Thermal Analysis of Nigerian Oil Palm Biomass with Sachet-Water Plastic Wastes for Sustainable Production of Biofuel
Abstract
1. Introduction
2. Materials and Methods
2.1. Biomass Samples
2.2. Elemental and Proximate Analysis
2.3. Thermal Analysis Using Thermogravimetric Analysis (TGA) and Differential Scanning Calorimetry (DSC)
2.4. Kinetic Reaction
3. Results and Discussion
3.1. Characteristic properties of OPEFB and SWP
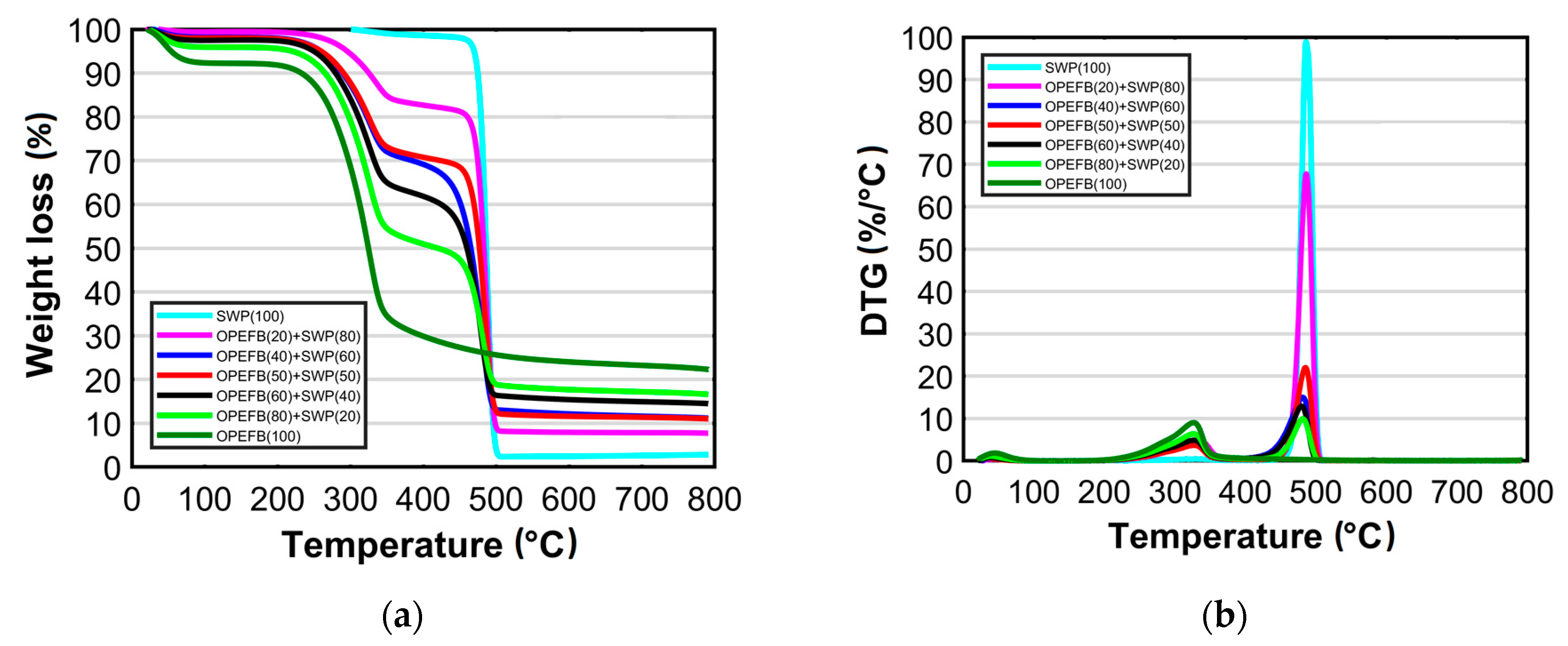
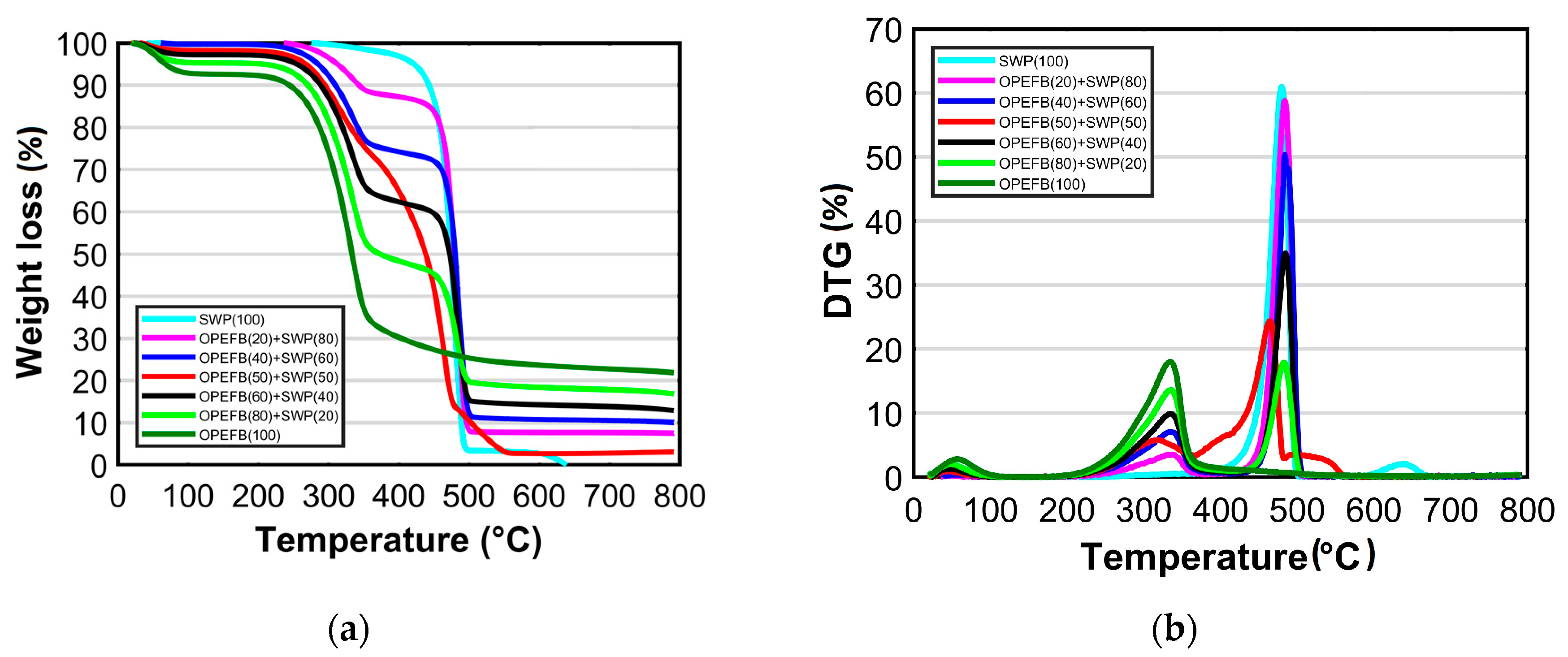
3.2. Thermal Characteristics
3.3. TGA Kinetic Analysis
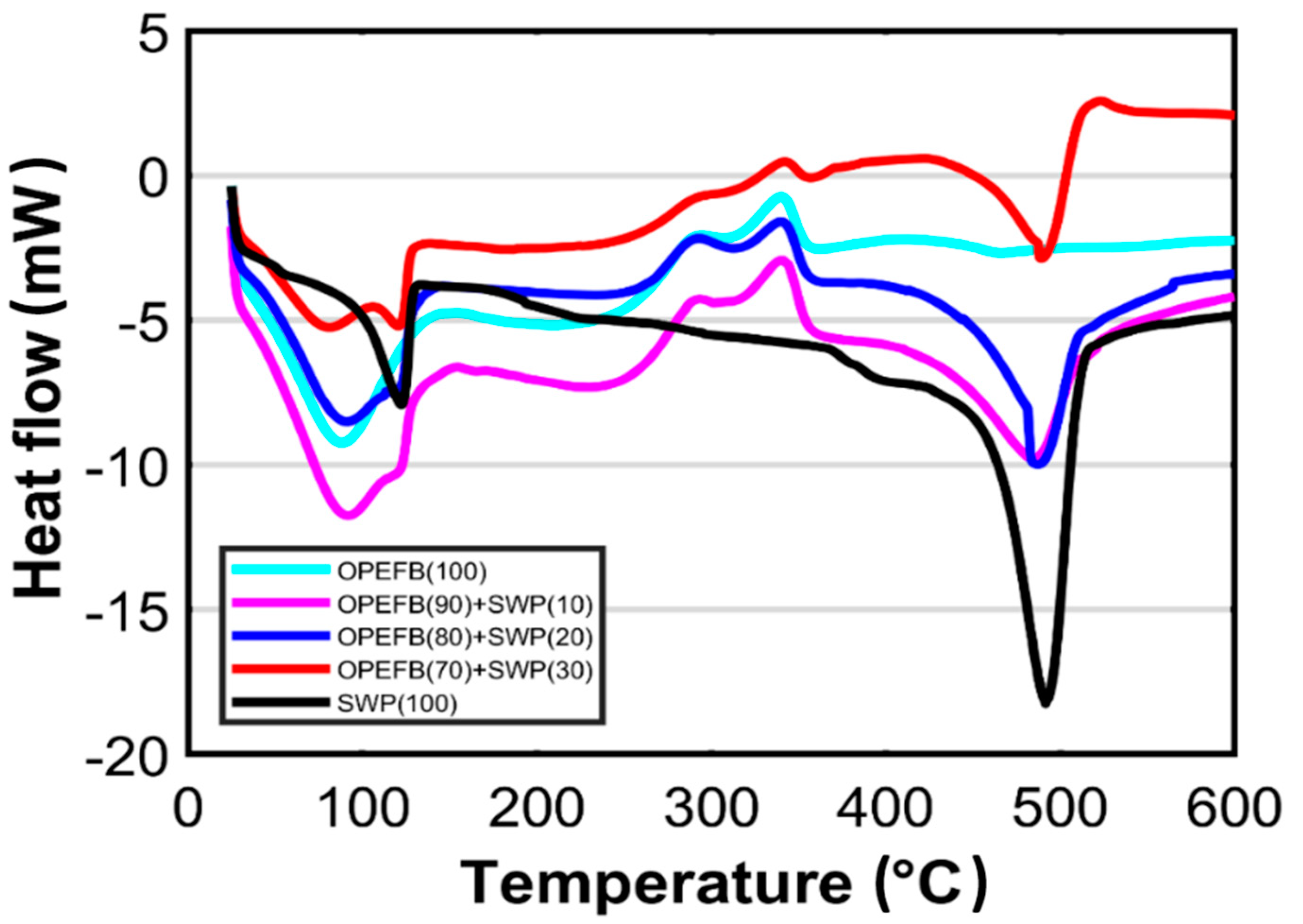
3.4. DSC Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Norhasyima, R.S.; Mahlia, T.M.I. Advances in CO2 utilization technology: A patent landscape review. J. CO2 Util. 2018, 26, 323–335. [Google Scholar] [CrossRef]
- IEA. Excerpt from CO2 Emissions from Fuel Combustion; IEA: Paris, France, 2015. [Google Scholar]
- Power Africa. Nigeria Power Africa Fact Sheet; USAID: Washington, DC, USA, 2019.
- Aliyu, A.S.; Ramli, A.T.; Saleh, M.A. Nigeria electricity crisis: Power generation capacity expansion and environmental ramifications. Energy 2013, 61, 354–367. [Google Scholar] [CrossRef]
- Salman, B.; Neshaeimoghaddam, H. An Evaluation of The Nigeria Electricity Sector Post Privatisation. J. Energy Environ. 2017, 9, 33–37. [Google Scholar]
- Financie, R.; Moniruzzaman, M.; Uemura, Y. Enhanced enzymatic delignification of oil palm biomass with ionic liquid pretreatment. Biochem. Eng. J. 2016, 110, 1–7. [Google Scholar] [CrossRef]
- Bustos, G.; Arcos, U.; Vecino, X.; Cruz, J.M.; Moldes, A.B. Recycled Lactobacillus pentosus biomass can regenerate biosurfactants after various fermentative and extractive cycles. Biochem. Eng. J. 2018, 132, 191–195. [Google Scholar] [CrossRef]
- Martínez-Patiño, J.C.; Ruiz, E.; Cara, C.; Romero, I.; Castro, E. Advanced bioethanol production from olive tree biomass using different bioconversion schemes. Biochem. Eng. J. 2018, 137, 172–181. [Google Scholar] [CrossRef]
- Uddin, M.N.; Techato, K.; Taweekun, J.; Mofijur, M.; Rasul, M.G.; Mahlia, T.M.I.; Ashrafur, S.M. An Overview of Recent Developments in Biomass Pyrolysis Technologies. Energies 2018, 11, 3115. [Google Scholar] [CrossRef]
- Rocha, E.P.A.; Sermyagina, E.; Vakkilainen, E.; Colodette, J.L.; de Oliveira, I.M.; Cardoso, M. Kinetics of pyrolysis of some biomasses widely available in Brazil. J. Therm. Anal. Calorim. 2017, 130, 1445–1454. [Google Scholar] [CrossRef]
