Resonance-Driven Ultrasound-Assisted Germination of Cucurbita pepo: A Multiphysics-Based Process Intensification Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Seed Material
2.2. Mathematical Modeling
- , with representing the local acoustic intensity.
- , with as the thermal resistance and the seed volume.
- .
- is the thermal capacitance of the seed.
2.3. Multiphysics Simulation Considerations
- Software used: Simulations were performed using COMSOL Multiphysics®, Software Version 5.3a employing the Pressure Acoustics and Heat Transfer interfaces. These modules were coupled to resolve acoustic wave propagation and thermo-acoustic energy deposition within the seed.
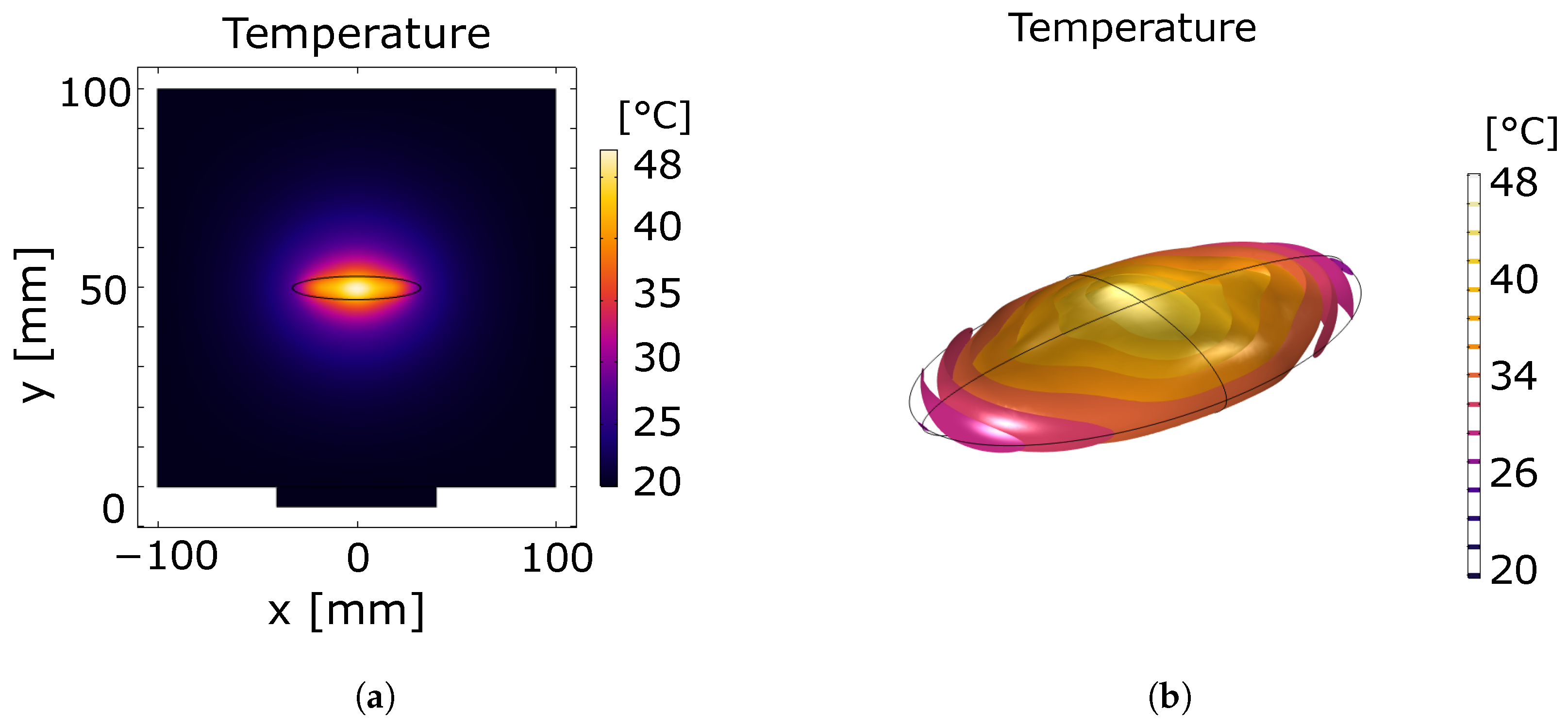
- Geometry and mesh configuration: A 2D rectangular domain was defined to represent the water-filled germination chamber. A piezoelectric transducer with a diameter of 5 cm was placed at one boundary to emit ultrasound signals. The seed was modeled as an elliptical inclusion centered within the domain (see Figure 2). Mesh resolution was set to [40].
