Improving Ammonium Sorption of Bayah Natural Zeolites by Hydrothermal Method
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization
2.3. Adsorption Test
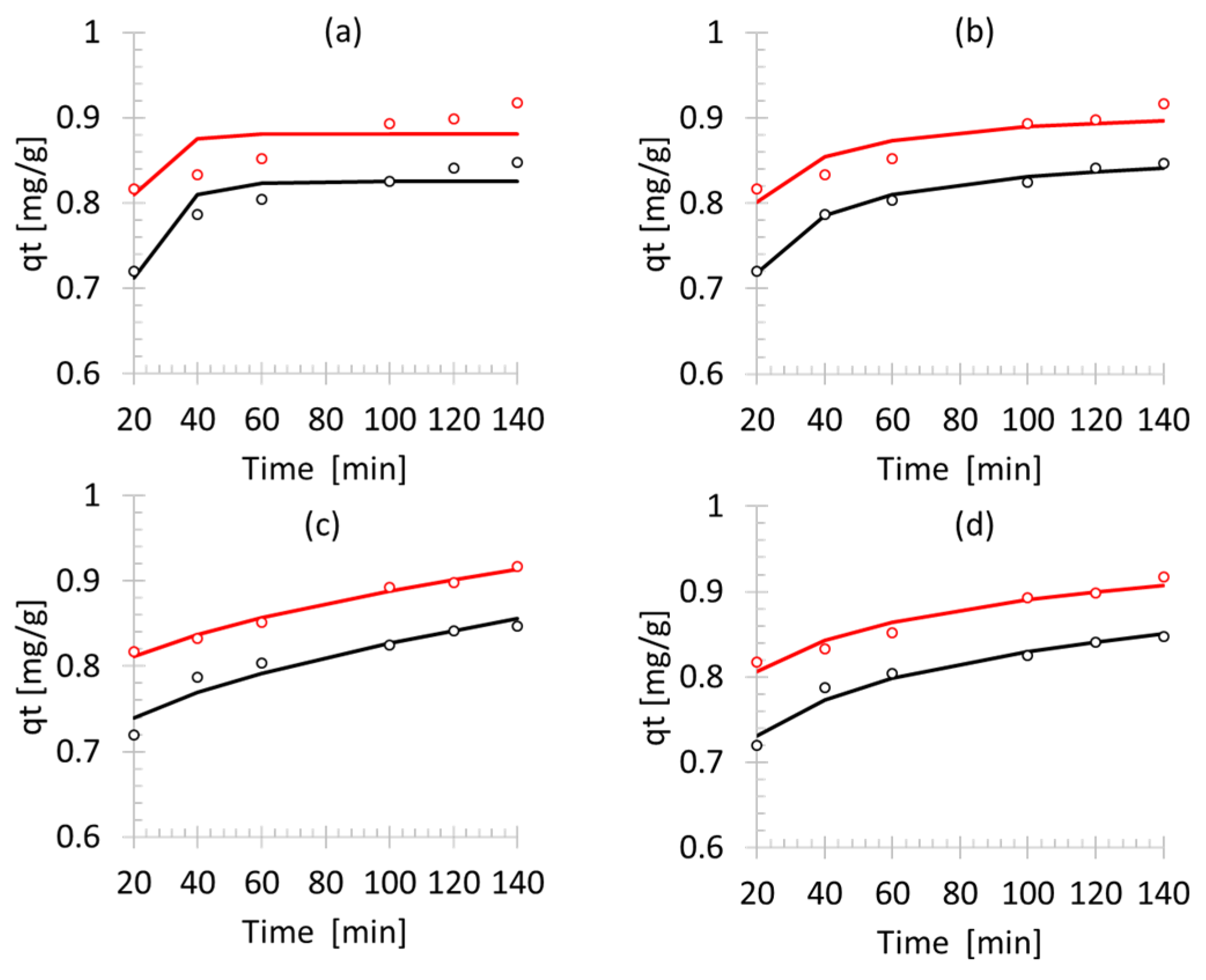
2.4. Kinetic Model
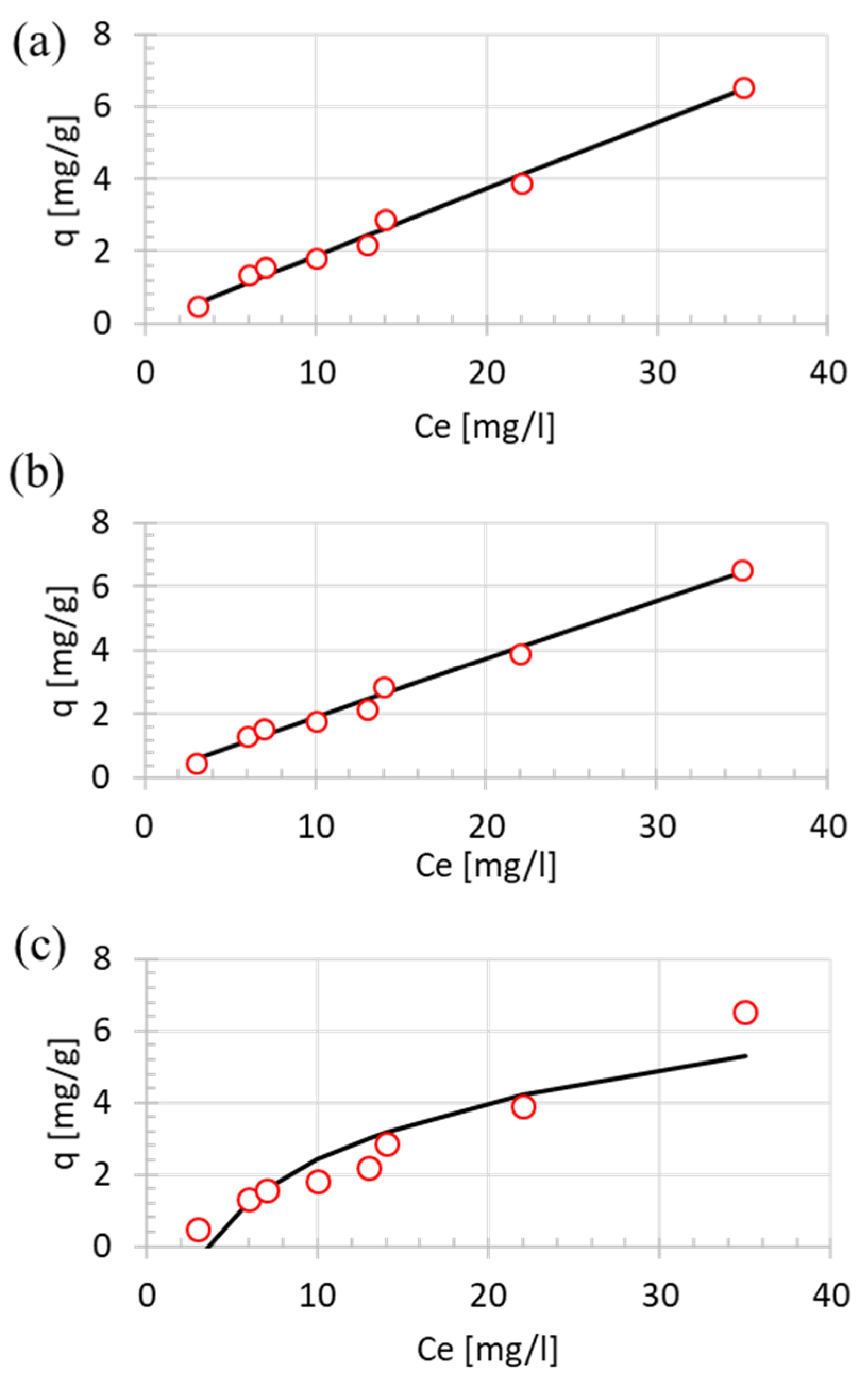
2.5. Isotherm Model
3. Results and Discussion
3.1. Characterization
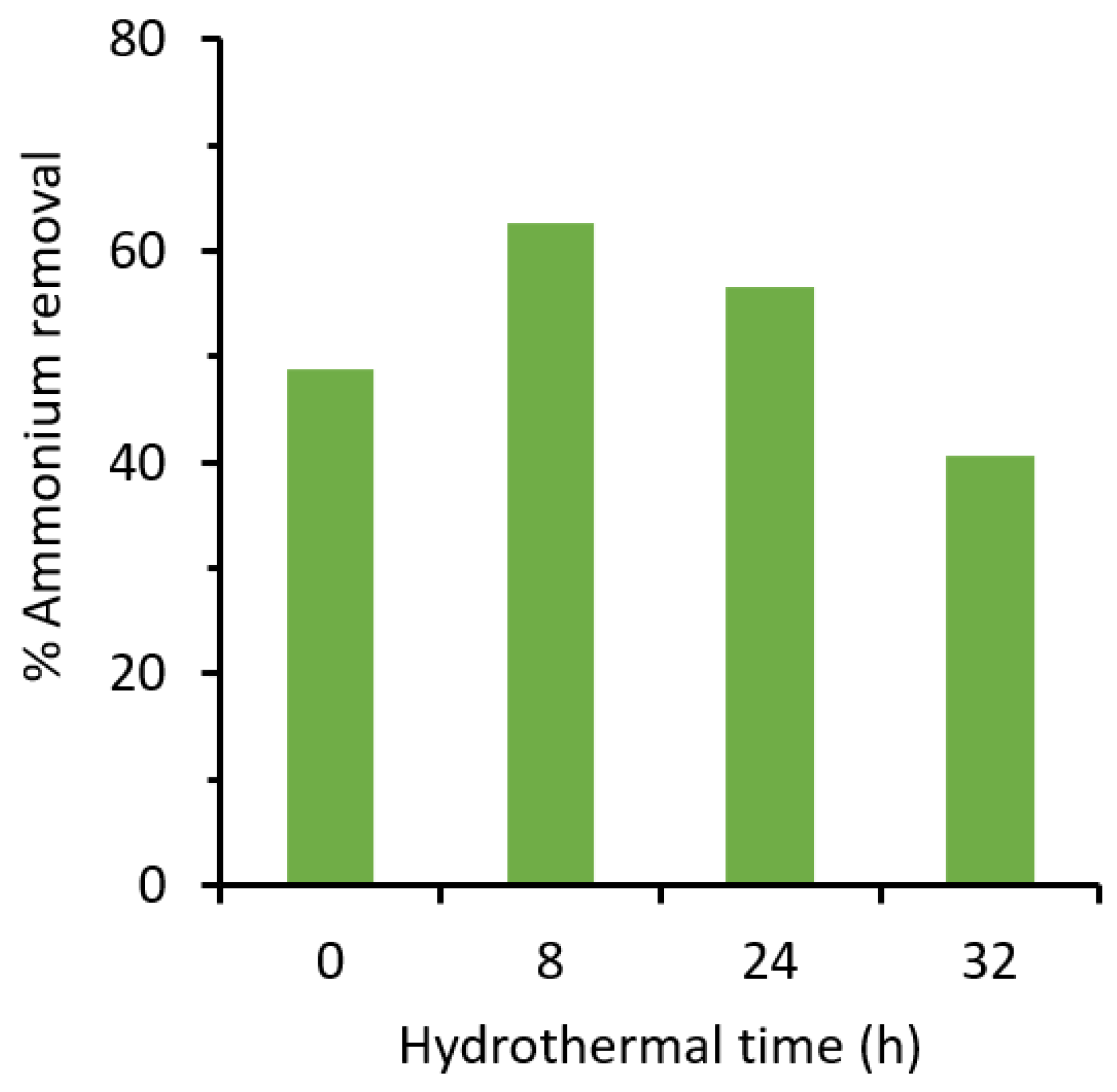
3.2. Ammonium Removal
3.3. Kinetic Model
3.4. Isotherm Model
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gultom, F.; Wirjosentono, B.; Nainggolan, H. Preparation and characterization of North Sumatera natural zeolite polyurethane nanocomposite foams for light-weight engineering materials. Procedia Chem. 2016, 19, 1007–1013. [Google Scholar] [CrossRef]
- Puspitasari, T.; Kadja, G.; Radiman, C.; Darwis, D.; Mukti, R. Two-step preparation of amidoxime-functionalized natural zeolites hybrids for the removal of Pb2+ ions in aqueous environment. Mater. Chem. Phys. 2018, 216, 197–205. [Google Scholar] [CrossRef]
- Djubaedah; Wulandari, D.A.; Nasruddin, N. Preliminary study of natural zeolite from Bayah for solar powered cooling application. IOP Conf. Ser. Earth Environ. Sci. 2018, 105, 012001. [Google Scholar] [CrossRef]