- Ben-Iwo, J.; Manovic, V.; Longhurst, P. Biomass resources and biofuels potential for the production of transportation fuels in Nigeria. Renew. Sustain. Energy Rev. 2016, 63, 172–192. [Google Scholar] [CrossRef]
- Stoler, J. From curiosity to commodity: A review of the evolution of sachet drinking water in West Africa. Wiley Interdiscip. Rev. Water 2017, 4, e1206. [Google Scholar] [CrossRef]
- De Caprariis, B.; Bavasso, I.; Bracciale, M.P.; Damizia, M.; De Filippis, P.; Scarsella, M. Enhanced bio-crude yield and quality by reductive hydrothermal liquefaction of oak wood biomass: Effect of iron addition. J. Anal. Appl. Pyrolysis 2019, 139, 123–130. [Google Scholar] [CrossRef]
- Chianese, S.; Fail, S.; Binder, M.; Rauch, R.; Hofbauer, H.; Molino, A.; Blasi, A.; Musmarra, D. Experimental investigations of hydrogen production from CO catalytic conversion of tar rich syngas by biomass gasification. Catal. Today 2016, 277, 182–191. [Google Scholar] [CrossRef]
- Nomanbhay, S.; Salman, B.; Hussain, R.; Ong, M.Y. Microwave pyrolysis of lignocellulosic biomass—A contribution to power Africa. Energy Sustain. Soc. 2017, 7, 23. [Google Scholar] [CrossRef]
- Abnisa, F.; Wan Daud, W.M.A. A review on co-pyrolysis of biomass: An optional technique to obtain a high-grade pyrolysis oil. Energy Convers. Manag. 2014, 87, 71–85. [Google Scholar] [CrossRef]
- Xiong, S.; Zhuo, J.; Zhou, H.; Pang, R.; Yao, Q. Study on the co-pyrolysis of high density polyethylene and potato blends using thermogravimetric analyzer and tubular furnace. J. Anal. Appl. Pyrolysis 2015, 112, 66–73. [Google Scholar] [CrossRef]
- Chen, L.; Wang, S.; Meng, H.; Wu, Z.; Zhao, J. Synergistic effect on thermal behavior and char morphology analysis during co-pyrolysis of paulownia wood blended with different plastics waste. Appl. Therm. Eng. 2017, 111, 834–846. [Google Scholar] [CrossRef]
- Xu, Z.-X.; Zhang, C.-X.; He, Z.-X.; Wang, Q. Pyrolysis Characteristic and kinetics of Polyvinylidene fluoride with and without Pine Sawdust. J. Anal. Appl. Pyrolysis 2017, 123, 402–408. [Google Scholar] [CrossRef]
- Shen, J.; Igathinathane, C.; Yu, M.; Pothula, A.K. Biomass pyrolysis and combustion integral and differential reaction heats with temperatures using thermogravimetric analysis/differential scanning calorimetry. Bioresour. Technol. 2015, 185, 89–98. [Google Scholar] [CrossRef]
