRef]
- Mebrahtu, C.; Abate, S.; Perathoner, S.; Chen, S.; Centi, G. CO2 methanation over Ni catalysts based on ternary and quaternary mixed oxide: A comparison and analysis of the structure-activity relationships. Catal. Today 2018, 304, 181–189. [Google Scholar] [CrossRef]
- Djinović, P.; Galletti, C.; Specchia, S.; Specchia, V. CO Methanation Over Ru–Al2O3 Catalysts: Effects of Chloride Doping on Reaction Activity and Selectivity. Top Catal. 2011, 54, 1042. [Google Scholar] [CrossRef]
- Konishcheva, M.V.; Potemkin, D.I.; Snytnikov, P.V.; Stonkus, O.A.; Belyaev, V.D.; Sobyanin, V.A. The insights into chlorine doping effect on performance of ceria supported nickel catalysts for selective CO methanation. Appl. Catal. B 2018, 221, 413–421. [Google Scholar] [CrossRef]
- Gao, Z.; Wang, L.; Ma, H.; Li, Z. Durability of catalytic performance of the chlorine-doped catalyst Ni(Clx)/ZrO2 for selective methanation of CO in H2-rich gas. Appl. Catal. A 2017, 534, 78–84. [Google Scholar] [CrossRef]
- Serrer, M.-A.; Kalz, K.F.; Saraҫi, E.; Lichtenberg, H.; Grunwaldt, J.-D. Role of Iron on the Structure and Stability of Ni3.2Fe/Al2O3 during Dynamic CO2 Methanation for P2X Applications. ChemCatChem 2019, 11, 5018–5021. [Google Scholar] [CrossRef]
- Mutz, B.; Belimov, M.; Wang, W.; Sprenger, P.; Serrer, M.-A.; Wang, D.; Pfeifer, P.; Kleist, W.; Grunwaldt, J.-D. Potential of an Alumina-Supported Ni3Fe Catalyst in the Methanation of CO2: Impact of Alloy Formation on Activity and Stability. ACS Catal. 2017, 7, 6802–6814. [Google Scholar] [CrossRef]
- Burger, T.; Ewald, S.; Niederdränk, A.; Wagner, F.E.; Köhler, K.; Hinrichsen, O. Enhanced activity of co-precipitated NiFeAlOx in CO2 methanation by segregation and oxidation of Fe. Appl. Catal. A 2020, 604, 117778. [Google Scholar] [CrossRef]
- Rosid, S.J.M.; Toemen, S.; Iqbal, M.M.A.; Bakar, W.A.W.A.; Mokhtar, W.N.A.W.; Aziz, M.M.A. Overview performance of lanthanide oxide catalysts in methanation reaction for natural gas production. Environ. Sci. Pollut. Res. 2019, 26, 36124–36140. [Google Scholar] [CrossRef]
- Bernardon, C.; Ben Osman, M.; Laugel, G.; Louis, B.; Pale, P. Acidity versus metal-induced Lewis acidity in zeolites for Friedel–Crafts acylation. Comptes Rendus Chim. 2017, 20, 20–29. [Google Scholar] [CrossRef]
- Zamani, A.H.; Ali, R.; Abu Bakar, W.A.W. Optimization of CO2 methanation reaction over M*/Mn/Cu–Al2O3 (M*: Pd, Rh and Ru) catalysts. J. Ind. Eng. Chem. 2015, 29, 238–248. [Google Scholar] [CrossRef]
- Franken, T.; Terreni, J.; Borgschulte, A.; Heel, A. Solid solutions in reductive environment – A case study on improved CO2 hydrogenation to methane on cobalt based catalysts derived from ternary mixed metal oxides by modified reducibility. J. Catal. 2020, 382, 385–394. [Google Scholar] [CrossRef]
- Daroughegi, R.; Meshkani, F.; Rezaei, M. Enhanced activity of CO2 methanation over mesoporous nanocrystalline Ni-Al2O3 catalysts prepared by ultrasound-assisted co-precipitation method. Int. J. Hydrogen Energy 2017, 42, 15115–15125. [Google Scholar] [CrossRef]
- Xu, Y.; Chen, Y.; Li, J.; Zhou, J.; Song, M.; Zhang, X.; Yin, Y. Improved low-temperature activity of Ni-Ce/gamma-Al2O3 catalyst with layer structural precursor prepared by cold plasma for CO2 methanation. Int. J. Hydrogen Energy 2017, 42, 13085–13091. [Google Scholar] [CrossRef]
- Song, F.; Zhong, Q.; Yu, Y.; Shi, M.; Wu, Y.; Hu, J.; Song, Y. Obtaining well-dispersed Ni/Al2O3 catalyst for CO2 methanation with a microwave-assisted method. Int. J. Hydrogen Energy 2017, 42, 4174–4183. [Google Scholar] [CrossRef]
- Schubert, M.; Pokhrel, S.; Thome, A.; Zielasek, V.; Gesing, T.M.; Roessner, F.; Maedler, L.; Baeumer, M. Highly active Co-Al2O3-based catalysts for CO2 methanation with very low platinum promotion prepared by double flame spray pyrolysis. Catal. Sci. Technol. 2016, 6, 7449–7460. [Google Scholar] [CrossRef]
- Aljishi, A.; Veilleux, G.; Lalinde, J.A.H.; Kopyscinski, J. The effect of synthesis parameters on ordered mesoporous nickel alumina catalyst for CO2 methanation. Appl. Catal. A 2018, 549, 263–272. [Google Scholar] [CrossRef]
- Xu, L.; Wang, F.; Chen, M.; Zhang, J.; Yuan, K.; Wang, L.; Wu, K.; Xu, G.; Chen, W. CO2 methanation over a Ni based ordered mesoporous catalyst for the production of synthetic natural gas. RSC Adv. 2016, 6, 28489–28499. [Google Scholar] [CrossRef]
- Shang, X.; Deng, D.; Wang, X.; Zou, X.; Ding, W.; Lu, X. Low Temperature Synthesis of Mesoporous gamma-Alumina supported Nickel Oxides and their Catalytic Application for CO2 Methanation. In Proceedings of the 2015 International Symposium on Energy Science and Chemical Engineering (isesce 2015); He, Y., Ed.; Atlantis Press: Paris, France, 2016; Volume 45, pp. 117–123, ISBN 978-94-6252-140-7. [Google Scholar]
- Le, T.A.; Kim, T.W.; Lee, S.H.; Park, E.D. CO and CO2 methanation over Ni catalysts supported on alumina with different crystalline phases. Korean J. Chem. Eng. 2017, 34, 3085–3091. [Google Scholar] [CrossRef]
- Beuls, A.; Swalus, C.; Jacquemin, M.; Heyen, G.; Karelovic, A.; Ruiz, P. Methanation of CO2: Further insight into the mechanism over Rh/γ-Al2O3 catalyst. Appl. Catal. B 2012, 113–114, 2–10. [Google Scholar] [CrossRef]
- Zhang, Z.; Tian, Y.; Zhang, L.; Hu, S.; Xiang, J.; Wang, Y.; Xu, L.; Liu, Q.; Zhang, S.; Hu, X. Impacts of nickel loading on properties, catalytic behaviors of Ni/γ–Al2O3 catalysts and the reaction intermediates formed in methanation of CO2. Int. J. Hydrogen Energy 2019, 44, 9291–9306. [Google Scholar] [CrossRef]