- Sumantri, I.; Buchori, L.; Mukti, F.A.W.; Ramadhani, F.; Anggoro, D.D. (Eds.) Study of the rate of adsorption of toxic gases in shrimp ponds using Sukabumi natural zeolite, AIP Publishing LLC. AIP Conf. Proc. 2020, 2197, 120005. [Google Scholar]
- Prasetya, A.; Prihutami, P.; Warisaura, A.D.; Fahrurrozi, M.; Petrus, H.T.B.M. Characteristic of Hg removal using zeolite adsorption and Echinodorus palaefolius phytoremediation in subsurface flow constructed wetland (SSF-CW) model. J. Environ. Chem. Eng. 2020, 8, 103781. [Google Scholar] [CrossRef]
- Kusuma, R.I.; Hadinoto, J.P.; Ayucitra, A.; Soetaredjo, F.E.; Ismadji, S. Natural zeolite from Pacitan Indonesia, as catalyst support for transesterification of palm oil. Appl. Clay Sci. 2013, 74, 121–126. [Google Scholar] [CrossRef]
- Rosalia, S.S.; Wilhelmus, L.H.; Nunuk, H.S.; Paulina, T. (Eds.) Study of the Use of Mamasa Natural Zeolite which is Activated by Acid as a Catalyst for Cracking Palm Oil Methyl Esters. Mater. Sci. Forum Trans. Tech. Publ. 2019, 967, 155–160. [Google Scholar] [CrossRef]
