Development and Validation of an Automated Stirred-Tank Photobioreactor for Astaxanthin Production from Haematococcus pluvialis
Abstract
1. Introduction
2. Materials and Methods
2.1. Haematococcus pluvialis and Cultivation Conditions
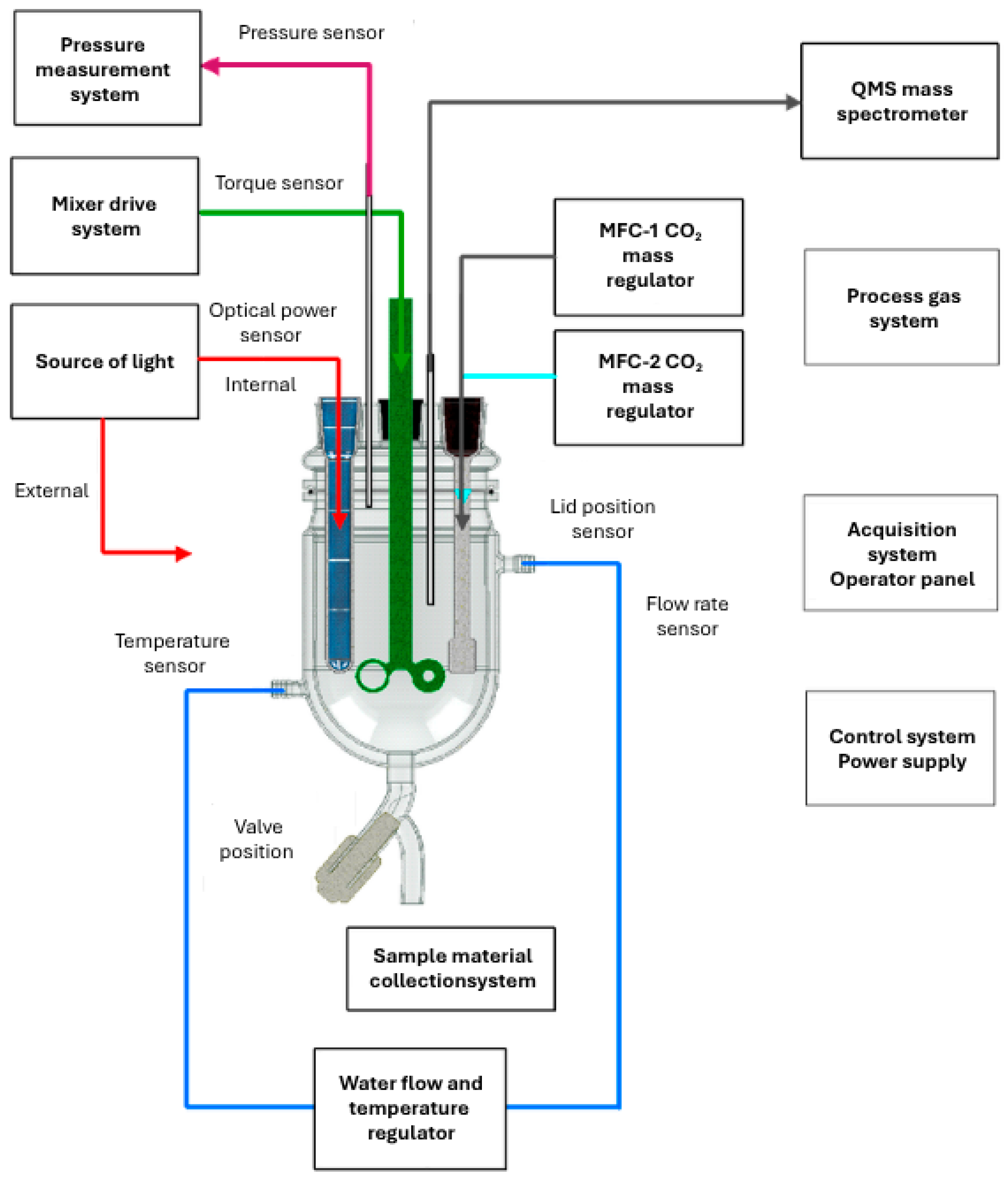
2.2. Construction and Mixing System
2.3. Lighting and Thermoregulation System
2.4. CO2 Dosing System
2.5. Control System
2.6. Analytical Methods
2.6.1. Biomass Growth Determination
2.6.2. Extraction and Enzymatic Hydrolysis of Astaxanthin Esters
2.6.3. HPLC Analysis
2.6.4. Bioactivity Assessment
2.6.5. Statistical Analysis
3. Results
3.1. Bioreactor Performance Characteristics and Process Stability
3.2. Growth Kinetics and Phase Transition
3.3. Biomass Production Yield and Metabolic Profile
3.4. Bioactivity and Cytotoxicity
4. Discussion
4.1. Bioprocess Efficiency in the Context of Advanced Control
4.2. Solving the Hydrodynamics Problem in ST-PBRs
4.3. Innovation in the Gas Dosing System
4.4. Product Bioactivity and the Extraction Method
5. Conclusions
- Validation of the ST-PBR concept for sensitive cultures: It was demonstrated that appropriate modification of the mixing system (application of a dual marine-type impeller and a brushless drive with precise torque control) allows for the efficient cultivation of H. pluvialis in a stirred-tank reactor. This dispels the common belief regarding the unsuitability of classical ST-PBRs for algal bioprocesses due to shear forces.
