Laser-Synthesized Vanadium-Based Nanoparticles on TiO2 Nanotubes for Photocatalytic Degradation of Acid Yellow 23
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Chemicals
2.2. Synthesis Processes
2.2.1. Synthesis of V Nanoparticles
2.2.2. Synthesis of TiO2 Nanotubes
2.2.3. Synthesis of V-TiO2 Samples
2.3. Characterization of Prepared V Nanoparticles
2.4. Characterization of Prepared V-TiO2 Samples
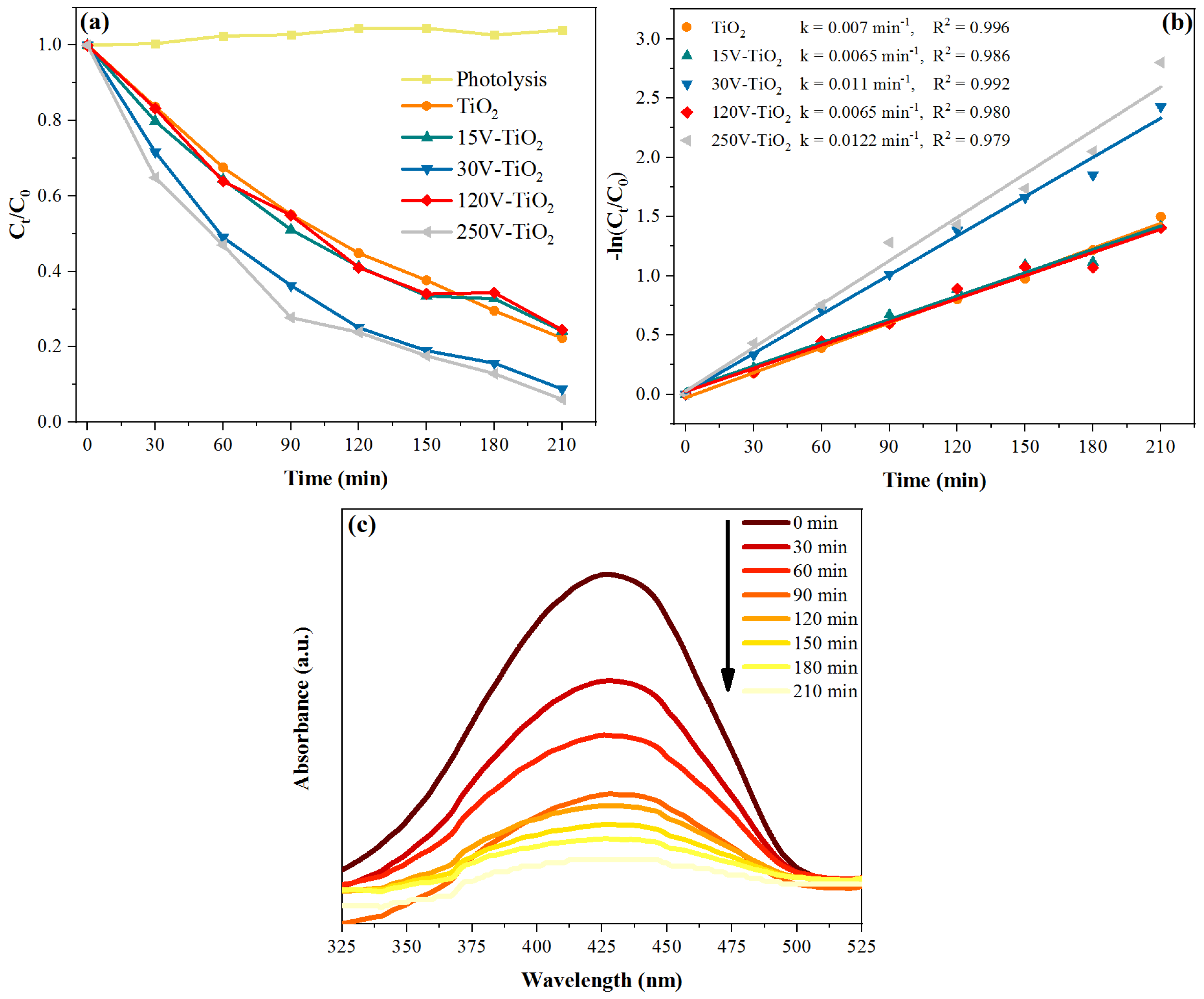
2.5. Photocatalytic Degradation of Acid Yellow 23
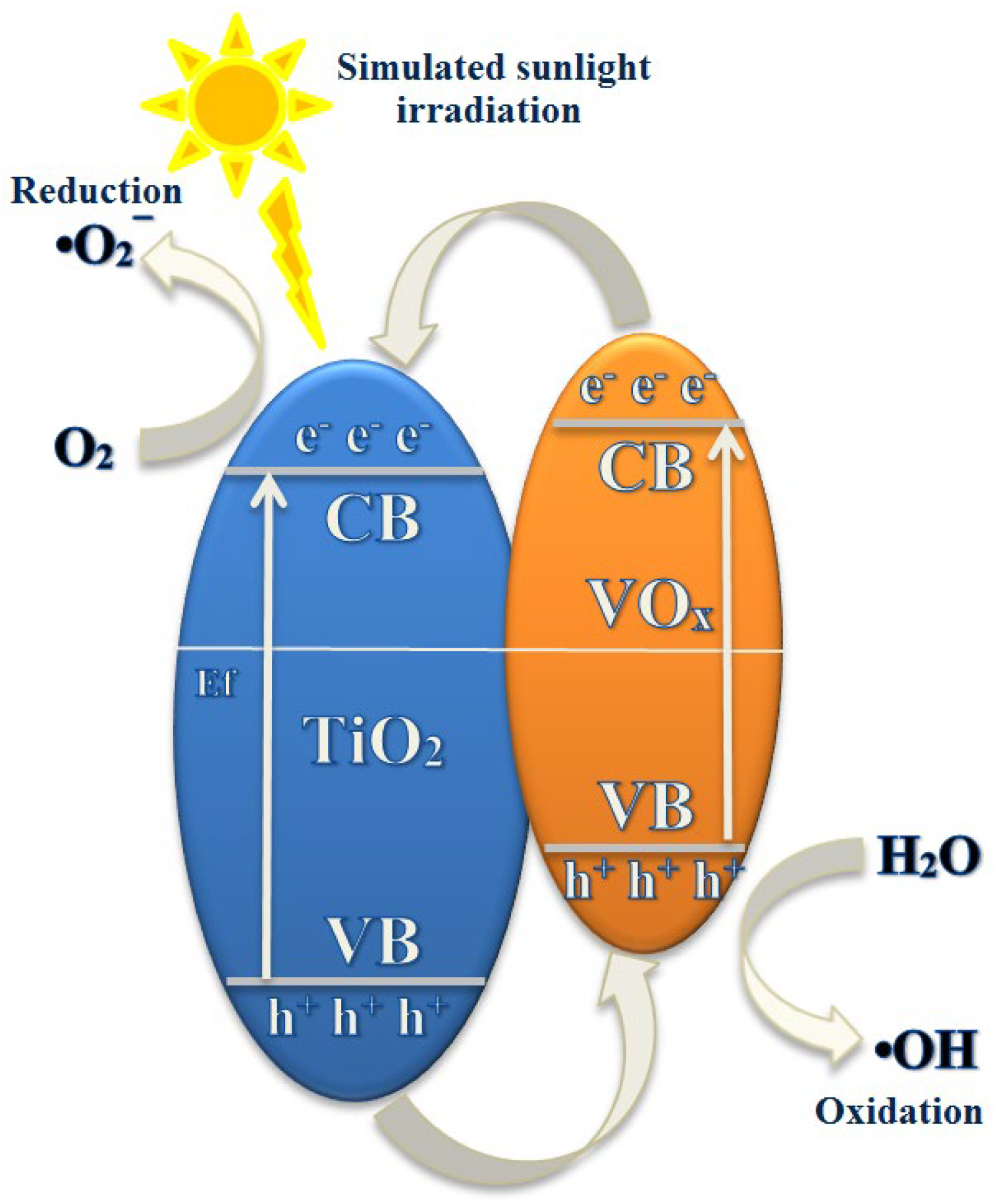
3. Results and Discussion
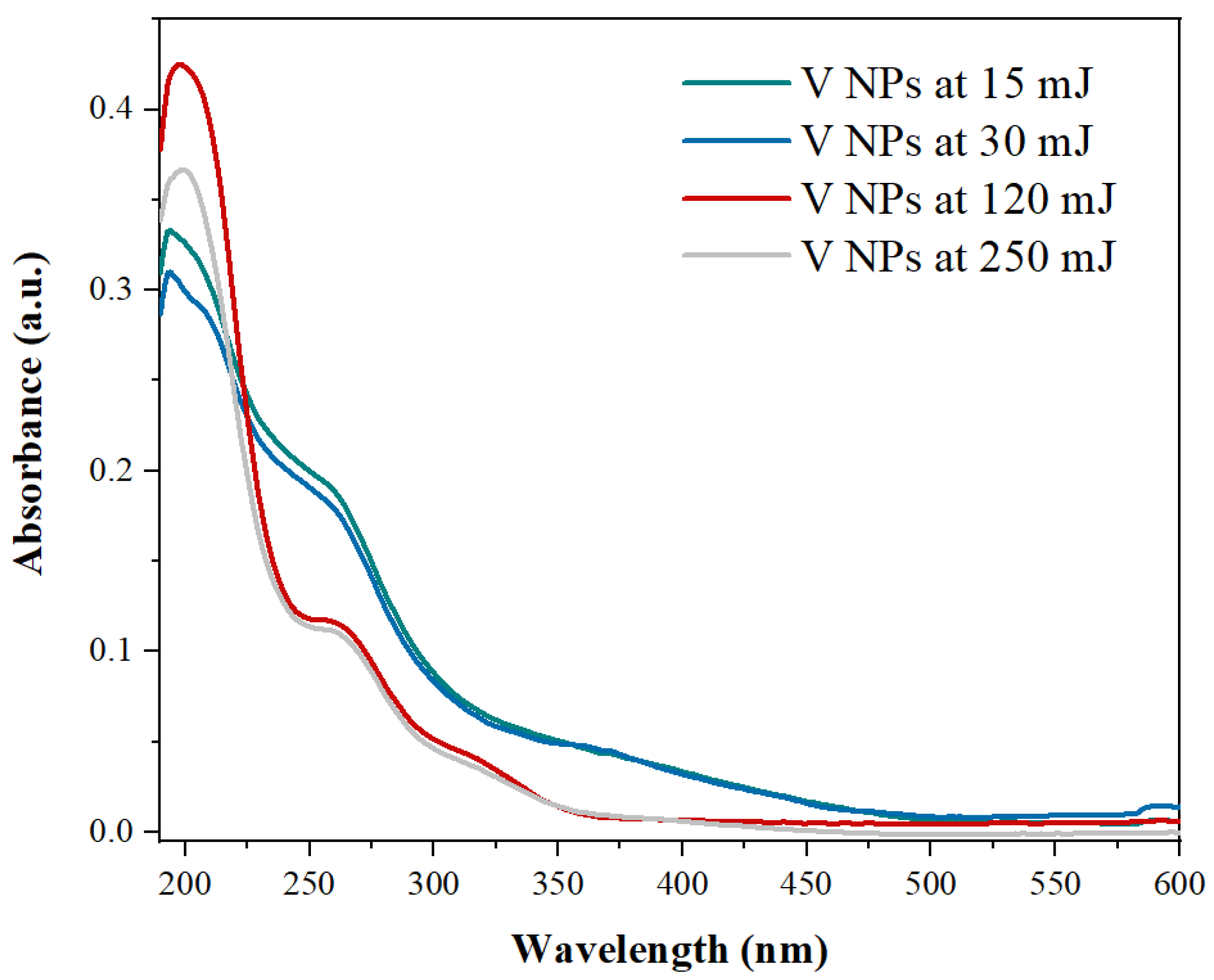
3.1. Characterization of V Nanoparticles
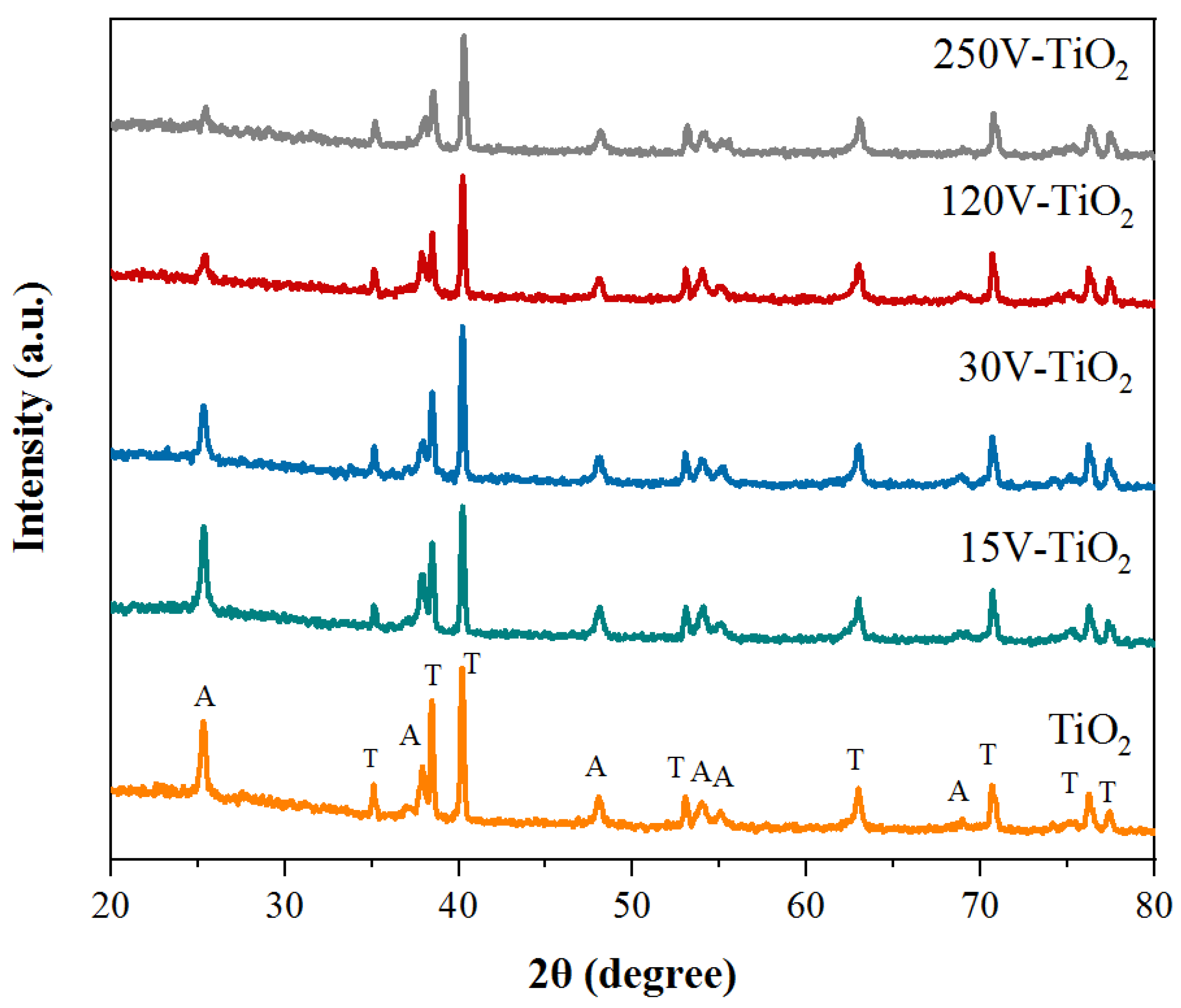
3.2. Examination of V-TiO2 Synthesis Parameters
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tkaczyk, A.; Mitrowska, K.; Posyniak, A. Synthetic Organic Dyes as Contaminants of the Aquatic Environment and Their Implications for Ecosystems: A Review. Sci. Total Environ. 2020, 717, 137222. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.M.; Aidid, A.R.; Mohshin, J.N.; Mondal, H.; Ganguli, S.; Chakraborty, A.K. A Critical Review on Textile Dye-Containing Wastewater: Ecotoxicity, Health Risks, and Remediation Strategies for Environmental Safety. Clean. Chem. Eng. 2025, 11, 100165. [Google Scholar] [CrossRef]
- Benkhaya, S.; M’rabet, S.; El Harfi, A. Classifications, Properties, Recent Synthesis and Applications of Azo Dyes. Heliyon 2020, 6, e03271. [Google Scholar] [CrossRef]
- Behnajady, M.A.; Modirshahla, N.; Hamzavi, R. Kinetic Study on Photocatalytic Degradation of C.I. Acid Yellow 23 by ZnO Photocatalyst. J. Hazard. Mater. 2006, 133, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.; Ko, J.; Ko, W. Synthesis of Gd2O3 Nanoparticles and Their Photocatalytic Activity for Degradation of Azo Dyes. Catalysts 2021, 11, 742. [Google Scholar] [CrossRef]
- Navidpour, A.H.; Abbasi, S.; Li, D.; Mojiri, A.; Zhou, J.L. Investigation of Advanced Oxidation Process in the Presence of TiO2 Semiconductor as Photocatalyst: Property, Principle, Kinetic Analysis, and Photocatalytic Activity. Catalysts 2023, 13, 232. [Google Scholar] [CrossRef]
