Adsorptive Removal of Pyridine in Simulation Wastewater Using Coke Powder
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Batch Adsorption Tests
2.2.2. Adsorption Isotherm Analysis
3. Results and Discussion
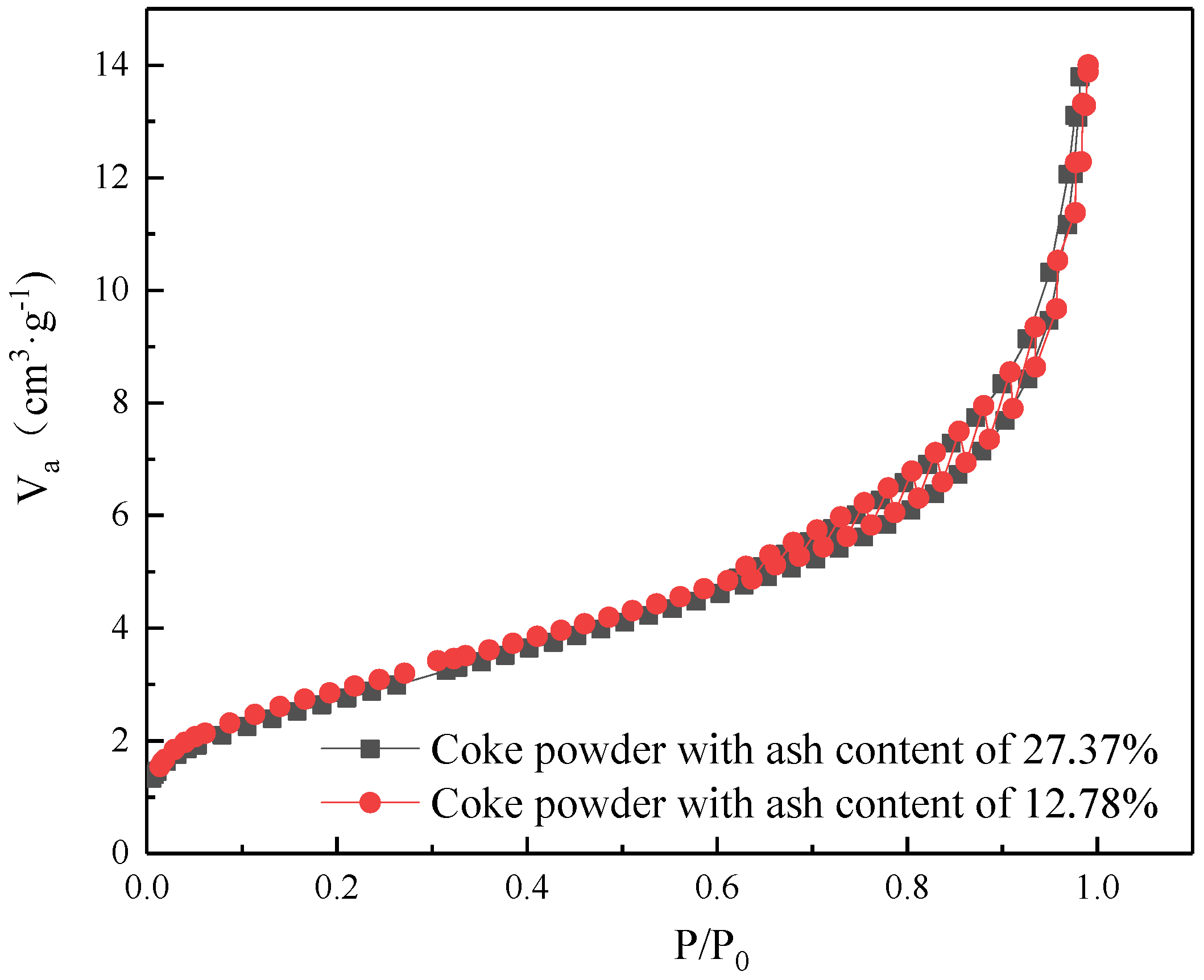
3.1. Coke Powder Properties
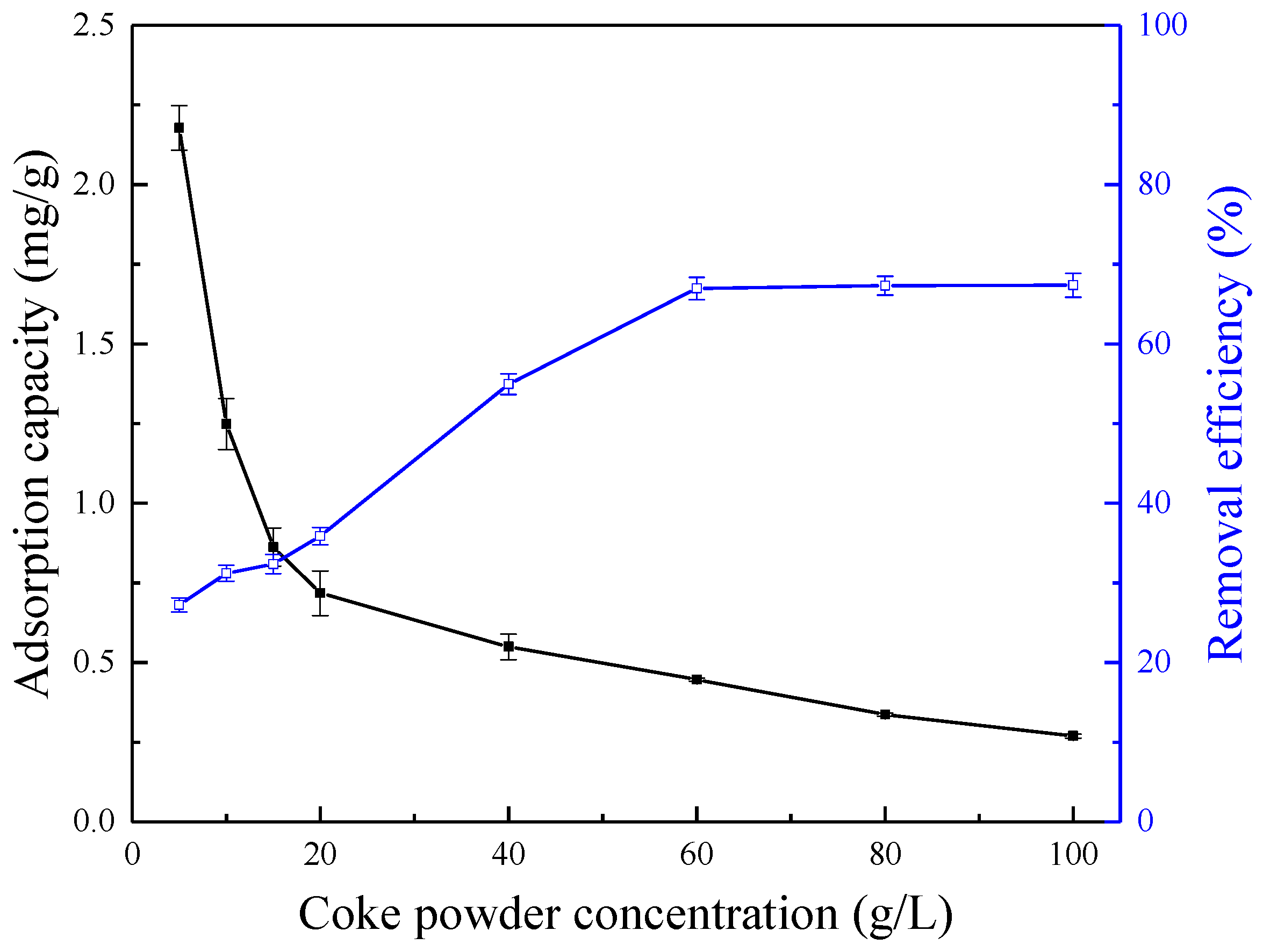
3.2. Effect of Coke Powder Concentration
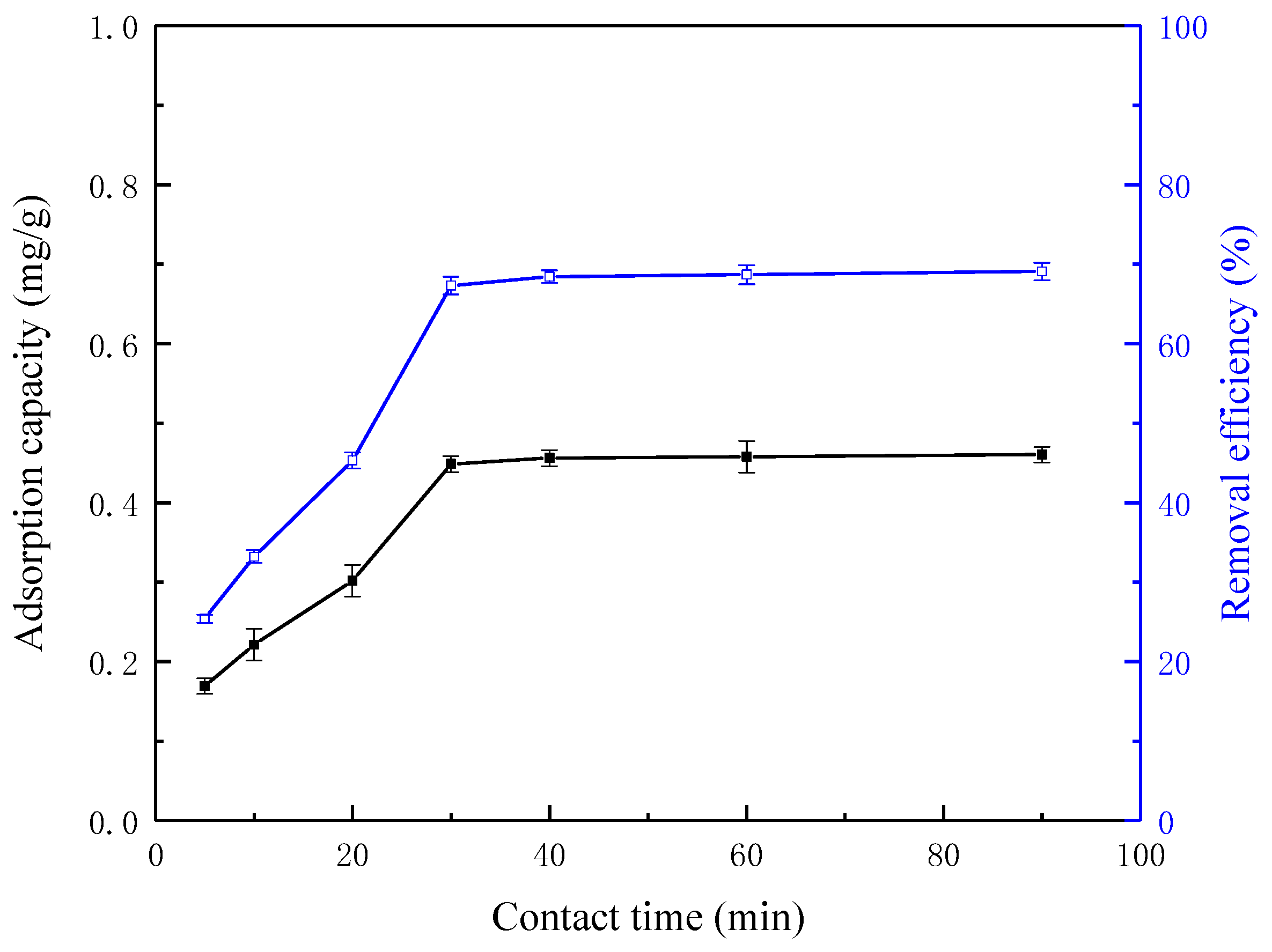
3.3. Effect of Contact Time
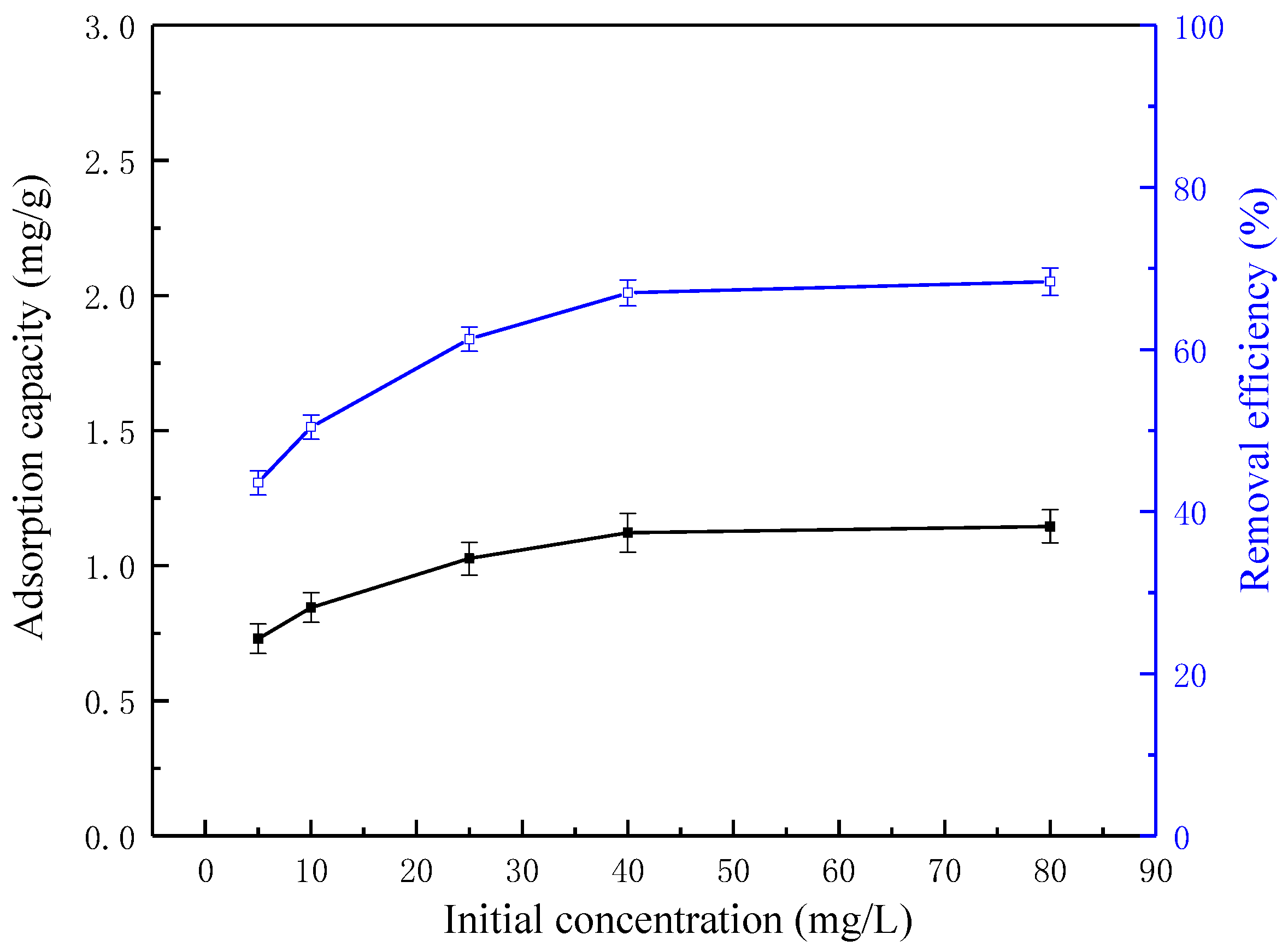
3.4. Effect of Initial Pyridine Concentration
3.5. Effect of Ash Content in Coke Powder
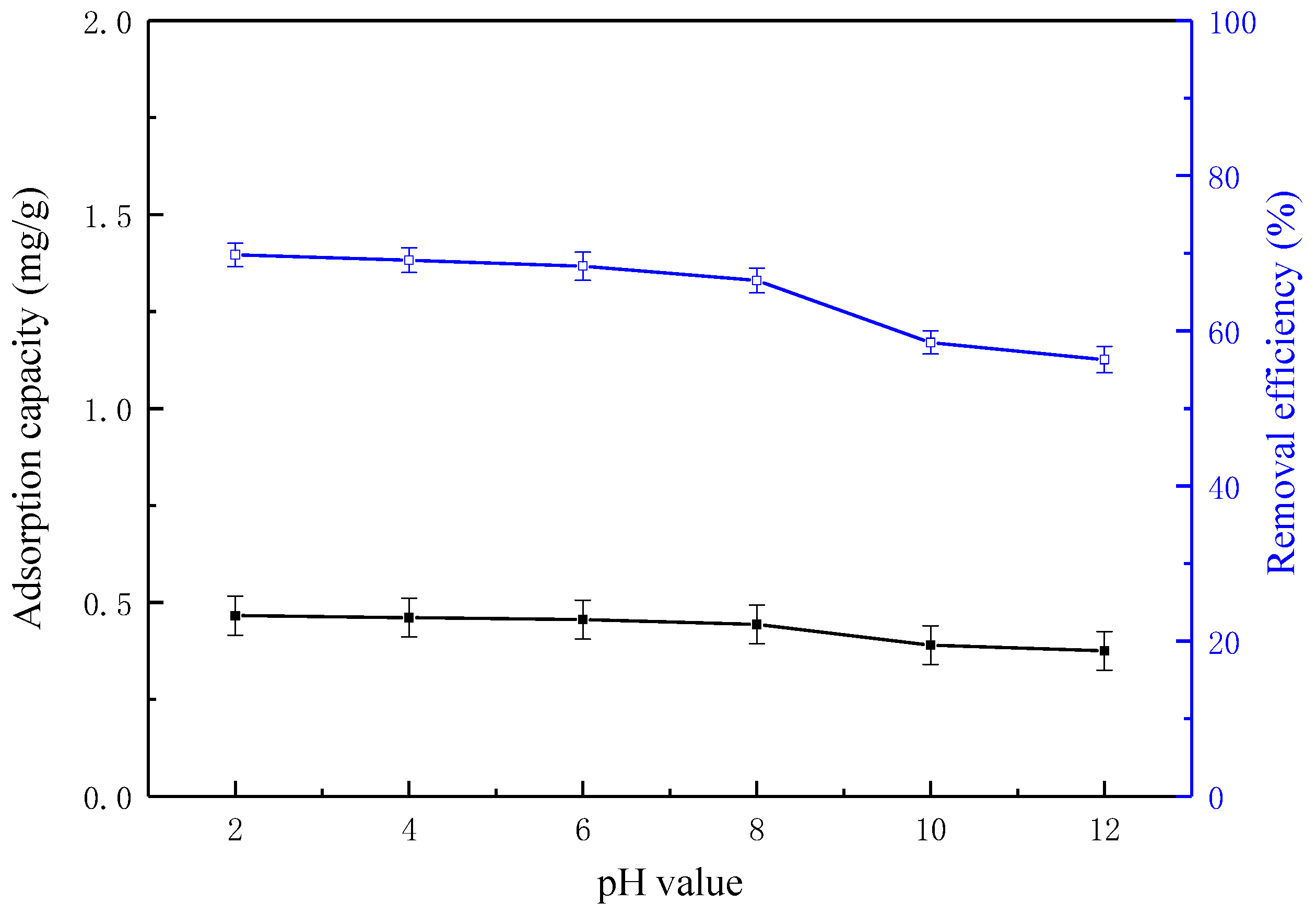
3.6. Effect of Solution pH
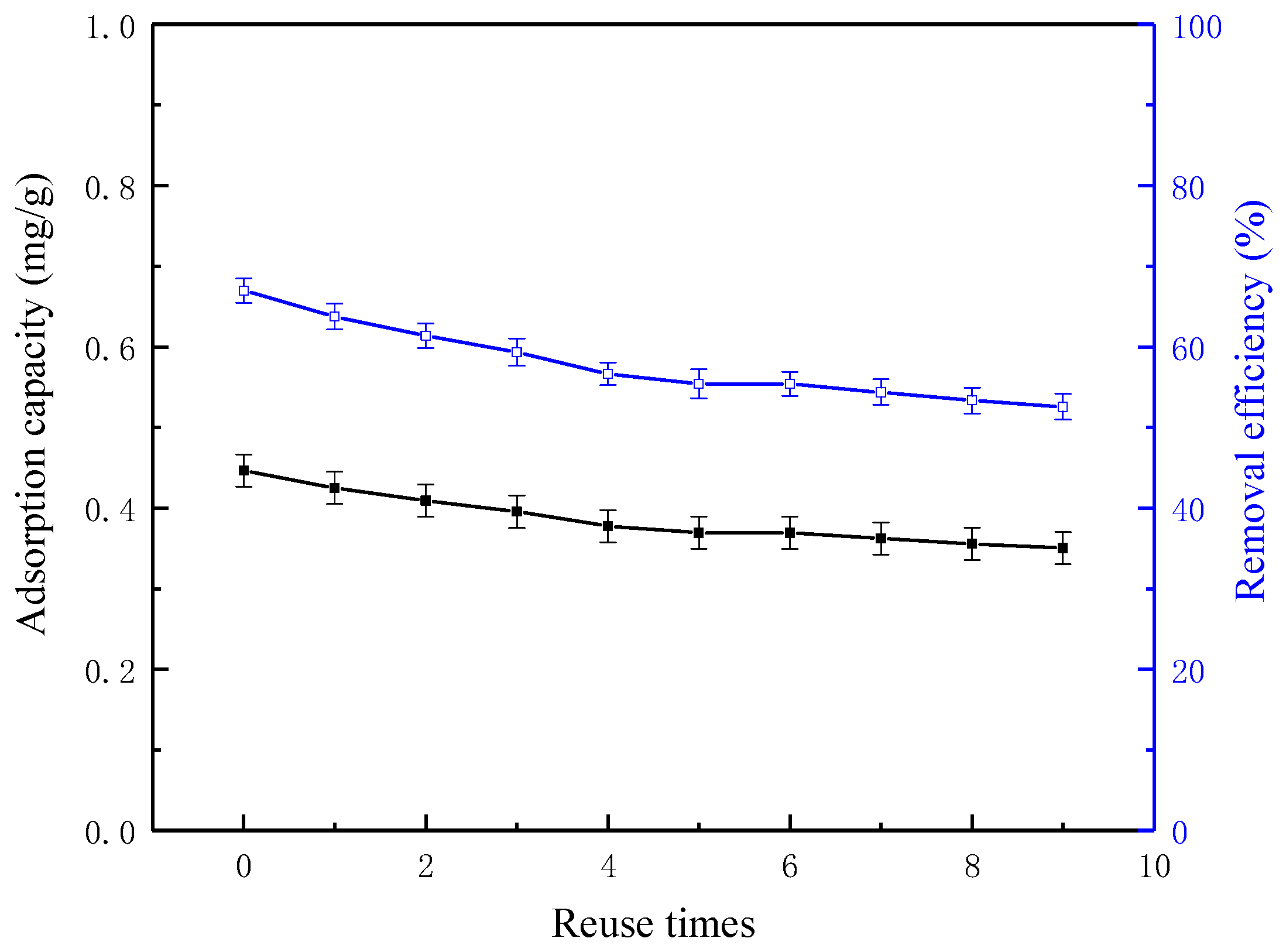
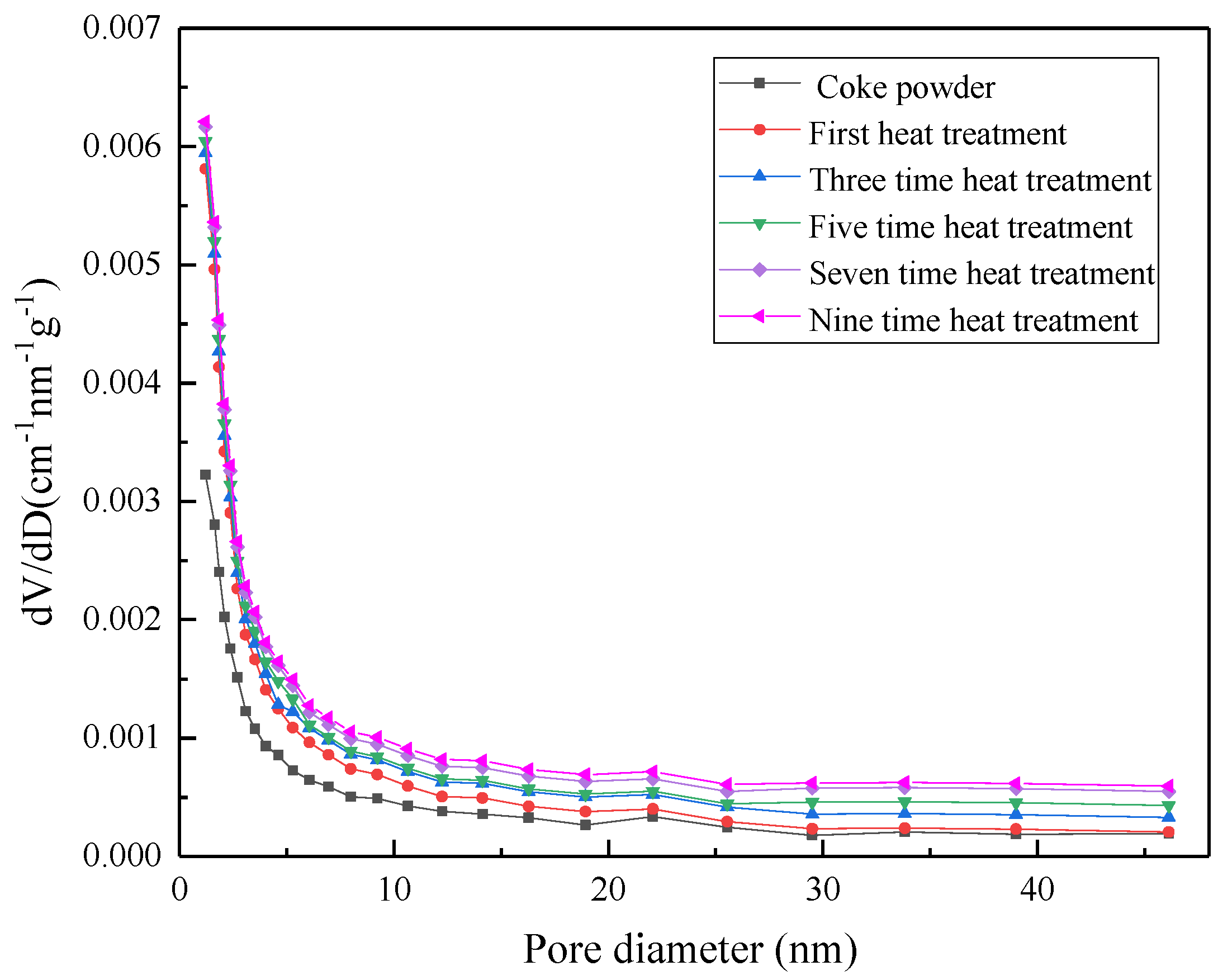
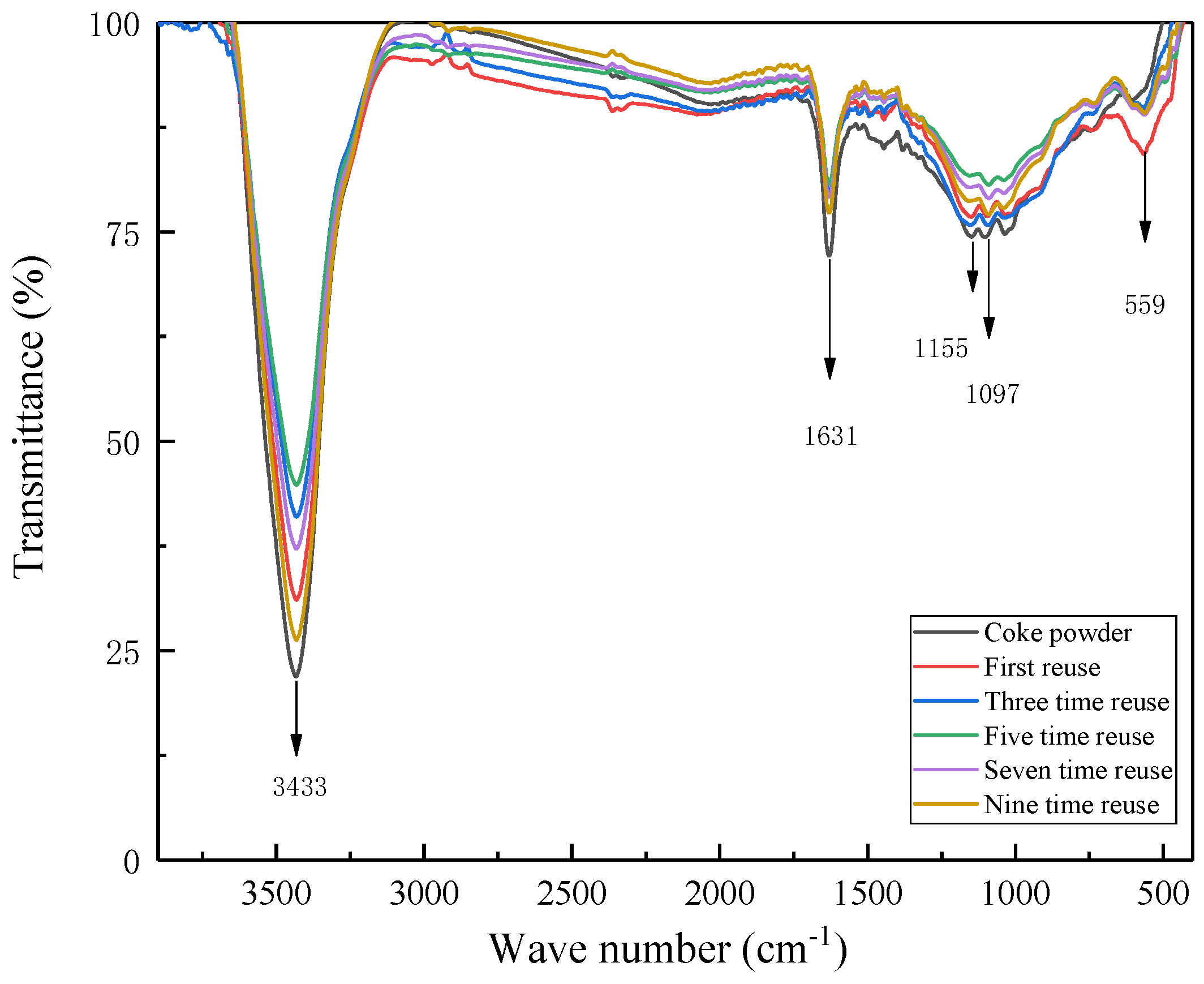
3.7. Reuse of Regenerated Coke Powder After Heat Treatment
3.8. Adsorption Isotherm Modeling
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zalat, O.A.; Elsayed, M.A. A study on microwave removal of pyridine from wastewater. J. Environ. Chem. Eng. 2013, 1, 137–143. [Google Scholar] [CrossRef]
- Hong, Z.; Liang, L.; Hong, W.L. Fast photo-catalytic degradation of pyridine in nano aluminum oxide suspension systems. J. Environ. Sci. 2011, 23 (Suppl S), S156–S158. [Google Scholar]