- Cárdenas-Arenas, A.; Quindimil, A.; Davó-Quiñonero, A.; Bailón-García, E.; Lozano-Castelló, D.; De-La-Torre, U.; Pereda-Ayo, B.; González-Marcos, J.A.; González-Velasco, J.R.; Bueno-López, A. Isotopic and in situ DRIFTS study of the CO2 methanation mechanism using Ni/CeO2 and Ni/Al2O3 catalysts. Appl. Catal. B 2020, 265, 118538. [Google Scholar] [CrossRef]
- Tada, S.; Shimizu, T.; Kameyama, H.; Haneda, T.; Kikuchi, R. Ni/CeO2 catalysts with high CO2 methanation activity and high CH4 selectivity at low temperatures. Int. J. Hydrogen Energy 2012, 37, 5527–5531. [Google Scholar] [CrossRef]
- Atzori, L.; Cutrufello, M.G.; Meloni, D.; Cannas, C.; Gazzoli, D.; Monaci, R.; Sini, M.F.; Rombi, E. Highly active NiO-CeO2 catalysts for synthetic natural gas production by CO2 methanation. Catal. Today 2018, 299, 183–192. [Google Scholar] [CrossRef]
- Ratchahat, S.; Sudoh, M.; Suzuki, Y.; Kawasaki, W.; Watanabe, R.; Fukuhara, C. Development of a powerful CO2 methanation process using a structured Ni/CeO2 catalyst. J. CO2 Util. 2018, 24, 210–219. [Google Scholar] [CrossRef]
- Cárdenas-Arenas, A.; Quindimil, A.; Davó-Quiñonero, A.; Bailón-García, E.; Lozano-Castelló, D.; De-La-Torre, U.; Pereda-Ayo, B.; González-Marcos, J.A.; González-Velasco, J.R.; Bueno-López, A. Design of active sites in Ni/CeO2 catalysts for the methanation of CO2: Tailoring the Ni-CeO2 contact. Appl. Mater. Today 2020, 19, 100591. [Google Scholar] [CrossRef]
- Sharma, S.; Hu, Z.; Zhang, P.; McFarland, E.W.; Metiu, H. CO2 methanation on Ru-doped ceria. J. Catal. 2011, 278, 297–309. [Google Scholar] [CrossRef]
- Vita, A.; Italiano, C.; Pino, L.; Frontera, P.; Ferraro, M.; Antonucci, V. Activity and stability of powder and monolith-coated Ni/GDC catalysts for CO2 methanation. Appl. Catal. B 2018, 226, 384–395. [Google Scholar] [CrossRef]
- Aldana, P.A.U.; Ocampo, F.; Kobl, K.; Louis, B.; Thibault-Starzyk, F.; Daturi, M.; Bazin, P.; Thomas, S.; Roger, A.C. Catalytic CO2 valorization into CH4 on Ni-based ceria-zirconia. Reaction mechanism by operando IR spectroscopy. Catal. Today 2013, 215, 201–207. [Google Scholar] [CrossRef]
- Ocampo, F.; Louis, B.; Roger, A.-C. Methanation of carbon dioxide over nickel-based Ce0.72Zr0.28O2 mixed oxide catalysts prepared by sol–gel method. Appl. Catal. A 2009, 369, 90–96. [Google Scholar] [CrossRef]
- Ashok, J.; Ang, M.L.; Kawi, S. Enhanced activity of CO2 methanation over Ni/CeO2-ZrO2 catalysts: Influence of preparation methods. Catal. Today 2017, 281, 304–311. [Google Scholar] [CrossRef]
- Le, T.A.; Kim, T.W.; Lee, S.H.; Park, E.D. Effects of Na content in Na/Ni/SiO2 and Na/Ni/CeO2 catalysts for CO and CO2 methanation. Catal. Today 2018, 303, 159–167. [Google Scholar] [CrossRef]
- Pastor-Pérez, L.; Patel, V.; Le Saché, E.; Reina, T.R. CO2 methanation in the presence of methane: Catalysts design and effect of methane concentration in the reaction mixture. J. Energy Inst. 2020, 93, 415–424. [Google Scholar] [CrossRef]
- Sun, H.; Zhang, Y.; Guan, S.; Huang, J.; Wu, C. Direct and highly selective conversion of captured CO2 into methane through integrated carbon capture and utilization over dual functional materials. J. CO2 Util. 2020, 38, 262–272. [Google Scholar] [CrossRef]
- Konishcheva, M.V.; Potemkin, D.I.; Badmaev, S.D.; Snytnikov, P.V.; Paukshtis, E.A.; Sobyanin, V.A.; Parmon, V.N. On the Mechanism of CO and CO2 Methanation Over Ni/CeO2 Catalysts. Top. Catal. 2016, 59, 1424–1430. [Google Scholar] [CrossRef]
- da Silva, D.C.D.; Letichevsky, S.; Borges, L.E.P.; Appel, L.G. The Ni/ZrO2 catalyst and the methanation of CO and CO2. Int. J. Hydrogen Energy 2012, 37, 8923–8928. [Google Scholar] [CrossRef]
- Lu, H.; Yang, X.; Gao, G.; Wang, K.; Shi, Q.; Wang, J.; Han, C.; Liu, J.; Tong, M.; Liang, X.; et al. Mesoporous zirconia-modified clays supported nickel catalysts for CO and CO2 methanation. Int. J. Hydrogen Energy 2014, 39, 18894–18907. [Google Scholar] [CrossRef]
- Jia, X.; Zhang, X.; Rui, N.; Hu, X.; Liu, C. Structural effect of Ni/ZrO2 catalyst on CO2 methanation with enhanced activity. Appl. Catal. B 2019, 244, 159–169. [Google Scholar] [CrossRef]
- Zhao, K.; Wang, W.; Li, Z. Highly efficient Ni/ZrO2 catalysts prepared via combustion method for CO2 methanation. J. CO2 Util. 2016, 16, 236–244. [Google Scholar] [CrossRef]
- Li, W.; Nie, X.; Jiang, X.; Zhang, A.; Ding, F.; Liu, M.; Liu, Z.; Guo, X.; Song, C. ZrO2 support imparts superior activity and stability of Co catalysts for CO2 methanation. Appl. Catal. B 2018, 220, 397–408. [Google Scholar] [CrossRef]
- Li, W.; Liu, Y.; Mu, M.; Ding, F.; Liu, Z.; Guo, X.; Song, C. Organic acid-assisted preparation of highly dispersed Co/ZrO2 catalysts with superior activity for CO2 methanation. Appl. Catal. B 2019, 254, 531–540. [Google Scholar] [CrossRef]
- Nagase, H.; Naito, R.; Tada, S.; Kikuchi, R.; Fujiwara, K.; Nishijima, M.; Honma, T. Ru nanoparticles supported on amorphous ZrO2 for CO2 methanation. Catal. Sci. Technol. 2020, 10, 4522–4531. [Google Scholar] [CrossRef]
- Takano, H.; Izumiya, K.; Kumagai, N.; Hashimoto, K. The effect of heat treatment on the performance of the Ni/(Zr-Sm oxide) catalysts for carbon dioxide methanation. Appl. Surf. Sci. 2011, 257, 8171–8176. [Google Scholar] [CrossRef]
- Takano, H.; Kirihata, Y.; Izumiya, K.; Kumagai, N.; Habazaki, H.; Hashimoto, K. Highly active Ni/Y-doped ZrO2 catalysts for CO2 methanation. Appl. Surf. Sci. 2016, 388, 653–663. [Google Scholar] [CrossRef]