- Milivojević, A.; Ćorović, M.; Carević, M.; Banjanac, K.; Vujisić, L.; Veličković, D.; Bezbradica, D. Highly efficient enzymatic acetylation of flavonoids: Development of solvent-free process and kinetic evaluation. Biochem. Eng. J. 2017, 128, 106–115. [Google Scholar] [CrossRef]
- Costa, R.S.; Vinga, S. Control analysis of the impact of allosteric regulation mechanism in a Escherichia coli kinetic model: Application to serine production. Biochem. Eng. J. 2016, 110, 59–70. [Google Scholar] [CrossRef]
- Martínez, I.; Santos, V.E.; Garcìa-Ochoa, F. Metabolic kinetic model for dibenzothiophene desulfurization through 4S pathway using intracellular compound concentrations. Biochem. Eng. J. 2017, 117, 89–96. [Google Scholar] [CrossRef]
- Coats, A.W.; Redfern, J.P. Kinetic Parameters from Thermogravimetric Data. Nature 1964, 201, 68–69. [Google Scholar] [CrossRef]
- Mishra, R.K.; Mohanty, K. Pyrolysis kinetics and thermal behavior of waste sawdust biomass using thermogravimetric analysis. Bioresour. Technol. 2018, 251, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Samaržija-Jovanović, S.; Jovanović, V.; Marković, G.; Marinović-Cincović, M.; Budinski-Simendić, J.; Janković, B. Ethylene–Propylene–Diene Rubber-Based Nanoblends: Preparation, Characterization and Applications. In Rubber Nano Blends; Springer: Cham, Switzerland, 2017; pp. 281–349. [Google Scholar]
- Oyedun, A.O.; Tee, C.Z.; Hanson, S.; Hui, C.W. Thermogravimetric analysis of the pyrolysis characteristics and kinetics of plastics and biomass blends. Fuel Process. Technol. 2014, 128, 471–481. [Google Scholar] [CrossRef]
- Abdullah, N.; Gerhauser, H. Bio-oil derived from empty fruit bunches. Fuel 2008, 87, 2606–2613. [Google Scholar] [CrossRef]
- Sulaiman, F.; Abdullah, N. Optimum conditions for maximising pyrolysis liquids of oil palm empty fruit bunches. Energy 2011, 36, 2352–2359. [Google Scholar] [CrossRef]
- Yang, J.; Rizkiana, J.; Widayatno, W.B.; Karnjanakom, S.; Kaewpanha, M.; Hao, X.; Abudula, A.; Guan, G. Fast co-pyrolysis of low density polyethylene and biomass residue for oil production. Energy Convers. Manag. 2016, 120, 422–429. [Google Scholar] [CrossRef]
- Lawson, O.E.; Lawson, E.O. Physico-Chemical Parameters and Heavy Metal Contents of Water from the Mangrove Swamps of Lagos Lagoon, Lagos, Nigeria. Adv. Biol. Res. 2011, 5, 8–21. [Google Scholar]
