Efficient Simulation of Chromatographic Processes Using the Conservation Element/Solution Element Method
Abstract
1. Introduction
2. Mathematical Models of Chromatographic Processes
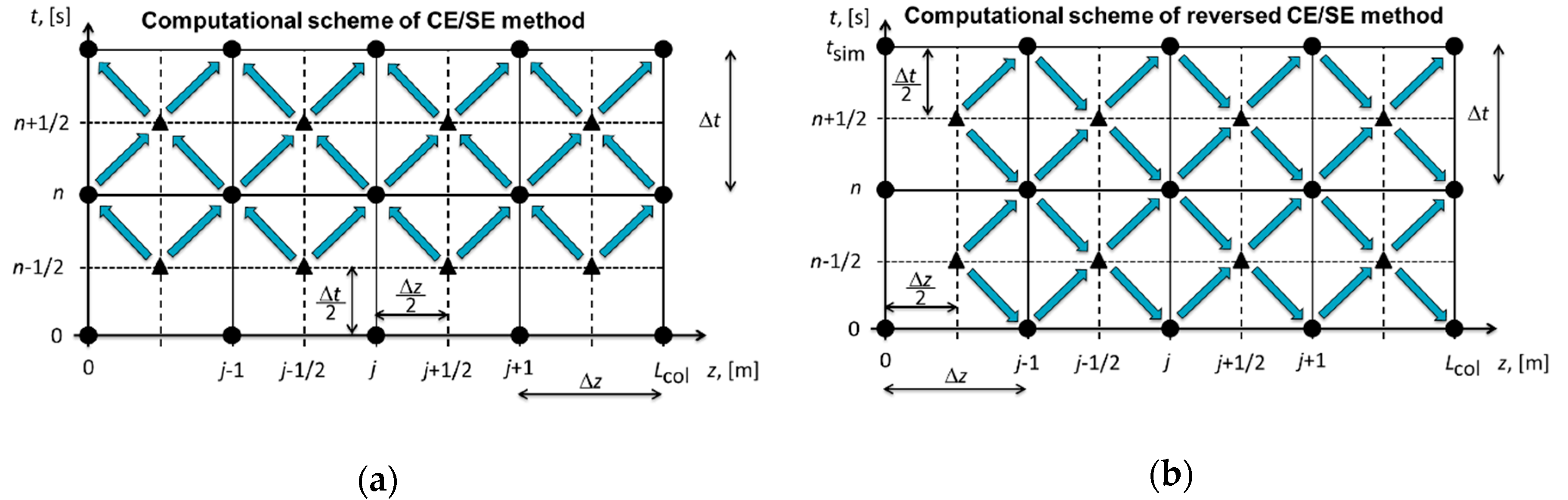
3. Conservation Element/Solution Element (CE/SE) Method
4. Results
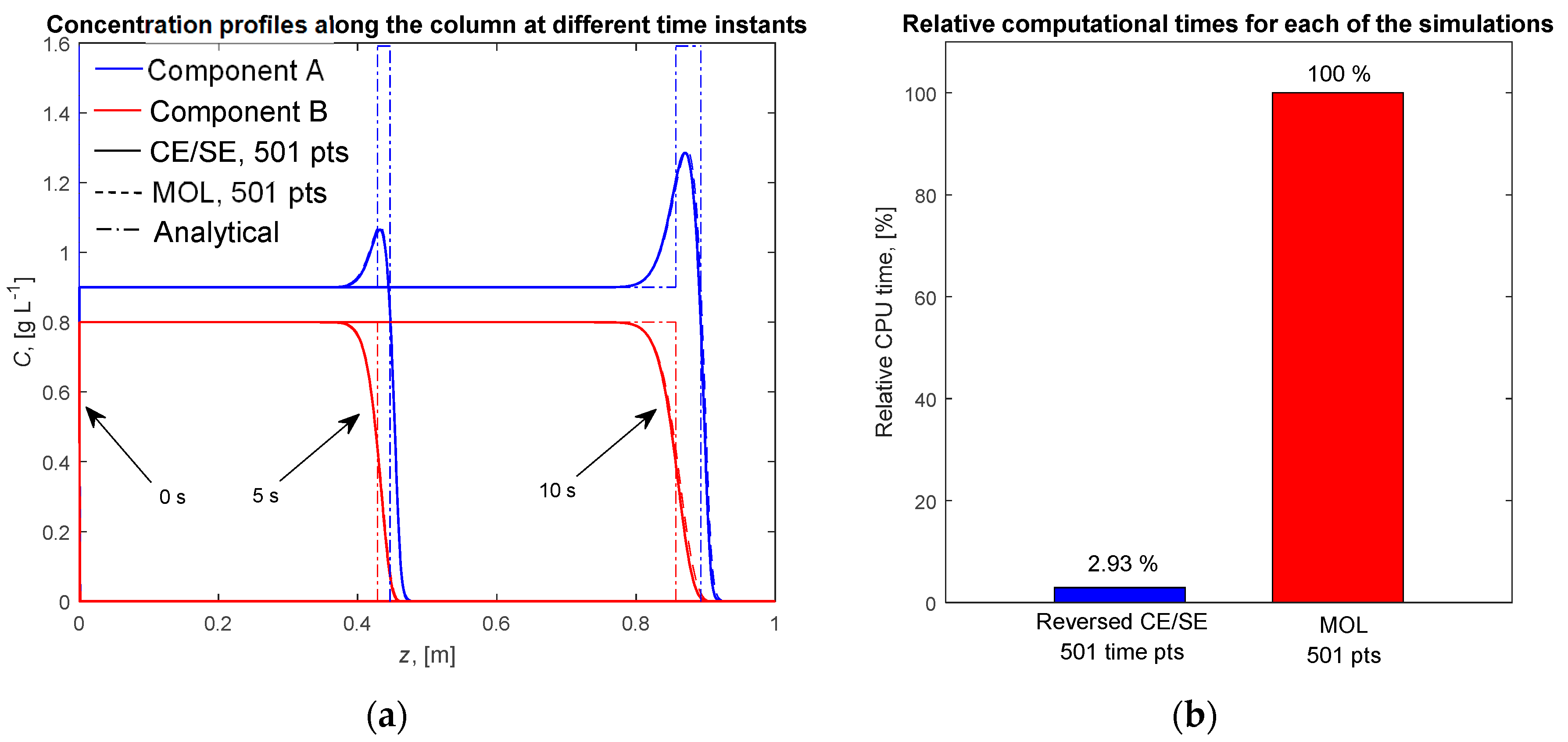
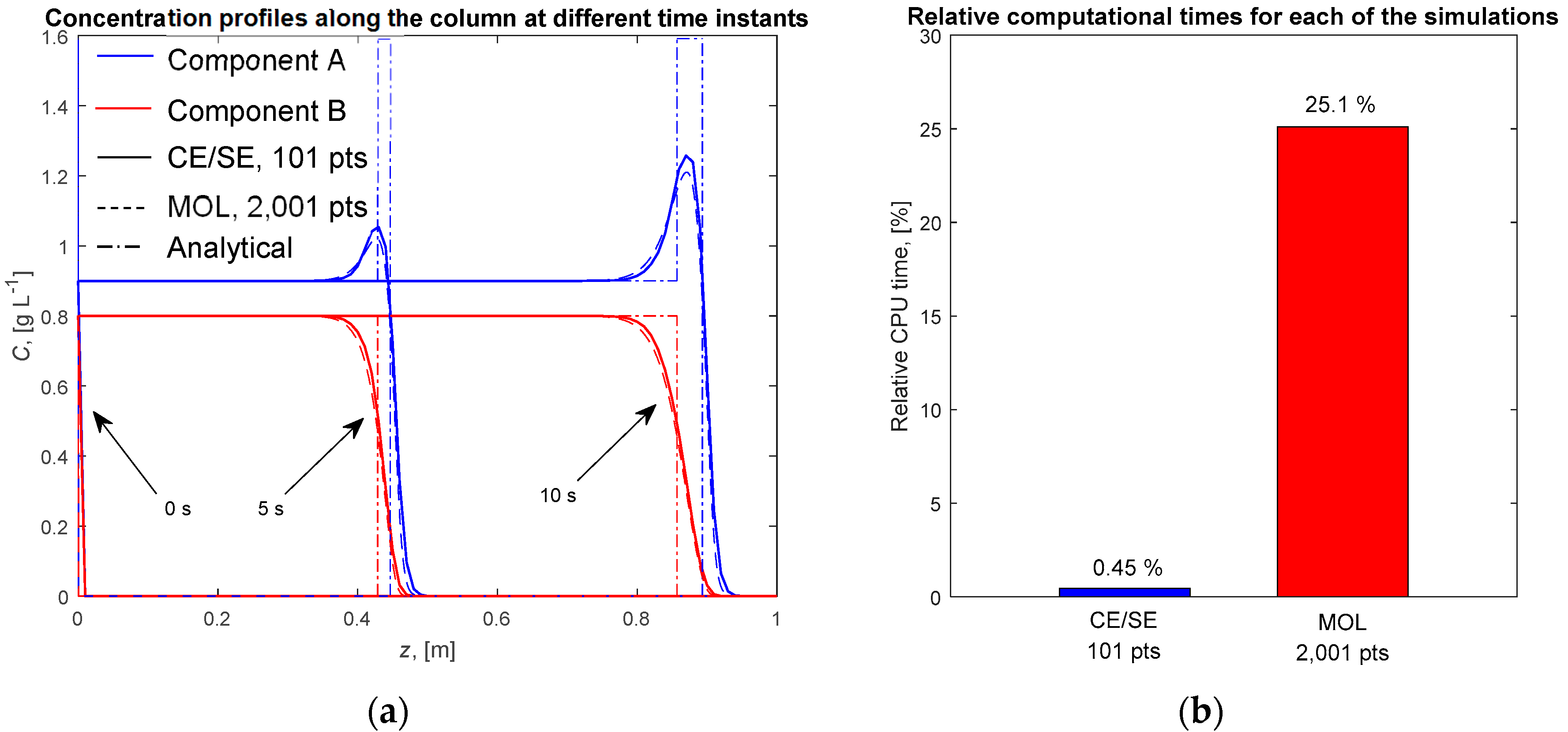
4.1. Single Column with the Ideal Equilibrium Model
- (i)
- Specify the simulation time tsim;
- (ii)
- Specify the number of time steps Nt, i.e., the number of conservation elements;
- (iii)
- Calculate the time step size ;
- (iv)
- Specify (;
- (v)
- Calculate the spatial step size from Equation (20);
- (vi)
- Continue with the reversed CE/SE method.
