1. Introduction
In recent years, the large-scale emission of greenhouse gases, especially CO
2, has intensified global climate change and threatens the sustainable development of human society [
1]. Carbon Capture, Utilization, and Storage (CCUS) is a key technology for achieving deep decarbonization and is studied in both research and policy contexts worldwide [
2]. CO
2 geological storage is a core component of CCUS technology, utilizing geological formations such as depleted oil and gas reservoirs or deep saline aquifers to store the captured CO
2 [
3]. These geological settings are generally evaluated in terms of storage capacity, injectivity, containment, and geomechanical integrity, which together determine their suitability for long-term CO
2 storage [
4,
5]. During CO
2 storage, complex physical and chemical interactions occur between the injected supercritical CO
2 and the saline aquifer, and secondary trapping mechanisms, particularly dissolution trapping, can enhance long-term storage security by immobilizing CO
2 and reducing buoyant migration [
6].
Despite the vast potential of geological storage, practical engineering faces challenges due to complex geological conditions. Previous studies often simplified the numerical simulations by assuming that the reservoir was homogeneous or had a simple layered structure [
7,
8,
9]. Among these, sedimentological heterogeneity and stratigraphic baffling within sandstone reservoirs may create local low-permeability barriers that influence CO
2 migration pathways and retention behavior [
10,
11]. Unlike continuous caprocks, intra-reservoir discontinuous lenticular shale layers act as local baffles rather than complete seals and delay vertical migration by redirecting flow pathways [
12].
Previous studies have examined how intra-reservoir shale barriers and layered heterogeneity influence CO
2 migration and dissolution [
13]. Hansen et al. [
14] showed through large-scale numerical simulations that permeability heterogeneity and background flow can substantially alter supercritical CO
2 dissolution behavior in the deep subsurface. Fang et al. [
15] found that in randomly multilayered heterogeneous formations, gravity segregation and permeability variability jointly control plume migration and dissolution efficiency. In formations with strong vertical permeability contrast, CO
2 tends to migrate laterally along high-permeability layers, which suppresses vertical plume rise but can enhance dissolution efficiency [
16]. Cavanagh and Haszeldine [
17] showed that plume geometry in the Sleipner storage formation is controlled by thin intra-formational shale barriers, underscoring the role of internal low-permeability layers in delaying vertical migration and redistributing lateral flow. Reservoir-scale studies of discontinuous shale barrier systems further indicate that barrier continuity and abundance strongly control plume geometry and pressure buildup, with longer or more laterally continuous barriers tending to suppress vertical migration and promote lateral spreading [
12]. Local blockage by intra-reservoir barriers may also enhance pressure buildup, which should be considered when evaluating safe injection conditions. In addition, site-screening studies have shown that sedimentological heterogeneity and stratigraphic baffling potential are important considerations in evaluating CO
2 migration behavior and storage-site performance [
11].
CO
2 migration and dissolution in deep saline aquifers are governed by buoyancy-driven upward rise and pressure-gradient-driven radial spreading. Reservoir heterogeneity reshapes the two-phase front and interfacial area, and it redistributes the plume by changing the pathways of pressure propagation [
18]. In layered reservoirs with strong contrast, permeability differences produce distinct plume spreading fronts and shift both plume footprint and dissolved fraction. Field observations and interpretations show that thin intra-reservoir shale barriers split CO
2 into stacked layers and delay bulk upward migration [
17]. They also promote lateral spreading within individual units and allow bypass into overlying strata through barrier terminations or weakly connected zones [
19]. When the barriers are discontinuous, local blockage forces CO
2 to follow more tortuous paths. This process creates uneven pressure gradients and localized pressure focusing above and below the barriers, which alters far-field pressure dissipation and vertical pressure connectivity [
20]. Mechanistic models further show that cross-layer flux and filling sequence depend on barrier properties and injection velocity, so shale geometry and petrophysical contrast affect pressure response and plume channeling in a measurable way [
21].
Beyond permeability contrast, capillary and structural heterogeneity associated with discontinuous low-permeability interlayers such as lenticular shale layers redirect CO
2 migration and modify plume-scale spreading under buoyancy-dominated conditions [
22]. Reservoir-scale simulations in randomly multilayered formations have shown that structural heterogeneity reorganizes plume footprint by guiding CO
2 along preferential layers, which changes the spatial distribution of free-phase CO
2 and the regions available for dissolution [
15]. Structural heterogeneity influences dissolution not only through plume shape but also through the timing of contact with fresh brine, because lateral spreading and slower post-injection migration can increase gas–brine contact area and residence time [
23]. Existing studies have often examined plume redistribution in layered media separately from quantitative analyses of CO
2 dissolution in heterogeneous reservoirs, rather than assessing how both processes interact within the same structural setting [
24]. Fewer studies have quantified how the longer bypass path and pressure redistribution produced by discontinuous lenticular shale interlayers modify CO
2-brine contact under radial injection, and how these changes affect dissolution trapping efficiency [
18,
22]. Against this background, the present study focuses on the coupled control of lenticular shale interlayers on plume pathway, pressure field, and dissolution trapping.
Although recent studies have examined capillary heterogeneity, multilayer migration, and vertical CO
2 flow beneath shale barriers, fewer investigations have isolated the coupled effects of idealized discontinuous lenticular shale layers on plume migration, pressure response, and dissolution trapping in radial saline-aquifer models [
22,
25]. In this study, we construct a geological model that includes lenticular shale layers through numerical simulations to investigate the effects of lenticular shale layer geometry, reservoir permeability anisotropy, and injection velocity on CO
2 migration. We also establish a sequestration efficiency factor and a nonlinear multi-parameter power-law correlation model, providing guidance for site selection and injection-strategy optimization in engineering applications.
2. Theoretical Model
This study employs a two-phase flow and mass transfer coupling model to describe the migration and dissolution processes of supercritical CO2 in brine, while considering the effects of reservoir rock structure heterogeneity. The model is based on the laws of mass conservation and Darcy’s law and introduces a mass transfer equation to describe CO2 dissolution and migration.