- Lee, S.T.; Lee, S.B.; Park, Y.H. Characterization of a pyridine-degrading branched gram-positive bacterium isolated from the anoxic zone of an oil shale column. Appl. Microbiol. Biotechnol. 1991, 35, 824–829. [Google Scholar] [CrossRef]
- Mohan, S.V.; Sistla, S.; Guru, R.K.; Prasad, K.K.; Kumar, C.S.; Ramakrishna, S.V. Microbial degradation of pyridine using pseudomonas sp. and isolation of plasmid responsible for degradation. Waste Manag. 2003, 23, 167–171. [Google Scholar] [CrossRef]
- Watson, G.K.; Cain, R.B. Microbial metabolism of the pyridine ring. metabolic pathways of pyridine biodegradation by soil bacteria. Biochem. J. 1975, 146, 157–172. [Google Scholar] [CrossRef] [PubMed]
- Khasaeva, F.; Vasilyuk, N.; Terentyev, P.; Troshina, M.; Lebedev, A.T. A novel soil bacterial strain degrading pyridines. Environ. Chem. Lett. 2011, 9, 439–445. [Google Scholar] [CrossRef]
- Kim, M.K.; Singleton, I.; Yin, C.R.; Quan, Z.X.; Lee, M.; Lee, S.T. Influence of phenol on the biodegradation of pyridine by freely suspended and immobilized pseudomonas putida mk1. Lett. Appl. Microbiol. 2010, 42, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.; Lee, S.; Lee, K.; Chung, J. Degradation of pyridine by nocardioides sp. strain os4 isolated from the oxic zone of a spent shale column. Can. J. Microbiol. 1997, 43, 205–209. [Google Scholar] [CrossRef]
