Eden Model Simulation of Re-Epithelialization and Angiogenesis of an Epidermal Wound
Abstract
1. Introduction
2. The Eden Model
2.1. Epithelialization Algorithm:
- The size of the simulation cell and the domain where growth occurs is defined for a given wound geometry. Each cell is represented by a grid point on the surface. A grid point can only be occupied by a single cell and is either seeded (occupied) or a growth site (empty).
- Since wounds heal by contraction, grid points on the wound border are delineated and initial seeds are placed along the wound edge. A cell, that is an occupied grid point, can only grow into an immediate neighboring vacant growth site.
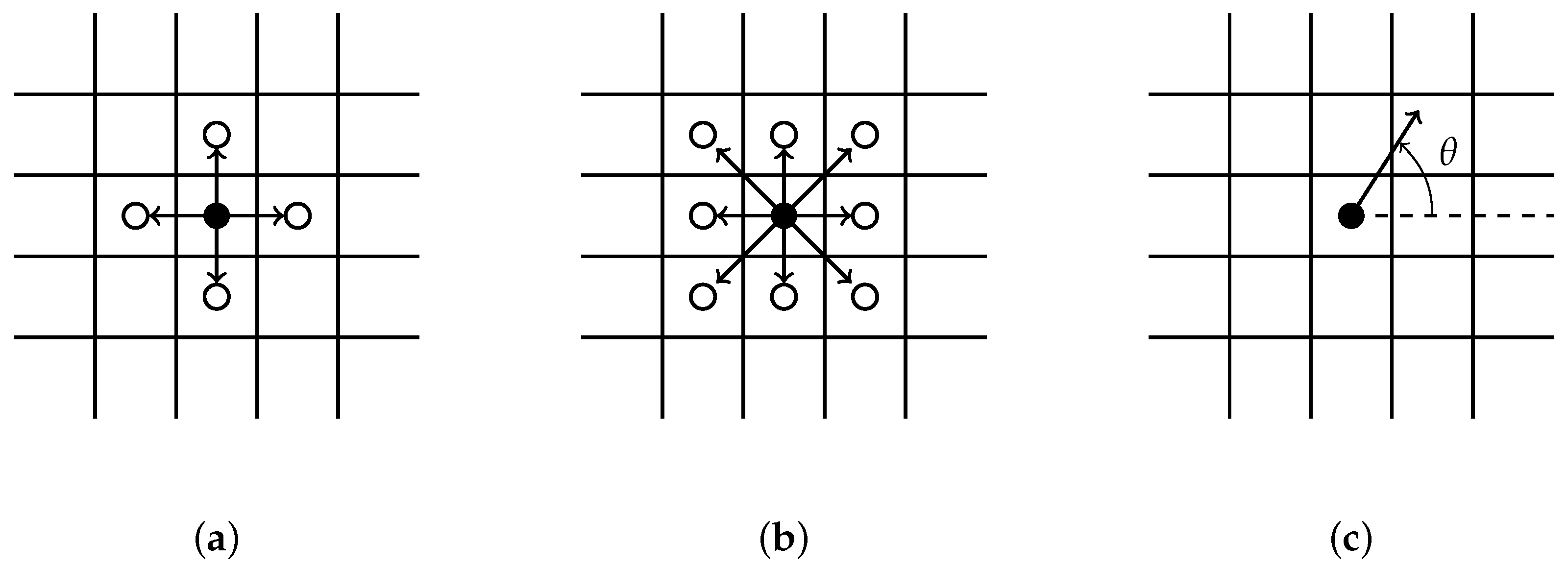
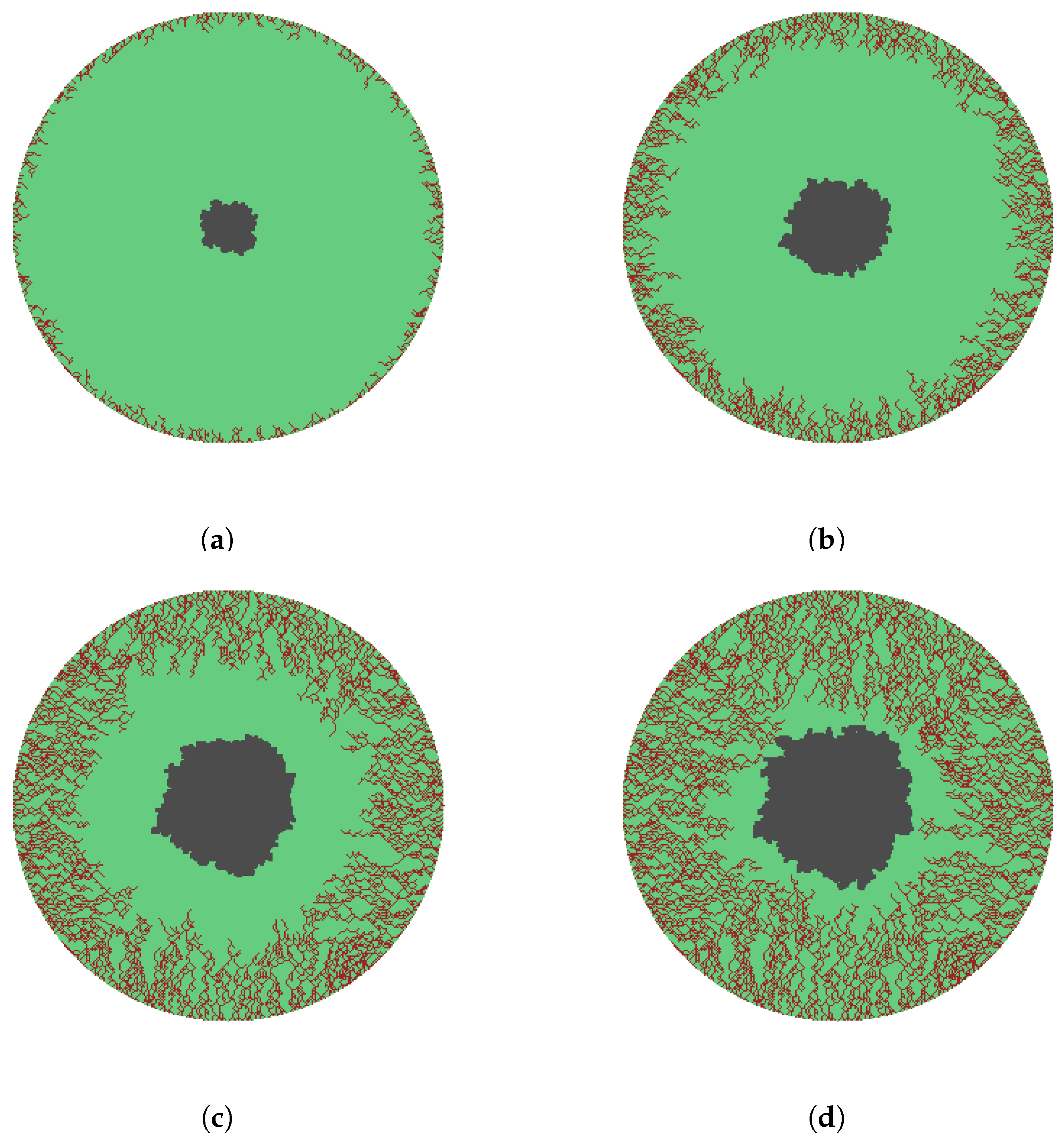
- All clusters originating along the wound edge grow simultaneously into wound space. An occupied grid point for each cluster grows by randomly selecting a growth site from its immediate neighbors. We implement a selection criterion that includes diagonal representation, illustrated by Figure 1b.
