Natural Products Extraction of the Future—Sustainable Manufacturing Solutions for Societal Needs
Abstract
1. Introduction
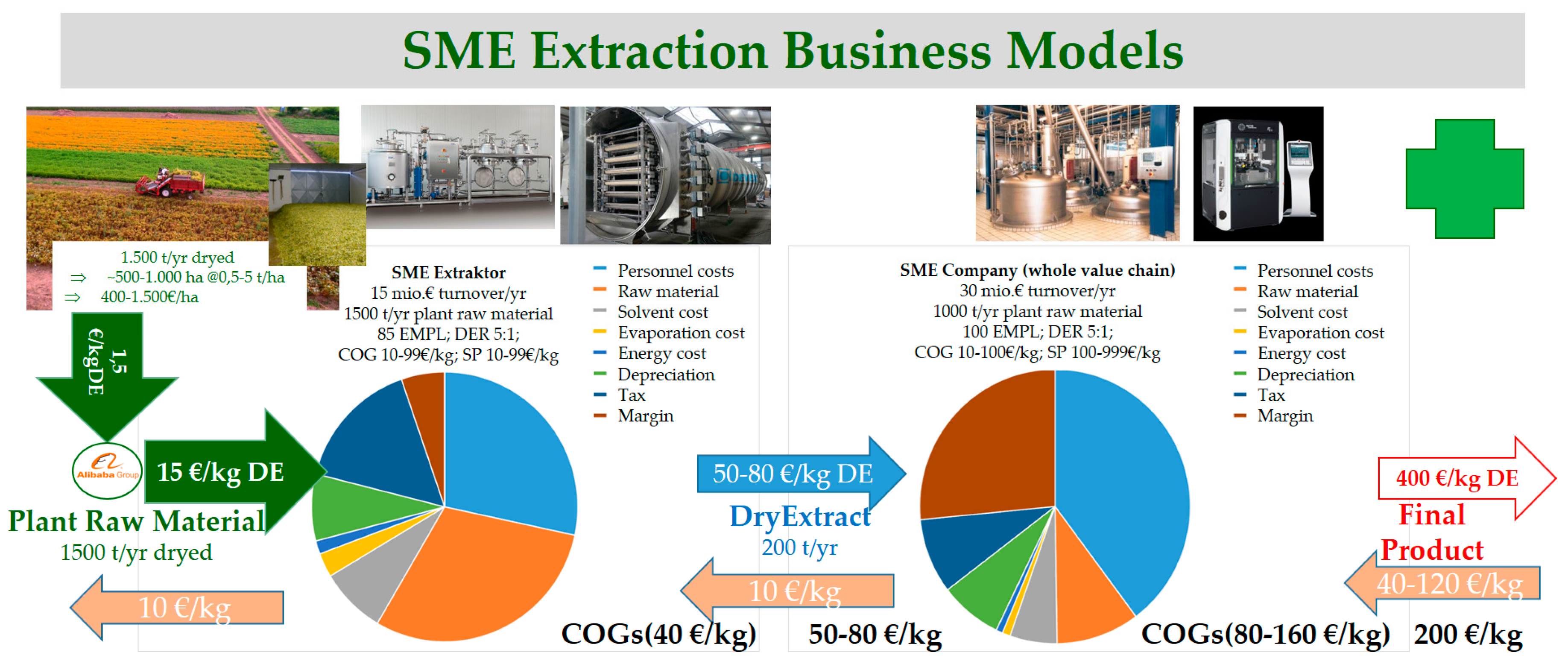
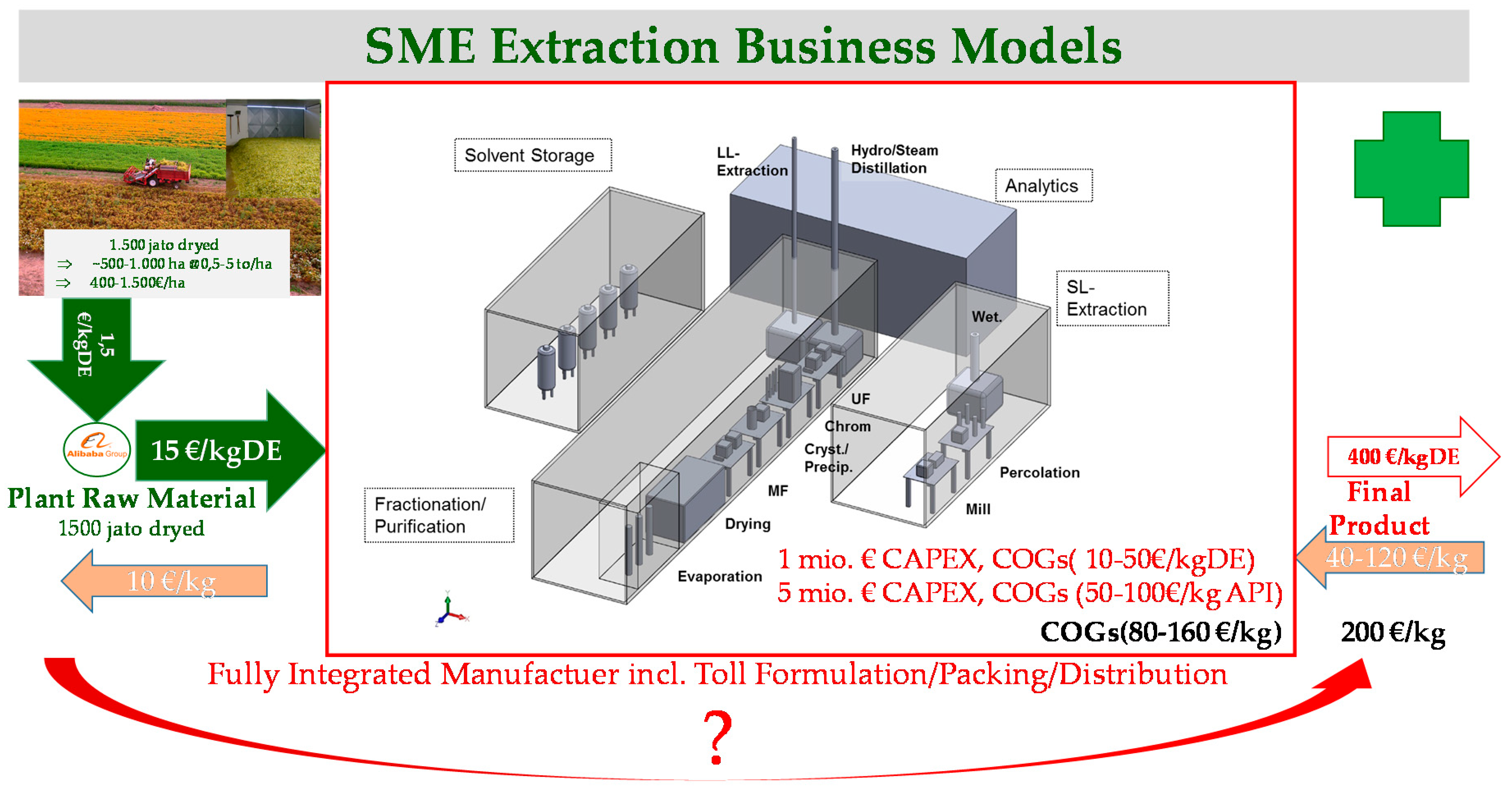
2. Products and Business Models
3. Regulatory of Herbal Products
- Registration Category 1: An active ingredient obtained from herbal raw, animal or mineral materials and its preparations that have not been marketed in China.
- Registration Category 2: A newly-discovered Chinese crude drug and its preparations.
- Registration Category 3: A new substitute for Chinese crude drug.
- Registration Category 4: A new part for medicinal use from currently-used Chinese crude drugs and their preparations.
- Registration Category 5: Active fraction(s) extracted from herbal raw, animal or mineral materials and its preparations that have not been marketed in China.
- Registration Category 6: A combination preparation of TCM or natural medicinal product, which has not been marketed in China.
- Registration Category 7: A preparation with changed administration route of a marketed TCM or natural medicinal product.
- Registration Category 8: A preparation with changed dosage form of a marketed TCM or natural medicinal product.
- Registration Category 9: Generic TCMs or natural medicinal products
- Factor 10–100 larger scale of production compared to pharma,
- regulated environment, but more freedom regarding extraction process,
- semi-purified products
- Factor 100–1000 larger scale of production compared to pharma,
- regulated environment regarding quality, but more freedom regarding extraction process,
- semi-purified products
- Pharma: Regulations specify the origin of herbal raw material; most resources have to be collected from natural habitats, which can problematic because of higher natural variability, environmental impact, risks in supply chain management etc.
- Nutritional supplements, herbicides, and crop protection: Cultivated herbal raw material is preferred due to advantages regarding quality, logistics, and secured supply chains.