- Boundary conditions and physical models: The transducer boundary was defined as a harmonic pressure source with amplitude MPa. Non-reflective boundary conditions were applied elsewhere to minimize artificial reflections. The acoustic field was resolved using the classical wave equation, while the thermal field was governed by a heat transfer model incorporating ultrasound-induced flux as a volumetric source.
- Simulation parameters: Water properties were defined as speed of sound m/s, density kg/m3, and acoustic absorption coefficient Np/m. Seed properties were defined according to Table 1. A temperature threshold of 60 °C was imposed to prevent thermal damage and ensure physiological viability.
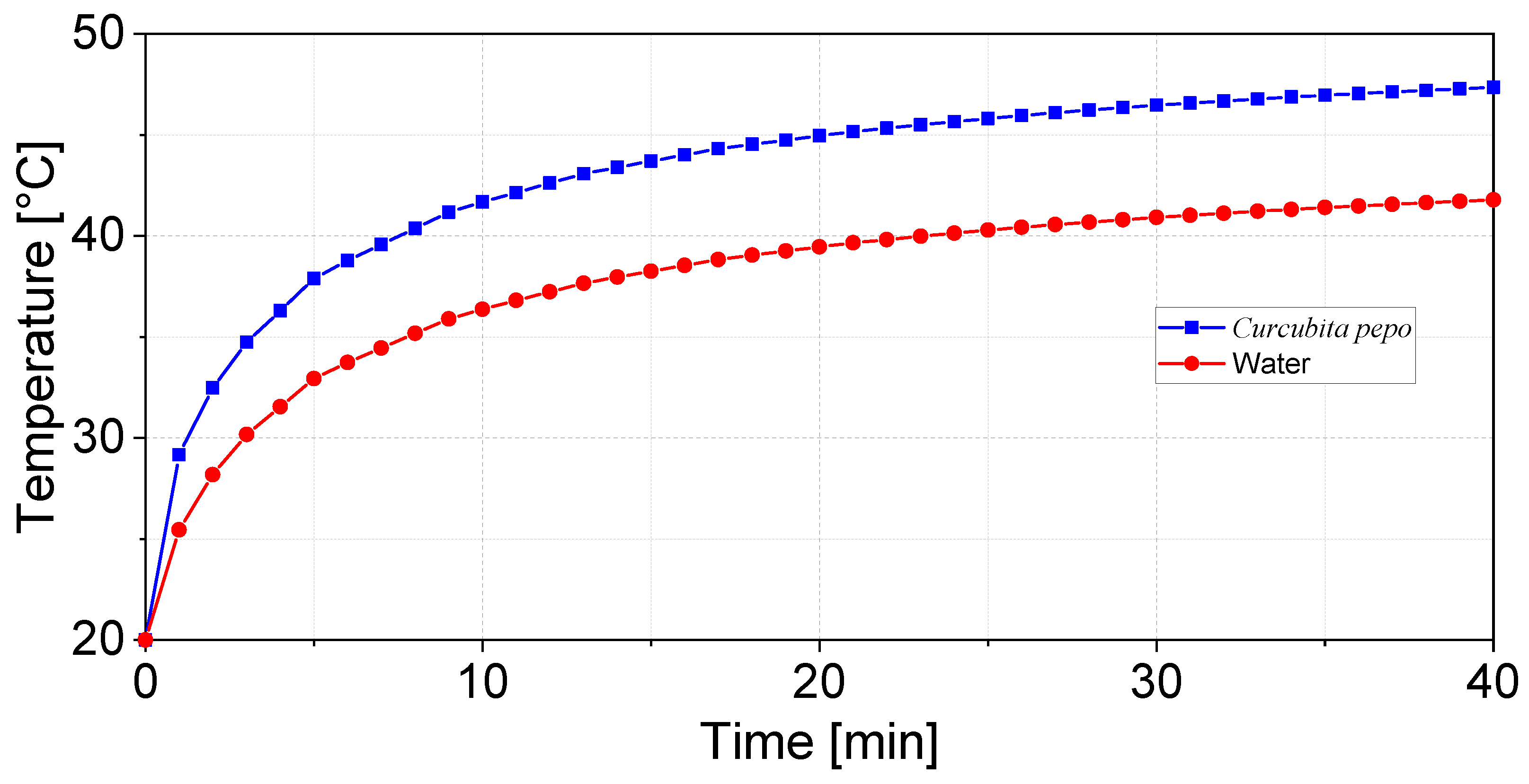
- Simulation workflow: A structured multiphysics simulation pipeline was implemented to characterize ultrasound–seed interactions across spectral, spatial, and temporal domains. First, a high-resolution spectral analysis was conducted over a frequency range of 20–50 kHz with a 1 Hz increment to identify the resonance frequency of the seed. Subsequently, a spatial analysis of the acoustic pressure field was performed at the seed–medium interface to resolve energy localization patterns, enabling the identification of pressure nodes and antinodes and their influence on the seed structure. Finally, coupled temporal and spatial analyses of temperature gradients were carried out to quantify transient thermal effects induced by ultrasonic excitation within both the seed and the surrounding propagation medium.
2.4. Ultrasound Treatment Setup
2.5. Germination Conditions
2.6. Ultrasound-Assisted Germination Setup
2.7. Germination Performance Assessment
2.8. Statistical Analysis
2.9. Energy and Water Consumption Assessment
3. Results
3.1. Thermoacoustic Simulation of Ultrasound–Seed Interaction
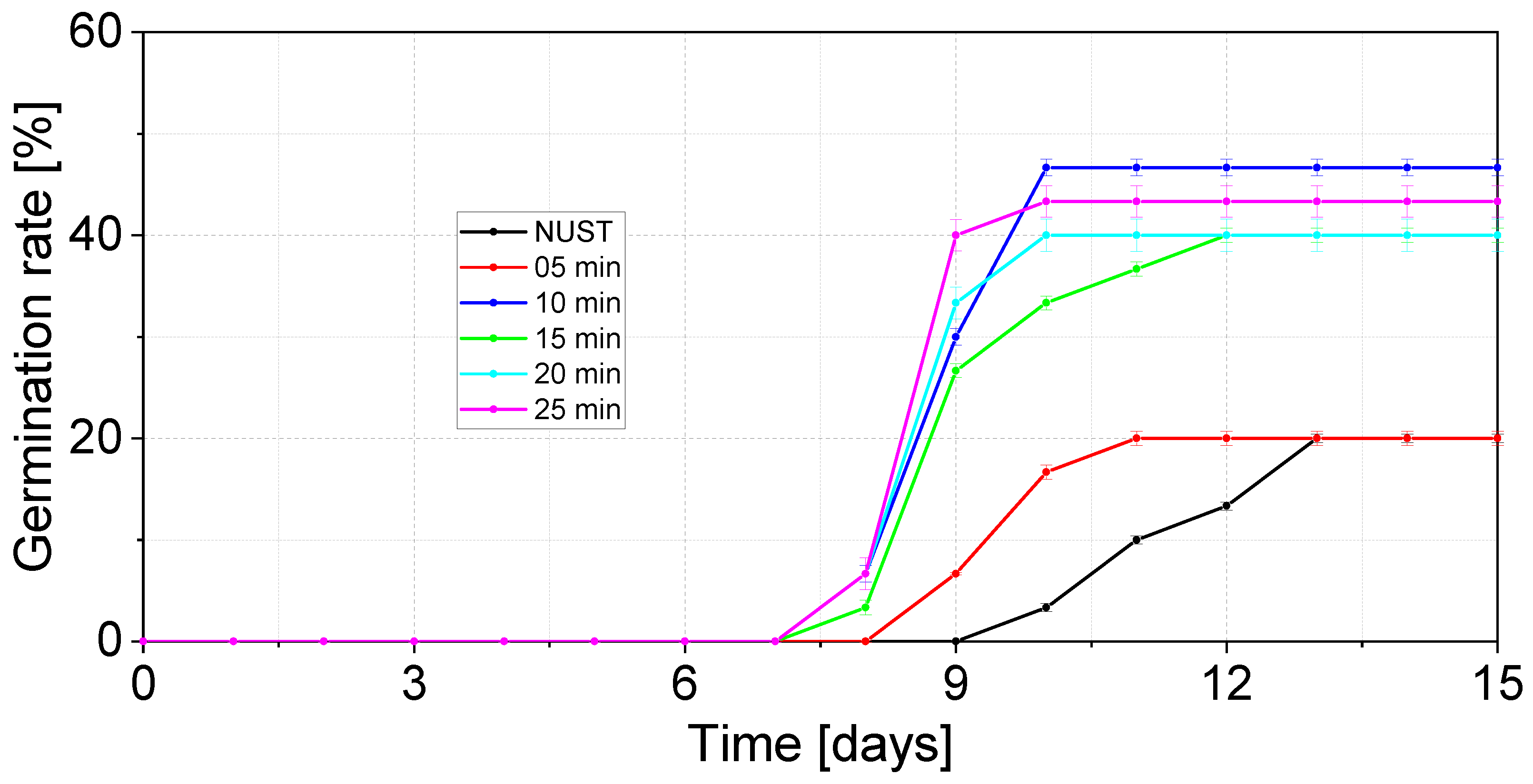
3.2. Baseline Germination Performance
3.3. Ultrasound-Assisted Germination Response
3.4. Water and Energy Consumption Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| UAG | Ultrasound–assisted germination |
| ABA | Abscisic acid |