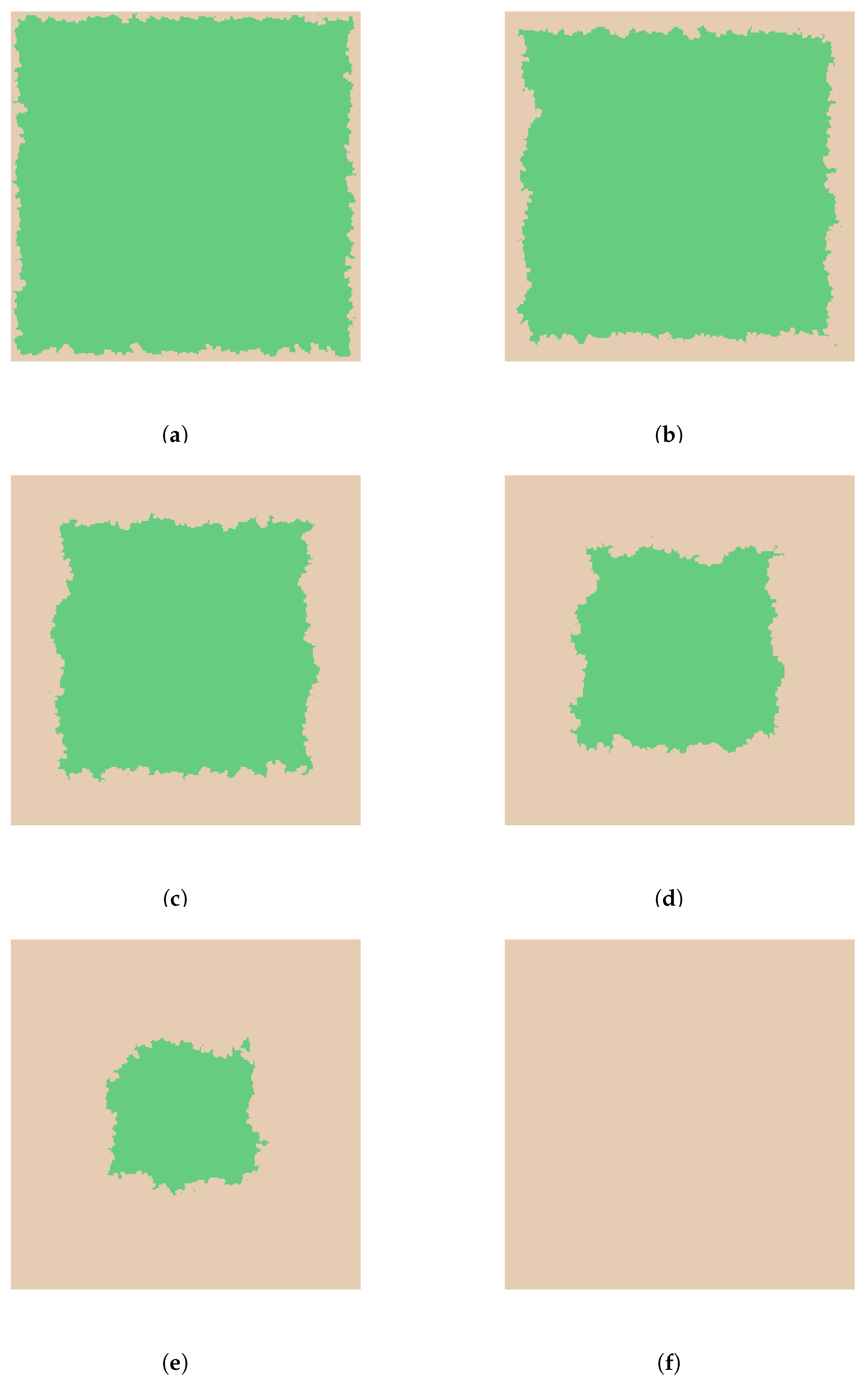
- A cell stops growing in a randomly chosen direction if there is no available immediate growth site. Growth terminates when all possible directions are exhausted. The growth process ends and wound considered re- epithelialized when the entire domain is covered.