- Fujishima, A.; Zhang, X.; Tryk, D.A. TiO2 Photocatalysis and Related Surface Phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Martin, S.T.; Choi, W.; Bahnemann, D.W. Environmental Applications of Semiconductor Photocatalysis. Chem. Rev. 1995, 95, 69–96. [Google Scholar] [CrossRef]
- Moreno-Vargas, J.M.; Echeverry-Cardona, L.M.; Torres-Ceron, D.A.; Amaya-Roncancio, S.; Restrepo-Parra, E.; Castillo-Delgado, K.J. Photocatalysis as an Alternative for the Remediation of Wastewater: A Scientometric Review. ChemEngineering 2024, 8, 95. [Google Scholar] [CrossRef]
- Chalatsi-Diamanti, P.; Isari, E.A.; Grilla, E.; Kokkinos, P.; Kalavrouziotis, I.K. Recent Prospects, Challenges and Advancements of Photocatalysis as a Wastewater Treatment Method. Water Emerg. Contam. Nanoplastics 2025, 4, 12. [Google Scholar] [CrossRef]
- Liu, H.; Wu, Y.; Zhang, J. A New Approach toward Carbon-Modified Vanadium-Doped Titanium Dioxide Photocatalysts. ACS Appl. Mater. Interfaces 2011, 3, 1757–1764. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, G.; Leng, X.; Sun, Z.; Zheng, S. Characterization and Improved Solar Light Activity of Vanadium Doped TiO2/Diatomite Hybrid Catalysts. J. Hazard. Mater. 2015, 285, 212–220. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Nghiem, T.T.; Hoang, H.H.; Cao, H.H.; Nguyen, V.A. Vanadium-Doped TiO2 Adsorbent-Photocatalyst for Organic Dye Treatment. Vietnam J. Catal. Adsorpt. 2024, 12, 13–18. [Google Scholar] [CrossRef]
- Christoforidis, K.C.; Fernández-García, M. Photoactivity and Charge Trapping Sites in Copper and Vanadium Doped Anatase TiO2 Nano-Materials. Catal. Sci. Technol. 2016, 6, 1094–1105. [Google Scholar] [CrossRef]
- Šuligoj, A.; Povirk, N.; Maver, K.; Mavrič, A.; Lavrenčič Štangar, U.; Surca, A.K. Transparent Vanadium Doped Titania-Silica Films: Structural Characterization and Self-Cleaning Properties. J. Environ. Chem. Eng. 2024, 12, 113904. [Google Scholar] [CrossRef]