- Qiao, L.; Wang, J.L. Microbial degradation of pyridine by paracoccus sp. isolated from contaminated soil. J. Hazard. Mater. 2010, 176, 220–225. [Google Scholar] [CrossRef]
- Yao, H.; Ren, Y.; Deng, X.; Wei, C. Dual substrates biodegradation kinetics of m-cresol and pyridine by lysinibacillus cresolivorans. J. Hazard. Mater. 2011, 186, 1136–1140. [Google Scholar] [CrossRef]
- Sun, J.Q.; Xu, L.; Tang, Y.Q. Degradation of pyridine by one Rhodococcus, strain in the presence of chromium (VI) or phenol. J. Hazard. Mater. 2011, 191, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Padoley, K.V.; Mudliar, S.N.; Banerjee, S.K. Fenton oxidation: A pretreatment option for improved biological treatment of pyridine and 3-cyanopyridine plant wastewater. Chem. Eng. J. 2011, 166, 1–9. [Google Scholar] [CrossRef]
- Xu, Z.; Liu, Z.; Lu, X.; Xu, Y. Pretreatment process of organic wastewater containing pyridines. J. Environ. Eng. ASCE 2014, 8, 1863–1868. [Google Scholar]
- Liu, S.; Wang, Q.; Sun, T.; Wu, C.; Shi, Y. The effect of different types of micro-bubbles on the performance of the coagulation flotation process for coke waste-water. J. Chem. Technol. Biotechnol. 2012, 87, 10. [Google Scholar] [CrossRef]