2.2. Angiogenesis Algorithm
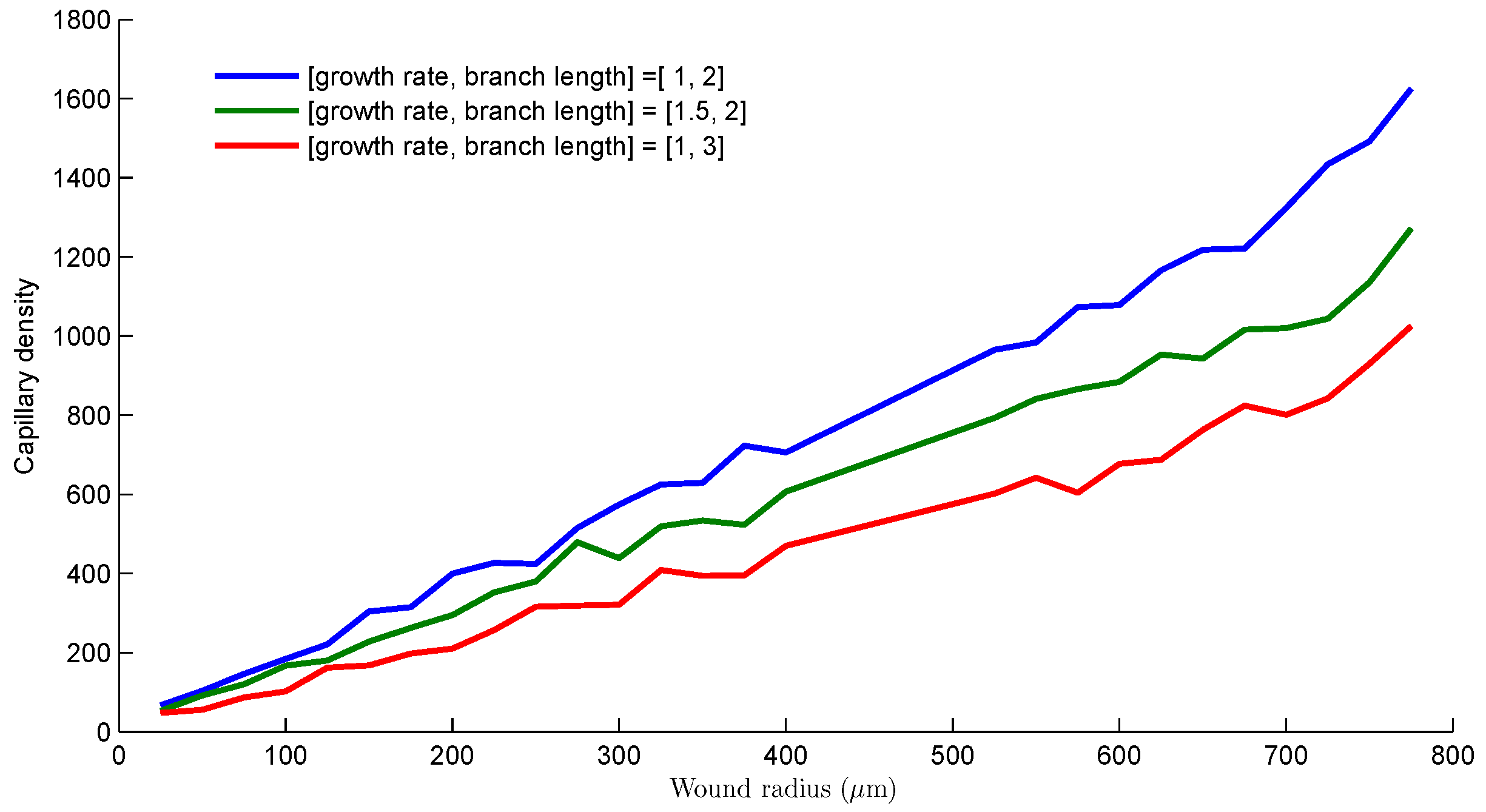
- The size of the simulation cell and the domain where growth occurs is defined for a given wound geometry. The growth rate of sprouts per generation, that is the length (number of grid spaces) is specified. The branching and merging conditions are also specified.