- Mumpton, F.A. La roca magica: Uses of natural zeolites in agriculture and industry. Proc. Natl. Acad. Sci. USA 1999, 96, 3463–3470. [Google Scholar] [CrossRef] [PubMed]
- Jha, V.K.; Hayashi, S. Modification on natural clinoptilolite zeolite for its NH4+ retention capacity. J. Hazard. Mater. 2009, 169, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Lei, Z.; Wang, L.; Liu, X.; Zhang, Y.; Wan, C.; Lee, D.J.; Tay, J.H. Adsorption mechanisms of high-levels of ammonium onto natural and NaCl-modified zeolites. Sep. Purif. Technol. 2013, 103, 15–20. [Google Scholar] [CrossRef]
- Soetardji, J.P.; Claudia, J.C.; Ju, Y.-H.; Hriljac, J.A.; Chen, T.-Y.; Soetaredjo, F.E.; Santoso, S.P.; Kurniawan, A.; Ismadji, S. Ammonia removal from water using sodium hydroxide modified zeolite mordenite. RSC Adv. 2015, 5, 83689–83699. [Google Scholar] [CrossRef]
- Wakihara, T.; Ichikawa, R.; Tatami, J.; Endo, A.; Yoshida, K.; Sasaki, Y.; Komeya, K.; Meguro, T. Bead-Milling and Postmilling Recrystallization: An Organic Template-free Methodology for the Production of Nano-zeolites. Cryst. Growth Des. 2011, 11, 955–958. [Google Scholar] [CrossRef]
