Process Modeling and Evaluation of Plasma-Assisted Ethylene Production from Methane
Abstract
1. Introduction
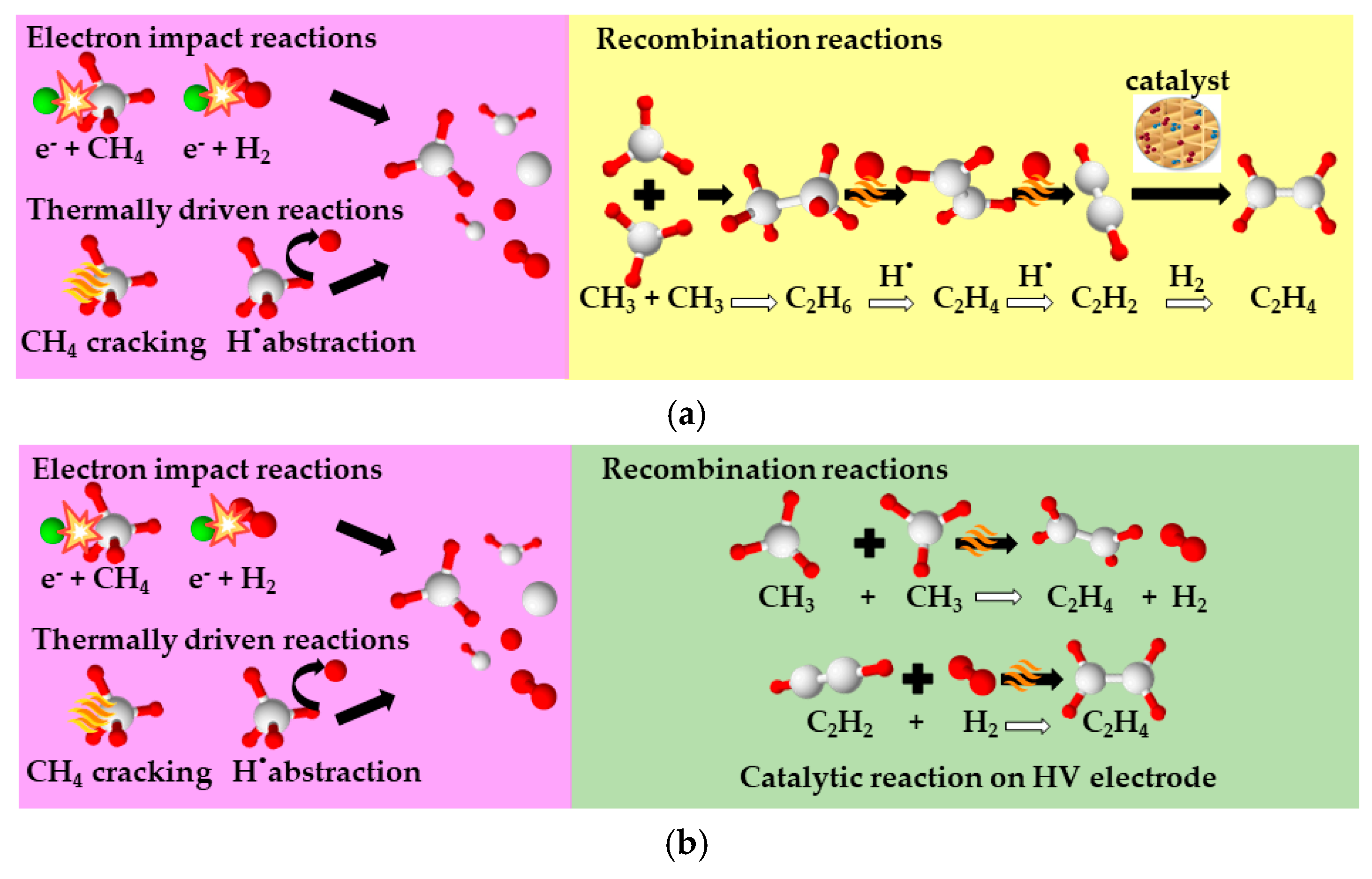
2. Plasma-Assisted Non-Oxidative Methane Coupling
3. Process Model
3.1. Modeling Inputs and Assumptions
- -
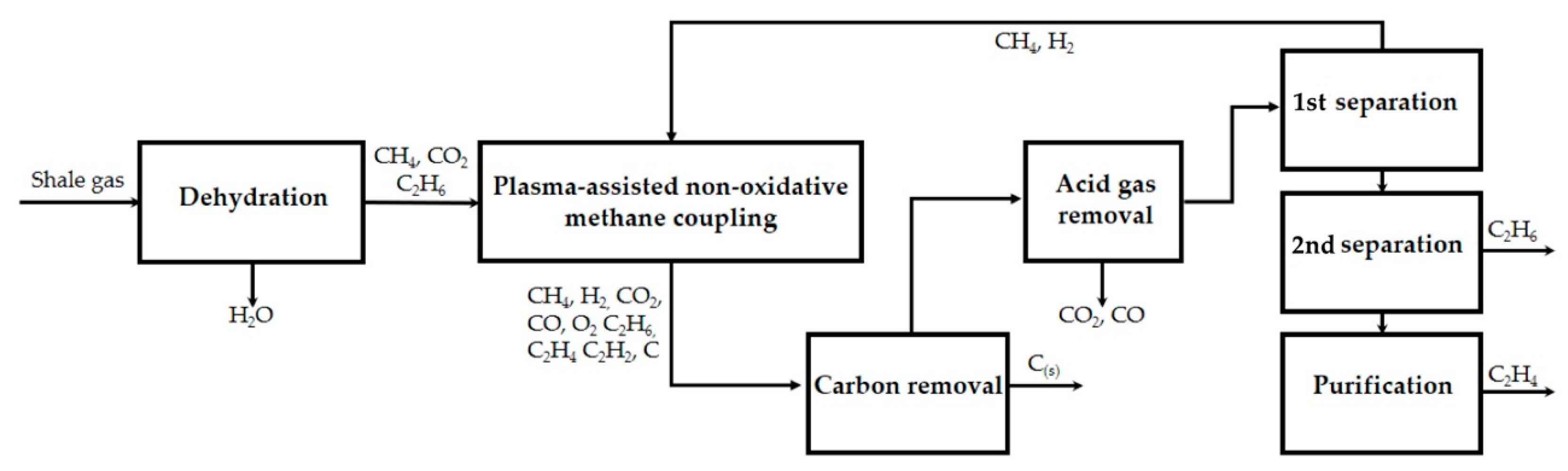
- Shale gas, a major source of natural gas rich-in methane, is considered as the main feedstock for ethylene production. The composition of shale gas varies depending on the extraction region [20], thus a stream containing 79% CH4, 15% C2H6, 3% CO2, and 3% H2O on a molar basis is considered as feedstock for the simulation. N2 and H2S impurities are not taken into consideration since they are usually present in negligible amounts. The plasma activity and high dilution with H2 (CH4:H2 = 1:1 mol) in the inlet plasma reactor stream guarantee that the feed stream meets the pipeline, safety, and quality standards [21,22].