- Initial seeds representing capillary sprouts are placed at prescribed points along the wound border. All sprouts originating along the wound edge grow simultaneously into wound space.
- Each sprout randomly selects a single direction for growth from possible growth sites. Two random growth directions are chosen when conditions for branching are satisfied. A backward growth direction is not allowed.
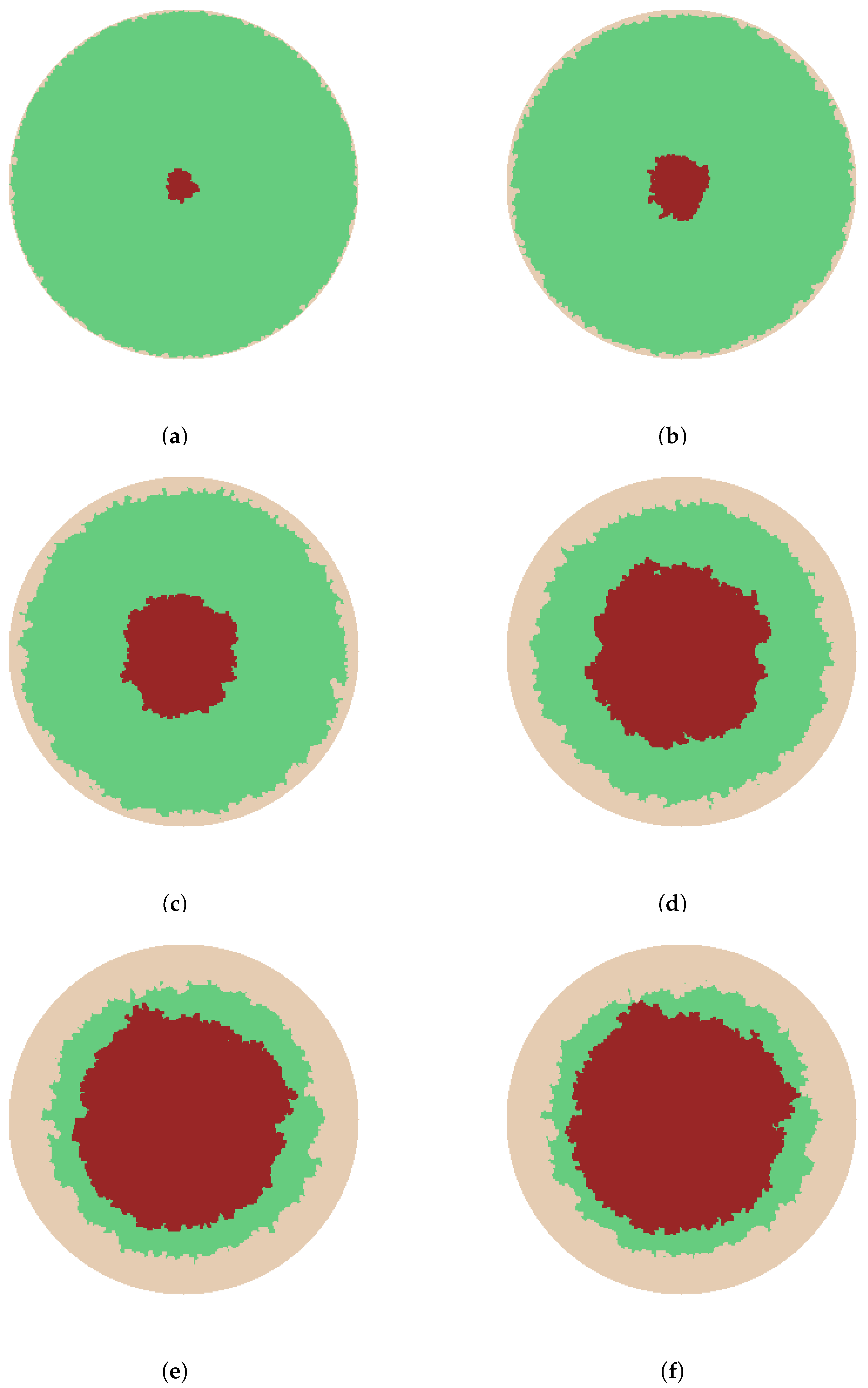
- The sprouts grow by diffusing into the wound space depending on the growth rate, the branching and merging strategies. The vascularization process is completed when sprouts reach the center of the wound.
