An Integrated Analytical Approach for the Evaluation of Low-THC Cannabis sativa Products
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Samples of C. sativa
2.3. Extraction
2.4. Ultraviolet Spectroscopy (UV–Vis)
2.5. High-Performance Thin-Layer Chromatography (HPTLC)
2.6. Fourier Transform Infrared Spectroscopy (FTIR)
2.7. Raman Spectroscopy
2.8. Gas Chromatography–Mass Spectrometry (GC–MS)
2.9. Image Analysis of HPTLC Chromatogram
2.10. Multivariate Analysis and Data Preprocessing
3. Results and Discussion
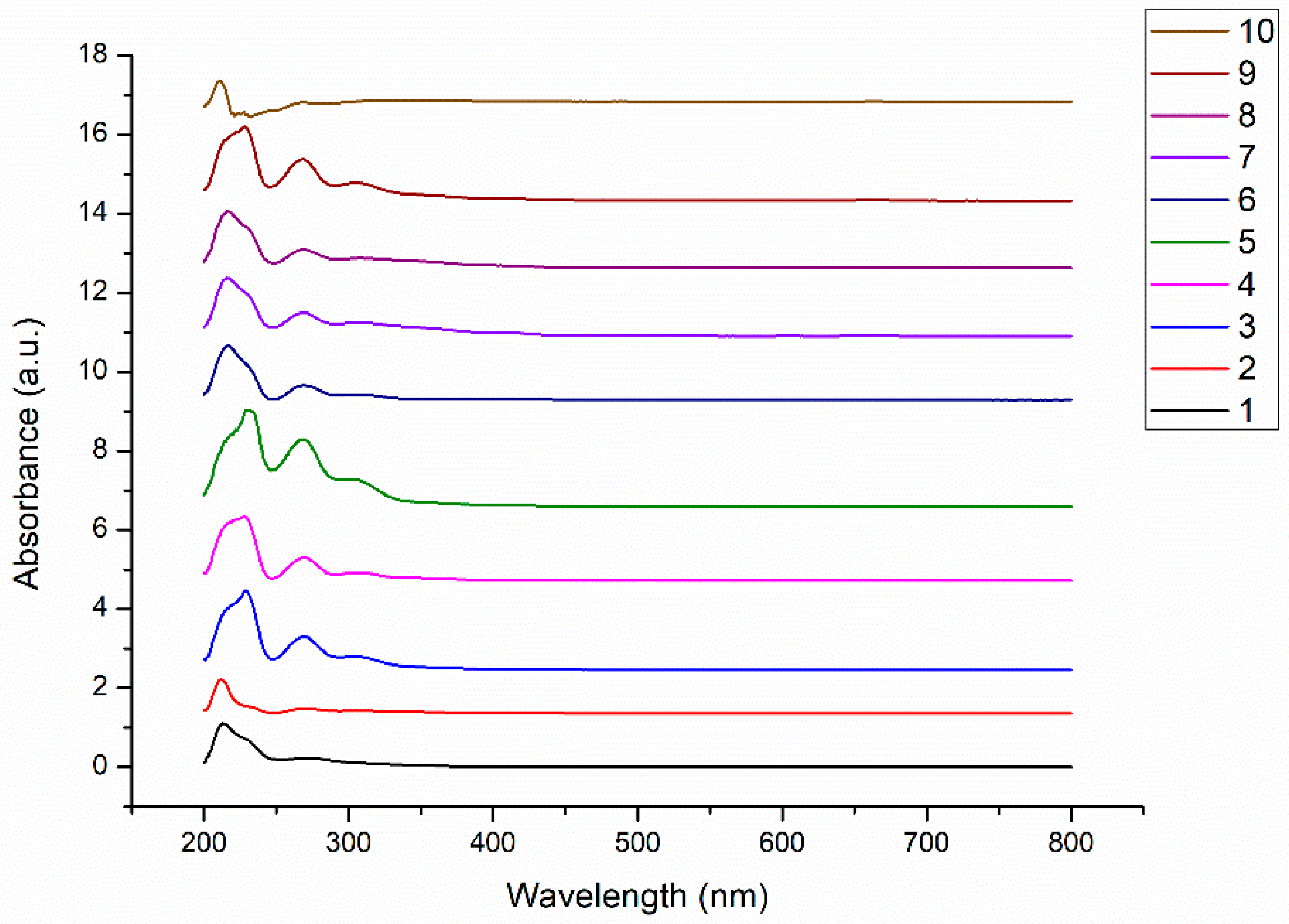
3.1. UV–Vis Spectra of C. sativa Extracts
3.2. HPTLC Fingerprint Analysis of C. sativa Extracts
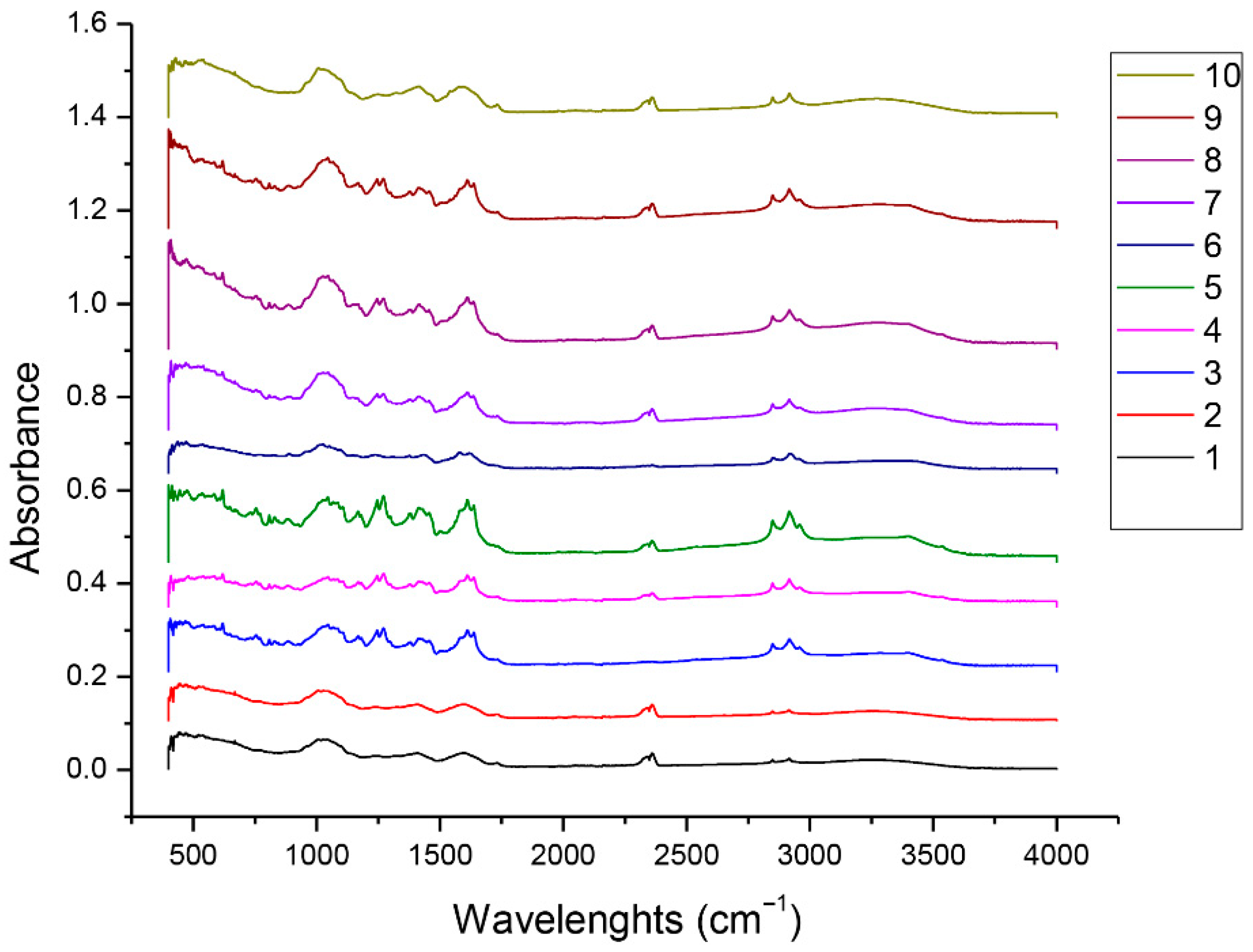
3.3. FTIR Analysis of C. sativa Samples