- Zhang, D.; Gökce, B.; Barcikowski, S. Laser Synthesis and Processing of Colloids: Fundamentals and Applications. Chem. Rev. 2017, 117, 3990–4103. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Chrisey, D.B. Pulsed Laser Ablation in Liquid for Micro-/Nanostructure Generation. J. Photochem. Photobiol. C Photochem. Rev. 2012, 13, 204–223. [Google Scholar] [CrossRef]
- Krstulović, N.; Salamon, K.; Budimlija, O.; Kovač, J.; Dasović, J.; Umek, P.; Capan, I. Parameters Optimization for Synthesis of Al-Doped ZnO Nanoparticles by Laser Ablation in Water. Appl. Surf. Sci. 2018, 440, 916–925. [Google Scholar] [CrossRef]
- Gurbandurdyyev, B.; Annamuradov, B.; Er, S.B.; Gross, B.; Er, A.O. Advances in Pulsed Liquid-Based Nanoparticles: From Synthesis Mechanism to Application and Machine Learning Integration. Quantum Beam Sci. 2025, 9, 32. [Google Scholar] [CrossRef]
- Niu, S.; Wang, W.; Liu, P.; Zhang, Y.; Zhao, X.; Li, J.; Xiao, M.; Wang, Y.; Li, J.; Shao, X. Recent Advances in Applications of Ultrafast Lasers. Photonics 2024, 11, 857. [Google Scholar] [CrossRef]
- Nyabadza, A.; Vazquez, M.; Brabazon, D. A Review of Bimetallic and Monometallic Nanoparticle Synthesis via Laser Ablation in Liquid. Crystals 2023, 13, 253. [Google Scholar] [CrossRef]
- Balachandran, A.; Sreenilayam, S.P.; Madanan, K.; Thomas, S.; Brabazon, D. Nanoparticle Production via Laser Ablation Synthesis in Solution Method and Printed Electronic Application—A Brief Review. Results Eng. 2022, 16, 100646. [Google Scholar] [CrossRef]
- Blažeka, D.; Radičić, R.; Maletić, D.; Živković, S.; Momčilović, M.; Krstulović, N. Enhancement of Methylene Blue Photodegradation Rate Using Laser Synthesized Ag-Doped ZnO Nanoparticles. Nanomaterials 2022, 12, 2677. [Google Scholar] [CrossRef] [PubMed]
- Radičić, R.; Jurov, A.; Zavašnik, J.; Kovač, J.; Brusar, V.; Vdović, S.; Novko, D.; Krstulović, N. UV and Solar-Driven Photocatalysis of Organic Dyes Using ZnO-Ag Heterojunction Nanoparticles Synthesized by One-Step Laser Synthesis in Water. Appl. Surf. Sci. 2024, 669, 160498. [Google Scholar] [CrossRef]