2.1. Governing Equations for the Two-Phase Flow Field
The flow fields of the CO
2 phase and the brine phase are governed by their respective mass conservation equations and the extended Darcy’s law. For two-phase flow in porous media (CO
2 phase
i =
g and brine phase
i =
w), the mass conservation equation is expressed as [
26]:
Here,
φp is the porosity of the formation,
ρw and
ρg are the densities of brine and gas,
sw and
sg are the saturations of brine and gas, and
uw and
ug represent the Darcy fluxes of the wetting and non-wetting phases, respectively. The Darcy fluxes are determined by Darcy’s law [
27]:
Here, k is the absolute permeability of the reservoir matrix (m2), kri is the relative permeability of phase i, μi is the dynamic viscosity of phase i (Pa·s), P is the pressure of phase i (Pa), ρi is the density of phase i (kg·m−3), and g is the gravitational acceleration vector (m·s−2).
In the two-phase flow formulation, CO
2 and brine saturations satisfy the following relation:
The interaction between the two phases is described by capillary pressure
Pc and relative permeability
kri, where the pressure difference between the non-wetting phase and the wetting phase is the capillary pressure:
Here,
Pnw is the pressure of the non-wetting phase, and
Pw is the pressure of the wetting phase. To analyze the changes in gas-liquid phase saturations, this study introduces the Brooks and Corey model [
28]:
Here,
Se is the effective saturation,
Srw and
Srg are the residual saturations of the brine and gas phases, respectively,
Pec is the capillary entry pressure, and
λ is the pore size distribution index [
9].
All verification cases in this chapter follow the conventional model settings reported in the corresponding references. The geometries, boundary conditions, initial conditions, and key parameters are kept consistent with those of the benchmark studies. Because multiple validation cases are included, the full parameter set for each case is not repeated here. Instead, emphasis is placed on whether the present model reproduces the reference results under the same setup. All validation cases are solved within the same numerical framework as the main model to ensure a consistent basis for comparison.
To validate the capillary pressure-saturation submodel, the study uses Berea sandstone core data to simulate the process of CO
2 displacing brine [
29]. The model boundary conditions are set as impermeable boundaries at the top and bottom, CO
2 injection on the left, and a far-field boundary on the right. The capillary pressure-brine saturation curve is obtained through simulation and compared with the experimental data fitting curve from Pini et al. [
29].
As shown in
Figure 1a, within the capillary pressure range from 0 to 30 kPa, the curve predicted by the model follows the trend of the fitted experimental curve reported by Pini et al. These results indicate that the adopted Brooks–Corey type capillary pressure model represents the relationship between saturation and capillary pressure in two-phase flow.
The relative permeability of the wetting phase is given by:
The relative permeability of the non-wetting phase is given by:
After establishing the relative permeability model, typical relative permeability saturation data from the literature were selected as benchmarks to validate the model [
30,
31]. By calculating the relative permeability values at different saturations and comparing the simulation results with the experimental curves, the results show that the model’s predictions are in good agreement with the experimental data. This validation demonstrates that the two-phase flow model is reliable and can serve as a foundation for constructing the coupled mass transfer model.
By combining the above conservation equations, we have:
The changes in thermophysical properties such as viscosity and density of supercritical CO
2 depend on temperature and pressure. This study uses equations applicable for a pressure range of 15 MPa to 40 MPa and a temperature range of 273 K to 553 K [
32].
To validate the accuracy of the overall two-phase flow displacement model, the simulation results were compared with the experimental data from Vivek et al. [
8] and the analytical solution of the Buckley-Leverett model [
33]. As shown in
Figure 2, the numerical model captured the saturation profiles at different time scales, which were consistent with both the analytical solution of the Buckley-Leverett model and the simulation results from Vivek et al. [
8]. The results confirm that the developed numerical model can reproduce the CO
2-brine displacement behavior across different scales.
2.2. Mass-Transfer Model
In a multicomponent multiphase system, the migration of the dissolved phase in porous media is controlled by advection and diffusion. The advection-diffusion equation is established based on the law of mass conservation. A transport model for dissolved CO
2 is used to describe the distribution and evolution of CO
2 in the liquid phase [
34]:
In the equation,
Cgi is the mass fraction of the gas phase in phase
i.
Di is the diffusion coefficient of the gas component in phase
i, in units of m
2·s
−1, and is calculated using the Wilke-Chang equation while neglecting mechanical dispersion caused by velocity fluctuations.
Igi is the mass transfer rate of the gas component from the phase interface to phase
i (kg·m
−3·s
−1) [
34]. Because interfacial resistance controls mass transfer in soluble or slightly soluble systems, a non-equilibrium interphase mass transfer model is used to represent this behavior [
35,
36]. In this model, the mass transfer rate depends on the interphase mass transfer resistance, specific surface area, and the equilibrium concentration of the gas components, and is expressed by the following equation:
Here, represents the interphase mass transfer rate of the component, in units of m/s. a represents the interfacial specific surface area, in units of m−1. ρw represents the density of the wetting phase, in units of kg·m−3. is a constant known as the total interphase mass transfer rate, in units of s−1. is the gas phase equilibrium concentration in the wetting phase, which refers to the concentration of the gas component in the liquid phase when it reaches equilibrium.
In this study, to facilitate the analysis and discussion of the subsequent results, CO
2 dissolution characteristics are represented in the form of normalized concentration. The normalized concentration is defined as the ratio of the dissolved CO
2 concentration predicted by the non-equilibrium mass transfer model to the equilibrium concentration in the liquid phase.