| ET | Ethylene |
| JA | Jasmonate |
| Gmax | Maximum germination capacity |
| Lag phase | |
| FEM | Finit element method |
| 2D | Two dimensions |
| 3D | Three dimensions |
| PET | Polyethylene Terephthalate |
| Gr | Germination rate |
| Gi | Germination index |
| E | Total energy input |
| SIL | Sound Intensity Level |
| tmax | Maximum germination time |
| NUST | Non-ultrasound-treated |
References
- Bewley, J.D.; Bradford, K.J.; Hilhorst, H.W.; Nonogaki, H. Seeds: Physiology of Development, Germination and Dormancy, 3rd ed.; Springer: New York, NY, USA, 2013. [Google Scholar] [CrossRef]
- Paparella, S.; Araújo, S.S.; Rossi, G.; Wijayasinghe, M.; Carbonera, D.; Balestrazzi, A. Seed priming: State of the art and new perspectives. Plant Cell Rep. 2015, 34, 1281–1293. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.; Wahid, A.; Siddique, K.H.M. Micronutrient application through seed treatments: A review. J. Soil Sci. Plant Nutr. 2012, 12, 125–142. [Google Scholar] [CrossRef]
- Javed, T.; Afzal, I.; Shabbir, R.; Ikram, K.; Saqlain Zaheer, M.; Faheem, M.; Haider Ali, H.; Iqbal, J. Seed coating technology: An innovative and sustainable approach for improving seed quality and crop performance. J. Saudi Soc. Agric. Sci. 2022, 21, 536–545. [Google Scholar] [CrossRef]
- Araújo, S.d.S.; Paparella, S.; Dondi, D.; Bentivoglio, A.; Carbonera, D.; Balestrazzi, A. Physical Methods for Seed Invigoration: Advantages and Challenges in Seed Technology. Front. Plant Sci. 2016, 7, 646. [Google Scholar] [CrossRef] [PubMed]
- Jarrar, H.; El-Keblawy, A.; Ghenai, C.; Abhilash, P.; Bundela, A.K.; Abideen, Z.; Sheteiwy, M.S. Seed enhancement technologies for sustainable dryland restoration: Coating and scarification. Sci. Total Environ. 2023, 904, 166150. [Google Scholar] [CrossRef]
- Mason, T.J.; Lorimer, J.P. Applied Sonochemistry: Uses of Power Ultrasound in Chemistry and Processing; Wiley: Hoboken, NJ, USA, 2002. [Google Scholar] [CrossRef]
- Rifna, E.; Ratish Ramanan, K.; Mahendran, R. Emerging technology applications for improving seed germination. Trends Food Sci. Technol. 2019, 86, 95–108. [Google Scholar] [CrossRef]