- Kurniawan, T.; Muraza, O.; Hakeem, A.S.; Al-Amer, A.M. Mechanochemical route and recrystallization strategy to fabricate mordenite nanoparticles from natural zeolites. Cryst. Growth Des. 2017, 17, 3313–3320. [Google Scholar] [CrossRef]
- Kurniawan, T.; Muraza, O.; Bakare, I.A.; Sanhoob, M.A.; Al-Amer, A.M. Isomerization of n-butane over cost-effective mordenite catalysts fabricated via recrystallization of natural zeolites. Ind. Eng. Chem. Res. 2018, 57, 1894–1902. [Google Scholar] [CrossRef]
- Millar, G.J.; Winnett, A.; Thompson, T.; Couperthwaite, S.J. Equilibrium studies of ammonium exchange with Australian natural zeolites. J. Water Process Eng. 2016, 9, 47–57. [Google Scholar] [CrossRef]
- Kang, S.-J.; Egashira, K. Modification of different grades of Korean natural zeolites for increasing cation exchange capacity. Appl. Clay Sci. 1997, 12, 131–144. [Google Scholar] [CrossRef]
- Watanabe, Y.; Yamada, H.; Tanaka, J.; Moriyoshi, Y. Hydrothermal modification of natural zeolites to improve uptake of ammonium ions. J. Chem. Technol. Biotechnol. 2005, 80, 376–380. [Google Scholar] [CrossRef]