- Han, B.; Chen, Y.; Wu, Y.; Hua, D.; Chen, Z.; Feng, W.; Yang, M.; Xie, Q. Co-pyrolysis behaviors and kinetics of plastics–biomass blends through thermogravimetric analysis. J. Therm. Anal. Calorim. 2014, 115, 227–235. [Google Scholar] [CrossRef]
- Uzun, B.B.; Yaman, E. Pyrolysis kinetics of walnut shell and waste polyolefins using thermogravimetric analysis. J. Energy Inst. 2017, 90, 825–837. [Google Scholar] [CrossRef]
- Bartoli, M.; Rosi, L.; Frediani, M.; Undri, A.; Frediani, P. Depolymerization of polystyrene at reduced pressure through a microwave assisted pyrolysis. J. Anal. Appl. Pyrolysis 2015, 113, 281–287. [Google Scholar] [CrossRef]
- Chang, S.H. An overview of empty fruit bunch from oil palm as feedstock for bio-oil production. Biomass Bioenergy 2014, 62, 174–181. [Google Scholar] [CrossRef]
- Ro, D.; Kim, Y.-M.; Lee, I.-G.; Jae, J.; Jung, S.-C.; Kim, S.C.; Park, Y.-K. Bench scale catalytic fast pyrolysis of empty fruit bunches over low cost catalysts and HZSM-5 using a fixed bed reactor. J. Clean. Prod. 2018, 176, 298–303. [Google Scholar] [CrossRef]
- Chandrasekaran, A.; Ramachandran, S.; Subbiah, S. Determination of kinetic parameters in the pyrolysis operation and thermal behavior of Prosopis juliflora using thermogravimetric analysis. Bioresour. Technol. 2017, 233, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Jin, Q.; Wang, X.; Li, S.; Mikulčić, H.; Bešenić, T.; Deng, S.; Vujanović, M.; Tan, H.; Kumfer, B.M. Synergistic effects during co-pyrolysis of biomass and plastic: Gas, tar, soot, char products and thermogravimetric study. J. Energy Inst. 2019, 92, 108–117. [Google Scholar] [CrossRef]
- Kai, X.; Li, R.; Yang, T.; Shen, S.; Ji, Q.; Zhang, T. Study on the co-pyrolysis of rice straw and high density polyethylene blends using TG-FTIR-MS. Energy Convers. Manag. 2017, 146, 20–33. [Google Scholar] [CrossRef]
- Chong, Y.Y.; Thangalazhy-Gopakumar, S.; Gan, S.; Ng, H.K.; Lee, L.Y.; Adhikari, S. Kinetics and Mechanisms for Copyrolysis of Palm Empty Fruit Bunch Fiber (EFBF) with Palm Oil Mill Effluent (POME) Sludge. Energy Fuels 2017, 31, 8217–8227. [Google Scholar] [CrossRef]
- Fang, T.W.; Asyikin, N.; Syasya, N.S.; Shawkataly, A.K.H.; Kassim, M.H.M.; Syakir, M.I. Water Absorption and Thickness Swelling of Oil Palm Empty Fruit Bunch (OPEFB) and Seaweed Composite for Soil Erosion Mitigation. J. Phys. Sci. 2017, 28, 1–17. [Google Scholar] [CrossRef]