4. Manufacturing Operation of Extracts
- Analytical fingerprint, characterization, lead substances definition
- Efficacy study of lead substances range
- QbD approach to determine operation parameter and
- Submission of Design Space, i.e., new approval
- Economic optimal operation at maximal therapeutic value product
- Analytical quantification (not characterization)
- Lead efficacy substance value (not range) i.e., efficacy studies
- Design Space by QbD approach much narrow i.e., robust process i.e., reliable product
- New approval
- The steps described above, with efficacy based on single substances or well characterized substance groups
- Purification process development
- New approval
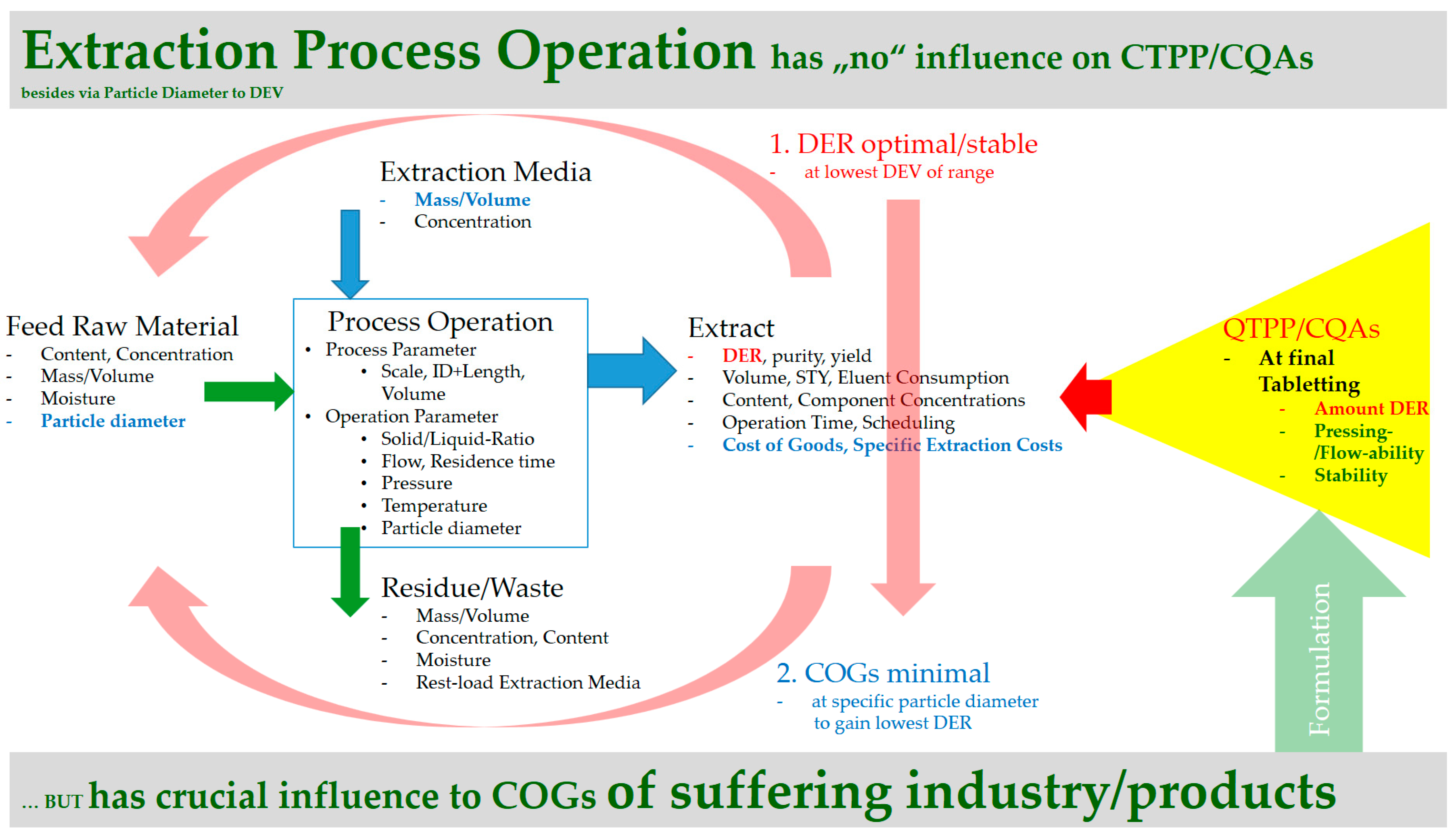
5. Process Design Proposal for Efficient Manufacturing
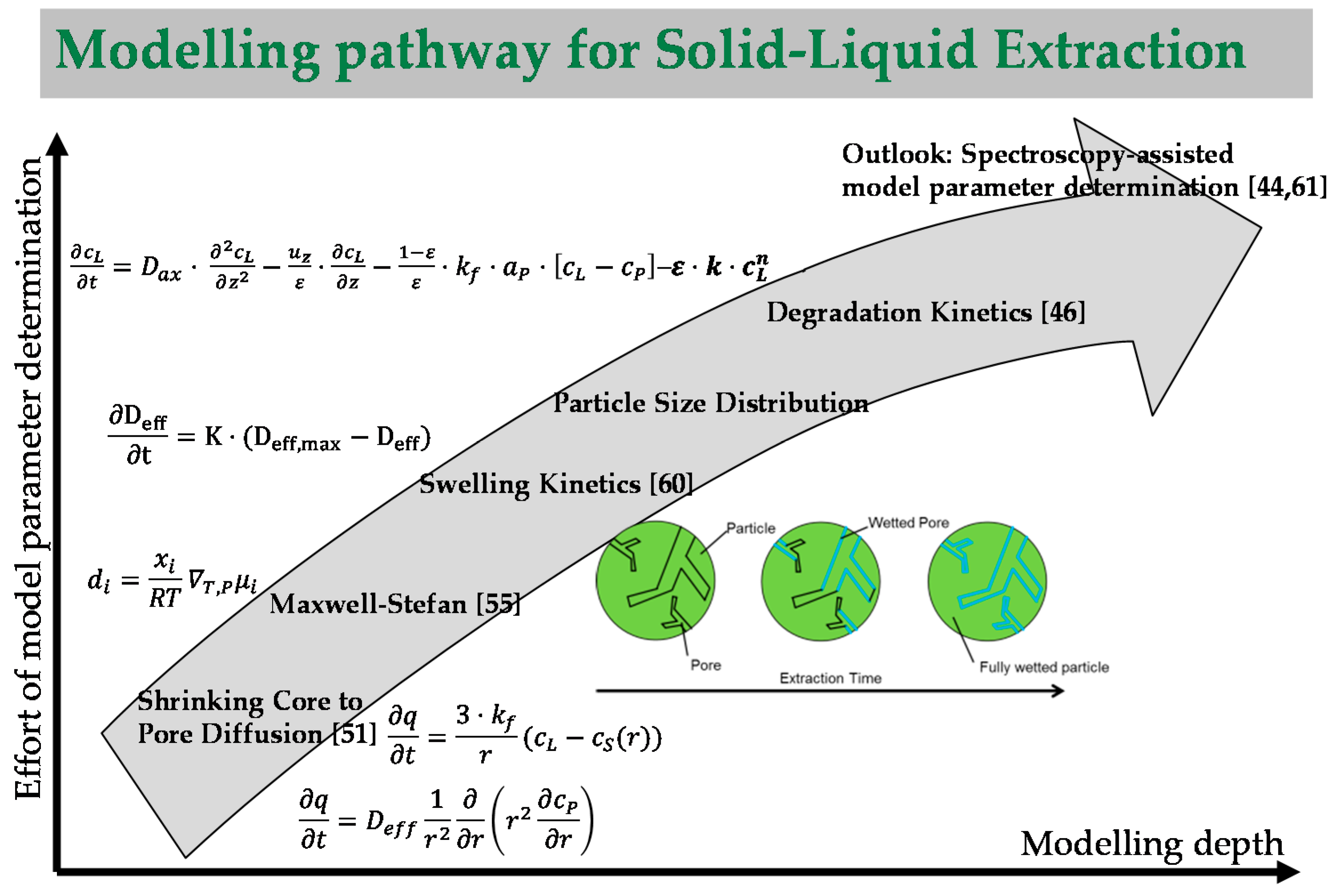
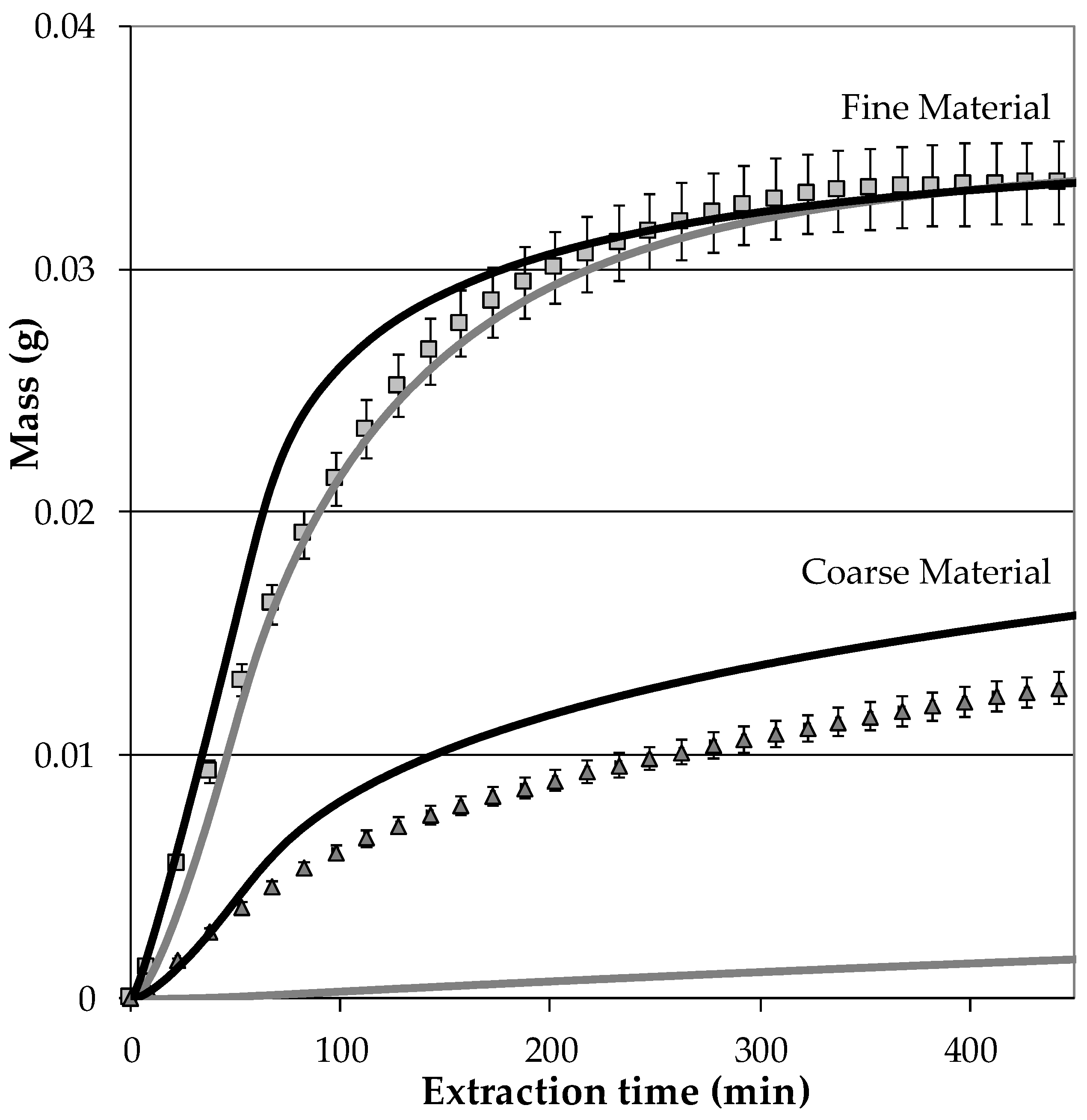