- Biosynthesis yield: The integrated system for controlling stress parameters (light/nutrients) resulted in an astaxanthin yield of 4.12% DW. This result surpasses the standard performance of open systems and is competitive with advanced tubular photobioreactors, while maintaining lower investment costs.
- Innovation in pH control: Replacing chemical buffers with precise CO2 dosing (using medical regulators) enabled residue-free pH stabilization. This strategy not only simplifies process operation but also eliminates the risk of product contamination by salts formed during acid-base neutralization.
- Product quality: The application of mild processing conditions and enzymatic extraction preserved the high biological activity of astaxanthin. The increase in cell viability to 128.1% in the MTS assay proves that the obtained product possesses proliferative potential and is free from toxic degradation products.
- Implementation potential: Through the application of industrial standards, the developed prototype constitutes a viable research platform, scalable to pilot volumes. It aligns with Industry 4.0 requirements through its capacity for multiparameter virtualization and automated cultivation control, facilitating future scale-up to industrial conditions. Furthermore, future research will focus on a direct, parallel comparative analysis between the developed ST-PBR and an air-lift photobioreactor to comprehensively evaluate their respective hydrodynamic, energetic, and mass transfer efficiencies.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ST-PBR | Stirred-Tank Photobioreactor |
| DW | Dry weight |
| TdST | Time-dependent Scheduled Tasks |
| PBR | Photobioreactor |
| CAPEX | Capital expenditures |
| MFCs | Mass flow controllers |
| BBM | Bold’s Basal Medium |
| PAR | Photosynthetically active radiation |
| PTFE | Polytetrafluoroethylene |
| HPLC | High-performance liquid chromatography |
| DMSO | Dimethyl sulfoxide |
| MTS | 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium |
References
- Wilawan, B.; Chan, S.S.; Ling, T.C.; Show, P.L.; Ng, E.-P.; Jonglertjunya, W.; Phadungbut, P.; Khoo, K.S. Advancement of carotenogenesis of astaxanthin from Haematococcus pluvialis: Recent insight and way forward. Mol. Biotechnol. 2024, 66, 402–423. [Google Scholar] [CrossRef]
- Ambati, R.R.; Phang, S.M.; Ravi, S.; Aswathanarayana, R.G. Astaxanthin: Sources, extraction, stability, biological activities and its commercial applications—A review. Mar. Drugs 2014, 12, 128–152. [Google Scholar] [CrossRef]
- Konwar, G.; Banik, R.; Geed, S.R. Bioprocess strategies for bioethanol production from Algae-Derived Carbohydrates: Scalable solutions and industry prospects in a circular bioeconomy. Chem. Eng. J. 2025, 515, 163456. [Google Scholar] [CrossRef]
- Capelli, B.; Bagchi, D.; Cysewski, G.R. Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and in terms of safety. Nutrafoods 2013, 12, 153–162. [Google Scholar] [CrossRef]