- Khalid, M.; Ratnam, C.T.; Luqman, C.A.; Salmiaton, A.; Choong, T.S.Y.; Jalaludin, H. Thermal and Dynamic Mechanical Behavior of Cellulose- and Oil Palm Empty Fruit Bunch (OPEFB)-Filled Polypropylene Biocomposites. Polym.-Plast. Technol. Eng. 2009, 48, 1244–1251. [Google Scholar] [CrossRef]
- Banat, R.; Fares, M.M. Thermo-Gravimetric Stability of High Density Polyethylene Composite Filled with Olive Shell Flour. Am. J. Ploym. Sci. 2015, 5, 65–74. [Google Scholar]
- Yahiaoui, M.; Hadoun, H.; Toumert, I.; Hassani, A. Determination of kinetic parameters of Phlomis bovei de Noé using thermogravimetric analysis. Bioresour. Technol. 2015, 196, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ma, D.; Jin, Q.; Deng, S.; Stančin, H.; Tan, H.; Mikulčić, H. Synergistic effects of biomass and polyurethane co-pyrolysis on the yield, reactivity, and heating value of biochar at high temperatures. Fuel Process. Technol. 2019, 194, 106127. [Google Scholar] [CrossRef]
- Romero Millán, L.M.; Sierra Vargas, F.E.; Nzihou, A. Kinetic Analysis of Tropical Lignocellulosic Agrowaste Pyrolysis. BioEnergy Res. 2017, 10, 832–845. [Google Scholar] [CrossRef]
- Nyakuma, B.B. Kinetic analysis of oil palm empty fruit bunch (OPEFB) pellets as feedstock for pyrolysis. PeerJ PrePrints 2015, 3, e1150v1. [Google Scholar]
- Wang, S.; Dai, G.; Yang, H.; Luo, Z. Lignocellulosic biomass pyrolysis mechanism: A state-of-the-art review. Prog. Energy Combust. Sci. 2017, 62, 33–86. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, P.; Liu, S.; Peng, P.; Min, M.; Cheng, Y.; Anderson, E.; Zhou, N.; Fan, L.; Liu, C.; et al. Effects of feedstock characteristics on microwave-assisted pyrolysis—A review. Bioresour. Technol. 2017, 230, 143–151. [Google Scholar] [CrossRef]
- Uzoejinwa, B.B.; He, X.; Wang, S.; El-Fatah Abomohra, A.; Hu, Y.; Wang, Q. Co-pyrolysis of biomass and waste plastics as a thermochemical conversion technology for high-grade biofuel production: Recent progress and future directions elsewhere worldwide. Energy Convers. Manag. 2018, 163, 468–492. [Google Scholar] [CrossRef]
- Hassan, H.; Lim, J.K.; Hameed, B.H. Recent progress on biomass co-pyrolysis conversion into high-quality bio-oil. Bioresour. Technol. 2016, 221, 645–655. [Google Scholar] [CrossRef]