3.4. Raman Spectra of C. sativa Samples
3.5. GC–MS Data of C. sativa Samples
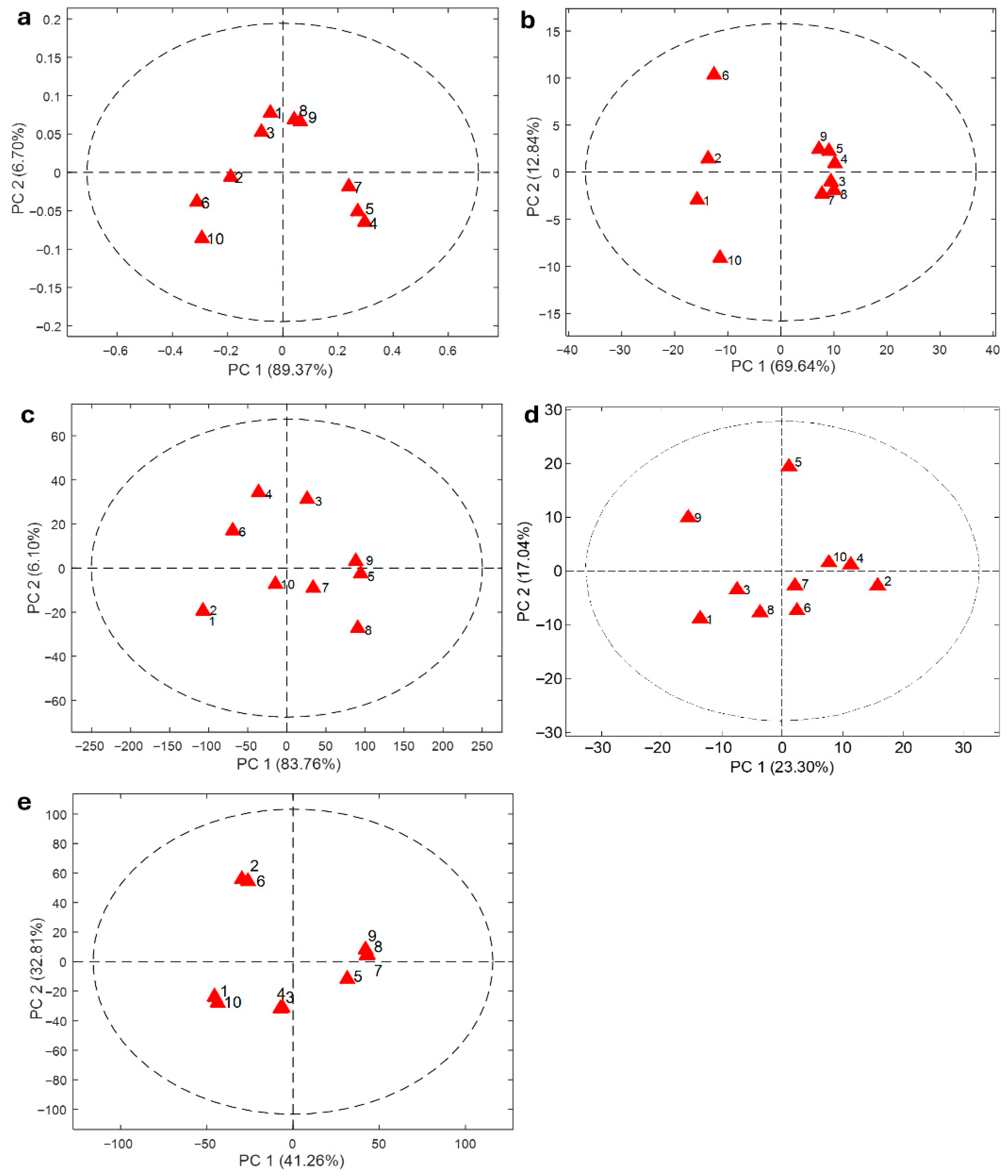
3.6. Principal Component Analysis (PCA)
3.6.1. PCA of UV–Vis Data
3.6.2. PCA Based on HPTLC Data
3.6.3. PCA of FTIR Data
3.6.4. PCA of Raman Data
3.6.5. PCA of CG–MS Data
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Vereinte Nationen. Recommended Methods for the Identification and Analysis of Cannabis and Cannabis Products: Manual for the Use by National Drug Analysis Laboratories; Rev. and updated; United Nations: New York, NY, USA, 2009; ISBN 978-92-1-148242-3. [Google Scholar]
- Knežević, N.; Pirvulescu, I. The Science behind Medical Marijuana and Research Challenges. Zdr. Zaštita 2021, 50, 7–20. [Google Scholar] [CrossRef]