- Panis, G.; Carreon, J.R. Commercial astaxanthin production derived by green alga Haematococcus pluvialis: A microalgae process model and a techno-economic assessment all through production line. Algal Res. 2016, 18, 175–190. [Google Scholar] [CrossRef]
- Shah, M.M.R.; Liang, Y.; Cheng, J.J.; Daroch, M. Astaxanthin-producing green microalga Haematococcus pluvialis: From single cell to high value commercial products. Front. Plant Sci. 2016, 7, 531. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhu, D.; Niu, J.; Shen, S.; Wang, G. An economic assessment of astaxanthin production by large scale cultivation of Haematococcus pluvialis. Biotechnol. Adv. 2011, 29, 568–574. [Google Scholar] [CrossRef] [PubMed]
- Suh, I.S.; Joo, H.N.; Lee, C.G. A novel double-layered photobioreactor for simultaneous Haematococcus pluvialis cell growth and astaxanthin accumulation. J. Biotechnol. 2006, 125, 540–546. [Google Scholar] [CrossRef]
- Palladino, F.; Marcelino, P.R.F.; Schlogl, A.E.; José, Á.H.M.; Rodrigues, R.C.L.B.; Fabrino, D.L.; Santos, I.J.B.; Rosa, C.A. Bioreactors: Applications and innovations for a sustainable and healthy future—A critical review. Appl. Sci. 2024, 14, 9346. [Google Scholar] [CrossRef]
- Garcia-Ochoa, F.; Gomez, E. Bioreactor scale-up and oxygen transfer rate in microbial processes: An overview. Biotechnol. Adv. 2009, 27, 153–176. [Google Scholar] [CrossRef]
- Wang, B.; Lan, C.Q.; Horsman, M. Closed photobioreactors for production of microalgal biomasses. Biotechnol. Adv. 2012, 30, 904–912. [Google Scholar] [CrossRef] [PubMed]
- Borowiak, D.; Lenartowicz, P.; Grzebyk, M.; Wiśniewski, M.; Lipok, J.; Kafarski, P. Novel, automated, semi-industrial modular photobioreactor system for cultivation of demanding microalgae that produce fine chemicals—The next story of H. pluvialis and astaxanthin. Algal Res. 2021, 53, 102151. [Google Scholar] [CrossRef]
- Enzing, C.; Ploeg, M.; Barbosa, M.; Sijtsma, L. Microalgae-Based Products for the Food and Feed Sector: An Outlook for Europe; JRC Scientific and Policy Reports; Joint Research Centre: Seville, Spain, 2014. [Google Scholar] [CrossRef]
- Khoo, K.S.; Lee, S.Y.; Ooi, C.W.; Fu, X.; Miao, X.; Ling, T.C.; Show, P.L. Recent advances in biorefinery of astaxanthin from Haematococcus pluvialis. Bioresour. Technol. 2019, 288, 121606. [Google Scholar] [CrossRef]
- Benner, P.; Lüdtke, F.J.; Beyer, N.; von den Eichen, N.; Oropeza Vargas, J.E.; Weuster-Botz, D. LED illumination modules enable automated photoautotrophic cultivation of microalgae in parallel milliliter-scale stirred-tank bioreactors. Appl. Sci. 2023, 13, 5064. [Google Scholar] [CrossRef]
- Ren, Y.; Deng, J.; Huang, J.; Wu, Z.; Yi, L.; Bi, Y.; Chen, F. Using green alga Haematococcus pluvialis for astaxanthin and lipid co-production: Advances and outlook. Bioresour. Technol. 2021, 340, 125736. [Google Scholar] [CrossRef]