- Aguilar-Torres, D.; Jiménez-Ramírez, O.; Perdomo, F.; Vázquez-Medina, R. Advanced Sustainable Food Processing: Ultrasound-Assisted Germination of Cucurbita pepo Seeds. In Proceedings of the 4th International Electronic Conference on Processes, Online, 20–22 October 2025. [Google Scholar]
- Goussous, S.J.; Samarah, N.H.; Alqudah, A.M.; Othman, M.O. Enhancing Seed Germination of Four Crop Species Using an Ultrasonic Technique. Exp. Agric. 2010, 46, 231–242. [Google Scholar] [CrossRef]
- Yaldagard, M.; Mortazavi, S.A.; Tabatabaie, F. Influence of ultrasonic stimulation on the germination of barley seed and its alpha-amylase activity. Afr. J. Biotechnol. 2008, 7, 2465–2471. [Google Scholar]
- Nogueira, A.; Teixeira, A.; Gerós, H.; Puga, H. Ultrasound Prototype for Improving Germination and Seedling Growth in Tomato and Maize Seeds. J. Plant Growth Regul. 2023, 43, 1216–1229. [Google Scholar] [CrossRef]
- Gong, M.; Kong, M.; Huo, Q.; He, J.; He, J.; Yan, Z.; Lu, C.; Jiang, Y.; Song, J.; Han, W.; et al. Ultrasonic treatment can improve maize seed germination and abiotic stress resistance. BMC Plant Biol. 2024, 24, 758. [Google Scholar] [CrossRef]
- Nogueira, A.; Puga, H.; Gerós, H.; Teixeira, A. Ultrasound-enhanced seed hydration: Impacts on seedling vigor, gene expression and absorption kinetics in maize, bean and pepper seeds. J. Sci. Food Agric. 2025, 105, 7227–7241. [Google Scholar] [CrossRef]
- Huang, S.; Ashraf, U.; Duan, M.; Ren, Y.; Xing, P.; Yan, Z.; Tang, X. Ultrasonic seed treatment improved seed germination, growth, and yield of rice by modulating associated physio-biochemical mechanisms. Ultrason. Sonochem. 2024, 104, 106821. [Google Scholar] [CrossRef]
- Nogueira, A.; Puga, H.; Gerós, H.; Teixeira, A. Seed germination and seedling development assisted by ultrasound: Gaps and future research directions. J. Sci. Food Agric. 2023, 104, 583–597. [Google Scholar] [CrossRef]
- Király, A.; Farkas, D.; Dobránszki, J. Ultrasound in Plant Life and Its Application Perspectives in Horticulture and Agriculture. Horticulturae 2025, 11, 318. [Google Scholar] [CrossRef]