- Kesavan, J.K.; Luisetto, I.; Tuti, S.; Meneghini, C.; Iucci, G.; Battocchio, C.; Mobilio, S.; Casciardi, S.; Sisto, R. Nickel supported on YSZ: The effect of Ni particle size on the catalytic activity for CO2 methanation. J. CO2 Util. 2018, 23, 200–211. [Google Scholar] [CrossRef]
- Kosaka, F.; Yamaguchi, T.; Ando, Y.; Mochizuki, T.; Takagi, H.; Matsuoka, K.; Fujishiro, Y.; Kuramoto, K. Effect of Ni content on CO2 methanation performance with tubular-structured Ni-YSZ catalysts and optimization of catalytic activity for temperature management in the reactor. Int. J. Hydrogen Energy 2020, 45, 12911–12920. [Google Scholar] [CrossRef]
- Ren, J.; Qin, X.; Yang, J.-Z.; Qin, Z.-F.; Guo, H.-L.; Lin, J.-Y.; Li, Z. Methanation of carbon dioxide over Ni–M/ZrO2 (M = Fe, Co, Cu) catalysts: Effect of addition of a second metal. Fuel Process. Technol. 2015, 137, 204–211. [Google Scholar] [CrossRef]
- Lu, H.; Yang, X.; Gao, G.; Wang, J.; Han, C.; Liang, X.; Li, C.; Li, Y.; Zhang, W.; Chen, X. Metal (Fe, Co, Ce or La) doped nickel catalyst supported on ZrO2 modified mesoporous clays for CO and CO2 methanation. Fuel 2016, 183, 335–344. [Google Scholar] [CrossRef]
- Dumrongbunditkul, P.; Witoon, T.; Chareonpanich, M.; Mungcharoen, T. Preparation and characterization of Co-Cu-ZrO2 nanomaterials and their catalytic activity in CO2 methanation. Ceram. Int. 2016, 42, 10444–10451. [Google Scholar] [CrossRef]
- Tan, J.; Wang, J.; Zhang, Z.; Ma, Z.; Wang, L.; Liu, Y. Highly dispersed and stable Ni nanoparticles confined by MgO on ZrO2 for CO2 methanation. Appl. Surf. Sci. 2019, 481, 1538–1548. [Google Scholar] [CrossRef]
- Takano, H.; Shinomiya, H.; Izumiya, K.; Kumagai, N.; Habazaki, H.; Hashimoto, K. CO2 methanation of Ni catalysts supported on tetragonal ZrO2 doped with Ca2+ and Ni2+ ions. Int. J. Hydrogen Energy 2015, 40, 8347–8355. [Google Scholar] [CrossRef]
- Loder, A.; Siebenhofer, M.; Lux, S. The reaction kinetics of CO2 methanation on a bifunctional Ni/MgO catalyst. J. Ind. Eng. Chem. 2020, 85, 196–207. [Google Scholar] [CrossRef]
- Wierzbicki, D.; Baran, R.; Debek, R.; Motak, M.; Grzybek, T.; Galvez, M.E.; Da Costa, P. The influence of nickel content on the performance of hydrotalcite-derived catalysts in CO2 methanation reaction. Int. J. Hydrogen Energy 2017, 42, 23548–23555. [Google Scholar] [CrossRef]
- Bette, N.; Thielemann, J.; Schreiner, M.; Mertens, F. Methanation of CO2 over a (Mg,Al)O-x Supported Nickel Catalyst Derived from a (Ni,Mg,Al)-Hydrotalcite-like Precursor. ChemCatChem 2016, 8, 2903–2906. [Google Scholar] [CrossRef]
- Abate, S.; Barbera, K.; Giglio, E.; Deorsola, F.; Bensaid, S.; Perathoner, S.; Pirone, R.; Centi, G. Synthesis, Characterization, and Activity Pattern of Ni-Al Hydrotalcite Catalysts in CO2 Methanation. Ind. Eng. Chem. Res. 2016, 55, 8299–8308. [Google Scholar] [CrossRef]
- Yan, Y.; Dai, Y.; He, H.; Yu, Y.; Yang, Y. A novel W-doped Ni-Mg mixed oxide catalyst for CO2 methanation. Appl. Catal. B 2016, 196, 108–116. [Google Scholar] [CrossRef]
- Varun, Y.; Sreedhar, I.; Singh, S.A. Highly stable M/NiO–MgO (M = Co, Cu and Fe) catalysts towards CO2 methanation. Int. J. Hydrogen Energy 2020. [Google Scholar] [CrossRef]
- Ho, P.H.; de Luna, G.S.; Angelucci, S.; Canciani, A.; Jones, W.; Decarolis, D.; Ospitali, F.; Aguado, E.R.; Rodríguez-Castellón, E.; Fornasari, G.; et al. Understanding structure-activity relationships in highly active La promoted Ni catalysts for CO2 methanation. Appl. Catal. B 2020, 278, 119256. [Google Scholar] [CrossRef]
- Wierzbicki, D.; Debek, R.; Motak, M.; Grzybek, T.; Galvez, M.E.; Da Costa, P. Novel Ni-La-hydrotalcite derived catalysts for CO2 methanation. Catal. Commun. 2016, 83, 5–8. [Google Scholar] [CrossRef]
- Wierzbicki, D.; Motak, M.; Grzybek, T.; Gálvez, M.E.; Da Costa, P. The influence of lanthanum incorporation method on the performance of nickel-containing hydrotalcite-derived catalysts in CO2 methanation reaction. Catal. Today 2018, 307, 205–211. [Google Scholar] [CrossRef]
- Zhang, L.; Bian, L.; Zhu, Z.; Li, Z. La-promoted Ni/Mg-Al catalysts with highly enhanced low-temperature CO2 methanation performance. Int. J. Hydrogen Energy 2018, 43, 2197–2206. [Google Scholar] [CrossRef]
- Wang, X.; Zhen, T.; Yu, C. Application of Ni-Al-hydrotalcite-derived catalyst modified with Fe or Mg in CO2 methanation. Appl. Petrochem. Res. 2016, 6, 217–223. [Google Scholar] [CrossRef]
- Mebrahtu, C.; Perathoner, S.; Giorgianni, G.; Chen, S.; Centi, G.; Krebs, F.; Palkovits, R.; Abate, S. Deactivation mechanism of hydrotalcite-derived Ni–AlOx catalysts during low-temperature CO2 methanation via Ni-hydroxide formation and the role of Fe in limiting this effect. Catal. Sci. Technol. 2019, 9, 4023–4035. [Google Scholar] [CrossRef]
- He, L.; Lin, Q.; Liu, Y.; Huang, Y. Unique catalysis of Ni-Al hydrotalcite derived catalyst in CO2 methanation: Cooperative effect between Ni nanoparticles and a basic support. J. Energy Chem. 2014, 23, 587–592. [Google Scholar] [CrossRef]
- Mebrahtu, C.; Krebs, F.; Perathoner, S.; Abate, S.; Centi, G.; Palkovits, R. Hydrotalcite based Ni–Fe/(Mg, Al)Ox catalysts for CO2 methanation – tailoring Fe content for improved CO dissociation, basicity, and particle size. Catal. Sci. Technol. 2018, 8, 1016–1027. [Google Scholar] [CrossRef]
- Jiménez, V.; Sánchez, P.; Panagiotopoulou, P.; Valverde, J.L.; Romero, A. Methanation of CO, CO2 and selective methanation of CO, in mixtures of CO and CO2, over ruthenium carbon nanofibers catalysts. Appl. Catal. A 2010, 390, 35–44. [Google Scholar] [CrossRef]