- Lončarić-Katušin, M.; Arbanas, D.; Žilić, A.; Jančić, E.; Žunić, J. Kanabis u Liječenju Karcinomske Boli. Acta Medica Croat. 2019, 73, 65–69. [Google Scholar]
- Elsohly, M. (Ed.) Cannabis Chemistry and Biology: Fundamentals; De Gruyter: Berlin, Germany, 2023; ISBN 978-3-11-071836-2. [Google Scholar]
- Markus Klarić, M.; Klarić, D.; Brborović, O.; Capak, K. Marihuana–Zlouporaba i Medicinska Uporaba. J. Appl. Health Sci. 2020, 6, 137–151. [Google Scholar] [CrossRef]
- Otašević, B.; Kolarević, D. Characteristics of Illegal Laboratories for Marijuana Production in Serbia. Bezb. Beogr. 2020, 62, 5–27. [Google Scholar] [CrossRef]
- Koturevic, B.; Brankovic, A. A Rapid Method for the Extraction of Cannabionoids from Cannabis sativa Using Microwave Heating Technique. Nauka Bezb. Polic. 2014, 109–123. [Google Scholar] [CrossRef]
- Cmpameшкa Пpoцeнa Jaвнe Бeзбeднocmu; Министарствo Унутрашњих пoслoва Републике Србије: Belgrade, Serbia, 2026.
- Krivični Zakonik 2019; Službeni glasnik Republike Srbije, br. 85/2005, 88/2005–ispr., 107/2005–ispr., 72/2009, 111/2009, 121/2012, 104/2013, 108/2014, 94/2016 i 35/2019; Belgrade, Serbia, 2019.
- Zakon o Psihoaktivnim Kontrolisanim Supstancama 2018; Službeni glasnik Republike Srbije, br. 99/2010 i 57/2018; Belgrade, Serbia, 2018.
- Pravilnik o Uslovima Gajenja Konoplje 2013; Službeni glasnik Republike Srbije, br. 64/2013; Belgrade, Serbia, 2013.