- Lorenz, R.T.; Cysewski, G.R. Commercial potential for Haematococcus microalgae as a natural source of astaxanthin. Trends Biotechnol. 2000, 18, 160–167. [Google Scholar] [CrossRef]
- Sangeetha, J.; Thangadurai, D. (Eds.) Microalgal Biomass for Bioeconomy Applications; Woodhead Publishing: Duxford, UK, 2023. [Google Scholar]
- Li, X.; Wang, X.; Duan, C.; Yi, S.; Gao, Z.; Xiao, C.; Agathos, S.N.; Wang, G.; Li, J. Biotechnological production of astaxanthin from the microalga Haematococcus pluvialis. Biotechnol. Adv. 2020, 43, 107602. [Google Scholar] [CrossRef]
- Vandenberghe, L.P.S.; Soccol, C.R.; Medeiros, A.B.P.; Spier, M.R. Application of different types of bioreactors in bioprocesses. In Bioreactors: Design, Properties and Applications; Antolli, P.G., Liu, Z., Eds.; Nova Science Publishers: Hauppauge, NY, USA, 2011; pp. 55–90. [Google Scholar]
- Koller, M.; Salerno, A.; Tuffner, P.; Koinigg, M.; Böchzelt, H.; Schober, S.; Pieber, S.; Schnitzer, H.; Mittelbach, M.; Braunegg, G. Characteristics and potential of micro algal cultivation strategies: A review. J. Clean. Prod. 2012, 37, 377–388. [Google Scholar] [CrossRef]
- Jerney, J.; Spilling, K. Large scale cultivation of microalgae: Open and closed systems. Methods Mol. Biol. 2020, 2096, 71–87. [Google Scholar] [CrossRef]
- Tredici, M.R.; Rodolfi, L.; Biondi, N.; Bassi, N.; Sampietro, G. Energy balance of algal biomass production in a 1-ha “Green Wall Panel” plant: How to produce algal biomass in a closed reactor achieving a high net energy ratio. Appl. Energy 2015, 154, 1103–1111. [Google Scholar] [CrossRef]
- Posten, C. Design principles of photo-bioreactors for cultivation of microalgae. Eng. Life Sci. 2009, 9, 165–177. [Google Scholar] [CrossRef]
- Uyar, B.; Djibrine Ali, M.; Ozer Uyar, G.E. Design parameters comparison of bubble column, airlift and stirred tank photobioreactors for microalgae production. Bioprocess Biosyst. Eng. 2024, 47, 195–209. [Google Scholar] [CrossRef] [PubMed]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef]
- Aslanbay Guler, B.; Deniz, I.; Demirel, Z.; Suphi, S.O.; Imamoglu, E. Computational fluid dynamics modelling of stirred tank photobioreactor for Haematococcus pluvialis production: Hydrodynamics and mixing conditions. Algal Res. 2020, 47, 101854. [Google Scholar] [CrossRef]
- Xu, C.; Liu, X.; Gu, X. Computational studies of airlift and stirred airlift bioreactors with non-Newtonian fluid: A comparison of hydrodynamics and gas-liquid mass transfer. J. Environ. Chem. Eng. 2023, 11, 110800. [Google Scholar] [CrossRef]
- Yoo, J.J.; Choi, S.P.; Kim, B.W.; Sim, S.J. Optimal design of scalable photo-bioreactor for phototropic culturing of Haematococcus pluvialis. Bioprocess Biosyst. Eng. 2012, 35, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Deniz, I. Scaling-up of Haematococcus pluvialis production in stirred tank photobioreactor. Bioresour. Technol. 2020, 310, 123434. [Google Scholar] [CrossRef]