- -
- Oil, natural gas liquids (NGLs), and most of the water, which natural gas is mixed with in the reservoir, are most often separated in equipment installed at or near the wellhead. Therefore, system boundaries include the dehydration step required prior to the plasma reactor and the plasma-assisted ethylene production process itself. Unlike the thermally driven processes, gas sweetening is not required since plasma reactors operation is robust in CO2 and other impurities presence. In addition, the concentration of impurities may be reduced via electron-impact dissociation reactions occurring in the plasma zone [23].
- -
- The Peng-Robinson (PR) equation of state is employed to calculate the vapor-liquid equilibria and the chemical compounds thermodynamic properties. The PR equation is commonly applied to hydrocarbon mixture systems and are specifically used for hydrocarbon fluids found in underground reservoirs [24].
- -
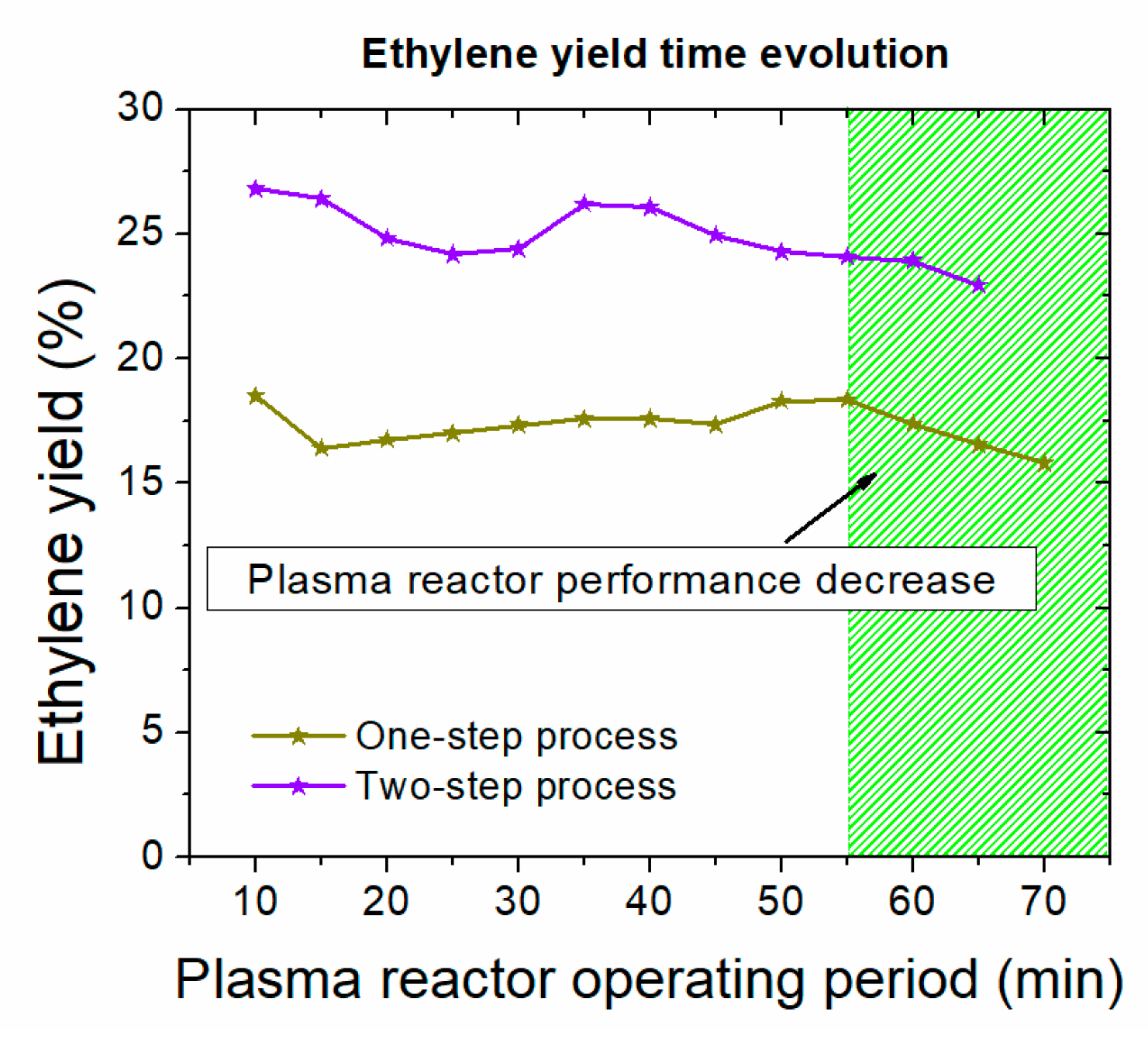
- The reactants conversion and products selectivity set in the simulation of plasma reactors for both process alternatives are based on linear extrapolation of the experimental data shown in Figure 2. The experiments reported in our previous works [11] and [12] were repeated by co-feeding C2H6 and CO2, the most important compounds, after methane, in shale gas reservoirs, to simulate a more realistic case. A feed stream containing 82% CH4, 15% C2H6, and 3% CO2 on a molar basis, which is aligned with the composition attained after the dehydration unit in the simulation, was fed into the NPD reactor. The plasma-assisted reaction was run at the optimum operating window, as is also defined in the reference works [11,12]. No essential differences are observed in the NPD reactor performance after C2H6 and CO2 addition. Moreover, C2H6 and CO2 concentrations decrease as they are cracked in the plasma zone. CH4, C2H6, and CO2 conversions of ~35%, ~60%, and ~25%, respectively, and C2H4 selectivity of ~54% are attained in the one-step process consuming ~2020 kJ/molC2H4 electric energy. CH4, C2H6, and CO2 conversions of ~33%, ~45%, and ~25%, respectively, and C2H4 selectivity of ~74% are attained in the two-step process consuming ~1642 kJ/molC2H4 electric energy.
- -
- The stochiometric reactor (RSTOIC) model is used to simulate the plasma reactor at steady state in the Aspen Plus process simulator (Version 10, AspenTech, Bedford, MA, USA) since the stoichiometry and molar extent of the reactions taking place have been defined by the experiments.
- -
- Numbering up the small-scale NPD reactors is foreseen as the most possible way to achieve higher capacities. Current industrially launched plasma reactors are based on this approach [25]. Therefore, linear extrapolation of the experimental data is considered a valid assumption. The reactor feed used in the simulation corresponds to a rate ~104 higher than the one tested in our experiments. Given that the industrially launched units can host a bundle of ~750 plasma reactors, nine units placed in parallel would satisfy the selected capacity.