- Alfalahi, A.O.; Alobaidy, B.S.; Almarie, A.A.; Dhanoon, O.M.; Qasem, J.R.; Almehemdi, A.F.; Najda, A. Ultrasonic Treatment Enhances Germination and Affects Antioxidant Gene Expression in Soybean (Glycine max L. Merr). Agronomy 2022, 12, 2446. [Google Scholar] [CrossRef]
- Tufail, T.; Ul Ain, H.B.; Ashraf, J.; Saeed, F.; Basharat, Z.; Ahmed, Z.; Waseem, M.; Xu, B.; Manzoor, M.F.; Mugabi, R. Effects of germination and ultrasound treatment on the thermodynamics, nutritional and structural quality of highland barley fractions. Ultrason. Sonochem. 2025, 123, 107652. [Google Scholar] [CrossRef]
- Villamiel, M.; García-Pérez, J.V.; Montilla, A.; Carcel, J.A.; Benedito, J. Ultrasound in Food Processing: Recent Advances; Wiley Blackwell: Hoboken, NJ, USA, 2017. [Google Scholar]
- Dhatt, A.; Pandey, S.; Garch, K.S.; Verma, N.; Sagar, V.; Sharma, M. Comprehensive review of pumpkin (Cucurbita spp.): Domestication, global distribution, genetic characterization, breeding strategies, and genomic insights. Veg. Sci. 2024, 51, 196–210. [Google Scholar] [CrossRef]
- Paris, H.S. Historical records, origins, and development of the edible cultivar groups of Cucurbita pepo (Cucurbitaceae). Econ. Bot. 1989, 43, 423–443. [Google Scholar] [CrossRef]
- Decker, D.S. Origin(s), evolution, and systematics of Cucurbita pepo (Cucurbitaceae). Econ. Bot. 1988, 42, 4–15. [Google Scholar] [CrossRef]
- Acila, S.; Derouiche, S.; Allioui, N. Embryo growth alteration and oxidative stress responses in germinating Cucurbita pepo seeds exposed to cadmium and copper toxicity. Sci. Rep. 2024, 14, 8608. [Google Scholar] [CrossRef]
- Irik, H.A.; Bikmaz, G. Effect of different salinity on seed germination, growth parameters and biochemical contents of pumpkin (Cucurbita pepo L.) seeds cultivars. Sci. Rep. 2024, 14, 6929. [Google Scholar] [CrossRef]