- Li, W.; Zhang, A.; Jiang, X.; Chen, C.; Liu, Z.; Song, C.; Guo, X. Low Temperature CO2 Methanation: ZIF-67-Derived Co-Based Porous Carbon Catalysts with Controlled Crystal Morphology and Size. ACS Sustain. Chem. Eng. 2017, 5, 7824–7831. [Google Scholar] [CrossRef]
- Gödde, J.; Merko, M.; Xia, W.; Muhler, M. Nickel nanoparticles supported on nitrogen–doped carbon nanotubes are a highly active, selective and stable CO2 methanation catalyst. J. Energy Chem. 2021, 54, 323–331. [Google Scholar] [CrossRef]
- Romero-Sáez, M.; Dongil, A.B.; Benito, N.; Espinoza-González, R.; Escalona, N.; Gracia, F. CO2 methanation over nickel-ZrO2 catalyst supported on carbon nanotubes: A comparison between two impregnation strategies. Appl. Catal. B 2018, 237, 817–825. [Google Scholar] [CrossRef]
- Wang, W.; Chu, W.; Wang, N.; Yang, W.; Jiang, C. Mesoporous nickel catalyst supported on multi-walled carbon nanotubes for carbon dioxide methanation. Int. J. Hydrogen Energy 2016, 41, 967–975. [Google Scholar] [CrossRef]
- Le, M.C.; Le Van, K.; Nguyen, T.H.T.; Nguyen, N.H. The Impact of Ce-Zr Addition on Nickel Dispersion and Catalytic Behavior for CO2 Methanation of Ni/AC Catalyst at Low Temperature. J. Chem. 2017, 4361056. [Google Scholar] [CrossRef]
- Gaidai, S.V.; Gryn’ko, V.S.; Zhludenko, M.G.; Dyachenko, A.G.; Tkach, V.M.; Ishchenko, O.V. Activity of Carbon-Fiber-Supported Fe-Co Catalysts in the CO2 Methanation Reaction. J. Superhard Mater. 2017, 39, 122–128. [Google Scholar] [CrossRef]
- Lapidus, A.L.; Gaidai, N.A.; Nekrasov, N.V.; Tishkova, L.A.; Agafonov, Y.A.; Myshenkova, T.N. The mechanism of carbon dioxide hydrogenation on copper and nickel catalysts. Pet. Chem. 2007, 47, 75–82. [Google Scholar] [CrossRef]
- Hussain, I.; Jalil, A.A.; Hassan, N.S.; Hambali, H.U.; Jusoh, N.W.C. Fabrication and characterization of highly active fibrous silica-mordenite (FS@SiO2-MOR) cockscomb shaped catalyst for enhanced CO2 methanation. Chem. Eng. Sci. 2020, 228, 115978. [Google Scholar] [CrossRef]
- Franken, T.; Heel, A. Are Fe based catalysts an upcoming alternative to Ni in CO2 methanation at elevated pressure? J. CO2 Util. 2020, 39, 101175. [Google Scholar] [CrossRef]
- Kitamura Bando, K.; Soga, K.; Kunimori, K.; Ichikuni, N.; Okabe, K.; Kusama, H.; Sayama, K.; Arakawa, H. CO2 hydrogenation activity and surface structure of zeolite-supported Rh catalysts. Appl. Catal. A 1998, 173, 47–60. [Google Scholar] [CrossRef]
- Bacariza, M.C.; Graça, I.; Lopes, J.M.; Henriques, C. Enhanced activity of CO2 hydrogenation to CH4 over Ni based zeolites through the optimization of the Si/Al ratio. Microporous Mesoporous Mater. 2018, 267, 9–19. [Google Scholar] [CrossRef]
- Bacariza, M.C.; Maleval, M.; Graça, I.; Lopes, J.M.; Henriques, C. Power-to-methane over Ni/zeolites: Influence of the framework type. Microporous Mesoporous Mater. 2019, 274, 102–112. [Google Scholar] [CrossRef]
- Goodarzi, F.; Kang, L.; Wang, F.R.; Joensen, F.; Kegnæs, S.; Mielby, J. Methanation of Carbon Dioxide over Zeolite-Encapsulated Nickel Nanoparticles. ChemCatChem 2018, 10, 1566–1570. [Google Scholar] [CrossRef]
- Guo, X.; Traitangwong, A.; Hu, M.; Zuo, C.; Meeyoo, V.; Peng, Z.; Li, C. Carbon Dioxide Methanation over Nickel-Based Catalysts Supported on Various Mesoporous Material. Energy Fuels 2018, 32, 3681–3689. [Google Scholar] [CrossRef]
- Isah, A.; Akanyeti, İ.; Oladipo, A.A. Methanation of CO2 over zeolite–promoted Ni/Al2O3 nanocatalyst under atmospheric pressure. Reac. Kinet. Mech. Cat. 2020, 130, 217–228. [Google Scholar] [CrossRef]
- Quindimil, A.; De-La-Torre, U.; Pereda, B.; González-Marcos, J.A.; González-Velasco, J.R. Ni catalysts with La as promoter supported over Y- and Beta- zeolites for CO2 methanation. Appl. Catal. B 2018, 238, 393–403. [Google Scholar] [CrossRef]
- Czuma, N.; Zarębska, K.; Motak, M.; Gálvez, M.E.; Da Costa, P. Ni/zeolite X derived from fly ash as catalysts for CO2 methanation. Fuel 2020, 267, 117139. [Google Scholar] [CrossRef]
- Wei, L.; Haije, W.; Kumar, N.; Peltonen, J.; Peurla, M.; Grenman, H.; de Jong, W. Influence of nickel precursors on the properties and performance of Ni impregnated zeolite 5A and 13X catalysts in CO2 methanation. Catal. Today 2020. [Google Scholar] [CrossRef]
- da Costa-Serra, J.F.; Cerdá-Moreno, C.; Chica, A. Zeolite-Supported Ni Catalysts for CO2 Methanation: Effect of Zeolite Structure and Si/Al Ratio. Appl. Sci. 2020, 10, 5131. [Google Scholar] [CrossRef]
- Bacariza, M.C.; Graça, I.; Bebiano, S.S.; Lopes, J.M.; Henriques, C. Magnesium as Promoter of CO2 Methanation on Ni-Based USY Zeolites. Energy Fuels 2017, 31, 9776–9789. [Google Scholar] [CrossRef]
- Graça, I.; González, L.V.; Bacariza, M.C.; Fernandes, A.; Henriques, C.; Lopes, J.M.; Ribeiro, M.F. CO2 hydrogenation into CH4 on NiHNaUSY zeolites. Appl. Catal. B 2014, 147, 101–110. [Google Scholar] [CrossRef]
- Bacariza, M.C.; Graça, I.; Lopes, J.M.; Henriques, C. Ni-Ce/Zeolites for CO2 Hydrogenation to CH4: Effect of the Metal Incorporation Order. ChemCatChem 2018, 10, 2773–2781. [Google Scholar] [CrossRef]
- Boix, A.V.; Ulla, M.A.; Petunchi, J.O. Promoting effect of Pt on Co mordenite upon the reducibility and catalytic behavior of CO2 hydrogenation. J. Catal. 1996, 162, 239–249. [Google Scholar] [CrossRef]