4.2. Single Column with the LDF Model
4.3. Binary SMB Process with the LDF Model
- − Desorbent node
- − Extract node
- − Feed node
- − Raffinate node
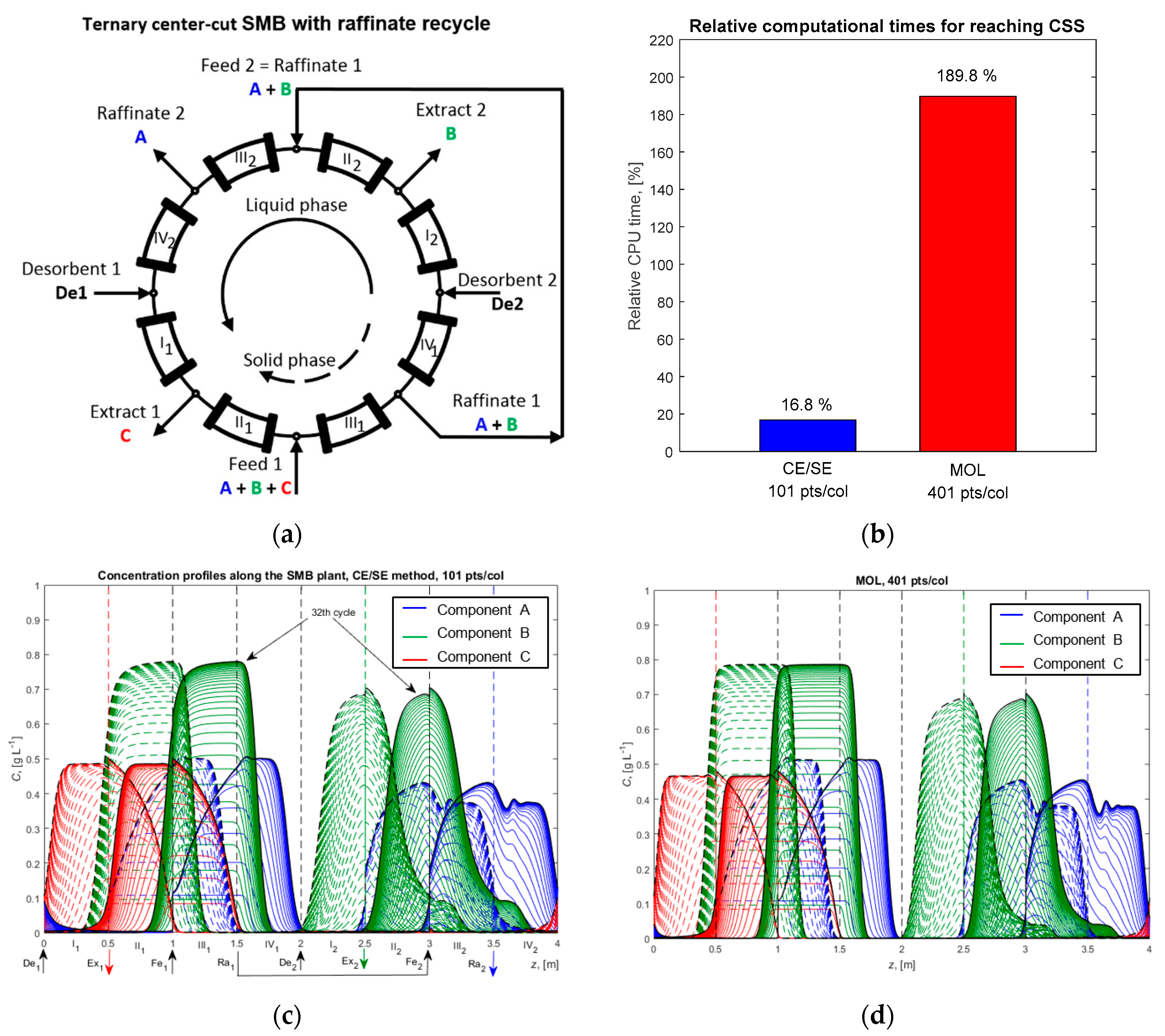
4.4. Ternary Center-Cut Eight-Zone SMB Process with Linear Isotherms and the Ideal Equilibrium Model
- − First desorbent node
- − First extract node
- − First feed node
- − First raffinate node
- − Second desorbent node
- − Second extract node
- − Second feed node
- − Second raffinate node
4.5. Ternary Center-Cut Eight-Zone SMB Process with Langmuir Isotherms and the LDF Model
5. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Nomenclature
| bi,k | retention factor in the Langmuir isotherm expression (L·g−1) |
| Ci,k | liquid phase concentration (g·L−1) |
| Courant–Friedrichs–Lewy number | |
| Courant–Friedrichs–Lewy number in the reversed CE/SE method | |
| Dax | axial dispersion coefficient (m2·s−1) |
| Dcol | column diameter (m) |
| Dpipe | pipe diameter (m) |
| f | vector of fluxes |
| Hi,k | adsorption Henry coefficient |
| mass transfer coefficient (s−1) | |
| Lcol | column length (m) |
| m | dimensionless flowrate ratio of liquid and solid phases |
| N | number of theoretical stages of the cell model |
| Ncol | number of columns in the SMB plant |
| Ncomp | number of components |
| Nt | number of time steps |
| Nz | number of spatial steps |
| p | vector of source terms |
| qi,k | solid phase concentration (g·L−1) |
| solid phase concentration at the interphase in equilibrium with the liquid phase (g·L−1) | |
| Q | volumetric flowrate (m3·s−1) |
| t | time coordinate (s) |
| tsim | simulation time (s) |
| tsw | switching time (s) |
| u | vector of state variables |
| Vcol | column volume (m3) |
| vk | liquid phase velocity (m·s−1) |
| z | spatial coordinate (m) |
| Δt | time step size (s) |
| Δz | spatial step size (m) |
| ε | column void fraction |
| τ | adjusted time |
| A, B, C | different components |
| De | desorbent stream |
| Ex | extract stream |
| Fe | feed stream |
| i, r | component indices |
| in | column inlet |
| int | internal flowrate |
| j | spatial coordinate index |
| k | column index |
| n | time coordinate index |
| out | column outlet |
| p | zone index |
| Ra | raffinate stream |
Appendix A. Direct Conversion of the Equilibrium Model to the Form Given by Equation (10)
References
- Schmidt-Traub, H.; Schulte, M.; Seidel-Morgenstern, A. Preparative Chromatography, 3rd ed.; Wiley-VCH Verlag: Weinheim, Germany, 2020. [Google Scholar]