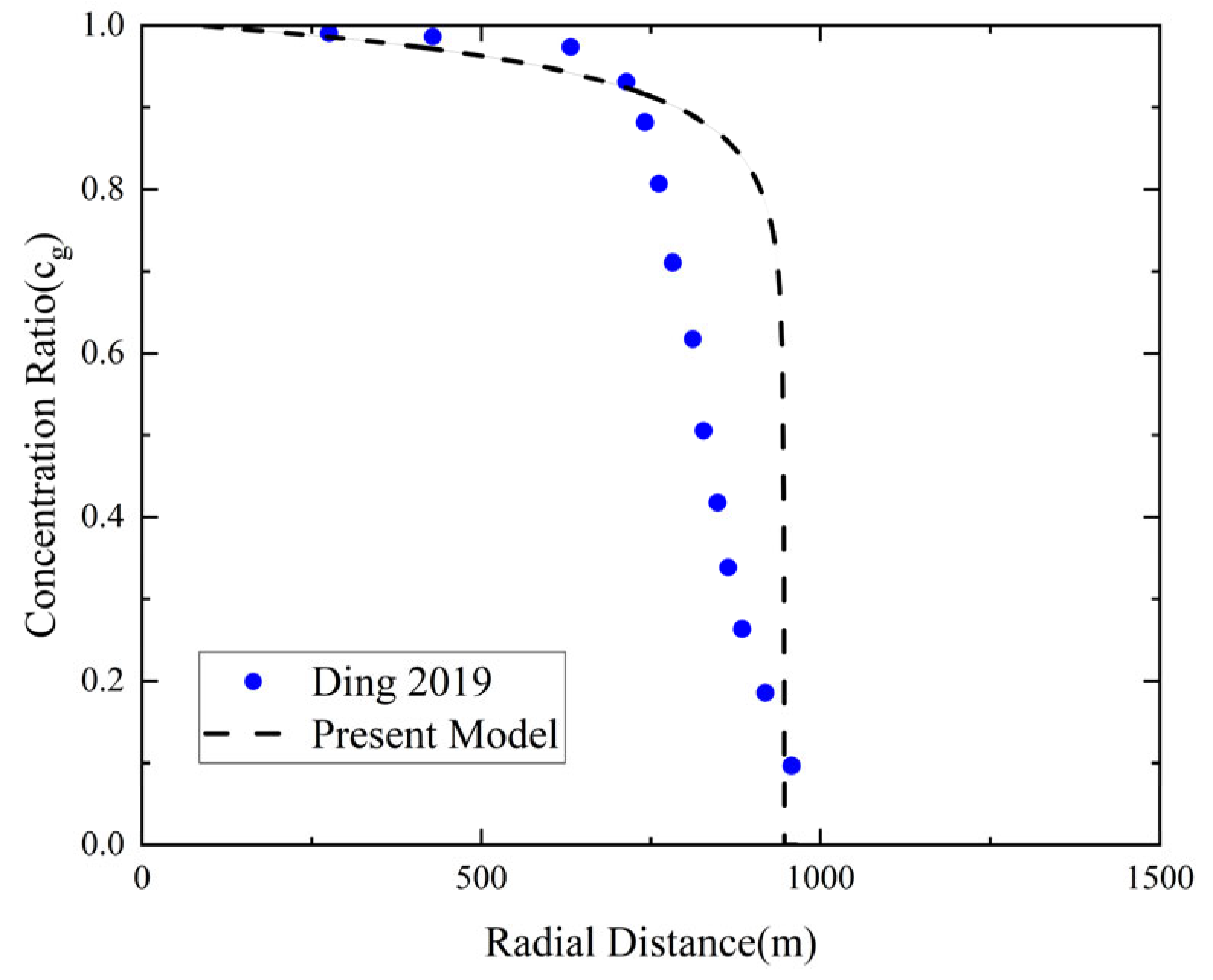
To validate the accuracy of the mass transfer model, the simulation results were compared with the results from Ding et al. [
9].
Figure 3 shows the trend of normalized CO
2 concentration along the radial profile with respect to reservoir length. The initial normalized CO
2 concentration is near 1, and it decreases to 0 as CO
2 migrates to about 1000 m, indicating limited dissolution into brine. The simulation results are consistent with the trend of the data from Ding et al., confirming the applicability of the constructed mass transfer model.
4. Results and Discussion
4.1. Temporal Evolution of Pressure in the Saline Aquifer
Figure 5 shows the spatial evolution of pore pressure distribution in the formation during continuous CO
2 injection from 250 to 1000 days. The high-pressure region is concentrated near the injection end in the lower-left corner of the model, and the low-pressure region lies in the far-field area in the upper-right corner. The pressure fields at different times show that, although injection continues from 250 to 1000 days, the spatial pattern and magnitude of pressure remain consistent. The pressure distribution suggests that pressure perturbations propagate faster than advective fluid migration and reach the model boundaries early. The system then approaches dynamic equilibrium, and the flow field remains near a quasistatic state. This provides a stable potential field for subsequent fluid displacement. Additionally, the vertical pressure distribution exhibits significant spatial heterogeneity. The pressure in the near-well region on the left shows a decreasing trend, transitioning from the high-pressure area at the bottom to the low-pressure area at the top, while an anomalous local pressure inversion occurs in the far-well area on the right.
To further quantify the pressure distribution patterns and gain deeper insight into the underlying flow mechanisms, this study selected radial cross-sections at a vertical distance of 40 m and vertical cross-sections at radial distances of 50 m and 300 m. The changes in pore pressure with distance were plotted at these locations to analyze the pressure response differences at various structural positions, as shown in
Figure 6.