- Sacco, O.; Sannino, D.; Matarangolo, M.; Vaiano, V. Room Temperature Synthesis of V-Doped TiO2 and Its Photocatalytic Activity in the Removal of Caffeine under UV Irradiation. Materials 2019, 12, 911. [Google Scholar] [CrossRef]
- Iketani, K.; Sun, R.D.; Toki, M.; Hirota, K.; Yamaguchi, O. Sol-Gel-Derived VχTi1-ΧO2 Films and Their Photocatalytic Activities under Visible Light Irradiation. Mater. Sci. Eng. B 2004, 108, 187–193. [Google Scholar] [CrossRef]
- Nguyen, D.M.; Ding, S.; Nghiem, T.T.; Nguyen, V.A.; Mejía, E. Visible Light-Driven Degradation of Trichloroethylene in Aqueous Phase with Vanadium-Doped TiO2 Photocatalysts. Sol. RRL 2023, 7, 2200938. [Google Scholar] [CrossRef]
- Tošić, M.; Rajić, V.; Pjević, D.; Stojadinović, S.; Perović, I.; Krstulović, N.; Momčilović, M. Vanadium-Modified TiO2 Nanotubes Synthesized Via Electrochemical Anodization and Nanosecond Pulsed Laser Deposition for Improved Photocatalytic Degradation of p-Nitrophenol. J. Clust. Sci. 2026, 37, 1. [Google Scholar] [CrossRef]
- Ramos, B.; Ookawara, S.; Matsushita, Y.; Yoshikawa, S. Intensification of Solar Photocatalysis with Immobilised TiO2 by Using Micro-Structured Reaction Spaces. J. Environ. Chem. Eng. 2015, 3, 681–688. [Google Scholar] [CrossRef]
- Manassero, A.; Satuf, M.L.; Alfano, O.M. Photocatalytic Reactors with Suspended and Immobilized TiO2: Comparative Efficiency Evaluation. Chem. Eng. J. 2017, 326, 29–36. [Google Scholar] [CrossRef]
- Souza, T.G.F.; Ciminelli, V.S.T.; Mohallem, N.D.S. A Comparison of TEM and DLS Methods to Characterize Size Distribution of Ceramic Nanoparticles. J. Phys. Conf. Ser. 2016, 733, 012039. [Google Scholar] [CrossRef]
- Zeng, H.; Du, X.W.; Singh, S.C.; Kulinich, S.A.; Yang, S.; He, J.; Cai, W. Nanomaterials via Laser Ablation/Irradiation in Liquid: A Review. Adv. Funct. Mater. 2012, 22, 1333–1353. [Google Scholar] [CrossRef]
- Forsythe, R.C.; Cox, C.P.; Wilsey, M.K.; Müller, A.M. Pulsed Laser in Liquids Made Nanomaterials for Catalysis. Chem. Rev. 2021, 121, 7568–7637. [Google Scholar] [CrossRef] [PubMed]