- -
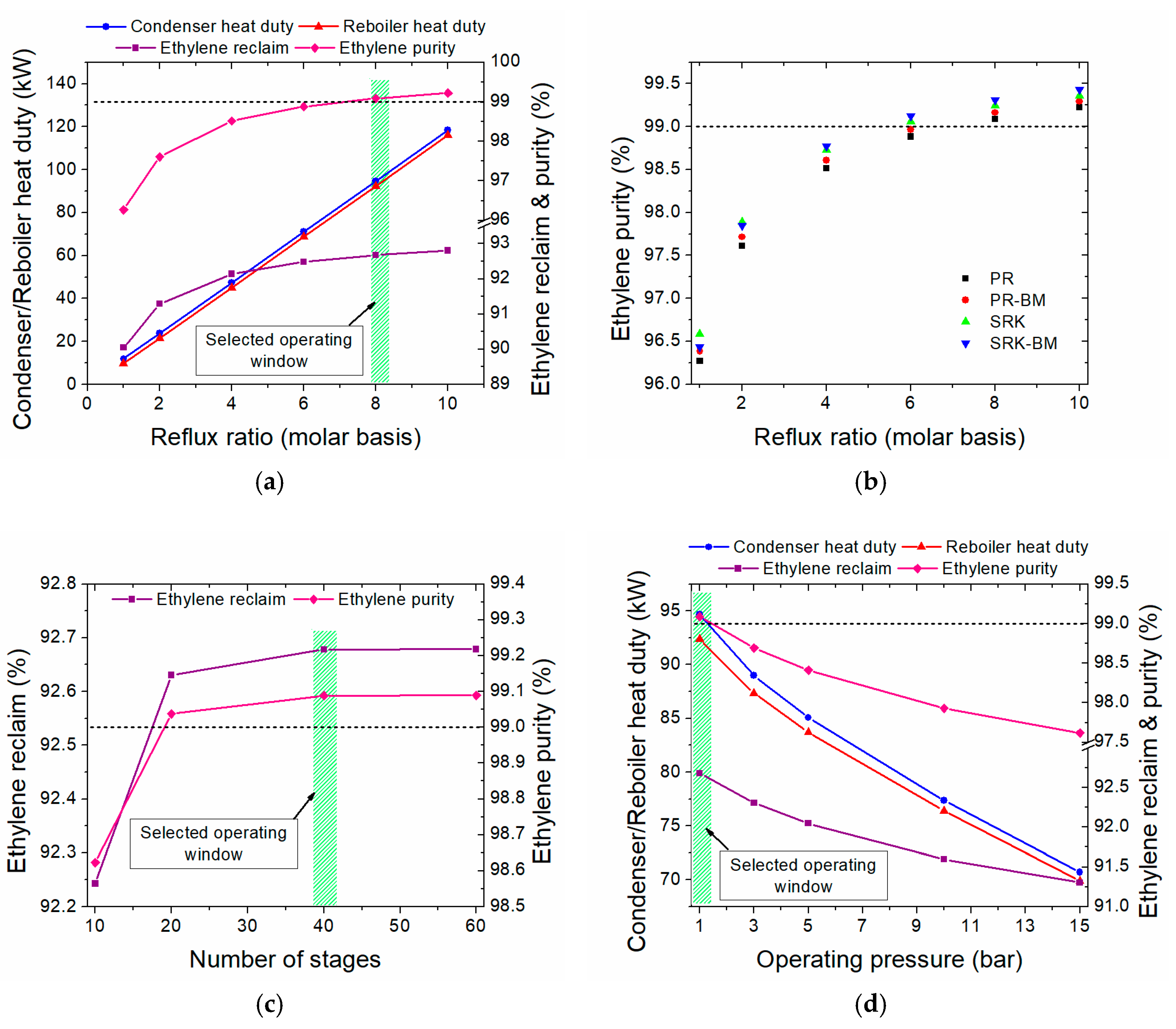
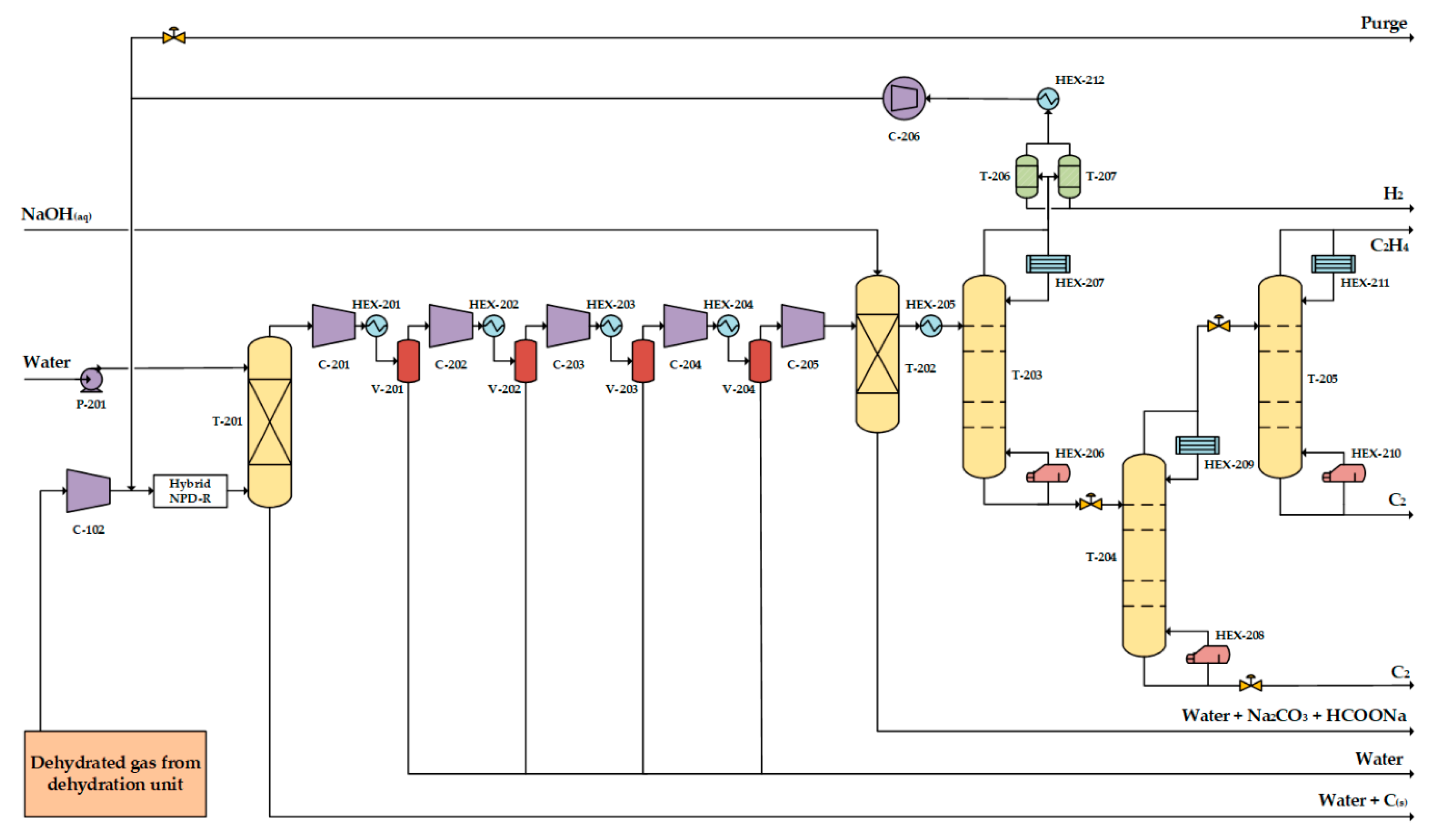
- Ethylene for polymers production is aimed to be produced via the plasma-assisted process; thus, polymer-grade ethylene (≥99% mol purity [26]) is delivered.
- -
- While polymerization is hardly activated at mild temperature conditions in the absence of catalyst, compressors outlet stream temperature does not exceed 120 °C to prevent olefins polymerization.