- Wei, L.; Kumar, N.; Haije, W.; Peltonen, J.; Peurla, M.; Grénman, H.; de Jong, W. Can bi-functional nickel modified 13X and 5A zeolite catalysts for CO2 methanation be improved by introducing ruthenium? Mol. Catal. 2020, 494, 111115. [Google Scholar] [CrossRef]
- Bacariza, M.C.; Graça, I.; Westermann, A.; Ribeiro, M.F.; Lopes, J.M.; Henriques, C. CO2 Hydrogenation Over Ni-Based Zeolites: Effect of Catalysts Preparation and Pre-reduction Conditions on Methanation Performance. Top Catal. 2015, 59, 314–325. [Google Scholar] [CrossRef]
- Bacariza, M.C.; Amjad, S.; Teixeira, P.; Lopes, J.M.; Henriques, C. Boosting Ni Dispersion on Zeolite-Supported Catalysts for CO2 Methanation: The Influence of the Impregnation Solvent. Energy Fuels 2020, 34, 14656–14666. [Google Scholar] [CrossRef]
- Bacariza, M.C.; Bértolo, R.; Graça, I.; Lopes, J.M.; Henriques, C. The effect of the compensating cation on the catalytic performances of Ni/USY zeolites towards CO2 methanation. J. CO2 Util. 2017, 21, 280–291. [Google Scholar] [CrossRef]
- Delmelle, R.; Duarte, R.B.; Franken, T.; Burnat, D.; Holzer, L.; Borgschulte, A.; Heel, A. Development of improved nickel catalysts for sorption enhanced CO2 methanation. Int. J. Hydrogen Energy 2016, 41, 20185–20191. [Google Scholar] [CrossRef]
- Borgschulte, A.; Gallandat, N.; Probst, B.; Suter, R.; Callini, E.; Ferri, D.; Arroyo, Y.; Erni, R.; Geerlings, H.; Züttel, A. Sorption enhanced CO2 methanation. Phys. Chem. Chem. Phys. 2013, 15, 9620–9625. [Google Scholar] [CrossRef] [PubMed]
- Walspurger, S.; Elzinga, G.D.; Dijkstra, J.W.; Sarić, M.; Haije, W.G. Sorption enhanced methanation for substitute natural gas production: Experimental results and thermodynamic considerations. Chem. Eng. J. 2014, 242, 379–386. [Google Scholar] [CrossRef]
- Westermann, A.; Azambre, B.; Bacariza, M.C.; Graça, I.; Ribeiro, M.F.; Lopes, J.M.; Henriques, C. Insight into CO2 methanation mechanism over NiUSY zeolites: An operando IR study. Appl. Catal. B 2015, 174–175, 120–125. [Google Scholar] [CrossRef]
- Westermann, A.; Azambre, B.; Bacariza, M.C.; Graça, I.; Ribeiro, M.F.; Lopes, J.M.; Henriques, C. The promoting effect of Ce in the CO2 methanation performances on NiUSY zeolite: A FTIR In Situ/Operando study. Catal. Today 2017, 283, 74–81. [Google Scholar] [CrossRef]
- Lu, X.; Gu, F.; Liu, Q.; Gao, J.; Liu, Y.; Li, H.; Jia, L.; Xu, G.; Zhong, Z.; Su, F. VOx promoted Ni catalysts supported on the modified bentonite for CO and CO2 methanation. Fuel Process. Technol. 2015, 135, 34–46. [Google Scholar] [CrossRef]
- Le, T.A.; Kang, J.K.; Park, E.D. CO and CO2 Methanation Over Ni/SiC and Ni/SiO2 Catalysts. Top Catal. 2018, 61, 1537–1544. [Google Scholar] [CrossRef]
- Zhi, G.; Guo, X.; Wang, Y.; Jin, G.; Guo, X. Effect of La2O3 modification on the catalytic performance of Ni/SiC for methanation of carbon dioxide. Catal. Commun. 2011, 16, 56–59. [Google Scholar] [CrossRef]
- Kirchner, J.; Anolleck, J.K.; Loesch, H.; Kureti, S. Methanation of CO2 on iron based catalysts. Appl. Catal. B-Environ. 2018, 223, 47–59. [Google Scholar] [CrossRef]
- Baysal, Z.; Kureti, S. CO2 methanation on Mg-promoted Fe catalysts. Appl. Catal. B 2020, 262, 118300. [Google Scholar] [CrossRef]
- Wang, Y.; Arandiyan, H.; Scott, J.; Dai, H.; Amal, R. Hierarchically Porous Network-Like Ni/Co3O4: Noble Metal-Free Catalysts for Carbon Dioxide Methanation. Adv. Sustain. Syst. 2018, 2. [Google Scholar] [CrossRef]
- Wang, K.; Li, W.; Huang, J.; Huang, J.; Zhan, G.; Li, Q. Enhanced active site extraction from perovskite LaCoO3 using encapsulated PdO for efficient CO2 methanation. J. Energy Chem. 2021, 53, 9–19. [Google Scholar] [CrossRef]
- Zhen, W.; Li, B.; Lu, G.; Ma, J. Enhancing catalytic activity and stability for CO2 methanation on Ni@MOF-5 via control of active species dispersion. Chem. Commun. 2015, 51, 1728–1731. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Rui, N.; Fan, Z.; Liu, C. Effect of the structure of Ni/TiO2 catalyst on CO2 methanation. Int. J. Hydrogen Energy 2016, 41, 22017–22025. [Google Scholar] [CrossRef]
- Liu, J.; Li, C.; Wang, F.; He, S.; Chen, H.; Zhao, Y.; Wei, M.; Evans, D.G.; Duan, X. Enhanced low-temperature activity of CO2 methanation over highly-dispersed Ni/TiO2 catalyst. Catal. Sci. Technol. 2013, 3, 2627–2633. [Google Scholar] [CrossRef]
- Petala, A.; Panagiotopoulou, P. Methanation of CO2 over alkali-promoted Ru/TiO2 catalysts: I. Effect of alkali additives on catalytic activity and selectivity. Appl. Catal. B 2018, 224, 919–927. [Google Scholar] [CrossRef]
- Qin, Z.; Wang, X.; Dong, L.; Su, T.; Li, B.; Zhou, Y.; Jiang, Y.; Luo, X.; Ji, H. CO2 methanation on Co/TiO2 catalyst: Effects of Y on the support. Chem. Eng. Sci. 2019, 210, 115245. [Google Scholar] [CrossRef]
- Cerdá-Moreno, C.; Chica, A.; Keller, S.; Rautenberg, C.; Bentrup, U. Ni-sepiolite and Ni-todorokite as efficient CO2 methanation catalysts: Mechanistic insight by operando DRIFTS. Appl. Catal. B 2020, 264, 118546. [Google Scholar] [CrossRef]
- Liang, C.; Gao, Z.; Lian, H.; Li, X.; Zhang, S.; Liu, Q.; Dong, D.; Hu, X. Impacts of metal loading in Ni/attapulgite on distribution of the alkalinity sites and reaction intermediates in CO2 methanation reaction. Int. J. Hydrogen Energy 2020, 45, 16153–16160. [Google Scholar] [CrossRef]
- Branco, J.B.; Ferreira, A.C. Methanation of CO2 Over Bimetallic Ni–5f Block Element (Th, U) Oxides. Eur. J. Inorg. Chem. 2019, 2019, 1039–1045. [Google Scholar] [CrossRef]