- Michels, M.H.; van der Goot, A.J.; Norsker, N.H.; Wijffels, R.H. Effects of shear stress on the microalgae Chaetoceros muelleri. Bioprocess Biosyst. Eng. 2010, 33, 921–927. [Google Scholar] [CrossRef]
- Wu, K.; Ying, K.; Zhou, J.; Liu, D.; Liu, L.; Tao, Y.; Hanotu, J.; Zhu, X.; Cai, Z. Optimizing the growth of Haematococcus pluvialis based on a novel microbubble-driven photobioreactor. iScience 2021, 24, 103461. [Google Scholar] [CrossRef]
- Saccardo, A.; Bezzo, F.; Sforza, E. Microalgae growth in ultra-thin steady-state continuous photobioreactors: Assessing self-shading effects. Front. Bioeng. Biotechnol. 2022, 10, 977429. [Google Scholar] [CrossRef]
- Lucato, V. Capturing CO2 and N2 from the Atmosphere: Nitrogen-Fixing Cyanobacteria for Sustainable Industrial Production of Proteins. Ph.D. Thesis, University of Padua, Padua, Italy, 2025. [Google Scholar]
- Catapano, G.; Czermak, P.; Eibl, R.; Eibl, D.; Pörtner, R. Bioreactor design and scale-up. In Cell and Tissue Reaction Engineering; Eibl, R., Eibl, D., Pörtner, R., Catapano, G., Czermak, P., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 173–259. [Google Scholar] [CrossRef]
- Fernandes, B.D.; Mota, A.; Teixeira, J.A.; Vicente, A.A. Continuous cultivation of photosynthetic microorganisms: Approaches, applications and future trends. Biotechnol. Adv. 2015, 33, 1228–1245. [Google Scholar] [CrossRef]
- Lakaniemi, A.-M. Microalgal Cultivation and Utilization in Sustainable Energy Production. Ph.D. Thesis, Tampere University of Technology, Tampere, Finland, 2012. [Google Scholar]
- Dong, S.; Huang, Y.; Zhang, R.; Wang, S.; Liu, Y. Four different methods comparison for extraction of astaxanthin from green alga Haematococcus pluvialis. Sci. World J. 2014, 2014, 694305. [Google Scholar] [CrossRef] [PubMed]
- Machmudah, S.; Shotipruk, A.; Goto, M.; Sasaki, M.; Hirose, T. Extraction of astaxanthin from Haematococcus pluvialis using supercritical CO2 and ethanol as entrainer. Ind. Eng. Chem. Res. 2006, 45, 3652–3657. [Google Scholar] [CrossRef]
- Sarada, R.; Vidhyavathi, R.; Usha, D.; Ravishankar, G.A. An efficient method for extraction of astaxanthin from green alga Haematococcus pluvialis. J. Agric. Food Chem. 2006, 54, 7585–7588. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, H.W.; Bold, H.C. Phycological studies IV. Some soil algae from Enchanted Rock and related algal species. Univ. Tex. Publ. 1963, 6318, 1–95. [Google Scholar]
- Fábregas, J.; Domínguez, A.; Regueiro, M.; Maseda, A.; Otero, A. Optimization of culture medium for the continuous cultivation of the microalga Haematococcus pluvialis. Appl. Microbiol. Biotechnol. 2000, 53, 530–535. [Google Scholar] [CrossRef]