- -
- Compressors inlet stream is free of carbon particles to prevent cylinder lubrication removal and excessive wear [21]. Moreover, neither condensates, nor liquids are contained to avoid major compressor damages (i.e., slug flow, carryover from interstage coolers and flow changes [27]) and valves failure [28]).
- -
- Liquified shale gas of −162 °C is used as cooling medium, which turns to the gas phase after heat exchange with the hot streams. To maintain sufficient heat exchange rates, temperatures <−125 °C are never reached.
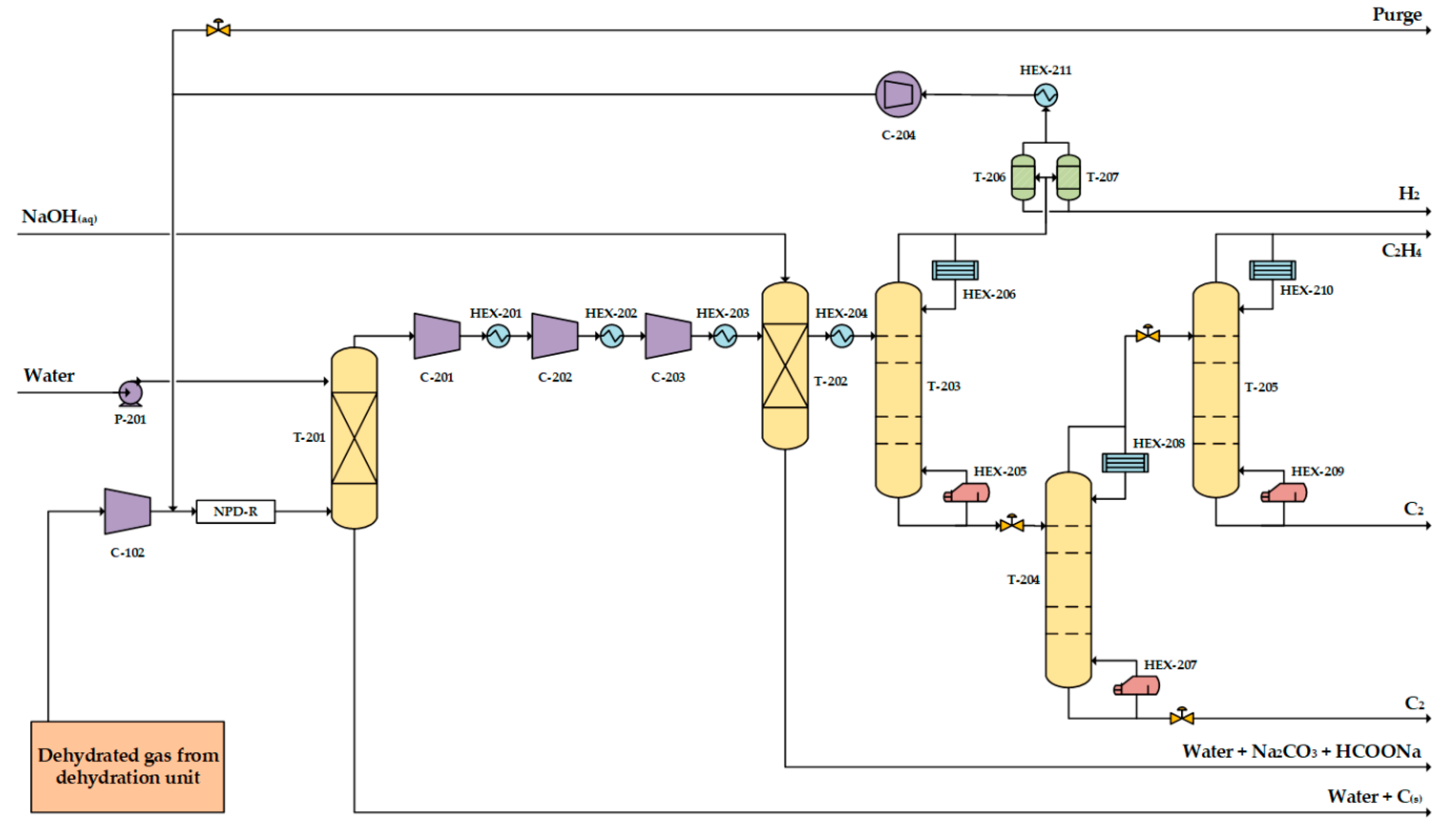
3.2. One-Step Process
3.2.1. Process Design of the One-Step Process
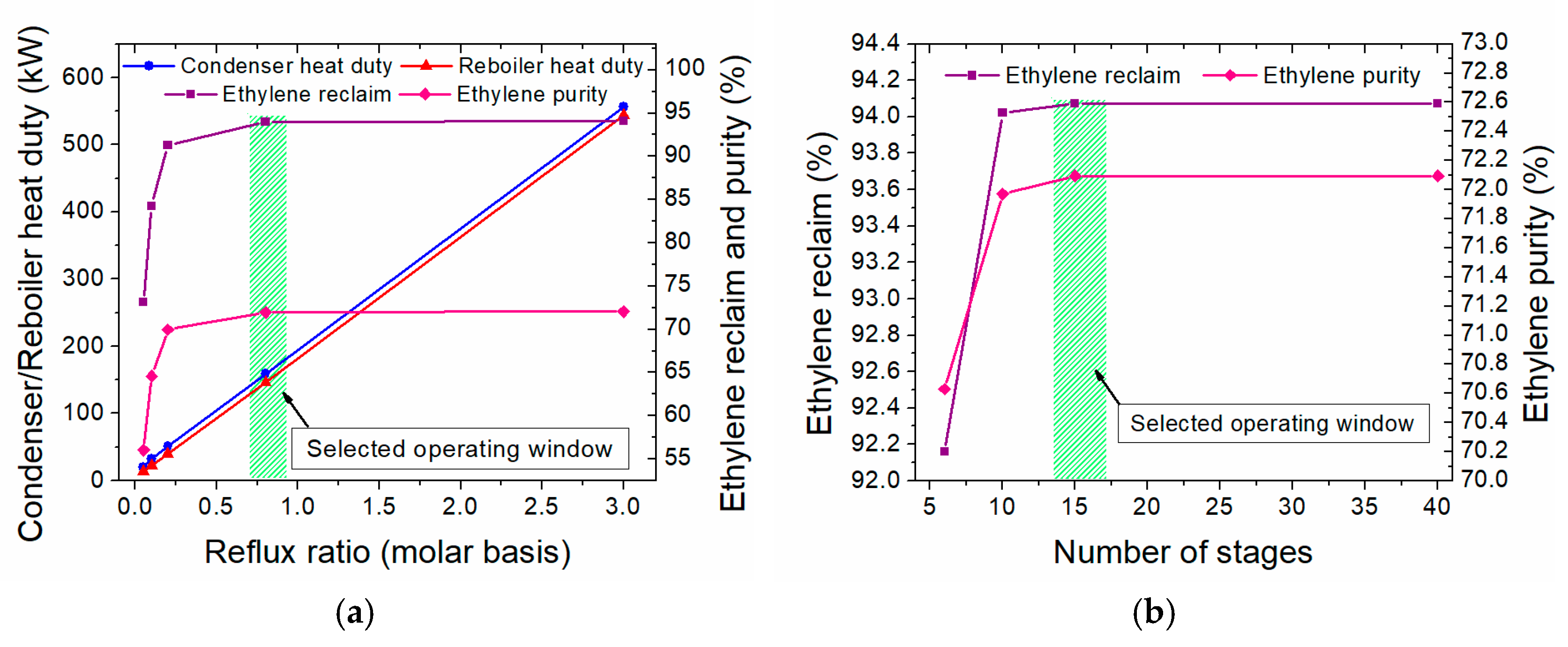
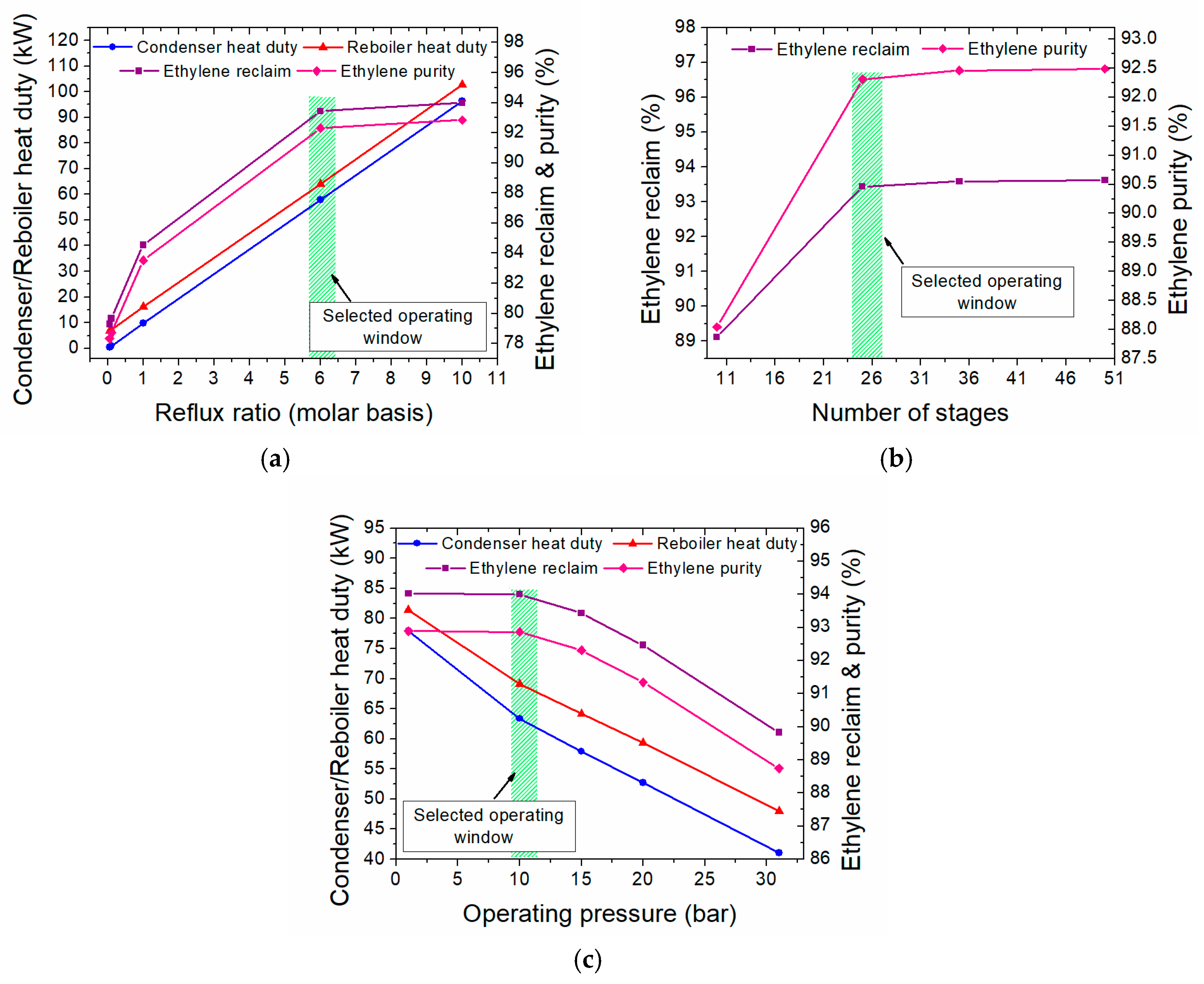
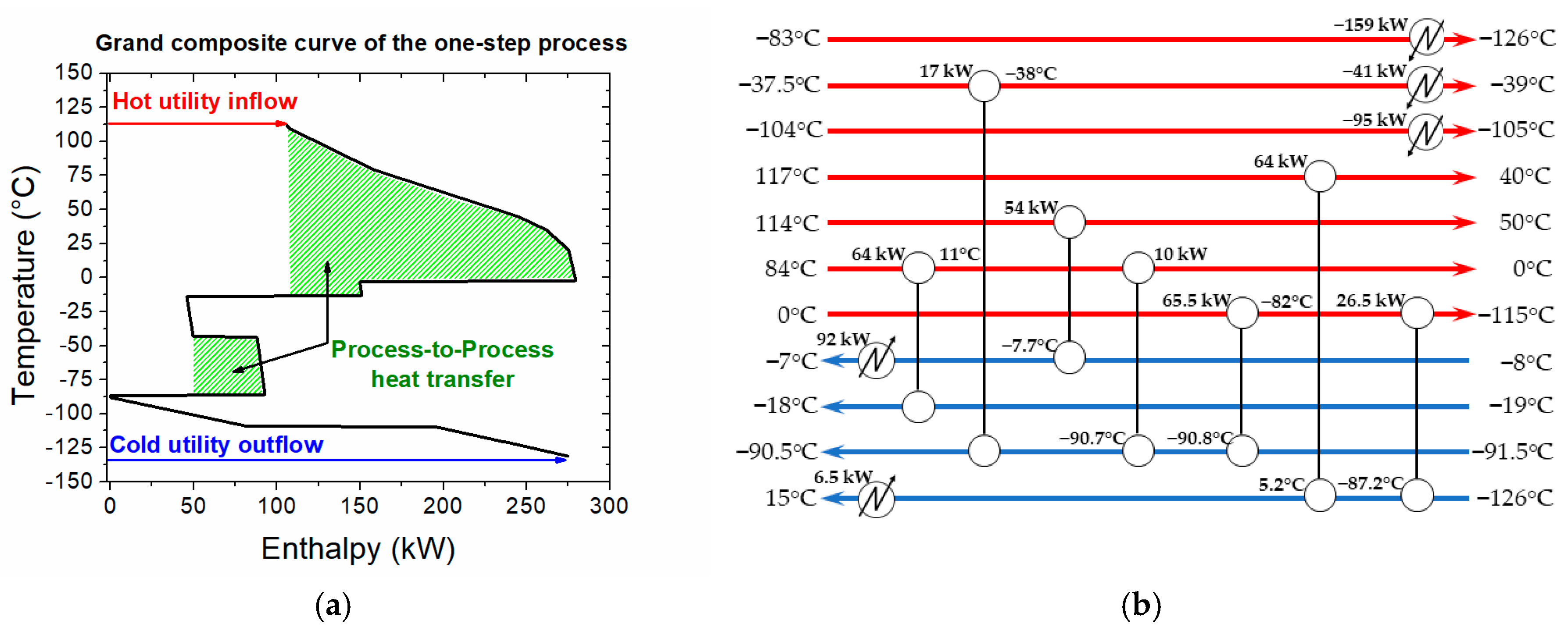
3.2.2. Heat Integration in the One-Step Process
- -
- The minimum temperature difference (ΔΤmin) is set at 10 °C
- -
- The streams enthalpy is based on the energy balances obtained by the process simulation.