- Lai, P.; Zhao, H.Z.; Wang, C.; Ni, J.R. Advanced treatment of coking wastewater by coagulation and zero-valent iron processes. J. Hazard. Mater. 2007, 147, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Yan-Bin, Z.; Ai-Wu, Z. Separation of pyridine and toluene by extractive distillation. Chem. Eng. 2012, 40, 13–16. [Google Scholar]
- Lei, K.; Ming, L.I. Recovering pyridine from industrial waste water by means of continuous distillation. Pesticides 2005, 02, 69–71. [Google Scholar]
- Zhu, Q.; Moggridge, G.D.; Ainte, M.; Mantle, M.D.; Gladden, L.F.; D’Agostino, C. Adsorption of pyridine from aqueous solutions by polymeric adsorbents mn 200 and mn 500. Part 1: Adsorption performance and pfg-nmr studies. Chem. Eng. J. 2016, 306, 67–76. [Google Scholar] [CrossRef]
- Larson, R.A.; Weber, E.J. Reaction Mechanisms in Environmental Organic Chemistry; CRC Press: Boca Raton, FL, USA, 1994; pp. 433–434. [Google Scholar]
- Zhu, L.; Tian, S.; Shi, Y. Adsorption of volatile organic compounds onto porous clay heterostructures based on spent organobentonites. Clay Clay Min. 2005, 53, 123–136. [Google Scholar] [CrossRef]
- Lataye, D.H.; Mishra, I.M.; Mall, I.D. Removal of pyridine from aqueous solution by adsorption on bagasse fly ash. Ind. Eng. Chem. Res. 2006, 45, 3934–3943. [Google Scholar] [CrossRef]
- Gosu, V.; Gurjar, B.R.; Rao, Y.S.; Zhang, T.C. Treatment of pyridine-bearing wastewater by nano zero-valent iron supported on activated carbon derived from agricultural waste. Desalin. Water Treat. 2015, 57, 1–11. [Google Scholar] [CrossRef]