Figure 6a shows the evolution of pore pressure with radial distance and injection time in the lower injection area at a vertical distance of 40 m. The pressure decreases monotonically from the injection well toward the far-field boundary, creating a radial pressure gradient that drives fluid migration. In the near-well zone with radial distances from 0 to 200 m, the pressure response indicates a gradual decrease in pore pressure as injection time increases. This phenomenon is attributed to the improved flow behavior caused by the viscosity difference between the gas and brine phases in two-phase flow. Since the viscosity of supercritical CO
2 is lower than that of formation brine, the low-viscosity gas plume expands radially in the near-well area and displaces the high-viscosity brine. Given that the flow resistance of the fluid is proportional to its viscosity, the expansion of the high-saturation gas phase region reduces the flow resistance in the near-well area. Under constant injection velocity, the driving pressure difference required to maintain flow decreases, leading to a gradual decline in pressure levels in the near-well area. Additionally, the pressure curves at different times converge at a radial distance of 200 m, corresponding to the geometric end of lenticular shale layer 1. Beyond 200 m, the fluid distribution remains dominated by brine, and the pressure wave propagates rapidly, establishing a quasi-steady state distribution of the flow field, causing the pressure curves in this region to overlap, with minimal time dependence.
Figure 6b shows the evolution of pore pressure with vertical distance and injection time in the near-well area at a radial distance of 50 m. Overall, the pressure curves exhibit a monotonically decreasing trend with increasing vertical height and its main gradient aligns with the hydrostatic pressure gradient of the formation fluid. Heterogeneity of the lithologic structure introduces local high-gradient features in the pressure curve. In the vertical distance intervals of 50 to 56 m and 90 to 96 m, corresponding to the lenticular shale layers 1 and 3, the absolute value of the slope increases to show steeper pressure drops. Low permeability reduces the efficiency of energy transfer in the pressure field across the lenticular shale layers. To maintain the continuity of potential energy across layers, large pressure gradients form within the layers to create local pressure drop zones.
Similar to the radial distribution, pressure curves at different times converge near a vertical distance of 96 m corresponding to the upper interface of lenticular shale layer 3. Below shale layer 3, the pressure curves remain distinct due to persistently high gas-phase saturation, which causes formation pressure to decline progressively with injection time, as low-viscosity CO2 displaces brine to reduce flow resistance. Above shale layer 3, the four pressure curves overlap, which indicates that the gas-phase plume is trapped beneath the layer and has not broken through shale layer 3 on a large scale.
Figure 6c shows the evolution of pore pressure with vertical distance and injection time in the far-well area at a radial distance of 300 m. Unlike the characteristics in the near-well zone, the pressure distribution here exhibits time independence and spatial non-monotonicity. The pressure curves at different times overlap in the vertical distribution, which indicates that a stable gradient distribution of the pressure field has been established in the far-well area, unaffected by changes in the upstream gas-phase saturation. A local pressure spike occurs within the vertical distance range of 70 to 76 m, where the pressure above lenticular shale layer 2 is higher than the pressure below the layer. This phenomenon results from the differences in the propagation of pressure waves through different flow paths.
In the near-well area, the upward diffusion of the pressure wave is blocked by lenticular shale layer 3, which causes the pressure energy to deflect and propagate radially. The confined space between lenticular shale layers 1 and 3 focuses and directs the pressure energy to create a local high-pressure zone above lenticular shale layer 2. Conversely, the area below shale layer 2 in the far-well region experiences pressure reduction due to geometric shielding by shale layer 1 and radial flow divergence, where the flow cross-section increases linearly with radial distance to maintain a lower pressure level. Shale layer 2 acts as a low-permeability medium connecting two different pressure systems and exhibits a linear transition of pressure from the lower interface to the upper interface, resulting in a pressure step in the curve.
4.2. Temporal Evolution of Gas-Phase Saturation in Saline Aquifers
Figure 7 shows the spatial distribution of gas-phase saturation during continuous CO
2 injection from day 250 to day 1000. CO
2 is less dense than brine. Injection pressure and buoyancy displace resident brine and produce simultaneous vertical migration and radial spreading. Lenticular shale layers constrain flow and generate a heterogeneous response. Early in the injection period, CO
2 accumulates beneath lenticular shale layer 1 within the basal brine aquifer, forming a high gas-saturation zone. The low-permeability and low-porosity shale acts as a capillary barrier that inhibits upward gas movement and redirects migration from vertical to lateral pathways.
Over time, the CO2 plume extends radially and enters the middle and upper saline aquifers through leakage across the shale matrix and bypass flow around shale edges. CO2 accumulation exhibits pronounced spatial variability. Near the well, at the lower interface of lenticular shale layer 1, CO2 accumulates rapidly and forms a high gas-saturation zone. At the distal location, at the lower interface of lenticular shale layer 2, saturation increases with time but remains below the near-well peak. This decline from left to right reflects two mechanisms. First, axisymmetric geometry increases flow cross-sectional area with radial distance. As the plume migrates outward, a fixed mass of CO2 occupies a larger pore volume, which lowers local gas-phase saturation. Second, the near-well shale forms a local trap. Only after the storage capacity beneath it approaches a saturation threshold does excess CO2 spill laterally and supply the distal reservoir. This delayed supply produces a lagged increase in distal saturation.
To quantify radial spreading and post-barrier migration, we extracted one radial transect at a vertical distance of 40 m and two vertical transects at radial distances of 50 m and 200 m.
Figure 8a shows gas-phase saturation along the basal injection interval at a vertical distance of 40 m as a function of radial distance and injection time. Gas-phase saturation remains high near the well indicating that brine has been displaced in this region. From day 250 to day 1000, the saturation front migrates outward and shows an abrupt drop consistent with an immiscible displacement front. The saturation profiles differ across injection stages. At day 250, limited injection time causes migration to be controlled by buoyancy and pressure gradients. CO
2 accumulates upward along the lower surface of lenticular shale layer 1, so a continuous high-saturation zone has not formed along the sampled transect, and near-well saturation is lower and decreases with radial distance. At day 1000, the profile shows a distinct saturation drop near the radial distance of 200 m, which differs from the trends at earlier times.
The right termination of lenticular shale layer 1 occurs at a radial distance of 200 m, which defines a transition in the migration pathway. Upstream of this point, between radial distances of 0 m and 200 m, the overlying lenticular shale confines CO2 beneath the layer to promote a high gas-saturation accumulation zone. When the plume crosses the shale termination at 200 m, the constraint from the overlying shale is removed. Buoyancy diverts a large fraction of CO2 upward around the shale edge into the overlying saline aquifers. This diversion reduces the lateral CO2 flux along the basal saline aquifer, which lowers local gas-phase saturation and produces the pronounced drop in the profile.