- -
- For the streams in which phase change occurs (i.e., condensers and reboilers), a pseudo CPM is calculated, accounting for both the latent and sensible heat; for the latter, a temperature difference of 1 °C is always considered.
3.3. Two-Step Process
3.3.1. Process Design of the Two-Step Process
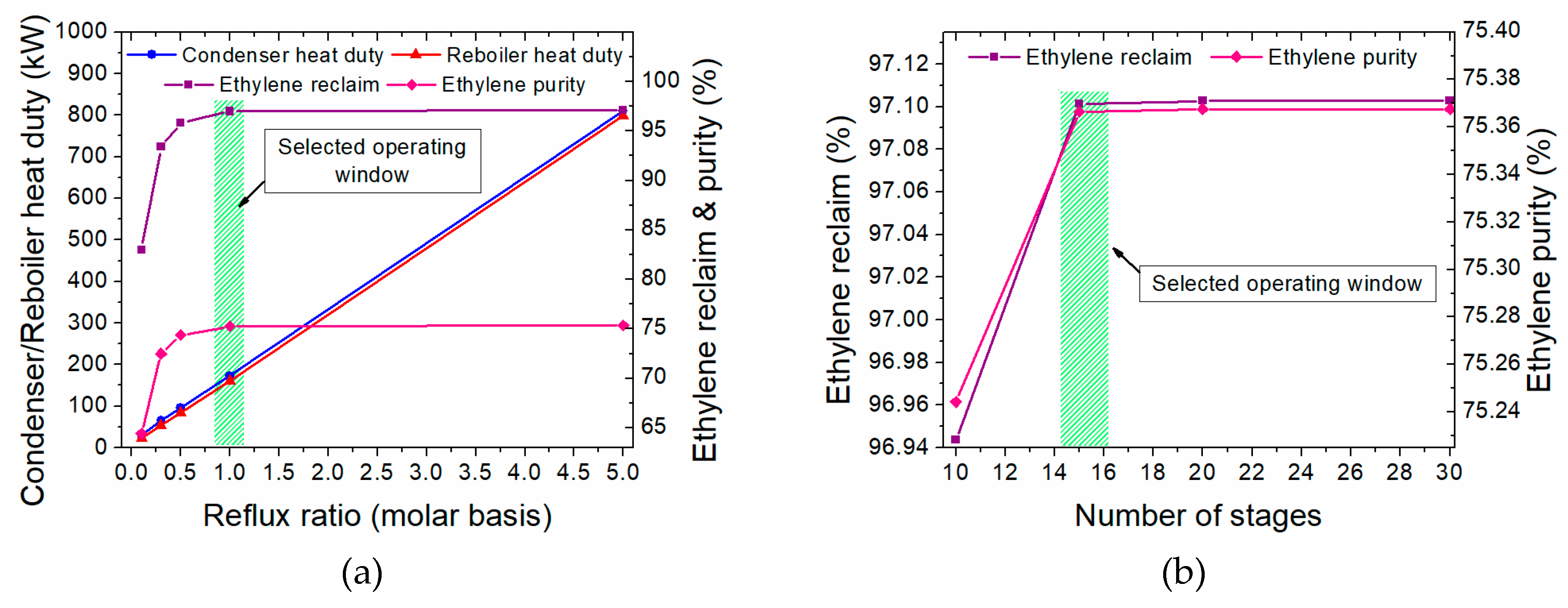
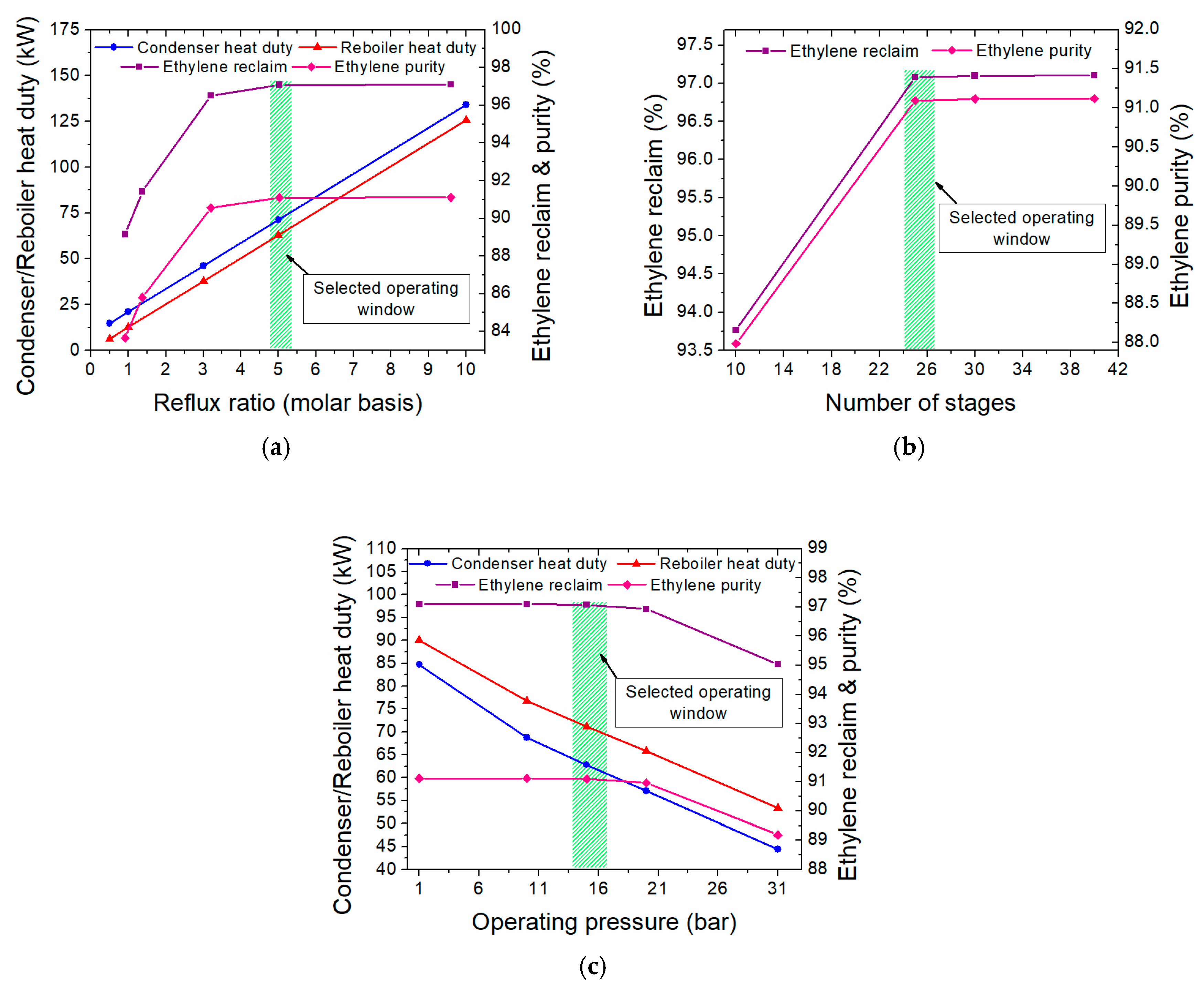
3.3.2. Heat Integration of the Two-Step Process
4. Evaluation of the Plasma-Assisted Process Alternatives
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Glossary
| HV | High voltage | GE | Ground electrode |
| GHGs | Greenhouse gas emissions | NPD | Nanosecond pulsed discharge |
| NGLs | Natural gas liquids | PR | Peng-Robinson |
| TEG | Triethylene glycol | PFD | Process flow diagram |
| NPD-R | Nanosecond pulsed discharge reactor | PSA | Pressure swing adsorption |
| HEX | Heat exchanger | KPIs | Key performance indicators |
| ΔΤmin | Minimum temperature difference | CPM | Specific heat capacity content |
| VLE | Vapor-liquid equilibrium | EOS | Equation of state |
References
- Convention on Climate Change: Climate Agreement of Paris; United Nations: Paris, France, 2015; pp. 1–27.