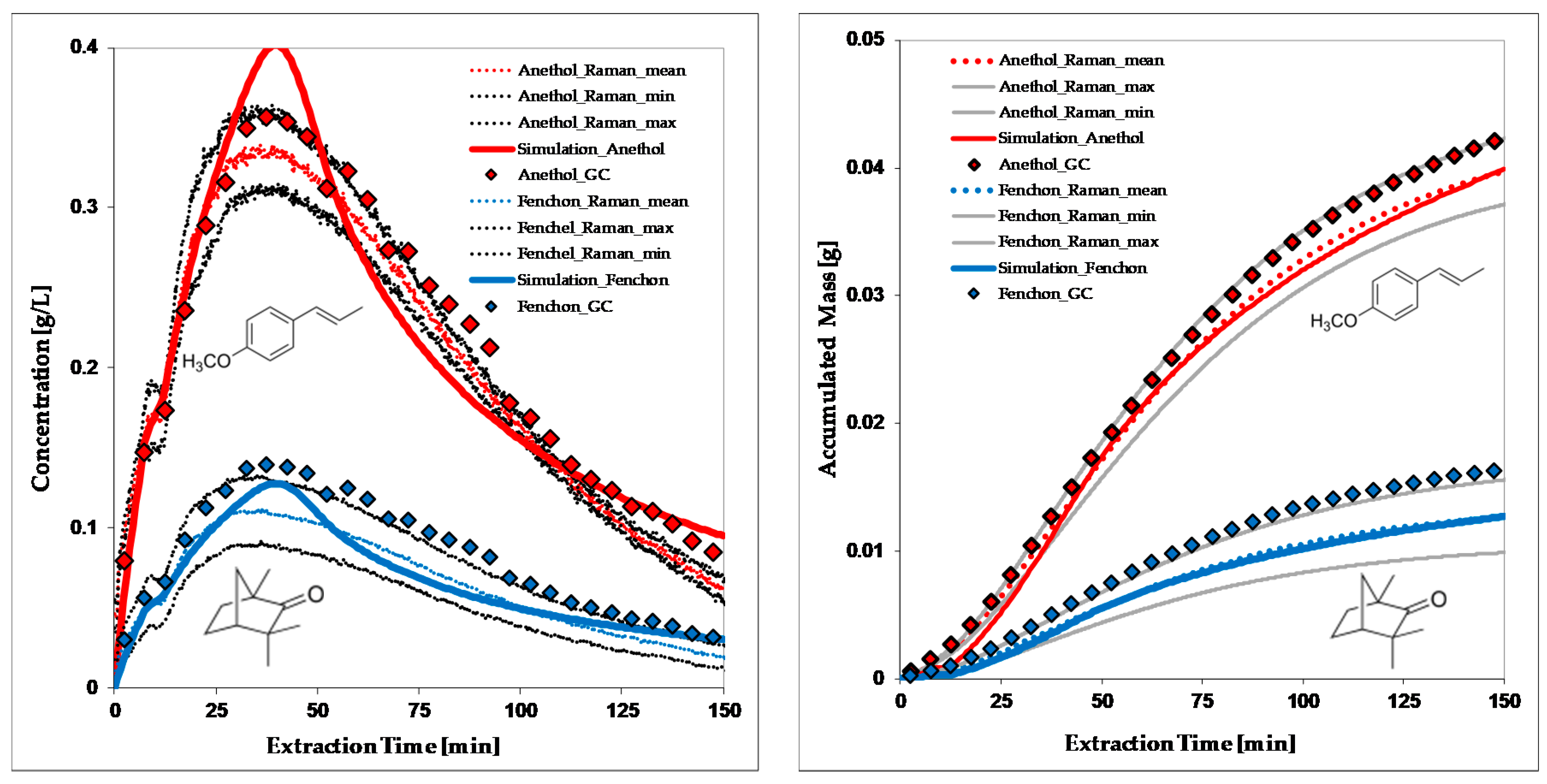
5.1. Modelling of the Extraction Process
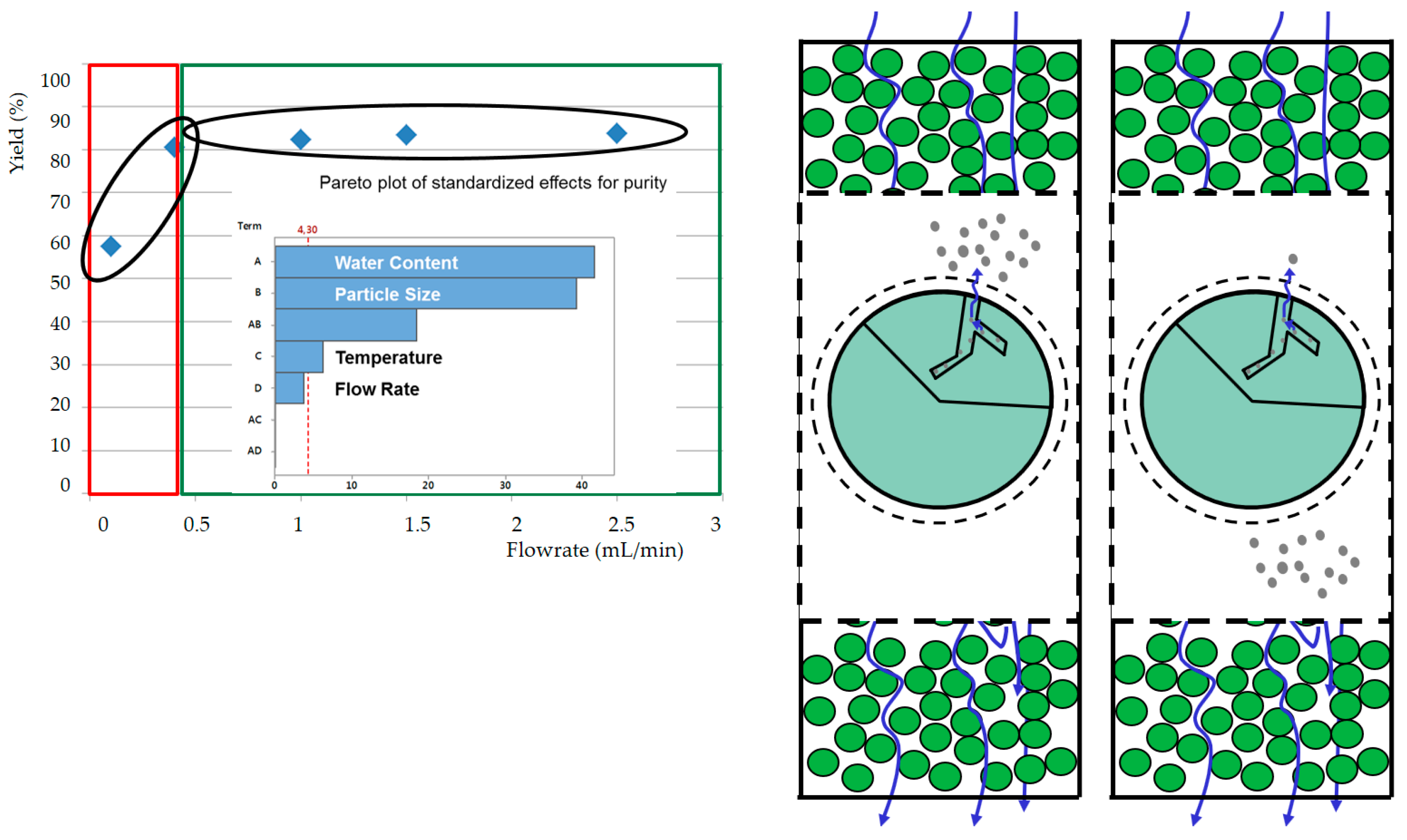
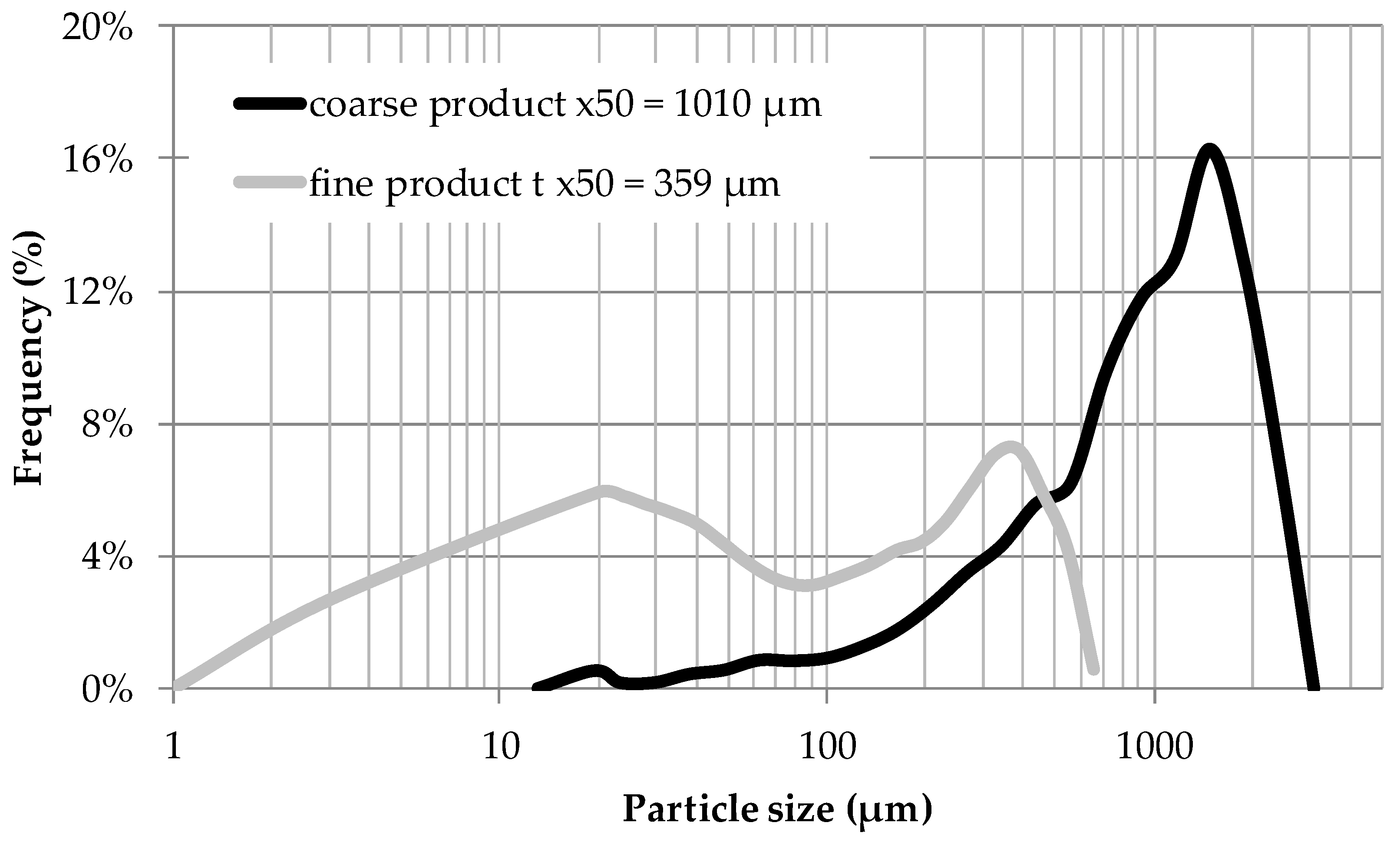
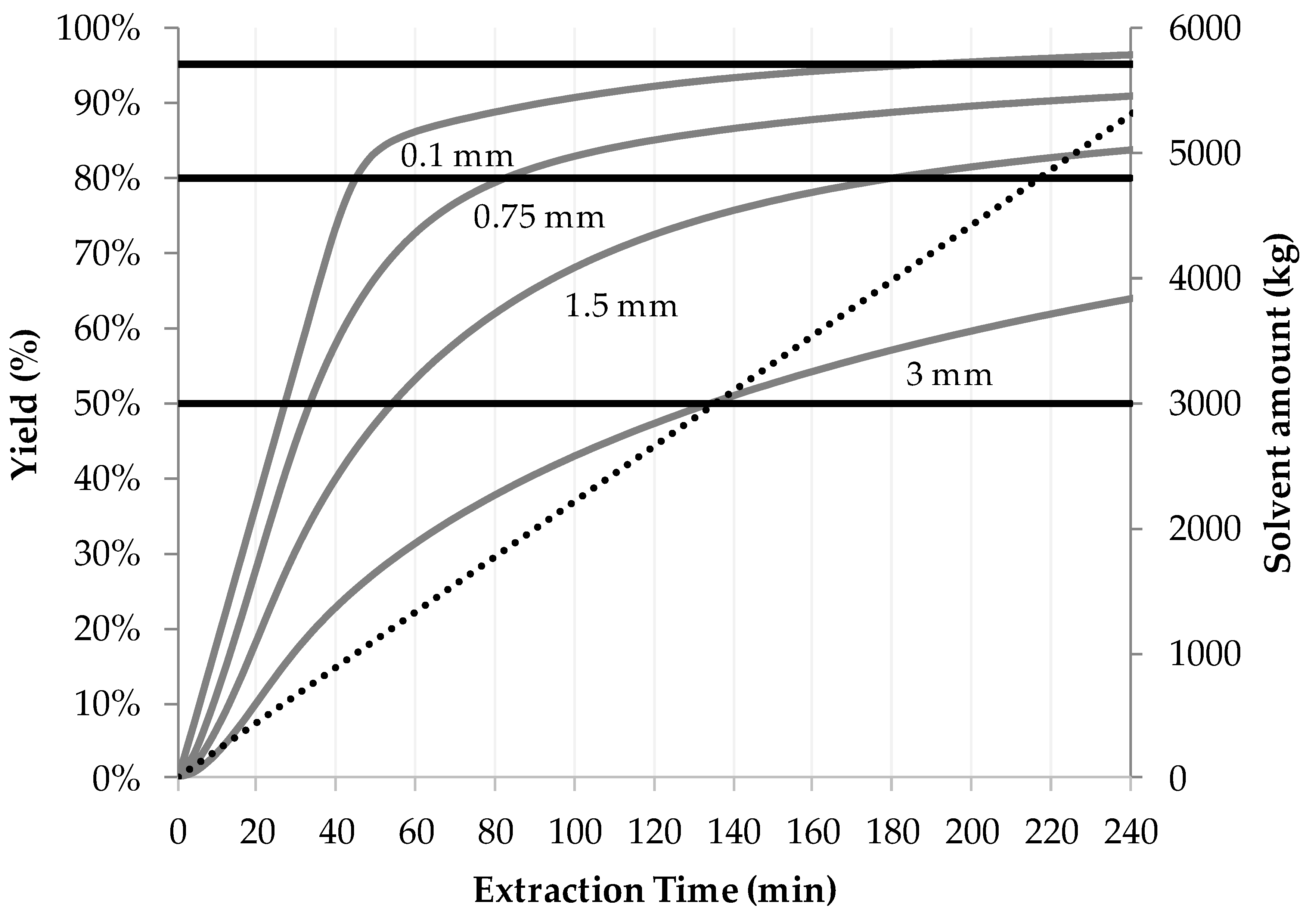
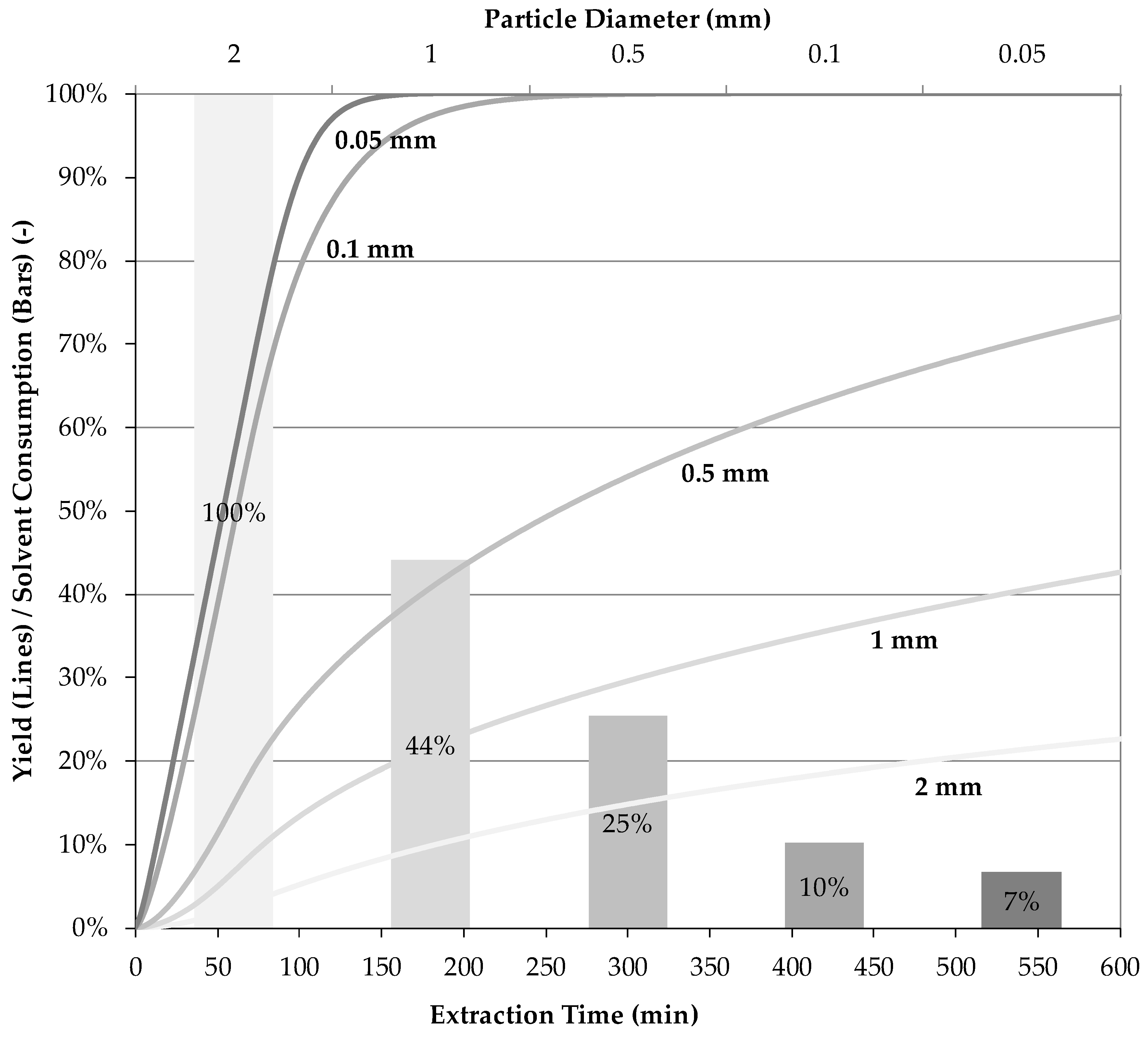
- If a targeted component is within the inner plant particle (e.g., yew, whitethorn, bearberry), then it is recommended that small particles be used to minimize the flow rate required to achieve high extraction yields. In this way, diffusion limitations can be minimized, and a highly concentrated extract can be obtained.
- Particle size is not significant if the target component is located on the outer surface (e.g., mugwort, salvia). As a consequence, high flow rates can be used to utilize the fast extraction kinetics. Overestimation of extraction kinetics can result in diluted extracts and a waste of solvent.