- Ning, K.; Wang, J.; Xu, H.; Sun, X.; Huang, G.; Liu, G. Effects and mechanistic aspects of absorbing organic compounds by coking coal. Water Sci. Technol. 2017, 76, 2280–2290. [Google Scholar] [CrossRef]
- Gao, L.; Li, S.; Wang, Y. Effect of different ph coking wastewater on adsorption of coking coal. Water Sci. Technol. 2016, 73, 582–587. [Google Scholar] [CrossRef]
- He-Ming, L.; Shu-Rong, Y.U.; Hui-Xia, F.; Jian-Qiang, Z.; Xia, Z.; Yi, W. The mechanism and properties of methylene blue adsorption on modified coke powder. J. China Coal Soc. 2009, 34, 971–976. [Google Scholar]
- Peng, D.; Xiao-Rui, W. Research on adsorption performance of fluorin ion over modified coke. Contemp. Chem. Ind. 2014, 43, 1163–1165. [Google Scholar]
- Mall, I.D.; Srivastava, V.C.; Agarwal, N.K.; Mishra, I.M. Adsorptive removal of malachite green dye from aqueous solution by bagasse fly ash and activated carbon-kinetic study and equilibrium isotherm analyses. Coll. Surface A 2005, 264, 17–28. [Google Scholar] [CrossRef]
- Basar, C.A. Applicability of the various adsorption models of three dyes adsorption onto activated carbon prepared waste apricot. J. Hazard. Mater. 2006, 135, 232–241. [Google Scholar] [CrossRef]