Figure 8b shows gas-phase saturation in the near-well region at a radial distance of 50 m as a function of vertical position and injection time, which indicates that lithologic layering controls vertical distribution. The profiles show alternating peaks and troughs that align with the lithologic layering. Peaks occur at the base of each shale layer and the top of the underlying saline aquifer, while near-vertical drops coincide with shale tops, and troughs occur within the middle saline aquifer. Injected CO
2 migrates upward and accumulates beneath lenticular shale layer 1 to raise local pressure. Once this pressure exceeds the capillary entry pressure of the shale, CO
2 enters the low-permeability shale.
Saturation drops sharply at the upper interfaces of lenticular shale layers 1 and 3 to produce near-vertical decreases in the profiles. Although CO2 enters the shale, the low permeability limits upward flux and causes CO2 to remain within shale pores. When CO2 reaches the overlying saline aquifer, buoyancy and pressure gradients redistribute the fluid to prevent high saturation at the interface and produce a sharp drop. The gas then accumulates beneath the next shale layer and forms a new peak. These processes indicate that in the near-well high-pressure zone, shale layers do not fully block upward migration but act as high-saturation low-flux barriers.
Figure 8c shows gas-phase saturation in the distal region at a radial distance of 200 m as a function of vertical position and injection time. Unlike the layered accumulation near the well in
Figure 8b, the profiles here show a single peak, with the maximum between vertical distances of 20 m and 60 m. In time, saturation remains zero at day 250, indicating that the CO
2 plume has not reached this location. Nonzero saturation appears after day 500. When CO
2 migrating along the basal aquifer reaches the termination of lenticular shale layer 1, buoyancy and pressure gradients drive upward leakage. The rising flow is then constrained by lenticular shale layer 2 on the right. The pressure field indicates a vertical pressure inversion across shale layer 2, with an upward adverse pressure gradient within the shale and at its interfaces. Gas becomes trapped beneath shale layer 2 and spreads laterally. Thickening of the gas layer promotes connection with upward spill flow supplied from below. As a result, saturation along this transect reflects the combined effects of upward spill supply from below and accumulation beneath the overlying shale. This process connects gas across the two stratigraphic levels and forms a continuous high-saturation zone, which produces the single-peak profile.
Figure 7 and
Figure 8 indicate that the staggered lenticular shale layers act analogously to baffles in process equipment. This discontinuous architecture allows upward migration of CO
2, but forces a tortuous pathway that includes lateral spreading, followed by bypass flow around the shale edge, and renewed spreading above. This mechanism lengthens the migration path and increases residence time to enhance sweeping of the upper portions of each saline aquifer. The resulting increase in contact area and contact time between CO
2 and brine promotes storage and improves macroscopic sweep efficiency.
4.3. Temporal Evolution of Dissolved CO2 Distribution in a Saline Aquifer
Figure 9 shows the spatial distribution and evolution of normalized CO
2 concentration during continuous injection from day 250 to day 1000. The normalized CO
2 concentration distribution follows the gas saturation pattern, indicating enhanced dissolution beneath the lenticular shale layers. Acting as structural traps, these shale layers restrict upward CO
2 escape, confining the gas plume beneath the barriers and promoting lateral spreading along their bases. This accumulation increases CO
2-brine contact time and expands the interfacial area between gas and undersaturated brine. As the plume front migrates outward, CO
2 encounters undersaturated brine, sustaining a steep concentration gradient that drives a high dissolution flux and enhances local dissolution.
In addition, steep concentration gradients occur near the shale interfaces. This indicates that, although shale limits gas migration, brine enriched in dissolved CO
2 still diffuses into shale micropores. Over long periods, dissolution allows CO
2 to enter low-permeability media independently of capillary flow by transporting it as a solute into shale micropores, which increases the overall storage potential. Based on the normalized CO
2 concentration field in
Figure 9, four grid points A(10,10), B(10,49), C(10,70) and D(10,82) are selected to examine the temporal evolution of dissolved CO
2 concentration.
Figure 10a shows a rapid increase in normalized CO
2 concentration at points A and B during the early injection period from day 0 to day 50. Both points lie in the near-well high-pressure zone, where rapid gas invasion increases gas saturation and interfacial areas. Dissolution increases and concentrations approach equilibrium rapidly. Thereafter, normalized CO
2 concentration at point B remains higher than at point A because B is adjacent to the lower interface of a lenticular shale layer, where gas accumulates and sustains gas–brine contact, enhancing local dissolution mass transfer. At point A, migration pathways are less confined and fluids exchange continuously, which limits long-term gas residence and keeps normalized CO
2 concentration at a lower level.
Figure 10b shows the temporal evolution of normalized CO
2 concentration at two locations in the middle saline aquifer. Early in time, normalized CO
2 concentration increases faster at point C than at point D because of their position along the migration pathway. Point C lies in the lower part of the middle saline aquifer and sits on the main pathway of the upward flow supplied from below. Under buoyancy, the CO
2 plume passes point C before rising to point D, so point C encounters the gas source earlier, and dissolution begins there first. The curves cross at about day 580. After that time, normalized CO
2 concentration at point D continues to rise and exceeds point C, whereas point C shows a modest decline after reaching its peak and then remains lower. Point D is near the base of lenticular shale layer 3. When CO
2 reaches this depth, the shale limits upward flux and promotes secondary accumulation beneath the layer. Brine at point D remains in contact with CO
2 and continues mass transfer, which drives a sustained increase in dissolved concentration. In contrast, point C lies in the ascent pathway. As gas rises and accumulates near the aquifer top, gas-phase saturation along the flow path past point C decreases.