- Rhee, H.-K.; Aris, R.; Amundson, N.R. First-Order Partial Differential Equations: Volume II—Theory and Application of Hyperbolic Systems of Quasilinear Equations; Prentice Hall: Englewood Cliffs, NJ, USA, 1989. [Google Scholar]
- Mazzotti, M.; Rajendran, A. Equilibrium theory-based analysis of nonlinear waves in separation processes. Ann. Rev. Chem. Biomol. Eng. 2013, 4, 119–141. [Google Scholar] [CrossRef] [PubMed]
- Mazzotti, M.; Storti, S.; Morbidelli, M. Optimal operation of simulated moving bed units for nonlinear chromatographic separations. J. Chromatogr. A 1997, 769, 3–24. [Google Scholar] [CrossRef]
- Migliorini, C.; Mazzotti, M.; Morbidelli, M. Continuous chromatographic separation through simulated moving beds under linear and nonlinear conditions. J. Chromatogr. A 1998, 827, 161–173. [Google Scholar] [CrossRef]
- Kaspereit, M.; Seidel-Morgenstern, A.; Kienle, A. Design of simulated moving bed processes under reduced purity requirements. J. Chromatogr. A 2007, 1162, 2–13. [Google Scholar] [CrossRef]
- Sainio, T.; Kaspereit, M. Analysis of steady state recycling chromatography using equilibrium theory. Sep. Purif. Technol. 2009, 6, 9–18. [Google Scholar] [CrossRef]
- Siitonen, J.; Sainio, T. Unified design of chromatographic separation processes. Chem. Eng. Sci. 2015, 122, 436–451. [Google Scholar] [CrossRef]
- Rhee, H.-K.; Aris, R.; Amundson, N.R. Shock layer in two solute chromatography: Effect of axial dispersion and mass transfer. Chem. Eng. Sci. 1974, 29, 2049–2060. [Google Scholar] [CrossRef]
- Mazzotti, M.; Storti, G.; Morbidelli, M. Shock layer analysis in multicomponent chromatography and countercurrent adsorption. Chem. Eng. Sci. 1994, 49, 1337–1355. [Google Scholar] [CrossRef]
- Marquardt, W. Traveling waves in chemical processes. Int. Chem. Eng. 1990, 30, 585–606. [Google Scholar]
- Lim, Y.I.; Chang, S.-C.; Jørgensen, S.B. A novel partial differential algebraic equation (PDAE) solver: Iterative space-time conservation element/solution element (CE/SE) method. Comput. Chem. Eng. 2004, 28, 1309–1324. [Google Scholar] [CrossRef]
- Von Lieres, E.; Andersson, J. A fast and accurate solver for the general rate model of column liquid chromatography. Comp. Chem. Eng. 2010, 34, 1180–1191. [Google Scholar] [CrossRef]
- Javeed, S.; Qamar, S.; Seidel-Morgenstern, A.; Warnecke, G. Efficient and accurate numerical simulation of nonlinear chromatographic processes. Comput. Chem. Eng. 2011, 35, 2294–2305. [Google Scholar] [CrossRef]