- Foschi, M.L.; Juan, M.; Pascual, B.; Pascual-Seva, N. Effects of High Intensity Ultrasound Stimulation on the Germination Performance of Caper Seeds. Plants 2023, 12, 2379. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.; Zhang, L.; Yin, H.; Yu, J.; Hu, S.; Zhang, Z.; Liu, J. Enhancing malting performance of harder barley varieties through ultrasound treatment. Ultrason. Sonochem. 2024, 105, 106860. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Pacheco, B.S.; Peralta-Guevara, D.E.; Yauris-Silvera, C.R.; Medina-Gutierrez, C.A.; Loayza-Buleje, J.J. Ultrasound-Assisted Germination: Impact of Time and Frequency on the Physical, Chemical, Thermal Properties and Bioactive Compounds of High Andean Quinoa. Processes 2025, 14, 98. [Google Scholar] [CrossRef]
- Rashid, M.T.; Liu, K.; Muzaffar, N.; Alohali, B.M.; Jatoi, M.A.; Usman, H.; Aadil, R.M. Impact of ultrasound-assisted germination and varied drying methods on phenolic biosynthesis, antioxidant capacity, and oxidative enzyme activity in selenium-biofortified black rice. Ultrason. Sonochem. 2025, 123, 107692. [Google Scholar] [CrossRef]
- dos Santos, M.A.; Pacheco, F.C.; Gusmão, M.H.A.; Andressa, I.; Pacheco, A.F.C.; Paiva, P.H.C.; Gomes, F.T. Ultrasound-assisted Hydration Improves Germination and Early Growth of Moringa oleifera Under Salinity Stress. J. Soil Sci. Plant Nutr. 2026. [Google Scholar] [CrossRef]
- Khalaf, E.M.; Raizada, M.N. Taxonomic and functional diversity of cultured seed associated microbes of the cucurbit family. BMC Microbiol. 2016, 16, 131. [Google Scholar] [CrossRef]
- Iglesias-Moya, J.; Cebrián, G.; Garrido, D.; Martínez, C.; Jamilena, M. The ethylene receptor mutation etr2b reveals crosstalk between ethylene and ABA in the control of Cucurbita pepo germination. Physiol. Plant. 2023, 175, e13864. [Google Scholar] [CrossRef]
- Alonso, S.; Gautam, K.; Iglesias-Moya, J.; Martínez, C.; Jamilena, M. Crosstalk between Ethylene, Jasmonate and ABA in Response to Salt Stress during Germination and Early Plant Growth in Cucurbita pepo. Int. J. Mol. Sci. 2024, 25, 8728. [Google Scholar] [CrossRef]
- Macedo, C.; Silva, A.M.; Ferreira, A.S.; Moreira, M.M.; Delerue-Matos, C.; Rodrigues, F. Microwave- and Ultrasound-Assisted Extraction of Cucurbita pepo Seeds: A Comparison Study of Antioxidant Activity, Phenolic Profile, and In-Vitro Cells Effects. Appl. Sci. 2022, 12, 1763. [Google Scholar] [CrossRef]