Overall, the lenticular shale layers promote gas accumulation beneath the layers, which increases residence time for gas–brine mass transfer. This heterogeneous architecture redirects migration from vertical ascent to lateral spreading and expands the contact area between the plume and undersaturated brine, which increases dissolution trapping efficiency.
4.4. Effects of Permeability Anisotropy on CO2 Dissolution
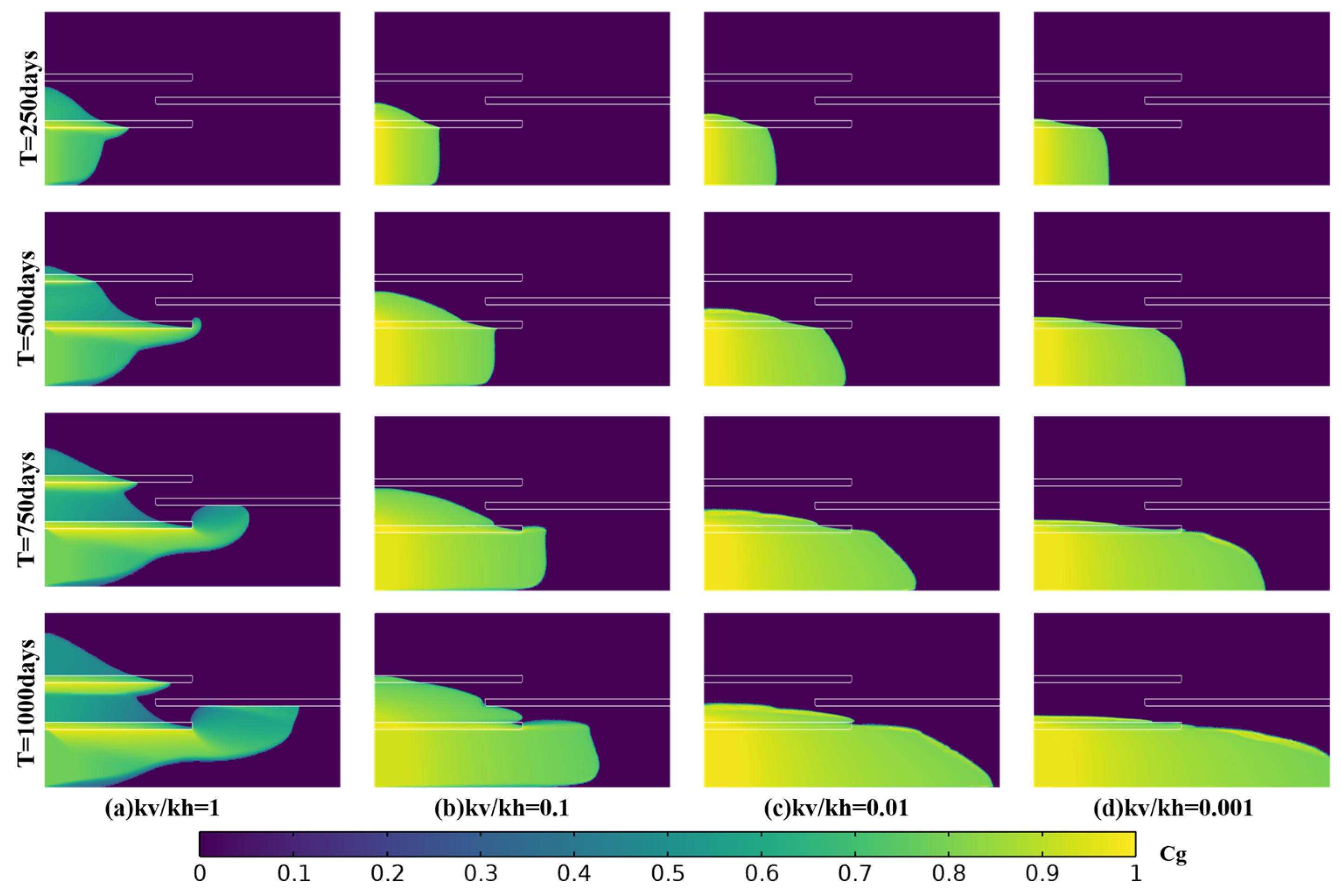
Figure 11 shows the spatiotemporal evolution of normalized CO
2 concentration in the reservoir matrix for four permeability anisotropy ratios. In subsurface reservoirs, horizontal and vertical matrix permeabilities may differ by orders of magnitude, which changes the balance between buoyant rise and lateral pressure-driven spreading. We define the permeability anisotropy ratio
Γ as the ratio of vertical to horizontal permeability:
Here,
kv is the vertical permeability of the reservoir matrix, and
kh is the horizontal permeability of the reservoir matrix.
Figure 11a shows that, under isotropic conditions, migration is dominated by vertical flow, and the high-concentration region exhibits pronounced vertical penetration. From day 500 to day 1000, high-concentration CO
2 does not accumulate extensively in the basal aquifer. Instead, it crosses the lenticular shale layer, migrates upward with time, and accumulates beneath the second lenticular shale layer.
Comparing
Figure 11b,c shows that the high-concentration retention layer beneath the lenticular shale thickens as
Γ decreases. Although CO
2 still crosses the lenticular shale, concentrations decrease after upward migration, indicating that part of the CO
2 is retained in the lower aquifers during transport. This behavior indicates that heterogeneity dilutes and delays the upward transfer of high CO
2 concentrations. When
Γ equals 0.001,
Figure 11d shows migration dominated by lateral spreading, with the high-concentration region confined beneath lenticular shale layer 1. Low vertical permeability increases resistance to upward flow and directs both fluid migration and pressure propagation toward the radial direction. The high-concentration layer remains continuous with a nearly uniform thickness and advances laterally in a piston-like manner. Relative to
Figure 11a, the much lower vertical permeability suppresses vertical mixing and limits upward transport, so most injected CO
2 remains trapped at depth with high saturation.