- Vande Wouwer, A.; Saucez, P.; Vilas, C. Simulation of ODE/PDE Models with MATLAB, OCTAVE and SCILAB. Scientific and Engineering Applications; Springer International Publishing Switzerland: Cham, Switzerland, 2014; pp. 125–202. [Google Scholar]
- Köhler, R.; Mohl, K.D.; Schramm, H.; Zeitz, M.; Kienle, A.; Mangold, M.; Stein, E.; Gilles, E.-D. Methods of lines within the simulation environment DIVA© for chemical processes. In Adaptive Method of Lines; Vande Wouwer, A., Saucez, P., Schiesser, W.E., Eds.; CRC Press: New York, NY, USA, 2001; pp. 371–406. [Google Scholar]
- Chang, S.-C. New Developments in the Method of Space-Time Conservation Element and Solution Element: Applications to the Euler and Navier-Stokes Equations; NASA TM 106226; The SAO/NASA Astrophysics Data System: Cleveland, OH, USA, 1993. [Google Scholar]
- Chang, S.-C. The method of space-time conservation element and solution element—A new approach for solving the Navier–Stokes and Euler equations. J. Comp. Phys. 1995, 119, 295–324. [Google Scholar] [CrossRef]
- Chang, S.-C. Courant number insensitive CE/SE schemes. In Proceedings of the 38th AIAA Joint Propulsion Conference, Indianapolis, Indiana, 7–10 July 2002; AIAA-2002-3890. AIAA: Indianapolis, IN, USA, 2012. [Google Scholar]
- Lim, Y.I.; Jørgensen, S.B. A fast and accurate numerical method for solving simulated moving bed (SMB) chromatographic separation problems. Chem. Eng. Sci. 2004, 59, 1931–1947. [Google Scholar] [CrossRef]
- Yao, C.; Tang, S.; Lu, Y.; Yao, H.-M.; Tade, M.O. Combination of space-time conservation element/solution element method and continuous prediction technique for accelerated simulation of simulated moving bed chromatography. Chem. Eng. Process 2015, 96, 54–61. [Google Scholar] [CrossRef]
- Keßler, L.C.; Seidel-Morgenstern, A. Theoretical study of multicomponent continuous countercurrent chromatography based on connected 4-zone units. J. Chromatogr. A 2006, 1126, 323–337. [Google Scholar] [CrossRef]
- Da Silva, F.V.S.; Seidel-Morgenstern, A. Evaluation of center-cut separations applying simulated moving bed chromatography with 8 zones. J. Chromatogr. A 2016, 1456, 123–136. [Google Scholar] [CrossRef]
- Kiwala, D.; Mendrella, J.; Antos, D.; Seidel-Morgenstern, A. Center-cut separation of intermediately. adsorbing target component by 8-zone simulated moving bed chromatography with internal recycle. J. Chromatogr. A 2016, 1453, 19–33. [Google Scholar] [CrossRef]
- MATLAB R2017a (Version 9.2.0); The MathWorks Inc.: Natick, MA, USA, 2017.