- Gao, Y.; Gao, X.; Zhang, X. The 2 °C Global Temperature Target and the Evolution of the Long-Term Goal of Addressing Climate Change—From the United Nations Framework Convention on Climate Change to the Paris Agreement. Engineering 2017, 3, 272–278. [Google Scholar] [CrossRef]
- De Pee, A.; Pinner, D.; Roelofsen, O.; Somers, K.; Speelman, E.; Witteveen, M. Decarbonization of Industrial Sectors: The Next Frontier; McKinsey & Company: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Schiffer, Z.J.; Manthiram, K. Electrification and Decarbonization of the Chemical Industry. Joule 2017, 1, 10–14. [Google Scholar] [CrossRef]
- Edenhofer, O.; Pichs-Madruga, R.; Sokona, Y.; Farahani, E.; Kadner, S.; Seyboth, K.; Adler, A.; Baum, I.; Brunner, S.; Eickemeier, P. (Eds.) IPCC, 2014: Climate Change 2014: Mitigation of Climate Change. Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014; ISBN 9780444627483. [Google Scholar]
- Lechtenböhmer, S.; Nilsson, L.J.; Åhman, M.; Schneider, C. Decarbonising the energy intensive basic materials industry through electrification—Implications for future EU electricity demand. Energy 2016, 115, 1623–1631. [Google Scholar] [CrossRef]
- Bataille, C.; Åhman, M.; Neuhoff, K.; Nilsson, L.J.; Fischedick, M.; Lechtenböhmer, S.; Solano-Rodriquez, B.; Denis-Ryan, A.; Stiebert, S.; Waisman, H.; et al. A review of technology and policy deep decarbonization pathway options for making energy-intensive industry production consistent with the Paris Agreement. J. Clean. Prod. 2018, 187, 960–973. [Google Scholar] [CrossRef]
- Wesseling, J.H.; Lechtenböhmer, S.; Åhman, M.; Nilsson, L.J.; Worrell, E.; Coenen, L. The transition of energy intensive processing industries towards deep decarbonization: Characteristics and implications for future research. Renew. Sustain. Energy Rev. 2017, 79, 1303–1313. [Google Scholar] [CrossRef]
- Åhman, M.; Nilsson, L.J.; Johansson, B. Global climate policy and deep decarbonization of energy-intensive industries. Clim. Policy 2017, 17, 634–649. [Google Scholar] [CrossRef]
- Scapinello, M.; Delikonstantis, E.; Stefanidis, G.D. The panorama of plasma-assisted non-oxidative methane reforming. Chem. Eng. Process. Process Intensif. 2016, 117, 120–140. [Google Scholar] [CrossRef]
- Scapinello, M.; Delikonstantis, E.; Stefanidis, G.D. Direct methane-to-ethylene conversion in a nanosecond pulsed discharge. Fuel 2018, 222, 705–710. [Google Scholar] [CrossRef]
- Delikonstantis, E.; Scapinello, M.; Stefanidis, G.D. Low energy cost conversion of methane to ethylene in a hybrid plasma-catalytic reactor system. Fuel Process. Technol. 2018, 176, 33–42. [Google Scholar] [CrossRef]
- Van Rooij, G.J.; Akse, H.N.; Bongers, W.A.; Van De Sanden, M.C.M. Plasma for electrification of chemical industry: A case study on CO2 reduction. Plasma Phys. Control. Fusion 2018, 60, 014019. [Google Scholar] [CrossRef]
- Anastasopoulou, A.; Wang, Q.; Hessel, V.; Lang, J. Energy Considerations for Plasma-Assisted N-Fixation Reactions. Processes 2014, 2, 694–710. [Google Scholar] [CrossRef]
- Anastasopoulou, A.; Butala, S.; Patil, B.; Suberu, J.; Fregene, M.; Lang, J.; Wang, Q.; Hessel, V. Techno-Economic Feasibility Study of Renewable Power Systems for a Small-Scale Plasma-Assisted Nitric Acid Plant in Africa. Processes 2016, 4, 54. [Google Scholar] [CrossRef]
- Delikonstantis, E.; Scapinello, M.; Stefanidis, G.D. Investigating the plasma-assisted and thermal catalytic dry methane reforming for syngas production: Process design, simulation and evaluation. Energies 2017, 10, 1429. [Google Scholar] [CrossRef]
- Scapinello, M.; Martini, L.M.; Dilecce, G.; Tosi, P. Conversion of CH4/CO2 by a nanosecond repetitively pulsed discharge. J. Phys. D Appl. Phys 2016, 49, 075602. [Google Scholar] [CrossRef]
- Scapinello, M.; Delikonstantis, E.; Stefanidis, G.D. A study on the reaction mechanism of non-oxidative methane coupling in a nanosecond pulsed discharge reactor using isotope analysis. Chem. Eng. J. 2019, 360, 64–74. [Google Scholar] [CrossRef]
- Ravasio, S.; Cavallotti, C. Analysis of reactivity and energy efficiency of methane conversion through non thermal plasmas. Chem. Eng. Sci. 2012, 84, 580–590. [Google Scholar] [CrossRef]
- Bullin, K.; Krouskop, P. Composition Variety Complicates Processing Plans for US Shale Gas. In Proceedings of the Annual Forum; Gas Processors Association: Houston, TX, USA, 2008. [Google Scholar]
- Engineering Data Book, 13th ed.; Gas Processors Suppliers Association: Tulsa, Oklahoma, 2012.