Quantitatively, the storage efficiency factor decreases only from 0.294 to 0.291 as the permeability anisotropy ratio increases from 0.001 to 1. This small change indicates that the effect of anisotropy on storage efficiency remains limited, even across three orders of magnitude.
4.5. Effects of Injection Rate on CO2 Dissolution
Figure 12 shows the evolution of normalized CO
2 concentration from day 250 to day 1000. At low injection rates, buoyancy controls migration as shown in
Figure 12a. At day 1000, CO
2 migrates primarily upward, and the main gas body accumulated beneath lenticular shale layer 1 exhibits the shortest lateral extent. At low injection rates, viscous pressure gradients are small, and CO
2 does not overcome capillary pressure to drive extensive lateral displacement. Instead, buoyancy favors migration along vertical pathways.
In
Figure 12d, the plume transitions toward a pressure-driven regime as injection rate increases. The basal high-concentration zone thickens and expands laterally beneath the shale, indicating stronger viscous forcing and a longer reach of the pressure perturbation. In the highest-rate case, the CO
2 front propagates across a larger radial distance by day 1000 to approach the domain boundary, and the stacked structure beneath successive shale layers becomes more pronounced. The lenticular shale layers still impose local accumulation due to high capillary entry pressure, but the larger imposed pressure gradient promotes earlier edge bypass and faster replenishment of upper-layer accumulations.
The high-rate cases also show multi-lobed and tongue-like concentration patterns near shale edges at late times. These features reflect competition between lateral sweep beneath the barrier and focused upward leakage at discrete bypass locations. As the injection rate rises, higher pore pressure near the barrier edges increases cross-layer flow through the most conductive pathways and strengthens interlayer connectivity of the plume. This rate dependence implies a tradeoff between lateral footprint and vertical migration risk and provides a mechanistic basis for linking operational rate to plume morphology and the spatial distribution of dissolution opportunity [
54].
Quantitatively, the storage efficiency factor increases from 0.142 to 0.410 as the injection velocity rises from 0.1 × 10−6 m/s to 0.75 × 10−6 m/s. Within the investigated range, injection velocity has the strongest effect on storage efficiency.
4.6. Influence of Lenticular Shale Layer Geometry on CO2 Dissolution Characteristics
This section evaluates how geometric parameters of lenticular shale layers control CO
2 plume evolution and storage security. We designed two sets of cases to isolate the effects of shale vertical thickness and radial length, with a simulation duration of 1000 days for all cases.
Figure 13 shows simulations with a fixed shale radial length and shale vertical thicknesses of 6 m, 8 m, and 10 m. The three cases show that the large-scale migration pathway of the high-concentration plume remains similar from day 250 to day 1000. In all cases, CO
2 rises from the injection side, accumulates beneath the shale, and then bypasses at the shale edge to supply the overlying unit. Because changing thickness does not alter the radial location of the shale edge, the plume must traverse almost the same lateral distance beneath the barrier before an upward pathway develops, which explains the weak sensitivity of the macroscopic concentration field to thickness.
Although the overall pathway is consistent, shale thickness modifies the local structure of the accumulation beneath the barrier. In the thinnest shale case, the high-concentration tongue beneath the shale becomes established earlier, and the onset of edge bypass appears sooner, which yields a more elongated along-interface band at intermediate times. As thickness increases, CO2 must build a larger local saturation and pressure anomaly beneath the shale before bypass initiates. This requirement shifts the basal accumulation from a more rounded front to a more boundary-aligned, quasi-rectangular pool with a thicker high-concentration core.
These differences indicate that shale thickness acts as a local control on the timing and sharpness of bypass rather than a primary control on plume reach. Over the simulated period, thickness changes influence where high concentrations persist beneath the shale, but they do not strongly change how far the plume migrates laterally or how the upper accumulation develops once bypass has begun.
Figure 14 shows how the radial length of the lenticular shale controls plume evolution under otherwise identical conditions. Increasing shale length increases the lateral distance that CO
2 must travel beneath the barrier before reaching an edge, and increase residence time of high-concentration CO
2 in the lower unit and delay the formation of an upward bypass pathway. This geometric control is evident across the time sequence, where the shortest shale case exhibits early edge bypass and a boundary-attached upward finger, while the longer shale cases retain a larger fraction of the high-concentration core beneath the shale for a longer period.
In the short-shale scenario, the plume rapidly reaches the barrier’s edge, where buoyancy forces a swift upward bypass. This rapid vertical migration feeds the overlying layer, creating an elongated, high-concentration zone right next to the shale edge. As shale length increases, the upward finger weakens and the lower-layer accumulation expands. Continued injection mainly increases the volume of the basal pool beneath the shale to yield a more compact quasi-hemispherical retention zone that grows laterally and thickens over time.
By day 1000, the short-shale case shows a more extensive retention zone beneath the second shale due to earlier bypass, whereas the long-shale cases retain most CO2 beneath the first barrier to demonstrate sustained storage at depth. These results indicate that shale length is a first-order geometric lever that regulates pathway tortuosity, controls the timing of vertical escape, and redistributes high concentrations between lower and upper aquifers.
Quantitatively, the storage efficiency factor increases from 0.289 to 0.292 as the shale radial length increases from 150 m to 250 m. It then rises from 0.292 to 0.295 as the shale thickness increases from 6 m to 10 m. These results indicate that shale geometry promotes storage efficiency, but the overall effect remains limited.