- Mori, A.; Maksimov, I.L. On the temkin model of solid–liquid interface. J. Cryst. Growth 1999, 200, 297–304. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, Y.; Liu, J. Utilization of mineral processing flocculants in dewatering of coal slimes. China Coal 2003, 29, 46–47. [Google Scholar]
- Zhou, X.; Ma, L.; Yang, J.; Huang, B.; Zou, Y.; Tang, J. Properties of graphitized boron-doped coal-based coke powders as anode for lithium-ion batteries. J. Electroanal. Chem. 2013, 698, 39–44. [Google Scholar] [CrossRef]
- Pandey, H.D.; Prasad, M.S. Effect of moisture on the accuracy of coke-ash determination by x-ray backscattering. Spectrochim. Acta Part B Atom. Spectr. 1984, 39, 943–946. [Google Scholar] [CrossRef]






| Conditions | Pyridine Concentration (mg/L) | Coke Powder Concentration (g/L) | Oscillation Speed (r/min) | Contact Time (min) | Ash Content (%) | Temperature (K) | pH | |
|---|---|---|---|---|---|---|---|---|
| Parameters | ||||||||
| Coke powder concentration | 40 | 5–100 | 150 | 30 | 27.37 | 298 | – | |
| Contact time | 40 | 60 | 150 | 5–90 | 27.37 | 298 | – | |
| Initial pyridine concentration | 5–80 | 60 | 150 | 30 | 27.37 | 298 | – | |
| Ash content | 40 | 60 | 150 | 30 | 27.37, 12.78 | 298 | – | |
| pH | 40 | 60 | 150 | 30 | 27.37 | 298 | 2-12 | |
| Regeneration of adsorbents | 40 | 60 | 150 | 30 | 27.37 | 298 | – | |