- Laurenzi, I.J.; Jersey, G.R. Life cycle greenhouse gas emissions and freshwater consumption of marcellus shale gas. Environ. Sci. Technol. 2013, 47, 4896–4903. [Google Scholar] [CrossRef] [PubMed]
- Gusev, A.V.; Kornev, R.A.; Sukhanov, A.Y. Behavior of carbon-containing impurities during plasma synthesis of trichlorosilane. High Energy Chem. 2008, 42, 56–58. [Google Scholar] [CrossRef]
- Ashour, I.; Al-Rawahi, N.; Fatemi, A.; Vakili-Nezhaad, G. Applications of Equations of State in the Oil and Gas Industry. In Thermodynamics-Kinetics of Dynamic Systems; InTech: Rijeka, Croatia, 2011; pp. 165–178. ISBN 978-953-307-627-0. [Google Scholar]
- Kogelschatz, U. Dielectric-barrier discharges: Their History, Discharge Physics, and Industrial Applications. Plasma Chem. Plasma Process. 2003, 23, 1–46. [Google Scholar] [CrossRef]
- Petrochemical Industry Ethylene Plant. Process Gas Chromatography Application Note; SiemensAG: Karlsruhe, Germany, 2016; pp. 1–7.
- Bloch, H.P.; Hoefner, J.J. Reciprocating Compressors: Operation and Maintenance; Gulf Professional Publishing Co.: Houston, TX, USA, 1996. [Google Scholar]
- Bauer, F. The Influence of Liquids on Compressor Valves. Int. Compress. Eng. Conf. 1990, 647–653. [Google Scholar]
- Hoffman, A.; Gustaf, O.; Andreas, L. Shale Gas and Hydraulic Fracturing: Framing the Water Issue; Stockholm International Water Institute: Stockholm, Sweden, 2014; Volume 34, ISBN 9789198186017. [Google Scholar]
- Olds, R.H.; Sage, B.H.; Lacey, W.N. Phase Equilibria in Hydrocarbon Systems. Composition of the Dew-Point Gas of the Methane-Water System. Ind. Eng. Chem. 1942, 34, 1223–1227. [Google Scholar] [CrossRef]
- Petropoulou, E.G.; Voutsas, E.C. Thermodynamic Modeling and Simulation of Natural Gas Dehydration Using Triethylene Glycol with the UMR-PRU Model. Ind. Eng. Chem. Res. 2018, 57, 8584–8604. [Google Scholar] [CrossRef]
- Smith, R.M. Chemical Process: Design and Integration; John Wiley & Sons Ltd.: Chichester, UK, 2005; ISBN 0470011912. [Google Scholar]
- Zimmermann, H.; Walzl, R. Ethylene. Ullmann’s Encycl. Ind. Chem. 2012, 13, 62. [Google Scholar]
- Mamrosh, D.L.; McIntush, K.E.; Fisher, K. Caustic Scrubber Designs for H2S Removal from Refinery Gas Streams. In 2014 AFPM Annual Meeting; American Fuel & Petrochemical Manufacturers: Orlando, FL, USA, 2014; pp. 1–26. [Google Scholar]
- Boswell, M.C.; Dickson, J.V. The action of sodium hydroxide on carbon monoxide, sodium formate and sodium oxalate. J. Am. Chem. Soc. 1918, 40, 1779–1786. [Google Scholar] [CrossRef]
- Zhao, P.; Zhang, G.; Sun, Y.; Xu, Y. A review of oxygen removal from oxygen-bearing coal-mine methane. Environ. Sci. Pollut. Res. 2017, 24, 15240–15253. [Google Scholar] [CrossRef] [PubMed]
- Moon, D.K.; Kim, Y.H.; Ahn, H.; Lee, C.H. Pressure swing adsorption process for recovering H2 from the effluent gas of a melting incinerator. Ind. Eng. Chem. Res. 2014, 53, 15447–15455. [Google Scholar] [CrossRef]
- Li, X.; Li, J.; Yang, B. Design and control of the cryogenic distillation process for purification of synthetic natural gas from methanation of coke oven gas. Ind. Eng. Chem. Res. 2014, 53, 19583–19593. [Google Scholar] [CrossRef]
- Luyben, W.L. Optimum product recovery in chemical process design. Ind. Eng. Chem. Res. 2014, 53, 16044–16050. [Google Scholar] [CrossRef]
- Ren, T.; Patel, M.; Blok, K. Olefins from conventional and heavy feedstocks: Energy use in steam cracking and alternative processes. Energy 2006, 31, 425–451. [Google Scholar] [CrossRef]
- Salerno, D.; Arellano-Garcia, H.; Wozny, G. Ethylene separation by feed-splitting from light gases. Energy 2011, 36, 4518–4523. [Google Scholar] [CrossRef]
- Salgado, H.J.; Valbuena, G. Technical and economic evaluation of the separation of light olefins (ethylene and propylene) by using π-complexation with silver salts. CT F Cienc. Tecnol. Futuro 2011, 4, 73–88. [Google Scholar]
- Comparing Electricity Prices for Industry. An elusive task—illustrated by the German Case; Agora Energiewende: Berlin, Germany, 2014.
- Ilas, A.; Ralon, P.; Rodriguez, A.; Taylor, M. Renewable Power Generation Costs in 2017; International Renewable Energy Agency: Abu Dhabi, UAE, 2018. [Google Scholar]
- Industrial electricity prices. German Association of the Automotive Industry. Available online: www.vda.de/en/topics/economic-policy-and-infrastructure/energy/industrial-electricity-prices (accessed on 29 January 2019).





| Reaction | Fractional Conversion (%) | |
|---|---|---|
| One-Step | Two-Step | |
| 2CH4 → C2H4 + 2H2 | 12.8 | 17.5 |
| 2CH4 → C2H2 + 3H2 | 0.3 | 0.3 |
| 2CH4 → C2H6 + H2 | 1.8 | 1.2 |
| CH4 → C(s) + 2H2 | 10.3 | 2.8 |
| C2H4 → 2C(s) + 2H2 ¥ | 100.0 | 100.0 |
| C2H6 → C2H4 + H2 | 50.0 | 45.0 |
| CO2 → CO + ⅟2O2 | 20.0 | 20.0 |
| Utility (kW) | Before Heat Integration | After Heat Integration | Utility Saving |
|---|---|---|---|
| Hot utility demand | 400 | 99 | −75% |
| Cold utility demand | −596 | −295 | −51% |
| Utility (kW) | Before Heat Integration | After Heat Integration | Utility Saving |
|---|---|---|---|
| Hot utility demand | 448 | 0 | −100% |
| Cold utility demand | −797 | −348 | −56% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delikonstantis, E.; Scapinello, M.; Stefanidis, G.D. Process Modeling and Evaluation of Plasma-Assisted Ethylene Production from Methane. Processes 2019, 7, 68. https://doi.org/10.3390/pr7020068
Delikonstantis E, Scapinello M, Stefanidis GD. Process Modeling and Evaluation of Plasma-Assisted Ethylene Production from Methane. Processes. 2019; 7(2):68. https://doi.org/10.3390/pr7020068
Chicago/Turabian StyleDelikonstantis, Evangelos, Marco Scapinello, and Georgios D. Stefanidis. 2019. "Process Modeling and Evaluation of Plasma-Assisted Ethylene Production from Methane" Processes 7, no. 2: 68. https://doi.org/10.3390/pr7020068
APA StyleDelikonstantis, E., Scapinello, M., & Stefanidis, G. D. (2019). Process Modeling and Evaluation of Plasma-Assisted Ethylene Production from Methane. Processes, 7(2), 68. https://doi.org/10.3390/pr7020068