- Liang, L.; Chen, L.; Liu, G.; Zhang, F.; Linhardt, R.J.; Sun, B.; Li, Q.; Zhang, Y. Optimization of germination and ultrasonic-assisted extraction for the enhancement of aminobutyric acid in pumpkin seed. Food Sci. Nutr. 2022, 10, 2101–2110. [Google Scholar] [CrossRef]
- Pacheco, F.C.; Cunha, J.S.; Andressa, I.; dos Santos, F.R.; Pacheco, A.F.C.; Nalon, G.A.; Paiva, P.H.C.; Tribst, A.A.L.; Augusto, P.E.D.; Leite Júnior, B.R.d.C. Ultrasound-Assisted Intermittent Hydration of Pumpkin Seeds: Improving the Water Uptake, Germination, and Quality of a Clean Label Ingredient. Food Bioprocess Technol. 2024, 18, 618–632. [Google Scholar] [CrossRef]
- Blackstock, D.T. Fundamentals of Physical Acoustics; John Wiley & Sons: Hoboken, NJ, USA, 2000. [Google Scholar]
- Aguilar-Torres, D.; Jiménez-Ramírez, O.; Hernández-Gama, R.; Perdomo-Hurtado, F.A.; Vázquez-Medina, R. Multiphysics analysis for ultrasound settings that can disinfect food: Inactivation of E. coli in apples. Food Control 2025, 172, 111191. [Google Scholar] [CrossRef]
- Aguilar-Torres, D.; Jiménez-Ramírez, O.; Camacho-Martínez, J.L.; Vázquez-Medina, R. Multiphysics analysis of a high-intensity ultrasound system applied to a three-layer animal tissue. WFUMB Ultrasound Open 2024, 2, 100039. [Google Scholar] [CrossRef]
- Prinn, A.G. A Review of Finite Element Methods for Room Acoustics. Acoustics 2023, 5, 367–395. [Google Scholar] [CrossRef]
- Ranal, M.A.; Santana, D.G.d. How and why to measure the germination process? Braz. J. Bot. 2006, 29, 1–11. [Google Scholar] [CrossRef]
- Chiapusio, G.; Sánchez, A.M.; Reigosa, M.J.; González, L.; Pellissier, F. Do Germination Indices Adequately Reflect Allelochemical Effects on the Germination Process? J. Chem. Ecol. 1997, 23, 2445–2453. [Google Scholar] [CrossRef]






| Species | E | l | w | h | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| [kg/m3] | [m/s] | [MPa] | [kPa·s/m] | [mm] | [mm] | [mm] | [W/(m·K)] | [kJ/(kg·K)] | ||
| Cucurbita pepo | 1152.00 | 201.29 | 46.65 | 125.71 | 0.90 | 16.10 | 8.40 | 2.93 | 0.50 | 2000.00 |
| Treatment | Germination Rate (Gr) [%] |