| Content (%) | SiO2 | CaO | Al2O3 | Fe2O3 | MgO | K2O | Na2O |
|---|---|---|---|---|---|---|---|
| Coke powder (Ash 27.37%) | 6.15 | 4.58 | 5.36 | 2.62 | 0.52 | 0.14 | 0.11 |
| Coke powder (Ash 12.78%) | 2.67 | 2.13 | 2.24 | 0.98 | 0.31 | 0.10 | 0.09 |
| Specific Surface Area (m2/g) | Average Pore Size (nm) | Mesoporous Volume (cm3/g) | Total Pore Volume (cm3/g) | |
|---|---|---|---|---|
| Coke powder (Ash 27.37%) | 51.54 | 3.23 | 0.019 | 0.019 |
| Coke powder (Ash 12.78%) | 63.46 | 3.55 | 0.020 | 0.020 |
| Coke Powder Concentration (g/L) | Pyridine Removal Efficiency by Coke Powder with 27.37% of Ash Content (%) | Pyridine Removal Efficiency by Coke Powder with 12.78% of Ash Content (%) |
|---|---|---|
| 5 | 27.23 | 28.50 |
| 10 | 31.21 | 35.89 |
| 20 | 35.89 | 47.65 |
| 40 | 54.96 | 68.54 |
| 60 | 66.97 | 85.20 |
| 80 | 67.32 | 84.06 |
| Adsorbent | Number of Reuses | Specific Surface Area (m2/g) | Average Pore Size (nm) | Mesoporous Volume (m3/g) | Total Pore Volume (m3/g) |
|---|---|---|---|---|---|
| Coke powder | 0 | 51.54 | 3.23 | 0.019 | 0.019 |
| 1 | 51.34 | 7.43 | 0.021 | 0.025 | |
| 3 | 50.52 | 8.23 | 0.025 | 0.027 | |
| 5 | 50.23 | 9.56 | 0.031 | 0.036 | |
| 7 | 50.15 | 10.01 | 0.040 | 0.043 | |
| 9 | 50.09 | 12.32 | 0.052 | 0.055 |