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A. Psuedocode for Regular Growth
Appendix B. Psuedocode for Capillary Growth
References
- Clark, R.A.F. Overview and general considerations of wound repair. In The Molecular and Cellular Biology of Wound Repair; Clark, R.A.F., Hendson, P.M., Eds.; Plenum: New York, NY, USA, 1988; pp. 3–34. [Google Scholar]
- De Oliveira Gonzalez, A.C.; Costa, T.F.; de Andrade, Z.; Medrado, A.R.A.P. Wound healing—A literature review. Anais Bras. Dermatol. 2016, 91, 614–620. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, S.; Rosowski, E.E.; Huttenlocher, A. Neutrophil migration in infection and wound repair: Going forward in reverse. Nat. Rev. Immunol. 2016, 16, 378–391. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Pamer, E.G. Monocyte recruitment during infection and inflammation. Nat. Rev. Immunol. 2011, 11, 762–774. [Google Scholar] [CrossRef] [PubMed]
- Heng, M.C. Wound healing in adult skin: Aiming for perfect regeneration. Int. J. Dermatol. 2011, 50, 1058. [Google Scholar] [CrossRef] [PubMed]
- Werner, S.; Grose, R. Regulation of wound healing by growth factors and cytokines. Physiol. Rev. 2003, 83, 835. [Google Scholar] [CrossRef] [PubMed]
- Pastar, I.; Stojadinovic, O.; Yin, N.C.; Ramirez, H.; Nusbaum, A.G.; Sawaya, A.; Patel, S.B.; Khalid, L.; Isseroff, R.R.; Tomic-Canic, M. Epithelialization in Wound Healing: A Comprehensive Review. Adv. Wound Care 2014, 3, 445–464. [Google Scholar] [CrossRef] [PubMed]
- Bauer, S.M.; Bauer, R.J.; Velazquez, O.C. Angiogenesis, vasculogenesis, and induction of healing in chronic wounds. Vasc. Endovasc. Surg. 2005, 39, 293. [Google Scholar] [CrossRef] [PubMed]
- Banks, R.E.; Forbes, M.A.; Kinsey, S.E.; Stanley, A.; Ingham, E.; Walters, C.; Selby, P.J. Release of the angiogenic cytokine vascular endothelial growth factor (VEGF) from platelets: Significance for VEGF measurements and cancer biology. Br. J. Cancer 1998, 77, 956. [Google Scholar] [CrossRef] [PubMed]
- Eden, M. A Probabilistic Model for Morphogenesis, Chapter 4: Information Networks; Pergamon Press: New York, NY, USA, 1958; pp. 359–370. [Google Scholar]
- Eden, M. A two-dimensional growth process. In Proceedings of the Fourth Berkeley Symposium on Mathematics Statistics and Probability, Berkeley, CA, USA, 20 June–30 July 1960; University of California Press: Berkeley, CA, USA, 1961; Volume 4, pp. 223–239. [Google Scholar]
- Jullien, R.; Botet, R. Surface thickness in the Eden model. Phys. Rev. Lett. 1985, 54, 2055. [Google Scholar] [CrossRef]
- Wang, C.J.; Bassingthwaighte, J.B. Biological Growth on a Surface. Math. Biosci. 1997, 142, 91–106. [Google Scholar] [CrossRef]
- Wagner, G.; Halvorsrud, R.; Meakin, P. Extended Eden model reproduces growth of an acellular slime mold. Phys. Rev. E 1999, 60, 5879–5887. [Google Scholar] [CrossRef]
- Block, M.; Schöll, E.; Drasdo, D. Classifying the expansion kinetics and critical surface dynamics of growing cell populations. Phys. Rev. Lett. 2007, 99, 248101. [Google Scholar] [CrossRef] [PubMed]