- Liu, Y.; Brettell, T.A.; Victoria, J.; Wood, M.R.; Staretz, M.E. High Performance Thin-Layer Chromatography (HPTLC) Analysis of Cannabinoids in Cannabis Extracts. Forensic Chem. 2020, 19, 100249. [Google Scholar] [CrossRef]
- Grafinger, K.E.; Krönert, S.; Broillet, A.; Weinmann, W. Cannabidiol and Tetrahydrocannabinol Concentrations in Commercially Available CBD E-Liquids in Switzerland. Forensic Sci. Int. 2020, 310, 110261. [Google Scholar] [CrossRef]
- Ramos-Guerrero, L.; Montalvo, G.; Cosmi, M.; García-Ruiz, C.; Ortega-Ojeda, F.E. Classification of Various Marijuana Varieties by Raman Microscopy and Chemometrics. Toxics 2022, 10, 115. [Google Scholar] [CrossRef] [PubMed]
- Hazekamp, A.; Peltenburg, A.; Verpoorte, R.; Giroud, C. Chromatographic and Spectroscopic Data of Cannabinoids from Cannabis sativa L. J. Liq. Chromatogr. Relat. Technol. 2005, 28, 2361–2382. [Google Scholar] [CrossRef]
- Dos Santos, N.A.; Souza, L.M.; Domingos, E.; França, H.S.; Lacerda, V.; Beatriz, A.; Vaz, B.G.; Rodrigues, R.R.T.; Carvalho, V.V.; Merlo, B.B.; et al. Evaluating the Selectivity of Colorimetric Test (Fast Blue BB Salt) for the Cannabinoids Identification in Marijuana Street Samples by UV–Vis, TLC, ESI(+)FT-ICR MS and ESI(+)MS/MS. Forensic Chem. 2016, 1, 13–21. [Google Scholar] [CrossRef]
- Herrera, J.; Rolim, L.; Honorato, R.; Pimentel, M.F. Analysis of Cannabinoids in Medicinal Cannabis Products: A Comprehensive Review. J. Braz. Chem. Soc. 2024, 35, e-20240129. [Google Scholar] [CrossRef]
- Radosavljević-Stevanović, N.; Andrić, F.; Manojlović, D.; Ristivojević, P.M. Green Analytical Strategy for Distinguishing Cannabis sativa L. Chemotypes via Planar Chromatography and Partial Least Squares Regression. JPC—J. Planar Chromatogr.–Mod. TLC 2024, 37, 499–509. [Google Scholar] [CrossRef]
- Radosavljević-Stevanović, N.; Kovačević, A.; Manojlović, D.; Ristivojević, P. High-Performance Thin-Layer Chromatography Hyphenated with Image Processing and Chemometrics as a Tool for Forensic Discrimination of Cannabis sativa L. Chemotypes. Forensic Chem. 2023, 36, 100528. [Google Scholar] [CrossRef]
- Ristivojević, P.M.; Tahir, A.; Malfent, F.; Opsenica, D.M.; Rollinger, J.M. High-Performance Thin-Layer Chromatography/Bioautography and Liquid Chromatography-Mass Spectrometry Hyphenated with Chemometrics for the Quality Assessment of Morus Alba Samples. J. Chromatogr. A 2019, 1594, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Seccamani, P.; Franco, C.; Protti, S.; Porta, A.; Profumo, A.; Caprioglio, D.; Salamone, S.; Mannucci, B.; Merli, D. Photochemistry of Cannabidiol (CBD) Revised. A Combined Preparative and Spectrometric Investigation. J. Nat. Prod. 2021, 84, 2858–2865. [Google Scholar] [CrossRef] [PubMed]
- Poletto, M.; Ornaghi, H.; Zattera, A. Native Cellulose: Structure, Characterization and Thermal Properties. Materials 2014, 7, 6105–6119. [Google Scholar] [CrossRef]