- Ferreira, A.C.; Branco, J.B. Methanation of CO2 over nanostructured nickel-4f block element bimetallic oxides. Int. J. Hydrogen Energy 2019, 44, 6505–6513. [Google Scholar] [CrossRef]
- Ferreira, A.C.; Branco, J.B. New approach for the synthesis of nanostructured binary f-block intermetallic compounds: CO2 methanation studies. Intermetallics 2019, 108, 32–38. [Google Scholar] [CrossRef]
- Branco, J.B.; da Silva, R.P.; Ferreira, A.C. Methanation of CO2 over Cobalt-Lanthanide Aerogels: Effect of Calcination Temperature. Catalysts 2020, 10, 704. [Google Scholar] [CrossRef]
- Kim, A.; Debecker, D.P.; Devred, F.; Dubois, V.; Sanchez, C.; Sassoye, C. CO2 methanation on Ru/TiO2 catalysts: On the effect of mixing anatase and rutile TiO2 supports. Appl. Catal. B 2018, 220, 615–625. [Google Scholar] [CrossRef]
- Marwood, M.; Doepper, R.; Renken, A. In-situ surface and gas phase analysis for kinetic studies under transient conditions The catalytic hydrogenation of CO2. Appl. Catal. A 1997, 151, 223–246. [Google Scholar] [CrossRef]
- Wei, W.; Jinlong, G. Methanation of carbon dioxide: An overview. Front. Chem. Sci. Eng. 2010, 5, 2–10. [Google Scholar] [CrossRef]
- Remya, V.R.; Kurian, M. Synthesis and catalytic applications of metal–organic frameworks: A review on recent literature. Int. Nano. Lett. 2019, 9, 17–29. [Google Scholar] [CrossRef]
- Safaei, M.; Foroughi, M.M.; Ebrahimpoor, N.; Jahani, S.; Omidi, A.; Khatami, M. A review on metal-organic frameworks: Synthesis and applications. TrAC Trends Anal. Chem. 2019, 118, 401–425. [Google Scholar] [CrossRef]
- Liang, J.; Liang, Z.; Zou, R.; Zhao, Y. Heterogeneous Catalysis in Zeolites, Mesoporous Silica, and Metal–Organic Frameworks. Adv. Mater. 2017, 29, 1701139. [Google Scholar] [CrossRef]
- Balakrishnan, M.S.; Batra, V.J.; Hargreaves, J.S.; Pulford, D.I. Waste materials – catalytic opportunities: An overview of the application of large scale waste materials as resources for catalytic applications. Green Chem. 2011, 13, 16–24. [Google Scholar] [CrossRef]



| Catalyst | Preparation Method | Tred (°C) | H2:CO2 | QT/W (mL g−1 h−1) | Best Catalytic Performances | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| T (°C) | XCO2 (%) | SCH4 (%) | ||||||
| 10Ni/SiO2 | Impregnation | 500 | 4:1 | 120,000 | 350 | 10 | 90 | [33] |
| 40Ni/SiO2 | Impregnation | 500 | 4:1 | 10,000 | 370 | 62 | 90 | [34] |
| 40Ni/SiO2 | Ammonia-evaporation | 500 | 4:1 | 10,000 | 370 | 80 | 95 | [34] |
| 10Ni-MgO/SiO2 | Co-impregnation | 450 | 4:1 | 24,000 | 400 | 72 | 98 | [35] |
| 6.2Pd-MgO/SiO2 | Reverse microemulsion | 450 | 4:1 | 7320 | 450 | 59 | 95 | [36,37] |
| 80Ni-Co/SiO2 | Co-precipitation | 350 | 4:1 | 30,000 | 350 | 49 | 98 | [38] |
| 1Rh/CeO2/SiO2 | Impregnation | 500 | 3:1 | n.a. | 230 | 10 | n.a. | [39] |
| 6Ni-La-Mo/SiO2 | Impregnation | 700 | 4:1 | 15,000 | 300 | 75 | 100 | [40] |
| Catalyst | Preparation Method | SA a (m2 g−1) | Tred (°C) | H2:CO2 | QT/W (mL g−1 h−1) | Best Catalytic Performances | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|
| T (°C) | XCO2 (%) | SCH4 (%) | |||||||
| 5Rh/MSN | Impregnation | 933 (3.6) | 500 | 4:1 | 50,000 | 350 | 99 | 100 | [43] |
| 10Ni/MSN | Impregnation | 662 (3.5) | 500 | 4:1 | 50,000 | 350 | 85 | 100 | [44] |
| 50Ni/MSN | Impregnation | 134 (2.3) | 450 | 4:1 | 15,000 | 375 | 96 | 100 | [45] |
| 20Co/KIT–6 | Impregnation | 369 (6.4) | 400 | 4.6:1 | 22,000 | 280 | 49 | 100 | [46,47] |
| 25Co–KIT–6 | Impregnation | 323 (~8) | 400 | 4:1 | 60,000 | 360 | 53 | 95 | [48,49] |
| Fe–KIT–6 | HS b | 435 (6.5) | 350 | 4:1 | 50,000 | 500 | 16 | 3 | [50] |
| 20Ni–V2O5/KIT–6 | Impregnation | n.a. | 550 | 4:1 | 96,000 | 350 | 87 | 100 | [51] |
| 15Ni-SBA–15 | One-pot HS b | 574 (4.5) | 500 | 4:1 | 10,000 | 420 | 76 | 96 | [52] |
| 10Ni/SBA–15 | Grafting | 551 (6.3) | 550 | 4:1 | 20,000 | 450 | 80 | 92 | [53] |
| 15Ni/SBA–15 | Impregnation | 235 (8.7) | 470 | 4:1 | 86,200 | 400 | 65 | 93 | [54] |
| CeO2/15Ni/SBA–15 | Impregnation | 320 (8.3) | 470 | 4:1 | 86,200 | 350 | 70 | 97 | [54] |
| 74Ni–Pd/SBA–15 | Impregnation | 535 (6.4) | 600 | 4:1 | 6000 | 430 | 96 | 97 | [55] |
| 3Ni–MCM–41 | One-pot HS b | 1480 (2.9) | 700 | 4:1 | 5760 | 400 | 17 | 96 | [56] |
| 15Ni/MCM–41 | Impregnation | 847 (1.9) | 470 | 4:1 | 86,200 | 400 | 70 | 93 | [54] |
| CeO2/15Ni/MCM–41 | Impregnation | 589 (2.0) | 470 | 4:1 | 86,200 | 400 | 75 | 95 | [54] |
| 20Ni–CeO2/MCM–41 | Deposition-precipitation | 302 (4.6) | 470 | 4:1 | 3000 | 380 | 86 | 100 | [57] |
| Ni–Y2O3/MgO–MCM–41 | DS method c | 445 (3.4) | 600 | 4:1 | 9000 | 400 | 65 | 85 | [58] |
| KCC–1 | Microemulsion | 773 (4.6) | 550 | 4:1 | 50,000 | 450 | 49 | 84 | [59] |
| 5Co/KCC–1 | Impregnation | 318 (4.9) | 500 | 4:1 | 4500 | 400 | 72 | n.a. | [60] |
| 5Ni/KCC–1 | Impregnation | 537 (4.7) | 500 | 4:1 | 4500 | 400 | 93 | >95 | [60] |
| 20Ni/KCC–1 | Impregnation | 216 (3.4) | 500 | 4:1 | 12,000 | 375 | 82 | 98 | [61] |
| 10Ni/FDU–12 | Impregnation | 506 (8.6) | 600 | 4:1 | 60,000 | 425 | 79 | 97 | [62] |
| 10Ni–CeO2/FDU–-12 | Impregnation | 500 (8.7) | 600 | 4:1 | 60,000 | 400 | 81 | 97 | [62] |
| Catalyst | Preparation Method | Tred (°C) | H2:CO2 | QT/W (mL g−1 h−1) | Best Catalytic Performances | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| T (°C) | XCO2 (%) | SCH4 (%) | ||||||
| 55Ni/Al2O3 | Impregnation | n.a. | 5:1 | n.a. | 500 | 71 | 86 | [72,73] |
| 12Ni/Al2O3 | Impregnation | 500 | 5:1 | 30,000 | 425 | 80 | 100 | [74] |
| 3Ru/Al2O3 | Impregnation | n.a. | 5:1 | n.a. | 350 | 95 | 96 | [75] |