| Adsorbate | Adsorbent | Isotherm Models | Constants and Correlation Coefficients | |||
|---|---|---|---|---|---|---|
| Pyridine | Coke powder | Langmuir | KL (L/mg) | qm (mg/g) | RLa | R2 |
| 0.0529 | 31.7460 | 0.8921 | 0.9240 | |||
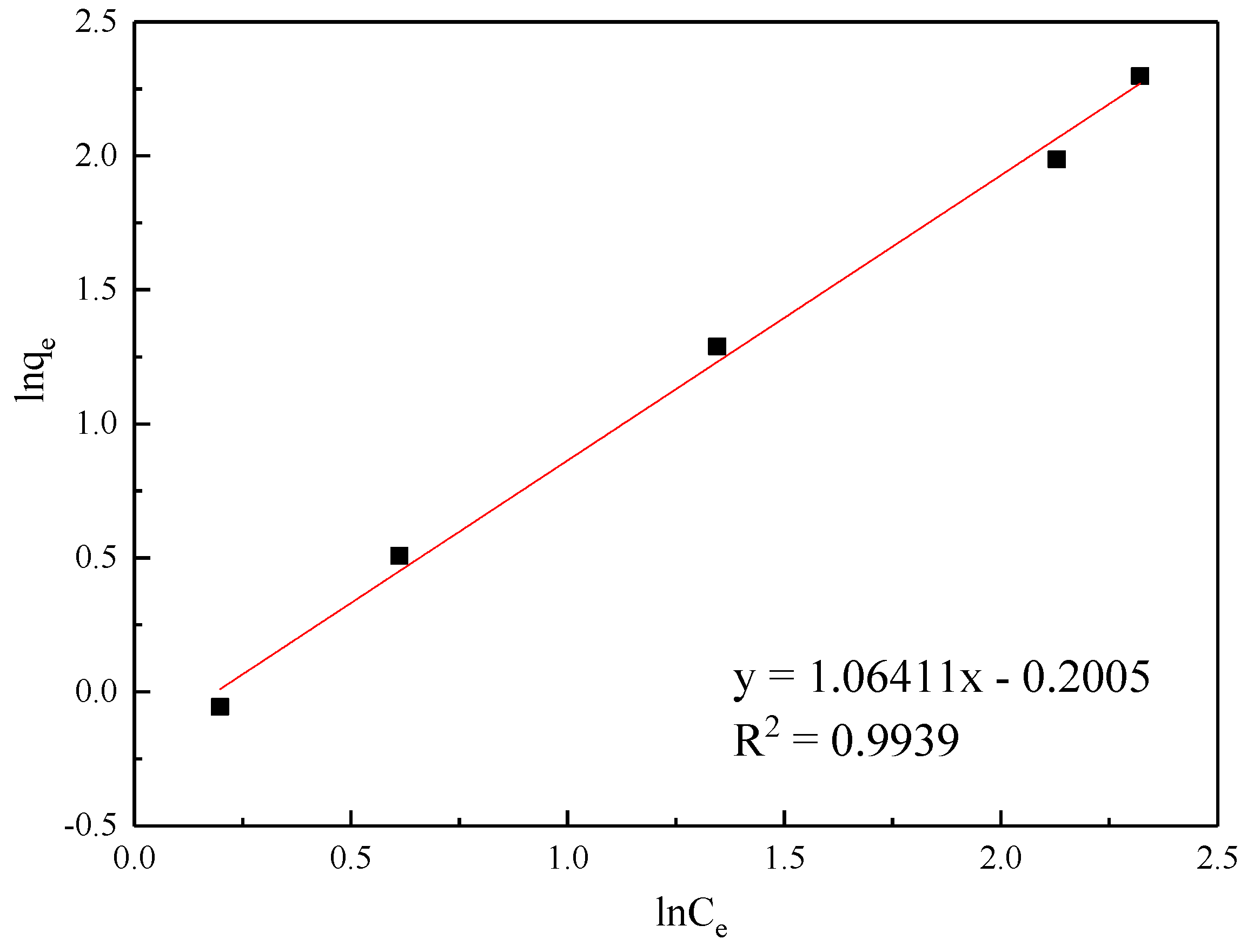
| Freundlich | KF (L/mg) | 1/n | - | R2 | ||
| 0.9518 | 0.3531 | - | 0.9939 | |||
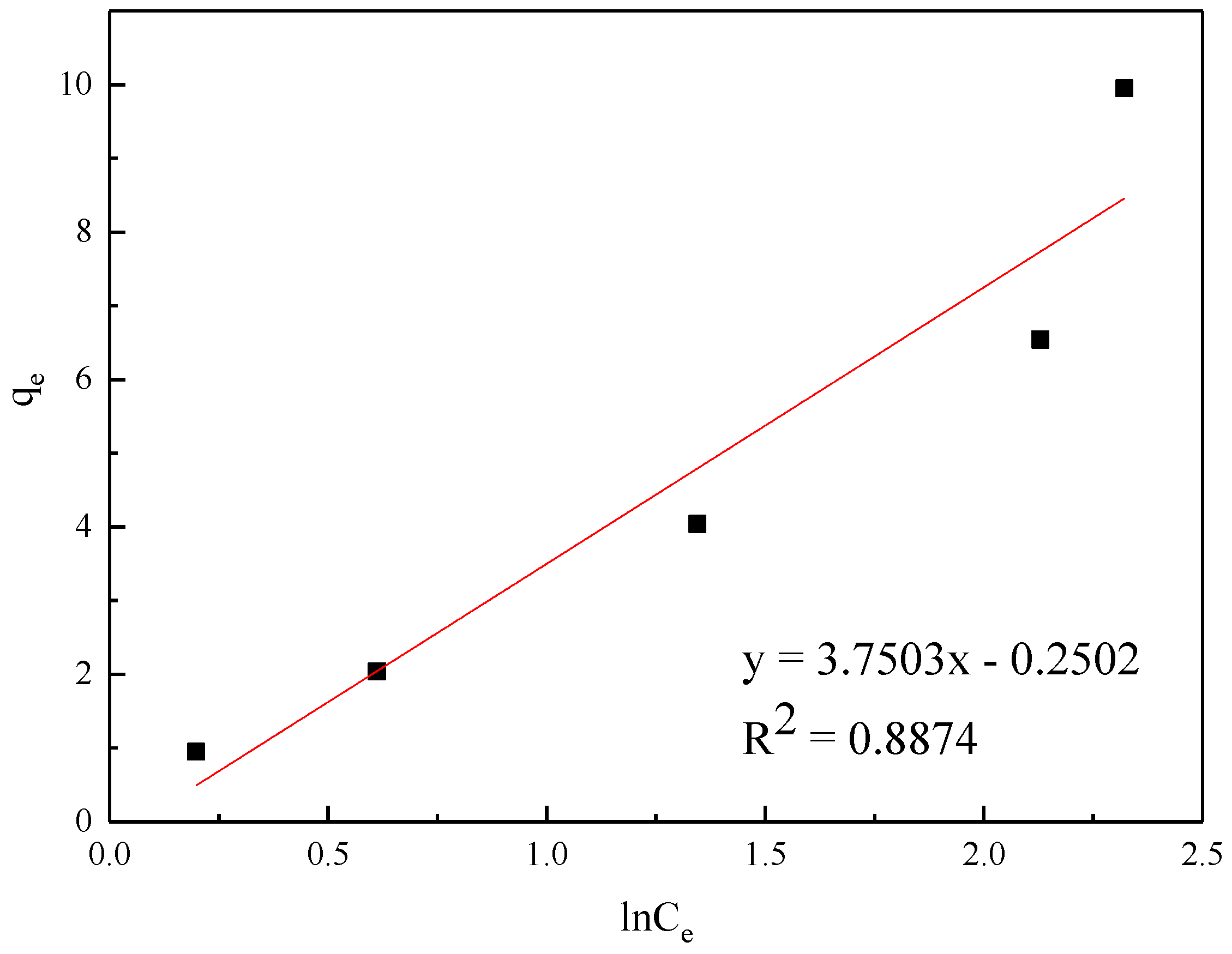
| Temkin | KT (L/mg) | bT (KJ/mol) | - | R2 | ||
| 3.1571 | 3.0592 | - | 0.8874 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, Q.; Wang, L.; Li, Z.; Xie, Y.; He, Q.; Wang, Y. Adsorptive Removal of Pyridine in Simulation Wastewater Using Coke Powder. Processes 2019, 7, 459. https://doi.org/10.3390/pr7070459
Gao Q, Wang L, Li Z, Xie Y, He Q, Wang Y. Adsorptive Removal of Pyridine in Simulation Wastewater Using Coke Powder. Processes. 2019; 7(7):459. https://doi.org/10.3390/pr7070459
Chicago/Turabian StyleGao, Qieyuan, Lei Wang, Zhipeng Li, Yaqi Xie, Qiongqiong He, and Yongtian Wang. 2019. "Adsorptive Removal of Pyridine in Simulation Wastewater Using Coke Powder" Processes 7, no. 7: 459. https://doi.org/10.3390/pr7070459
APA StyleGao, Q., Wang, L., Li, Z., Xie, Y., He, Q., & Wang, Y. (2019). Adsorptive Removal of Pyridine in Simulation Wastewater Using Coke Powder. Processes, 7(7), 459. https://doi.org/10.3390/pr7070459