- Guiochon, G.; Lin, B. Modeling for Preparative Chromatography, 1st ed.; Academic Press: San Diego, CA, USA, 2003. [Google Scholar]
- Rhee, H.-K.; Aris, R.; Amundson, N.R. First-Order Partial Differential Equations: Volume I—Theory and Application of Single Equations; Prentice Hall: Englewood Cliffs, NJ, USA, 1986. [Google Scholar]
- Agrawal, G.; Kawajiri, Y. Comparison of various ternary simulated moving bed separation schemes by multi-objective optimization. J. Chromatogr. A 2012, 1238, 105–113. [Google Scholar] [CrossRef]
- Pishkari, R.; Kienle, A. Fast and accurate simulation of simulated moving bed chromatographic processes with linear adsorption isotherms. Comp. Aided Chem. Eng. 2020, 48, 487–492. [Google Scholar]
- Nicolaos, A.; Muhr, L.; Gotteland, P.; Nicoud, R.M.; Bailly, M. Application of equilibrium theory to ternary moving bed configurations (4 + 4, 5 + 4, 8 and 9 zones) II. Langmuir case. J. Chromatogr. A 2001, 908, 87–109. [Google Scholar] [CrossRef]
- Suvarov, P.; Vande Wouwer, A.; Kienle, A.; Nobre, C.; De Weireld, G. Cycle to cycle adaptive control of simulated moving bed chromatographic separation processes. J. Proc. Control 2014, 24, 357–367. [Google Scholar] [CrossRef]
- Suvarov, P.; Vande Wouwer, A.; Lee, J.-W.; Seidel-Morgenstern, A.; Kienle, A. Control of incomplete separation in simulated moving bed chromatographic processes. In Proceedings of the 11th IFAC Symposium on Dynamics and Control of Process Systems, including Biosystems, Trondheim, Norway, 6–8 June 2016. [Google Scholar]