- Savakis, A.E.; Maggelakis, S.A. Models of Shrinking Clusters with Applications to Epidermal Wound Healing. Math. Comput. Model. 1997, 25, 1–6. [Google Scholar] [CrossRef]
- Callaghan, T.; Khain, E.; Sander, L.M.; Ziff, R.M. A stochastic model for wound healing. J. Stat. Phys. 2006, 122, 909–924. [Google Scholar] [CrossRef]
- Stern, J.R.; Ziraldo, C.; Vodovotz, Y.; An, G. Agent-based models of wound healing. In Complex Systems and Computational Biology Approaches to Acute Inflammation; Vodovotz, Y., An, G., Eds.; Springer: New York, NY, USA, 2013; pp. 209–228. [Google Scholar]
- Walker, D.C.; Hill, G.; Wood, S.M.; Smallwood, R.H.; Southgate, J. Agent-based computational modeling of wounded epithelial cell monolayers. IEEE Trans. Nanobiosci. 2004, 3, 153–163. [Google Scholar] [CrossRef]
- Kareva, I.; Abou-Slaybi, A.; Dodd, O.; Dashevsky, O.; Klement, G.L. Normal Wound Healing and Tumor Angiogenesis as a Game of Competitive Inhibition. PLoS ONE 2016, 11, e0166655. [Google Scholar] [CrossRef] [PubMed]
- Stern, J.R.; Christley, S.; Zaborina, O.; Alverdy, J.C.; An, G. Integration of TGF-β- and EGFR-based signaling pathways using an agent-based model of epithelial restitution. Wound Repair Regen. 2012, 20, 862–871. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, S.N.; Sanders, J.R. Mathematical models of wound healing and closure: A comprehensive review. Med. Biol. Eng. Comput. 2016, 1297–1316. [Google Scholar] [CrossRef] [PubMed]
- Valero, C.; Javierre, E.; García-Aznr, J.M.; Menzel, A.; Gómez-Benito, M.J. Challenges in the modeling of wound healing mechanisms in soft biological tissues. Ann. Biomed. Eng. 2015, 43, 1654–1665. [Google Scholar] [CrossRef] [PubMed]
- Cochet-Escartin, O.; Ranft, J.; Silberzan, P.; Marcq, P. Border Forces and Friction Control Epithelial Closure Dynamics. Biophys. J. 2014, 106, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Eichler, M.J.; Carlson, M.A. Modeling dermal granulation tissue with the linear fibroblast-populated collagen matrix: A comparison with the round matrix model. J. Dermatol. Sci. 2006, 41, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Ravasio, A.; Cheddadi, I.; Chen, T.; Pereira, T.; Ong, H.T.; Bertocchi, C.; Brugues, A.; Jacinto, A.; Kabla, A.J.; Toyama, Y.; et al. Gap geometry dictates epithelial closure efficiency. Nat. Commun. 2015, 6, 7683. [Google Scholar] [CrossRef] [PubMed]
- Flegg, J.A.; Menon, S.N.; Maini, P.K.; SeanMcElwain, D.L. On the mathematical modeling of wound healing angiogenesis in skin as areaction-transport process. Front. Physiol. 2015, 6, 262. [Google Scholar] [CrossRef] [PubMed]
- Li, D.S.; Zimmermann, J.; Levine, H. Modeling closure of circular wounds through coordinated collective motion. Phys. Biol. 2016, 13, 016006. [Google Scholar] [CrossRef] [PubMed]
- BenAmar, M.; Wu, M. Re-epithelialization:advancing epithelium frontier during wound healing. J. R. Soc. Interface 2014, 11, 20131038. [Google Scholar] [CrossRef] [PubMed]