- Luo, X.-M.; Yin, W.-Z.; Wang, Y.-F.; Sun, C.-Y.; Ma, Y.-Q.; Liu, J. Effect and mechanism of siderite on reverse anionic flotation of quartz from hematite. J. Cent. South Univ. 2016, 23, 52–58. [Google Scholar] [CrossRef]
- Hosaka, M.; Miyata, T.; Sunagawa, I. Growth and morphology of quartz crystals synthesized above the transition temperature. J. Cryst. Growth 1995, 152, 300–306. [Google Scholar] [CrossRef]
- Wu, F.-C.; Tseng, R.-L.; Juang, R.-S. Characteristics of Elovich equation used for the analysis of adsorption kinetics in dye-chitosan systems. Chem. Eng. J. 2009, 150, 366–373. [Google Scholar] [CrossRef]
- Fu, H.; Li, Y.; Yu, Z.; Shen, J.; Li, J.; Zhang, M.; Ding, T.; Xu, L.; Lee, S.S. Ammonium removal using a calcined natural zeolite modified with sodium nitrate. J. Hazard Mater. 2020, 393, 122481. [Google Scholar] [CrossRef] [PubMed]
- Kotoulas, A.; Agathou, D.; Triantaphyllidou, I.E.; Tatoulis, T.I.; Akratos, C.S.; Tekerlekopoulou, A.G.; Vayenas, D.V. Zeolite as a Potential Medium for Ammonium Recovery and Second Cheese Whey Treatment. Water 2019, 11, 136. [Google Scholar] [CrossRef]