- Hotze, E.M.; Phenrat, T.; Lowry, G.V. Nanoparticle Aggregation: Challenges to Understanding Transport and Reactivity in the Environment. J. Environ. Qual. 2010, 39, 1909–1924. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 Photocatalysis: Mechanisms and Materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef]
- Bhattacharjee, S. DLS and Zeta Potential—What They Are and What They Are Not? J. Control. Release 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Bañares, M.A. Operando Spectroscopy: The Knowledge Bridge to Assessing Structure-Performance Relationships in Catalyst Nanoparticles. Adv. Mater. 2011, 23, 5293–5301. [Google Scholar] [CrossRef]
- Weckhuysen, B.M.; Keller, D.E. Chemistry, Spectroscopy and the Role of Supported Vanadium Oxides in Heterogeneous Catalysis. Catal. Today 2003, 78, 25–46. [Google Scholar] [CrossRef]
- Radko, M.; Kowalczyk, A.; Bidzińska, E.; Witkowski, S.; Górecka, S.; Wierzbicki, D.; Pamin, K.; Chmielarz, L. Titanium Dioxide Doped with Vanadium as Effective Catalyst for Selective Oxidation of Diphenyl Sulfide to Diphenyl Sulfonate. J. Therm. Anal. Calorim. 2018, 132, 1471–1480. [Google Scholar] [CrossRef]
- Stojadinović, S.; Radić, N.; Vasilić, R.; Tadić, N.; Tsanev, A. Photocatalytic Degradation of Methyl Orange in the Presence of Transition Metals (Mn, Ni, Co) Modified TiO2 Coatings Formed by Plasma Electrolytic Oxidation. Solid State Sci. 2022, 129, 106896. [Google Scholar] [CrossRef]
- Sondezi, N.; Njengele-Tetyana, Z.; Matabola, K.P.; Makhetha, T.A. Sol-Gel-Derived TiO2 and TiO2/Cu Nanoparticles: Synthesis, Characterization, and Antibacterial Efficacy. ACS Omega 2024, 9, 15959–15970. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, Y.; Ye, J.; Guo, M.; Chen, J.; Chen, W. Nonenzymatic Glucose Biosensors Based on Silver Nanoparticles Deposited on TiO2 Nanotubes. J. Nanotechnol. 2016, 6, 9454830. [Google Scholar] [CrossRef]
- Nguyen, N.T.; Altomare, M.; Yoo, J.E.; Taccardi, N.; Schmuki, P. Noble Metals on Anodic TiO2 Nanotube Mouths: Thermal Dewetting of Minimal Pt Co-Catalyst Loading Leads to Significantly Enhanced Photocatalytic H2 Generation. Adv. Energy Mater. 2016, 6, 1501926. [Google Scholar] [CrossRef]