- Suvarov, P.; Lee, J.-W.; Vande Wouwer, A.; Seidel-Morgenstern, A.; Kienle, A. Online estimation of optimal operating conditions for simulated moving bed chromatographic processes. J. Chrom. A 2019, 1602, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Helfferich, F.G.; Klein, G. Multicomponent Chromatography: Theory of Interference; Marcel Dekker: New York, NY, USA, 1970. [Google Scholar]

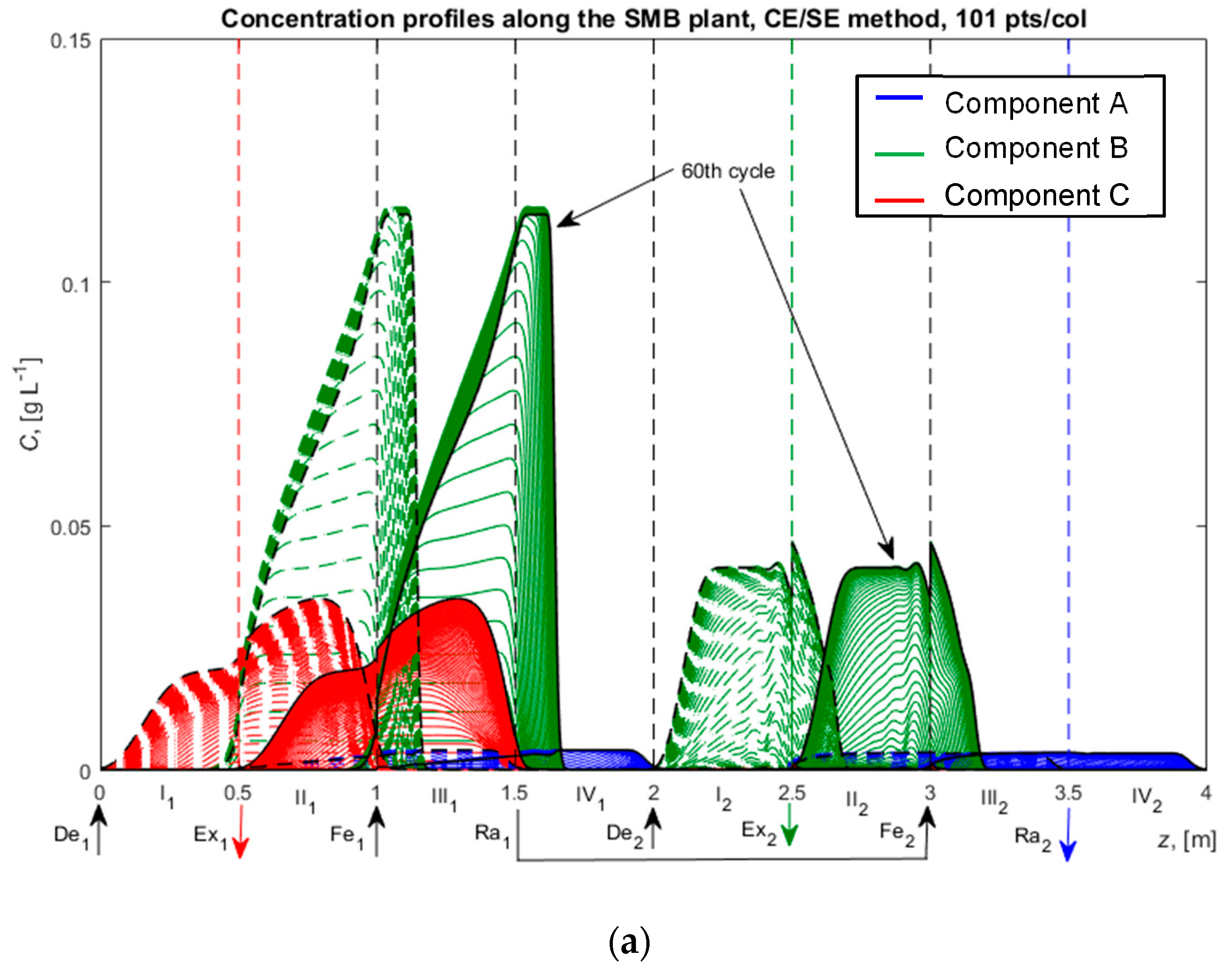
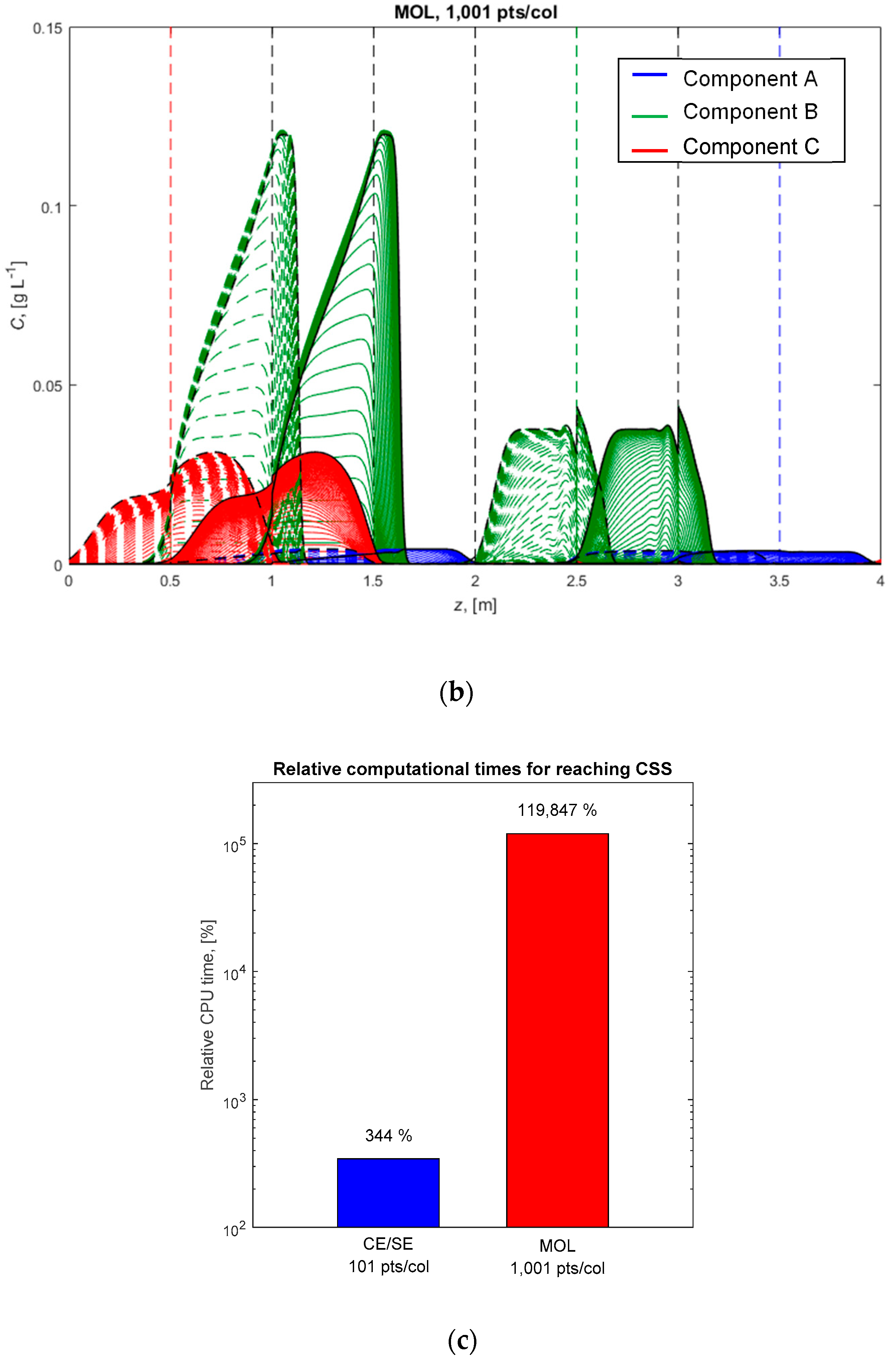
| Quantity | Value | Quantity | Value | Quantity | Value |
|---|---|---|---|---|---|
| Lcol (m) | 1.0 | HA | 2 | HB | 4 |
| v (m·s−1) | 0.1 | bA (L·g−1) | 2 | bB (L·g−1) | 4 |
| ε | 0.8 | CFe,A (g·L−1) | 0.9 | CFe,B (g·L−1) | 0.8 |
| tsim (s) | 10 | Nt | 501 | 0.4 |
| Quantity | Value | Quantity | Value |
|---|---|---|---|
| km,A (s−1) | 10 | Nz | 101 |
| km,B (s−1) | 10 | CFL | 0.4 |
| Quantity | Value | Quantity | Value | Quantity | Value | Quantity | Value |
|---|---|---|---|---|---|---|---|
| Lcol (m) | 0.5 | mI | 5 | HA | 2 | HB | 4 |
| Dcol (m) | 0.02 | mII | 1.8 | bA (L·g−1) | 0.2 | bB (L·g−1) | 0.4 |
| ε | 0.8 | mIII | 2.8 | km,A (s−1) | 10 | km,B (s−1) | 10 |
| Dpipe (m) | 0.002 | mIV | 1.3 | CFe,A (g·L−1) | 0.9 | CFe,B (g·L−1) | 0.7 |
| tsw (s) | 40 |
| Quantity | Value | Quantity | Value | Quantity | Value | Quantity | Value | Quantity | Value |
|---|---|---|---|---|---|---|---|---|---|
| Lcol (m) | 0.5 | mI,1 | 2.55 | mI,2 | 1.82 | HA | 1.1 | CFe,A (g·L−1) | 0.9 |
| Dcol (m) | 0.02 | mII,1 | 1.57 | mII,2 | 1.22 | HB | 1.7 | CFe,B (g·L−1) | 0.8 |