- Mazloomi, F.; Jalali, M. Ammonium removal from aqueous solutions by natural Iranian zeolite in the presence of organic acids, cations and anions. J. Environ. Chem. Eng. 2016, 4, 1664–1673. [Google Scholar] [CrossRef]
- He, W.; Gong, H.; Fang, K.; Peng, F.; Wang, K. Revealing the effect of preparation parameters on zeolite adsorption performance for low and medium concentrations of ammonium. J. Environ. Sci. 2019, 85, 177–188. [Google Scholar] [CrossRef]
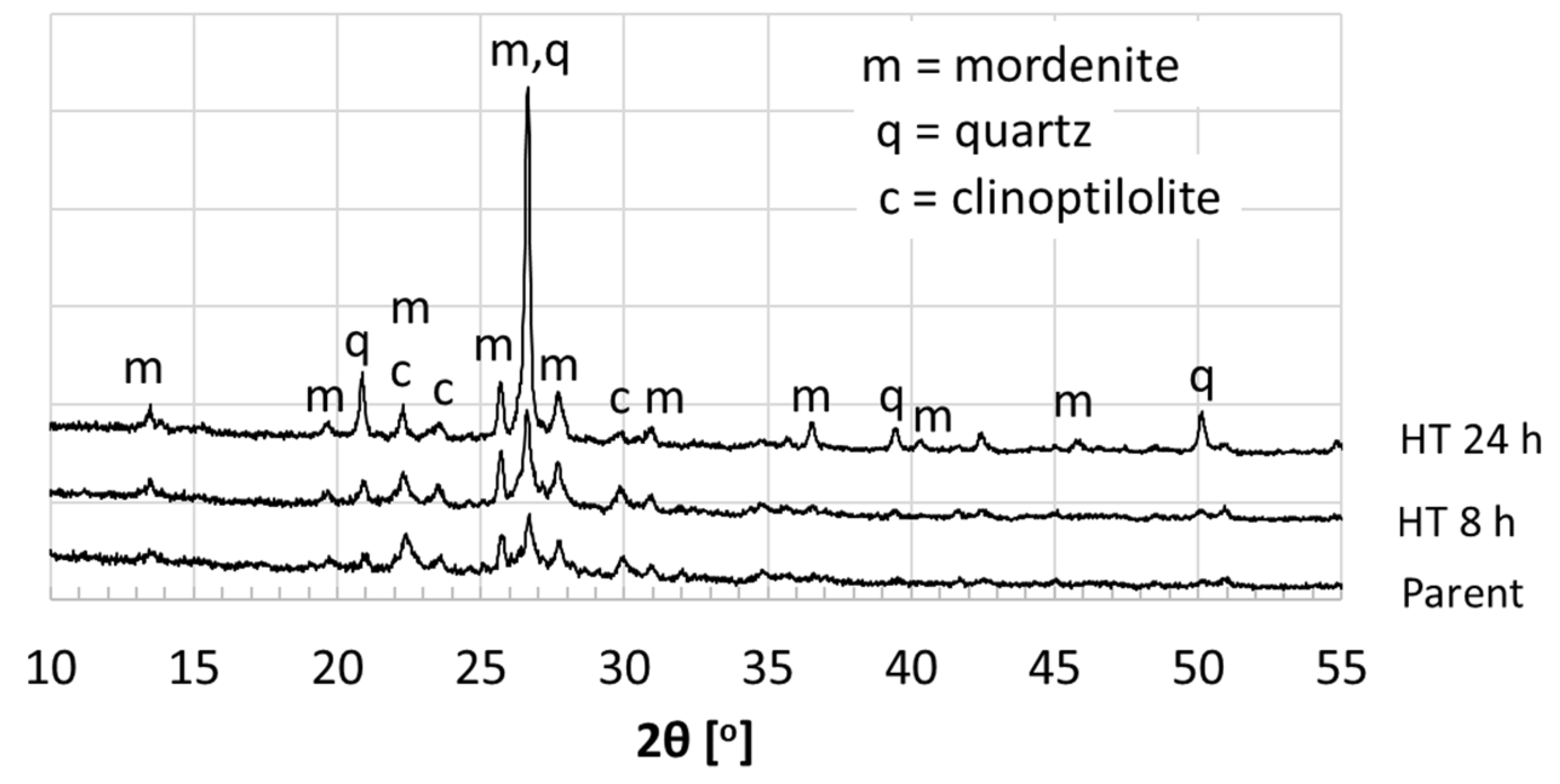
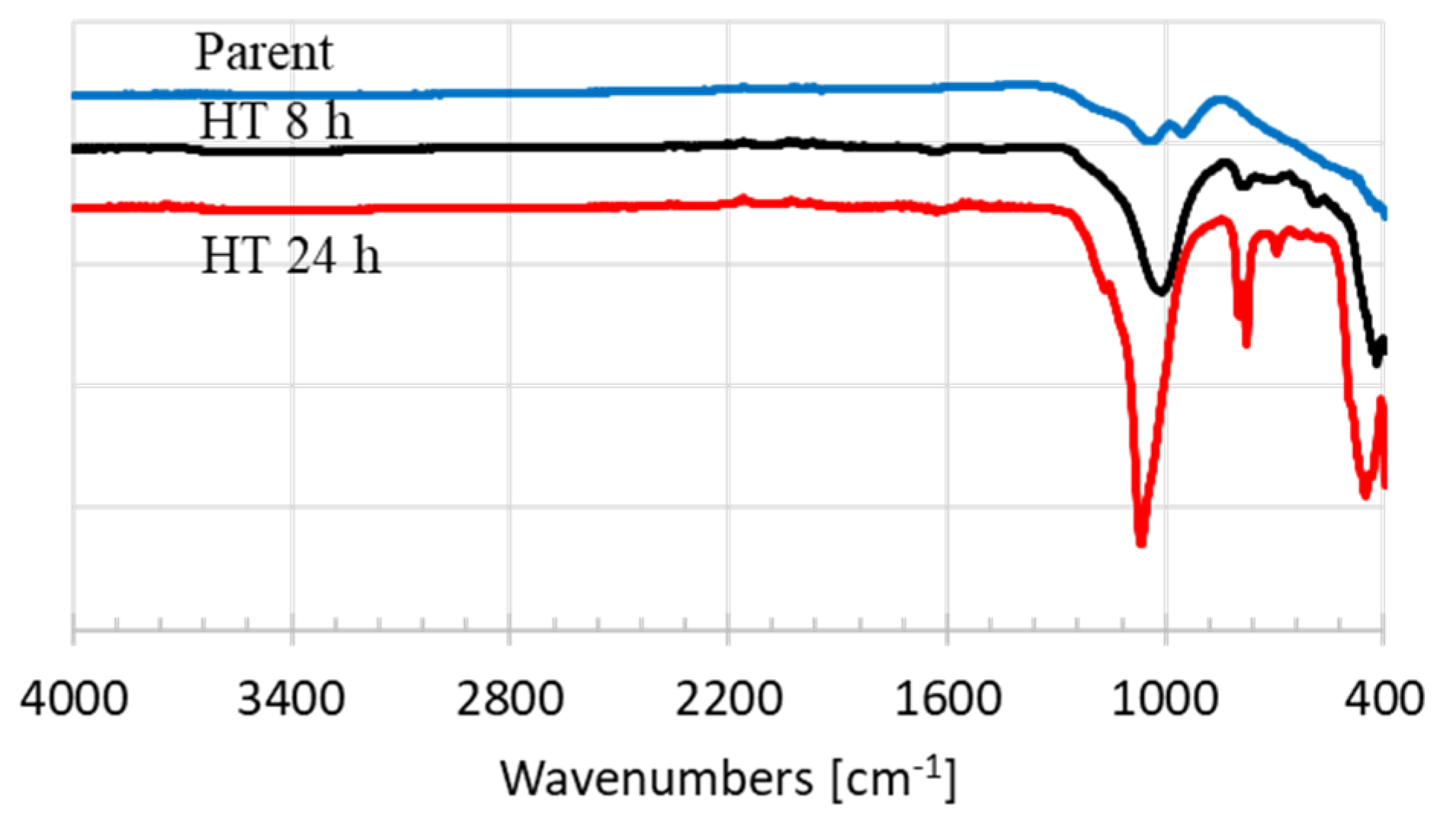
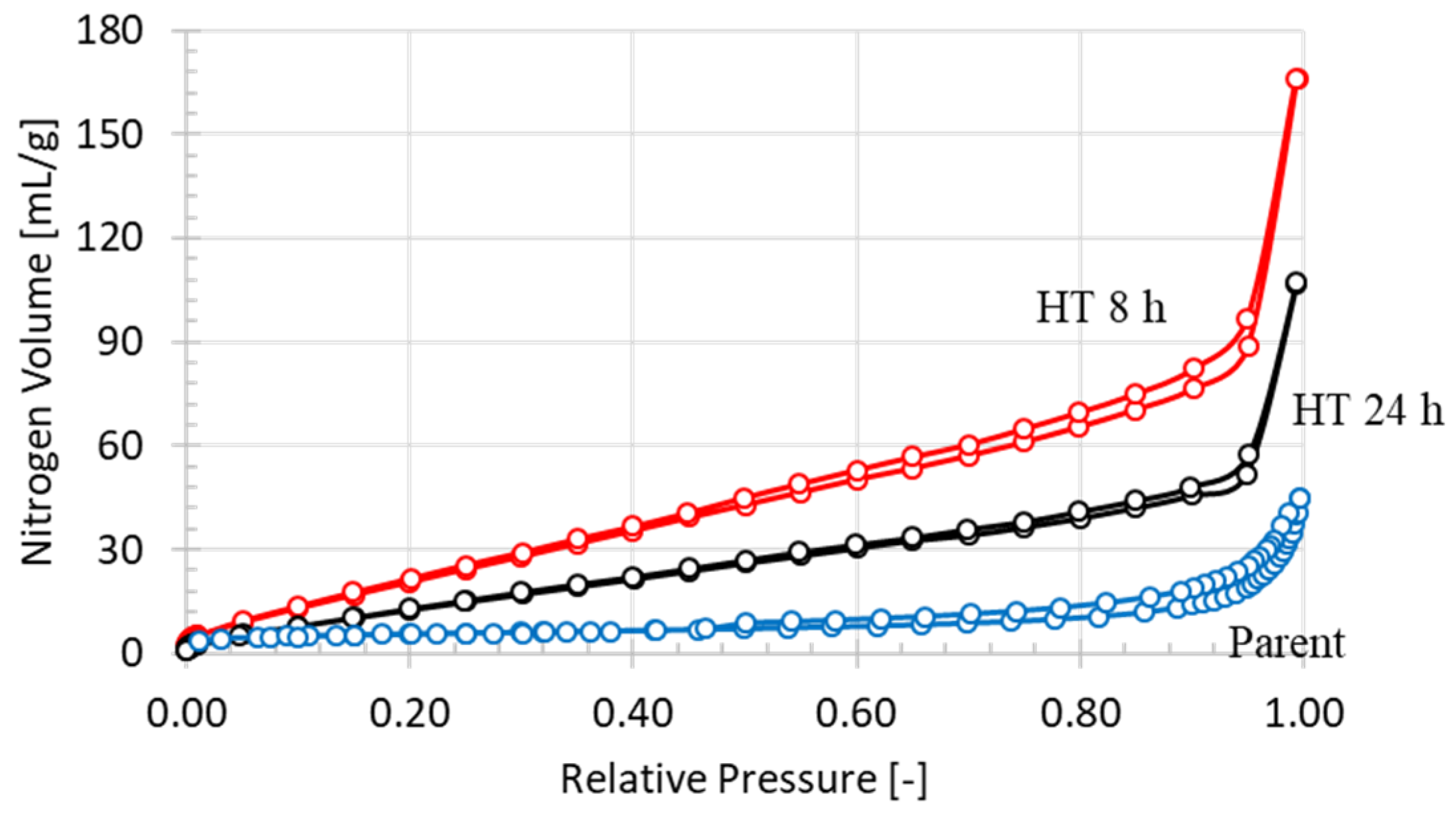

| Textural Properties | Parent | HT 8 h | HT 24 h |
|---|---|---|---|
| Surface area (m2/g) | 19 | 105 | 65 |
| Total pore volume (mL/g) | 0.06 | 0.23 | 0.15 |
| No | Origins of Zeolites | Phase | Treatment | Initial [NH4+] (mg/L) | % Ammonium Removal | Ref | |
|---|---|---|---|---|---|---|---|
| Before Modification | After Modification | ||||||
| 1 | Bayah, Indonesia | Mordenite, clinoptilolite, quartz | Hydrothermal | 100 | 48.9 | 62.6 | This work |
| 2 | Shimane Prefecture, Japan | Mordenite | Hydrothermal | 180 | 32 | 64 | [16] |
| 3 | Lingshou, China | Clinoptilolite-Ca, stilbite-Ca | NaNO3 impregnation followed by calcination | 5 | 41.8 | 81.7 | [21] |