| 4Ru/Al2O3 | Impregnation | 300 | 5:1 | 30,000 | 375 | 85 | 100 | [74] |
| 30Ni–Fe–Al2O3 | Co-precipitation | 700 | 4:1 | 9600 | 220 | 58 | 99 | [76] |
| 13Ni–Fe–γ–Al2O3 | Co-precipitation | 500 | 4:1 | 353,000 | 450 | 66 | 92 | [77] |
| 36Ni–Fe–Al2O3 | Co-precipitation | 500 | 4:1 | 150,000 | 325 | 98 | 99 | [78] |
| 36Ni–Mn–Al2O3 | Co-precipitation | 500 | 4:1 | 150,000 | 300 | 92 | 99 | [79] |
| 8Ni–Co–Al2O3 | EISA one-pot | 800 | 4:1 | 15,000 | 400 | 80 | 98 | [80] |
| 10Ni–Co/γ–Al2O3 | Impregnation | 400 | 4:1 | 130 | 325 | 90 | 100 | [81] |
| 20Ni/CeO2/γ–Al2O3 | Impregnation | 450 | 3.5:1 | 9000 | 350 | 80 | 100 | [82] |
| 15Ni–CeO2/Al2O3 | Co-impregnation | 500 | 4:1 | 15,000 | 350 | 85 | 100 | [83] |
| CeO2/42Ni–Al2O3 | Hydrothermal | 500 | 4:1 | n.a. | 250 | 90 | 99 | [84] |
| 2Ru/CeO2/Al2O3 | Impregnation | 500 | 4:1 | 72 | 350 | 90 | 100 | [85] |
| 12Ni–Pr/γ–Al2O3 | Impregnation | 750 | 4:1 | 6000 | 300 | 98 | 100 | [86] |
| 15Ni–Mg–Al2O3 | EISA one-pot | 800 | 4:1 | 86,100 | 400 | 70 | 96 | [87] |
| 15Ni–CeO2–ZrO2/γ–Al2O3 | Impregnation | 600 | 4:1 | 3000 | 300 | 90 | 100 | [88] |
| 5Ru/Mn/CeO2/Al2O3 | Impregnation | n.a. | 4:1 | 636 | 200 | 98 | 91 | [89] |
| 5Ru/Mn/Ni/Al2O3 | Impregnation | n.a. | 4:1 | 500 | 400 | 100 | 72 | [90] |
| (CaO/11NiTiO3)/γ–Al2O3 | Impregnation | 700 | 4:1 | n.a. | 400 | 53 | n.a. | [91] |
| 12Ni/(ZrO2–Al2O3) | Impregnation | 450 | 3.5:1 | 8100 | 360 | 70 | 70 | [92] |
| 10Ni/(Al2O3–ZrO2) | Epoxide-driven sol-gel | 500 | 4:1 | 6000 | 340 | 77 | 100 | [93] |
| 5Ru/(TiO2–Al2O3) | Impregnation | 400 | 4:1 | 60,000 | 375 | 82 | 100 | [94] |
| 15Ni/(CaO–Al2O3) | Impregnation | 500 | 4:1 | n.a. | 450 | 66 | 92 | [95] |
| 10Ni–Ru–(CaO–Al2O3) | EISA one-pot | 600 | 4:1 | 30,000 | 380 | 84 | 100 | [96] |
| 20Ni/(γ–Al2O3–ZrO2–TiO2–CeO2) | Impregnation | 500 | 4:1 | 20,000 | 300 | 82 | 98 | [97] |
| 20Ni/(γ–Al2O3–ZrO2–TiO2–CeO2) | Impregnation | 500 | 4:1 | 60,000 | 350 | 90 | n.a. | [98] |
| Catalyst | Preparation Method | Tred (°C) | H2:CO2 | QT/W (mL g−1 h−1) | Best Catalytic Performances | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| T (°C) | XCO2 (%) | SCH4 (%) | ||||||
| 10Ni/CeO2 | Impregnation | 600 | 4:1 | n.a. | 300 | 90 | 100 | [85,120] |
| 32Ni-CeO2 | Soft-template | 400 | 4:1 | 72,000 | 300 | 87 | 100 | [121] |
| 10Ni/CeO2 | Impregnation | 500 | 4:1 | n.a. | 300 | 92 | 100 | [122] |
| 8.5Ni/CeO2 | Impregnation | 500 | 4:1 | 30,000 | 375 | 80 | 100 | [123] |
| Ce0.95Ru0.05O2 | Combustion | 500 | 4:1 | n.a. | 450 | 55 | 99 | [124] |
| 50Ni/CexGd1-xO2 | Solution-combustion | 800 | 4:1 | n.a. | 450 | 72 | n.a. | [125] |
| 5Ni-Ce0.72Zr0.28O2 | Sol-gel | 400 | 4:1 | 22,000 | 400 | 85 | 99 | [126,127] |
| 10Ni-CexZr1-xO2 | Ammonia evaporation | 450 | 4:1 | 22,000 | 275 | 55 | 100 | [128] |
| 13Ni-NaO/CeO2 | Impregnation | 500 | 50:1 | 60,000 | 250 | 97 | 96 | [129] |
| 16Ni-Co/CeO2-ZrO2 | Impregnation | 450 | 4:1 | 12,000 | 350 | 70 | 98 | [130] |
| Catalyst | Preparation Method | Tred (°C) | H2:CO2 | QT/W (mL g−1 h−1) | Best Catalytic Performances | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| T (°C) | XCO2 (%) | SCH4 (%) | ||||||
| 10Ni/ZrO2 | Impregnation | n.a. | 125:1 | 140,000 | 450 | 100 | n.a. | [133] |
| 15Ni/ZrO2 | Impregnation | 500 | 4:1 | n.a. | 350 | 80 | 99 | [134] |
| 10Ni/ZrO2 | Impregnation a | 500 | 4:1 | 10,000 | 350 | 80 | 97 | [135] |
| 15Ni/ZrO2 | Combustion | 500 | 4:1 | 48,000 | 400 | 85 | 100 | [136] |
| 10Co/ZrO2 | Impregnation | 400 | 4:1 | 3600 | 400 | 93 | 100 | [137] |
| 2Co/ZrO2 | Impregnation | 400 | 4:1 | 72,000 | 400 | 85 | 99 | [138] |
| 3Ru/ZrO2 | Selective deposition method | 300 | 4:1 | n.a. | 300 | 82 | 100 | [139] |
| 50Ni/Sm-ZrO2 | Mechanical mixture | 300 | 4:1 | 3000 | 350 | 95 | 100 | [140] |
| Ni/Y–ZrO2 | Co-impregnation | 400 | 4:1 | 6300 b | 400 | 38 c | [141] | |
| 10Ni/Y–ZrO2 | Impregnation with EDTA | 500 | 4:1 | 60,000 | 375 | 60 | 96 | [142] |
| 75Ni/Y–ZrO2 | Extrusion | 600 | 4:1 | n.a. | 300d | 92 | 98 | [143] |
| 30Ni–Fe/ZrO2 | Co-impregnation | 400 | 4:1 | 4980 | 270 | 100 | 95 | [144] |
| 30Ni–Co/ZrO2 | Co-impregnation | 400 | 4:1 | 4980 | 270 | 100 | 95 | [144] |
| 30Ni–Cu/ZrO2 | Co-impregnation | 400 | 4:1 | 4980 | 330 | 88 | 88 | [144] |
| 15Ni–Fe/ZrO2 | Co-impregnation | 500 | 4:1 | n.a. | 400 | 78 | 96 | [145] |
| Co–Cu/ZrO2 | Co-precipitation | 300 | 3:1 | 14,400 | 300 | 68 | 83 | [146] |
| 6Ni–MgO/ZrO2 | Impregnation with citric acid | 450 | 4:1 | 15,000 | 300 | 95 | 100 | [147] |
| Ni–CaO/ZrO2 | Co-impregnation | 400 | 4:1 | 6742 b | 350 | 85 | 100 | [148] |
| Catalyst | Preparation Method | Tred (°C) | H2:CO2 | QT/W (mL g−1 h−1) | Best Catalytic Performances | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| T (°C) | XCO2 (%) | SCH4 (%) | ||||||
| 27Ni/MgO | Impregnation | n.a. | 4:1 | 3700 | 325 | 87 | >99 | [149] |
| 42.5Ni–HTderived | Co-precipitation | 900 | 4:1 | n.a. | 300 | 82 | 99 | [150] |
| 59Ni–HTderived | Co-precipitation | 900 | 4:1 | 1100 | 330 | 74 | 95 | [151] |
| 80Ni–HTderived | Co-precipitation | 500 | 4:1 | 20,000 | 300 | 85 | 100 | [152] |
| Ni–WOx–MgO | Co-precipitation | 500 | 4:1 | 60,000 | 300 | 85 | 100 | [153] |
| 2Co/NiO–MgO | Impregnation | 120 | 8:1 | 60,000 | 325 | 92 | 100 | [154] |
| 56Ni–La2O3–HTderived | Co-precipitation | 600 | 4:1 | 80,000 | 275 | 70 | 96 | [155] |
| 15Ni–La2O3–Ni–HTderived | Co-precipitation | 900 | 4:1 | n.a. | 300 | 75 | 98 | [156] |
| 21Ni–La2O3–HTderived | Ion-exchanged | 900 | 4:1 | n.a. | 300 | 82 | 100 | [157] |