- Sogancioglu, M.; Yel, E.; Ahmetli, G. Pyrolysis of waste high density polyethylene (HDPE) and low density polyethylene (LDPE) plastics and production of epoxy composites with their pyrolysis chars. J. Clean. Prod. 2017, 165, 369–381. [Google Scholar] [CrossRef]
- Scarfato, P.; Incarnato, L.; Di Maio, L.; Dittrich, B.; Schartel, B. Influence of a novel organo-silylated clay on the morphology, thermal and burning behavior of low density polyethylene composites. Compos. Part B Eng. 2016, 98, 444–452. [Google Scholar] [CrossRef]
- Zattini, G.; Leonardi, C.; Mazzocchetti, L.; Cavazzoni, M.; Montanari, I.; Tosi, C.; Benelli, T.; Giorgini, L. Pyrolysis of Low-Density Polyethylene. In Sustainable Design and Manufacturing 2017; SDM 2017; Smart Innovation, Systems and Technologies; Campana, G., Howlett, R., Setchi, R.C.B., Eds.; Springer: Cham, Switzerland, 2017; pp. 480–490. [Google Scholar]
- Li, B.; Chen, G.; Zhang, H.; Sheng, C. Development of non-isothermal TGA–DSC for kinetics analysis of low temperature coal oxidation prior to ignition. Fuel 2014, 118, 385–391. [Google Scholar] [CrossRef]
- Ozbas, K.E.; Kök, M.V.; Hicyilmaz, C. DSC study of the combustion properties of turkish coals. J. Therm. Anal. Calorim. 2003, 71, 849–856. [Google Scholar] [CrossRef]
- Mohan, D.J.; Kullová, L. A study on the relationship between preparation condition and properties/performance of polyamide TFC membrane by IR, DSC, TGA, and SEM techniques. Desalin. Water Treat. 2013, 51, 586–596. [Google Scholar] [CrossRef]
- Boycheva, S.; Zgureva, D.; Vassilev, V. Kinetic and thermodynamic studies on the thermal behaviour of fly ash from lignite coals. Fuel 2013, 108, 639–646. [Google Scholar] [CrossRef]
- Zhou, L.; Zou, H.; Wang, Y.; Le, Z.; Liu, Z.; Adesina, A.A. Effect of potassium on thermogravimetric behavior and co-pyrolytic kinetics of wood biomass and low density polyethylene. Renew. Energy 2017, 102, 134–141. [Google Scholar] [CrossRef]
| Component | Method | Composition (wt.%) | |
|---|---|---|---|
| OPEFB | SWP | ||
| Ultimate analysis | |||
| Carbon (C) | Elemental analyzer | 54.40 | 86.93 |
| Hydrogen (H) | Elemental analyzer | 7.64 | 16.54 |
| Oxygen (O) | By difference | 36.44 | 1.39 |
| Nitrogen (N) | Elemental analyzer | 1.04 | 0.09 |
| Sulphur (S) | Elemental analyzer | 0.48 | 0.12 |
| Proximate analysis | |||
| Volatile matter | ASTM E 897-82 | 81.4 | 99.6 |
| Fixed carbon | By difference | 18.6 | 0.0 |
| Ash | ASTM D 1102-84 | 4.6 | 0.4 |