| 4 | Kardzhali, Bulgaria | Clinoptilolite | Washing followed by drying | 100 | 95 | - | [22] |
| 5 | Semnan, Iran | Clinoptilolite | No modification | 100 | 87 | - | [23] |
| 6 | Ponorogo, Indonesia | Mordenite | NaOH | 10 | 30 | 50–80 | [11] |
| 7 | Synthetic | NaY | No Modification | 100 | 70 | - | [24] |
| Zeolite | Model | Parameter 1 | Parameter 2 | SSE |
|---|---|---|---|---|
| Parent | 1st order | kL = 0.83 | qe = 0.10 | 0.001660 |
| 2nd order | Ks = −0.28 | qe = 0.86 | 0.000139 | |
| Intraparticle | Ki = 0.016 | C = 0.67 | 0.000956 | |
| Elovich | A = 416.13 | β = 16.17 | 0.000367 | |
| HT 8 h | 1st order | kL = 0.88 | qe = 0.12 | 0.004379 |
| 2nd order | Ks = −0.387 | qe = 0.915 | 0.001608 | |
| Intraparticle | Ki = 0.014 | C = 0.75 | 0.000118 | |
| Elovich | A = 13,363 | β = 19.16 | 0.000433 |
| Isotherm | Parameter 1 | Parameter 2 | SSE |
|---|---|---|---|
| Langmuir | qmax = 353 | KL = 0.0005 | 0.29 |
| Freundlich | KF = 0.20 | n = 1.0235 | 0.28 |