- The extraction of oil requires breakage of oils seams of the particle. The phase equilibrium is practically immeasurable, because only solution mechanisms with extremely fast kinetics occur (e.g., fennel, caraway).
5.2. Resource Efficiency Optimization
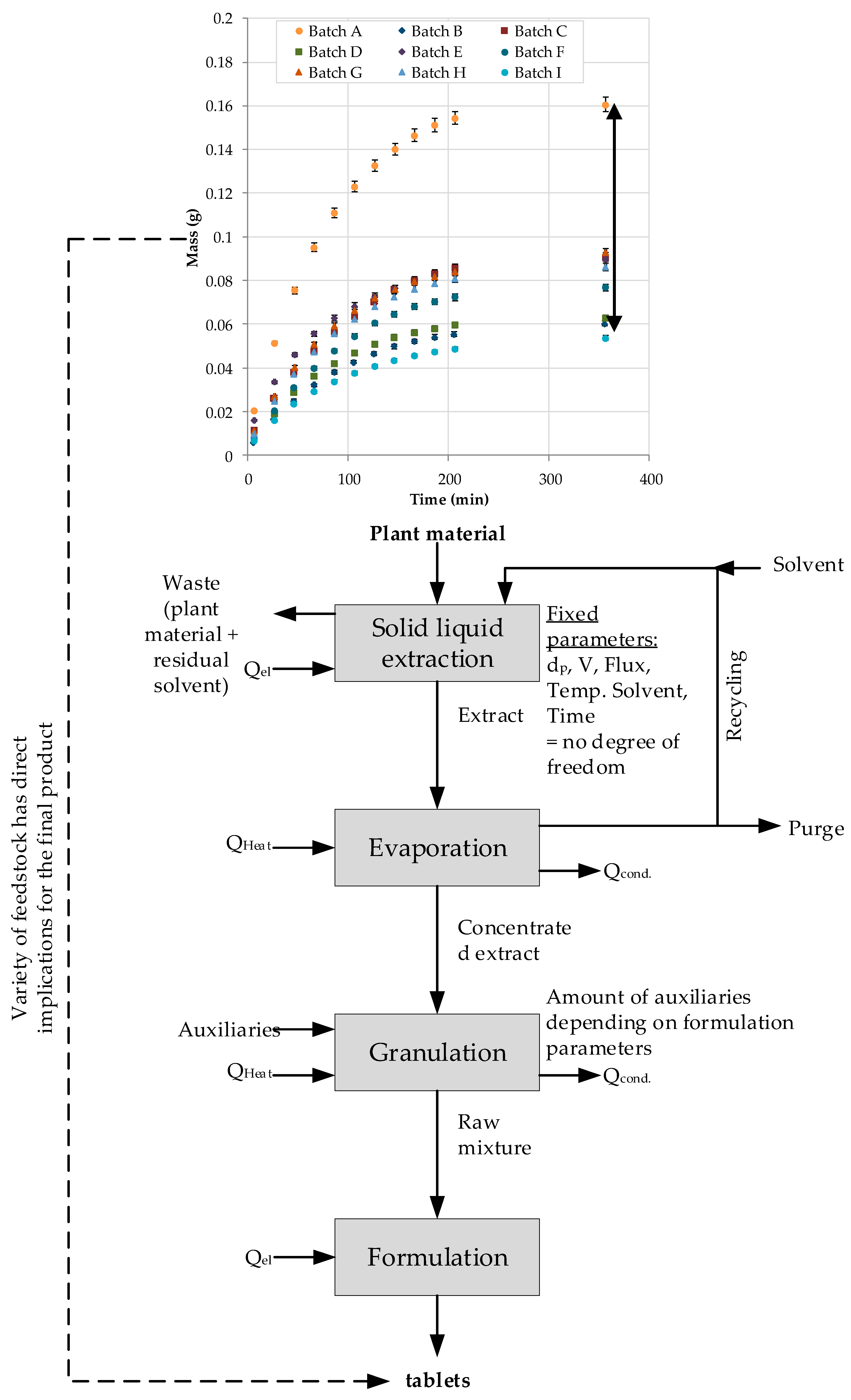
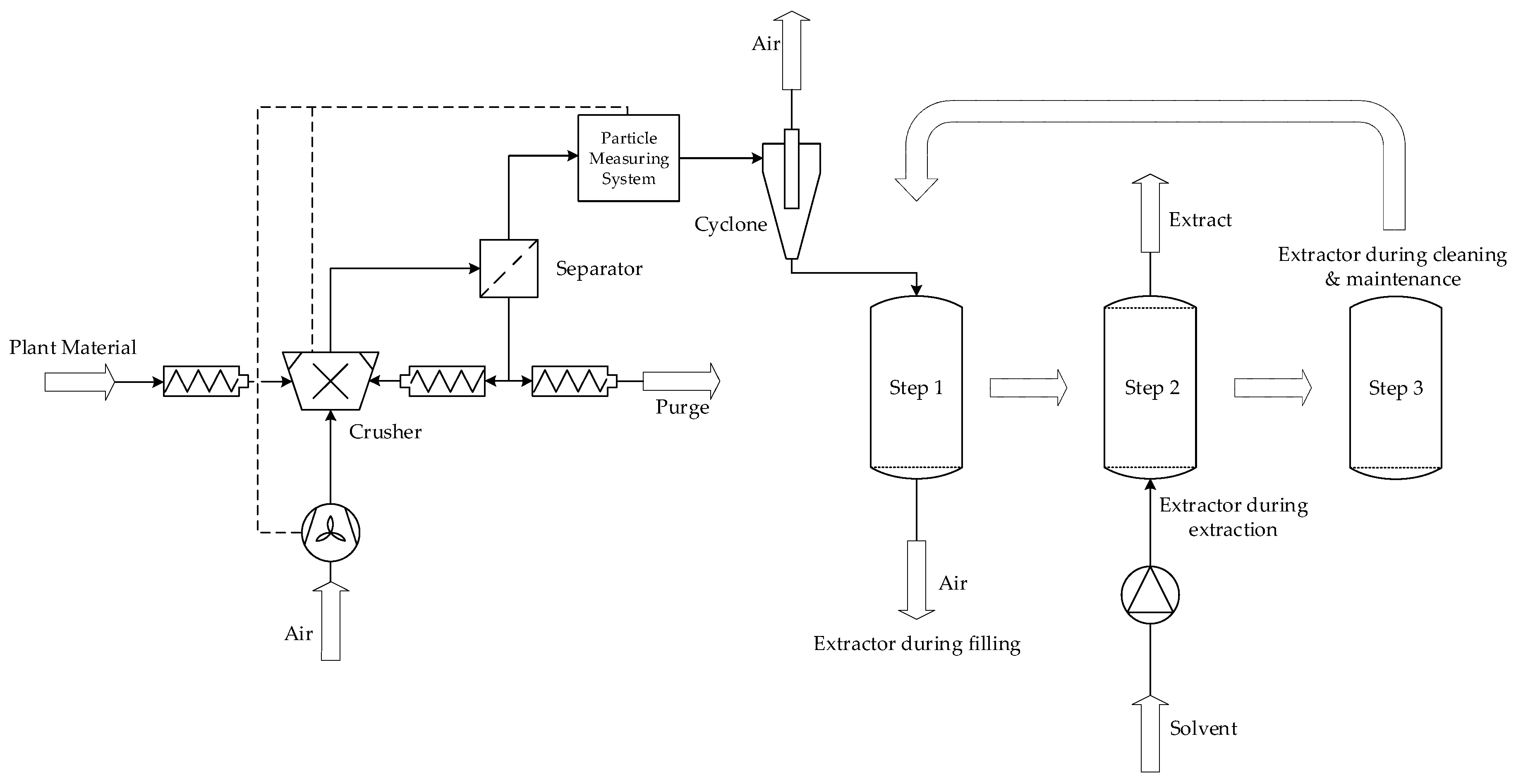
6. Integrated Continuous Pre-Treatment and Extraction (iCPE)
6.1. Integrated Continuous Pretreatment and Extraction (iCPE) Process
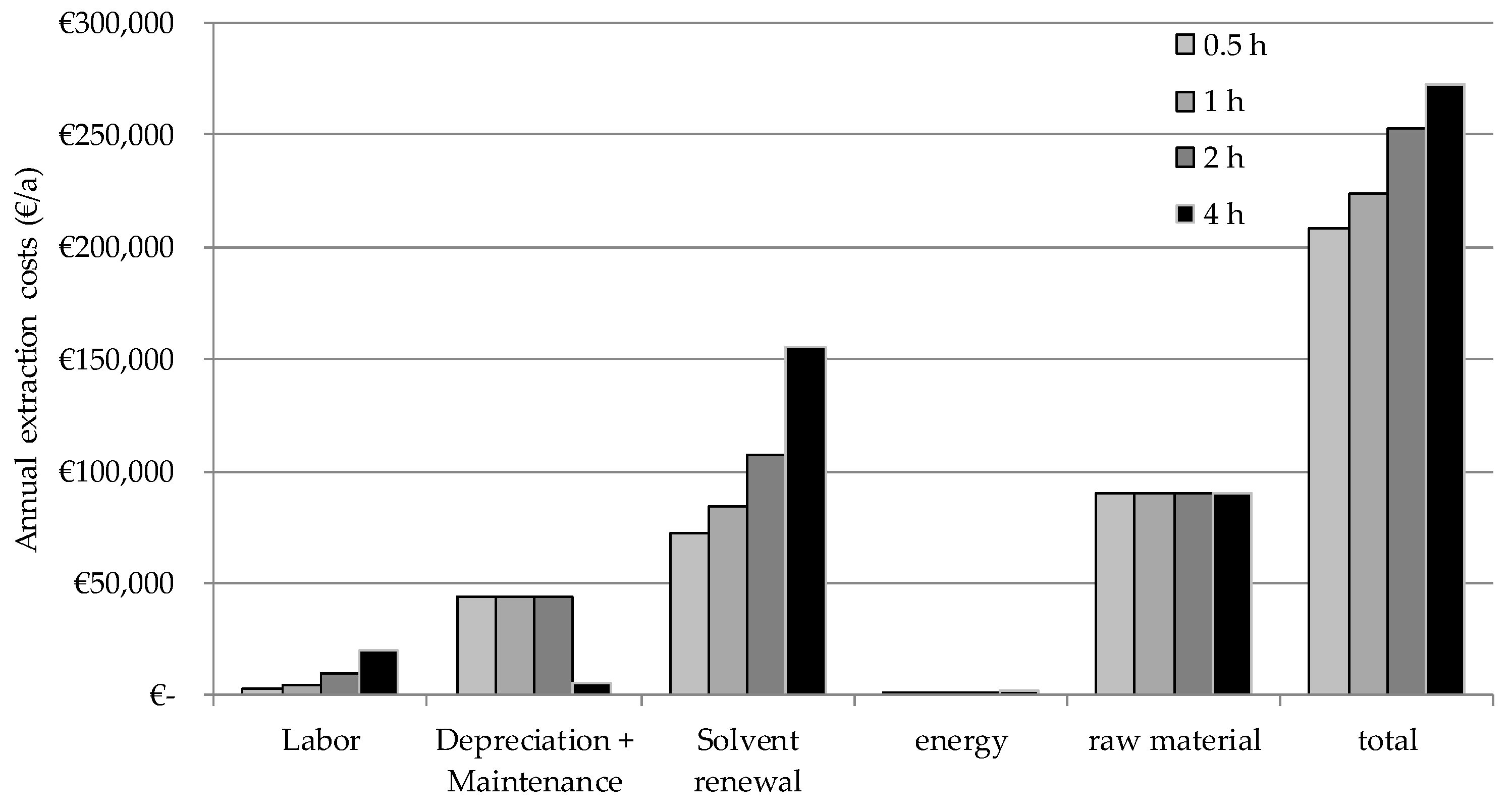
6.2. Cost Calculation and Results of the iCPE Process
- reducing the solvent amount,
- minimizing costs for solvent storage, recycling and replacement,
- continuously running fully automated solid-liquid extraction,
- replacing established processes with state-of-the art technology with comparable or even lower CAPEX,
- reducing COG.
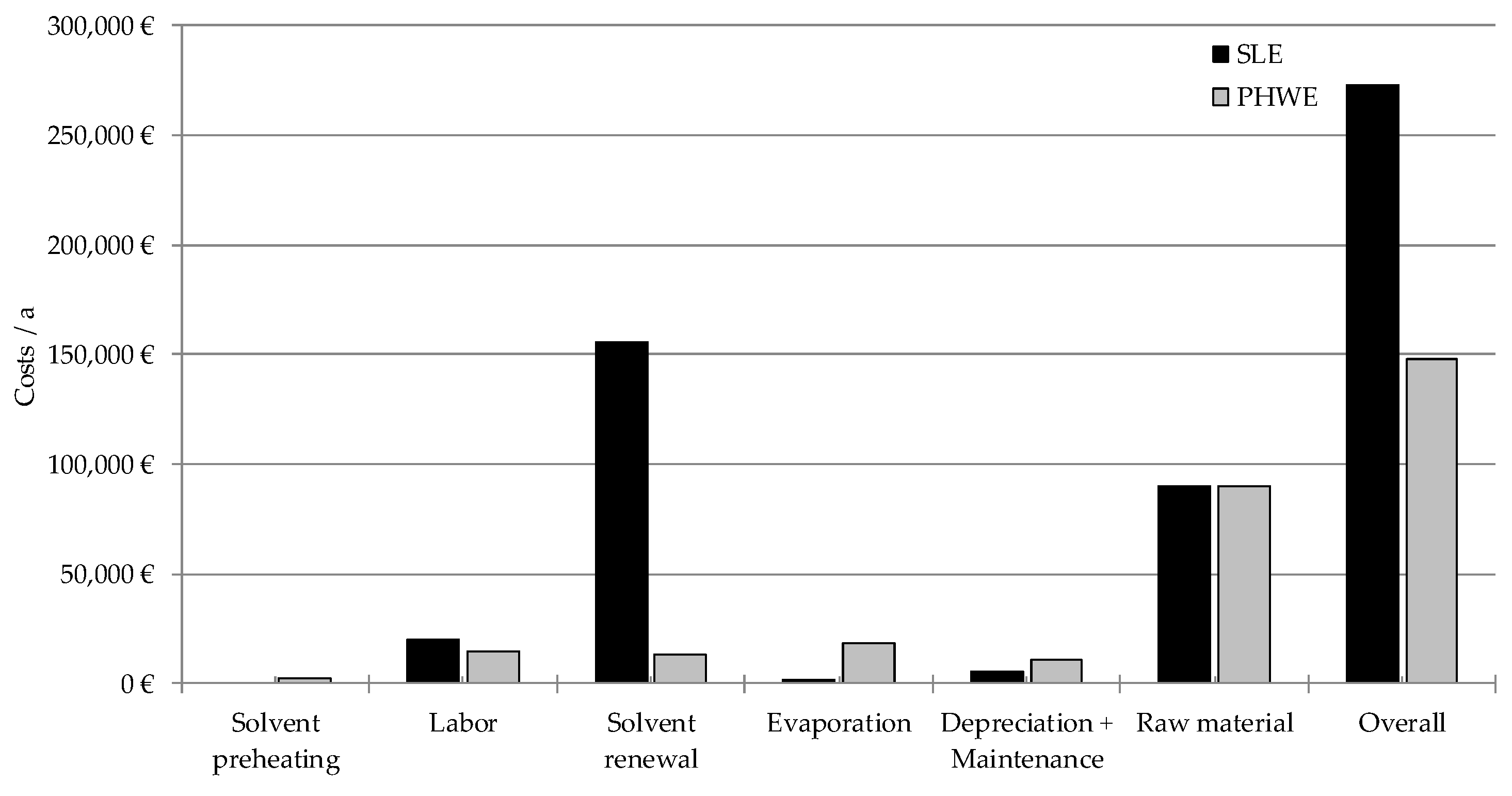
7. Water-Based Green Extraction Processing
- 30 t of leaves are extracted in 60 batches a year in a multi-purpose herbal raw. The costs for the herbal raw material is 3 €/kg and the yearly capacity of this product is 25%.
- An extraction equipment with 2 m3 of volume is used. The investment cost is 200,000 €.
- The extraction takes place for four hours and the solvent ratio is 2.7 kg Solvent/kg Herbal raw material/h.
- The extract is evaporated for solvent recycling purpose. Steam is used (120 °C, 5 bar, 2.7 MJ/kg) to operate the evaporator. The costs are 13 €/t which is typical for a site infrastructure.
- 10% of the solvent has to be renewed after each extraction due to loss. Moreover, the whole solvent (20 m3) is exchanged once a year to maintain a constant product quality. The solvent is priced at 3 €/kg.
- Labor costs are 100,000 €/a.
- The costs for yearly depreciation and maintenance are 2.5% each (multi-purpose herbal raw).