- Rochkind, M.; Pasternak, S.; Paz, Y. Using Dyes for Evaluating Photocatalytic Properties: A Critical Review. Molecules 2015, 20, 88–110. [Google Scholar] [CrossRef]
- Pooseekheaw, P.; Thongpan, W.; Panthawan, A.; Kantarak, E.; Sroila, W.; Singjai, P. Porous V2O5/TiO2 nanoheterostructure Films with Enhanced Visible-Light Photocatalytic Performance Prepared by the Sparking Method. Molecules 2020, 25, 3327. [Google Scholar] [CrossRef]
- Rossi, L.; Palacio, M.; Villabrille, P.I.; Rosso, J.A. V-Doped TiO2 Photocatalysts and Their Application to Pollutant Degradation. Environ. Sci. Pollut. Res. 2021, 28, 24112–24123. [Google Scholar] [CrossRef]
- Tian, B.; Li, C.; Gu, F.; Jiang, H.; Hu, Y.; Zhang, J. Flame Sprayed V-Doped TiO2 Nanoparticles with Enhanced Photocatalytic Activity under Visible Light Irradiation. Chem. Eng. J. 2009, 151, 220–227. [Google Scholar] [CrossRef]
- Teghil, R.; D’Alessio, L.; De Bonis, A.; Galasso, A.; Ibris, N.; Salvi, A.M.; Santagata, A.; Villani, P. Nanoparticles and Thin Film Formation in Ultrashort Pulsed Laser Deposition of Vanadium Oxide. J. Phys. Chem. A 2009, 113, 14969–14974. [Google Scholar] [CrossRef]
- Syrti, L.; Kumar, E.V.; Arjun, A.; Nagaraju, G.; Priyanka, K.P. Unveiling the Bifunctional Potentiality of TiO2/V2O5 Nanocomposite for Photodegradation of Industrial Dyes and Latent Fingerprint Visualization in Forensic Investigation. Surf. Interfaces 2025, 71, 106840. [Google Scholar] [CrossRef]
- Khan, H.; Berk, D. Sol-Gel Synthesized Vanadium Doped TiO2 Photocatalyst: Physicochemical Properties and Visible Light Photocatalytic Studies. J. Sol-Gel Sci. Technol. 2013, 68, 180–192. [Google Scholar] [CrossRef]
- Murila, G.I.; Mageto, M.J.; Wafula, H.B.; Gaitho, F.M.; Mutuma, B.K. Tailoring Structural Phases and Photocatalytic Activity of Vanadium-Doped TiO2 Nanoparticles Prepared by Modified Sol–Gel Technique. MRS Adv. 2025. [Google Scholar] [CrossRef]