| Temkin | B = 2.28 | Kt = 0.29 | 3.58 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kurniawan, T.; Bahri, S.; Diyanah, A.; Milenia, N.D.; Nuryoto, N.; Faungnawakij, K.; Thongratkaew, S.; Roil Bilad, M.; Huda, N. Improving Ammonium Sorption of Bayah Natural Zeolites by Hydrothermal Method. Processes 2020, 8, 1569. https://doi.org/10.3390/pr8121569
Kurniawan T, Bahri S, Diyanah A, Milenia ND, Nuryoto N, Faungnawakij K, Thongratkaew S, Roil Bilad M, Huda N. Improving Ammonium Sorption of Bayah Natural Zeolites by Hydrothermal Method. Processes. 2020; 8(12):1569. https://doi.org/10.3390/pr8121569
Chicago/Turabian StyleKurniawan, Teguh, Saiful Bahri, Anita Diyanah, Natasya D. Milenia, Nuryoto Nuryoto, Kajornsak Faungnawakij, Sutarat Thongratkaew, Muhammad Roil Bilad, and Nurul Huda. 2020. "Improving Ammonium Sorption of Bayah Natural Zeolites by Hydrothermal Method" Processes 8, no. 12: 1569. https://doi.org/10.3390/pr8121569
APA StyleKurniawan, T., Bahri, S., Diyanah, A., Milenia, N. D., Nuryoto, N., Faungnawakij, K., Thongratkaew, S., Roil Bilad, M., & Huda, N. (2020). Improving Ammonium Sorption of Bayah Natural Zeolites by Hydrothermal Method. Processes, 8(12), 1569. https://doi.org/10.3390/pr8121569