8. Inline Process Control in Phyto Extraction
9. Options and Opportunities for Future Value Generation
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| aF | Particle surface, m2 |
| CAPEX | Capital expentitures |
| cL | Concentration in the liquid phase, kg/m3 |
| COG | Cost of goods |
| cP | Concentration in the porous particle, kg/m3 |
| Dax | Axial dispersion coefficient, m/s2 |
| DE | dry extract |
| Deff | Effective diffusion coefficient, m2/s |
| DER | drug extract ratio |
| dF | Particle diameter m |
| DPF | Distributed plug flow |
| EMPL | employee |
| KL | Equilibrium constant, m3/kg |
| kf | Mass transport coefficient, m/s |
| Pe | Péclet number |
| PSD | Particle size distribution |
| q | Loading, kg/m3 |
| qmax | Maximum Loading, kg/m3 |
| Re | Reynolds number |
| r | Radius, m |
| Sc | Schmidt number |
| Sh | Sherwood number |
| SME | Small and medium-sized enterprise |
| SP | selling price |
| t | Time, s |
| uz | Superficial velocity, m/s |
| V | Volume flow, m3/s |
| z | Coordinate in axial direction, m |
| ε | Voids fraction, - |
| ρ | Density, kg/m3 |
References
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta 2013, 1830, 3670–3695. [Google Scholar] [CrossRef] [PubMed]
- Li, W.L.; Zheng, H.C.; Bukuru, J.; de Kimpe, N. Natural medicines used in the traditional Chinese medical system for therapy of diabetes mellitus. J. Ethnopharmacol. 2004, 92, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [PubMed]
- Ditz, R.; Gerard, D.; Hagels, H.; Igl, N.; Schäffler, M.; Schulz, H.; Stürtz, M.; Tegtmeier, M.; Treutwein, J.; Strube, J.; et al. Phytoextracts: Proposal towards a New Comprehensive Research Focus; DECHEMA Gesellschaft für Chemische Technik und Biotechnologie e.V.: Frankfurt, Germany, 2017. [Google Scholar]
- Bart, H.-J.; Pilz, S. Industrial Scale Natural Products Extraction; Wiley-VCH: Weinheim, Germany, 2011. [Google Scholar]
- Chémat, F.; Strube, J. (Eds.) Green Extraction of Natural Products: Theory and Practice; Wiley VCH: Weinheim, Germany, 2015. [Google Scholar]
- Veeresham, C. Natural products derived from plants as a source of drugs. J. Adv. Pharm. Technol. Res. 2012, 3, 200–201. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Lopez, S.N.; Ramallo, I.A.; Sierra, M.G.; Zacchino, S.A.; Furlan, R.L.E. Chemically engineered extracts as an alternative source of bioactive natural product-like compounds. Proc. Natl. Acad. Sci. USA 2007, 104, 441–444. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, H.Y.; Jamshidi, S.; Sutton, J.M.; Rahman, K.M. Current Advances in Developing Inhibitors of Bacterial Multidrug Efflux Pumps. Curr. Med. Chem. 2016, 23, 1062–1081. [Google Scholar] [CrossRef] [PubMed]
- Posadzki, P.; Watson, L.; Ernst, E. Herb-drug interactions: An overview of systematic reviews. Br. J. Clin. Pharmacol. 2013, 75, 603–618. [Google Scholar] [CrossRef] [PubMed]
- Newman, D. Screening and identification of novel biologically active natural compounds. F1000Research 2017, 6, 783. [Google Scholar] [CrossRef] [PubMed]
- Ramallo, I.A.; Salazar, M.O.; Mendez, L.; Furlan, R.L.E. Chemically Engineered Extracts: Source of Bioactive Compounds. Acc. Chem. Res. 2011, 44, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Ta, C.; Arnason, J. Mini Review of Phytochemicals and Plant Taxa with Activity as Microbial Biofilm and Quorum Sensing Inhibitors. Molecules 2016, 21, 29. [Google Scholar] [CrossRef] [PubMed]
- Kalia, V.C.; Wood, T.K.; Kumar, P. Evolution of Resistance to Quorum-Sensing Inhibitors. Microb. Ecol. 2014, 68, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Borges, A.; Abreu, A.; Dias, C.; Saavedra, M.; Borges, F.; Simões, M. New Perspectives on the Use of Phytochemicals as an Emergent Strategy to Control Bacterial Infections Including Biofilms. Molecules 2016, 21, 877. [Google Scholar] [CrossRef] [PubMed]
- Aparna, V.; Dineshkumar, K.; Mohanalakshmi, N.; Velmurugan, D.; Hopper, W. Identification of Natural Compound Inhibitors for Multidrug Efflux Pumps of Escherichia coli and Pseudomonas aeruginosa Using In Silico High-Throughput Virtual Screening and In Vitro Validation. PLoS ONE 2014, 9, e101840. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, G.G.F.; Locatelli, J.; Freitas, P.C.; Silva, G.L. Antibacterial activity of plant extracts and phytochemicals on antibiotic-resistant bacteria. Braz. J. Microbiol. 2000, 31, 247–256. [Google Scholar] [CrossRef]
- Efferth, T.; Kahl, S.; Paulus, K.; Adams, M.; Rauh, R.; Boechzelt, H.; Hao, X.; Kaina, B.; Bauer, R. Phytochemistry and pharmacogenomics of natural products derived from traditional Chinese medicine and Chinese materia medica with activity against tumor cells. Mol. Cancer Ther. 2008, 7, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Europäisches Zentrum für die Prävention und die Kontrolle von Krankheiten. The Bacterial Challenge, Time to React: A Call to Narrow the Gap between Multidrug-Resistant Bacteria in the EU and the Development of New Antibacterial Agents; ECDC: Stockholm, Sweden, 2009. [Google Scholar]
- DECHEMA e.V. Biobased World. Available online: https://www.biobasedworldnews.com/ (accessed on 22 June 2018).
- Bundesministerium für Bildung und Forschung (BMBF). Wegweiser Bioökonomie—Forschung für Biobasiertes und Nachhaltiges Wirtschaftswachstum; BMBF: Berlin, Germany, 2014. [Google Scholar]
- Jamshidi Aidji, M.; Morlock, G. Effect Directed Analysis of Salvia Officinalis, Poster Presentation; University of Giessen: Gießen, Germany, 2014. [Google Scholar]
- Morlock, G.; Schwack, W. Hyphenations in planar chromatography. J. Chromatogr. A 2010, 1217, 6600–6609. [Google Scholar] [CrossRef] [PubMed]
- Staniek, A.; Bouwmeester, H.; Fraser, P.D.; Kayser, O.; Martens, S.; Tissier, A.; van der Krol, S.; Wessjohann, L.; Warzecha, H. Natural products—Learning chemistry from plants. Biotechnol. J. 2014, 9, 326–336. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Assessment Report on Echinacea purpurea (L.) Moench, Radix; University of Ljubljana: Ljubljana, Slovenia, 2017. [Google Scholar]
- European Medicines Agency. European Union Herbal Monograph on Echinacea purpurea (L.) Moench, Radix; European Medicines Agency: London, UK, 2017. [Google Scholar]
- Council of Europe. European Pharmacopoeia, 8th ed.; Published in Accordance with the Convention on the Elaboration of a European Pharmacopoeia (European Treaty Series No. 50); Council of Europe: Strasbourg, France, 2013–2015. [Google Scholar]
- Food and Drug Administration (FDA). Guidance for Industry—Botanical Drug Development; FDA: Silver Spring, MD, USA, 2016. [Google Scholar]
- Food and Drug Administration (FDA). Dietary Supplements. Available online: https://www.fda.gov/Food/DietarySupplements/ProductsIngredients/default.htm (accessed on 21 September 2018).
- Chinese Food and Drug Administration (CFDA). Verification and Issuance of Registration Certificates for Imported (Incl. from Hong Kong, Macao and Taiwan) TCM and Natural Medicine. Available online: http://eng.sfda.gov.cn/WS03/CL0769/98141.html (accessed on 21 September 2018).
- Akerele, O. WHO traditional medicines programme: Progress and perspectives. WHO Chron. 1984, 38, 76–81. [Google Scholar] [PubMed]
- Chaing, H.S.; Merino-Chavez, G.; Yang, L.L.; Hafez, E.S.E. Medical plants: Conception/Contratception. Adv. Contracept Deliv Syst. 1994, 10, 355–363. [Google Scholar] [PubMed]
- Chotchoungchatchai, S.; Saralamp, P.; Jenjittikul, T.; Pornsiripongse, S.; Prathanturarug, S. Medicinal plants used with Thai Traditional Medicine in modern healthcare services: A case study in Kabchoeng Hospital, Surin Province, Thailand. J. Ethnopharmacol. 2012, 141, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Wagner, H.; Ulrich-Merzenich, G. (Eds.) Evidence and Rational Based Research on Chinese Drugs; Springer: Vienna, Austria, 2013. [Google Scholar]