- García-Malea, M.C.; Acién, F.G.; Fernández, J.M.; Cerón, M.C.; Molina, E. Continuous production of green cells of Haematococcus pluvialis: Modeling of the irradiance effect. Enzyme Microb. Technol. 2006, 38, 981–989. [Google Scholar] [CrossRef]
- Wang, S.; Meng, Y.; Liu, J.; Cao, X.; Xue, S. Accurate quantification of astaxanthin from Haematococcus pluvialis using DMSO extraction and lipase-aided hydrolysis pretreatment. Algal Res. 2018, 35, 427–431. [Google Scholar] [CrossRef]
- Jacobs, P.B.; LeBoeuf, R.D.; McCommas, S.A.; Tauber, J.D. The cleavage of carotenoid esters by cholesterol esterase. Comp. Biochem. Physiol. Part B Comp. Biochem. 1982, 72, 157–160. [Google Scholar] [CrossRef]
- Yuan, J.P.; Chen, F. Hydrolysis kinetics of astaxanthin esters and stability of astaxanthin of Haematococcus pluvialis during saponification. J. Agric. Food Chem. 1999, 47, 31–35. [Google Scholar] [CrossRef]
- Miao, F.; Lu, D.; Li, Y.; Zeng, M. Characterization of astaxanthin esters in Haematococcus pluvialis by liquid chromatography–atmospheric pressure chemical ionization mass spectrometry. Anal. Biochem. 2006, 352, 176–181. [Google Scholar] [CrossRef]
- Cory, A.H.; Owen, T.C.; Barltrop, J.A.; Cory, J.G. Use of an aqueous soluble tetrazolium/formazan assay for cell growth assays in culture. Cancer Commun. 1991, 3, 207–212. [Google Scholar] [CrossRef]
- Berridge, M.V.; Herst, P.M.; Tan, A.S. Tetrazolium dyes as tools in cell biology: New insights into their cellular reduction. Biotechnol. Annu. Rev. 2005, 11, 127–152. [Google Scholar] [CrossRef]
- Montgomery, D.C. Design and Analysis of Experiments, 9th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Aflalo, C.; Meshulam, Y.; Zarka, A.; Boussiba, S. On the relative efficiency of two- vs. one-stage production of astaxanthin by the green alga Haematococcus pluvialis. Biotechnol. Bioeng. 2007, 98, 300–305. [Google Scholar] [CrossRef]
- Boussiba, S. Carotenogenesis in the green alga Haematococcus pluvialis: Cellular physiology and stress response. Physiol. Plant. 2000, 108, 111–117. [Google Scholar] [CrossRef]
- Carvalho, A.P.; Meireles, L.A.; Malcata, F.X. Microalgal reactors: A review of enclosed system designs and performances. Biotechnol. Prog. 2006, 22, 1490–1506. [Google Scholar] [CrossRef]
- García-Malea, M.C.; Acién, F.G.; Del Río, E.; Fernández, J.M.; Cerón, M.C.; Molina, E. Comparative analysis of the outdoor culture of Haematococcus pluvialis in tubular and bubble column photobioreactors. J. Biotechnol. 2006, 123, 329–342. [Google Scholar] [CrossRef]
- He, H.; Liu, Y.; Zhang, A.; Yang, Z.; Liu, X.; Yang, R.; Tang, H.; Li, Z. Kinetic modeling and experimental verification of a swirl flocculation-enhanced reactor: A case study of coal chemical wastewater pretreatment. Sep. Purif. Technol. 2023, 326, 124852. [Google Scholar] [CrossRef]
- Ozkan, A.; Berberoglu, H. Adhesion of algal cells to surfaces. Biofouling 2013, 29, 469–482. [Google Scholar] [CrossRef]