|---|---|
| 0 min | 19.96 ± 0.152 |
| 5 min | 20.00 ± 0.707 |
| 10 min | 46.80 ± 0.837 |
| 15 min | 40.00 ± 0.707 |
| 20 min | 40.00 ± 1.581 |
| 25 min | 43.00 ± 1.571 |
| Cases | Sum of Squares | df | Mean Square | F | p |
|---|---|---|---|---|---|
| Treatments | 3522 | 5 | 704.40 | 628.70 | <0.001 |
| Residuals | 26.89 | 24 | 1.12 |
| Comparison | Mean Difference | |
|---|---|---|
| 05 min—0 min | 0.04 | 1.000 |
| 10 min—0 min | 26.84 | <0.001 |
| 15 min—0 min | 20.04 | <0.001 |
| 20 min—0 min | 20.04 | <0.001 |
| 25 min—0 min | 23.04 | <0.001 |
| Study | Cultivar | Gr [%] | Temperature [°C] |
|---|---|---|---|
| This work | NE | ∼47 | 41.6 |
| Liang et al. [35] | LR-1 | ∼15–17 | ∼30 |
| Liang et al. [35] | LR-2 | ∼9–11 | ∼30 |
| Liang et al. [35] | LR-3 | ∼7–9 | ∼30 |
| Liang et al. [35] | JH-4 | ∼53–56 | ∼30 |
| Liang et al. [35] | RF-9 | ∼45–48 | ∼30 |
| Liang et al. [35] | YH-3 | ∼88–91 | ∼30 |
| Liang et al. [35] | XC-2 | ∼95–98 | ∼30 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Aguilar-Torres, D.; Jiménez-Ramírez, O.; Perdomo, F.A.; Vázquez-Medina, R. Resonance-Driven Ultrasound-Assisted Germination of Cucurbita pepo: A Multiphysics-Based Process Intensification Approach. Processes 2026, 14, 1168. https://doi.org/10.3390/pr14071168
Aguilar-Torres D, Jiménez-Ramírez O, Perdomo FA, Vázquez-Medina R. Resonance-Driven Ultrasound-Assisted Germination of Cucurbita pepo: A Multiphysics-Based Process Intensification Approach. Processes. 2026; 14(7):1168. https://doi.org/10.3390/pr14071168
Chicago/Turabian StyleAguilar-Torres, Daniel, Omar Jiménez-Ramírez, Felipe A. Perdomo, and Rubén Vázquez-Medina. 2026. "Resonance-Driven Ultrasound-Assisted Germination of Cucurbita pepo: A Multiphysics-Based Process Intensification Approach" Processes 14, no. 7: 1168. https://doi.org/10.3390/pr14071168
APA StyleAguilar-Torres, D., Jiménez-Ramírez, O., Perdomo, F. A., & Vázquez-Medina, R. (2026). Resonance-Driven Ultrasound-Assisted Germination of Cucurbita pepo: A Multiphysics-Based Process Intensification Approach. Processes, 14(7), 1168. https://doi.org/10.3390/pr14071168