- Giovannini, P.; Howes, M.-J.R.; Edwards, S.E. Medicinal plants used in the traditional management of diabetes and its sequelae in Central America: A review. J. Ethnopharmacol. 2016, 184, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Govindaraghavan, S.; Sucher, N.J. Quality assessment of medicinal herbs and their extracts: Criteria and prerequisites for consistent safety and efficacy of herbal medicines. Epilepsy Behav. 2015, 52, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Pferschy-Wenzig, E.-M.; Bauer, R. The relevance of pharmacognosy in pharmacological research on herbal medicinal products. Epilepsy Behav. 2015, 52, 344–362. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.L. Ins and outs of dietary phytochemicals in cancer chemoprevention. Biochem. Pharmacol. 2007, 74, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Schuster, B.G. A new integrated program for natural product development. J. Altern. Complement. Med. 2001, 7, 61–72. [Google Scholar] [CrossRef]
- Food and Drug Administration (FDA). Guidance for Industry. PAT—A Framework for Innovative Pharmaceutical Development, Manufacturing, and Quality Assurance; FDA: Silver Spring, MD, USA, 2004. [Google Scholar]
- Food and Drug Administration (FDA). Pharmaceutical cGMP for the 21st Century: A Risk Based Approach; FDA: Silver Spring, MD, USA, 2004. [Google Scholar]
- Sixt, M.; Koudous, I.; Strube, J. Process design for integration of extraction, purification and formulation with alternative solvent concepts. C. R. Chim. 2016, 19, 733–748. [Google Scholar] [CrossRef]
- Sixt, M.; Gudi, G.; Schulz, H.; Strube, J. In-line Raman spectroscopy and advanced process control for the extraction of anethole and fenchone from fennel (Foeniculum vulgare L. MILL.). C. R. Chim. 2018, 21, 97–103. [Google Scholar] [CrossRef]
- Sixt, M.; Uhlenbrock, L.; Strube, J. Toward a Distinct and Quantitative Validation Method for Predictive Process Modelling—On the Example of Solid-Liquid Extraction Processes of Complex Plant Extracts. Processes 2018, 6, 66. [Google Scholar] [CrossRef]
- Sixt, M.; Strube, J. Pressurized hot water extraction of 10-deacetylbaccatin III from yew for industrial application. Resour. Effic. Technol. 2017, 3, 177–186. [Google Scholar] [CrossRef]
- Uhlenbrock, L.; Sixt, M.; Strube, J. Quality-by-Design (QbD) process evaluation for phytopharmaceuticals on the example of 10-deacetylbaccatin III from yew. Resour. Effic. Technol. 2017, 3, 137–143. [Google Scholar] [CrossRef]
- Wübbeke, J.; Meissner, M.; Zenglein, M.J.; Ives, J.; Conrad, B. Made in China 2025: The Making of a High-Tech Superpower and Consequences for Industrial Countries; Mercator Institute for China Studies: Berlin, Germany, 2016. [Google Scholar]
- Sixt, M.; Strube, J. Systematic Design and Evaluation of an Extraction Process for Traditionally Used Herbal Medicine on the Example of Hawthorn (Crataegus monogyna JACQ.). Processes 2018, 6, 73. [Google Scholar] [CrossRef]
- Sixt, M. Entwicklung von Methoden zur systematischen Gesamtprozessentwicklung und Prozessintensivierung von Extraktions- und Trennprozessen zur Gewinnung pflanzlicher Wertkomponenten; Clausthal University of Technology: Clausthal-Zellerfeld, Germany; Shaker: Aachen, Germany, 2018. [Google Scholar]
- Kassing, M.; Jenelten, U.; Schenk, J.; Hänsch, R.; Strube, J. Combination of Rigorous and Statistical Modeling for Process Development of Plant-Based Extractions Based on Mass Balances and Biological Aspects. Chem. Eng. Technol. 2012, 35, 109–132. [Google Scholar] [CrossRef]
- Kassing, M.; Jenelten, U.; Schenk, J.; Strube, J. A New Approach for Process Development of Plant-Based Extraction Processes. Chem. Eng. Technol. 2010, 33, 377–387. [Google Scholar] [CrossRef]
- Kaßing, M. Process Development for Plant-Based Extract Production; Clausthal University of Technology: Clausthal-Zellerfeld, Germany; Shaker: Aachen, Germany, 2012. [Google Scholar]
- Both, S.; Eggersglüß, J.; Lehnberger, A.; Schulz, T.; Schulze, T.; Strube, J. Optimizing Established Processes like Sugar Extraction from Sugar Beets—Design of Experiments versus Physicochemical Modeling. Chem. Eng. Technol. 2013, 36, 2125–2136. [Google Scholar] [CrossRef]
- Both, S. Systematische Verfahrensentwicklung für Pflanzlich Basierte Produkte im Regulatorischen Umfeld; Clausthal University of Technology: Clausthal-Zellerfeld, Germany; Shaker: Aachen, Germany, 2015. [Google Scholar]
- Both, S.; Chemat, F.; Strube, J. Extraction of polyphenols from black tea—Conventional and ultrasound assisted extraction. Ultrason Sonochem. 2014, 21, 1030–1034. [Google Scholar] [CrossRef] [PubMed]
- Ndocko Ndocko, E.; Bäcker, W.; Strube, J. Process Design Method for Manufacturing of Natural Compounds and Related Molecules. Sep. Sci. Technol. 2008, 43, 642–670. [Google Scholar] [CrossRef]
- Goedecke, R. Fluidverfahrenstechnik; Wiley-VCH: Hoboken, NI, USA, 2008. [Google Scholar]
- Sixt, M.; Strube, J. Systematic and Model-Assisted Evaluation of Solvent Based- or Pressurized Hot Water Extraction for the Extraction of Artemisinin from Artemisia annua L. Processes 2017, 5, 86. [Google Scholar] [CrossRef]
- Koudous, I. Stoffdatenbasierte Verfahrensentwicklung zur Isolierung von Wertstoffen aus Pflanzenextrakten; Clausthal University of Technology: Clausthal-Zellerfeld, Germany; Shaker: Aachen, Germany, 2016. [Google Scholar]
- Gudi, G.; Krähmer, A.; Koudous, I.; Strube, J.; Schulz, H. Infrared and Raman spectroscopic methods for characterization of Taxus baccata L.—Improved taxane isolation by accelerated quality control and process surveillance. Talanta 2015, 143, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Duke, M.V.; Paul, R.N.; Elsohly, H.N.; Sturtz, G.; Duke, S.O. Localization of Artemisinin and Artemisitene in Foliar Tissues of Glanded and Glandless Biotypes of Artemisia annua L. Int. J. Plant. Sci 1994, 155, 365–372. [Google Scholar] [CrossRef]
- Gudi, G.; Krähmer, A.; Krüger, H.; Hennig, L.; Schulz, H. Discrimination of Fennel Chemotypes Applying IR and Raman Spectroscopy: Discovery of a New γ-Asarone Chemotype. J. Agric. Food Chem. 2014, 62, 3537–3547. [Google Scholar] [CrossRef] [PubMed]
- Lucke, M.; Koudous, I.; Sixt, M.; Huter, M.J.; Strube, J. Integrating crystallization with experimental model parameter determination and modeling into conceptual process design for the purification of complex feed mixtures. Chem. Eng. Res. Des. 2018, 133, 264–280. [Google Scholar] [CrossRef]
- Helling, C.; Strube, J. Modeling and Experimental Model Parameter Determination with Quality by Design for Bioprocesses: Biopharmaceutical Production Technology; Wiley-VCH-Verl: Weinheim, Germany, 2012; pp. 409–445. [Google Scholar]
- Chemat, F.; Vian, M.A. (Eds.) Alternative Solvents for Natural Products Extraction; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Chémat, F.; Vorobiev, E.; Lebovka, N.I. Enhancing Extraction Processes in the Food Industry; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- European Medicines Agency, Food and Drug Administration. EMA-FDA Pilot Program for Parallel Assessment of Quality by Design Applications; European Medicines Agency, Food and Drug Administration: London, UK, 2011. [Google Scholar]
- Food and Drug Administration (FDA). Guideline for Implimentation of Q9; FDA: Silver Spring, MD, USA, 2006. [Google Scholar]
- ICH Expert Working Group (ICH EWG). Riskmanagement (Q9). ICH Harmonised Tripartite Guideline. Q9; ICH Expert Working Group: Geneva, Switzerland, 2005. [Google Scholar]
- Roggo, Y.; Duponchel, L.; Huvenne, J.-P. Quality Evaluation of Sugar Beet (Beta vulgaris) by Near-Infrared Spectroscopy. J. Agric. Food Chem. 2004, 52, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Chémat, F.; Fernandez, X. (Eds.) La Chimie des Huiles Essentielles: Tradition et Innovation; Vuibert: Paris, France, 2012. [Google Scholar]
- Forschungsvereinigung der Arzneimittel-Hersteller e.V. (FAH). (FAH). Trainings-Course on Phytoextraction—Process Design and Operation, Experiments and Modelling October 2017 at the Institute for Separation and Process Technology; Clausthal University of Technology: Clausthal-Zellerfeld, Germany, 2017. [Google Scholar]

| Category | Agrochemicals | Cosmetics | Aroma, Flavours and Nutrition | Pharma |
|---|---|---|---|---|
| Market volume | 1 Billion USD | 200 Billion USD | 10 Billion USD | 107 Billion USD (forecast 2017) [21] |
| Market growth | Double digit annual growth rate | Double digit annual growth rate | Double digit annual growth rate Market for nutrition additives decreases Market for aromas grows | Double digit annual growth rate Decline in prescription market Growth in over-the-counter market |