- Chang, P.Y.; Huang, C.H.; Doong, R. an Characterization and Photocatalytic Activity of Vanadium-Doped Titanium Dioxide Nanocatalysts. Water Sci. Technol. 2009, 59, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Li, H.; Zhang, J.; Chen, Z.; Han, G.; Song, B. Effects of Preparation Parameters on Room Temperature Formation of Vanadium-Doped TiO2 Nanocrystalline Powder. J. Am. Ceram. Soc. 2011, 94, 71–76. [Google Scholar] [CrossRef]






| 30 V-TiO2 (1 mg/L; 5 min) | 30 V-TiO2 (0.25 mg/L; 2.5 min) | |||
|---|---|---|---|---|
| Element | Weight % | Atom % | Weight % | Atom % |
| O | 37.088 | 63.853 | 38.695 | 65.410 |
| Ti | 61.973 | 35.639 | 60.581 | 34.205 |
| V | 0.939 | 0.508 | 0.724 | 0.384 |
| TiO2 | 15 V-TiO2 (0.25 mg/L; 2.5 min) | 30 V-TiO2 (0.25 mg/L; 2.5 min) | 120 V-TiO2 (0.25 mg/L; 2.5 min) | 250 V-TiO2 (0.25 mg/L; 2.5 min) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Element | Weight % | Atom % | Weigh % | Atom % | Weight % | Atom % | Weight % | Atom % | Weight % | Atom % |
| O | 30.564 | 56.856 | 25.935 | 51.185 | 38.695 | 65.410 | 32.714 | 59.239 | 21.033 | 43.388 |
| Ti | 69.436 | 43.144 | 73.861 | 48.689 | 60.581 | 34.205 | 66.439 | 40.255 | 77.735 | 54.796 |
| V | / | / | 0.203 | 0.126 | 0.724 | 0.384 | 0.794 | 0.452 | 1.231 | 0.816 |
| 15 V-TiO2 (0.25 mg/L; 2.5 min) | 15 V-TiO2 (0.5 mg/L; 2.5 min) | 120 V-TiO2 (0.25 mg/L; 2.5 min) | 120 V-TiO2 (0.5 mg/L; 2.5 min) | |||||
|---|---|---|---|---|---|---|---|---|
| Element | Weight % | Atom % | Weight % | Atom % | Weight % | Atom % | Weight % | Atom % |
| O | 25.935 | 51.185 | 11.542 | 28.110 | 32.714 | 59.239 | 31.595 | 58.053 |
| Ti | 73.861 | 48.689 | 87.041 | 70.806 | 66.439 | 40.255 | 67.446 | 41.393 |
| V | 0.203 | 0.126 | 1.417 | 1.084 | 0.794 | 0.452 | 0.960 | 0.554 |
| V-TiO2/ Synthesis Method | Catalyst Concentration/Area | Pollutant Concentration | Degradation/Light Source (Spectrum) | Ref. |
|---|---|---|---|---|
| V:Ti = 100:1/Sol–gel, calcined at 300 °C | 0.01 g in 250 mL | Methylene blue 0.026 mM | ~95%; 6 min/Hg lamp (Vis light) | [50] |
| V:Ti = 600:1/Sol–gel, calcined at 500 °C | 0.01 g in 250 mL | Methylene blue 0.026 mM | ~89%; 6 min lamp/Hg lamp (Vis light) | |
| V (5%)/Sol–gel | 50 mg | Methylene blue 5 mg/L | 95%; 60 min/(UV) | [51] |