| Component | Heating Rate (°C min−1) | Pyrolysis Range (°C) | Peak Temperature (°C) | Total Mass Loss (%) |
|---|---|---|---|---|
| OPEFB: SWP | ||||
| 100:0 | 10 | 219–380 | 328 | 59.98 |
| 20 | 216–364 | 335 | 58.4 | |
| 80:20 | 10 | 220–502 | 337 & 484 | 72.29 |
| 20 | 220–497 | 332 & 483 | 74.5 | |
| 60:40 | 10 | 232–498 | 339 & 480 | 77.33 |
| 20 | 220–502 | 337 & 485 | 81.5 | |
| 50:50 | 10 | 243–503 | 331 & 485 | 83.40 |
| 20 | 237–552 | 304 & 460 | 93.16 | |
| 40:60 | 10 | 235–503 | 329 & 481 | 83.96 |
| 20 | 237–502 | 338 & 483 | 85.7 | |
| 20:80 | 10 | 262–501 | 329 & 486 | 89.14 |
| 20 | 273–501 | 329 & 484 | 90.75 | |
| 0:100 | 10 | 464–507 | 486 | 94.45 |
| 20 | 411–497 | 480 | 92.55 |
| Reaction | Heating Rate (°C min−1) | Tp (°C) | R2 | Ea (kJ mol−1) | A (min−1) | Reaction Rate Constant at Tp (min−1) | |
|---|---|---|---|---|---|---|---|
| Average | Standard Deviation | ||||||
| OPEFB 100 | 10 | 327 | 0.9821 | 46.83 | 1.29 × 103 | 0.15 | 0.0386 |
| 20 | 334 | 0.9655 | 44.21 | 1.18 × 103 | |||
| OPEFB 80, SWP 20-First stage | 10 | 331 | 0.9934 | 48.42 | 1.10 × 103 | 0.10 | 0.0332 |
| 20 | 332 | 0.9894 | 49.97 | 2.84 × 103 | |||
| OPEFB 80, SWP 20-Second stage | 10 | 486 | 0.9467 | 75.14 | 3.05 × 104 | 4.22 | 4.01 |
| 20 | 484 | 0.9659 | 72.33 | 8.05 × 105 | |||
| OPEFB 60, SWP 40-First stage | 10 | 320 | 0.9948 | 49.89 | 1.10 × 103 | 0.07 | 0.027 |
| 20 | 334 | 0.9926 | 50.85 | 2.34 × 103 | |||
| OPEFB 60, SWP 40-Second stage | 10 | 480 | 0.9523 | 93.25 | 7.03 × 105 | 0.42 | 0.1763 |
| 20 | 484 | 0.9281 | 123.65 | 2.01 × 108 | |||
| OPEFB 50, SWP 50-First stage | 10 | 327 | 0.9934 | 45.81 | 3.28 × 102 | 0.04 | 0.0107 |
| 20 | 316 | 0.9910 | 49.28 | 1.29 × 103 | |||
| OPEFB 50, SWP 50-Second stage | 10 | 485 | 0.9627 | 183.00 | 1.72 × 1012 | 0.54 | 0.1202 |
| 20 | 464 | 0.9862 | 112.16 | 5.89 × 107 | |||
| OPEFB 40, SWP 60-First stage | 10 | 326 | 0.9748 | 45.46 | 3.11 × 102 | 0.06 | 0.0223 |
| 20 | 334 | 0.9992 | 61.75 | 1.61 × 104 | |||
| OPEFB 40, SWP 60-Second stage | 10 | 479 | 0.9587 | 217.67 | 4.30 × 107 | 0.40 | 0.4002 |
| 20 | 484 | 0.9463 | 195.99 | 2.66 × 1013 | |||
| OPEFB 20, SWP 80-First stage | 10 | 327 | 0.9066 | 40.08 | 3.91 × 101 | 0.02 | 0.0092 |
| 20 | 335 | 0.9683 | 55.97 | 1.99 × 103 | |||
| OPEFB 20, SWP 80–Second stage | 10 | 481 | 0.9395 | 240.26 | 1.55 × 1016 | 0.62 | 0.2648 |
| 20 | 482 | 0.9393 | 229.86 | 7.02 × 1015 | |||
| SWP 100 | 10 | 486 | 0.9092 | 346.93 | 2.95 × 1023 | 0.71 | 0.3111 |