| Challenges | Market dominated by SMEs as well as global players Small volume/low cost products bulk High cost/low volume niche products | Significant amount of products with natural claims but up to 75% synthetic ingredients; No uniform and binding standards for natural, fair-trade, organic labels | Low cost products (in the order of 1–10 €/kg) Many products with small volumes (100–1000 kg/a) | Most products are OTC Only few blockbusters Restrictive regulatory hamstringed R&D |
| Medium-term research demands | Efficient total process design for SMEs; Integrate process intensification Methods for SMEs and scale-up of infrastructure to fully integrated manufacturers; Energy efficient and low waste processes for decentralised utilization of natural resources [21,22] * | Efficient ways of finding new natural ingredients [23,24]; * Ensuring sustainability of supply | Apply and adopt more often scCO2, bio-based solvents *, PHWE Biomass valorization, e.g., carrot, broccoli, artichoke etc. do have 30–80% herbal raw material waste | Speed up of development of herbal raw cell fermentation by omics [25] * Process Analytical Technology for inline-analysis of extraction processes; Parametric defined release at manufacturing of herbal raw extracts; * Homogeneity at production of extracts in large-scale Lyophilisation instead of vacuum-belt drying; Fresh herbal raws instead of dried raw material; HGACP instead of GMP on field incl. extraction media and pomace to be deposited on field again |
| Long-term research demands | Development of new products | Shift from wild collection to greenhouse or field cultivation in Europe; Energy efficient and low waste processes for decentralised utilization of natural resources [21,22] | Energy efficient and low waste processes for decentralised utilization of natural resources [21,22] | Determination of distribution behaviour of herbal raw ingredients in “single pot model” with herbal raw cell membranes and a gastrointestinal membrane for fast prediction of bioavailable components; Efficacy studies for new herbal raws and products which enable IP protection to cover the costs via patents on the new processes |
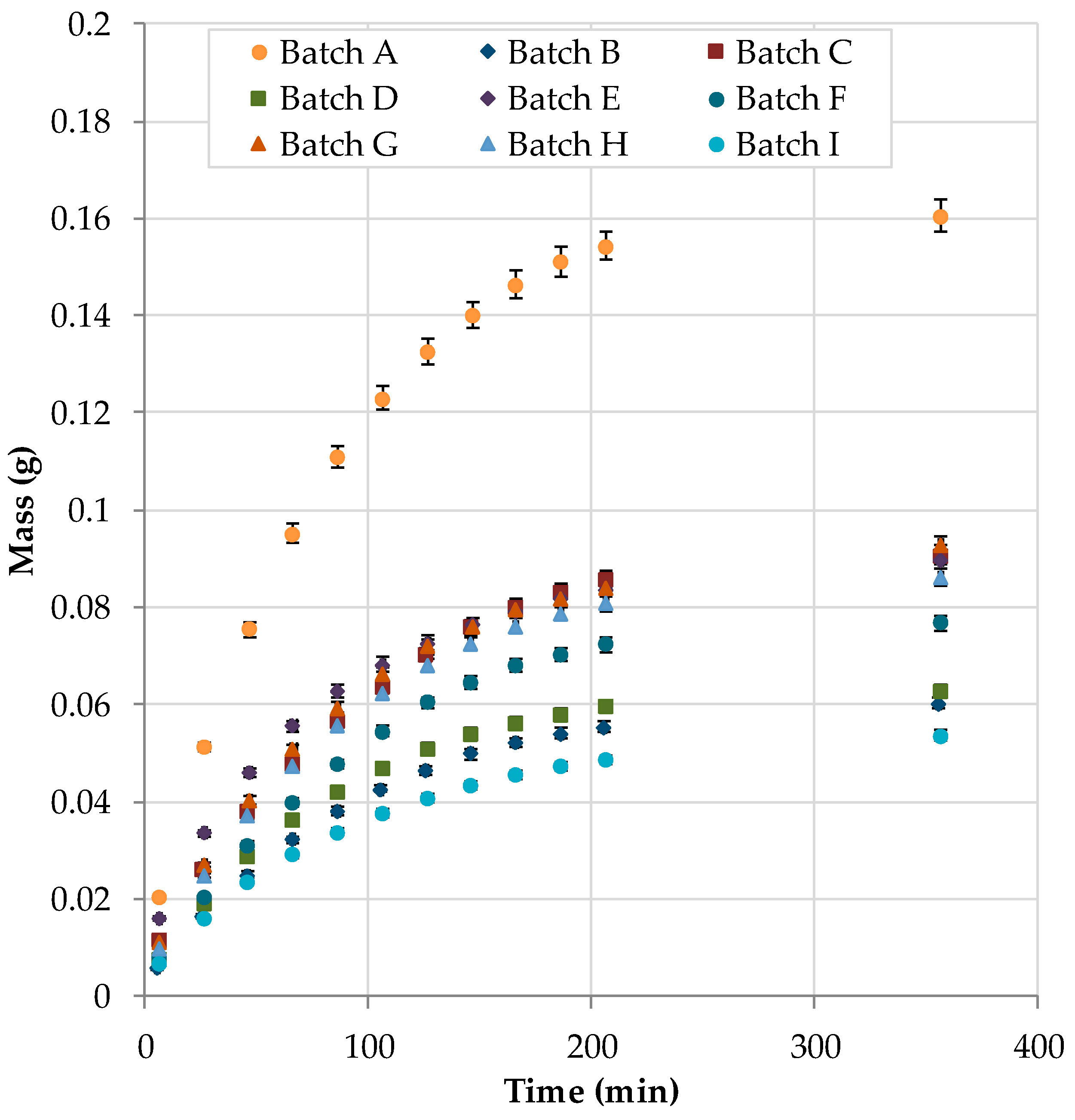
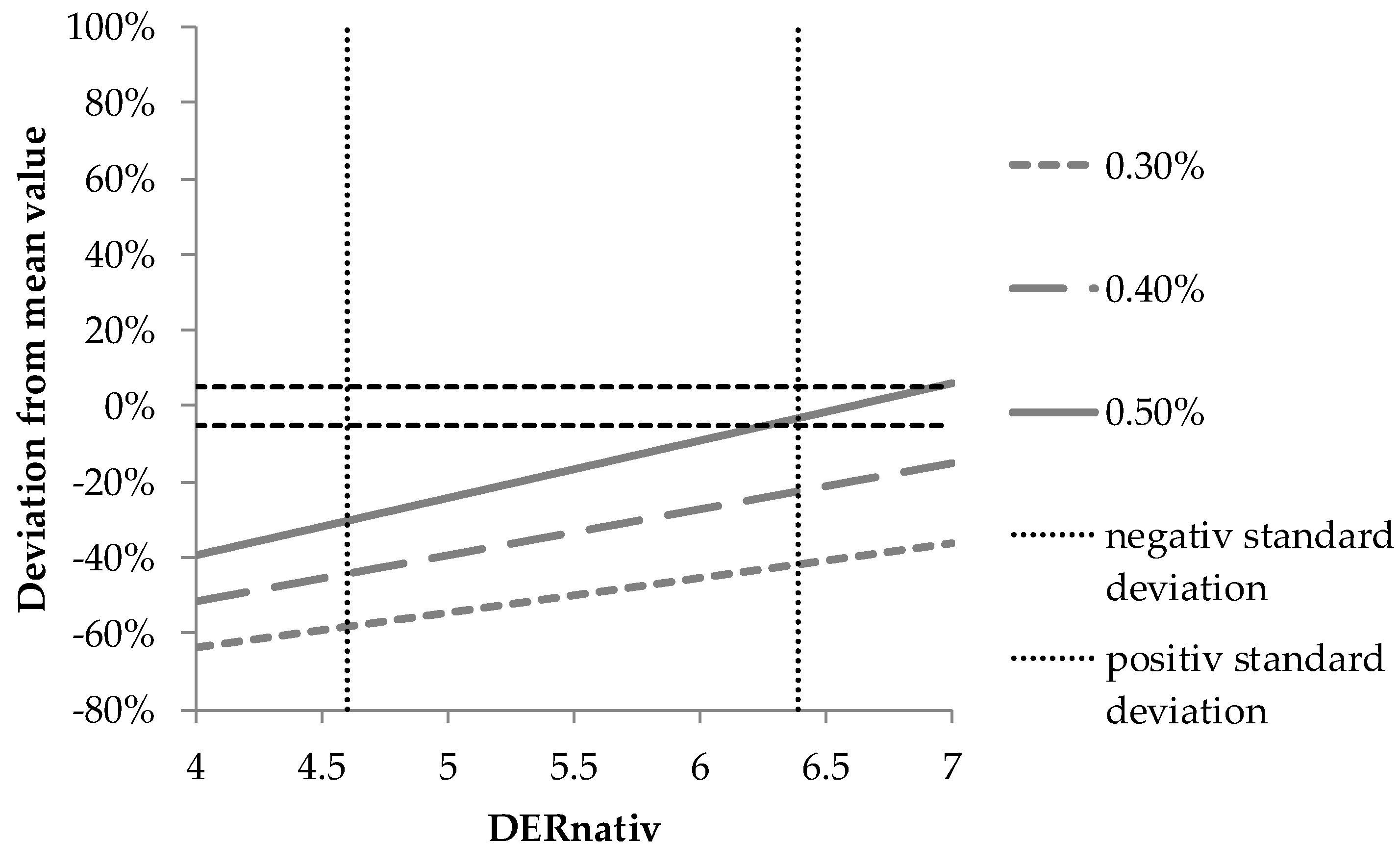
| Lot | Year | Origin | Overall Amount | Deviation Referred to Lot I |
|---|---|---|---|---|
| A | 2017 | Southeast Europe | 0.87% | 146% |
| B | 2016 | Macedonia | 0.35% | −1% |
| C | 2017 | Bulgaria | 0.58% | 64% |
| D | 2017 | Rumania | 0.41% | 16% |
| E | 2014 | Bulgaria | 0.60% | 71% |
| F | 2017 | Albania | 0.42% | 20% |
| G | 2017 | Southeast Europe | 0.57% | 62% |
| H | 2017 | Serbia | 0.51% | 43% |
| I | 2017 | Germany | 0.35% | - |
| Category | Taxus baccata L. | Crataegus monogyna JACQ. | Foeniculum vulgare L. Mill. | Carum carvi L. | Artemisia annua L. | Arctostaphylos uvaursi (L.) SPRING. | Azadirachta indica A. Juss. |
|---|---|---|---|---|---|---|---|
| Use | Pharma (cancer treatment) | Pharma (extract for cardiac insufficiency) | Aroma/fragrance | Aroma/fragrance | Pharma (Malaria treatment) | Pharma (extract for bladder infection) | Agro (pest control) |
| Target component | 10-Deacetylbaccatin III (0.1–0.4% w) | Hyperosid (0.3–0.7% w) | Anethole (5.3%), Fenchone (2.9%) essential oil (~8% w) | Carvone, Limonene essential oil (~2% w) | Artemisinin (~0.4% w) | Arbutin (~15% w) | Azadirachtin |
| Molecular structure and weight | 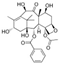 | 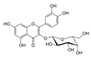 | 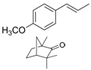 | 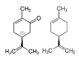 | 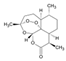 | 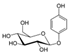 |  |
| 544.59 Da | 464.38 Da | 148.2/152.23 Da | 150.22 Da | 282.33 Da | 272.25 Da | 720.71 Da | |
| Side component | Unknown | Unknown | Estragole (0.2%) | Unknown | Hydroquinone | Unknown | |
| Molecular structure and weight | 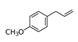 | 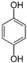 | |||||
| 148.2 Da | 110.11 Da | ||||||
| Location | Needle Diffusion limitation | Leaf Diffusion limitation | Fruit Oil channels | Fruit Oil channels | Trichoma cells [62] | Leaf | |
| Solvent | Acetone/Water (80/20 v/v) [43] PHWE (120 °C, moderate decomposition) [46] | Ethanol/Water (70/30 v/v) PHWE (140 °C, no decomposition) [50] | Ethanol [60] | Acetone PHWE (80 °C, fast decomposition) [59,60] | Water (25 °C, fast degradation during maceration) PHWE (140 °C, no decomposition) [50] | Water (25 °C pH 4) | |
 |  | 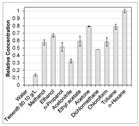 | 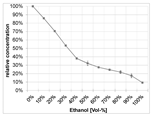 | ||||
| Modelling | Pore diffusion | Pore diffusion | Broken Cells | Broken Cells | Film diffusion | Pore diffusion | |
| Equilibrium |  |  |  | 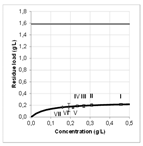 |  | ||
| Optimization | small dp [60] | small dp [50] | Hydro distillation preferred | Hydro distillation preferred | high flow rate [6] | [50] | |
| Purification | Benchmark, lab-scale [43] | None | Conceptional | None | Benchmark, Lab-scale, Pilot-scale [50] | None | Lab-scale, Pilot-scale |
| Basic research | FTIR process control, Raman-mapping [61] | Lot variety [50] | Inline spectroscopy, APC, Raman-Mapping [63] | CLSM, FTIR | Process integration crystallization [50] | Lot variety, decomposition |
| Category | Vanilla planifolia Jacks. Ex Andrews | Piper nigrum L. | Camellia sinensis (L.) KUNTZE | Salvia officinalis L. | Beta vulgaris L. | Zea mays L. | Larix decidua Mill. |
|---|---|---|---|---|---|---|---|
| Use | Aroma/Food | Aroma/Food | Aroma/Food | Food (preserving agent) | Food | Food | Agro (pest control) |
| Target component | Vanillin (3–7% w) | Piperine (~6.5%) | Caffeine (3–6%) | Carnosol (0.1% w) | Succrose (14–20% w) | Tricin (55 ppm) | Larixol Larixylacetat |
| Molecular structure and weight | 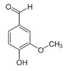 |  | 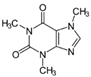 | 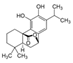 | 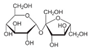 |  | 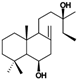 |
| 152.15 Da | 285.34 Da | 194.19 Da | 330.42 Da | 343.3 Da | 306.49 Da | ||
| Side component | Polyphenoles | Carnosoic acid (1.7%) | Ions (Mg, Na, K) Proteins | Tannines | |||
| Molecular structure and weight |  | 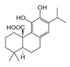 | |||||
| >1000 Da | 332.42 Da | ||||||
| Location | Fruit | Leaf | Trichoma cells, Film diffusion limitation | Tuber | Bark | ||
| Solvent | Ethanol [51] | Ethyl acetate [51] | Water, Ethanol, US Extraction [56] | Acetone [60] | Water [54] | Ethanol/Isopropanol | |
 | |||||||
| Modelling | Pore diffusion | Pore diffusion | Pore diffusion | Film diffusion | Pore diffusion | Pore diffusion | Pore diffusion |
| Equilibrium | 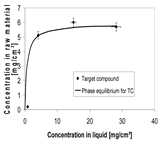 | 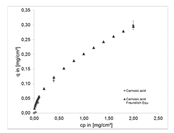 | |||||
| Optimization | [51] | small dp | high flow rate [60] | small dp [9] | |||
| Purification | None | None | None | Benchmark, Lab-scale [60] | None | Conceptional | None |
| Basic research | Raman-mapping, crystallization [64] | CLSM [51] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uhlenbrock, L.; Sixt, M.; Tegtmeier, M.; Schulz, H.; Hagels, H.; Ditz, R.; Strube, J. Natural Products Extraction of the Future—Sustainable Manufacturing Solutions for Societal Needs. Processes 2018, 6, 177. https://doi.org/10.3390/pr6100177
Uhlenbrock L, Sixt M, Tegtmeier M, Schulz H, Hagels H, Ditz R, Strube J. Natural Products Extraction of the Future—Sustainable Manufacturing Solutions for Societal Needs. Processes. 2018; 6(10):177. https://doi.org/10.3390/pr6100177
Chicago/Turabian StyleUhlenbrock, Lukas, Maximilian Sixt, Martin Tegtmeier, Hartwig Schulz, Hansjörg Hagels, Reinhard Ditz, and Jochen Strube. 2018. "Natural Products Extraction of the Future—Sustainable Manufacturing Solutions for Societal Needs" Processes 6, no. 10: 177. https://doi.org/10.3390/pr6100177
APA StyleUhlenbrock, L., Sixt, M., Tegtmeier, M., Schulz, H., Hagels, H., Ditz, R., & Strube, J. (2018). Natural Products Extraction of the Future—Sustainable Manufacturing Solutions for Societal Needs. Processes, 6(10), 177. https://doi.org/10.3390/pr6100177