| ε | 0.75 | mIII,1 | 2.19 | mIII,2 | 2.55 | HC | 2.5 | CFe,C (g·L−1) | 0.7 |
| Dpipe (m) | 0.002 | mIV,1 | 0.86 | mIV,2 | 1.01 | tsw (s) | 60 |
| Quantity | Value | Quantity | Value | Quantity | Value | Quantity | Value | Quantity | Value |
|---|---|---|---|---|---|---|---|---|---|
| mI,1 | 2.55 | mI,2 | 2.10 | HA | 1 | HB | 2 | HC | 2.5 |
| mII,1 | 1.893 | mII,2 | 0.928 | bA (L·g−1) | 1 | bB (L·g−1) | 2 | bC (L·g−1) | 2.5 |
| mIII,1 | 1.90 | mIII,2 | 1.99 | km,A (s−1) | 10 | km,B (s−1) | 10 | km,C (s−1) | 10 |
| mIV,1 | 0.915 | mIV,2 | 0.85 | CFe,A (g·L−1) | 0.5 | CFe,B (g·L−1) | 5.0 | CFe,C (g·L−1) | 1.5 |
| tsw (s) | 40 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chernev, V.P.; Vande Wouwer, A.; Kienle, A. Efficient Simulation of Chromatographic Processes Using the Conservation Element/Solution Element Method. Processes 2020, 8, 1316. https://doi.org/10.3390/pr8101316
Chernev VP, Vande Wouwer A, Kienle A. Efficient Simulation of Chromatographic Processes Using the Conservation Element/Solution Element Method. Processes. 2020; 8(10):1316. https://doi.org/10.3390/pr8101316
Chicago/Turabian StyleChernev, Valentin Plamenov, Alain Vande Wouwer, and Achim Kienle. 2020. "Efficient Simulation of Chromatographic Processes Using the Conservation Element/Solution Element Method" Processes 8, no. 10: 1316. https://doi.org/10.3390/pr8101316
APA StyleChernev, V. P., Vande Wouwer, A., & Kienle, A. (2020). Efficient Simulation of Chromatographic Processes Using the Conservation Element/Solution Element Method. Processes, 8(10), 1316. https://doi.org/10.3390/pr8101316