- Arciero, J.C.; Mi, Q.; Branca, M.F.; Hackman, D.J.; Swigon, D. Continuum Model of Collective Cell Migration in Wound Healing and Colony Expansion. Biophys. J. 2011, 100, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Zahouani, H.; Assoul, M.; Janod, P.; Mignot, J. Theoretical and experimental study of wound healing: Application to leg ulcers. Med. Biol. Eng. Comput. 1992, 30, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Agyingi, E.; Maggelakis, S.; Ross, D. The effect of bacteria on epidermal wound healing. Math. Model. Nat. Phenom. 2010, 5, 28–39. [Google Scholar] [CrossRef]
- Agyingi, E.; Ross, D.; Maggelakis, S. Modeling the effect of topical oxygen therapy on wound healing. AIP Conf. Proc. 2011, 1368, 159. [Google Scholar]
- Gaffney, E.A.; Pugh, K.; Maini, P.K.; Arnold, F. Investigating a simple model of cutaneous wound healing angiogenesis. J. Math. Biol. 2002, 45, 337–374. [Google Scholar] [CrossRef] [PubMed]
- Schugart, R.C.; Friedman, A.; Zhao, R.; Sen, C.K. Wound angiogenesis as a function of tissue oxygen tension: a mathematical model. Proc. Natl. Acad. Sci. USA 2008, 105, 2628–2633. [Google Scholar] [CrossRef] [PubMed]
- Xue, C.; Friedman, A.; Sen, C.K. A mathematical model of ischemic cutaneous wounds. Proc. Natl. Acad. Sci. USA 2009, 106, 16782–16787. [Google Scholar] [CrossRef] [PubMed]
- Machado, M.J.; Watson, M.G.; Devlin, A.H.; Chaplain, M.A.; McDougall, S.R.; Mitchell, C.A. Dynamics of angiogenesis during wound healing: A coupled In Vivo and In Silico study. Mircocirculation 2011, 18, 183–197. [Google Scholar] [CrossRef] [PubMed]
- Valero, C.; Javierre, E.; García-Aznr, J.M.; Gómez-Benito, M.J. Numerical modelling of the angiogenesis process in wound contraction. Biomech. Model. Mechanobiol. 2012, 12, 349–360. [Google Scholar] [CrossRef] [PubMed]






| Bacteria Growth Rate | Amount of Epithelialization | Amount of Bacteria |
|---|---|---|
| half that of epithelialization growth rate | 53% | 27% |
| equal to the epithelialization growth rate | 42% | 41% |
| one and half times epithelialization growth rate | 34% | 46% |
| twice that of epithelialization growth rate | 30% | 53% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agyingi, E.; Wakabayashi, L.; Wiandt, T.; Maggelakis, S. Eden Model Simulation of Re-Epithelialization and Angiogenesis of an Epidermal Wound. Processes 2018, 6, 207. https://doi.org/10.3390/pr6110207
Agyingi E, Wakabayashi L, Wiandt T, Maggelakis S. Eden Model Simulation of Re-Epithelialization and Angiogenesis of an Epidermal Wound. Processes. 2018; 6(11):207. https://doi.org/10.3390/pr6110207
Chicago/Turabian StyleAgyingi, Ephraim, Luke Wakabayashi, Tamas Wiandt, and Sophia Maggelakis. 2018. "Eden Model Simulation of Re-Epithelialization and Angiogenesis of an Epidermal Wound" Processes 6, no. 11: 207. https://doi.org/10.3390/pr6110207
APA StyleAgyingi, E., Wakabayashi, L., Wiandt, T., & Maggelakis, S. (2018). Eden Model Simulation of Re-Epithelialization and Angiogenesis of an Epidermal Wound. Processes, 6(11), 207. https://doi.org/10.3390/pr6110207