| 20 | 480 | 0.9540 | 234.36 | 1.83 × 1016 | |||
| Sample (%) | TOnset (°C) | TPeak (°C) | TEndset (°C) | Enthalpy, H (J g−1) |
|---|---|---|---|---|
| First stage | ||||
| OPEFB, 100 | 42.06 | 87.64 | 131.32 | 147.69 |
| OPEFB, 90: SWP 10 | 45.21 | 91.04 | 130.31 | 155.55 |
| OPEFB, 80: SWP, 20 | 42.04 | 90.42 | 146.40 | 144.45 |
| OPEFB, 70: SWP, 30 | 89.66 | 120.77 | 127.50 | 83.54 |
| SWP, 100 | 95.30 | 122.15 | 122.15 | 117.39 |
| Second stage | ||||
| OPEFB, 100 | 147.23 | 240.96 | 292.32 | 88.41 |
| OPEFB, 90: SWP, 10 | - | - | - | 84.42 |
| OPEFB, 80: SWP, 20 | - | - | - | 59.83 |
| OPEFB, 70: SWP, 30 | 152.08 | 251.87 | 332.97 | 88.97 |
| SWP, 100 | - | - | - | - |
| Third stage | ||||
| OPEFB, 100 | 315.25 | 340.20 | 353.66 | 22.28 |
| OPEFB, 90: SWP, 10 | 287.90 | 339.55 | 355.90 | 158.21 |
| OPEFB, 80: SWP, 20 | 295.60 | 339.89 | 354.82 | 161.04 |
| OPEFB, 70: SWP, 30 | - | - | - | 145.40 |
| SWP, 100 | - | - | - | - |
| Forth stage | ||||
| OPEFB, 100 | 437.11 | 465.29 | 520.42 | 18.44 |
| OPEFB, 90: SWP, 10 | 423.74 | 484.73 | 517.18 | - |
| OPEFB, 80: SWP, 20 | 476.52 | 487.21 | 515.89 | - |
| OPEFB, 70: SWP, 30 | 476.17 | 489.27 | 510.80 | - |
| SWP, 100 | 455.68 | 491.56 | 511.48 | 1030.19 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salman, B.; Ong, M.Y.; Nomanbhay, S.; Salema, A.A.; Sankaran, R.; Show, P.L. Thermal Analysis of Nigerian Oil Palm Biomass with Sachet-Water Plastic Wastes for Sustainable Production of Biofuel. Processes 2019, 7, 475. https://doi.org/10.3390/pr7070475
Salman B, Ong MY, Nomanbhay S, Salema AA, Sankaran R, Show PL. Thermal Analysis of Nigerian Oil Palm Biomass with Sachet-Water Plastic Wastes for Sustainable Production of Biofuel. Processes. 2019; 7(7):475. https://doi.org/10.3390/pr7070475
Chicago/Turabian StyleSalman, Bello, Mei Yin Ong, Saifuddin Nomanbhay, Arshad Adam Salema, Revathy Sankaran, and Pau Loke Show. 2019. "Thermal Analysis of Nigerian Oil Palm Biomass with Sachet-Water Plastic Wastes for Sustainable Production of Biofuel" Processes 7, no. 7: 475. https://doi.org/10.3390/pr7070475
APA StyleSalman, B., Ong, M. Y., Nomanbhay, S., Salema, A. A., Sankaran, R., & Show, P. L. (2019). Thermal Analysis of Nigerian Oil Palm Biomass with Sachet-Water Plastic Wastes for Sustainable Production of Biofuel. Processes, 7(7), 475. https://doi.org/10.3390/pr7070475