| 15Ni–La2O3–HTderived | Urea hydrolysis | 700 | 4:1 | 45000 | 300 | 90 | 95 | [158] |
| Ni–Fe–HTderived | Co-precipitation | 700 | 4:1 | 12000 | 300 | 95 | 100 | [159] |
| 12Ni–Fe–HTderived | Co-precipitation | 500 | 4:1 | n.a. | 300 | 83 | 97 | [160] |
| 78Ni–K2O–Al2O3–HTderived | Co-precipitation | 500 | 4:1 | 75,000 | 350 | 85 | 100 | [161] |
| Catalyst | Preparation Method | Tred (°C) | H2:CO2 | QT/W (mL g−1 h−1) | Best Catalytic Performances | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| T (°C) | XCO2 (%) | SCH4 (%) | ||||||
| 0.5Ru/CNFs | Impregnation | 300 | 3.3:1 | 80,000 | 500 | 52 | 100 | [163] |
| 32.5Co–PC | Carbonization | 400 | 4:1 | 72,000 | 270 | 53 | 99 | [164] |
| 30Ni/NCNTs | Impregnation | 420 | 4:1 | 50,000 | 340 | 51 | 96 | [165] |
| 5Ni/ZrO2/CNTs | Impregnation | 500 | 5:1 | 20,000 | 400 | 55 | 96 | [166] |
| 12Ni–CeO2/CNTs | Ultrasonic-assisted co-impregnation | 350 | 4:1 | 30,000 | 350 | 84 | 100 | [167] |
| 7Ni/Ce0.2Zr0.8O2/AC | Impregnation | 600 | 4:1 | 40,000 | 300 | 85 | 100 | [168] |
| 30Fe–Co/CF | Uniform infiltration | 300 | 27:1 | 6600 | 390 | 65 | 88 | [169] |
| Catalyst | Preparation Method | Tred (°C) | H2:CO2 | QT/W (mL g−1 h−1) | Best Catalytic Performances | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| T (°C) | XCO2 (%) | SCH4 (%) | ||||||
| SiO2–MOR a | Microemulsion | 500 | 4:1 | 45,000 | 500 | 65 | 68 | [171] |
| 1Fe/13X | Impregnation | 400 | 4:1 | n.a. | 350 | 89 | 76 | [172] |
| 6Rh–Y | Ion exchange | 450 | 3:1 | 6000 | 150 | 6 | 100 | [173] |
| 15Ni/USY | Impregnation | 470 | 4:1 | 86,200 | 400 | 73 | 97 | [174] |
| 15Ni/MOR | Impregnation | 470 | 4:1 | 86,200 | 440 | 69 | 95 | [175] |
| 15Ni/ZSM-5 | Impregnation | 470 | 4:1 | 86,200 | 450 | 68 | 94 | [175] |
| 5Ni/d-S1 | Impregnation | 500 | 4:1 | 72,000 | 450 | 57 | 91 | [176] |
| 10Ni/ZSM-5 | Impregnation | 500 | 4:1 | n.a. | 400 | 76 | 99 | [177] |
| 10Ni–ZSM-5 | Hydrothermal method b | 400 | 3:1 | n.a. | 400 | 66 | 100 | [178] |
| 10Ni/BEA | Impregnation | 500 | 4:1 | 30,000 | 450 | 73 | 97 | [179] |
| 15Ni/X | Impregnation | 470 | 4:1 | 12,000 | 450 | 53 | 90 | [180] |
| 5Ni/13X | Evaporation impregnation | 500 | 4:1 | 13,333 | 320 | 80 | 100 | [181] |
| 5Ni/5A | Evaporation impregnation | 500 | 4:1 | 13,333 | 400 | 65 | 95 | [181] |
| 5Ni/ITQ-2 | Impregnation | 450 | 4:1 | 9000 | 400 | 82 | 99 | [182] |
| MgO/13Ni/USY | Impregnation | 700 | 4:1 | 86,200 | 400 | 63 | 93 | [183] |
| CeO2/14Ni/USY | Impregnation | 470 | 4:1 | 86,200 | 400 | 68 | 95 | [184] |
| 20Ni–CeO2/USY | Co-impregnation | 470 | 4:1 | 86,200 | 305 | 78 | 99 | [185] |
| 0.5Pt–Co–MOR | Ion exchange | 350 | 4:1 | n.a. | 350 | 41 | 15 | [186] |
| 10Ni/La2O3/USY | Impregnation | 500 | 4:1 | 30,000 | 400 | 75 | 100 | [179] |
| 2.5Ni–2.5Ru/13X | Evaporation impregnation | 500 | 4:1 | 13,333 | 400 | 65 | 92 | [187] |
| 4Ni–1Ru/5A | Evaporation impregnation | 500 | 4:1 | 13,333 | 400 | 60 | 94 | [187] |
| Catalyst | Preparation Method | Tred (°C) | H2:CO2 | QT/W (mL g−1 h−1) | Best Catalytic Performances | Ref. | ||
|---|---|---|---|---|---|---|---|---|
| T (°C) | XCO2 (%) | SCH4 (%) | ||||||
| 20Ni/Bentonite | Impregnation | 550 | 4:1 | 30,000 | 400 | 74 | 90 | [196] |
| 20Ni–VOx/Bentonite | Impregnation | 550 | 4:1 | 30,000 | 380 | 89 | 87 | [196] |
| 20Ni/SiC | Deposition-precipitation | 600 | 50:1 | 60,000 | 325 | 100 | 100 | [197] |
| 15Ni/SiC | Impregnation | 400 | 4:1 | n.a. | 400 | 80 | 99 | [198] |
| 15Ni–La2O3/SiC | Co-impregnation | 400 | 4:1 | n.a. | 350 | 85 | 99 | [198] |
| γ–Fe2O3 | Commercial | 450 | 200:1 | 150,000 | 400 | 50 | 70 | [199] |
| Mg/α–Fe2O3 | Impregnation | 350 | 4:1 | 150,000 | 400 | 49 | 65 | [200] |
| 12Ni/Co3O4 | Impregnation | 450 | 4:1 | 48,000 | 250 | 100 | 100 | [201] |
| 3Pd–LaCoO3 | One pot | n.a. | 3:1 | 18,000 | 300 | 62 | >99 | [202] |
| 10Ni/MOF-5 | Impregnation | n.a. | 4:1 | 7500 | 320 | 75 | 100 | [203] |
| 6.2Ni/TiO2 | Impregnation a | 700 | 4:1 | n.a. | 350 | 73 | 100 | [204] |
| 15Ni/TiO2 | Deposition-precipitation | 450 | 4:1 | 2400 | 260 | 96 | 99 | [205] |
| 5Ru/TiO2 | Impregnation | 300 | 4:1 | 90,000 | 300 | 80 | 100 | [206] |
| 20Co/Y2O3–TiO2 | Deposition-precipitation | 400 | 4:1 | 3600 | 350 | 86 | 100 | [207] |
| 5Ni–Sepiolite | Precipitation | 450 | 4:1 | 9000 | 400 | 88 | 100 | [208] |
| 15Ni–Todorokite | Sequential method | 450 | 4:1 | 9000 | 300 | 90 | 100 | [208] |
| 25Ni/Attapulgite | Impregnation | 600 | 4:1 | 11,400 | 400 | 85 | 99 | [209] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bacariza, M.C.; Spataru, D.; Karam, L.; Lopes, J.M.; Henriques, C. Promising Catalytic Systems for CO2 Hydrogenation into CH4: A Review of Recent Studies. Processes 2020, 8, 1646. https://doi.org/10.3390/pr8121646
Bacariza MC, Spataru D, Karam L, Lopes JM, Henriques C. Promising Catalytic Systems for CO2 Hydrogenation into CH4: A Review of Recent Studies. Processes. 2020; 8(12):1646. https://doi.org/10.3390/pr8121646
Chicago/Turabian StyleBacariza, M. Carmen, Daniela Spataru, Leila Karam, José M. Lopes, and Carlos Henriques. 2020. "Promising Catalytic Systems for CO2 Hydrogenation into CH4: A Review of Recent Studies" Processes 8, no. 12: 1646. https://doi.org/10.3390/pr8121646
APA StyleBacariza, M. C., Spataru, D., Karam, L., Lopes, J. M., & Henriques, C. (2020). Promising Catalytic Systems for CO2 Hydrogenation into CH4: A Review of Recent Studies. Processes, 8(12), 1646. https://doi.org/10.3390/pr8121646