- Le Troedec, M.; Sedan, D.; Peyratout, C.; Bonnet, J.P.; Smith, A.; Guinebretiere, R.; Gloaguen, V.; Krausz, P. Influence of Various Chemical Treatments on the Composition and Structure of Hemp Fibres. Compos. Part A Appl. Sci. Manuf. 2008, 39, 514–522. [Google Scholar] [CrossRef]
- Dai, D.; Fan, M. Characteristic and Performance of Elementary Hemp Fibre. Mater. Sci. Appl. 2010, 01, 336–342. [Google Scholar] [CrossRef]
- Ona, A.; Vârban, R.; Crișan, I.; Vârban, D.; Pop, C.R.; Filip, M.; Stoie, A. Characterization of Hemp (Cannabis sativa L.) Raw Bast Fibers of Monoecious versus Dioecious Cultivars from Flowering to Senescence: FTIR of Lignocelluloses and Extract Bioactivities. Int. J. Biol. Macromol. 2025, 307, 141852. [Google Scholar] [CrossRef]
- Komisar, D.; Kutsyk, A.; Vasyliev, O.; Aulin, Y.; Boisen, S.; Stergiou, K.; Pilhun, Y.; Duelund, L.; Bahij, R.; Hedegaard, M.A.B.; et al. Portable Raman Spectroscopy for Non-Invasive Chemotype Identification and pCB Profiling in Cannabis. J. Raman Spectrosc. 2025, 56, 1498–1507. [Google Scholar] [CrossRef]
- Agarwal, U.P.; Ralph, S.A. Raman Spectra of Delignified Plant Fibers: Exploring the Impact of Xylan’s Presence on the Spectral Features of Cellulose. Fibers 2023, 12, 5. [Google Scholar] [CrossRef]
- Sanchez, L.; Baltensperger, D.; Kurouski, D. Raman-Based Differentiation of Hemp, Cannabidiol-Rich Hemp, and Cannabis. Anal. Chem. 2020, 92, 7733–7737. [Google Scholar] [CrossRef] [PubMed]



| No. | Name | Form | Year |
|---|---|---|---|
| 1 | Gramina 22 | Dried plant leaves | 2022 |
| 2 | Gramina 25 | Dried plant leaves | 2025 |
| 3 | Lemon Haze 21 | Dried Flower | 2021 |
| 4 | Lemon Haze 22 | Dried Flower | 2022 |
| 5 | Lemon Haze 25 | Dried Flower | 2025 |
| 6 | Amnezia | Dried Flower | 2025 |
| 7 | Lemon Haze Preroll | Rolled in a joint | 2025 |
| 8 | Gorilla Glue Preroll | Rolled in a joint | 2025 |
| 9 | Amnezia Preroll | Rolled in a joint | 2025 |
| 10 | Botanikos | Tea bags | 2021 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cumbo, A.; Otašević, B.; Radosavljević-Stevanović, N.; Jankov, M.; Tasić, G.; Ristivojević, P.; Branković, A. An Integrated Analytical Approach for the Evaluation of Low-THC Cannabis sativa Products. Processes 2026, 14, 1172. https://doi.org/10.3390/pr14071172
Cumbo A, Otašević B, Radosavljević-Stevanović N, Jankov M, Tasić G, Ristivojević P, Branković A. An Integrated Analytical Approach for the Evaluation of Low-THC Cannabis sativa Products. Processes. 2026; 14(7):1172. https://doi.org/10.3390/pr14071172
Chicago/Turabian StyleCumbo, Ana, Božidar Otašević, Nataša Radosavljević-Stevanović, Milica Jankov, Gvozden Tasić, Petar Ristivojević, and Ana Branković. 2026. "An Integrated Analytical Approach for the Evaluation of Low-THC Cannabis sativa Products" Processes 14, no. 7: 1172. https://doi.org/10.3390/pr14071172
APA StyleCumbo, A., Otašević, B., Radosavljević-Stevanović, N., Jankov, M., Tasić, G., Ristivojević, P., & Branković, A. (2026). An Integrated Analytical Approach for the Evaluation of Low-THC Cannabis sativa Products. Processes, 14(7), 1172. https://doi.org/10.3390/pr14071172