4.7. Storage Efficiency Factor and a Nonlinear Multi-Parameter Power-Law Correlation Model
Based on the previously defined storage efficiency factor and the analyses of different controlling factors in
Section 4.4,
Section 4.5 and
Section 4.6, a multi-parameter correlation model for the storage efficiency factor is further developed in this section. To represent the dominant mechanisms and eliminate dimensional effects, the relevant independent variables are further reformulated into dimensionless forms. We use a macroscopic capillary number
Ca* to represent the competition between viscous pressure gradients and the capillary threshold pressure:
Here,
μCO2 is the dynamic viscosity of CO
2 (Pa·s),
k is the matrix permeability (mD), and
L is a characteristic length scale for fluid migration (m). Using the total reservoir thickness
Hres as the characteristic length, we define the normalized shale radial length
WD and normalized shale thickness
HD as:
The recast dimensionless multi-parameter nonlinear power-law model is:
Because geologic and engineering controls couple nonlinearly to influence storage efficiency, direct nonlinear fitting may lead to convergence difficulties and nonunique solutions. A logarithmic transformation is applied to convert the multiplicative nonlinear relationship into an additive linear form and then fit a multiple linear regression model:
Then the equation can be written in the standard multiple linear form:
Here, ln(ϵ) is the response variable, and ln(Ca*), ln(WD), ln(HD), and ln(Γ) are predictors. We fit the regression using ordinary least squares on the simulation dataset by minimizing the sum of squared residuals between observed and predicted log values, which yields unbiased parameter estimates with minimum variance under standard assumptions. We evaluate model performance using the coefficient of determination R2 for goodness of fit and the root mean square error RMSE for predictive accuracy.
Regression on the simulation dataset shows good model fit, with R
2 greater than 0.92.
In the dimensionless storage efficiency correlation shown in
Figure 15, the constant 0.073 represents the characteristic storage coefficient for this geologic configuration. The exponents in the correlation reflect competition among the controlling mechanisms during CO
2 storage in deep saline aquifers. Storage efficiency
ϵ increases with
Ca* following a power law with an exponent of 0.543. Higher
Ca* indicates that viscous pressure gradients become more important relative to capillary thresholds. Once
Ca* exceeds a threshold, the macroscopic pressure drop can overcome capillary entry pressures in small pore throats. The exponent 0.543 indicates nonlinear gain. Increasing injection velocity increases both injected mass and reservoir pressure and promotes access to low-permeability pore space that would otherwise remain poorly connected.
In addition, the exponents for the normalized shale radial length WD and normalized shale thickness HD are both 0.020, indicating a robust positive association between lenticular shale geometry and storage efficiency. The mechanism is that shale layers act as physical baffles that increase pathway tortuosity and gas-brine contact time, which promotes residual gas trapping. The exponent for the permeability anisotropy ratio is −0.003, indicating a weak suppressing effect. This suggests that, in this heterogeneous setting, although Γ influences vertical migration in homogeneous media, flow steering by the lenticular geometry dominates the flow field and reduces the macroscopic contribution of anisotropy to storage efficiency. From an engineering perspective, this dimensionless correlation offers a practical basis for evaluating storage performance. The macroscopic capillary number Ca* is a key indicator for optimizing injection velocities. The geometric parameters WD and HD provide quantitative metrics for site selection. The permeability anisotropy ratio Γ has a limited quantitative effect under the investigated heterogeneous conditions. Even so, it remains relevant for characterizing the tendency for vertical flow.
4.8. Sensitivity Analysis of the Storage Efficiency Factor to Key Parameters
Figure 16 shows the sensitivity of the storage efficiency factor to
Ca*,
WD,
HD, and
Γ. Among these parameters,
Ca* has the strongest effect on the storage efficiency factor
ϵ. As
Ca* increases,
ϵ rises from 0.142 to 0.410 and shows a clear nonlinear increase. This trend suggests that stronger viscous forces under higher injection conditions improve the ability of CO
2 to enter and occupy the pore space, which increases storage efficiency. This result agrees with the dimensionless correlation analysis in
Section 4.7 and confirms that
Ca* is the dominant parameter controlling
ϵ.
By contrast, increases in WD and HD produce only small increases in ϵ, whereas changes in Γ cause little variation in ϵ. This result indicates that the geometric scale of the shale layers has a positive but limited effect on storage efficiency. Permeability anisotropy is also not a primary control on macroscopic storage efficiency within the investigated range. Overall, the sensitivity analysis shows that the relative influence of the investigated parameters on storage efficiency follows the order Ca* > WD ≈ HD > Γ.
4.9. Qualitative Discussion on Long-Term Geochemical Effects and Future Outlook
The current numerical simulations focus on multiphase flow and dissolution processes over short to medium timescales. However, geochemical interactions between CO
2, brine, and minerals influence long-term storage security in deep saline aquifers [
55]. Over long time scales, lenticular shale layers may influence the spatial distribution of reaction zones and the progression of the chemical weathering front by modifying the residence time and migration pathways of CO
2-rich brine. Low-permeability barriers promote lateral plume spreading beneath the barrier before upward migration resumes [
56], which implies that CO
2-rich, low-pH brine may persist and spread along the underside of lenticular layers. As a result, the chemical weathering front is expected to extend preferentially along the shale base rather than remaining confined to a near-well vertical pathway.
On a long-term scale, these fluid stagnation regions at the shale bottom and the bypass channels at the edges may evolve into concentrated geochemical reaction zones. Within these local reaction bands, prolonged fluid-rock interactions initially drive the dissolution of primary minerals such as feldspar or carbonate cements, leading to a slight increase in local porosity [
57]. As the reactions proceed, the precipitation of secondary carbonate minerals becomes dominant. This dissolution-and-precipitation process in localized areas induces heterogeneous changes in reservoir porosity and permeability. Long-term secondary mineral precipitation may clog the micro pore throats of the bypass channels at the edges of the lenses, causing a further reduction in local permeability [
58]. From a long-term perspective, such heterogeneous changes in physical properties induced by local concentrated reactions reinforce the physical sealing capacity of the lenticular formations, improving the overall safety of CO
2 sequestration.
The computational framework of this study does not include quantitative simulations of the complex mineral dissolution and precipitation processes mentioned above. Future work may introduce coupled thermal, hydrological, mechanical, and chemical models. Such models would provide an in-depth quantitative assessment of the heterogeneous evolution mechanisms of porosity and permeability along with the long-term distribution of reaction zones under the constraints of heterogeneous lenses.