| V (0.084 mol%)/ Hydrothermal | 20 mg in 10 mL | Trichloroethylene 1000 ppm | 72%; 210 min/LEDs lamp (UV) | [27] |
| V (0.134 mol%)/ Hydrothermal | 40 mg in 20 mL | Trichloroethylene 1000 ppm | 49%; 190 min/Xenon lamp (Visible light) | |
| V (0.036 mol%)/Sol–gel | 0.3 g in 100 mL | Caffeine 25 mg/L | 96%; 360 min/UV-LED strip | [25] |
| V (1 wt.%)/Sol–gel | 15 mg in 15 mL | Methylene blue 1 mM | 69%; 350 min/Sunlight simulation | [52] |
| V (0.15 mol%)/ Cohydrolysis | 10 mg in 15 mL | Methyl orange 10−4 mol/L | 75%; 12 d/Daylight irradiation | [53] |
| V (2.5 wt.%)/Electrochemical anodization, pulsed laser deposition, calcined at 450 °C | 1 cm2 in 20 mL | p-Nitrophenol 2 mg/L | 87.6%; 300 min/Sunlight simulation | [28] |
| V (0.724 wt.%)/Electrochemical anodization, immersion, calcined at 450 °C | 1 cm2 in 20 mL | Acid Yellow 23 2 mg/L | 93.9%; 210 min/Sunlight simulation | This study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tošić, M.; Radenković, M.; Radičić, R.; Stojadinović, S.; Živković, S.; Krstulović, N.; Momčilović, M. Laser-Synthesized Vanadium-Based Nanoparticles on TiO2 Nanotubes for Photocatalytic Degradation of Acid Yellow 23. Processes 2026, 14, 1188. https://doi.org/10.3390/pr14081188
Tošić M, Radenković M, Radičić R, Stojadinović S, Živković S, Krstulović N, Momčilović M. Laser-Synthesized Vanadium-Based Nanoparticles on TiO2 Nanotubes for Photocatalytic Degradation of Acid Yellow 23. Processes. 2026; 14(8):1188. https://doi.org/10.3390/pr14081188
Chicago/Turabian StyleTošić, Miloš, Marina Radenković, Rafaela Radičić, Stevan Stojadinović, Sanja Živković, Nikša Krstulović, and Miloš Momčilović. 2026. "Laser-Synthesized Vanadium-Based Nanoparticles on TiO2 Nanotubes for Photocatalytic Degradation of Acid Yellow 23" Processes 14, no. 8: 1188. https://doi.org/10.3390/pr14081188
APA StyleTošić, M., Radenković, M., Radičić, R., Stojadinović, S., Živković, S., Krstulović, N., & Momčilović, M. (2026). Laser-Synthesized Vanadium-Based Nanoparticles on TiO2 Nanotubes for Photocatalytic Degradation of Acid Yellow 23. Processes, 14(8), 1188. https://doi.org/10.3390/pr14081188