- Hoshan, L.; Jiang, R.; Moroney, J.; Bui, A.; Zhang, X.; Hang, T.-C. Effective bioreactor pH control using only sparging gases. Biotechnol. Prog. 2019, 35, e2743. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Kao, P.-C.; Tan, C.H.; Show, P.L.; Cheah, W.Y.; Lee, W.L.; Ling, T.C.; Chang, J.-S. Using an innovative pH-stat CO2 feeding strategy to enhance cell growth and C-phycocyanin production from Spirulina platensis. Biochem. Eng. J. 2016, 112, 78–85. [Google Scholar] [CrossRef]
- Guerin, M.; Huntley, M.E.; Olaizola, M. Haematococcus astaxanthin: Applications for human health and nutrition. Trends Biotechnol. 2003, 21, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.P.; Peng, J.; Yin, K.; Wang, J.H. Potential health-promoting effects of astaxanthin: A high-value carotenoid mostly from microalgae. Mol. Nutr. Food Res. 2011, 55, 150–165. [Google Scholar] [CrossRef] [PubMed]
- Kidd, P. Astaxanthin, cell membrane nutrient with diverse clinical benefits and anti-aging potential. Altern. Med. Rev. 2011, 16, 355–364. [Google Scholar]
- Higuera-Ciapara, I.; Felix-Valenzuela, L.; Goycoolea, F.M. Astaxanthin: A review of its chemistry and applications. Crit. Rev. Food Sci. Nutr. 2006, 46, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Fassett, R.G.; Coombes, J.S. Astaxanthin: A potential therapeutic agent in cardiovascular disease. Mar. Drugs 2011, 9, 447–465. [Google Scholar] [CrossRef]


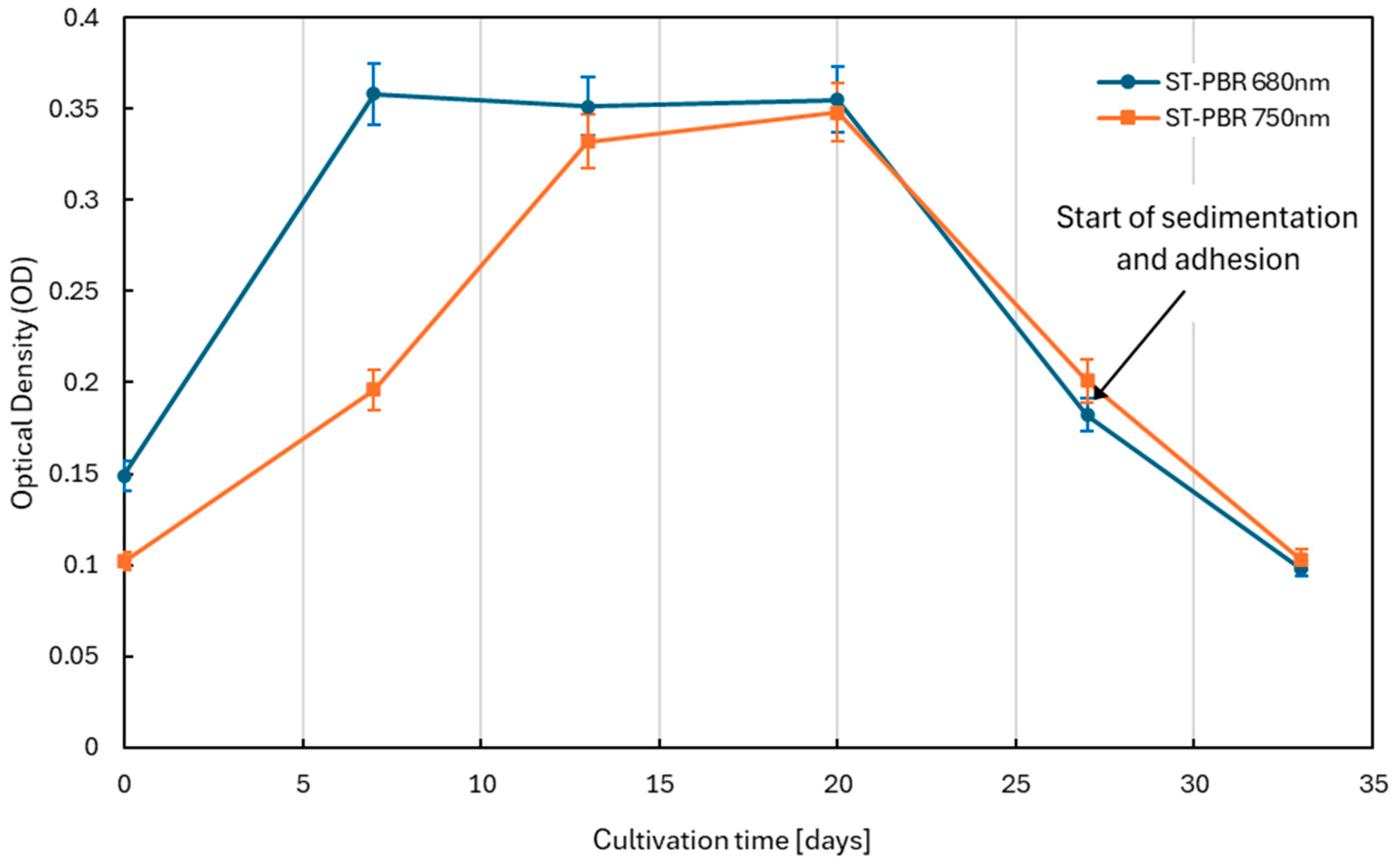


| Cultivation Day | Cultivation Stage | Dry Biomass Weight (20 mL Sample) [g] |
|---|---|---|
| Day 1 | Start | 0.0029 ± 0.0002 |
| Day 8 | Exponential growth | 0.0175 ± 0.0011 |
| Day 14 | Max. green phase | 0.0241 ± 0.0010 |
| Day 21 | Stress induction/Onset of adhesion | 0.0249 ± 0.0013 |
| Day 28 | Red phase | 0.0256 ± 0.0015 |
| Day 34 | Final harvest | 0.0263 ± 0.0011 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Rudnicki, P.; Wiewiórski, P.; Kowalik, A.; Kaleta, J. Development and Validation of an Automated Stirred-Tank Photobioreactor for Astaxanthin Production from Haematococcus pluvialis. Processes 2026, 14, 1230. https://doi.org/10.3390/pr14081230
Rudnicki P, Wiewiórski P, Kowalik A, Kaleta J. Development and Validation of an Automated Stirred-Tank Photobioreactor for Astaxanthin Production from Haematococcus pluvialis. Processes. 2026; 14(8):1230. https://doi.org/10.3390/pr14081230
Chicago/Turabian StyleRudnicki, Piotr, Przemysław Wiewiórski, Adam Kowalik, and Jerzy Kaleta. 2026. "Development and Validation of an Automated Stirred-Tank Photobioreactor for Astaxanthin Production from Haematococcus pluvialis" Processes 14, no. 8: 1230. https://doi.org/10.3390/pr14081230
APA StyleRudnicki, P., Wiewiórski, P., Kowalik, A., & Kaleta, J. (2026). Development and Validation of an Automated Stirred-Tank Photobioreactor for Astaxanthin Production from Haematococcus pluvialis. Processes, 14(8), 1230. https://doi.org/10.3390/pr14081230







